Abstract
A 10-month-old female Parson Jack Russell Terrier was euthanatized because of therapy-resistant ataxia, hypermetria, and deafness that had first been observed at 10 weeks of age. Severe, bilateral, symmetrical neuronal degeneration and mineralization of the brain were found in the cochlear and cerebellar nuclei, dorsal areas of the medulla oblongata, the vestibulocochlear nerve, plexus choroideus, and within the granule cell layer of the ventral cerebellar hemispheres. The mineralized deposits were located free in the parenchyma, around intact or degenerate neurons, in myocytes of small- and medium-sized arteries, and around capillaries. Hepatocytes and cardiac myocytes showed oncocytotic change with increased numbers of enlarged or misshapen mitochondria filled with densely packed cristae and electron-dense inclusions. Skeletal myocytes had only minor increases in the number of mitochondria. The microscopic and ultrastructural lesions were consistent with mitochondrial encephalopathy with similarities to mitochondrial encephalomyopathy with lactic acidosis and strokelike episodes in humans.
Keywords
Mitochondriopathies represent an extremely heterogeneous group of disorders that result from genetically defective mitochondrial pathways. These may be caused by mutations in the nuclear or mitochondrial deoxyribonucleic acid (DNA) that occur spontaneously or by maternal, Mendelian, or complex inheritance patterns.4,9 The essential role of mitochondrial oxidative phosphorylation in cellular energy production leads to a particular vulnerability of tissues with high energy requirements, including the nervous system and cardiac and skeletal musculature.8 In contrast to the large number of distinct mitochondrial disease entities that have been identified in humans throughout the last 40 years, it appears that spontaneous mitochondrial disorders are rare findings in animals. A condition with severe neuronal degeneration, necrosis, and astrocytosis in the cortex and cerebellar vermis occurs in the Alaskan Husky breed with striking similarities to Leigh's disease in humans.3 A systemic myopathy has been reported in Old English Sheepdog littermates, resembling mitochondrial myopathies in humans.1 An English Springer Spaniel had encephalomyopathy with morphologic abnormalities in the mitochondria.2 Finally, a mitochondriopathy restricted to the musculature was reported in a Jack Russell Terrier.7 A more comprehensive overview on mitochondriopathies reported in animals so far is listed in table 2 of Brenner et al.3 Here, we report a case of focal neural and vascular degeneration and mineralization in a Jack Russell Terrier, reminiscent of lesions that are typically seen in the human mitochondrial encephalomyopathy with lactic acidosis and strokelike episodes (MELAS) syndrome.
A female Parson Jack Russell Terrier had gait abnormalities and increased sleepiness that had first been observed at 10 weeks of age. The physical examination at 10 months of age including electrocardiography was normal. The neurologic examination revealed an alert dog that barked repeatedly and was unable to locate sources of noise, suggesting bilateral blindness. Gait abnormalities included symmetric generalized ataxia and hypermetric movements of all four legs. A permanent, generalized, fine tremor was observed, which worsened during feeding. The menace response was absent, conjugated eye movements were delayed, and a horizontal positional nystagmus was noticed. Proprioception and spinal reflexes were normal. Clinical pathology findings including the measurement of electrolytes and the examination of the cerebrospinal fluid revealed no abnormalities. No changes in size or structure of the cerebellum were detected by thecography and computed tomography scans. Recordings of brainstem auditory–evoked potentials indicated complete bilateral deafness. Attempts to improve the clinical signs by repeated systemic administration of corticosteroids remained unsuccessful. The dog was euthanatized at 10 months of age with a presumptive clinical diagnosis of cerebellar encephalopathy. The parents of the dog were clinically healthy, the dog had no siblings, and similar clinical abnormalities have not been observed within the dog's kindred.
A complete necropsy revealed no significant gross findings. Histologic examination was performed on hematoxylin and eosin (HE)-stained tissue sections prepared from formalin-fixed, paraffin-embedded tissues from all major organ systems. The most significant findings were detected in the brain. There was a remarkable bilateral mineralization of select structures, mainly located around the fourth ventricle. The cochlear nuclei, dorsal medulla, cerebellar nuclei (Figs. 1, 2), granular layer of ventral parts of the cerebellar cortex (Fig. 3), and major portions of the vestibulocochlear nerve (Fig. 4) contained fine granular to coalescing basophilic deposits suggestive of mineralization, which was later confirmed by special stains. Four distinct patterns of deposits were observed in the brain: fine granular to coarse deposits free throughout the neuropil (Fig. 5), intracellularly within myocytes of small- to medium-sized arteries up to 250 μm in diameter (Fig. 6), surrounding capillaries (Fig. 7), and perineuronal (Fig. 8). The deposits stained strongly with the von Kossa stain (Figs. 1–7) and the periodic acid–Schiff (PAS) reaction (Fig. 8). A Turnbull stain of the deposits for iron was variable and only weakly positive in select regions with a random distribution. Adjacent to areas with heavy mineral deposits, there were occasional degenerate neurons (Fig. 9) and spheroids. With the exception of a single medium-sized artery that was surrounded by a rim of monocytes and macrophages within a severely affected area of the vestibulocochlear nerve, there was no evidence of inflammation. Throughout the cerebral cortex and hippocampus, there were moderate numbers of single or clustered Alzheimer type II–astrocytes surrounding neurons or isolated in the neuropil. No lesions were found in other structures of the brain, eyes, inner ear, or spinal cord. Microscopic changes in other tissues were minor. In hepatocytes and cardiac and, to a much lesser degree, skeletal muscle cells, a granular eosinophilic quality to the cytoplasm was noticed surrounding the nucleus (muscle cells; Fig. 10) or throughout the entire cytosol (hepatocytes; Fig. 11). The eosinophilic granules appeared as distinct structures, 1 to 3 μm in diameter, suggestive of mitochondria. A mild increase of small- to medium-sized clear vacuoles was observed intracellularly in hepatocytes (Fig. 11). Sudan-red stain identified these vacuoles as lipid droplets. Endocrine glands including parathyroids and adrenals were normal. There was no evidence of mineral deposition in any other tissue, such as kidney, lung, stomach, or major arteries (von Kossa stain), and the structure of bones was normal. Myofibers with increased amounts of red-staining sarcoplasm in the intermyofibrillar and subsarcolemal areas (so-called ragged-red fibers) were not observed in sections of skeletal or cardiac muscle cells (Gomori's modified trichrome stain).
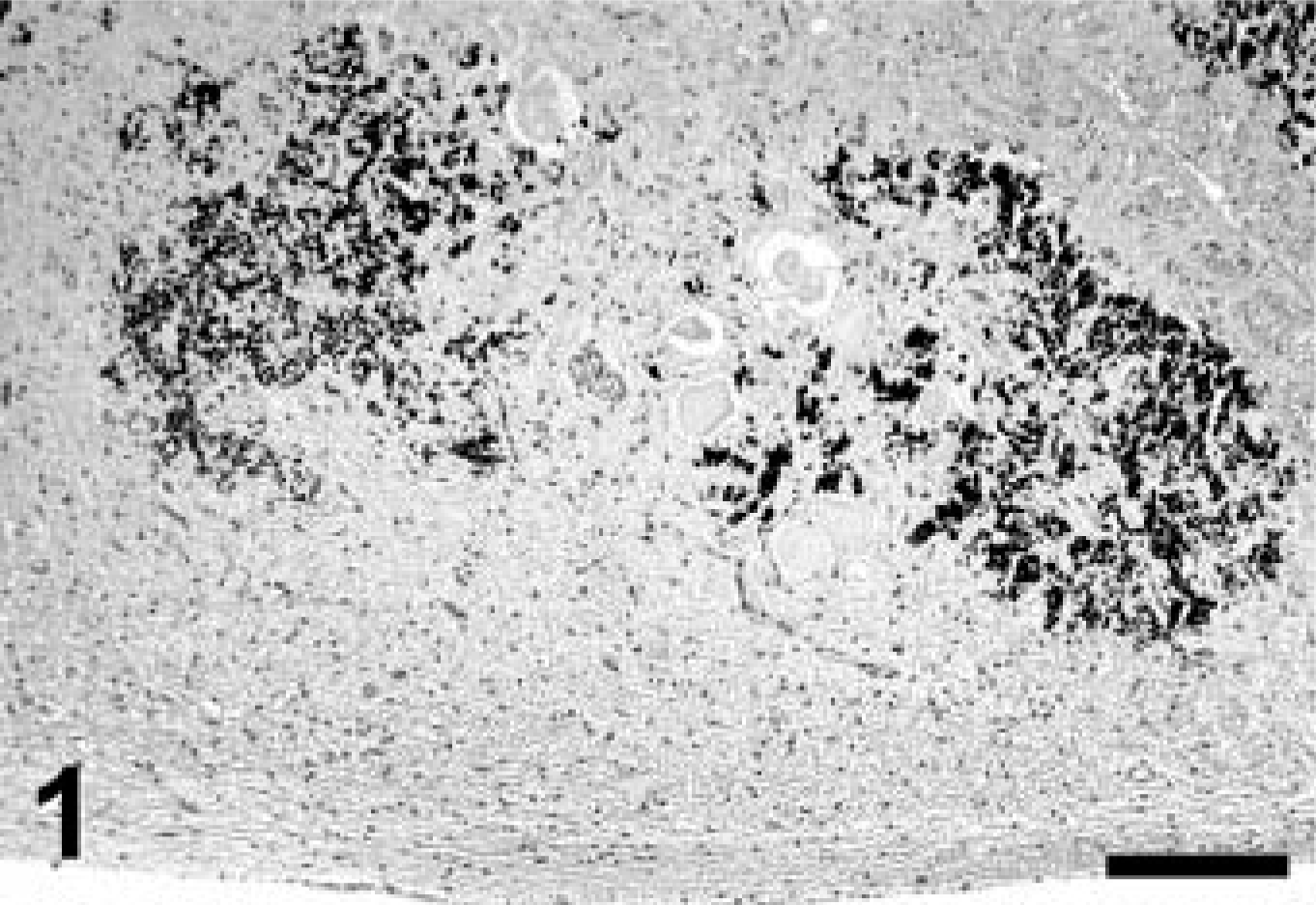
Cerebellum; Jack Russell Terrier. Marked mineralization (dark, granular deposits) of two adjacent cerebellar nuclei. The edge of the fourth ventricle is seen at the bottom. Von Kossa stain. Bar = 200 μm.
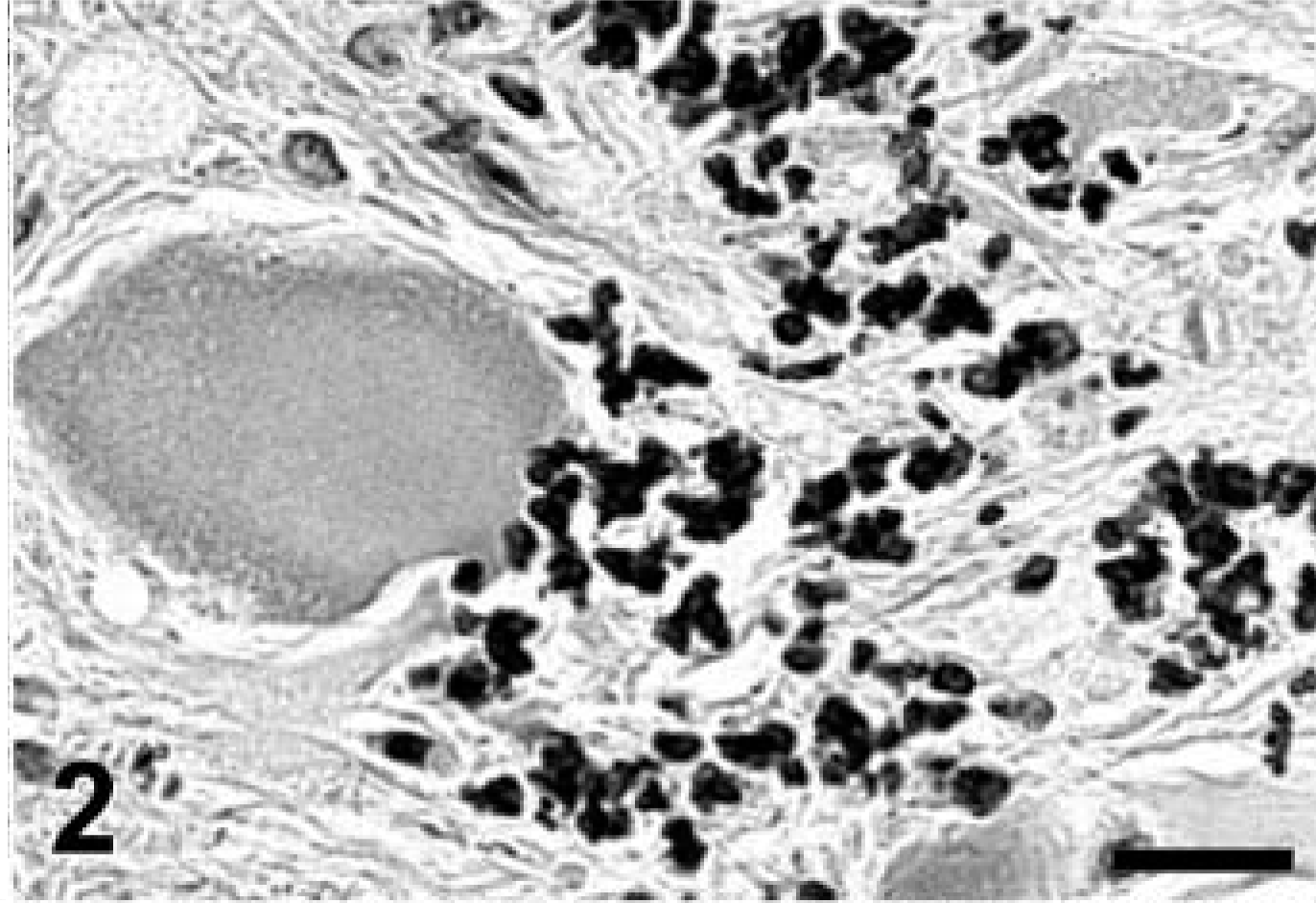
Cerebellum; Jack Russell Terrier. Higher magnification of Fig. 1. Marked mineralization appears to be extracellular in the paraffin-embedded tissue. Von Kossa stain. Bar = 20 μm.
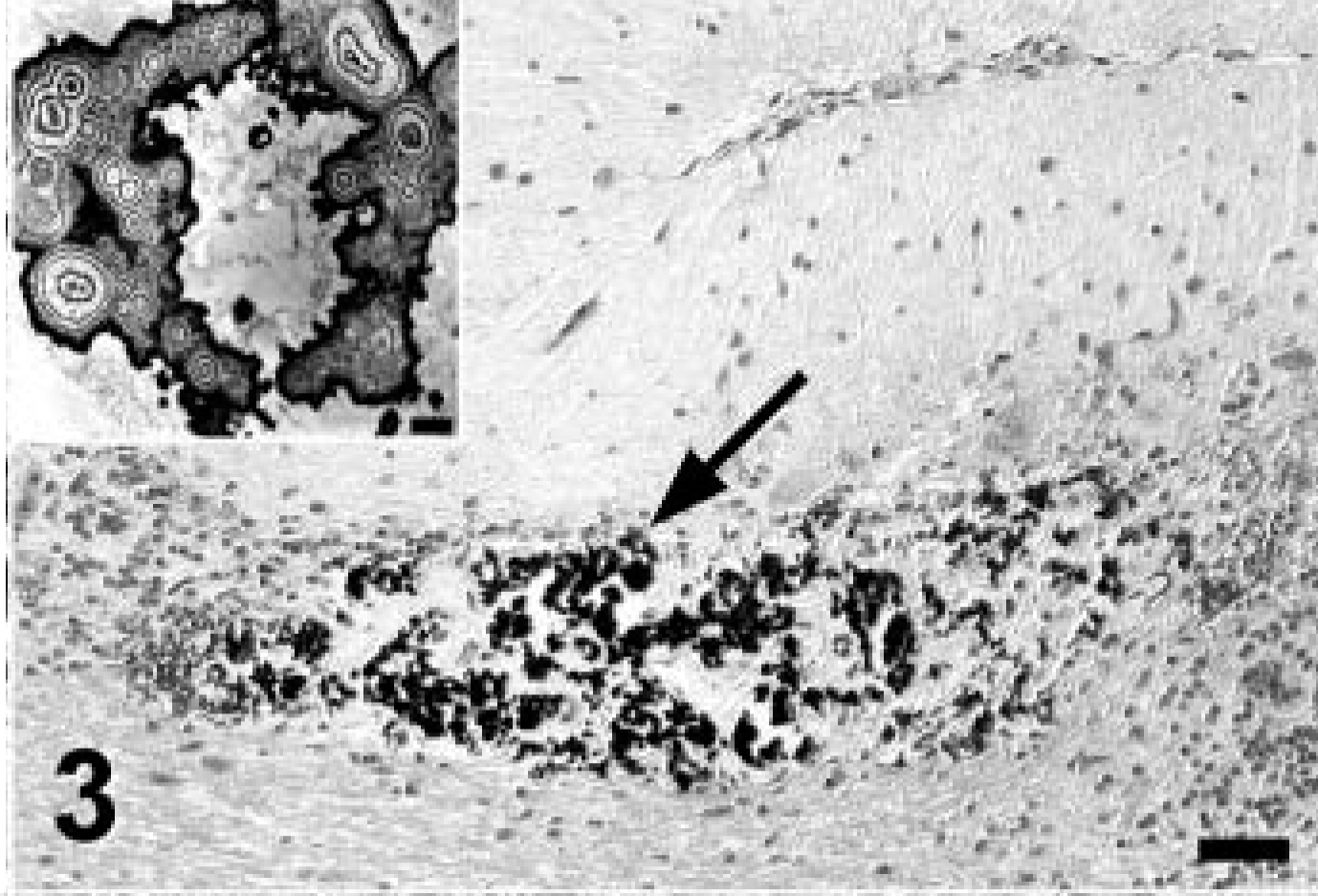
Cerebellum; Jack Russell Terrier. Focal mineralization (arrow) of the granular layer in the cerebellum with mild focal loss of Purkinje cells. Von Kossa stain. Bar = 100 μm. Inset: The ultrastructure of mineral deposits in the brain were reminiscent of fractals with multilayered circular cores. Electron micrograph of formalin-fixed, paraffin-embedded tissue. Bar = 2 μm.
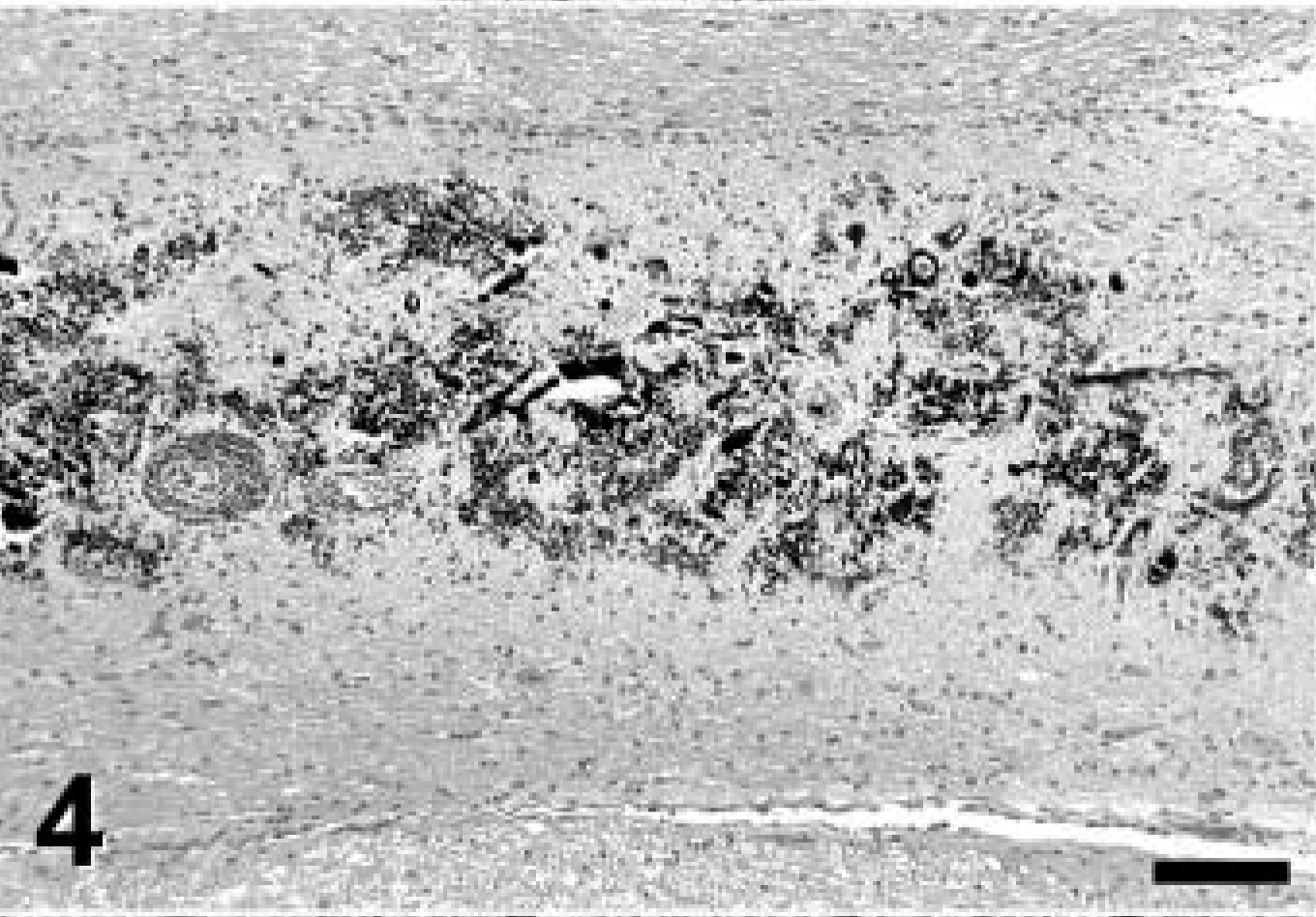
Root of left vestibulocochlear nerve; Jack Russell Terrier. Note the severe granular mineralization of the nerve. Von Kossa stain. Bar = 200 μm.
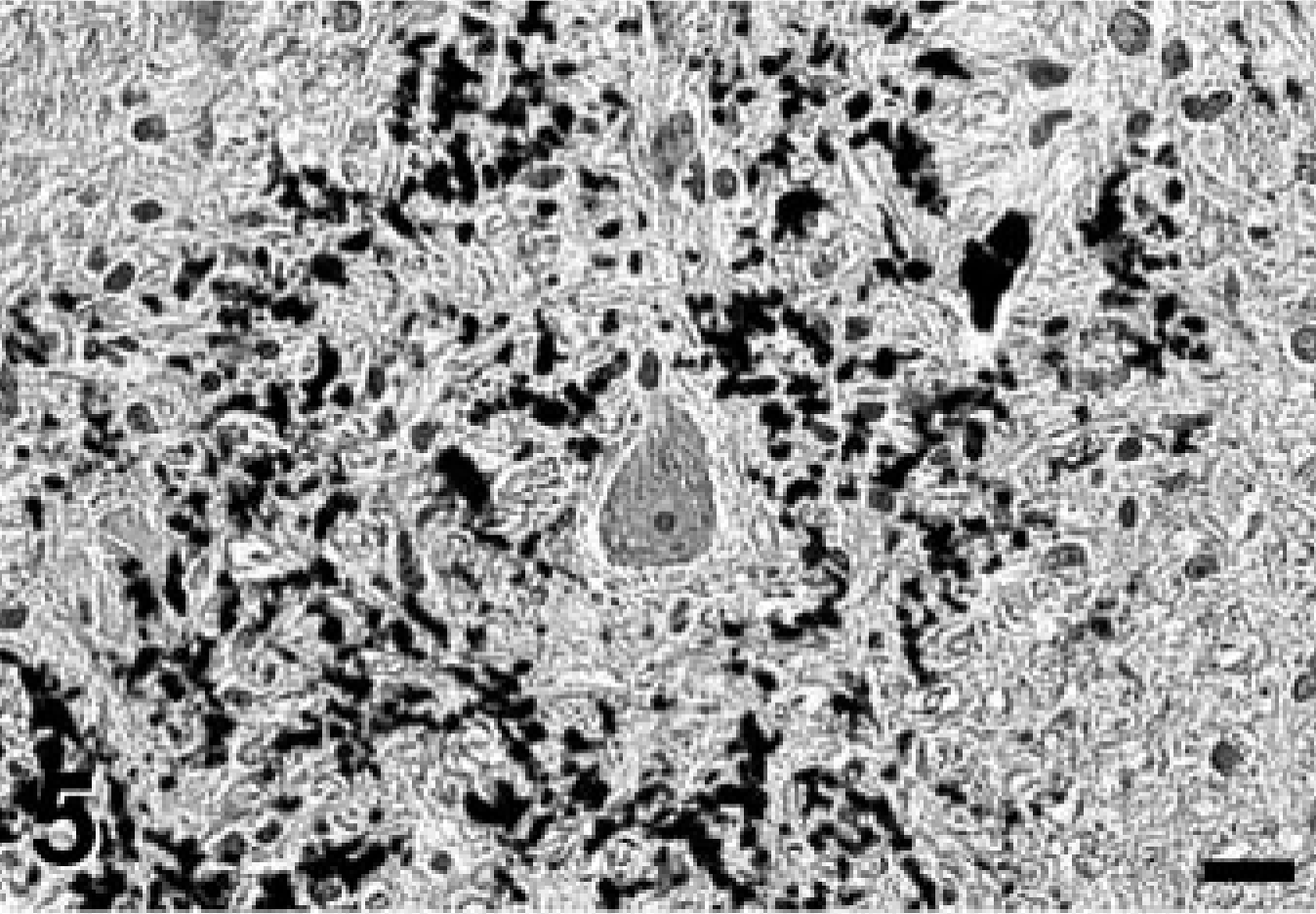
Medulla oblongata; Jack Russell Terrier. Fine granular mineral deposits free in the neuropil, surrounding an intact neuron. Von Kossa stain. Bar = 50 μm.
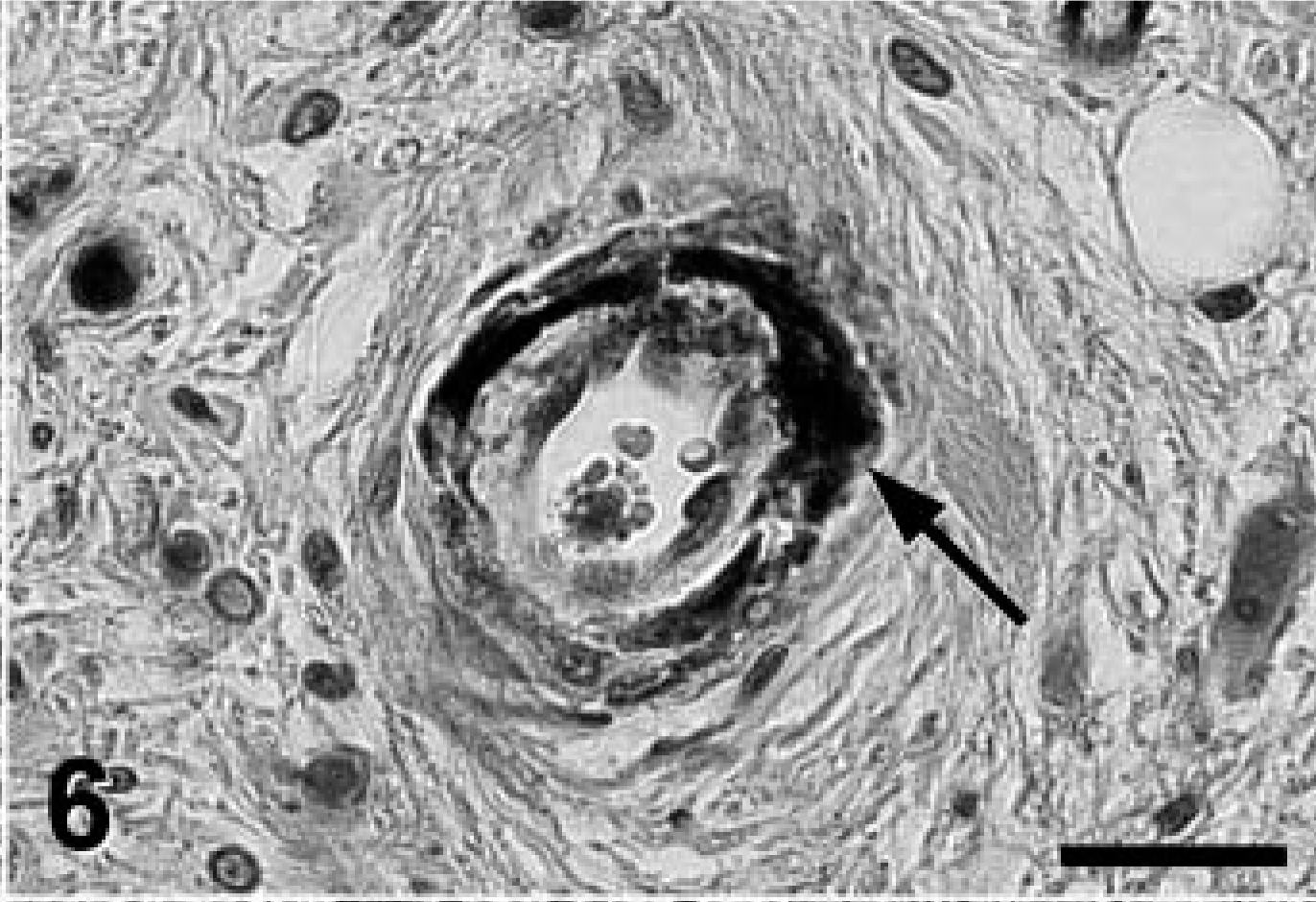
Medulla oblongata; Jack Russell Terrier. In small- and medium-sized arteries up to 250 μm in diameter, mineral deposits were associated with smooth muscle cells of the vessel wall (arrow). Endothelial cells and vascular elastic and basement membranes were unaffected. Von Kossa stain. Bar = 50 μm.
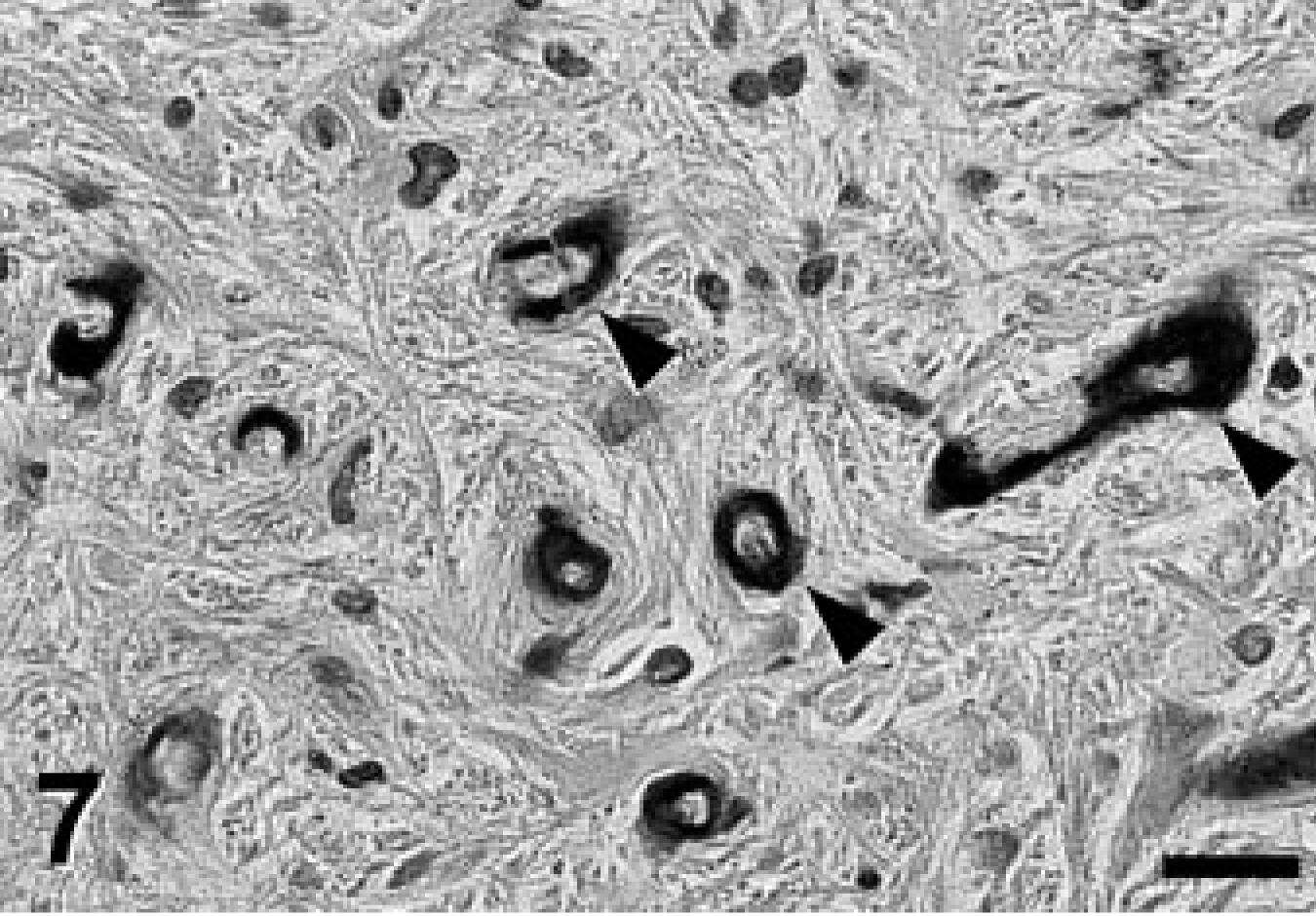
Medulla oblongata; Jack Russell Terrier. Mineral deposits surrounding capillaries (arrowheads) in the neuropil. Von Kossa stain. Bar = 50 μm.
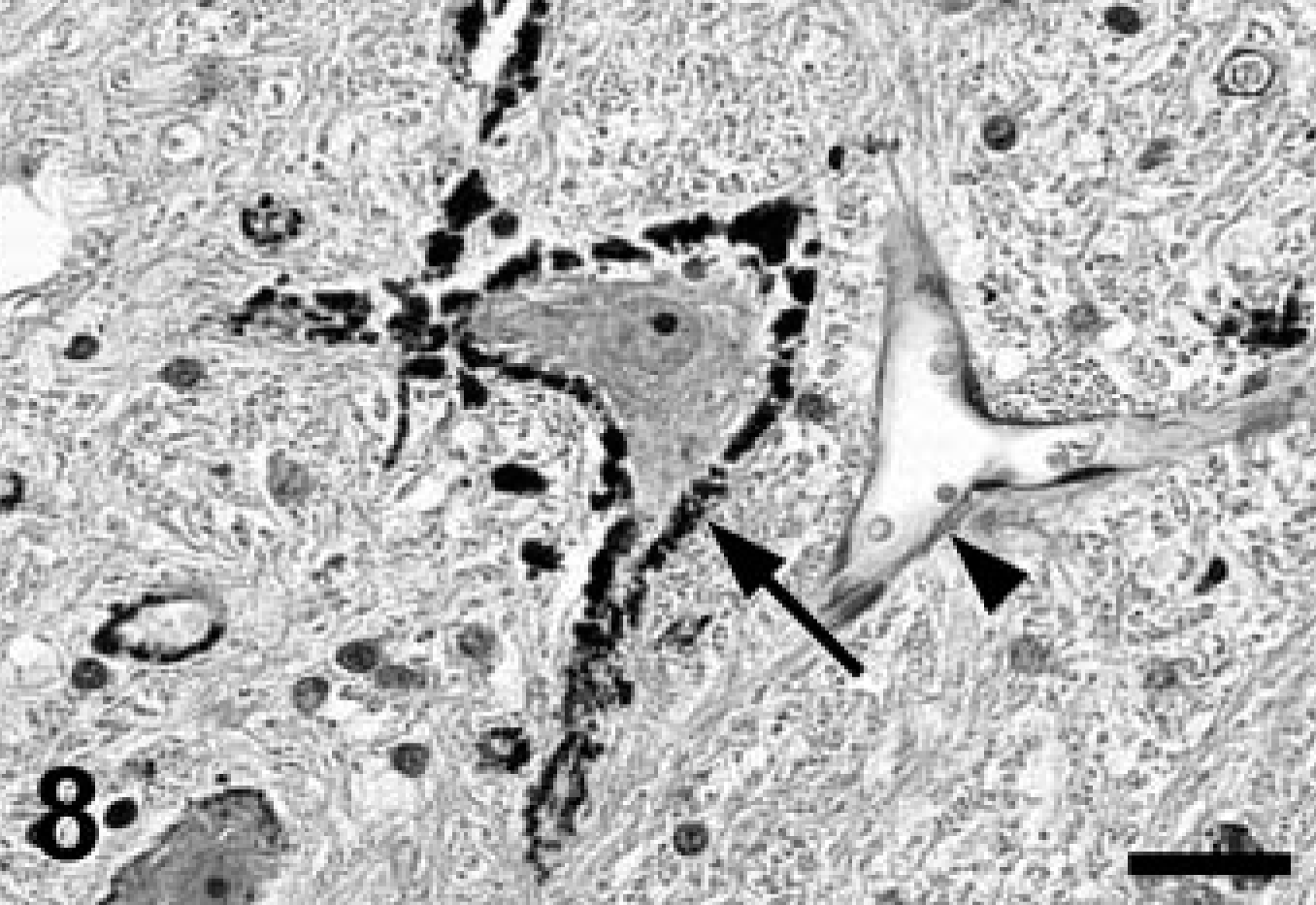
Left cochlear nucleus; Jack Russell Terrier. A distinct perineuronal pattern of mineralization (arrow) was frequently seen in the affected areas with no evidence of neuronal damage. No mineral deposits were noticed in the wall of veins (arrowhead). PAS stain. Bar = 50 μm.
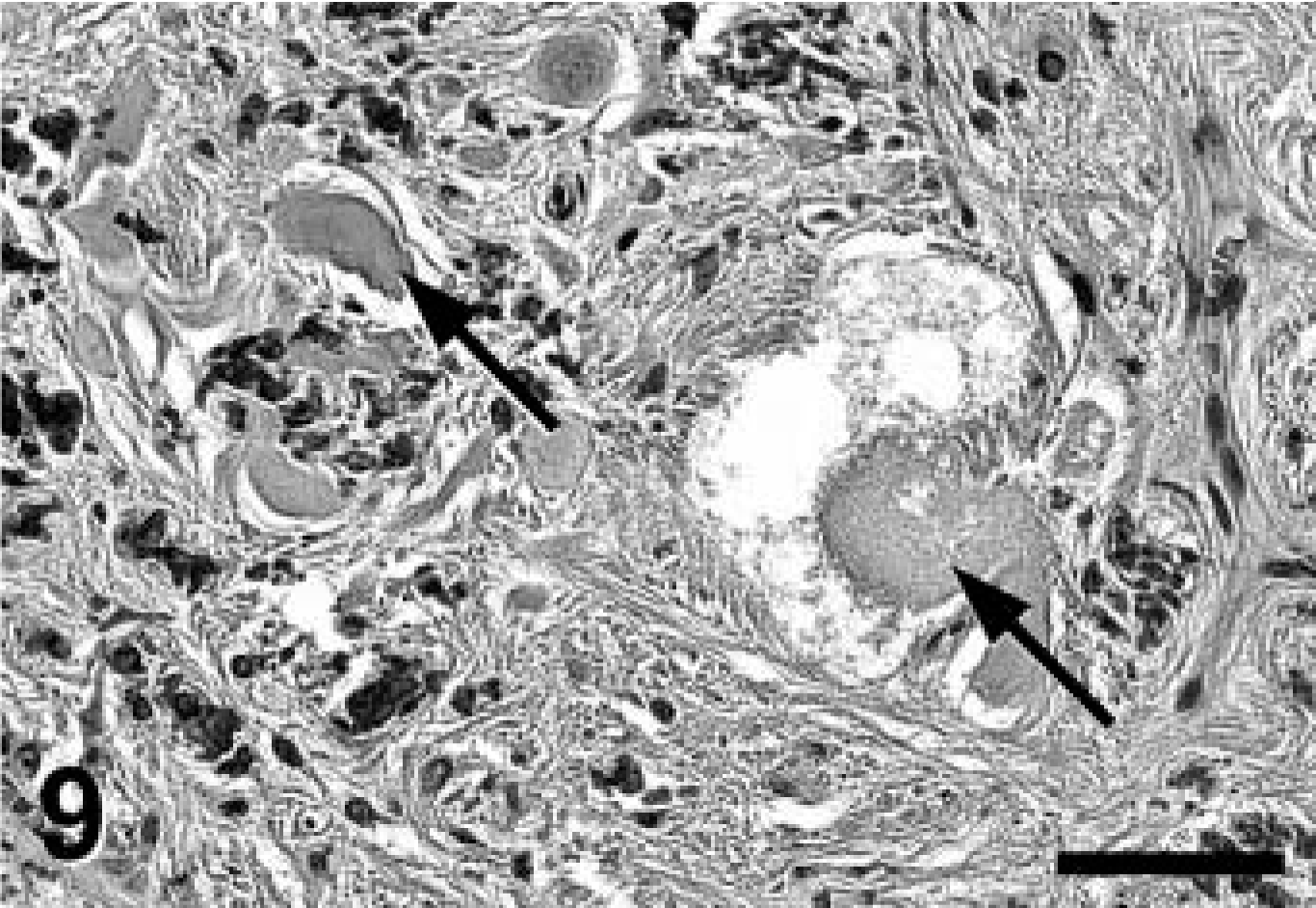
Cerebellar nucleus; Jack Russell Terrier. In select areas adjacent to heavy mineral deposits, marked degeneration of neurons was observed including chromatolysis and ballooning degeneration (arrows). PAS stain. Bar = 100 μm.
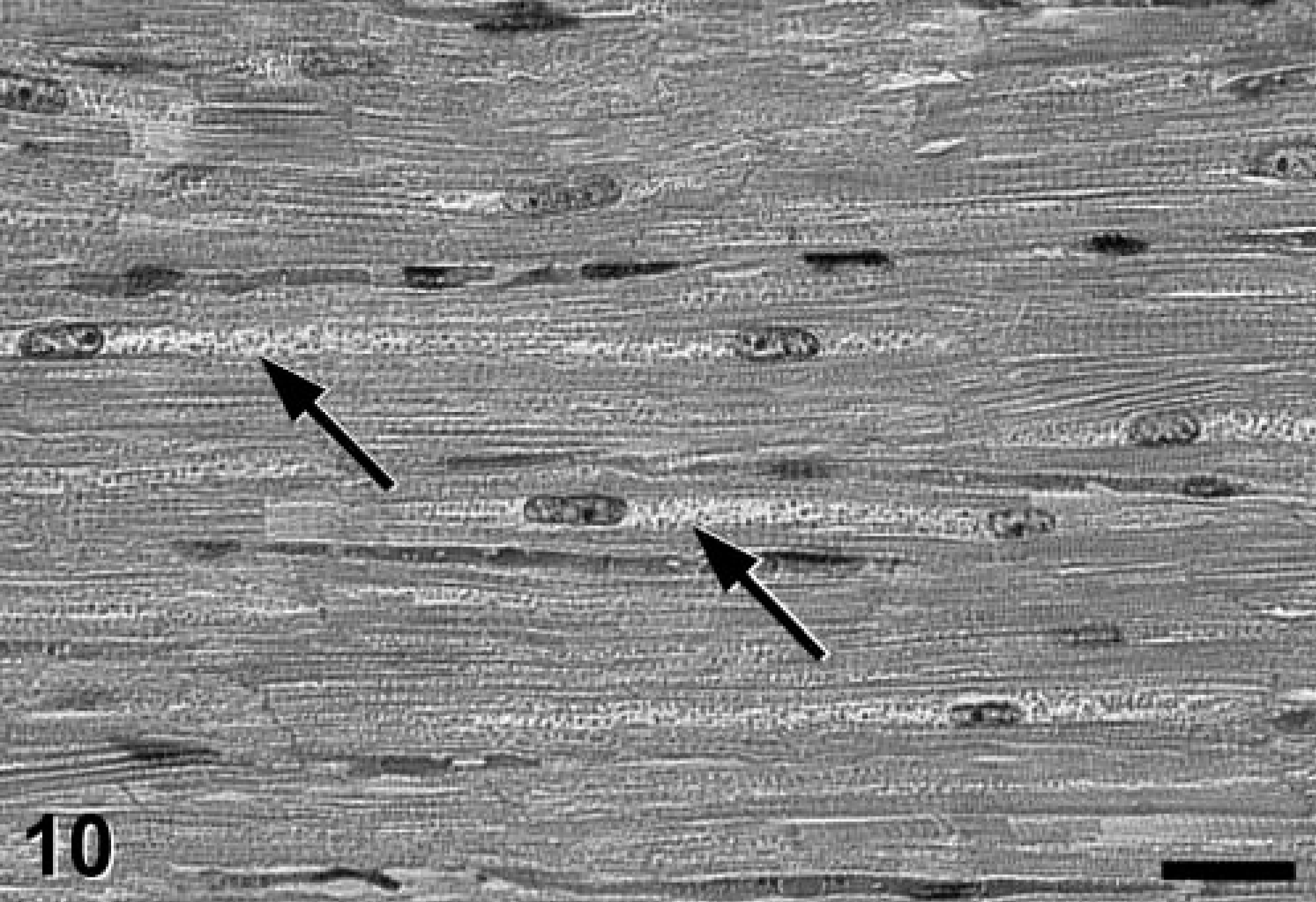
Heart; Jack Russell Terrier. An eosinophilic, granular (oncocytotic) pattern was noticed in cardiac myocytes diffusely throughout the cells and extending longitudinally on both sides of the nuclei (arrows). HE stain. Bar = 50 μm.
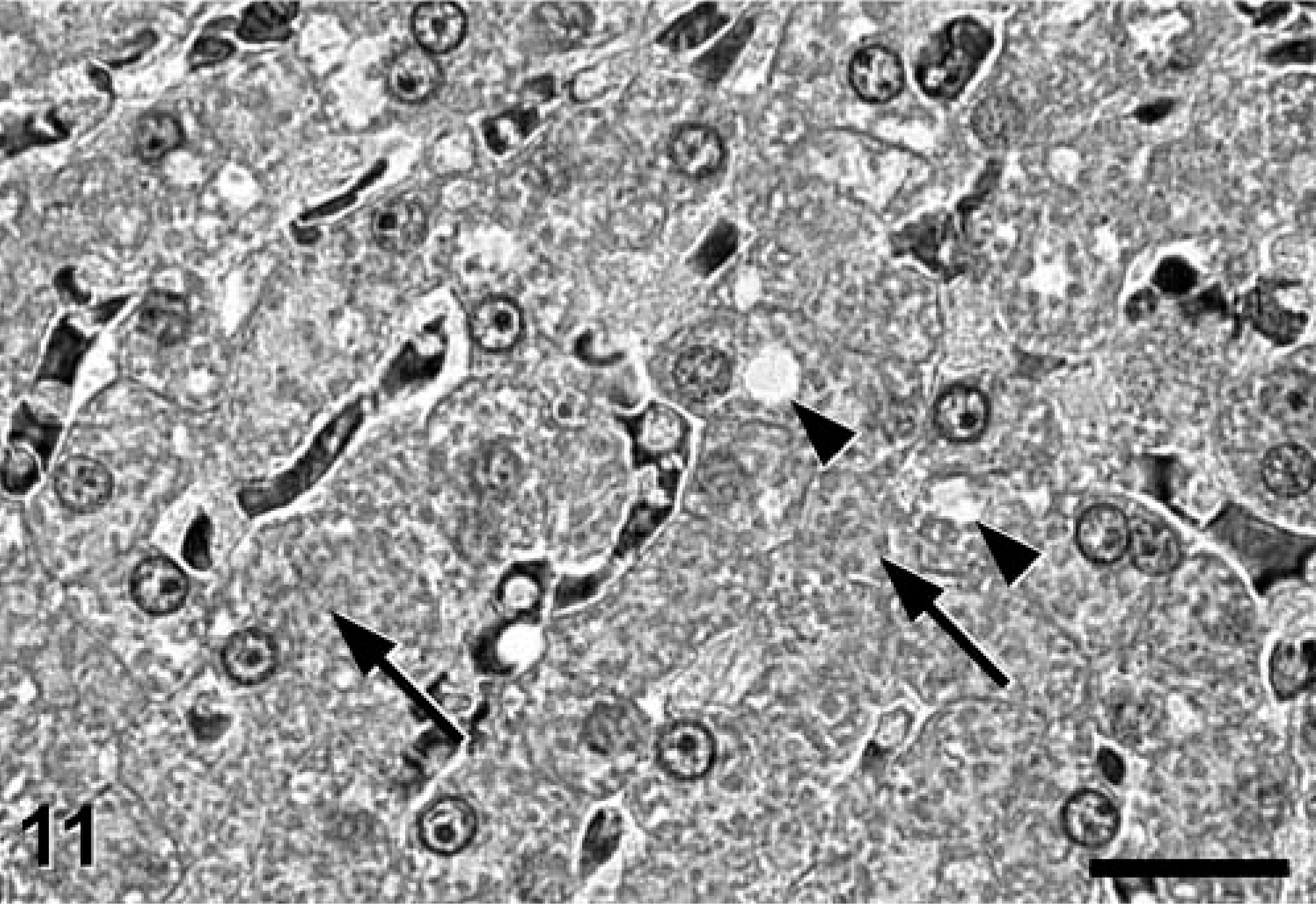
Liver; Jack Russell Terrier. Hepatocytes had a similar diffuse, eosinophilic, granular (oncocytotic) pattern (arrows). Arrowheads indicate lipid droplets. HE stain. Bar = 50 μm.
Because tissues specifically prepared for electron microscopy were unavailable, electron microscopy was performed using the formalin-fixed, paraffin-embedded tissues after deparaffination, postfixation with 5% glutaraldehyde, contrasting with osmium tetroxide, and standard embedding in epon. Ultrastructurally, the eosinophilic cytosolic globules within hepatocytes and cardiac muscle cells seen in HE sections were identified as mitochondria with various abnormalities. Mitochondria were significantly increased in number, enlarged, and contained densely packed cristae (Figs. 12, 13). The majority of mitochondria had numerous, randomly distributed, round to irregularly shaped, non–membrane-bound electron dense inclusions of variable sizes. When compared with mitochondrial inclusions of similarly processed tissues from two normal control dogs, the Jack Russell Terrier's inclusions were significantly more numerous, more heterogeneous, and larger. Examination of the mitochondria in neurons and encephalic vascular myocytes in the affected brain areas was complicated by neuronal degenerative lesions, autolysis, and artifacts caused by the mineralization, but similar numerical and structural mitochondrial abnormalities were seen in select regions where the tissue morphology was appropriately conserved. Basement membranes could not be evaluated by electron microscopy because of their loss during paraffin embedding. The ultrastructural appearance of the encephalic mineral deposits resembled fractal-like patterns with multilayered circular cores (inset of Fig. 3).

Heart, Jack Russell Terrier. Cardiac myocytes had increased numbers of mitochondria filled with tight stacks of cristae and electron-dense inclusions. Electron micrograph of formalin-fixed, paraffin-embedded tissue. Bar = 500 nm.
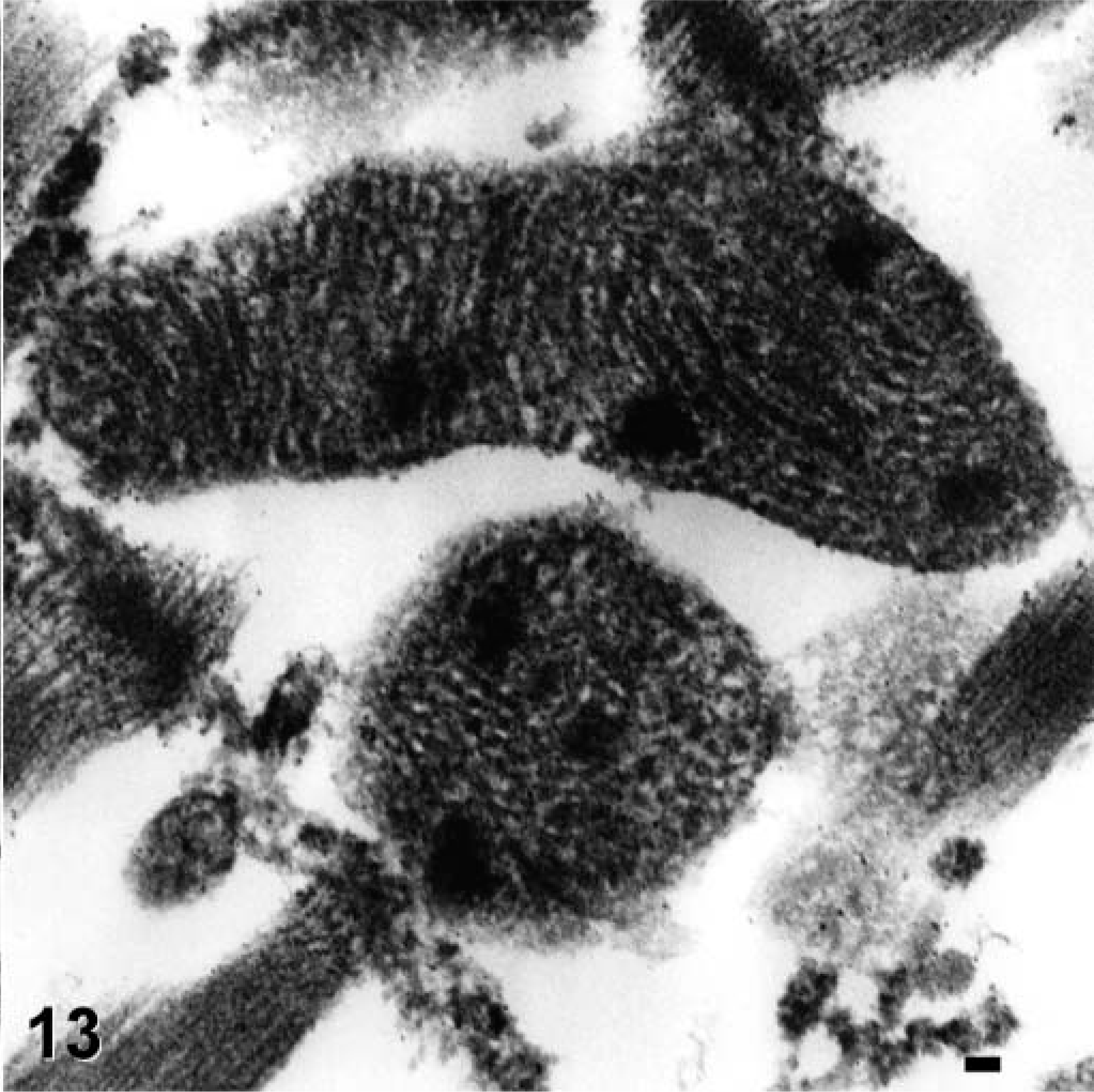
Heart, Jack Russell Terrier. Misshapen mitochondria with tightly stacked cristae and electron-dense inclusions. Electron micrograph of formalin-fixed, paraffin-embedded tissue. Bar = 50 nm.
The clinical signs in this dog including progressive ataxia and deafness that ultimately lead to euthanasia can be accounted for by the extensive mineralization and degeneration of select structures of the cerebellum, medulla, and vestibulocochlear nerve. Mineralization of the brain may be associated with various systemic endocrine, metabolic, or neurodegenerative disorders. In young dogs tissue mineralization is commonly seen as a sequelum of vitamin D3 toxicity, whereas in older dogs the most frequent cause is renal failure with subsequent hyperparathyroidism. In these conditions, however, a systemic metastatic calcification is usually present that was not found in this Jack Russell Terrier puppy. Furthermore, there was no evidence of extraencephalic disease including disturbances of mineral homeostasis, endocrine, or degenerative maladies. Discrete vascular encrustation in the brain is seen in some aged animals of various species such as the horse,10 but this animal was young. Thus, the lesions found in this puppy cannot be easily explained by any condition known in the canine species.
In humans, select mineralization of basal nuclei is commonly seen in MELAS and other mitochondriopathies. Interestingly, the combination of mineral deposits within the media of small- and medium-sized arteries, around capillaries, and free in the neuropil as seen in this case is also a typical feature of MELAS and is thought to arise from mitochondrial dysfunctions in encephalic vascular myocytes and, to a lesser degree, in endothelial cells as well as in neurons.5 A complex composition of the mineral deposits as indicated by positive von Kossa, Turnbull, and PAS stains is also a feature of the entity in humans. The material is thought to represent remnants of degenerated and mineralized mitochondria including iron-containing enzymes of redox pathways.5,8,9 Several additional findings in this puppy are reminiscent of the spectrum of lesions encountered in human mitochondriopathies, especially of the MELAS syndrome. These include the young age (10 weeks) at the onset of clinical signs, hearing loss, and clinical signs of cerebellar dysfunction.4–6 Most importantly, the oncocytotic appearance (so-called eosinophilic granularity) seen in HE-stained sections of several extraencephalic cell types (including hepatocytes and cardiac myocytes) that is caused by an increased number of mitochondria in MELAS and other mitochondriopathies was clearly present in this case. In addition to their increase in number, electron microscopy revealed structural mitochondrial abnormalities typical of mitochondrial dysfunction, such as densely packed cristae and electron-dense inclusions. Similar to MELAS in humans, the presence of mitochondrial abnormalities in these organs need not necessarily be associated with obvious organ pathology as seen in this dog. Clinical signs of myopathy and ragged-red muscle fibers that are a hallmark of mitochondriopathies with muscle involvement (e.g., myoclonic epilepsy with ragged-red fibers) but are usually absent in MELAS were not present. Thus, among the different mitochondriopathies in humans, the lesions in this puppy appear to be most closely related to the MELAS syndrome. But the distribution pattern of mineral deposits in the brain is different from that in MELAS in which the basal nuclei are usually affected.5,6 It is conceivable that this difference may represent sequelae of different mitochondrial or neuronal metabolism in the human and canine species, analogous to differences between the human and murine mitochondrial diseases.8 Alternatively, these differences are well in line with the wide heterogeneity of mitochondriopathies and numerous reports on individual variations and overlaps or transition forms of different mitochondrial syndromes in humans that are thought to arise from a wide spectrum of underlying genetic defects and varying individual modulating factors.4–6,8
Although the clinical and morphologic abnormalities in this Jack Russell Terrier puppy are clearly consistent with mitochondrial encephalopathy, not all questions can be addressed with the data obtained. None of the previously reported suspected cases of mitochondriopathy in dogs1–3,7 resembled the symptoms in this Jack Russell Terrier, and thus a mitochondriopathy was not suspected clinically. Therefore, the lactate and pyruvate levels that may help to diagnose MELAS in humans were not measured in the serum or cerebrospinal fluid. As a further consequence, appropriate unfixed tissues were not collected for enzyme histochemistry, such as the succinate dehydrogenase stain, which would have been helpful in further characterizing mitochondrial abnormalities. Thus, awareness of mitochondrial disorders among veterinary neurologists and pathologists is an essential prerequisite for the diagnosis and more comprehensive studies of the clinical features and morphology in future cases of mitochondriopathies in dogs and other domestic animals. Besides functional studies, the ultimate confirmation of mitochondrial disorders in dogs will require DNA sequence information of the affected individuals as well as exhaustive knowledge of the respective nuclear and mitochondrial DNA sequences and normal allelic variations between individuals and between different breeds, all of which is unavailable at this point.
Footnotes
Acknowledgements
We thank Käthe Franke and Kerstin Rohn for their excellent technical assistance.
