Abstract
The mountain peacock pheasant (Polyplectron inopinatum), the Malayan peacock pheasant (Polyplectron malacense), and the Congo peafowl (Afropavo congensis) are all listed as vulnerable to extinction under the International Union for Conservation of Nature Red List of Threatened Species. Here the authors report fatal infection with a novel herpesvirus in all 3 species of birds. DNA was extracted from the livers of birds with hepatocellular necrosis and intranuclear eosinophilic inclusions consistent with herpesvirus infection. Based on degenerate herpesvirus primers and polymerase chain reaction, 220– and 519–base pair products of the herpes DNA polymerase and DNA terminase genes, respectively, were amplified. Sequence analysis revealed that all birds were likely infected with the same virus. At the nucleotide level, the pheasant herpesvirus had 92% identity with gallid herpesvirus 3 and 77.7% identity with gallid herpesvirus 2. At the amino acid level, the herpes virus had 93.8% identity with gallid herpesvirus 3 and 89.4% identity with gallid herpesvirus 2. These findings indicate that the closest relative to this novel herpesvirus is gallid herpesvirus 3, a nonpathogenic virus used widely in a vaccine against Marek's disease. In situ hybridization using probes specific to the peacock pheasant herpesvirus DNA polymerase revealed strong intranuclear staining in the necrotic liver lesions of an infected Malayan peacock pheasant but no staining in normal liver from an uninfected bird. The phasianid herpesvirus reported here is a novel member of the genus Mardivirus of the subfamily Alphaherpesvirinae and is distinct from other galliform herpesviruses.
Herpesviruses are among the largest and most complex of viruses. Their double-stranded DNA genome, ranging between 125 and 250 kilobases, is encapsulated within an icosahedral protein capsid and a tegument matrix. 6,7,13 Viral DNA replication and assembly of capsid structures occurs in the nucleus of the cells, and viral particles become enveloped by lipids as they transit out the nucleus. 2 In some host cells, the virus can remain dormant for long periods through the active transcription of genes associated with latency. During symptomatic or lytic infection or reactivation, the production and release of the virions result in destruction and necrosis of the host cells. The virions are subsequently released, leading to infection of other cells. In nature, each virus is associated with a distinct host species, suggesting that herpesviruses coevolve with their hosts over long periods and are thus extremely well adapted to them. 6,13
Herpesviridae are divided into 3 distinct subfamilies—Alphaherpesvirinae, Betaherpesvirinae, and Gammaherpesvirinae—based on biological and molecular properties and genomic sequence divergence. 6,13 Alphaherpesviruses have a more rapid cytolytic productive cycle than β- and gammaherpesviruses. 8 All taxonomically assigned avian herpesviruses belong to the Alphaherpesvirinae subfamily and are grouped in the Iltovirus or Mardivirus genus. 20 Phylogenetic estimates suggest that avian iltoviruses appeared about 200 million years ago and that avian mardiviruses and mammalian simplex viruses appeared between 100 and 150 million years ago. 13 Mardiviruses—such as gallid herpesvirus 2 (GaHV-2, or Marek's disease virus 1), gallid herpesvirus 3 (GaHV-3, or Marek's disease virus 2), and meleagrid herpesvirus 1 (MeHV-1)—and iltoviruses (notably, gallid herpesvirus 1 [GaHV-1]) have been characterized in several species of gallinaceous birds. 1,11,17,20 Herpesvirus infections have also been reported, causing disease in peacock pheasants and Congo peafowl; the same gross and microscopic lesions seen in pheasants were produced in healthy chickens inoculated with the pheasant viral isolate. 14 Work by Gunther et al attempted to classify a tragopan herpesvirus using restriction endonuclease analysis; however, the unique restriction pattern exhibited by the virus revealed a high dissimilarity to other known avian herpesviruses. 9 To date, no studies have taxonomically classified a pheasant herpesvirus based on molecular phylogeny. 9,14 We describe the molecular characterization of a novel alphaherpesvirus of the genus Mardivirus that resulted in death in 3 species of pheasants.
Materials and Methods
Animals and Histology
Archived pathology records for gallinaceous birds from the Wildlife Conservation Society collection that died between 1994 and 2002 were reviewed. Birds with a histologic diagnosis of hepatocellular necrosis with intranuclear eosinophilic inclusions suggestive of herpesvirus infection met the inclusion criteria. These cases included 8 mountain peacock pheasants (case Nos. 1–4, 8–10, 12), 3 Malayan peacock pheasants (case Nos. 5, 6, 11), and 1 Congo peafowl (case No. 7).
The pheasants were all housed at the Bronx Zoo, New York, singly or in pairs with conspecifics. Some were cohoused in the same enclosures with or adjacent to other genera of birds. Enclosures were located in 2 buildings, and birds were occasionally moved between those buildings to achieve management goals. Birds were housed in enclosures with free access to indoor or outdoor environments with the exception of periods of inclement weather during which birds were housed indoors. No common exposure to other taxa or individual birds was found in affected birds. No recent introductions of birds into adjacent enclosures occurred. Affected birds either died or presented acutely with nonspecific clinical signs. Supportive treatment, including subcutaneous fluids, antibiotics and vitamins, were administered to early cases. After herpesvirus infections were documented, antiviral treatment with acyclovir was initiated as well. Treatment did not affect outcome. Upon death, each bird was submitted for gross necropsy, and tissue from the liver, spleen, intestines, kidneys, brain, adrenals, thymus, lungs, bone marrow, heart, tongue, trachea, crop, ventriculus, proventriculus, eyes, and pancreas was fixed in 10% formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin for routine bright-field microscopy. Two Malayan peacock pheasants that died of noninfectious causes, including trauma and tracheal obstruction (case Nos. 13 and 14), were used as nonherpes infection controls for histology and in situ hybridization.
Herpesvirus Polymerase Chain Reaction
To determine if the diseased pheasants were infected with herpesviruses, DNA was extracted from a total of 25 μm of formalin-fixed paraffin-embedded liver tissue (5 sections × 5 μm each) using the QIAamp DNA extraction kit (Qiagen Inc., Valencia, California). The DNA was subjected to nested polymerase chain reaction (PCR) using degenerate primers designed to detect herpesvirus DNA polymerase and DNA terminase. DNA extracted from livers of 2 normal Malayan peacock pheasants (case Nos. 13 and 14) was used as negative controls. Primers were purchased from Eurofins (Eurofins MWG Operon, Huntsville, Alabama), and all PCR reagents were purchased from Qiagen unless otherwise specified. Nested PCR was used to amplify 2 regions of DNA terminase—first round, 519 base pairs (bp); second round, 419 bp 5 —and a 220-bp fragment of DNA polymerase. 21 Human herpesvirus 1 DNA was used as a positive control. The following reagents were added to each 50-μl PCR reaction: 5 μl of extracted DNA (> 20 ng/μl) for the first round (or 5 μl of primary PCR mixture for the second round), 1μM each primer, 2mM MgCl2, 200μM each dNTP, 1.5 U of Hotstart Taq Polymerase, 5 μl of 10× PCR buffer, and 5% DMSO (Sigma-Aldrich, St Louis, Missouri). Cycling parameters for both methods of PCR used an initial denaturation at 95°C for 5 minutes, followed by 45 cycles of 94°C for 20 seconds, 46°C for 60 seconds, 72°C for 60 seconds, and a final extension at 72°C for 10 minutes. Primary and secondary PCR products of correct molecular weight (220 bp for DNA polymerase and 519 bp for DNA terminase) were spliced out of the agarose gel and purified using the MinElute Gel Extraction Kit (Qiagen), followed by direct sequencing in both directions using ABI 3730x/DNA analyzers for capillary electrophoresis and fluorescent dye terminator detection (Genewiz Inc., South Plainfield, New Jersey).
Nucleotide and protein sequences were submitted to BLASTn and BLASTx to identify sequence homologies within the Genbank database (GenBank, National Center for Biotechnology Information). Sequences from the herpesvirus-positive peacock pheasants (n = 8) and the positive peacock (n = 1) were aligned at the nucleotide and protein level (Geneious Pro 5.1.7, Biomatters Ltd., Auckland, New Zealand).
Phylogenetic Analysis
To determine the phylogenetic relationship of peacock herpesvirus to described alpha-, beta-, and gammaherpesviruses, protein sequences from representative species were retrieved from GenBank and aligned with the translated Malayan peacock (case No. 11) herpesvirus DNA terminase or DNA polymerase sequence using the Geneious alignment tool (Geneious Pro 5.1.7). Sequences for comparison were trimmed to 162 amino acids for the DNA terminase and 46 amino acids for the DNA polymerase. To obtain pairwise identities among herpesvirus species, the protein alignment for DNA terminase was run through PAUP (Phyogenetic Analaysis Using Parsimony 4b10osx, Sinauer Associates, Inc., Publishers, Sunderland, Massachusetts) to generate a running P-distance pairwise comparison matrix. Bayesian analysis was performed using MrBayes 3.1 plug-in on Geneious Pro using gamma-distributed rate variation and a Poisson rate matrix. 10 The first 25% of a 1 100 000 chain length was discarded as burn-in, and 4 heated chains were run with a subsampling frequency of 200 (GenBank accession numbers for the DNA terminase listed later). The nucleotide sequence for phasianid herpesvirus DNA terminase was deposited in GenBank under accession No. JN127369. For DNA polymerase, GenBank accession numbers are as follows: AF168792_3, GaHV-1; ACR02982, GaHV-2; BAB16540, GaHV-3; HVT038, MeHV-1; ABM92130, anatid herpesvirus 1; AAU88096, cercopithecine herpesvirus 2; NP_851890, macacine herpesvirus 1; YP_443877, papiine herpesvirus 2; CAA26941, human herpesvirus 1; ADM52140, human herpesvirus 2; AAS45914, equid herpesvirus 1; ACT88328, felid herpesvirus 1; AAN64026, psittacid herpesvirus 1.
In Situ Hybridization
Probes to a 151-nucleotide sequence of DNA polymerase from Polyplectron malacense malacense and an 872-nucleotide sequence of β-actin from Maleagris gallopavo, used as an internal housekeeping gene control, were hybridized to formalin-fixed paraffin-embedded sections (5 μm) from infected or noninfected Malayan peacocks (Affymetrix, QuantiGene ViewRNA, Santa Clara, California). Sections on positively charged glass microscope slides (Surgipath Medical Industries, Inc., Richmond, Illinois) were heated at 60°C for 30 minutes in a Statspin Thermobrite slide hybridizer (Iris Sample Processing, Westwood, Massachusetts) to increase adhesion of the tissue to the slide. The slides were then fixed in 10% neutral buffer formalin solution for 1 hour and washed twice with phosphate buffered saline (PBS) and allowed to air-dry. The slides were subsequently heated at 80°C for 3 minutes to melt the paraffin, which was dissolved and removed using citrate. The slides were washed in 95% ethanol and air-dried, submerged in 1× pretreatment solution (Affymetrix) at subboiling temperatures (between 95°C and 99°C) for 10 minutes to unmask the DNA and then transferred to deionized water and washed twice. Prewarmed slides were treated for 10 minutes in protease solution (Affymetrix) to allow for target accessibility at 40°C and washed twice with PBS. Slides were fixed in a 4% formalin solution for 5 minutes and washed in PBS. Slides were then incubated for 3 hours with the appropriate probes (β-actin or peacock herpes) diluted in Hyb A buffer (Affymetrix) at 40°C in a humidified Thermobrite slide hybridizer. Slides were washed 4 times in wash buffer and then incubated for 25 minutes at 40°C in PreAmp1 (preamplifier) diluted in prewarmed Hyb B buffer (Affymetrix). Slides were washed 3 times in wash buffer and then incubated in Amp 1 (amplifier) in prewarmed Hyb B buffer for 15 minutes at 40°C. Slides were again washed 3 times and incubated in LP-AP (hybridize label probe) in prewarmed Hyb C buffer (Affymetrix) for 15 minutes at 40°C. Slides were washed again 3 times in wash buffer and incubated for 10 minutes in AP-Enhancer solution (Affymetrix) at room temperature. After decanting the enhancer solution, fast-red substrate (Affymetrix) was added to slides and incubated at 40°C for 30 minutes in the dark. After the incubation, the slides were washed in PBS and fixed in 4% formalin for 5 minutes at room temperature. Slides were counterstained with DAPI (4′,6-diamidino-2-phenylindole) fluorescent stain (Sigma-Aldrich), mounted, and sealed using nail polish. Sections were viewed using bright-field and fluorescent microscopy (Zeiss Axio fluorescent microscope, Scope AI, equipped with a Jenoptic ProgResC5 CCD camera; or Nikon Eclipse E600 fluorescent microscope equipped with a CFI Plan Fluor 100× oil objective, 12V/100W mercury lamp, and DVC 2000C high-resolution CCD camera). Images were acquired either using Mac Capture Pro 2.6.0 (Jenoptik, Jena, Germany) or Stereo Investigator (MBF Bioscience, Williston, Vermont).
Results
Between 1994 and 2002, 12 pheasants from 3 species—Polyplectron inopinatum (mountain peacock pheasant), Polyplectron malacense (Malayan peacock pheasant), and Afropavo congensis (Congo peafowl)—died acutely with multifocal necrotizing hepatitis with hepatocellular eosinophilic inclusions (Figs. 1–4) and necrotizing splenitis, after presenting briefly with clinical signs of lethargy and anorexia. Necrotizing enteritis with intraepithelial eosinophilic intranuclear inclusions was seen in 1 Malayan peacock pheasant and 3 mountain peacock pheasants.
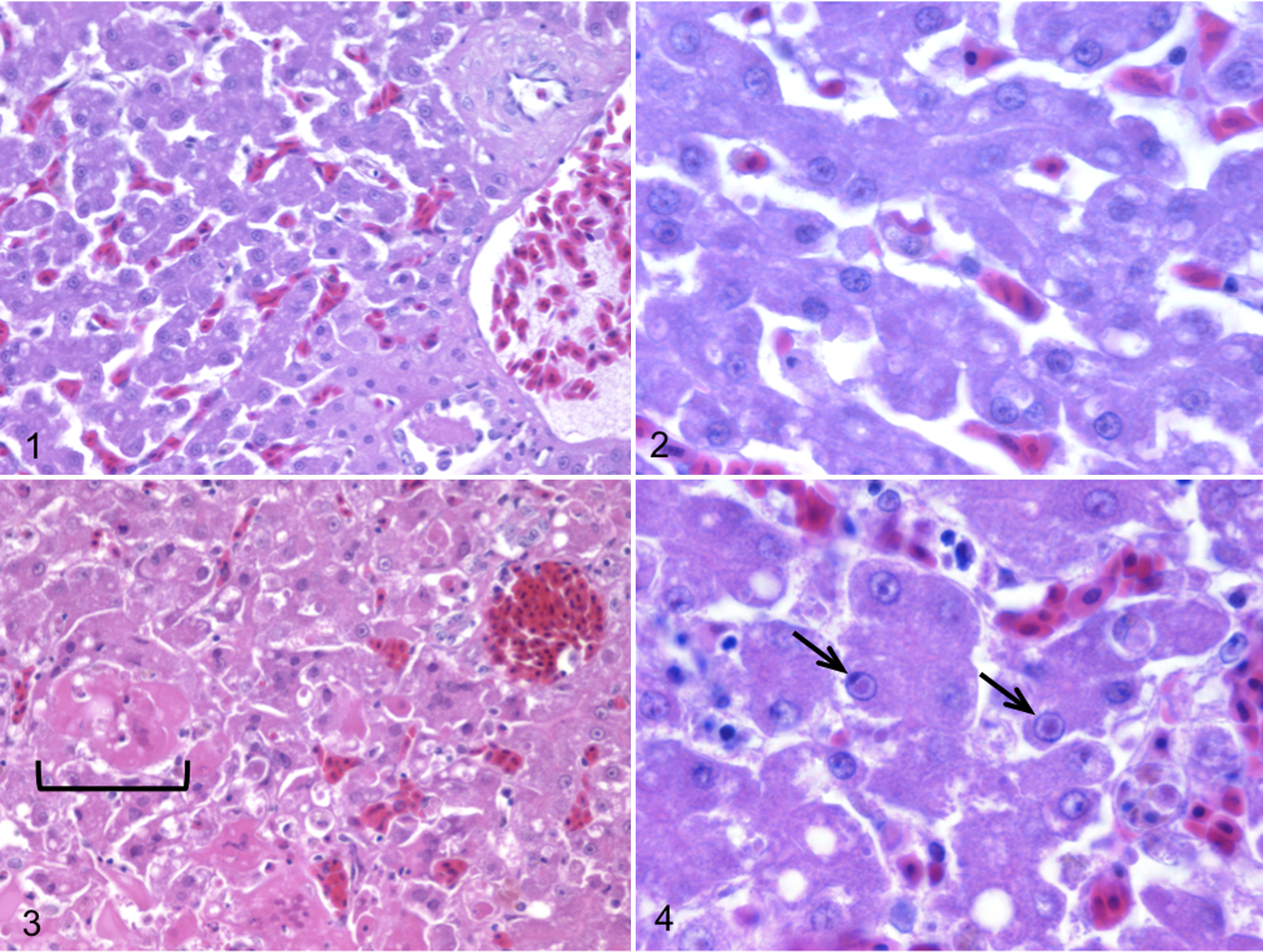
Liver, case No. 14; normal Malayan peacock pheasant. HE.
DNA samples were subjected to PCR analysis using degenerate primers specific for the DNA polymerase (UL30) and DNA terminase genes commonly found in all members of Herpesviridae. The nucleotide sequences of these PCR products were determined from 9 of 12 samples from symptomatic birds (case Nos. 1–3, 5–7, 10–12). In the other 3 cases, only the 220-bp product of the DNA polymerase was successfully amplified (case Nos. 4, 8, 9). The DNA sequences were translated, and a 162–amino acid sequence of the DNA terminase gene and a 46 amino acid sequence from the DNA polymerase gene were obtained. Viral DNA terminase sequences were nearly identical in all birds, indicating infection with the same herpesvirus (Fig. 5). The Malayan peacock pheasant DNA terminase had 92% identity at the nucleotide level with GaHV-3 and 77.7% identity with GaHV-2 (BLASTn). Figure 6 shows protein alignments comparing a translated 164–amino acid sequence of DNA terminase from the Malayan peacock herpesvirus with representative alpha-, beta-, and gammaherpesviruses sequences obtained from GenBank (accession numbers indicated). Bayesian phylogenetic analysis of the multiple protein alignment was performed and representative sequences clustered into the expected subfamilies for alpha-, beta-, and gammaherpesviruses. Based on the phylogenetic analysis, the peacock pheasant herpesvirus (phasianid herpesvirus) belonged to the Mardivirus genus in the Alphaherpesvirinae subfamily, and is most closely related to GaHV-3 (Fig. 7a). An additional phylogenetic analysis performed on a 46–amino acid multiple-protein sequence alignment of DNA polymerase within alphaherpesviruses resulted in a similar phylogenetic tree (Fig. 7b). Pairwise identity for DNA terminase was determined for all species in the protein alignment (Fig. 8). PAUP distance matrix analysis results show that the phasianid herpesvirus is 93.8% identical to GaHV-3 and 89.4% identical to GaHV-2, 84.4% identical to MeHV-1, and 74.4% to Anatid herpesvirus at the amino acid level. Although identity as high as 94% is not seen between other known mardiviruses, this level of identity is seen in other groups of alphaherpesviruses, such as simplex viruses in primates (Fig. 8). For example, cercopithecine herpesvirus 2 shares 95.6% identity with human herpesviruses 1 and 2, and papiine herpesvirus 2 shares 94.9% identity with human herpesvirus 2.
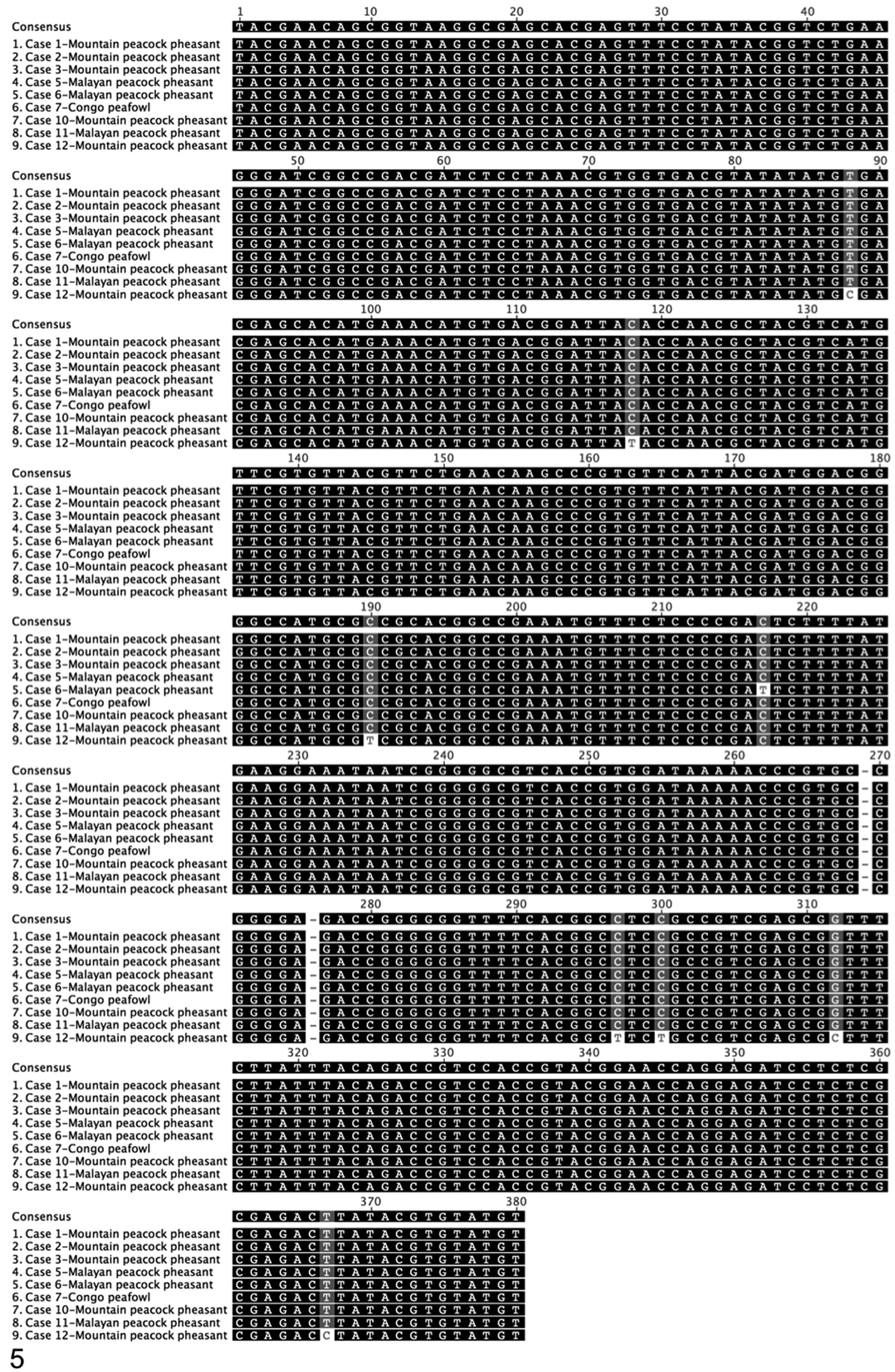
Consensus sequence and nucleotide alignment of a 380–base pair sequence of herpesvirus of DNA packaging subunit 1 gene (DNA terminase). Nucleotides indicated in black indicate 100% similarity; dark gray, 80%–100% similarity in the alignment; light gray, 60%–80% similarity; and white, less than 60% similarity using an identity score matrix (Geneious Pro).

Protein alignment of a 163–amino acid sequence of DNA terminase. Amino acids indicated in black indicate 100% similarity; dark gray, 80%–100% similarity; light gray, 60%–80% similarity; and white, less than 60% similarity using an identity score matrix (Geneious Pro). GenBank accession numbers used for all species precede the names of the herpesvirus species.
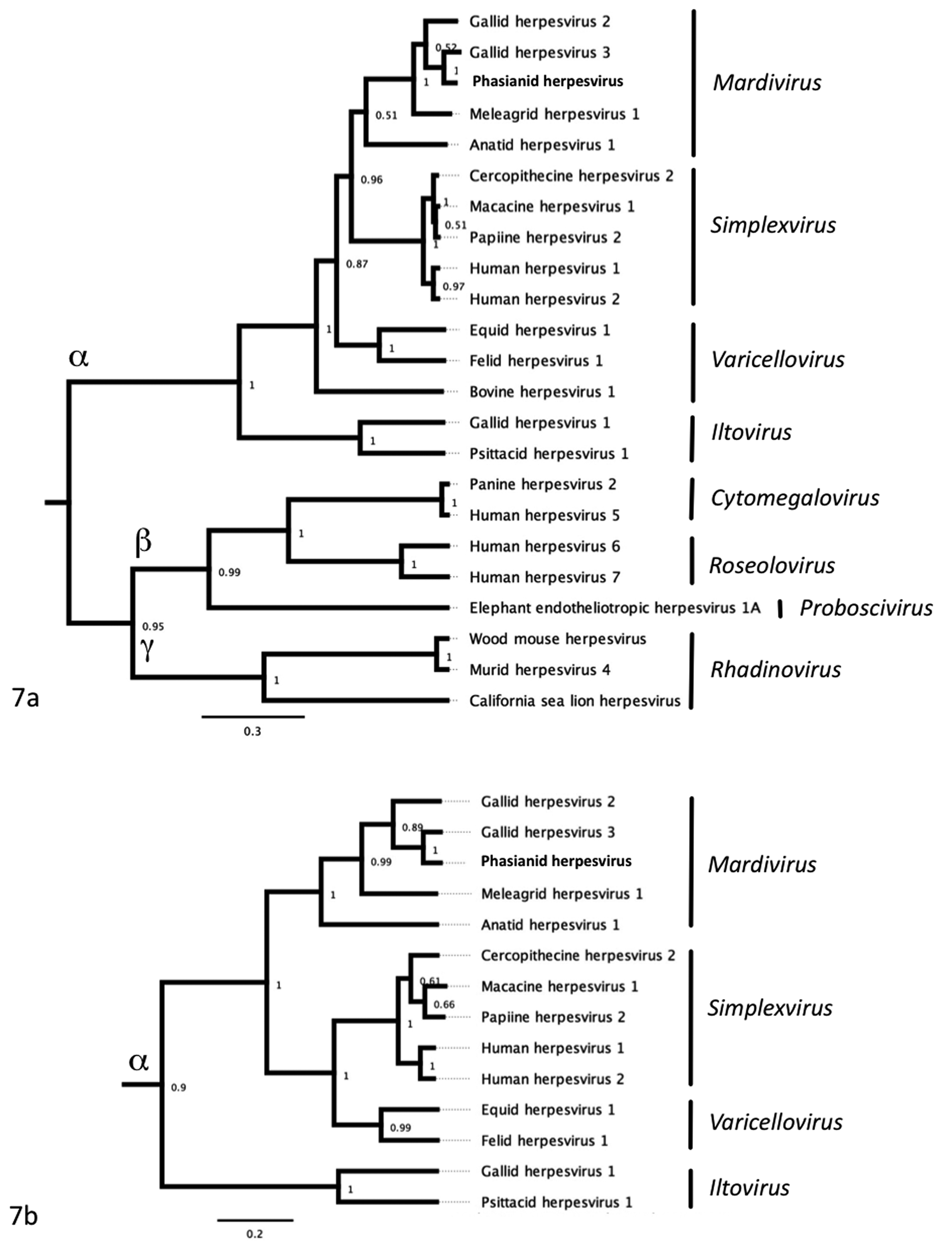
Bayesian phylogenetic tree of (a) herpesvirus DNA packaging subunit 1 gene (UL-15, DNA terminase) and (b) the catalytic subunit of DNA polymerase amino acid sequences from the Malayan peacock pheasant and representative sequences obtained from GenBank. Sequences were aligned using Geneious Pro. Bayesian posterior probabilities of branching demonstrate the robustness of the individual clades.
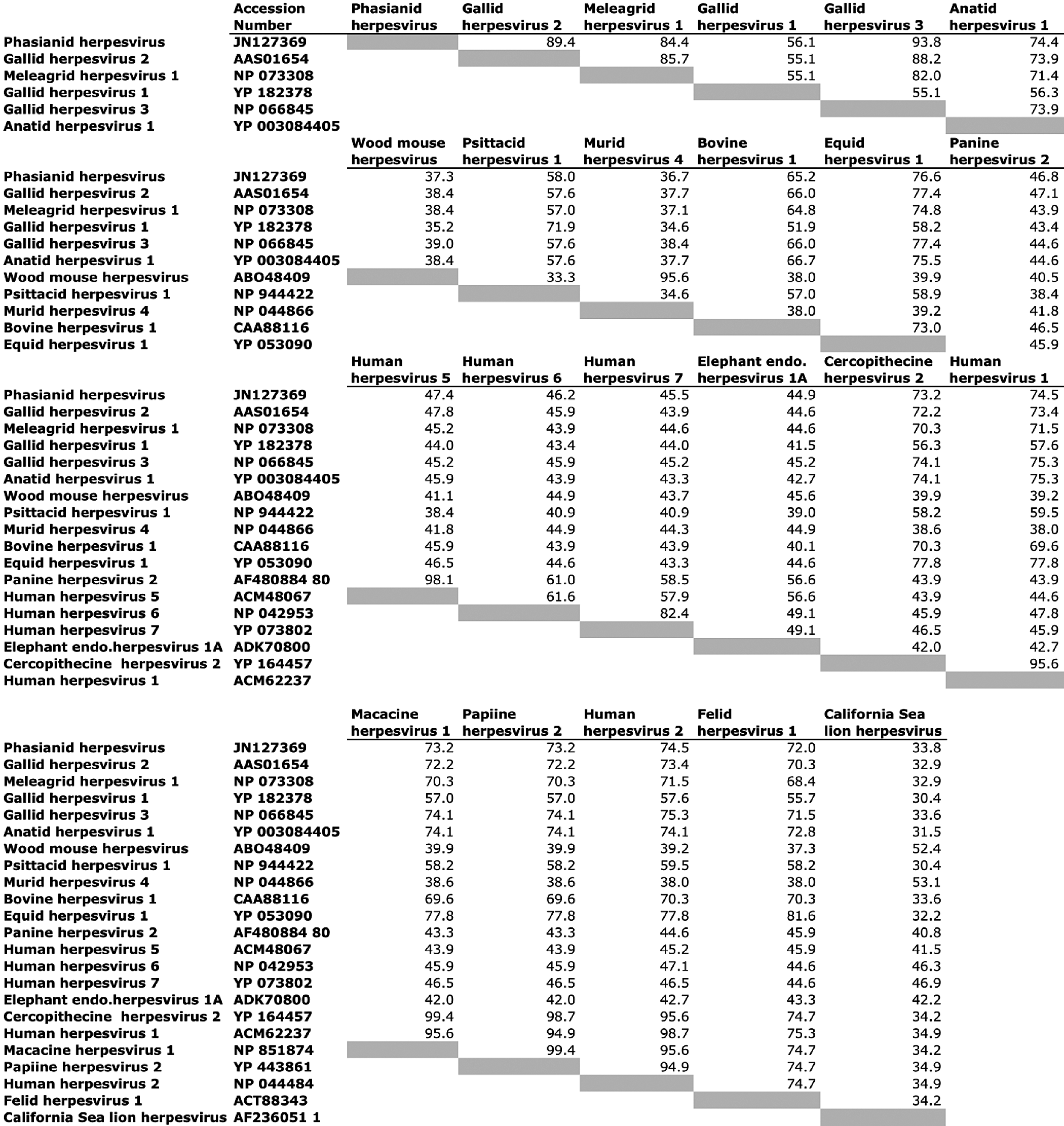
Pairwise identity of amino acids among different species of herpesviruses, generated from a pairwise distance matrix calculated with PAUP software.
The distribution of the phasianid herpesvirus nucleic acid in liver sections from Malayan peacocks with and without disease was assessed using branched in situ hybridization (Quantigene View RNA, Panomics), employing probes that hybridize to the mRNA of Malayan peacock pheasant herpesvirus DNA polymerase or a housekeeping gene, β-actin, as a control (Figs. 9–11). Signal for β-actin was observed in Malayan peacock pheasants with or without disease; however, the intensity of the hybridization signal was lower in the bird with disease. In contrast, hybridization signal with peacock herpesvirus probes was detected only in the bird with disease. The nuclear distribution of herpesvirus DNA polymerase was determined using DAPI staining.
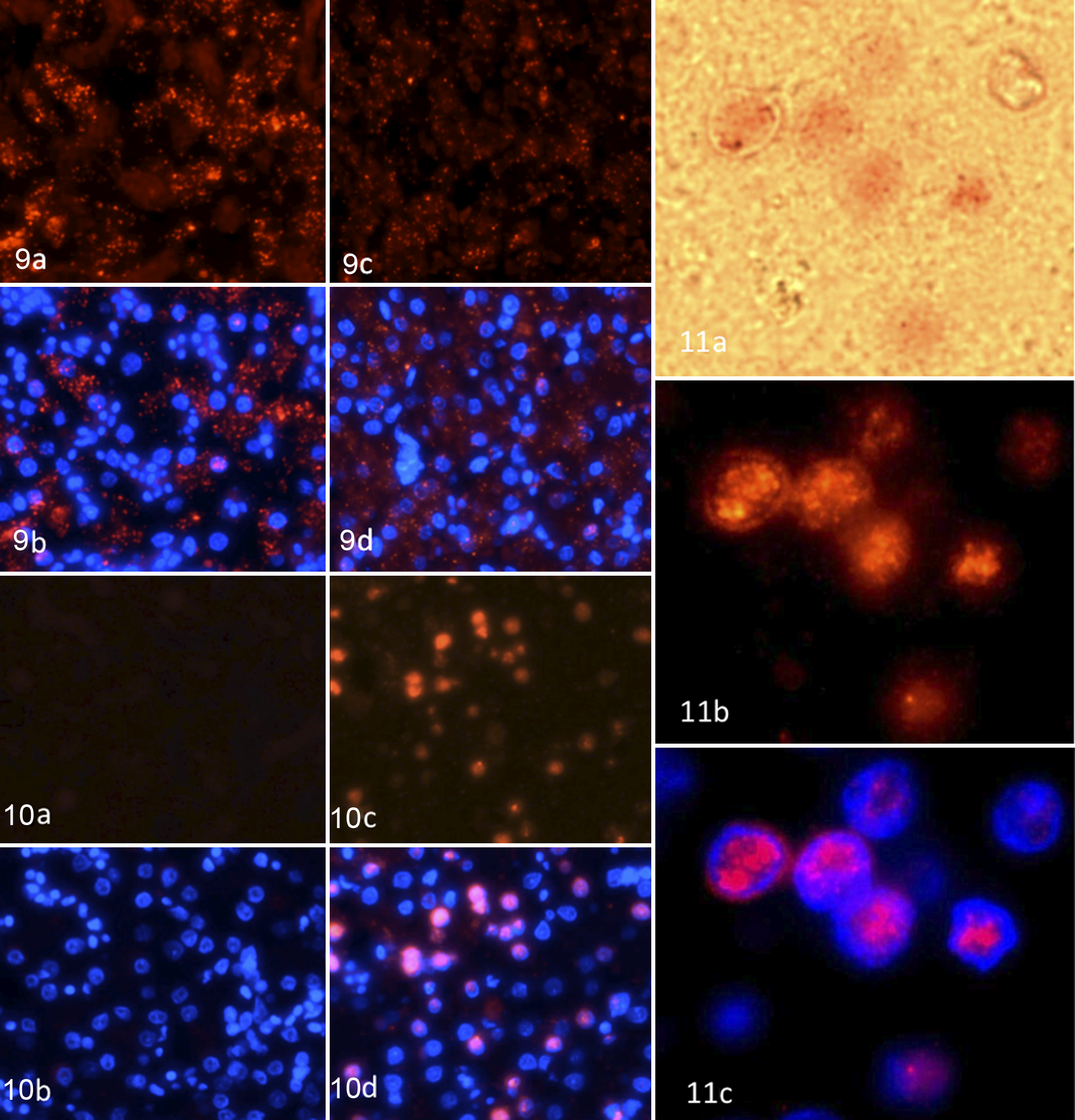
Liver, case No. 14; in situ hybridization for β-actin fluorescent staining (a) or β-actin and DAPI costaining (b) of normal Malayan peacock pheasant. The tissue was counterstained with DAPI (blue) to identify the nuclei, and images were merged to show the localization of β-actin and DNA polymerase relative to the nuclei. Liver, case No. 11; β-actin fluorescent staining (c) or β-actin and DAPI costaining (d) in herpesvirus-infected Malayan peacock pheasant.
Discussion
Our results support the identification of a novel alphaherpesvirus, tentatively named phasianid herpesvirus, in the genus Mardivirus. This virus was associated with severe disease and death in 3 species of pheasants. Chicken inoculation studies of the herpesvirus affecting the exotic pheasants in this zoological collection performed in 1996 produced the same gross and microscopic lesions seen in the naturally infected pheasants. 14 However, at that time, molecular characterization was not performed. 14
Phylogenetic and distance matrix analysis indicates that the closest relative to phasianid herpesvirus is GaHV-3, a genetically similar, nonpathogenic virus widely used as a vaccine against Marek's disease. 18,19,22 A neoplastic lymphoproliferative disease of gallinaceous birds, Marek's disease produces visceral and/or cutaneous lymphoma, immunosuppression, paralysis, and/or death. 17 The causative agent, GaHV-2, is distributed worldwide and is associated with severe economic losses in domestic poultry. 15,17 The disease has been reported in pheasants, quail, and game fowl. 16 GaHV-2 is closely related to but taxonomically distinct from GaHV-3 and MeHV-1. 11 –13
GaHV-3 strains are considered to be widely prevalent in the poultry industry, replicate in the host, and spread easily by contact. 4,18,19 Live vaccines for Marek's disease include GaHV-3, MeHV-1, and attenuated GaHV-2. However, the long-term effects of live vaccine strains such as MeHV-1 and GaHV-3 in other nondomesticated galliform species, particularly species that are cohoused in zoological or captive collections and farms, are largely unknown.
Herpesviruses are theorized to have coevolved with their hosts, and the severity of the lesions observed in the liver and spleen and the presence of the disease in 3 species of pheasants suggest that interspecies transfer of the virus may have occurred from a yet-unidentified host galliform species. Vaccination of birds against Marek's disease has not been performed in the Wildlife Conservation Society collection since 1985, and none of the pheasants in the collections were housed near vaccinated domestic poultry. In an effort to determine when this herpesvirus entered the collection, we retrospectively examined earlier cases of suspected herpesvirus infection and identified histologic hepatic necrosis with intranuclear eosinophilic inclusions consistent with herpesvirus infection in a Temminck’s tragopan that died in 1988. PCR analysis revealed DNA polymerase products identical to phasianid herpesvirus. However, we were unable to amplify DNA terminase gene products.
The mountain peacock pheasant (P. inopinatum), the Malayan peacock pheasant (P. malacense), and the Congo peafowl (A. congensis) are all listed as vulnerable to extinction under the International Union for Conservation of Nature Red List of Threatened Species due to decreasing population trends resulting from increased habitat fragmentation and deforestation, which are compounded by the bird’s restricted range. 3 In the eastern Democratic Republic of the Congo, the Congo peafowl population is small and scattered, and critical habitat is being lost to mining, subsistence agriculture, and logging. 3 Population growth is hindered by the long history of political conflict, war, increasing human population pressures, and poaching. 3 Maintaining healthy birds in captivity provides a conservation option for each species and ensures that viable breeding populations exist in the event that species rescue or recovery efforts are required. Future research aimed at testing the safety and efficacy of vaccination of pheasant species with closely related nonpathogenic strains of mardiviruses to protect against pathogenic herpesvirus infection will be beneficial for captive management and as a safeguard for these valuable species.
Footnotes
Acknowledgements
This work was supported by funds from the National Institutes of Health (NIAID AI57158 to W.I.L.), NIAID AI070411 to W.I.L.), and the US Department of Defense. We also acknowledge Dr Tracey McNamara and the support of the clinical, pathology, and ornithology departments at the Wildlife Conservation Society.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
