Abstract
Over the past 20 years, the conception of brain development has radically changed from a fixed and limited hierarchical process to a more plastic and continuous one. Most surprising, the field has learned that postnatal neurogenesis is not just a seasonal phenomenon in songbirds but a process that occurs across species and seasons. Astrocytes, whose primary role in the central nervous system was thought to be strictly supportive, have emerged as a heterogeneous population, a subset of which is the neural stem cell. Postnatal neurogenesis persists in specialized niches within the rostral subventricular zone and hippocampal dentate gyrus and, for a limited period, within the white matter tracts and external granular layer of the cerebellum. These specialized microenvironments are influenced by factors in the blood, cerebrospinal fluid, and local extracellular matrix. This article reviews the current understanding of adult neurogenesis, which is conserved across many vertebrate species, underscoring the value of animal models in past and present studies of human neurogenesis and neurogenic disease.
Keywords
In the 1960s, Altman was among the first to document neurogenesis within the postnatal rodent central nervous system (CNS). 2–4 Despite this body of work, postnatal neurogenesis was not widely accepted until 2 decades later when Nottebohm demonstrated neuronal replacement in the adult avian forebrain. 42,77 While evidence of postnatal neurogenesis accumulated in avian and mammalian species, another 15 years elapsed before adult neurogenesis was recognized in humans. 32 Thirty years after Altman’s first descriptions of adult neurogenesis, neural stem cells (NSCs) were finally isolated from the adult mammalian brain by selective response to epidermal growth factor (EGF) and basic fibroblast growth factor (FGF2). 94 ,95 Subsequently, multipotent NSCs have been isolated from the postnatal subventricular zone (SVZ) and its rostral and caudal extensions into the olfactory bulb and hippocampus, 67 ,68,84 as well as within the white matter tracts of the postnatal rodent cerebellum. 59 ,64,128,129 In addition, glial progenitors in regions not associated with neurogenesis demonstrate multipotency when exposed to appropriate growth factors in vitro. 9 ,60,78,83,105
The identification of neural stem and precursor cells in the adult brain broaches the possibility of regenerative medicine to improve brain function, learning, and memory. Apart from the ethical and moral implications associated with human embryonic research, postnatal NSCs are more readily available than embryonic stem cells for basic stem cell research and therapy and afford the opportunity for autologous stem cell transplantation.35, 46, 59, 64, 71, 79, 108, 119 In addition to replacement of dead or dying neural tissue, NSCs have the potential to elaborate trophic factors to rescue dysfunctional endogenous neurons, as well as inhibit inflammation in the transplanted areas. 31,81,89
Identification of postnatal NSCs and the environments in which they reside can provide insight into the pathogenetic mechanisms of various neurodegenerative and neoplastic diseases through in vitro and in vivo studies. To date, neural stem or precursor cells have been successfully isolated from rodents, primates, sheep, pigs, cats, and dogs.17,30,71,79,82,84,94,111,121–123,130 In vitro and in vivo studies of NSCs from multiple animal species have demonstrated remarkable species conservation of phenotype and neurogenic mechanisms. 21,27,41,67,97,124 Accordingly, rodents, domesticated animal species, and nonhuman primates are valuable models for human neurogenesis and neurogenic disease and are used in investigative and therapeutic studies of human neurologic diseases.
Neural Precursor Cells
NSCs are rigorously defined as multipotent, capable of extended self-renewal, and able to generate a large number of progeny. 93 ,101 Cells that demonstrate only multipotency, or are bipotent or unipotent, are more appropriately termed neural progenitor cells. 20,101 Neural stem and progenitor cells are both present in postnatal neurogenic regions. Thus, for the purposes of this review, unless a population is known to conform to the definition of a stem cell, the term neural precursor cell (NPC) is used to include both neural stem and progenitor cell populations.
Neurogenesis within the CNS occurs in the early postnatal period in the cerebellar external granule layer (EGL), 34,88 whereas routine adult neurogenesis is thought to occur only in the SVZ/olfactory bulb and subgranular zone (SGZ) of the hippocampal dentate gyrus. 131 Extra-CNS neurogenesis, such as that which occurs in the nasal mucosa, 8,53 is beyond the scope of this review. In the CNS of mice, adult neurogenesis maintains and reorganizes the olfactory bulb interneuron network, modulates and refines existing neuronal circuits in the dentate gyrus, and is required for hippocampal-dependent learning and memory. 22,49
Only within the past decade has the in vivo identity of postnatal NSCs gradually been ascertained. The origin of adult SVZ NSCs is proposed to be the ventricular zone (VZ) neuroepithelial (NE) cell. In this view, NSCs persist from development through adulthood, undergoing a morphologic transformation from NE cell to radial glial cell to astrocyte (Fig. 1 ). 24 During fetal development, NE cells of the developing neural tube VZ are NSCs. Similar to radial glia, NE cells have a radial morphology characterized by a bipolar shape with a short apical process and single cilium contacting the ventricle, as well as a long basalar process extending toward the pia. The NE cells also express radial glial markers, such as vimentin, RC1, RC2, nestin, and, in primates, glial fibrillary acidic protein (GFAP). 76 ,90,114 Radial glia arising from the NE cells differentiate into neurons, glia, and ependymal cells. 36 ,70,76,90,114 After birth, radial glia disappear by “differentiating” into astrocytes and ependymal cells. The ependyma replaces the VZ, and astrocytes localize beneath this layer in the SVZ. The adult SVZ lines the entire ventricular system; however, the majority of adult SVZ neurogenesis occurs rostrally, especially in the lateral ventricles of the frontal horn. 18,28
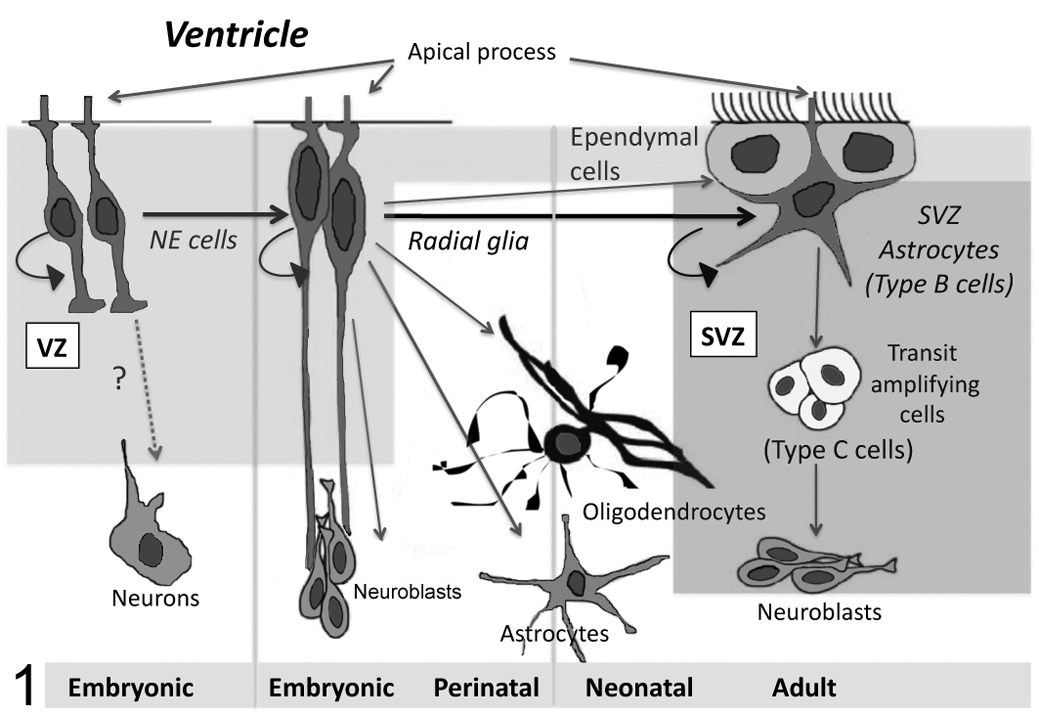
Current conception of neurogenesis in the rostral periventricular region throughout development. The neural stem cell niche shifts from the ventricular zone (VZ) to subventricular zone (SVZ) after birth. NE, neuroepithelial.
It was once thought that with the disappearance of radial glia, the ability for neurogenesis was lost. However, it is now recognized that the radial glia-derived astrocytes in the SVZ have the ability to self-renew as well as differentiate into neurons and glia. Moreover, like NE cells, they share some of the molecular markers and structural features of radial glia, such as the presence of GFAP, contact with the ventricle, and a single cilium extended into the ventricle. 90 ,114 In the lateral wall of the lateral ventricle, the apical process of SVZ astrocytes, occasionally containing a nucleus, squeezes between ependymal cells to allow contact with the ventricle (Fig. 1). Thus, the adult lateral wall ventricular surface actually comprises a mixture of ependymal cells and astrocytic NSCs, mimicking the embryonic VZ in which primary progenitors line the ventricular space. 72,107
The presence of a ventricle-contacting cilium in SVZ astrocytes obfuscated the initial identification of the SVZ stem cell. Ciliated ventricle-contacting ependymal cells were originally proposed to be the adult CNS stem cell. 52 Subsequently, it was shown that the ciliated “true” stem cells were in fact SVZ astrocytes and not ependymal cells. 26 Recent studies suggest that there is a subpopulation of ependymal cells with progenitor cell characteristics and properties in the adult SVZ that produces neurons and astrocytes in response to stroke. 16,19,87 However, this population is not self-renewing under conditions of stroke and therefore would not constitute a true stem cell population. 16
The astrocytic stem cells in the hippocampal SGZ are also derived from radial glia, are GFAP+, retain a radial morphology, and are thus of the neuroepithelial cell–radial glial cell–astrocyte lineage. 104 The GFAP+ astrocytic cells from neurogenic and nonneurogenic regions appear to have distinct identities as NSCs and astrocytes, respectively. 51 In rodent neurogenic regions, the GFAP+ cells with stem cell ability represent a morphologically distinct population of cells with bipolar or unipolar morphology. 38 Subsequent studies demonstrated that GFAP+ cells cultured with leukemia inhibitory factor have a bipolar/tripolar morphology and possess NSC markers and differentiation ability, whereas bone morphogenetic protein-generated GFAP+ cells are astrocytes. 10 Thus, the NSC in developing and adult vertebrates is a cell with markers and structural characteristics of an astrocyte.
In Vitro Propagation
NPCs grow in vitro as free-floating cellular clusters termed neurospheres or as adherent cell cultures (Fig. 2 ). 84 ,91,94,95 It was widely held that a neurosphere represented the clonal expansion of a single stem cell. However, there is evidence that neurospheres may also be generated by progenitor cells. 29 ,93 Thus, the standard neurosphere assay that assesses the ability of single cells to form neurospheres overestimates the proportion of true stem cells in a population. 93
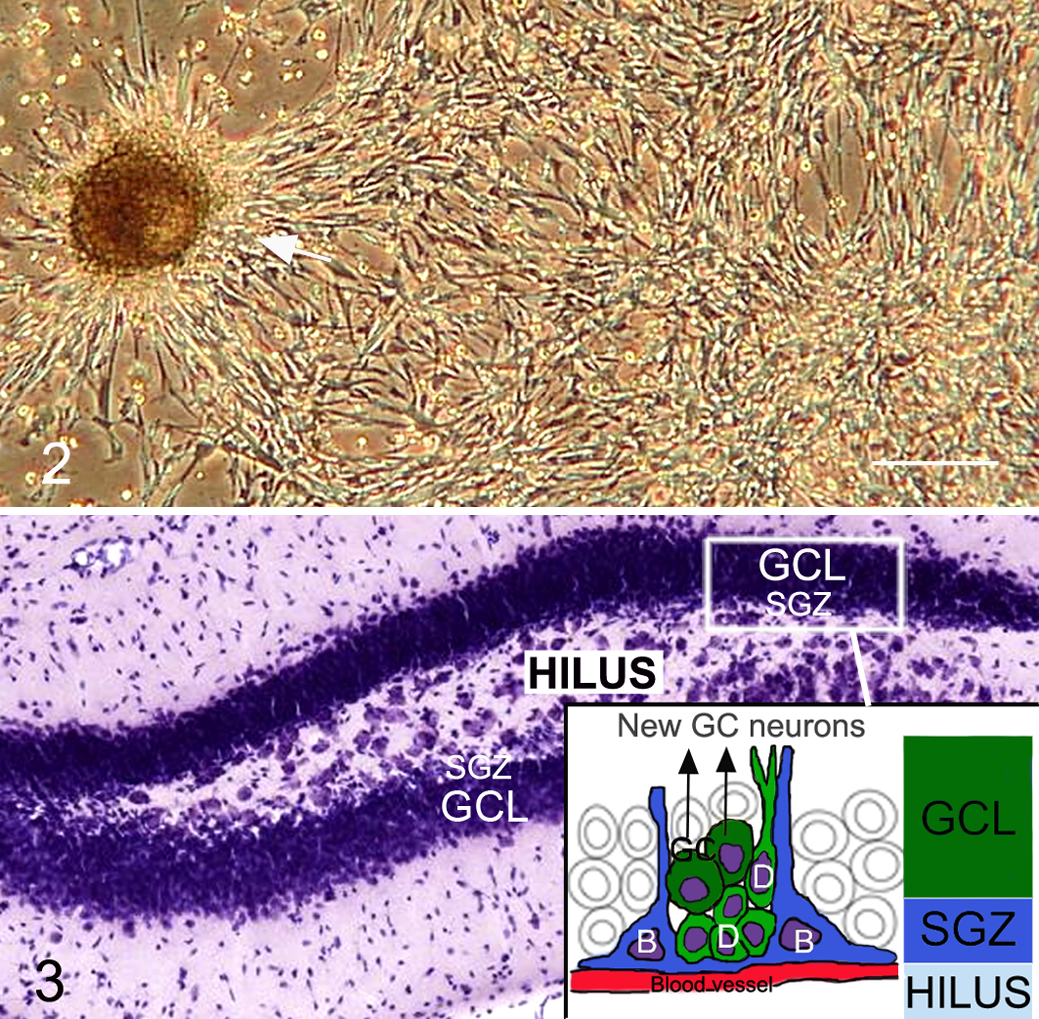
NPCs in the developing and postnatal brain were first isolated by their in vitro response to EGF or FGF2 with heparin, factors known to stimulate precursor proliferation in the developing neuroepithelium. 91 ,93,94,95 Subsequently it was shown that NSCs in the embryonic mouse telencephalic germinal zone are responsive to FGF as early as embryonic day 8.5, but EGF responsiveness does not occur until embryonic day 14.5. 115 This study also suggested that EGF-responsive NSCs were lineage descendants of the FGF-responsive NSCs. Indeed, later studies showed that FGF2 upregulates EGF receptor expression in NPCs. 14 ,66 In support of these findings, exhaustive in vivo evaluation of the EGF-responsive NPC type in the adult mouse SVZ showed that it corresponds to the rapidly cycling SVZ neuronal progenitor cell (Fig. 1; GFAP– type C cell), rather than the true NSC, which is quiescent (GFAP+ type B cell). 29
Protocols for in vitro NPC culture differ with respect to concentration and combination of growth factors and substrates.17,34,56,63,75,84,91,94,95 There is ample evidence that NPC response to growth factors varies not only with the developmental stage but also with factor composition and concentration. 125 ,132 For example, although NPCs grown in either EGF or FGF2 with heparin were multipotential, 2 separate studies showed a preferential gliogenic effect from EGF-derived cultures and a neurogenic effect in cultures derived from FGF2 and heparin. 125 ,132 In one study, 132 this effect was noted in NPCs derived from embryonic and early postnatal rats but not in adults, whereas a second group reported these results for adult rat NPCs. 125 The combination of EGF and FGF2 with heparin, rather than either alone, was shown to have a synergistic effect with embryonic rat and human NPCs and with adult mouse subventricular NPCs. 12 ,13,45,117 Addition of leukemia inhibitory factor or a laminin substrate with EGF and FGF2 had a preferential neurogenic effect in postnatal canine NPC cultures. 122 In light of these findings, direct comparisons of NPCs grown under variable culture conditions must consider the conditions under which the cells are grown. Moreover, optimal growth conditions must be determined for NPCs according to developmental stage, species, and intended use.
Neurogenic Regions
While NPCs have been isolated from multiple areas of the postnatal brain, it is currently under debate whether neurogenesis occurs under normal conditions in areas outside the SVZ and SGZ. There is evidence of low-level adult neurogenesis in other regions, such as the neocortex, amygdala, and hypothalamus, but results are not always replicable, possibly because of the relatively small number of new neurons produced compared with the SVZ and SGZ. 44 There is limited postnatal neurogenesis in the rodent cerebellum, although multipotential cerebellar NPCs have been isolated from adult mice. 64
The SVZ and Rostral Migratory Stream
The astrocytic type B cells of the forebrain SVZ give rise to neuroblasts via a rapidly dividing population of neuronal progenitors (type C cells; Fig. 1). 27 Remarkably, neuroblasts then undergo chain migration over long distances from the lateral ventricular wall to the olfactory bulb, where they differentiate into granule and periglomerular cell interneurons throughout life. This chain migration, known as the rostral migratory stream (RMS), has been well characterized in rodents and nonhuman primates; 21,27,61,68,86 new evidence suggests that this migration is guided by the flow of cerebrospinal fluid produced by the beating of ependymal cilia. 100 Until recently, the RMS was thought to be absent in humans 99 likely due to the fact that its orientation is slightly different from that of rodents. 21 There is now evidence of the RMS and olfactory bulb neurogenesis in humans. 7,21
SGZ of the Dentate Gyrus
The hippocampal SGZ (Fig. 3 ), located between the granule cell layer and hilus, contains a population of quiescent astrocytic stem cells (GFAP+ type B cells), which give rise to a proliferating population of GFAP– type D cells (analogous to the SVZ type C cells) that differentiate into new granule neurons. 103,104 Type D cells differ from the type C cells of the SVZ because they divide less frequently and express early markers of neuronal differentiation. 25 The astrocytic NSCs in the SGZ have basal processes running under the dentate gyrus along blood vessels and apical processes extending into the granule layer. Unlike the SVZ, the new neurons migrate only a very short distance to the adjacent granular layer. Another difference between the 2 neurogenic regions is the presence of endothelial cell precursors intermingled with NPCs in the SGZ, the first evidence of a relationship between angiogenesis and neurogenesis in the adult brain. 85
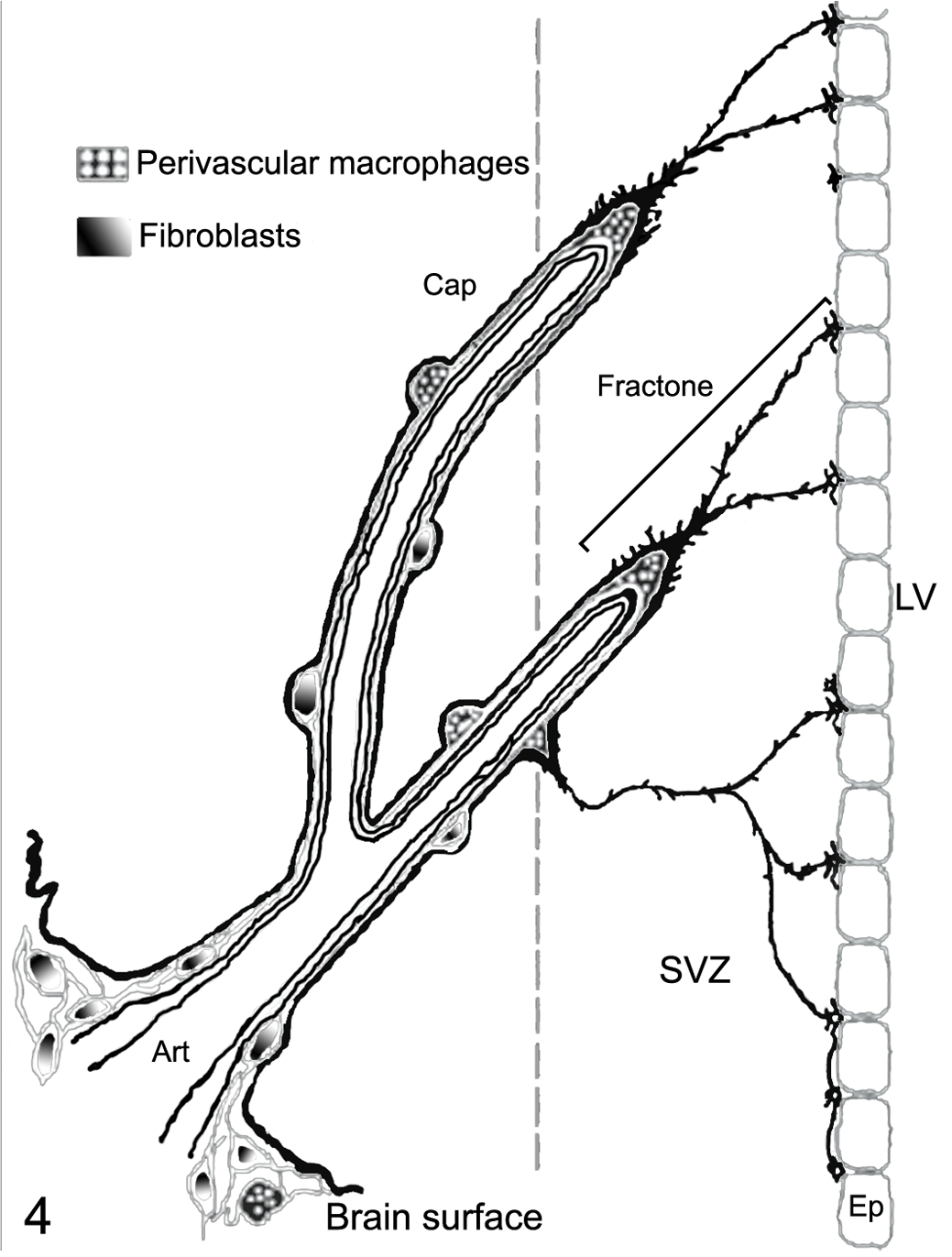
Schematic representation of fractones. Vascular and perivascular basal laminae and fractones are represented in dark black. The schema is based on the confocal distribution of laminin immunoreactivity, which defines the vascular/perivascular basal laminae and fractones. The depicted perivascular macrophages are part of a fibroblast/macrophage network originating from the meninges. Art, artery; Cap, capillary; Ep, ependyma; SVZ, subventricular zone; LV, lateral ventricle. Modified with permission from Mercier et al. 69
Cerebellar White Matter Tracts and the EGL
The cerebellum is the third location of postnatal neurogenesis. However, depending on species and location, neurogenesis appears to be limited to weeks to a year postnatally. Neuronal progenitors are present in the postnatal rodent cerebellar EGL up to 25 days and in the canine EGL until around 70 days; in humans, the EGL remains proliferative up to a year. 34,88 Neuronal progenitors of the EGL are restricted to generating granule cell neurons. 129 Cortical interneurons, as well as astrocytes and oligodendrocytes, are generated from multipotential NPCs that migrate from the VZ into white matter tracts after birth. The neurogenic potential of these NPCs appears limited to the first 2 postnatal weeks in rats. 128 It is of interest to note that cerebellar Bergmann glia, the only cells that retain radial glia morphology in the adult brain, originate from NPCs migrating from the VZ through the white matter within the first postnatal week. Moreover, adult Bergmann glia express the NSC markers Sox1, Sox2, and Sox9, as well as CD15 and GFAP. 1,5,6,37 Although overt neurogenesis has not been observed in the adult cerebellum, NSC potential is demonstrated in a subpopulation of cells isolated from the adult cerebellum in general 59 and Sox1-positive Bergmann glial cells in particular. 1
The NSC Niche
The concept of a stem cell niche arose from the observation that stem cells were evident in limited areas within adult tissues. The importance of the microenvironment’s role in cell fate is illustrated by in vitro experiments in which glial progenitors and NPCs from nonneurogenic areas demonstrate stem cell potential when exposed to the appropriate mixture of growth factors. 29,60,83 These studies suggest that neurogenesis is restricted to specific areas of the brain as a result of differentials in regulatory signals rather than an absence of NPCs in nonneurogenic regions. The SVZ and SGZ stem cell niches are microenvironments uniquely suited to the maintenance and regulation of stem cells. Fundamental niche properties identified to date include a specialized extracellular matrix, basal laminae, and NSC localized near blood vessels.
Vascular Niche
The unique relationship of NSCs to the vasculature was first identified in the SGZ of the hippocampal dentate gyrus. 85 Immunophenotyping of the proliferating cells in the SGZ revealed the presence of dividing cells in aggregates closely associated with blood vessels. Unexpectedly, more than a third of these proliferating cells were immunopositive for endothelial markers, and the surrounding area stained positively for vascular endothelial growth factor. The intimate association of NPCs and endothelial precursors around microvessels suggested a relationship between angiogenesis and neurogenesis. Further evidence of the importance of the vasculature in the NSC niche is provided by in vitro studies. In endothelial/NSC cocultures, diffusible factors released from endothelial cells inhibited NSC differentiation, stimulated self-renewal, and enhanced neurogenesis. Stem cell renewal appears to be promoted via Notch and Hes1 activation subsequent to enhanced neuroepithelial cell contact induced by soluble endothelial factors. Thus, while FGF2 is necessary for NSC proliferation, maintenance of self-renewal requires endothelial factors acting in concert with FGF2. 106
In the SVZ, the majority of proliferating type C cells and astrocytic stem cells are positioned within 10 μm of blood vessels, whereas most neuroblasts are found more distant to the microvasculature. 113 SVZ astrocytic NSCs (type B cells) are present in a superficial layer just under and sometimes intercalating with the ependyma; these astrocytes have apical membranes snaking between ependymal cells and a basal process terminating on blood vessels. 72,107 Type B cells possessing long processes running parallel to the ventricle are also found subjacent to the apical type B cell layer. 107 The blood-brain barrier, typified by tight junctions between endothelial cells and astrocyte endfeet, appears to be modified in the SVZ. Type B and C cells have direct contact with blood vessels in areas that do not have pericyte coverage or astrocyte endfeet; accordingly, small molecules circulating in the blood have access to the SVZ. This unique relationship of NPCs to SVZ blood vessels is not detected in other nonneurogenic regions. 113 The connection of type B cells to both the ventricle and the basal lamina of blood vessels provides 2 direct routes by which signal molecules can reach NSCs. However, unlike the SGZ, no evidence of proliferating endothelial precursors is detected in the adult SVZ. 107,113
Extracellular Matrix and Basal Laminae
Stem cells outside the brain are closely associated with basal laminae, which compartmentalize and store factors such as FGF2, bone morphogenetic proteins, vascular endothelial growth factor, and EGF. 40,69 Basal laminae are defined as aggregates of extracellular matrix rich in heparan sulfate glycosaminoglycans, fibronectin, laminin, and collagen IV. 69 In the SVZ, basal laminae are highly organized extravascular structures with a fractal arrangement, dubbed fractones. 69 Fractones have apical stems ending in bulbs immediately beneath the ependyma and extensively folded basal contacts with blood vessels. The fractal basal laminae enfold astrocytes, ependymal cells, NPCs, microglia, and the perivascular fibroblast/macrophage network (Fig. 4 ). Fractal basal laminae in the SVZ of mice, rats, and humans comprise laminin (β1 and γ1), collagen IV, the heparan sulfate proteoglycan (HSPG) perlecan, and glycoprotein nidogen. 55 An N-sulfated HSPG, seemingly distinct from perlecan, is present in a subpopulation of fractones and smaller subpopulation of blood vessels. Mitotic NPCs are highly associated with N-sulfated HSPG fractones compared with non-N-sulfated HSPG fractones, and mitotically active NPCs are located significantly closer to N-sulfated HSPG-containing fractones than blood vessels. 55 Of most interest is the finding that FGF2 binds mainly to N-sulfated HSPG fractones (and blood vessels) in situ and in vivo and that treatment with a heparitinase–heparanase mixture abolishes FGF2 binding in the SVZ. 55
Collagen I, usually associated with basal laminae and growth factors during developmental morphogenesis, is detected in the SVZ in a broad, diffuse band, as well as within some fractone stems and bulbs. 69 This parenchymal band of collagen I is not detected in the parenchyma of surrounding brain structures. Other extracellular matrix molecules that are strongly expressed in the adult SVZ but are rare to absent in surrounding regions include glial-derived tenascin C and chondroitin sulfate glycosaminoglycans, especially dermatan sulfate–dependent (DSD-1) proteoglycan. 40
Tenascin C, is produced by type B NSCs and forms a layer between the SVZ and adjacent striatum. 54 The glycoprotein functions as a developmental regulator, enhancing and diminishing sensitivity to FGF2 and bone morphogenetic protein 4, respectively, which accelerates the developmental expression of EGF receptors in NPCs. 39 In contrast, chondroitin sulfate glycosaminoglycans are necessary for the proliferation, self-renewal, and maintenance of FGF2-responsive, but not EGF-responsive, NPCs.109,110 In particular, the CS proteoglycan DSD-1, expressed on radial glia and retained on adult SVZ NPCs, is shown to have these properties. Targeted interference with the DSD-1 proteoglycan epitope recognized by the antibody 473HD results in significant diminution of neurosphere forming ability. 118
Laminin is abundant around vascular cells in the SVZ and comprises part of the fractal basal laminae. The importance of laminin in the neurogenic niche is demonstrated by the expression of the laminin receptor α6ß1 on NPCs. 14,107 The integrin α6 is thus far the only gene identified in common between embryonic, hematopoietic, and NSCs. 33 In the adult mouse, α6-integrin is expressed by NPCs in acutely dissociated SVZ cultures and within neurospheres. 107 When the α6-integrin is blocked in vivo by intraventricular anti-α6-antibody infusion, there is roughly a 50% increase in NPCs located > 10 μm from blood vessels and nearly a 35% increase in proliferative NPCs. 107 These findings suggest that α6-integrin expression is integral in maintaining NPCs within the vascular niche and regulating NPC proliferation and differentiation.
Immunomodulation of Neurogenesis
The NSC niche, like the rest of the brain, is not immunologically privileged as once thought but is an immunologically specialized region in which microglia and lymphocytes appear to interact with NPCs in both health and disease. 11,73,120,126,133 Recent studies have shown that in disease, inflammation may either enhance or suppress neurogenesis depending on how microglia, newly recruited blood monocytes, and/or astrocytes are activated. Activation of microglia with helper T-cell cytokines interferon γ and interleukin-4 (IL-4) is shown to promote neurogenesis and oligodendrogenesis, whereas tumor necrosis factor α, expressed by endotoxin-activated microglia, inhibits neurogenesis. 11,73 Subsequent investigations into the nondiseased adult neurogenic niche in rats revealed that increased hippocampal neurogenesis induced by environmental enrichment was associated with microglial activation and expression of MHC-II proteins, as well as insulin-like growth factor. 133 Microglial MHC-II expression suggests activation by Th2 cytokines (eg, IL-4); MHC-II is not expressed after innate immune activation of microglia (eg, endotoxin). 127 In contrast, exercise-induced increases in hippocampal neurogenesis in mice showed no evidence of microglial activation, microglial MHC II expression, or increase in T cells in the hippocampus. 80 The discrepancies between these 2 studies may be attributable to the differential effects of environmental enrichment versus physical activity on neurogenesis and/or to the species, rat versus mouse.
The importance of T cells in adult physiologic neurogenesis is supported by studies showing that neurogenesis is impaired in the hippocampus and SVZ of genetically immune-deficient mice and can be restored by systemic repopulation with T cells, specifically CD4+ T cells. 126,133 Moreover, immune-depleted and genetically immune-deficient mice show impaired spatial learning and memory compared with wild-type controls. 58,126,133 Interestingly, peripheral T-cell activation results in a transient increase in hippocampal neurogenesis and NPC proliferation. 127 In health, T cells are rare to absent in the CNS parenchyma but present in the meningeal spaces and choroid plexus/ventricular walls. Under physiologic conditions, the neurogenic effects of the adaptive immune response may originate largely from T cells within the meningeal spaces rather than the hippocampus; these effects appear to be mediated via IL-4. 23
While data indicate that adult neurogenesis is dependent on systemic CD4+ T-cell activity, controversy remains with respect to T-cell antigen specificity. Ziv et al showed that neurogenesis was enhanced in transgenic mice with an excess of monospecific T cells directed against a CNS-specific antigen (myelin basic protein) and reduced in transgenic mice with an excess of T cells directed against nonself antigen (ovalbumin) relative to matched wild-type controls. 133 Wolf et al found that repopulation of immune-deficient RAG2–/– mice with a heterogeneous population of CD4+ T cells was sufficient to increase neurogenesis. However, repopulation with CNS-specific T cells was not evaluated. 126
NPC Phenotype
Phenotypic characterization of NPCs aids in the identification of true stem cells, allows their prospective isolation and enrichment for therapeutic purposes, and helps in elucidating the mechanisms by which the nervous system develops and regenerates. Among the first NSC markers identified were nestin and vimentin, which are intracellular intermediate filament proteins. 47 ,65 Vimentin and nestin are not ideal NPC markers, because their expression is not restricted to NPCs. Vimentin is present in many other cell types in the CNS besides NPCs, and nestin is expressed in fibroblastic cell lines, reactive astrocytes, and skeletal muscle. 98 ,102,112 Moreover, neural progenitor/stem cells have been identified that do not express nestin. 62
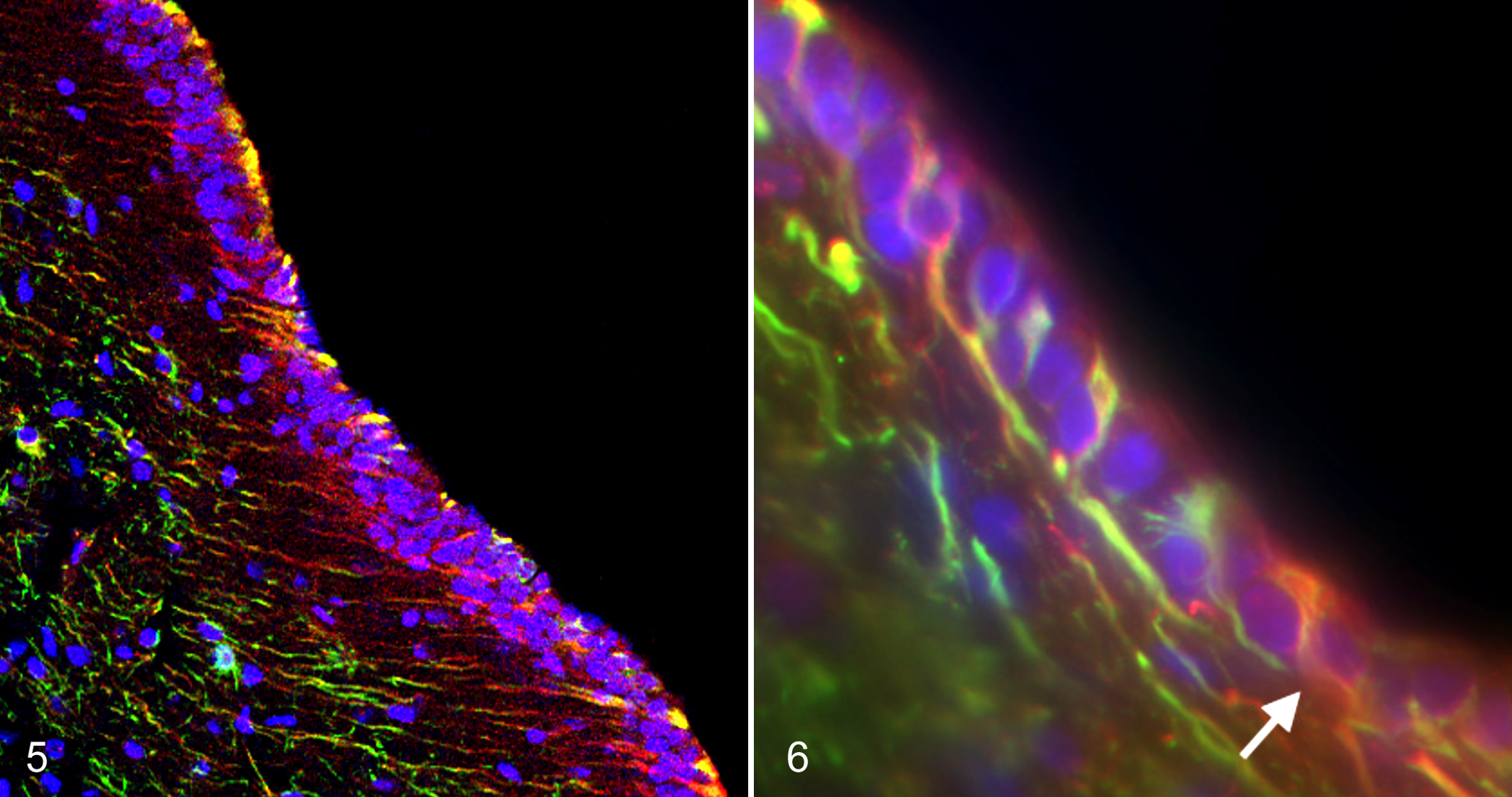
Work in the past decade has shown that NSCs in the developing fetus and in neurogenic regions of the postnatal brain express GFAP. 26 ,50,70,74,76,104 The quiescent astrocytic type B cell identified as the NSC in the adult mouse SVZ is both GFAP+ and nestin+. 27 Like nestin and vimentin, GFAP is an intracellular filament protein and is expressed in nonprecursor cells as well as NSCs. Although none of these proteins alone is a definitive NSC marker, labeling for combinations of markers in conjunction with cellular morphology and location may serve to accurately identify NSCs (Figs. 5, 6).
While the use of intracellular NPC markers is a valuable tool for in situ studies of neural development and regeneration, prospective isolation and enrichment of NPCs for in vitro and transplantation studies require a cell surface marker. Candidate cell surface markers used to enrich for NPCs include the hematopoietic stem cell marker CD133 and differential binding of peanut agglutinin and heat-stable antigen. 64,96,116 Human and murine hematopoietic stem cells can be fractionated using the cell surface antigen CD133 or the differential binding of peanut agglutinin and heat-stable antigen. When applied to single cell suspensions from the ependyma/subependyma of adult mice, approximately 80% of the cells that had poor peanut agglutinin binding and expressed very low levels of heat-stable antigen were capable of forming neurospheres; this represents nearly a 250-fold enrichment of NPC activity compared to unsorted cells. 96 The cell surface marker CD133 selected for a nearly 24-fold increase of NPC activity from human fetal brain tissue and prospectively identified a multipotent NPC in the postnatal mouse cerebellum. 64 ,116 Interestingly, CD133 expression in the murine postnatal SVZ is confined to a subpopulation of ependymal cells capable of forming neurospheres with limited self-renewal ability in vitro. 19,87
Three other cell surface markers associated with NPCs in developing and adult brains are Lewis X antigen (CD15), an epitope in phosphacan (DSD-1 proteoglycan) recognized by the monoclonal antibody 473HD, and the integrin β1—specifically, α6β1-integrin. 14,15,107,118 Enriching for CD15+ cells from the adult mouse subependyma resulted in a 25-fold increase in NPC activity, 15 whereas 473HD+ cells showed a 6-fold increase in neurosphere formation compared with unsorted cells. 118 Integrin β1-expressing cells from postnatal mice and rats generated 3 to 12 times as many neurospheres as β1-negative cells. 14
A method of hematopoietic stem cell purification, side population analysis, has been successfully used to enrich for NPCs. Hematopoietic stem cells exclude the nuclear dye Hoechst 33342, resulting in the presence of a side population of cells with low fluorescence emission. 43 Differential dye efflux was shown to be a property of NPCs, as it is with hematopoietic stem cells. 48 ,57 In the side population derived from adult neurospheres, there was a 7.5-fold enrichment in NPC activity. 57 Moreover, combining side population analysis with CD15 labeling further enriched NSC activity beyond that achieved by side population analysis alone. This enrichment was much more pronounced with embryonic NPCs (38% enrichment) than with adult NPCs (10% enrichment). 57
Conclusion
The current body of knowledge concerning developmental and adult human neurogenesis is largely attributable to research animals, including avian species. Postnatal neurogenesis was discovered in rats in the 1960s but was not widely accepted until 2 decades later when research studies in adult songbirds elegantly demonstrated seasonal neurogenesis in specialized regions. As more animal species are investigated, fundamental mechanisms of neurogenesis and NSC characteristics appear to be well conserved across diverse species. Interspecies conservation of neurogenesis (along with the existence of large and small animal models of neurogenic diseases) affords unique investigative and therapeutic research opportunities relevant to both humans and animals.
Footnotes
Acknowledgements
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author received no financial support for the research, authorship, and/or publication of this article.
