Abstract
Urothelial carcinomas (UCs), also known as transitional cell carcinomas, are the most common canine urinary tract neoplasms. Tyrosine kinases (TKs) are enzymes that tightly regulate cell growth and differentiation through phosphorylation. Receptor TK (RTK) inhibitors are currently used to treat UCs. Toceranib phosphate (Palladia; Pfizer) is an RTK inhibitor that blocks the activity of vascular endothelial growth factor receptor 2 (VEGFR2), platelet-derived growth factor receptor–alpha and –beta (PDGFR-α, -β), FMS-like tyrosine kinase 3, stem cell factor receptor (KIT, kinase inhibitor targeting), and colony stimulating factor receptor. To better understand UCs and validate treatment targets, we performed immunohistochemical staining for RTKs, as well as a novel target, cyclin-dependent kinase 4 (CDK4, a central regulator of the mammalian cell cycle), on formalin-fixed, paraffin-embedded tissues from bladder biopsies from 17 dogs with UCs, 17 dogs with cystitis (diseased controls), and 8 normal dogs (negative controls). Although immunohistochemical scores could not be extrapolated to prognostic value, response to treatment, and outcome of patients with UC, we demonstrated expression of PDGFR-β and VEGFR2 in UCs; all UC samples staining positively for VEGFR2. Minimal positive staining for KIT was noted in the tumor samples. CDK4 staining intensity was significantly weaker in UCs compared with normal and cystitis bladder samples. The intense staining of VEGFR2 in UC cells suggested that VEGFR2 may be of prognostic and/or therapeutic value in dogs with UC. Overexpression of VEGFR2 in UC cells validates this receptor as a treatment target in UC.
Urinary bladder cancer is common in dogs, accounting for ~2% of all reported malignancies. 19 Urothelial carcinoma (UC) is the most common form, and the disease is invariably fatal. 19 Canine UCs show striking similarities to human UC in terms of histopathology, biologic behavior, response to medical therapy, and prognostic factors. 14 In contrast to humans, in which ~75% of patients have noninvasive disease, most canine UCs are of the muscle-invasive phenotype. 13 Given the similarities of the clinical and pathology profiles, canine UC is believed to be a model for human invasive UC. 14
Treatment options available for UC are limited. 28 Owners who are reluctant to pursue aggressive chemotherapeutic protocols are left with NSAIDs, which are contraindicated in animals with renal failure, or receptor tyrosine kinase (RTK) inhibitors. Overexpression of RTKs has been noted to occur in cases of human and canine UC.21,31 Pharmacologic inhibition of RTKs have been explored in human 20 and canine 24 bladder cancer patients, although its success rate has been limited. A clinical trial has demonstrated that 3 of 4 dogs with UC treated with RTK inhibitors alone had stable disease for ≥10 wk. 17 Toceranib phosphate (Palladia; Pfizer), an RTK inhibitor that blocks the activity of vascular endothelial growth factor receptor 2 (VEGFR2), platelet-derived growth factor receptor–alpha and –beta (PDGFR-α and -β), FMS-like tyrosine kinase 3, stem cell factor receptor (KIT, kinase inhibitor targeting), and colony stimulating factor receptor, 17 has demonstrated activity against a variety of tumor types in dogs, including several carcinomas. 17
Overall, RTK inhibitors require further investigation as a treatment option for patients with UC. Preclinical studies continue to identify novel pathways, and thus therapeutic targets, that may be involved in the formation of UCs. Specifically, cyclin-dependent kinase 4 (CDK4) is a central regulator of the mammalian cell cycle. 12 Mechanistically, overexpression of CDK4 is commonly caused by deletion of p16INK4A, which occurs via gene mutation, deletion, 27 and/or promoter methylation. 11 Overexpression of CDK4 inhibits RB1 function, increasing cell signaling, and promoting cell cycle progression. Knowledge of the expression of CDK4 in healthy dog tissue is not available, and only one study has described expression of CDK4 in canine liposarcomas. 2 In humans, overexpression of CDK4 has been shown in many tumor types, including oral squamous cell carcinomas, 23 pancreatic endocrine tumors, 18 pulmonary neoplasia, 6 melanomas, 30 mammary carcinomas, 1 sarcomas, 11 and nasopharyngeal carcinomas. 16
Our goal was to assess RTK expression in canine UC, important because it will assist in more targeted therapy of canine UC and address its limited treatment options.
Materials and methods
Sample collection
We selected formalin-fixed, paraffin-embedded samples of UC, cystitis, and normal canine bladders from the archive at the Veterinary Pathology Diagnostic Services at the University of Sydney (New South Wales, Australia). An anatomic pathologist confirmed the diagnoses of UC, cystitis, and normal before immunohistochemical analysis was performed. Carcinoma cases had also been confirmed by histopathology for tumor origin in the kidney, bladder, or urethra. Samples without propria-submucosa were excluded. Information collected from the medical records included age, sex, breed, date of treatment initiation, treatment protocol, outcome, and, when available, cause of death. Cause of death was classified as: related to primary UC (such as acute renal failure, urinary obstruction, uremia), related to metastatic UC, or associated with other diseases unrelated to UC. All cases were assessed histologically and graded according to the World Health Organization (WHO) tumor classification system 2016 10 ; tumors are differentiated into 3 grades: G1, G2, and G3. The lowest grade (G1) displays slim papilla with no atypia; the highest grade (G3) displays major atypia with marked loss of normal architecture. A classification of G2 covers the wide spectrum of lesions seen between G1 and G3 and includes increasing layers within the papilla and rare atypia.
Immunohistochemistry
All procedures were carried out at room temperature (RT) in the Veterinary Pathology Diagnostic Services laboratory at the University of Sydney. Slides were rinsed between steps and first, 4-µm thick sections were dewaxed and rehydrated (Table 1). After antigen retrieval, slides were incubated with the appropriate antibody: CDK4 was applied overnight at 4°C; VEGFR2, KIT, and PDGFR-β were applied for 60 min at RT. All antibodies used in our study were validated by the manufacturers and/or were used in studies by other researchers.4,8,9,25,26,29,33 Sections were incubated for 30 min with the appropriate secondary antibody using a horseradish peroxidase–labeled polymer system (EnVision). Each run included a set of positive and negative controls. Following chromogen application, slides were counterstained for 5 s in hematoxylin. Slides were then dehydrated through ethanol, cleared with xylene, and coverslipped.
Details of immunohistochemical stains performed on sections of urinary bladder in dogs.
Immunohistochemical evaluation
Staining was compared among UC, inflammatory non-neoplastic (cystitis), and normal urinary bladder samples. A histologically confirmed canine metastatic hemangiosarcoma served as a positive control for VEGFR2; a canine liposarcoma served as a positive control for KIT; a canine squamous cell carcinoma served as a positive control for PDGFR-β; and a canine mesenchymal neoplasm was used as a positive control for CDK4 (Suppl. Figs. 1–4). A negative control that omitted incubation with the primary antibody (antibody diluent with no antibody) was included for each sample.
Samples were evaluated for urothelial expression of VEGFR2, KIT, PDGFR-β, and CDK4 in an anonymized study by 3 veterinary pathologists, each from a different institution. A qualitative immunohistochemical assessment was performed to evaluate staining intensity. As described previously, 32 staining intensity was assessed over the whole sample at 200 × magnification (0 = none, 1 = mild, 2 = moderate, 3 = intense). Staining distribution (% urothelial cells affected) was evaluated semi-quantitatively over 10 hpfs at 400 × magnification (0 = no staining, 1 = >0% to <10% positive, 2 = ≥10% to <25% positive, 3 = ≥25% to 50% positive, 4 = ≥50% to <75% positive, 5 = ≥75% positive). To calculate a score for every sample, the average scores were taken and called the standardized score. After averaging the 10 selected fields, a final immunohistochemical score for each sample was calculated by multiplying the intensity (qualitative) standardized score by the staining distribution (semi-quantitative) standardized score, as described previously.3,22
Statistical analysis
To determine the sample size for our study, we performed a power analysis for logistic regression using WebPower (https://webpower.psychstat.org/). The association of intensity score, staining distribution score, and final immunohistochemical score with sample groups (UC, cystitis, normal canine urinary bladder) for VEGFR2, KIT, PDGFR-β, and CDK4 was determined. Fisher exact tests were performed to evaluate for independence and for pairwise comparison (i.e., UC vs. cystitis, UC vs. normal, cystitis vs. normal). Risk factors for UC development were evaluated using conditional logistic regression analyses. Cox proportional hazard regression analysis was used to evaluate the correlation between the parameters under investigation and the clinical outcome. Dogs that were lost to follow-up were censored in the Cox proportional hazard regression analysis. Differences were considered significant at p ≤ 0.05. All statistical analyses were performed via computer software (R 4.0.2, https://www.r-project.org/). Logistic regression was used to identify risk factors for UC using a generalized linear model through the “glm” function in R. Binomial regression was used to evaluate the development of UC and cystitis relative to normal samples.
Results
Power analysis demonstrated little risk of type II error
A power analysis for simple logistic regression demonstrated a power of 0.55–0.61 for our study, indicating that there was a 55–61% probability that this analysis would not commit a type II error. The power was 0.61 at an alpha level of 0.05 comparing cystitis to normal canine urinary bladder samples; the power was 0.55 at an alpha level of 0.05 comparing UC with normal canine urinary bladder samples.
Analysis of study population demonstrated no significant risk factors
We selected samples from bladders of 17 dogs with UC, 17 dogs with cystitis, and 8 normal dogs. One case of UC was not assessed for expression of PDGFR-β because of limited tissue availability. Samples of cystitis and UC were derived from dogs 1–16-y-old and 1–15-y-old, respectively. The breed affected most commonly by UC was the Border Collie (n = 3; Table 2). Logistic regression analyses were used to evaluate the association of age, sex, and breed with risk for development of UC. No significant risk factor was identified using a significance level of 0.05. All risk factors had p values close to 1.
Patient population characteristics of canine cases with cystitis or urothelial carcinoma (UC).
CM = castrated male; F = intact female; M = intact male; SF = spayed female.
Most canine UCs were high-grade and infiltrative
The subgross tumor growth pattern could be assessed in 6 of 17 UC cases because these samples were transmural tissue sections. The tumor in all 6 cases invaded the basement membrane, consistent with a diagnosis of infiltrating UC per the WHO 2016 tumor classification (Table 2).
Normal and cystitis bladder samples expressed VEGFR2, PDGFR-β, and CDK4, but not KIT
Histopathologic findings in cystitis cases included lymphoplasmacytic-to-neutrophilic infiltrates within the lamina propria, accompanied by one or more of the following: urothelial erosion or ulceration, sub-urothelial proliferation (Brunn nests), granulation tissue formation within the lamina propria, submucosal inflammation (edema, fibrosis, blood vessel dilation, hemorrhage), and/or detrusor muscle fibrosis.
CDK4 was expressed in 15 of 17 cystitis samples, with weak (n = 3) to moderate (n = 11) to intense (n = 1) cytoplasmic and nuclear staining. The percentage of labeled urothelium ranged from ≥10% to <25% (n = 1) to ≥75% (n = 11). Of the normal urinary bladder samples, CDK4 was expressed in 7 of 8 cases with mild (n = 7) to moderate (n = 1) cytoplasmic and nuclear staining (Fig. 1). Of these, 2 cases had ≥50% to <75% of their urothelium stain for CDK4, and the others had ≥75%.
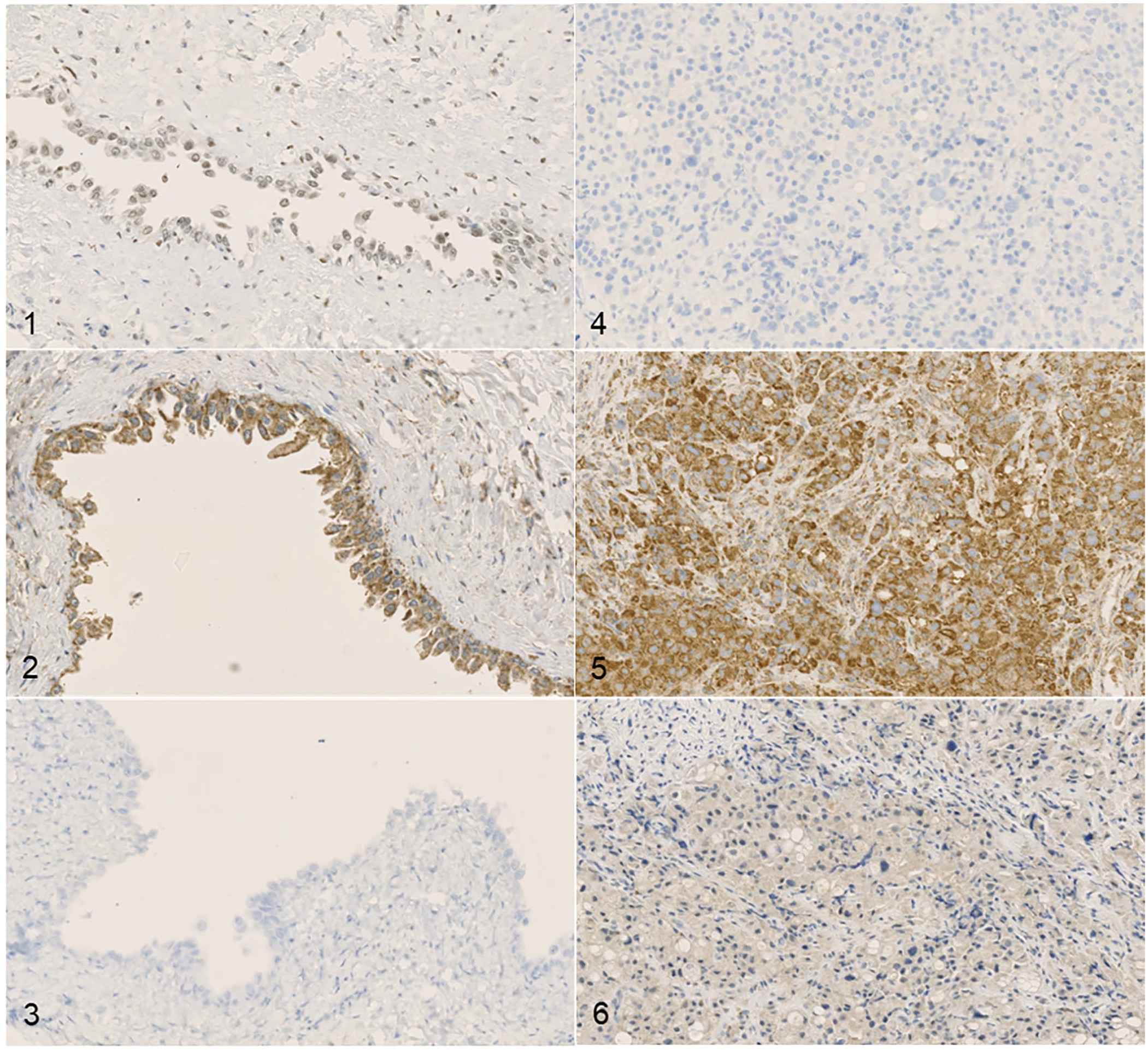
Immunohistochemical expression of VEGFR2, KIT, PDGFR-β, and CDK4 in canine urothelial carcinoma (UC).
VEGFR2 was expressed in 13 of 17 cystitis samples, with mild (n = 1) to moderate (n = 7) to intense (n = 5; Fig. 2) cytoplasmic staining. The percentage of labeled urothelium ranged from no staining (n = 4) to ≥75% (n = 11). Of the 8 normal urinary bladder samples, VEGFR2 was expressed in all cases with moderate (n = 3) to intense (n = 5) cytoplasmic staining; all 8 cases had ≥75% of their urothelium stain for VEGFR2.
Of the normal urinary bladder samples, KIT was expressed with moderate cytoplasmic staining in 1 of 8 cases; this case had ≥50% to <75% of its urothelium stain for KIT. KIT was expressed in 3 of 17 cystitis samples, with mild (n = 1) to moderate (n = 2) cytoplasmic staining. The percentage of labeled urothelium ranged from no staining (n = 14; Fig. 3) to ≥75% of its urothelium staining for KIT (n = 1).
Of the normal urinary bladder samples, PDGFR-β was expressed intensely in all 8 cases, with ≥75% of the urothelium staining for PDGFR-β. PDGFR-β was expressed in 17 of 17 cystitis samples, with weak (n = 6) to moderate (n = 10) to intense (n = 1) cytoplasmic staining. The percentage of labeled urothelium ranged from ≥25% to <50 % (n = 1) to ≥75% (n = 15).
UCs expressed VEGFR2, KIT, PDGFR-β, and CDK4
CDK4 was expressed in 14 of 17 UC samples, with weak (n = 12) to moderate (n = 2) to intense (n = 1) cytoplasmic staining. No CDK4 immunoreactivity was evident in 3 of 17 UC samples (Fig. 4). The percentage of labeled urothelium ranged from ≥25% to 50% (n = 1) to ≥75% (n = 12).
VEGFR2 was expressed in all cases of UC, with moderate (n = 5) and intense (n = 12; Fig. 5) cytoplasmic staining demonstrated. The percentage of labeled urothelium ranged from ≥50% to <75% (n = 1) to ≥75% (n = 16).
KIT was expressed in 3 of 20 UC samples, with weak (n = 2) to moderate (n = 1) cytoplasmic staining. Most samples (17 of 20) demonstrated no KIT immunoreactivity (Fig. 6). The percentage of labeled urothelium ranged from ≥25% to 50% (n = 1) to ≥75% (n = 2).
PDGFR-β was expressed in all 16 UCs with weak (n = 11) to moderate (n = 5) cytoplasmic staining. The percentage of labeled urothelium ranged from ≥25% to 50% (n = 2) to ≥75% (n = 11).
Intensity scores demonstrate PDGFR-β and CDK4 as possible biomarkers of UC
Significant negative association between the intensity of expression of PDGFR-β in UC, cystitis, and normal canine urinary bladder samples was demonstrated (p <0.02). Similarly, intensity of CDK4 expression was found to be differentially expressed significantly among all 3 groups (p <0.004), with CDK4 expressed weakly in more UC samples, compared to stronger expression in cystitis and normal canine urinary bladder samples. Intensity score and staining distribution of VEGFR2 had a weak association with UC, cystitis, and normal canine urinary bladder samples (p = 0.087 and p = 0.131, respectively). Virtually no statistically significant associations were apparent for KIT when comparing intensity score (p = 1) or staining distribution (p = 0.875) with UC, cystitis, and normal canine urinary bladder samples.
Chemotherapy agents and COX inhibitors were mainstay treatments for UC
At the time of analysis, 5 of 17 dogs were dead, and 12 of 17 dogs were lost to follow-up at a median of 23 d post-visit (range: 1–1,115 d). Seven of 17 were treated with cyclooxygenase (COX) inhibitors + chemotherapy (mitoxantrone), and 5 of 17 dogs were treated with COX inhibitors alone. Administered COX inhibitors included piroxicam in 9 of 17 dogs, meloxicam in 1 of 17 dogs, firocoxib in 1 of 17 dogs, and robenacoxib in 1 of 17 dogs. The median survival time (ST) for dogs in our study was 152 d (range: 1–1,427 d). Of the 5 dogs that died during our study period, the cause of death was available in all cases, and was related to primary UC (n = 2), related to metastatic UC (n = 1), or associated with other diseases unrelated to UC (osteoarthritis, n = 1; sedation-related, n = 1). Cox proportional hazard regression analysis was attempted, but coefficients did not converge, and the analysis failed to identify predictor variables associated with disease progression.
Discussion
We demonstrated expression of PDGFR-β and VEGFR2 in UCs, suggesting that the role of these RTKs in the progression of UC warrants further investigation. All UC and cystitis samples stained positively for PDGFR-β, and most exhibited mild-to-moderate diffuse cytoplasmic staining. Normal bladder samples had ≥75% PDGFR-β positivity, with mostly intense staining. A significant difference was found between PDGFR-β staining intensity among normal, cystitis, and UC groups. Importantly, there was marked variation in immunolabeling between UC samples, and clinical variance in this parameter is likely. This finding is further supported by a study that demonstrated marked variation in the relative mRNA transcript expression of PDGFR-β across 9 canine primary UC tumor samples. 15
VEGFR2 expression was demonstrated in neoplastic and non-neoplastic bladder samples. We found a weak association between VEGFR2 staining intensity among normal and UC groups; however, all UCs had intense cytoplasmic staining for VEGFR2 (FLK-1) in >50% of tumor cells. Although normal and UC sample groups both expressed VEGFR2, the intense staining of VEGFR2 in UC cells suggested that VEGFR2 may be of prognostic and/or therapeutic value in dogs with UC. Phosphorylation (activation) of VEGFR2 was detectable to various degrees in 3 of 9 canine primary UC tumor samples. An assessment of VEGFR2 activation in larger sample sizes would be of benefit to further explore its therapeutic efficacy. 15 When assessing prognostic biomarkers of UC, VEGF-A expression is known to be correlated with UC grade in humans. 15 VEGFR2, measured in our study, interacts mainly with VEGF-A. 7 High levels of VEGF-A are also correlated with worse survival and higher recurrence rates in humans with UC. 34
We found that only 25% of UC cases stained positively for KIT, with variable staining distribution. No significant difference was found between KIT staining intensity and staining distribution score among UC, cystitis, and normal groups. These findings suggest that KIT is unlikely to play a major role in tumor development and progression of UC.
Most UC, cystitis, and normal bladder samples stained for CDK4; however, staining intensity was generally weak in UCs. A significant difference was found between CDK4 staining intensity among UC, cystitis, and normal groups, with reduced expression of CDK4 in UC. CDK4 is likely to play a role in UC development and progression.
A 2017 study reported the immunohistochemical expression of VEGFR2, PDGFR-β, and KIT in urinary bladder UC cases in 30 dogs; tissue samples were stained and scored for staining intensity and staining distribution. 32 A discussion about staining intensity across the 2017 study and our study follows. Staining distribution could not be compared between the 2 studies because the different scaling methods used precluded comparison. PDGFR-β was the most promising biomarker in both studies, with a significant difference found in PDGFR-β staining intensity among UC, cystitis, and normal groups. Similar results across the 2 studies were found with regard to VEGFR2 and KIT intensity; most UC samples had intense cytoplasmic staining for VEGFR2 in ≥75% of tumor cells, and KIT demonstrated no significant association with UC, cystitis, and normal sample groups. Hence, KIT appeared to be a poor biomarker for UC or cystitis. These conclusions were consistent with findings of variable RTK expression in UCs. 15
Risk analysis failed to identify any significant risk factors; however, given our small sample size, this result may not be reliable. Unfortunately, given the retrospective nature of our study, treatment protocols were not standardized or known, and reason for euthanasia was not always noted in medical records. Therefore, immunohistochemical scores could not be extrapolated to prognostic value, response to treatment, and outcome of patients with UC. In humans, molecular gene expression signatures are helpful for determining prognosis; hence, it is thought that molecular grading may supplant traditional morphologic grading systems. 5 Molecular gene expression of canine UC, and correlation of molecular grading systems with traditional immunohistochemical analysis, may be worth exploring.5,19
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221146247 – Supplemental material for Immunohistochemical analysis of expression of VEGFR2, KIT, PDGFR-β, and CDK4 in canine urothelial carcinoma
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221146247 for Immunohistochemical analysis of expression of VEGFR2, KIT, PDGFR-β, and CDK4 in canine urothelial carcinoma by Laura C. Setyo, Shannon L. Donahoe, Patrick L. Shearer, Penghao Wang and Mark B. Krockenberger in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Karen Barnes, Elaine Chew, Andrew Fortis, and Huy Tran for making further investigations possible by providing technical help. Thank you to Wilson So (Veterinary Pathology Diagnostic Services, University of Sydney) for invaluable assistance with data collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
