Abstract
Actinobacillus suis–like organisms (ASLOs) have been isolated from the genital, respiratory, and digestive tracts of healthy adult horses, horses with respiratory disease, and septic foals. Two foals with congenital hypothyroidism–dysmaturity syndrome from separate farms developed ASLO infection. At necropsy, both had contracted carpal flexor tendons, thyroid hyperplasia, and thrombotic and necrotizing mesenteric lymphangitis and lymphadenitis; one foal also had mandibular prognathism. Numerous ASLOs were isolated from tissues from both foals, including intestine. Biochemical testing and mass spectrometric analysis of the two Actinobacillus isolates did not allow unequivocal identification. Comparative genetic analysis was done on these and similar isolates, including phylogeny based on 16S rRNA, rpoB and recN genes, as well as RTX (repeat in toxin) toxin typing of apxIA–apxIVA and aqxA genes. One isolate was identified as Actinobacillus suis sensu stricto, based on the presence of apxIA and apxIIA but not aqxA, whereas the other isolate had aqxA but neither apxIA nor apxIIA, consistent with A equuli ssp haemolyticus. Based on genotypic analysis of the isolates included for comparison, 3 of 3 equine ASLOs and 2 of 5 A equuli isolates were reclassified as A equuli subsp haemolyticus, emphasizing the importance of toxin genotyping in accurate classification of actinobacilli.
Mortality from sepsis is common in equine neonates, especially in those with poor colostral transfer of immunity. Escherichia coli and species of Actinobacillus, Klebsiella, Streptococcus, and Enterococcus are most commonly isolated from septic foals.15,26,30 Actinobacillus suis has been recovered from septic pigs and has been associated with pneumonia, pleuritis, pericarditis, and polyarthritis in this species. 33 In horses, A suis–like organisms (ASLOs) have been isolated from the genital and respiratory tracts and the oral cavity of apparently healthy adults, animals with respiratory disease, and septic neonates. 4 The taxonomic grouping of these equine isolates—which have been referred to as A suis, ASLO, A equuli variants, or Bisgaard taxon 11—and their relationship to A suis of porcine origin has been problematic.6,13,14,20 They are similar to, or even indistinguishable from, A suis sensu stricto by conventional microbiological methods. Based on results from DNA–DNA hybridization studies, 14 the organism has been reclassified as A equuli subsp haemolyticus; its hemolytic activity is specified by the RTX (repeat in toxin) Aqx protein. 5 RTX toxin genotyping can be utilized to distinguish A equuli subsp haemolyticus from A suis sensu stricto.20,23 Hemolysis and modified CAMP (Christie-Atkins-Munch-Petersen) test positivity differentiate A equuli subsp haemolyticus from A equuli subsp equuli: the cause of sepsis, nephritis, and arthritis in neonatal foals. 14 A equuli subsp haemolyticus seems to be of significance in only the horse, whereas A equuli subsp equuli has been isolated from diseased foals as well as pigs.13,14,23,32 To date, no equine ASLO has been confirmed as A suis sensu stricto. 20
Congenital hypothyroidism–dysmaturity syndrome (CHDS) is a recognized cause of equine abortion and neonatal death in the northwestern United States and western Canada.1,3,28 CHDS is characterized by marked thyroid hyperplasia, mandibular prognathism, and delayed or insufficient ossification of carpal and tarsal bones with bilateral flexural contraction of the forelimbs. Due to these lesions, the term thyroid hyperplasia and musculoskeletal deformities has also been applied. 1 The specific cause (or causes) of CHDS remains unknown, but dietary factors seem important in its pathogenesis, such as excessive nitrate or insufficient iodine during the first trimester of gestation. 19 Similar lesions have been reported in foals from mares that consumed locoweed (Astragalus mollisimus) or fescue infected with Acremonium coenophialum or Claviceps purpurea. 8 Partially thyroidectomized foals have anomalies that are comparable to those of CHDS, which has led to the conclusion that hypothyroidism is the primary event in the pathogenesis of the skeletal anomalies. 2
Localized lymphangitis is a feature of several diseases. Thrombotic lymphangitis develops in the localized form of anthrax in some species. 27 In horses, subcutaneous ulcerative lymphangitis can be seen after infection with Corynebacterium pseudotuberculosis, Rhodococcus equi, Pseudomonas aeruginosa, or Histoplasma farciminosum. 27 Enteritis can be associated with granulomatous lymphangitis in infections with R equi in the horse or Johne’s disease in ruminants. 27
We report pathologic and microbiologic findings in 2 cases of necrotizing mesenteric lymphangitis and lymphadenitis in foals with CHDS and ASLO sepsis. Results from the microbiologic, biochemical, protein, and molecular biological analyses of the ASLOs isolated from the 2 foals were compared with those of Actinobacillus spp isolates from foals and adult horses and with porcine A suis isolates.
Materials and Methods
Cases
Two unrelated foals developed weakness, apathy, and fever and were submitted for postmortem examination to the Washington Animal Disease Diagnostic Laboratory (WADDL), Pullman, Washington. In both cases, a clinical diagnosis of CHDS was based on the presence of contracted tendons (both foals), mandibular prognathism (foal No. 1), and a history of foals (5 of 24) born with contracted tendons in the herd of foal No. 2. Foal No. 1, a male 2-day-old 52-kg Quarter Horse, was in fair body and good postmortem condition. Foal No. 2, a female 3-day-old Quarter Horse, was in fair body and moderate postmortem condition.
Bacterial Isolates and Growth Conditions
Two ASLO isolates, WADDL1 and WADDL2, were obtained from foals No. 1 and No. 2, respectively. A third ASLO isolate, WADDL3, came from a 1.5-day-old 50-kg male Quarter Horse foal with sepsis. These 3 American isolates were compared to an A suis isolate from a septic foal (Fohlen 5/96) or the joint of a foal (89103/1/2000) in Germany, 5 A equuli strains (A57203, A equuli H, 8274GI, A6202, 912GI), and 1 ASLO isolated from the pleura of an adult horse (MCCM 02245). Two porcine A suis strains (52285/85; MCCM 00717) as well as an Actinobacillus pleuropneumoniae and Actinobacillus lignieresii isolate were included for comparison. The designations of the different isolates, their sources, and growth characteristics are presented in Supplementary Table 1 (available at http://vet.sagepub.com/supplemental). All isolates were cultivated on Columbia blood base agar (Oxoid Ltd, Cambridge, UK), with 5% bovine blood supplement and without, at 37°C for 18 to 24 hours under aerobic conditions. The CAMP test for cohemolytic activity according to Christie et al (1944) was performed on 5% blood agar plates using a β-hemolytic Staphylococcus aureus as described. 17
Biochemistry and Antimicrobial Sensitivity
The biochemical characteristics of the different isolates and strains were determined using the Biolog system (Oxoid Ltd) and API 50CHE or API ZYM (BioMérieux, Nürtingen, Germany). Adherence to agar media was tested by attempts to remove colonies with a bacteriologic loop from Columbia agar plates (Oxoid Ltd) without bovine blood after incubation at 37°C in air for 18 to 24 hours. To analyze chromogenicity, the isolates and strains were cultivated at 37°C under aerobic conditions on Chrom-Agar-Orientierung (Mast-Diagnostica GmbH, Reinfeld, Germany) for 24 to 48 hours. Antimicrobial sensitivity was performed as a disc test on Columbia agar without blood. The following antimicrobials were tested: ampicillin, chloramphenicol, gentamycin, enrofloxacin, tetracycline, neomycin, and nitrofurantoin.
Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis
Late-log-phase bacteria from broth were harvested by centrifugation at 10 000 × g and washed 3 times in phosphate buffered saline. Pelleted cells was resuspended in sample buffer and heated for 10 minutes at 95°C according to the manufacturer’s recommendations (BugBuster Protein Extraction Reagents, EMD Chemicals Inc, Merck KGaA, Darmstadt, Germany). Bacterial proteins were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) in 10% gels. Proteins were visualized with Coomassie blue stain.
Typing by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry
Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF) typing was performed on an Ultraflex I instrument (Bruker, Bremen, Germany) in linear positive mode in the mass range from 2000 to 15 000 Da. Acceleration voltage was 25 kV; the instrument was calibrated in the range from 4364 to 10 299 Da with reference masses of an extract of an E coli DH5-α strain. Samples were prepared from individual colonies as described. 34 In brief, the colony was transferred to a microcentrifuge tube, diluted in water, and precipitated with ethanol. The sediment was then extracted with 70% formic acid; an aliquot was spotted on a steel target; and the dried spot was overlaid with 1.5 μl of a saturated solution of α-cyano-4-hydroxycinnamic acid in 50% acetonitrile / 2.5% trifluoroacetic acid as matrix. Phylogenetic analysis was performed with Biotyper software as suggested by the manufacturer (Bruker). Six colonies were spotted per examined species or subspecies. Four spectra of 500 shots were registered from every spot. The resulting 24 spectra were processed to yield a single reference spectrum with 100 masses for every species or subspecies. For the construction of phylogenetic trees based on the reference spectra, “correlation” and “average” were selected in the Biotyper software as the distance measure and the linkage algorithm.
High-Performance Liquid Chromatography Analysis of Nucleotides
Guanine–cytosine content of DNA preparations was assayed according to published procedures 38 with the following modifications. An EC 250/4 Nucleosil 120-5 C18 column (Macherey & Nagel, Dueren, Germany) was used at a flow rate of 1.2 ml per minute. A 20-μl aliquot of standard or sample was applied; elution of the analyte was monitored at 260 nM. With standards containing 1.25 mM of dA, dT, dC, and dG each, baseline separations were achieved for all 4 nucleotides, including dT and dG. Data were evaluated with WinChrom 1.21 (GBC Scientific Equipment, Braeside, Australia). Guanine–cytosine content was calculated from the peak heights rather than from the area under the curve, because variations were lower than expected.
Phylogenetic Analyses
For template DNA preparation, a few bacterial colonies from pure cultures on tryptic soy broth or Choc-agar plates (Oxoid Ltd) were resuspended in 450 μl of lysis buffer (0.1 M Tris HCl, pH 8.5, 0.05% Tween 20, 240 μg/ml proteinase K), incubated at 60°C for 1 hour, then heat inactivated at 94°C for 15 minutes. Two μl of lysate were used for polymerase chain reaction (PCR) amplification and direct sequencing to determine almost full-length 16S rRNA gene (rrs) and recN as well as partial rpoB sequences.22,24,25 As such, 1360 base pairs of rrs, 1300 base pairs of recN, and 520 base pairs of rpoB were used for analyses. Sequences were edited with the software Sequencher™, version 4.6 (Gene Codes Corporation, Ann Harbor, MI), and phylogenetic analyses were carried out in BioNumerics 5.1 (Applied Maths, Kortijk, Belgium).
RTX Toxin Gene Typing
Two microliters of lysate were used for PCR detection of genes encoding RTX toxins. PCR tests for apxIA, apxIIA and apxIIIA, 23 apxIVA, 35 and aqxA were performed as published. 5
Results
Pathology
At necropsy, both foals had limited extension of the carpal joints, up to 140° in foal No. 1 and 120° in foal No. 2. Foal No. 1 also had moderate mandibular prognathism (Fig. 1 ). Insufficient ossification of carpal bones was confirmed by postmortem radiology in foal No. 2. In both foals, the orad third of the small intestinal mesentery had tortuous, distended (diameter, 1.0-1.5 mm), off-white to yellow, and somewhat rigid lymphatic vessels extending from the small intestinal wall to the mesenteric lymph nodes (Fig. 2), which were swollen and had a blurred corticomedullary junction. Foal No. 1 had serial fractures of the right ribs with corresponding subcutaneous hemorrhage, perforation of the underlying lung lobe and pericardium, and fibrinous pleuritis, pericarditis, and epicarditis. Bilaterally, the psoas and masseter muscles had multiple pale dry foci with a hemorrhagic wet periphery. A grossly normal loop of small intestine had herniated through the umbilicus. The thyroid weighed 16.8 g; each lobe measured 4 × 2 × 2 cm.
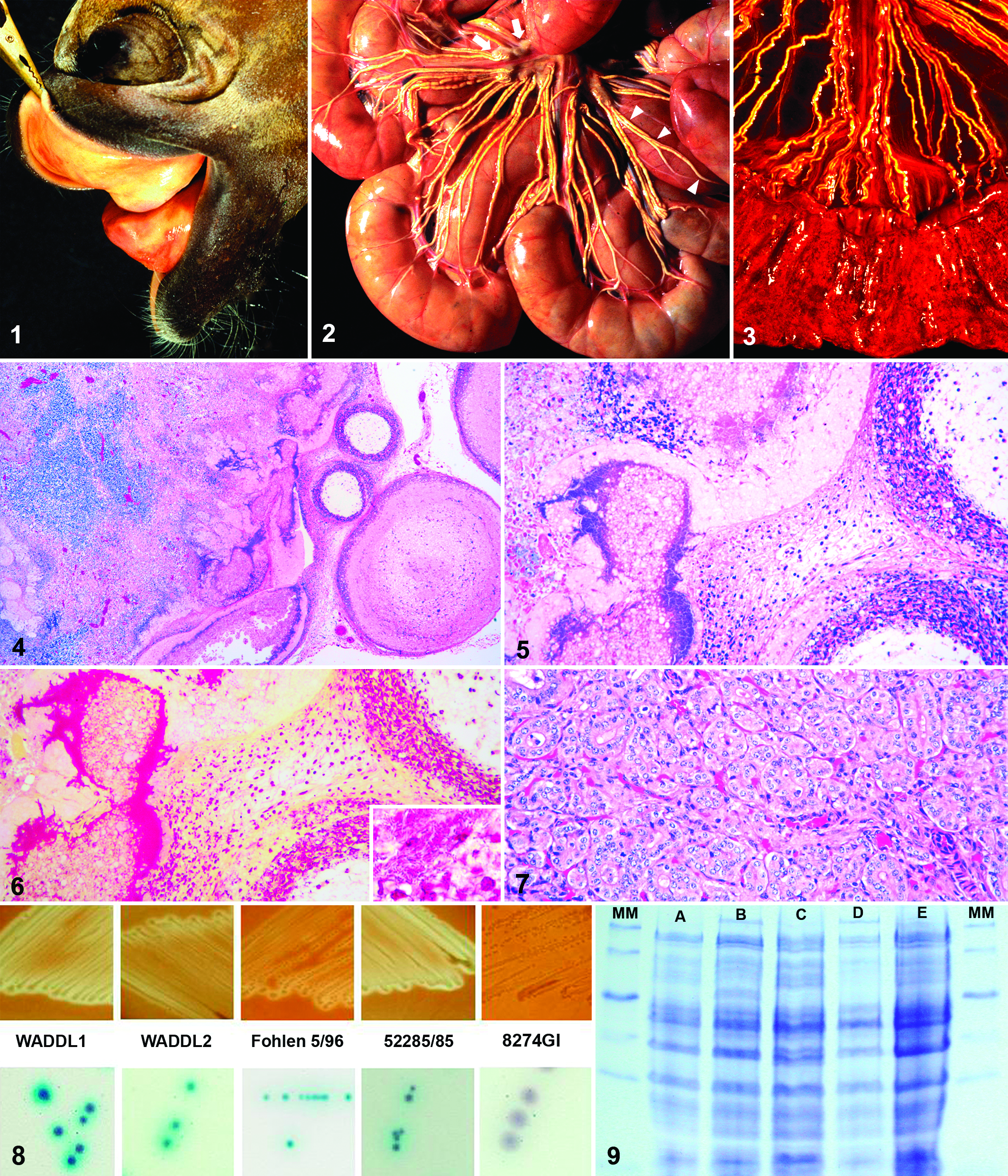
In foal No. 2, the orad portion of the small intestine was reddened and flaccid, with fine strands of fibrin on the serosa. This corresponded to severe hemorrhagic and necrotizing duodenitis and jejunitis with a thickened, rough, red mucosa, and bloody digesta (Fig. 3). The ileum had severe mural edema and fibrinonecrotizing enteritis with yellow-tan pseudomembranes. The mucosa of the colon and cecum was edematous with multifocal ulcers up to 1 cm in diameter. The thyroid lobes measured 3 × 2 × 1.5 cm each and together weighed 13 g.
On histology, intestinal and mesenteric lymphatic vessels and lymph nodes of both foals had thrombotic and necrotizing lymphangitis and lymphadenitis (Figs. 4, 5). Afferent mesenteric lymphatic vessels were dilated and occluded by fibrin thrombi, rare intact and numerous degenerate neutrophils in cellular debris, and large colonies of coccoid Gram-negative bacterial rods (Fig. 6). Necrosis extended through the lymphatic walls into the surrounding adipose and connective tissue. Similar changes were noted in the cortical sinuses of mesenteric lymph nodes of both foals, which also had lympholysis in germinal centers of foal No. 1 and sinus histiocytosis in foal No. 2.
In foal No. 1, Gram-negative bacterial rods were found in fibrinous exudate on the pleura, pericardium, and epicardium, as well as in thymic interstitium, terminal bronchioles, and hepatic sinusoids. Subepicardial myocardium had multifocal acute degeneration and necrosis. The lungs had multifocal suppurative alveolitis. Hepatocellular necrosis was focally associated with bacteria in the liver.
In foal No. 2, the duodenal and jejunal lamina epithelialis mucosae and superficial lamina propria mucosae were diffusely necrotic; the denuded submucosa was covered with myriads of short Gram-negative rods, as well as scattered long Gram-positive rods, rarely with terminal spores. Ileal submucosa and serosa were expanded by edema with scattered lymphocytes and plasma cells; remaining proprial and many submucosal blood vessels were occluded by fibrin thrombi. The lung had multifocal mild interstitial pneumonia with macrophages, lymphocytes, and plasma cells expanding interalveolar septa. A few small pulmonary arteries were occluded by fibrin thrombi containing colonies of coccoid bacteria, which were also in sinusoids of the congested liver.
Histologically, the hyperplastic thyroid gland of both foals had variably sized, generally small follicles with narrow lumens that lacked colloid (Fig. 7). Multifocally, the cuboidal follicular epithelial cells were piled up to 2 to 3 layers deep and had vacuolated cytoplasm.
Bacterial Cultures and Growth Characteristics
Numerous ASLOs were isolated from multiple tissues, including intestine from foal No. 1 (WADDL1) and foal No. 2 (WADDL2), and in pure culture from mesenteric lymph nodes of foal No. 1. Culture of the small intestinal contents from foal No. 2 also yielded moderate numbers of Clostridium difficile and few Clostridium perfringens. PCR toxin genotyping detected α and β-2 toxin genes in the C perfringens isolate. In addition to numerous ASLOs, moderate numbers of staphylococci and few E coli were isolated from the lung samples from both foals.
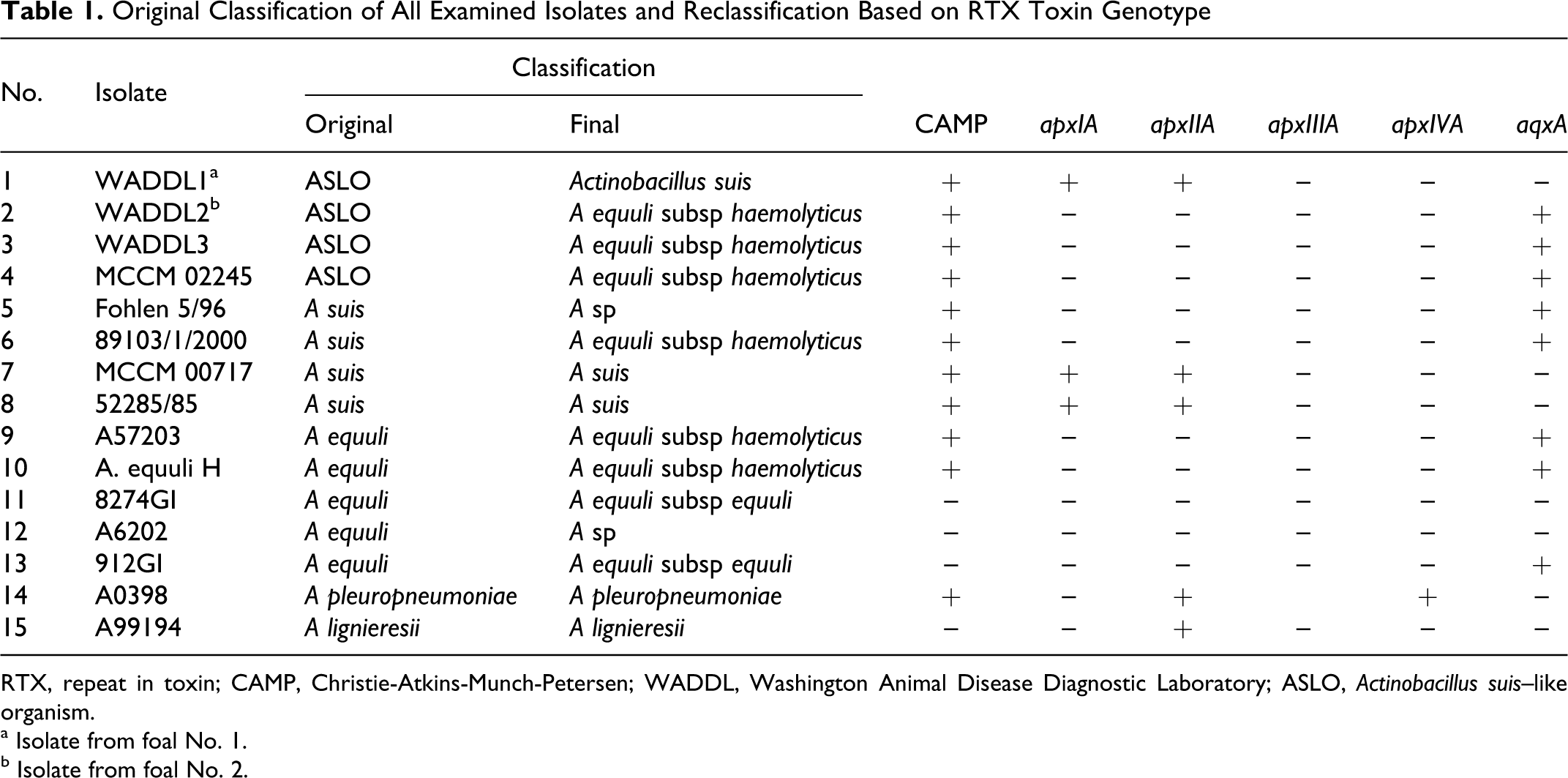
On bovine blood agar, isolates WADDL1 and WADDL2 grew as convex translucent gray colonies, 1 to 2 mm in diameter, and produced distinct zones of hemolysis (Fig. 8). The other equine ASLOs and the porcine A suis isolates, but not the A equuli isolates, had similar appearance on culture. On Chrom-Agar-Orientierung, WADDL1, WADDL2, and WADDL3 produced turquoise colonies; 1 other equine ASLO (Fohlen 5/96) and 1 porcine A suis isolate (52285/85) produced pale turquoise colonies, whereas 2 other equine ASLOs (MCCM 02245 and 89103/1/2000), the other porcine A suis isolate (MCCM 00717), and the single A equuli strain tested (8274GI) grew in violet colonies (Fig. 8, Supplementary Table 1). In the CAMP test, all equine ASLOs, all A suis, 2 A equuli, and A pleuropneumonia isolates tested positive, whereas all other isolates tested negative (Table 1).
Original Classification of All Examined Isolates and Reclassification Based on RTX Toxin Genotype
RTX, repeat in toxin; CAMP, Christie-Atkins-Munch-Petersen; WADDL, Washington Animal Disease Diagnostic Laboratory; ASLO, Actinobacillus suis–like organism.
a Isolate from foal No. 1.
b Isolate from foal No. 2.
Biochemical Characteristics and Antimicrobial Sensitivity
Supplementary Table 2 (available at http://vet.sagepub.com/supplemental) summarizes important biochemical characteristics of all isolates as evaluated by the API ZYM system. Equine ASLOs including WADDL1 and WADDL2 did not differ significantly from porcine A suis and A equuli isolates, except for the β-galactosidase activity, which was positive for most equine ASLOs and porcine A suis and negative for A equuli. All isolates were resistant to gentamycin and neomycin. In addition, WADDL 2 was resistant to ampicillin and chloramphenicol, and the isolate Fohlen 5/96 was resistant to ampicillin.
SDS-PAGE and MALDI-TOF Typing
Whole-cell SDS-PAGE (Fig. 9) and mass spectrometric analysis demonstrated no significant differences between WADDL1 and WADDL2 and the other examined isolates. The MALDI-TOF typing results underline the close relationship among the ASLOs, A suis, and A equuli. Due to the small score distances in the MALDI-TOF typing analysis, additional analyses would be required to explore possible differences.
Phylogenetic Analysis and High-Performance Liquid Chromatography Analysis of Nucleotides
Phylogenetic analysis was based on partial rrs, rpoB, and recN gene sequences. Whereas the rrs is generally used to delineate phylogenetic relationship, the rpoB and especially the recN have been proven to be ideal, high-resolution genes for phylogenetic analyses of Pasteurellaceae.22,25 However, species separation between A suis and A equuli and between A pleuropneumoniae and A lignieresii is not possible with rrs and not reliable on the basis of rpoB or recN sequences alone.22,25 Combining the sequence information of all 3 genes might increase the resolution. Figure 10 shows the combined phylogenetic tree based on all 3 genes determined for isolates listed in Supplementary Table 2 in comparison to the genera Pasteurellaceae. WADDL1 clusters closely with porcine A suis isolates and the A suis type strain, with the equine Actinobacillus sp isolate Fohlen 5/96 as a more distant relative. WADDL2 falls within 1 subcluster of A equuli subsp haemolyticus, whereas WADDL3 is most closely related to the other subscluster of A equuli subsp haemolyticus. No significant differences in the guanine–cytosine contents of bacterial DNA were observed among all isolates when assayed by quantification for deoxynucleotides following DNA digests. 38
RTX Toxin Genotype Determination
Table 1 summarizes the results of the RTX toxin genotype analysis. The presence of apxIA and apxIIA resulted in classification as A suis, whereas isolates that lacked these 2 genes and contained aqxA were classified as A equuli subsp haemolyticus. 23 The presence of apxA and aqxA genes corresponded with the phylogenetic clusters represented by A suis and A equuli, respectively. An exception is represented by the isolate Fohlen 5/96, which contained the aqxA gene but had no clear association with the other A equuli subsp haemolyticus strains (Fig. 10 , Table 1). The strain 912GI was positive for the aqxA gene but had a nonhemolytic phenotype. As expected, the RTX genes apxIA and apxIVA were amplified from the A pleuropneumonia and apxIIA from A lignieresii.23,35

Phylogenetic tree. Individual trees for rrs, rpoB, and recN were used to generate a consensus tree in Bionumerics. Jukes-Cantor correction and neighbor joining were used.
Discussion
The taxonomic grouping of equine A suis and ASLOs—including organisms reported as hemolytic A equuli, variants of A equuli, and Bisgaard taxon 11—and the relationship to porcine A suis isolates has been problematic.6,13,14,20 This is largely due to inconsistent and overlapping results obtained by serologic and biochemical analyses.4,6 Consequently, the usefulness of various approaches for differentiation of ASLOs by routine diagnostics has been questioned.6,14,20,23 Consistent with these observations, it was not possible to clearly distinguish the equine ASLOs examined here from porcine A suis and equine A equuli isolates based on phenotypic analysis alone.6,14,20,23 The results from the protein analysis by SDS-PAGE and MALDI-TOF typing further emphasize that these methods are too insensitive to discriminate among the examined Actinobacillus sp isolates. Due to the small score distances in MALDI-TOF typing, additional methods are required to further differentiate the strains.
Results from DNA-DNA hybridization studies, CAMP test, hemolysis, and epidemiologic data have led to the reclassification of equine A suis, A equuli, variants of A equuli, and Bisgaard taxon 11 with formation of 2 new A equuli subsp—namely, A equuli subsp haemolyticus and A equuli subsp equuli. 14 The hemolytic phenotype of A equuli subsp haemolyticus has been linked to the Aqx protein, a member of the RTX exotoxin family. 5 Genotypic analysis of the RTX toxin genes apxIA, apxIIA, apxIIIA, and apxIVA revealed a tight link between RTX toxin genotype of actinobacilli and the presence of a diffusible extracellular compound completing the hemolysis of sheep erythrocytes by S aureus (positive CAMP test). 23 Characterization of the RTX genotype allows for unequivocal differentiation among A equuli subsp haemolyticus, A equuli subsp equuli, and A suis. 23 Not surprising, several strains evaluated here—as initially classified by hemolysis, biochemistry, nucleotide content, and protein analysis—were reclassified after RTX genotyping. Chrom-Agar-Orientierung (CHROMagar, Paris, France), a general screening mechanism used to differentiate and presumptively identify certain bacterial genera and species on the basis of color and morphology alone,18,29 has not proven to be of value in differentiating between Actinobacillus sp or subsp.
The isolate from foal No. 1 was determined to be a true A suis (sensu stricto) based on hemolysis on ovine blood agar, a positive CAMP test result, and the presence of apxIA and apxIIA. 23 Since the reclassification of ASLOs, 14 the only nonporcine ASLO confirmed as A suis sensu stricto based on presence of apxICA and apxIICA were from a cat and a dog. 20 In contrast, the few genotypically characterized equine ASLOs have been identified as A equuli subsp haemolyticus based on absence of apxICA and apxIICA 20 or due to the presence of aqxA. 31 Our findings clearly disprove the notion that equine A suis isolates do not exist and are merely misnomers. 23
The isolate from the second foal was reclassified as A equuli subsp haemolyticus based on the presence of aqxA, absence of apxIA and apxIIA, and a positive CAMP test result.5,23 A equuli subsp haemolyticus has been implicated as a contributing factor to equine disease conditions.16,31 In the analysis of an isolate from an adult horse with pulmonary hemorrhage, PCR for aqx mRNA was used as a surrogate for direct testing for the toxin. 31 These results supported earlier implications of A equuli subsp haemolyticus as a contributing factor to the development of hemorrhagic conditions in the horse, with the caveat that classification of the ASLOs isolated from adult horses with pulmonary hemorrhage and infarction was based solely on phenotype. 12 Similarly, in a large retrospective study of mare reproductive loss syndrome, identification of A equuli subsp isolated from aborted fetuses and adult horses with pericarditis was based on phenotypic characterization and 16S rDNA (rrs) sequence similarity with the type strains. 16 This is problematic in that rrs sequence analyses only weakly define A equuli subsp, resulting in a comblike topology without consistency.7,14
In a large phylogenetic analysis of actinobacilli and related organisms, A equuli subsp haemolyticus dispersed into 2 subclusters: one clustering with porcine A suis isolates, the A suis type strain, and the type strain, equine and porcine isolates of A equuli subsp equuli; the other clustering with the A equuli subsp haemolyticus type strain but not porcine A suis isolates. 23 We obtained similarly confusing results in that the A equuli subsp haemolyticus isolates formed 2 subclusters, and the equine ASLO isolates from the 2 foals reported here and 2 additional isolates from septic foals represent a heterogeneous group of organisms. WADDL2 and the equine ASLO isolates (89103/1/2000), both reclassified as A equuli subsp haemolyticus, clustered with other A equuli subsp haemolyticus in the subcluster containing the A equuli subsp equuli type strain. WADDL3, reclassified as Actinobacillus sp, was not within but was most closely related to the second A equuli subsp haemolyticus subcluster, which contained 2 ASLOs from adult horses reclassified as A equuli subsp haemolyticus. WADDL1, confirmed as A suis sensu stricto (see above), clustered as expected, closely with porcine A suis isolates and the A suis type strain. 23 The unspeciated equine Actinobacillus isolate Fohlen 5/96 was also within this clad, albeit at considerable distance. Interestingly, the porcine and equine A suis isolates reported here did not fall into the same A equuli subsp haemolyticus subcluster containing A equuli subsp equuli, as previously reported. 24 The reason for these seemingly conflicting data may be too little sequence variation among taxa whereby random variation contributes too much noise to the analysis.14,23
All isolates examined in our study were resistant to gentamycin and neomycin. In addition, WADDL2 was resistant to ampicillin and chloramphenicol, and the isolate Fohlen 5/96 was resistant to ampicillin. Of the ASLOs reported as A suis (phenotypic analysis only) isolated from 2 septic foals, 1 isolate was sensitive to all tested antibiotics, and 1 had limited sensitivity to amikacin and penicillin. 30
CHDS is associated with severe musculoskeletal anomalies that impair the ability of affected foals to stand. 3 Foal Nos. 1 and 2 were presented with contracted tendons. Foal No. 1 also had prognathism, and foal No. 2 had delayed mineralization of carpal bones. Both foals had marked thyroid hyperplasia without increase in size of the gland. All these findings support the diagnosis of CHDS.1,3 The inability of either foal to stand could have led to decreased colostrum consumption with decreased passive transfer of immunity. This would have rendered the foals more susceptible to infectious diseases, including those caused by facultative pathogens such as ASLOs. Unfortunately, immunoglobulin levels were not determined in either foal. Interestingly, sepsis due to ASLOs reported as A suis (phenotypic analysis only) has been documented in 2 foals with adequate immunoglobulin levels. 30
A equuli is not a significant cause of equine abortion with the exception of the mare reproductive loss syndrome. In mare reproductive loss syndrome, Eastern tent caterpillar (Malacosoma americanum) exoskeleton and setae are suspected to cause mucosal damage and thus allow secondary invasion of the intestine by actinobacilli, which are isolated from many affected horses. 16 Foal No. 2 in our study had severe necrotizing to hemorrhagic enteritis, and moderate numbers of C difficile and a few C perfringens were isolated from the intestine, in addition to A equuli subsp haemolyticus. C difficile is recognized as a primary enteric pathogen and cause of necrotizing enteritis in the foal. 21 The C perfringens isolate carried α and β-2 toxin genes. Presence of the β-2 toxin gene in C perfringens is not considered a risk by itself, but selection of β-2 toxigenic C perfringens may lead to enteritis or enterotoxemia. 36 The damage to the mucosal barrier caused by the clostridial infection could have facilitated entry of A equuli subsp haemolyticus in foal No. 2. No evidence of enteric damage was identified in foal No. 1. Nonetheless, bacterial invasion through the digestive tract is most likely due to restriction of the lymphangitis to the mesentery. Bacterial invasion progressing to sepsis was not limited to actinobacilli in either foal, given that a few E coli and staphylococci were isolated from the lung of both foals.
The lymphangitis in both foals was necrotizing and thrombotic. Mesenteric lymphangitis is generally limited to infections with Rhodoccocus equi or Streptocccus equi subsp zooepidemicus, in which it has a granulomatous or suppurative character, respectively. 27 In other species, lymphangitis is usually granulomatous, as in Johne’s disease, 10 intestinal lymphangiectasia in dogs, 37 and intestinal emphysema in pigs, 9 with the exception of the thrombotic and necrotizing lymphangitis of restricted anthrax. 27 We found only a single report of thrombotic and pyonecrotizing lymphangiectasia and lymphangitis of the mesentery and mesenteric lymph nodes in a foal. 11 In that case, bacterial organisms were not identified in a cytology sample of the chylous abdominal effusion, and neither aerobic nor anaerobic cultures yielded bacterial growth. It was concluded that stasis of chyle rather than bacterial infection caused the lymphangitis. The authors speculated that the underlying problem may have been a congenital defect resulting in the failure of afferent and efferent lymphatics to connect or absence of efferent lymphatics. It cannot be completely ruled out that the 2 foals in our study may have had such an anomaly; however, this defect has not been reported in foals with CHDS,1–3 and it is more likely that infection with actinobacilli was responsible for the lymphangitis with secondary lymphangiectasia. The low success rate for isolation of bacterial pathogens from effusions in cases of parenchymal actinobacillary infections 12 could explain the failure to isolate bacteria from the chylous effusion in the reported case. 11 Toxic damage to endothelium is suspected as the primary cause of the lymphangitis in the 2 foals of our report, but presence of toxin was analyzed neither directly nor by surrogate methods, such as reverse transcription PCR, to detect the toxin gene transcript. 31
In summary, the cause of the thrombotic and necrotizing lymphangitis and lymphadenitis of the mesentery in the 2 foals presented here was probably an ascending bacterial infection as a sequel to intestinal colonization with A suis or A equuli subsp haemolyticus. Bacterial invasion through the digestive tract could have been facilitated by the necrotizing enteritis caused by C difficile in foal No. 2 and, possibly, by failure of passive transfer associated with CHDS in both foals. To the best of our knowledge, this is the first report of mesenteric lymphangitis associated with A suis sensu stricto or A equuli subsp haemolyticus infection in the horse. It was not possible to unequivocally classify the actinobacilli based on phenotypical characterization alone due to the insensitivity of these methods in differentiating between certain species and subspecies of this genus. These results emphasize the importance, if not necessity, of RTX toxin genotyping in the classification of this group of organisms.5,23
Footnotes
We dedicate this article to the late Dr J. Lindsay Oaks, an exceptional microbiologist with a strong interest in and commitment to wildlife conservation. We feel fortunate to have known and worked with Lindsay. He will be sorely missed.
Acknowledgments
We thank Drs Gary Lesamiz, Jason McFarland, and Melissa Hines for clinical workup of the foals necropsied at Washington Animal Disease Diagnostic Laboratory; the late Dr J. Lindsay Oaks for overseeing the routine microbiology of these cases; Drs Timothy Walsh and Kathleen Potter for assistance with postmortem evaluation. We acknowledge the contributions of the veterinarians and microbiologists to the isolation of the bacterial strains used for comparison.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
