Abstract
In humans, the glycosylation pattern of serum and of membrane glycoproteins is associated with invasiveness of tumors: specifically, α2,6-sialylation and α2,3-sialylation are associated with metastasizing and nonmetastasizing tumors, respectively. In turn, the type of sialylation depends on the activity of α2,6 or α2,3 sialyltransferase (ST) enzymes. Because of the high prevalence of metastasizing tumors with biological behavior similar to the human counterpart, female dogs with metastasizing neoplasms could provide a good animal model for investigating the potential roles of sialic acid (Sia) and ST enzymes in the pathogenesis of metastatic tumors. The aims of this study were (1) to validate a solid-phase method based on lectin staining of serum and tissue homogenates to investigate sialylation and ST activity and (2) to compare the results obtained with this method and with lectin staining and to collect preliminary information on sialylation and ST activity in dogs with (n = 8) and without (n = 8) mammary tumors. The data recorded in healthy dogs revealed that serum and tissue glycoproteins are prevalently characterized by a α2,6 sialylation, but ST-α2,3 seems to be the most active enzyme in both samples. Sia-α2,3 and ST-α2,3 activity decreases in serum and tissues of dogs with tumors, especially in a dog with metastasis, suggesting that the equilibrium between ST-α2,6 and ST-α2,3 activity shifts toward the former, as reported in humans.
Most of the glycoproteins expressed on the cell membranes contain sialic acid (Sia), which in mammals is mainly composed of a mixture of N-acetylneuraminic acid (NeuAc) and N-glycolylneuraminic acid (NeuGy). Specifically, Sia can bind C-3 or C-6 positions of galactose residues of glycoproteins (known as α2,3 or α2,6 glycosylation, respectively, also expressed as Sia-α2,3 and Sia-α2,6), C-6 position of N-acetylgalactosamine residues, or C-8 position of other Sia molecules. 3 In turn, the type of sialylation depends on the activity of the enzymes of the sialyltransferase (ST) family, which includes the enzymes β-galactoside-α2,3 sialyltransferase (ST3Gal I-VI or ST-α2,3) and β-galactoside-α2,6 sialyltransferase (ST6Gal I and II or ST-α2,6), responsible for α2,3 and α2,6 glycosylation, respec-tively.2,32
The expression of Sia-α2,3 and/or of Sia-α2,6 on cell membranes influences response of the cell to external stimuli. Specifically, sialylated glycoproteins play a chief role in viral infections and tumor invasiveness.3,33 Some viruses can bind sialylated receptors, and in most cases changes in viral virulence or in host spectrum affinity depend on the ability of viral strains to bind Sia-α2,3 instead of Sia-α2,6.18,21,29 As regards tumors, the altered glycosylation pattern of glycoproteins expressed on neoplastic leads to altered cell-to-cell adhesiveness and, ultimately, to the ability of neoplastic cells to detach from the primary site and acquire invasiveness and metastatic potential.4,16,25–28 In humans, metastasizing tumors are characterized by an increased expression of Sia-α2,6, whereas benign tumors express mainly Sia-α2,3. Moreover, a higher activity of ST-α2,6 has been reported in malignant colorectal cells5,9,35 and oral cancers 30 as well as in serum of patients with metastasizing tumors.6,13,30
Mammary tumors are frequent in dogs23,24 and share some common morphological and behavioral characteristics with humans. 17 For example, canine mammary neoplasms frequently metastasize, especially in lungs, as may occur in women.1,8,9,24,34 Thus, given the high prevalence of metastasizing tumors with biological behavior similar to the human counterpart, female dogs with metastasizing neoplasms could provide a good model to investigate the potential role of Sia and ST enzymes in the pathogenesis of metastatic tumors. Also, the diagnostic or prognostic role of Sia expression or ST activity in serum and tissues can be explored in the canine model. To our knowledge, no studies of Sia expression or ST activity in dogs are currently available, mostly because of the complexity of methods available to investigate Sia and ST activity.10,12,15 A less expensive and simpler method to investigate ST activity has been proposed,22,36 but its applicability to canine sera and tissues has never been tested.
The aim of this study is thus to investigate the presence of Sia-α2,3 or Sia-α2,6 in sera and tissues from dogs with mammary neoplasms with and without metastasis and to conduct preliminary assessment of the performance of a solid-phase method to evaluate the activity of ST-α2,3 and ST-α2,6 in the same dogs. This approach is performed on a limited number of samples (8 from dogs with tumors and 8 from clinically healthy dogs) to draw preliminary information about the possible role of these molecules in the pathogenesis of tumor metastases and, consequently, their possible use as diagnostic and/or prognostic markers in canine oncology.
Materials and Methods
Solid-Phase Method for Evaluation of Both Sia and ST Activity
The type of sialylation and the activity of ST were evaluated by modifying the methods proposed by Mattox et al 22 and Yeh and Cummings 36 to evaluate ST activity. These methods were based on the immobilization of nonsialylated molecules (asialofetuin) on ELISA plates, followed by the addition of samples in the presence of the donor of Sia cytosine 5′-monophospho-N-acetyl neuraminic acid (CMP-NeuAc). Sia released from the donor binds asialofetuin in position α2,3 or α2,6 depending, respectively, on the activity of ST-α2,3 or ST-α2,6 of the sample. The addition of the biotinylated lectins Maackia amurensis (MAL-I) or Sambucus nigra (SNA-I), specific for Sia-α2,3 and Sia-α2,6, respectively, in the presence of an avidin-conjugated enzyme and a chromogen generates a colored signal proportional to the amount of Sia bound in one or the other position.
The optical density (OD) recorded in sample wells cannot be converted into specific enzyme activity (micromole of product per minute per milligram of protein or units per milligram) because canine ST is not commercially available and it is thus impossible to build a standard curve to compare the results from individual dogs. Thus, the original method was partially modified by adding each sample either with or without CMP-NeuAc. The results obtained in the absence of CMP-NeuAc correspond to the baseline amount of Sia-α2,3 and Sia-α2,6; the results obtained in the presence of CMP-NeuAc correspond to the variation of Sia-α2,3 and Sia-α2,6 due to the activity of ST-α2,3 or ST-α2,6. Results regarding ST activities were thus expressed as percentage of variation of Sia-α2,3 and Sia-α2,6 compared with baseline values.
Additional modifications to the original protocol were the following: the amplification of the positive signal was done using the avidin–biotin–peroxidase complex (ABC) instead of streptavidin–alkaline phosphatase or aequorin, which were suggested by the original protocols;22,36 the optimal dilution of samples and lectins was modified compared with the original protocol, based on the results of a preliminary test on analytical performances of the method. This test was run on a pooled canine sample using a serum/buffer ratio ranging from 1 to 75% vol/vol followed by a linearity under dilution (LUD) test using blank wells (ie, wells without pooled samples) as a control; similarly, variable concentration of both lectins, ranging from 2 to 10 μg/ml, were tested. To complete the preliminary assessment of the method, for each experimental condition (eg, incubation with and without CMP-NeuAc for both SNA-I and MAL-I lectins) within-run (intra-assay) and between-run (interassay) imprecision were evaluated respectively by analyzing 5 replicates of the pooled canine serum in a single plate and by analyzing the pooled sample in 3 different plates.
The details of the method used in this study were thus:
Coating of the Microtiter Plate With Asialofetuin
Each well of a flat polystyrene microtiter plate (Nunc Apogen, Denmark) was filled with 100 μl of carbonate buffer pH 9.6 (Na2CO3 150 mM, NaHCO3 348 mM, 0.02% NaN3) containing 2 mg/ml bovine asialofetuin (asialofetuin from fetal calf serum, type I, Sigma Aldrich, Saint Louis, MO). The plate was incubated overnight at room temperature (RT) and then washed 3 times with 300 μl of phosphate-buffered solution (PBS) containing sodium azide (NaN3) using an automated plate washer (Bio-Rad Microtech, Hercules, CA). Each well was then filled with 300 μl of a blocking solution (PBS/NaN3 containing bovine serum albumin [BSA] 1%) and incubated 2 hours at RT. The plate was then washed as described before using the same washing solution added with 0.05% of Tween 20.
Assessment of Sia and ST Activity
The wells of the plate coated as described above were filled with 100 μl of serum samples or homogenized tissues (prepared as described below) diluted at the final concentration of 25% vol/vol in cacodylate buffer 50 mM, pH 6.5, to which was added 10 mg/ml BSA either with or without CMP-NeuAc (50 μM) and MnCl2 (10 mM). Specifically, for each serum or tissue sample, a sufficient amount to fill 8 wells was prepared. This allowed us to prepare duplicated wells for each sample and for each of the following experimental conditions: Incubation with CMP-NeuAc and detection with the lectin MAL-I Incubation without CMP-NeuAc and detection with the lectin MAL-I Incubation with CMP-NeuAc and detection with the lectin SNA-I Incubation without CMP-NeuAc and detection with the lectin SNA-I One row of each plate was used as a negative control, for which distilled water instead of samples was placed in the wells. Plates were incubated 2 hours at RT and then the wells were washed with 300 μl of PBS/NaN3/Tween 20 using the same procedure described above. To the wells dedicated to the evaluation of Sia-α2,3 and ST-α2,3, we added 100 μl of PBS containing 2 μg/ml biotinylated lectin MAL-I (Vector Laboratories, Burlingame, CA). To the wells dedicated to the evaluation of Sia-α2,6 and ST-α2,6, we added 100 μl of PBS containing 2 μg/ml biotinylated lectin SNA-I (Vector Laboratories). Plates were incubated 1 hour at RT, and then the wells were washed as described above. Each well was then filled with 100 μl of ABC (ABC kit, Vector Laboratories) diluted as suggested by the manufacturer (9 μl of reagent A and 9 μl of reagent B for each 1 ml of the buffer included in the kit). Plates were incubated 1 hour at RT, and then the wells were washed as described above. Then 100 μl of the chromogen substrate (peroxidase and tetra-methyl-benzidine) (R&D Systems, Minneapolis, MN) was added to each well. Plates were incubated 20 minutes at RT, and then the reaction was stopped by adding 50 μl of stop solution (sulfuric acid 0.2N) to each well. Plates were then read at 450 nm using an automated ELISA plate reader (Labsystems Multiskan MS, Dasit, Cornaredo, Italy), with 540 nm as reference wavelength.
Calculations
The OF of control wells was subtracted from the OF recorded in sample wells. Values recorded in wells incubated without Sia donor corresponded to the baseline Sia levels. The ST activity was calculated as percentage variation of OF of samples with Sia donor compared with the OF of samples without Sia donor.
Animals and Samples
This study was done on blood and tissues from 16 dogs sampled under informed consent of the owners following, with some modification, the study design described by Shah et al 30 in human patients with oral cancers. Blood was taken from 8 clinically healthy dogs during routine check-up visits (7 cases) or just before routine ovariectomy (1 case) and from 8 dogs with tumors during a presurgical workup. This workup included a complete physical examination (to collect information about number and size of neoplastic masses), the blood tests specified below, and a tomographic or radiographic examination of the chest to look for possible metastases.
Blood was collected from the cephalic vein, and part of the sample was transferred in tubes with EDTA to perform a routine hemogram. The remaining sample was transferred in plain tubes to obtain serum by centrifugation (500 × g for 15 minutes) to perform routine biochemistry and serum protein electrophoresis and to determine sialylation and ST activities, using the method described above. Serum samples were then frozen at –20°C until use.
Just after the presurgical workup, 1 clinically healthy dog was ovariectomized using the standard protocols followed at our institution: during surgery, under informed consent by the owner, a slice (approximately 1 cm × 1 cm × 0.2 cm) of mammary tissue was collected.
Seven dogs with tumors were mastectomized using the standard protocols followed at our institution: specifically, the whole mammary line and the draining lymph nodes were collected in all cases and processed as described below. The serum of 1 additional dog was included in this study. This dog was mastectomized in a private clinic that provided us with the results of histological examination. It was thus not possible to determine the type of sialylation and the ST activity on tissues samples of this dog.
The follow-up of dogs with tumors was monitored for 2 months (4 cases) or 6 months (4 cases): to this aim, clinical and radiological data and cytological results in the case of relapsing tumors were recorded.
Laboratory Assessment of the Health Status of the Dogs
Blood samples collected in EDTA were used to perform a complete cell blood count (CBC) using a laser counter (Sysmex XT2000iV) already validated for canine blood.19,20 The differential leukocyte count provided by the instrument was microscopically verified on blood smears stained with May Grünwald-Giemsa.
Serum samples were used to perform a basic panel of biochemical tests using an automated spectrophotometer (Cobas Mira, Roche Diagnostic, Basel, Switzerland). Specifically, the following parameters were evaluated, using reagents provided by Real Time Diagnostic System (Viterbo, Italy): lactate dehydrogenase (kinetic enzymatic method), γ-glutamyltransferase (GGT) (kinetic International Federation of Clinical Chemistry and Laboratory Medicine method), urea (urease method), glucose (Glucose Oxidase-Peroxidase method), total protein (biuret method), albumin (bromocresol method), Alkaline Phosphatase (ALP; kinetic IFCC method), creatinine (Jaffè method), alanine aminotransferase (ALT) (kinetic IFCC method), calcium (ortho-cresolphthalein method), inorganic phosphate (molybdate method), and creatine kinase (creatine kinase-N-Acetyl-Cysteine method).
To complete the diagnostic blood work, a serum protein agarose gel electrophoresis was performed using an automated apparatus (Hydrasis, Sebia Italia S.r.l., Bagno a Ripoli, Italy) and specific kits provided by the manufacturer of the instrument.
Tissue Processing
Immediately after surgery, mammary glands were macroscopically examined, and representative samples were obtained for each gland. Each sample was subsequently divided in multiple portions, half of which were immediately frozen in liquid nitrogen (–196°C) and stored at –80°C for histochemical staining of lectins and biochemical evaluation of Sia and of ST activity. The residual samples were fixed in buffered isotonic 10% formalin solution, routinely processed, and paraffin embedded using an automated tissue processing system (Sakura Tissue Tek VIP 5) (Sakura Finetek USA, Torrance, CA).
Formalin-fixed tissues were sectioned using a microtome (Leica RM 2125 RT) and frozen tissue samples using a rotating cryostat (Leica CM 1950) (Leica Microsystems Srl Milano, Italy). In both cases, multiple, 5-μm-thick serial sections were obtained from each sample.
Histology
Histological analysis was carried out on sections routinely stained with hematoxylin and eosin. In particular, slides were microscopically examined to classify the tumor according to the World Health Organization canine mammary tumors classification 23 and to evaluate the possible presence of micrometastases within the intra- or perineoplastic blood and lymphatic vessels.
Homogenization of Tissues
Nitrogen stored frozen mammary tissues was placed in an Eppendorf tube containing 300 μl of PBS and manually homogenized using an appropriate manual pestle. Tubes were then centrifuged (3000 × g for 10 minutes), and the supernatant obtained after centrifugation was transferred into another Eppendorf tube and frozen at –20°C.
Lectin Staining of Tissue Sections
Lectin histochemistry was carried out in double, both on microthomic (formalin-fixed, paraffin-embedded material) and on cryostatic (frozen in liquid nitrogen material) sections. Lectin were histochemically evaluated according to the following protocol:
14
Endogenous peroxidase was gently inhibited by placing the slides in Tris buffer, pH 7.6, to which was added 0.3% (vol/vol) H2O2 and 0.01% (wt/vol) NaN3 for 45 minutes. Slides were rinsed in Tris buffer and incubated 30 minutes at RT with a blocking solution composed of Tris buffer pH 7.6 with 1% wt/vol bovine serum albumin (BSA); then slides were washed in Tris (3 changes in 10 minutes). The biotinylated lectins MAL-I and SNA-I were diluted in the blocking solution. Preliminary experiments (data not shown) showed that optimal work dilutions were 1:500 and 1:1000 for MAL-I and SNA-I, respectively. Primary reagents were applied 1 hour at 37°C. One serial section of each sample was incubated with Tris buffer solution instead of lectins and was used as negative control. Slides were then washed in Tris, pH 7.6, for 10 minutes; then the ABC solution (prepared as described above) was applied on the slides for 30 minutes at RT. Slides were next washed as described above; then the chromogen 3,3′-diaminobenzidine (DAB, Peroxidase Substrate Kit, Vector Laboratories), prepared according to the manufacturer’s instructions, was applied for 45 seconds. Slides were rinsed in tap water for 5 minutes, counterstained with Mayer hematoxylin (5 minutes), rinsed in tap water, dehydrated in alcohol, and cleared in xylene. The coverslip was then mounted using an appropriate mounting medium (Eukitt, Darmstad, Germany).
Slides were microscopically analyzed to record the histological characteristics (eg, presence of mammary tissue, presence and type of neoplastic cells) and the characteristics of positivity. Lectin staining was semiquantitatively scored as follows: – no signal; +/– weak signal; + positive signal in fewer than 50% of the cells; ++ positive signal in more than 50% of the cells.
Statistical Analysis
Statistical analyses were performed on an Excel (Microsoft Corporation, Redmond, WA) spreadsheet with the Analyse-it set of macroinstructions (Analyse-it Software, Leeds, UK). For each experimental condition (eg, incubation with or without CMP-NeuAc and detection with MAL-I or SNA-I), the results obtained in controls and in dogs with tumors were compared with each other using the Mann-Whitney U-test. Within each group (controls and tumor) and for each detection system (MAL-I or SNA-I), the results obtained with and without CMP-NeuAc were compared with each other using a Wilcoxon paired test. The same test was used to compare, within each group, the OD recorded with MAL-I and SNA-I staining.
Results
Characteristic of the Dogs Included in the Study
None of the dogs classified as clinically healthy had laboratory abnormalities. Histology of the mammary tissue of the control dog did not reveal any morphological abnormality.
Data recorded from dogs with tumors are reported in Table 1 , where data are summarized considering the most malignant tumor recorded from each dog. When multiple nodules were retrieved, the tumors with most favorable grading are indicated in brackets. As shown in the table, 6 dogs had malignant neoplasms and 2 had lesions consistent with mammary hyperplasia/dysplasia. When multiple nodules were present, a different histotype (ranging from hyperplastic lesions to malignant lesions) was often recorded in different mammary glands. The degree of malignancy was higher in dog No. 1 than in others, as confirmed by a more aggressive histotype (simple anaplastic carcinoma) and by the presence of metastases, radiologically detected in the lung but likely present also in other organs, because the clinicopathological panel revealed severe hepatobiliary damage (increased ALT, ALP, and GGT). A metastasizing carcinoma was found also in dog No. 4 (carrying a simple tubulopapillary carcinoma), which had micrometastases in regional lymph nodes despite the lack of evident radiographic pulmonary changes and of biochemical or hematological changes. By contrast, all the other dogs with malignant tumors did not show clinicopathological abnormalities.
Health Status (Evidenced by Hematological and Biochemical Screening) of Dogs With Neoplastic Masses and Follow-Up of the 8 Dogs With Tumors
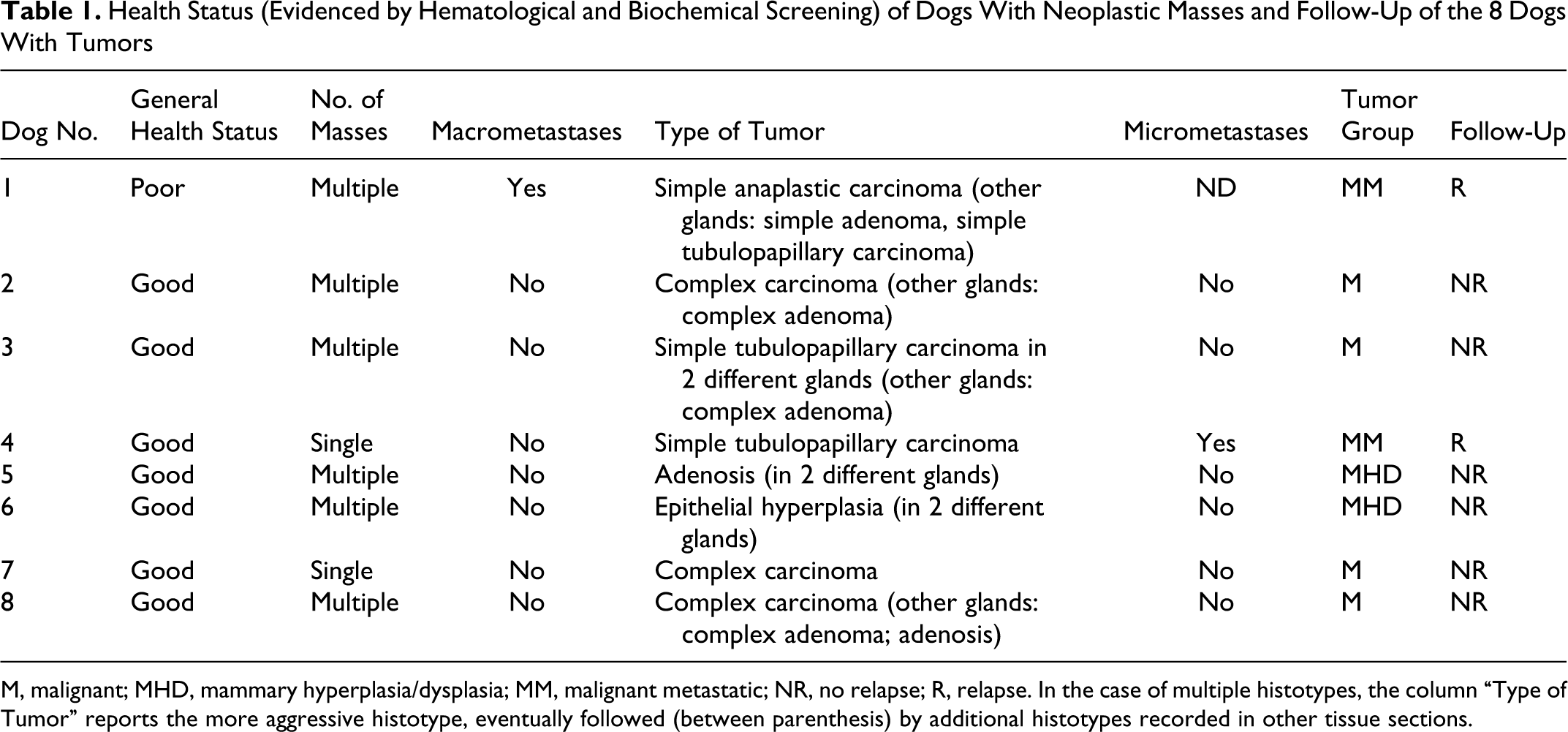
M, malignant; MHD, mammary hyperplasia/dysplasia; MM, malignant metastatic; NR, no relapse; R, relapse. In the case of multiple histotypes, the column “Type of Tumor” reports the more aggressive histotype, eventually followed (between parenthesis) by additional histotypes recorded in other tissue sections.
The aggressive behavior of tumors of dog Nos. 1 and 4 was confirmed by the follow-up: dog No. 1 was euthanatized 2 months after surgery because of relapse of the neoplasia, associated with a severe edema of the hind limb, likely due to a regional lymphadenopathy; dog No. 4 become dyspneic because of the presence of an abundant thoracic effusion, cytologically characterized by the presence of clusters of epithelial neoplastic cells, and the owner elected euthanasia of the animal.
Performances of the Solid-Phase Method to Assess Sialylation and ST Activity
The results of preliminary tests allowed us to obtain optimal characteristics of intensity of the colored products, without an excessive background in wells used for negative controls; using a serum concentration of 25% (vol/vol) in cacodylate buffer and using a final dilution of 2 μg/ml for both lectins provided good positive signal.
Both the repeatability and the reproducibility of the method were acceptable: intra-assay coefficients of variation (CVs) ranged from 3.7% (wells without CMP-NeuAc and with SNA-I) to 9.2% (wells with CMP-NeuAc and MAL-I); interassay CVs ranged from 7.8% (wells with CMP-NeuAc and MAL-I) to 12.8% (wells without CMP-NeuAc and with SNA-I).
Results of linearity under dilution tests are summarized in Fig. 1 . A dose-dependent increase of the OD was recorded in samples with increasing concentration of the pooled sample. This increase, however, was nonlinear and peaked at a concentration of 25% (vol/vol) of samples in cacodylate buffer. At the following concentration studied (50%), no further increases of OD were recorded or, in wells incubated with CMP-NeuAc followed by staining with the lectin MAL-I, the OD decreased.
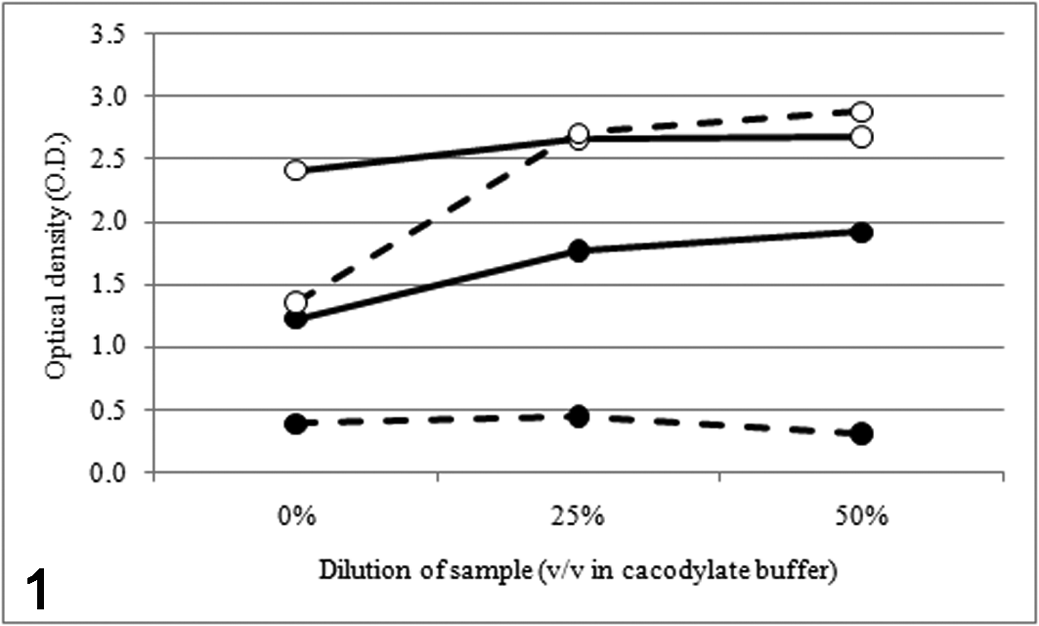
Results of the linearity under dilution tests using different dilutions of the sample in cacodylate buffer. Lines with solid circles refer to MAL-I reactivity (α2,3 sialylation); lines with open circles refer to SNA-I reactivity (α2,6 sialylation). Solid lines refer to baseline lectin reactivity (in the absence of the Sia donor); dotted lines refer to lectin reactivity in the presence of the Sia donor (CMP-NeuAc).
Sia and ST Activity in Serum
Both in controls and in dogs with tumors, the OD recorded in samples incubated without the donor of Sia was lower after labeling with MAL-I (0.82 ± 0.21 and 0.62 ± 0.19, respectively, without significant differences between groups) than after labeling with SNA-I (3.54 ± 0.27 and 3.61 ± 0.22, respectively, without significant differences between groups). As a consequence, the ratio between the OD recorded with MAL-I and SNA-I staining, ultimately corresponding to the Sia-α2,3/Sia-α2,6 ratio, was largely in favor of SNA-I (Sia-α2,6 sialylation) both in controls (0.23 ± 0.06) and in dogs with tumors (0.17 ± 0.05), with a significant difference (P < .05) between groups.
The mean OD recorded in the samples incubated with CMP-NeuAc and labeled with MAL-I was significantly higher than that recorded without CMP-NeuAc in controls (1.10 ± 0.35; P < .001), but not in dogs with tumors (0.67 ± 0.25), whereas both the values recorded after labeling with SNA-I were slightly lower (3.44 ± 0.23 and 3.58 ± 0.13, respectively) than those recorded without CMP-NeuAc. Also in the presence of CMP-NeuAc, the MAL-I/SNA-I ratio was significantly higher in controls (0.32 ± 0.10) than in dogs with tumor (0.19 ± 0.07). In controls, but not in dogs with tumors, the MAL-I/SNA-I ratio recorded in the presence of CMP-NeuAc was significantly higher than in the absence of CMP-NeuAc.
When the results obtained with CMP-NeuAc are expressed as percentage variation compared with those obtained without CMP-NeuAc (Fig. 2 ), the increase of MAL-I reactivity is much more evident in controls (34.7% ± 33.6%) than in dogs with tumors (8.1% ± 24.81%), whereas the slight decrease of signals detected in samples stained with SNA-I is similar in samples incubated with or without CMP-NeuAc (–2.5% ± 3.6% and –0.5% ± 4.4%, respectively). These differences were not statistically significant, but when data from individual dogs are considered (Fig. 3a), it is evident that this individual variability is basically associated with MAL-I reactivity that in the presence of CMP-NeuAc showed variable increases (ranging from 4 to about 90%), except in 2 pathologic dogs (Nos. 1 and 8), in which MAL-I reactivity decreased in the presence of CMP-NeuAc. In all the dogs, the variations in SNA-I reactivity recorded with CMP-NeuAc were minimal and basically were characterized by a decreased reactivity or by a slightly increased reactivity compared with that recorded in the absence of CMP-NeuAc. Specifically, the maximum increase of SNA-I reactivity in controls was 2%, whereas in 3 pathologic dogs the increase ranged from 3 to 4.1%.
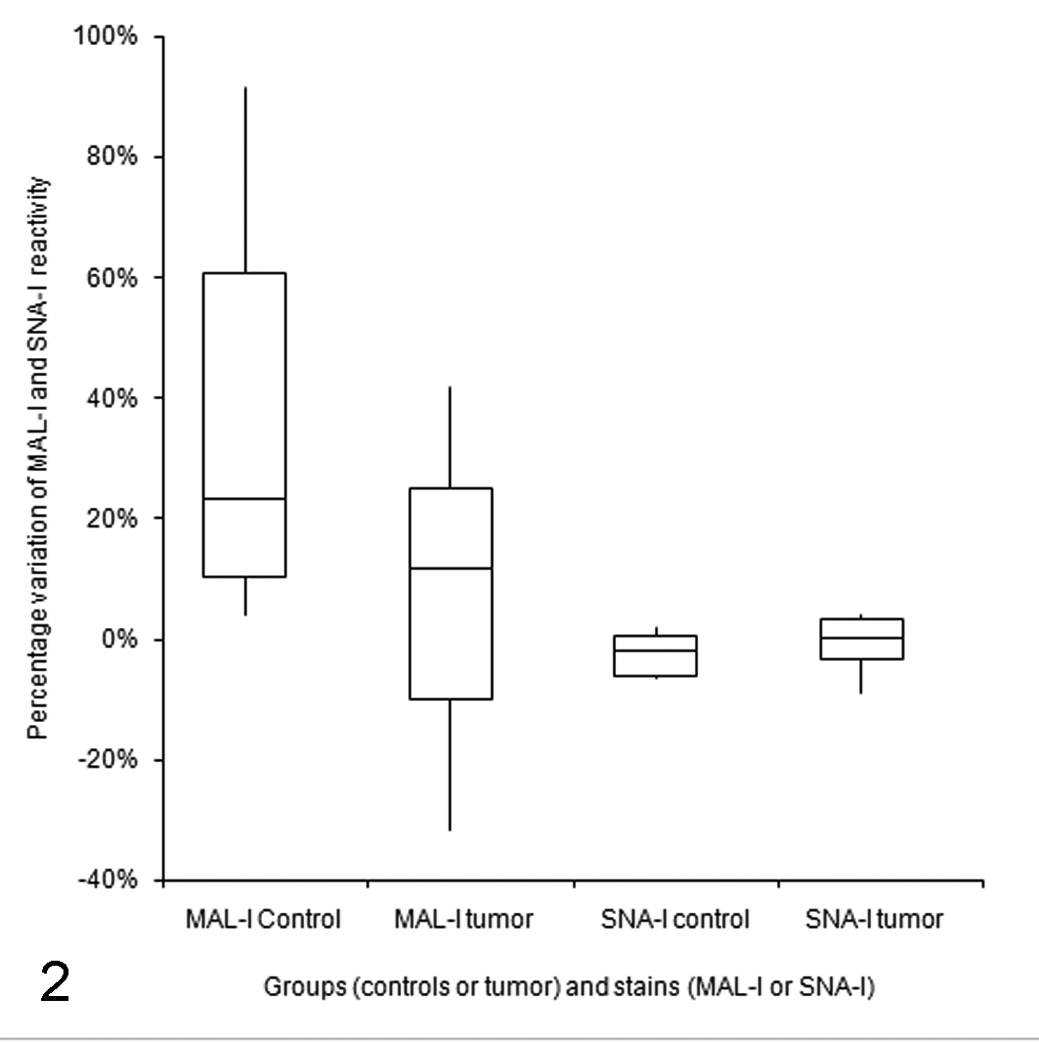
Distribution of results regarding the percentage variation of MAL-I and SNA-I reactivity in control dogs and in dogs with tumors. Scatterplots represent the I–III interquartile range, and the horizontal line shows the median value of each group.
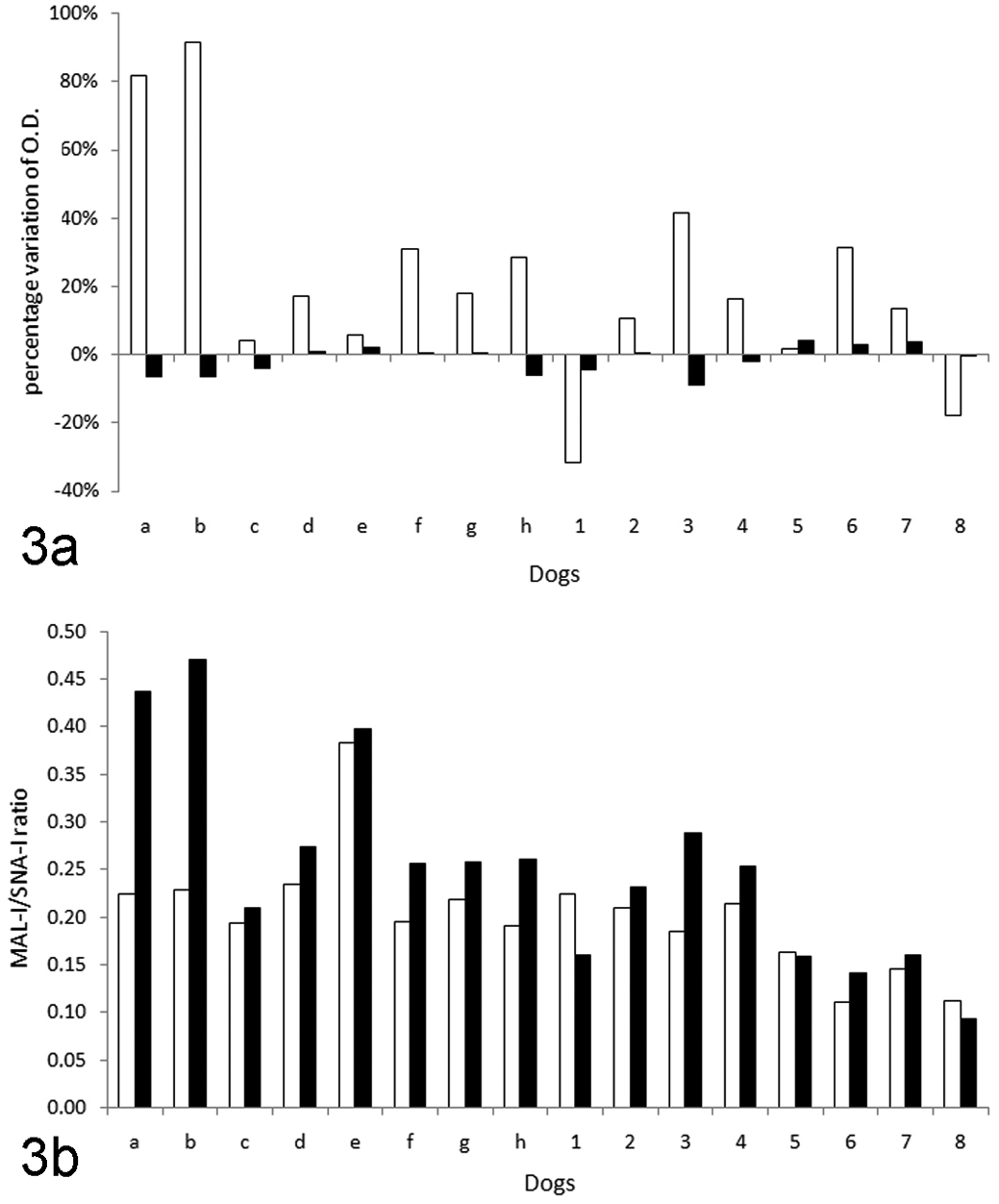
(a) Individual results (a–h = controls; 1–8 = dogs with tumors) regarding the percentage variation of optical density (OD) in the presence of Sia donor (CMP-NeuAc) compared with the OD recorded without CMP-NeuAc: white bars correspond to MAL-I reactivity (Sia-α2,3) and black bars correspond to SNA-I reactivity (Sia-α2,6). (b) MAL-I/SNA-I ratio recorded in individual dogs in the absence (white bars) or in the presence (black bars) of CMP-NeuAc.
The MAL-I/SNA-I ratios recorded in dog Nos. 1, 5, and 8 were lower in the presence than in the absence of CMP-NeuAc, whereas all the other dogs showed variable degrees of increase of the MAL-I/SNA-I ratio after addition of the Sia donor (Fig. 3b).
Sia and ST Activity in Tissues
The results of homogenized mammary tissues (Table 2 ) can be summarized as follows:
Optical Densities (OD) Recorded for Both MAL-I and SNA-I Lectins in Homogenized Tissues From a Control Dog and From Dogs With Tumors
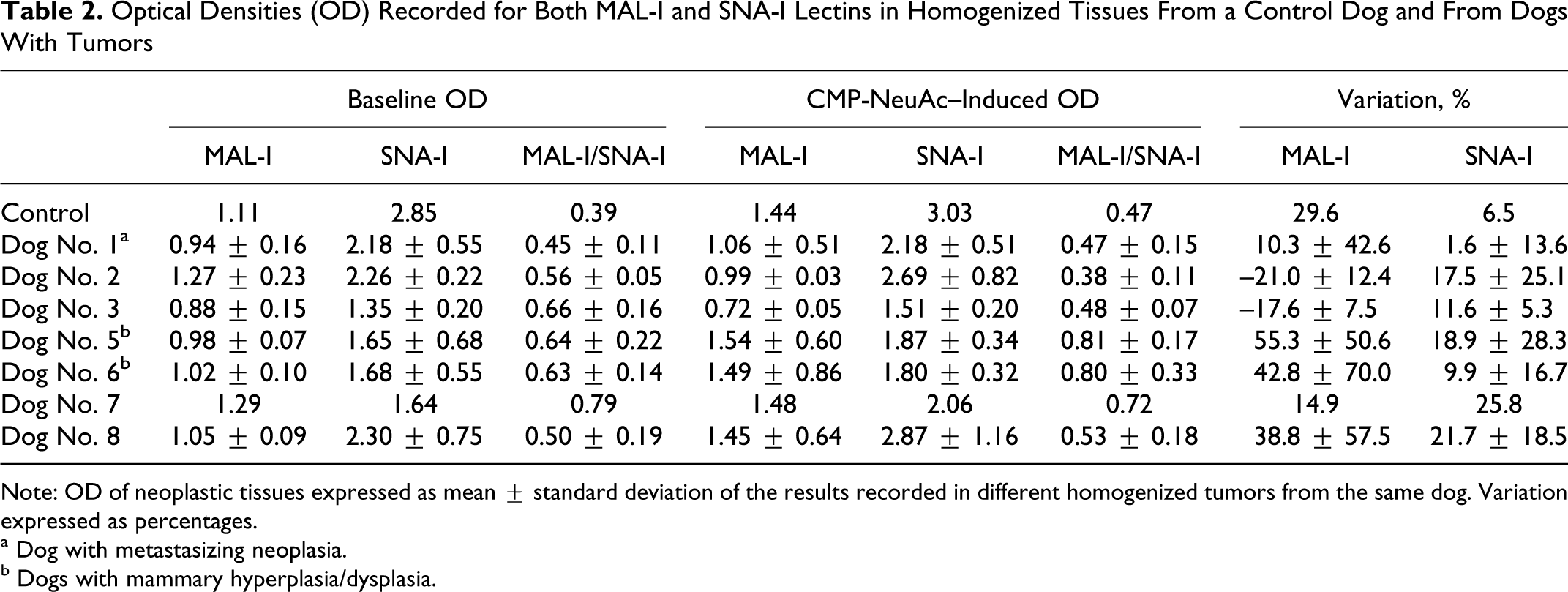
Note: OD of neoplastic tissues expressed as mean ± standard deviation of the results recorded in different homogenized tumors from the same dog. Variation expressed as percentages.
a Dog with metastasizing neoplasia.
b Dogs with mammary hyperplasia/dysplasia.
Sia content (baseline OD)
Baseline values were characterized by a prevalent SNA-I reactivity, indicating that Sia-α2,6 is more abundant than Sia-α2,3. The baseline MAL-I/SNA-I ratio of homogenized tumors, however, is higher than that of the control dog, due to both an increased Sia-α2,3 content and a decreased Sia-α2,6. The data reported in Table 2, however, were calculated on the basis of the results obtained from different masses and/or from different part of each single mass and are characterized by a certain degree of variability, as evidenced in Fig. 4 . This variability was confirmed in tissue sections stained with lectins. In particular, the distribution pattern of Sia was greatly variable depending on the site and on the tumor type as summarized in Table 3 . Lectin staining in paraffin embedded section did not provide satisfying results. Conversely, lectin staining on frozen sections provided excellent results in term of specificity of signal detection, especially for MAL-I (Fig. 5 ), even though sections stained with SNA-I often presented an intense background staining (Fig. 6). For both lectins, the positive signal was found in the cytoplasm of tubular epithelial cells in a perinuclear pattern (Fig. 7). Interestingly, spindle cells (myoepithelial and/or stromal cells) also stained with SNA-I (Fig. 8). Positive staining was evident in the amorphous material contained in tubular or cystic structures (Fig. 9) or on the cell membrane at luminal level (Fig. 10). With rare exceptions (see Table 3), MAL-I–positive cells were less abundant and less intensely stained than SNA-I–positive cells.
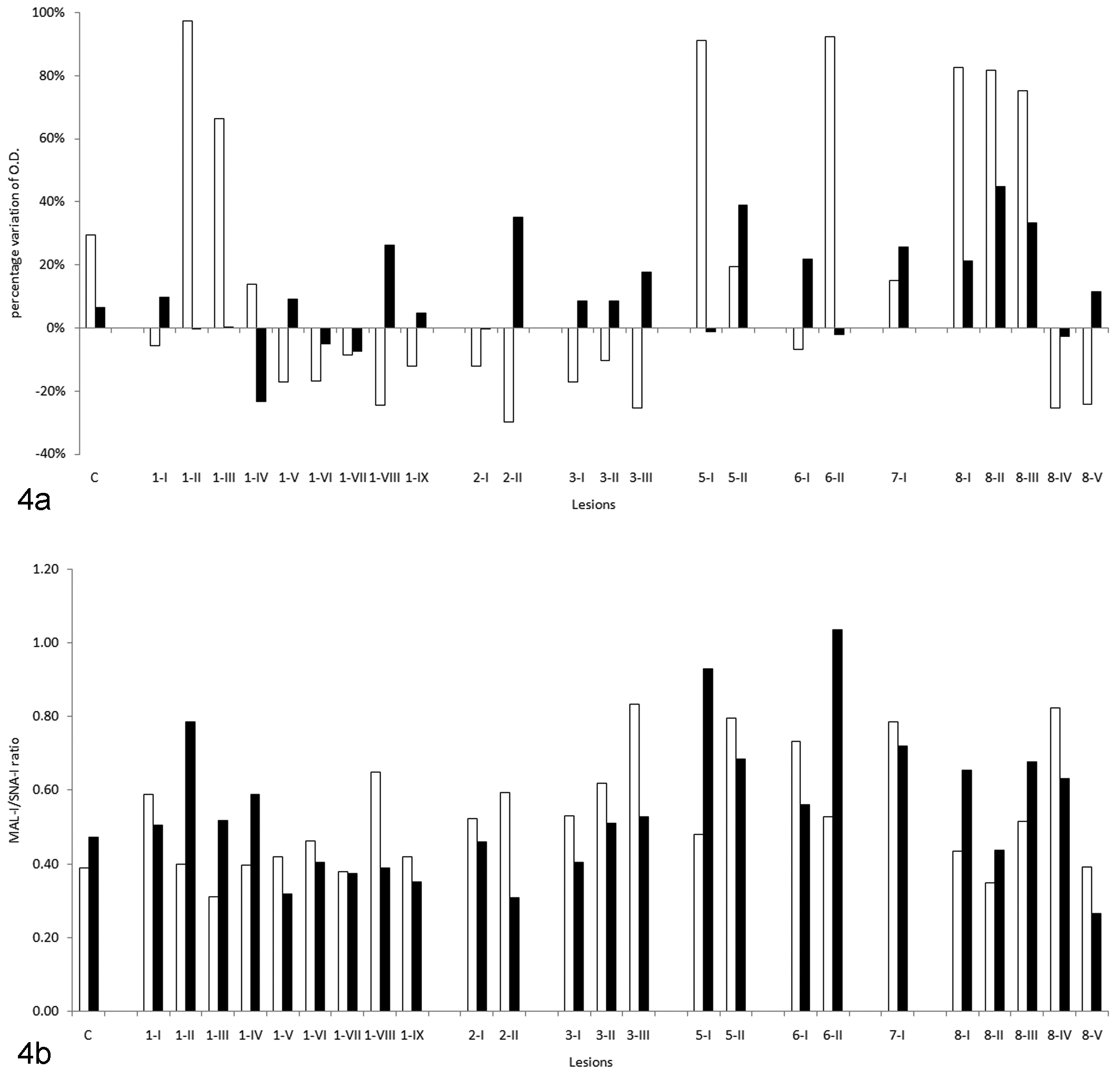
(a) Percentage variation of optical density (OD) in the presence of Sia donor (CMP-NeuAc) compared with the OD recorded without CMP-NeuAc in tissues from a control dog (C) and from dogs with tumors (1–8); I–IX indicate different lesions from the same dog. White bars correspond to MAL-I reactivity (Sia-α2,3) and black bars to SNA-I reactivity (Sia-α2,6). (b) MAL-I/SNA-I ratio recorded in the absence (white bars) or in the presence (black bars) of CMP-NeuAc.
Summary of the Positive Signals Recorded in Samples Collected From the Control Dog and From Different Areas (Lesion Number) of the Tumors in Pathologic Dogs (Mammary Tissue of Dog 4 Was Not Examined)
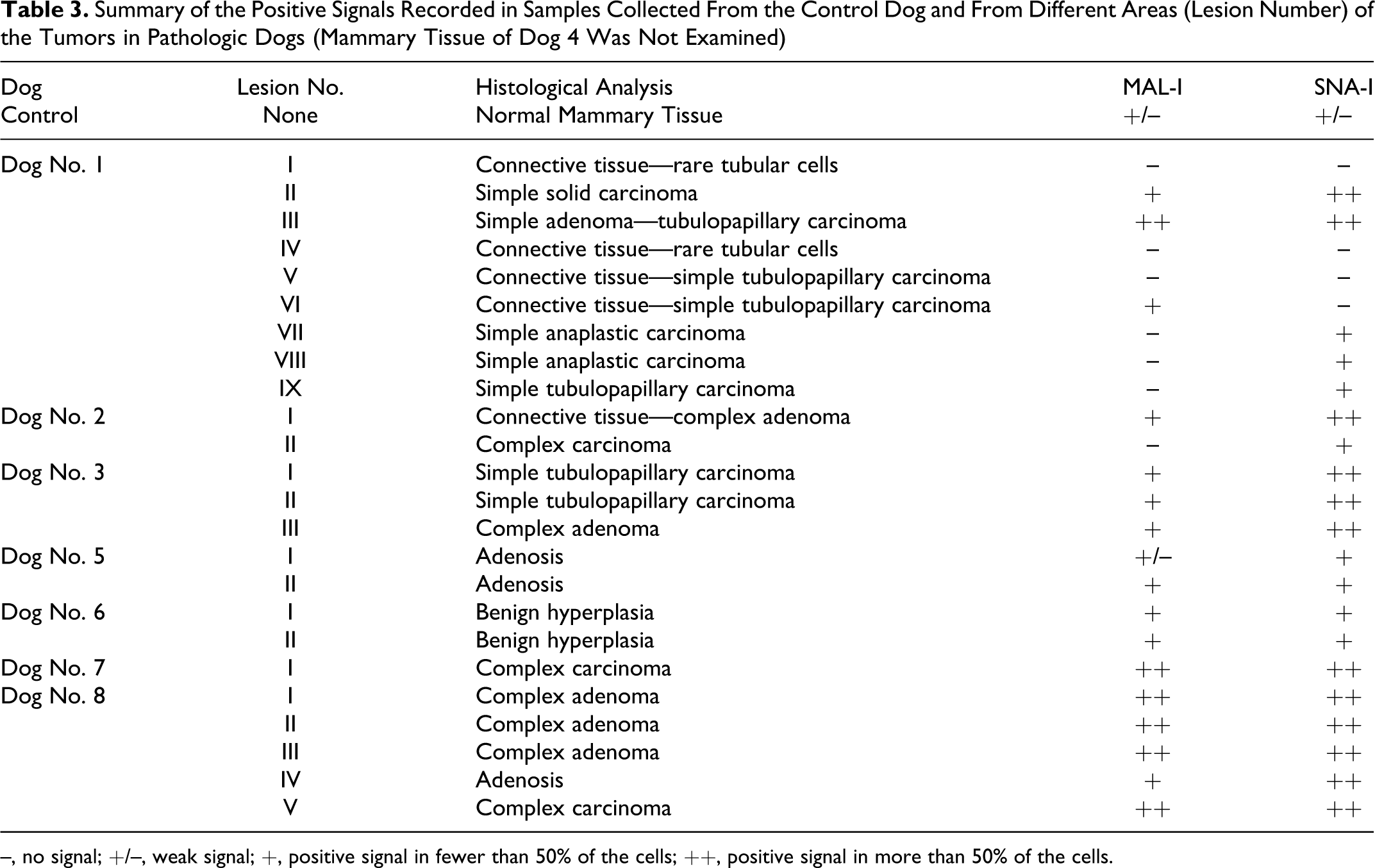
–, no signal; +/–, weak signal; +, positive signal in fewer than 50% of the cells; ++, positive signal in more than 50% of the cells.
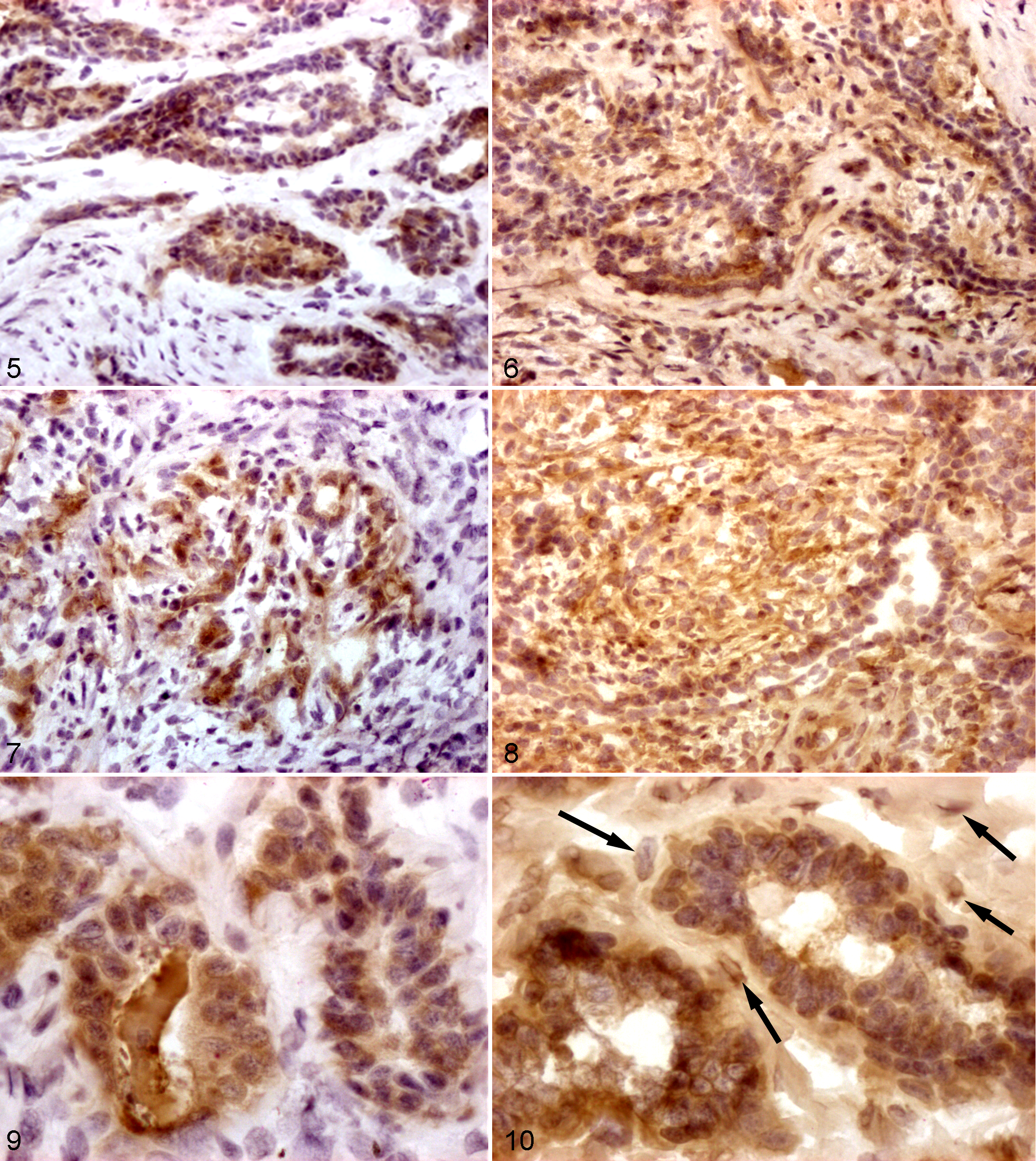
Mammary gland, mammary complex adenoma; dog No. 3. Intense MAL-I staining is restricted to neoplastic epithelial cells. Frozen serial sections: histochemistry with lectin MAL-I, which specifically recognizes Sia-α2,3. DAB chromogen; Mayer’s hematoxylin counterstain. 200×.
Sialyltransferase activity
The analysis of data recorded with CMP-NeuAc (Table 2) evidenced an increased MAL-I reactivity in the control dog, associated with a slight increase of SNA-I reactivity. Increases of MAL-I reactivity of different magnitude were found in other dogs with tumors, except for dog Nos. 2 and 3, that showed a decreased MAL-I reactivity. Conversely, increases of SNA-I activity were more intense in dogs with tumors than in the control dog, except for the dog with macrometastases (dog No. 1), in which only a moderate increase of SNA-I activity was found.
As a consequence, the MAL-I/SNA-I ratio of decreased compared with baseline values in dog Nos. 1 to 3, in which the ratio become similar to that of the control dog. In all the other dogs, this ratio was higher than the baseline ratio. In the case of ST activity, however, an extreme variability between the different homogenized samples was present (Fig. 4b).
Discussion
To our knowledge, this is the first study in which both the type of Sia and ST activity are investigated in serum and tissues of dogs with mammary tumors. A previous study revealed that the concentration of total Sia in serum increases in dogs with tumors but did not provide information about the type of sialylation or about ST activity. 31 The technique used in the present study has never been tested in dogs, and our preliminary validation tests demonstrated good repeatability and reproducibility, although the interpretation of data regarding ST activity in serum must be interpreted with caution, as specified below. The technique is inexpensive and easy to perform, but it is quite time consuming and is laborious in its preliminary phase (ie, preparation of buffers, dilutions, etc). This limits the possibility of a wide-scale application of the method unless preliminary information identifies practical advantages associated with this approach. For this reason, we limited the analysis of individual data. The low number of observations did not allow us to draw definitive conclusions about the role of both the type of sialylation and the ST activity in dogs with mammary tumors but provided promising results on the applicability of the method in research studies. Before we recommend a wider use of the technique, further modifications of the original protocol should be tested, such as the availability of canine ST to be used as a reference material to build a standard curve, thus avoiding excessive duplication of tests to determine the variation of reactivity due to the ST activity compared with baseline values.
This technique has been designed, in humans and in laboratory animals, to assess the ST activity based on the different reactivity to MAL-I and SNA-I lectins induced by the addition of CMP-NeuAc, which works as a donor of Sia, in comparison with a standard curve obtained using samples with known activity of ST. To our knowledge, there are no reports about using this technique, without the addition of donors of Sia, to establish the baseline type of sialylation (α2,3 and/or α2,6). This latter approach in the present study revealed that the large majority of Sia in blood is bound in position α2,6 on circulating glycoproteins, independent of the breed or age of the dogs. In this regard, it should be stressed that dogs were randomly selected because no information about a possible breed-associated genetic background on protein sialylation or ST activity is available in literature. The lower ratio recorded in dogs with tumors compared with controls could suggest that α2,6 sialylation is more active in this group, but the analysis of ST activity (see below) revealed that this likely depends on a decreased activity of ST-α2,3.
Results in tissues were extremely variable for both the analysis of homogenized tissues and the lectin staining of tissue sections. This variability, in turn, could depend on the tissue composition: mammary neoplasia in dog, in fact, presents peculiar growth patterns such as the coexistence of multiple masses with different histotypes or with malignant versus benignant patterns. As a consequence, a single mass can show different lectin phenotypes. Moreover, the extremely high stroma/epithelial ratio, with few malignant epithelial cells invading and inducing the proliferation of an abundant connective tissue presented by some malignant tumor, resulted in a low Sia positivity on homogenized tissues characterized by a clear positivity of malignant epithelial cells. In these cases, some spindle cells were SNAI-I positive. This especially occurred in complex tumors, suggesting that most of the positive spindle cells were myoepithelial: in the absence of immunohistochemical characterization, however, it is not possible to conclude whether SNA-I–positive cells were myoepithelial cells or stromal fibroblasts. Finally, some tumors were characterized by dilated, tubular structures, thus further reducing the number of epithelial cells in the section. Thus, gaining more precise information about Sia content requires a better standardization of samplings, for example, by using laser picking techniques to isolate single cells or glandular structures from their stromal support. 7 Despite these technical limitations, the analysis of tissues was basically in agreement with the results obtained in serum: both the mean Sia content of neoplastic tissues and lectin staining revealed a prevalence of Sia-α2,6. Sia-α2,3 was also present in a higher amount compared with normal tissues, although a sialylation pattern consistently associated with malignancy or with the presence of metastases was not found, likely because of the variable composition of tissue samples mentioned above.
As regards ST, the protocol used in this study determines ST activity rather than the simple presence of ST molecules or the expression of ST genes. These latter factors can be determined using immunohistochemistry or quantitative PCR,6,9,13 but ultimately both these approaches would have provided information about the presence of the enzymes but not about their actual activity. ST activity is usually assessed by adding CMP-NeuAc to the reaction mixture. This induces variations of MAL-I and SNA-I reactivity due to the binding of Sia provided by the donor to the acceptor (asialofetuin): the magnitude of variations in turn depends on the activity of ST-α2,3 and/or ST-α2,6. As mentioned above, in humans the ST activity is extrapolated from a standard curve built with a reference standard solution. This is not available for canine, and we thus modified the method by expressing the activity of ST enzymes in terms of changes of lectin reactivity after addition of the Sia donor compared with baseline values. Using this approach, we generated both positive and negative results (ie, ST activity higher and lower than baseline values, respectively). The significance of negative results theoretically could raise some doubt regarding the actual performance of the approach. Nevertheless, the dose-dependent increase of sialylation recorded in preliminary tests both in the presence and in the absence of the Sia donor suggests that enzyme activity is present in both types of samples. It is unlikely that the decrease recorded with CMP-NeuAc depends on the activation of sialidase enzymes, because sialidase is down-regulated in cancer. 36 In addition, sialidase would have exerted its effect on wells both with and without Sia donor. Ultimately, the most likely interpretation of the negative results obtained especially with MAL-I staining is that samples contain a prevalent ST-α2,6 activity, thus reducing the Sia-α2,3/Sia-α2,6 ratio compared with baseline values.
Apart from methodological considerations, serum samples of the 2 groups revealed evident differences of mean values in the presence of CMP-NeuAc. This difference was significant only for ST-α2,3 and not for ST-α2,6, likely because of the low number of animals per group and the high individual variability evidenced within both groups. Nevertheless, the analysis of individual data revealed a prevalent ST-α2,3 activity in healthy dogs, which showed an increased MAL-I reactivity (the Sia provided by the donor was prevalently bound in position α2,3) with minimal changes in the SNA-I reactivity, leading to an increased Sia-α2,3/Sia-α2,6 ratio. In most dogs with tumors, the increases of MAL-I reactivity and of the Sia-α2,3/Sia-α2,6 ratio were less intense than in controls. In 2 pathologic dogs, both the MAL-I reactivity and the Sia-α2,3/Sia-α2,6 ratio decreased, suggesting that the activity of ST-α2,3 is depressed, thus leading to a prevalent ST-α2,6 activity. Interestingly, this decrease was observed in the only dog that, when sampled, had macrometastases and in 1 of the 2 dogs with multiple malignant masses. This abnormal ST-α2,3/ST-α2,6 pattern thus could be associated with the degree of malignancy and with the metastatic process, as postulated in humans.5,6,13 It cannot be excluded that the depressed ST-α2,3 activity in dog No. 1 could be attributed to liver damage because impaired liver function compromises most of the protein synthesis in the body. In this case this hypothesis is true; however, the ST-α2,6 activity also should have been depressed in dog No. 1, as demonstrated in human patients with alcoholic liver disease. 11 In contrast, in the case of a direct association between the presence of metastases and depressed ST-α2,3 activity, dog Nos. 4 and 1 should have evidenced the same ST-α2,3/ST-α2,6 pattern. However, dog No. 4 did not have macrometastases at the time of sampling, and unfortunately we did not repeat the sampling 1 month later, when the pulmonary metastases become evident, to assess whether the pattern associated with overt metastases was similar to that of dog No. 1.
Taken together, these results suggest that in the presence of tumor with a higher degree of malignancy, the activity of ST-α2,3 in serum is moderately decreased, thus shifting the equilibrium toward a prevalent ST-α2,6 activity, in turn responsible for the higher Sia-α2,6 recorded in serum of dogs with tumors.
Similar to what was reported in serum, in most of the neoplastic tissues the ST-α2,3 activity was lower than in controls and this decrease was associated with increased ST-α2,6 activity. In contrast to serum, however, these changes were moderate in the dog with macrometastases and more evident in dogs without metastases. As for the type of sialylation, however, the results for ST activity in homogenized tissues were characterized by a huge variability between different samples for the same patient, likely due to the above-mentioned variable composition of different tissues samples (eg, amount of stroma).
In conclusion, this study revealed that both Sia content and ST activity can be studied by combining a solid-phase method of evaluation of both the parameters in serum or in homogenized tissues and the lectin staining of tissue sections. Some methodological limitations reduce the wide-scale applicability of the method. Better standardization of tissue samplings and the availability of reference material to standardize the analysis of ST activity in fluids are needed. Despite these technical limitations, the analysis of a few samples from dogs with and without tumors provided encouraging results. In both types of samples from healthy dogs, Sia-α2,6 was prevalent but ST-α2,3 seemed to be the most active enzyme. Both Sia-α2,3 and ST-α2,3 activity decreased in serum and tissues of dogs with tumors. In serum, this decrease was particularly evident in the dog with macrometastases, whereas in tissues it occurred mainly in dogs without metastases. These preliminary results suggest that in dogs, as in humans, metastasizing tumors can be associated with an altered sialylation pattern of glycoproteins and that this alteration depends on an imbalance between ST-α2,3 activity, which decreases in dogs with metastases, and ST-α2,6 activity, which has little variation in dogs with metastatic tumors. Given on these results, larger and more standardized studies on the possible pathogenic and diagnostic role of Sia and of ST activity in dogs with mammary tumors are advisable.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This study was funded by the University of Milan (Grant F.I.R.S.T. 2008). The authors are grateful to Dr D. Groppetti, who participated in the selection of patients.
