Abstract
Canine cutaneous histiocytoma (CCH) is a noninfectious tumor that spontaneously regresses. It is suggested that this regression is due to tumor cell maturation, which is responsible for CD8 lymphocyte activation and tumor cell destruction. Nevertheless, the possible role of the immune microenvironment in tumor regression has not been investigated to date. The aim of this study was to investigate the expression of CD208 and FoxP3 as markers of dendritic cells and regulatory T lymphocytes, respectively, and tumor cell expression of CD206 as a marker of Langerhans cell activation, and relate these parameters to the different phases of CCH and to intratumoral T cell infiltration. Formalin-fixed, paraffin-embedded samples from 31 CCH were evaluated. In each case, the mitotic count and regression phase were recorded. Within the tumor, a quantitative evaluation of immunolabeled CD208+ cells, FoxP3+ cells, and CD3+ lymphocytes was performed, as well as the CD206+ tumor cell location. Intratumoral CD208+ cells correlated with CD3+ lymphocytic infiltration. The possible role of dendritic cells in tumor regression was not confirmed since CD208 seemed to be a nonspecific marker for canine dendritic cells. FoxP3+ lymphocyte density was not correlated with any parameter. Neoplastic Langerhans cells presented progressive CD206 expression, from the bottom of the tumor to the epidermis, which correlated with the tumor regression phase and with intratumoral T lymphocyte infiltration. In conclusion, we confirmed a CD206 phenotype change in tumor cells in a spatial group-related pattern, supporting the hypothesis that tumoral Langerhans cells acquire a mature phenotype with tumor regression.
Keywords
Canine cutaneous histiocytoma (CCH) is a common neoplasm derived from cutaneous Langerhans cells, as demonstrated by immunophenotype and ultrastructural studies. 16,17,20 CCH affects mainly young dogs and its incidence decreases after 3 years of age. 12,15,20 Despite the rapid growth and high mitotic index of CCH, in most cases it spontaneously regresses, and rarely metastasizes. Histologically, CCH is a nodular lesion extending from the subcutaneous tissue and/or the deep dermis to the epidermis. Progressively, T and B lymphocytes infiltrate the tumor, starting from the deep periphery and moving toward the center and up to the surface of the tumor under the epidermis. 6,12,20,27 Although lymphocyte infiltration is correlated with tumor regression, the precise mechanism responsible for lymphocyte activation is unknown. The hypothesis of tumor Langerhans cell maturation is suggested based on the observation of spatial changes in the tumor cell phenotype. Specifically, paralleling the progression of lymphocyte infiltration from the depth toward the surface of the tumor, MHC II labeling of tumor cells increases from the deep periphery to the epidermis. 15,27 Moreover, membranous E-cadherin expression, a marker of immature Langerhans cells, decreases with the same spatial pattern. 15,26,27 Based on these observations, it is hypothesized that tumor cells become activated and mature, from deep margins to the epidermal surface. The maturation of tumor cells is associated with increased CD4+ T lymphocytes and the recruitment of antitumor effector cells, such as CD8+ T lymphocytes and M1 macrophages. 12,27 Nevertheless, the maturation hypothesis has never been definitively confirmed, and the possible role of the immune microenvironment in lymphocyte recruitment has not been investigated.
Tumors are dependent on the microenvironment. The balance between anti-tumoral and pro-tumoral activity of the tumor microenvironment is responsible for tumor progression and metastasis or tumor regression and eventually disappearance. 45 Tumor-infiltrating dendritic cells and lymphocytes are important actors in this balance.
Dendritic cells (DCs) are professional antigen presenting cells. In the immature state, DCs reside in peripheral organs, where they capture and process antigens. After encountering the antigens, DCs change their phenotype, secrete pro-inflammatory cytokines, and migrate toward afferent lymph nodes. 37 In the presence of costimulatory signals, they can present the antigens by MHC I to stimulate CD8+ cytotoxic T cells, and by MHC II to stimulate CD4+ T lymphocytes. In the absence of costimulatory signals, DCs induce an immunosuppressive T cell response. 1,4 In cancer, DCs play a critical role in priming T lymphocytes against tumoral antigens, and they represent an important target for cancer immunotherapy. CD208 is a lysosome-associated protein involved in antigen presentation. 32 CD208 is expressed in human, porcine, and bovine DCs, 10,31,32 and has been used in canine histiocytic sarcoma tumors and cell lines to demonstrate a dendritic cell phenotype. 39,40
Regulatory T lymphocytes (Tregs) are CD4+ T lymphocytes with an immunosuppressive role. Different subsets have been identified, including endogenous and inducible Tregs. Endogenous Tregs are naturally occurring cells that develop in the thymus. When stimulated, these cells suppress the activity of self-reacting lymphocytes in a cell-contact-dependent manner. Inducible Tregs are produced under antigen stimulation in peripheral tissues. These cells induce immunosuppression in a cell-contact-independent manner, by secreting cytokines such as interleukin-10 and transforming growth factor-β. 8 Tregs mediate immune tolerance in most immune cells, such as T and B lymphocytes, natural killer lymphocytes, and antigen presenting cells, including DCs and macrophages. 8 In some cancers, the presence of Tregs is correlated with an immune tolerant microenvironment and a worse prognosis. 28,33,47 Tregs express the transcription factor Forkhead box P3 (FoxP3), which is essential for Treg development and function. In dogs, FoxP3 is considered a specific marker for Tregs 5 and has been used in several studies to detect the presence of Tregs in canine cancer. 28,33,38,47,48
CCH are derived from dermal precursors of Langerhans cells. 19 Langerhans cells are epidermal dendritic cells that are involved in the cutaneous immune response. In normal skin, these cells are considered immature, with a high capability of antigen uptake and low antigen presentation. After encountering an antigen, Langerhans cells acquire a high antigen presentation ability and migrate to satellite lymph nodes. 19,37 In humans, Langerhans cells are characterized by the presence of intra-cytoplasmic Birbeck granules and the expression of langerin (CD207). In dogs, Birbeck granules are absent and Langerhans cells are langerin-negative. Therefore, there are no specific markers for canine Langerhans cells. 19 Iba-1 is an antigen expressed by most dendritic antigen-presenting cells and macrophage/monocyte lineage cells. Though not specific for canine Langerhans cells, anti-Iba-1 antibodies have the advantage of working on paraffin-embedded tissue. Thus, histologically, canine Langerhans cells can be recognized by cell morphology, anatomic location, and Iba-1 positivity. 24 Recently, in canine atopic dermatitis, but not in healthy skin, a population of CD206+ and MAC387− DCs, consistent with activated Langerhans cells or inflammatory epidermal DCs, has been described. 30 CD206 is a C-type mannose receptor 1 that has 2 main functions in humans: phagocytosis or endocytosis of mannose-coated particles, and receptor-mediated facilitated antigen presentation. 49 If neoplastic canine Langerhans cells acquire a mature phenotype during tumor progression, they could express CD206 in the regression phases of the tumor.
To the best of our knowledge, although CCH regression has been correlated with lymphocyte activation and MHC II tumor cell expression, there is no proof that tumor regression is directly induced by tumor cells without the participation and modulation of pro-tumoral and anti-tumoral immune cells. To contribute to the clarification of the role of the tumor microenvironment in CCH regression, the aim of this study was to investigate the expression of CD208 and FoxP3 as markers of dendritic cells and regulatory T lymphocytes, respectively, and tumor cell expression of CD206 as a marker of Langerhans cell activation, and relate these parameters to the different phases of CCH.
Material and Methods
Tissue Processing
Thirty-two tumors previously diagnosed as CCH were retrieved from the archive of the histopathology laboratory, VetAgro Sup, Lyon, and 1 block per case was selected. For each block, 4-µm sections were obtained: one was stained with hematoxylin and eosin (HE) and the others were used for immunohistochemistry.
Protein Extraction and Western Blot Analysis
For protein extraction, a mouse liver, used as a positive control, and a canine mesenteric lymph node were used. Briefly, 30 mg of tissue was minced using a scalpel and homogenized with 500 µl of RIPA buffer (ThermoFisher Scientific) with protease inhibitors, using TissueLyser II (Qiagen). Then, tubes were placed on ice in constant agitation for 2 hours. Afterwards, samples were centrifuged for 20 minutes at 12 000 rpm at 4 °C and the supernatant was stored at −80 °C for further analyses. Protein concentration was calculated using the Pierce BCA Protein Assay Kit (ThermoFisher Scientific). To evaluate CD206 antibody specificity, a Western blot analysis was performed as previously described. 34 Twenty-five micrograms of protein was denatured at 70 °C for 10 minutes. Proteins were resolved using NuPAGE 4-12% Bis-Tris gel (ThermoFisher Scientific) and transferred to a nitrocellulose membrane. Nonspecific binding sites were blocked for 90 minutes with 5% nonfat dry milk in TBS-T (TBS containing 0.05% Tween-20) at room temperature. The blot was incubated at 4 °C overnight with an antibody against CD206 (1:500, Abcam, #ab64693). Then, the membrane was incubated with anti-rabbit peroxidase conjugated secondary antibody (1:3000, ThermoFisher Scientific, #32260) for 1 hour at room temperature. Reactive bands were visualized with a chemiluminescent detection kit (SuperSignal West Pico Chemiluminescent Substrate, ThermoFisher Scientific) using the iBright instrument (ThermoFisher Scientific).
Immunohistochemistry
Immunohistochemistry was performed with the avidin-biotin-peroxidase complex method. Species specificity of Iba-1, CD3, CD208, and FoxP3 antibodies has been demonstrated in previous studies and by the manufacturers. 24,28,33,38 –40,47,48 CD206 species-specificity was validated by Western blot. The protocols for each antibody were validated as recommended by the literature (Suppl. Figs. S1, S2, S3, S4). 29
Anti-Iba-1, (ab5076, diluted 1:500, Abcam, Tokyo, Japan), which is a pan-marker for macrophages and DCs, but also labels Langerhans cells in CCH, 24 was applied to confirm the diagnosis of histiocytoma. CD208, CD3, FoxP3, and CD206 expression was evaluated using the following antibodies: anti-CD208 (CD208, clone 1010E1,01, diluted 1:200, Dendritics, Lyon, France), anti-CD3 epsilon (clone CD3-12, diluted 1:100, Bio Rad, Hercules, CA), anti-FoxP3 (clone FJK-16s, diluted 1:200, Ebioscience, San Diego, CA), and anti-CD206 (rabbit polyclonal anti-mannose receptor, diluted 1:200, Abcam). Antigen retrieval for CD208 and CD206 was performed at 95 °C for 40 minutes in a pH 9 solution (Dako target retrieval solution, pH 9, Dako, Carpinteria, CA) and in a citrate buffer solution at pH 4, respectively. For Iba-1, CD3, and FoxP3, antigen retrieval was performed by heating at 95 °C for 40 minutes in a pH 6 citrate buffer (ThermoScientific Dewax and HIER Buffer L, ThermoFisher, Runcorn, UK) followed by a 20-minute cooling. Labeling was amplified by the last step-product of the ultraTek HRP (anti-polyvalent) ready-to-use kit (ScyTek Laboratories, Logan, UT; 30 minutes at 20 °C) and revealed with the Vector NovaRED Peroxidase (HRP) Substrate kit (Vector, Burlingame, CA; 5 minutes). Hematoxylin counterstain (5 minutes) was applied; then sections were dehydrated and mounted. The negative control consisted of the replacement of the primary antibody with the antibody diluent solution (Emerald diluent antibody, Cellmarque, Sigma Aldrich, Rocklin, CA).
To better characterize CD208+ cells, double immunolabeling was performed on a CCH sample, using anti-CD208 and anti-CD204 antibodies. First and second primary antibodies, as well as substrates, are detailed in Supplementary Table S1. Species-specificity of the CD204 clone (SRA-E5) has been demonstrated in a previous article 14 and by several manufacturers, and the clone was used in multiple studies for detecting canine macrophages. 21,36,47,48 The antibody used in the present study was validated in our laboratory on different paraffin-embedded canine tissues, comprising skin and lymph nodes (Suppl. Figs. S5, S6). Slides were dewaxed and rehydrated. Antigen retrieval was performed by heating at 95 °C for 40 minutes in citrate buffer, pH 9, followed by 20-minute cooling. Blocking, secondary antibody, and amplification were performed with the ultraTek HRP (anti-polyvalent) ready-to-use kit (ScyTek Laboratories, Logan, UT; 30 minutes at 20 °C). The first reaction was revealed with the Vector NovaRED Peroxidase (HRP) Substrate kit (Vector; 5 minutes), while the second reaction was performed with Histogreen (Histoprime, Histogreen HRP-substrate kit, Linaris, Biologische Produkte, Germany). To remove the antibodies of the first reaction and avoid cross-reactions with the second primary antibody, after the first round of immunolabeling the slides were treated with LinBlock kit (Linaris, Biologische Produkte, Germany) in accordance with the manufacturer’s protocol. A supplemental treatment with an avidin-biotin blocker (Endogenous Avidin/Biotin Blocking system, Abcam) was added, before incubating the slides with the second primary antibody. A positive control for each antibody was obtained by omitting the second primary antibody, while the negative control was obtained by omitting both primary antibodies.
CCH Groups
Tumors were allocated into 4 groups, with each group representing a different phase of tumor development and regression, as described. 6 Briefly, group 1 was represented by tumors with scant peripheral lymphocytic infiltrate; group 2 was characterized by peripheral lymphocytic nodules; in group 3 lymphocytic nodules were within and at the periphery of the tumor; and group 4 was characterized by a diffuse lymphocytic infiltrate within the tumor.
Histological and Immunohistochemical Evaluation
On HE-stained slides, epidermal ulcers were recorded, and mitoses were counted in 2.37 mm2 areas. 18 Tumor cells were evaluated for positivity to Iba-1, to confirm the diagnosis of histiocytoma. For the evaluation of CD208, CD3, FoxP3, and CD206 immunolabeling, tumors were spatially divided into 3 zones: the deeper tier, generally in the subcutaneous tissue and/or dermis; the intermediate tier and the superficial tier, close to the epidermis. Positive cells were evaluated in the 3 zones as follows. CD208+ and FoxP3+ cells were counted in 3 high-power fields (0.237 mm2/field) in each zone. For intratumoral CD3+ T lymphocyte assessment, 3 images in the superficial tier and 3 images in the intermediate tier were taken using the Axiocam 506 Color camera (Zeiss) mounted on a Zeiss ImagerD2 microscope. In each image, corresponding to a surface area of 0.164 mm2, lymphocytes were counted using the Qupath open source software for digital image analysis. 3 The overall amount of positive cells was defined as the sum of positive cells in the superficial, mid and deep tiers, while the intratumoral amount of positive cells was defined as the sum of positive cells in the superficial and mid tiers. The distribution of tumor CD206+ cells was evaluated as follows: 0, lack of positive tumor cells; 1, positive tumor cells present only at the bottom of the tumor; 2, tumoral cells present at the bottom and in the center of the tumor; 3, positive tumoral cells present at the bottom, in the center, and near the epidermis.
Statistical Analyses
To evaluate the correlations between at least one ordinal variable, the Kendall test was applied (group, ulcer presence, CD206+ tumoral cell location). Correlations between numeric variables (number of CD208+, CD3+, and FoxP3+ cells and mitotic count) were evaluated with the Pearson test. The τ, for the Kendall test, and the ρ, for the Pearson test, give the intensity of the correlation: the closer the τ or the ρ is to −1 or +1, the stronger the correlation (inverse or direct correlation, respectively), while a τ or a ρ close to 0 indicates no correlation. A P value <.05 was considered to be significant. 7
Results
All of the tumors had morphological features of CCH, and all of the tumors except one were positive for Iba-1 labeling. The Iba-1-negative tumor was discarded from the evaluation of the immune infiltrate, even though it was morphologically consistent with a group 1 CCH. In tumor cells, Iba-1 labeling was membranous and occasionally cytoplasmic, and its intensity was variable. Near the epidermis, tumor cells were often Iba-1-negative or weakly positive, while deeper in the section, the immunolabeling intensity increased, reaching the highest levels at the periphery.
Based on the degree of lymphocytic infiltration on HE-stained slides, 6 8 (28%), 4 (13%), 12 (38%), and 7 (22%) samples were classified in groups 1, 2, 3, and 4, respectively. The amount of intratumoral CD3+ lymphocytes was positively correlated to the tumor groups (P= 9.7 × 10− 7 ; τ: 0.69; Fig. 1).
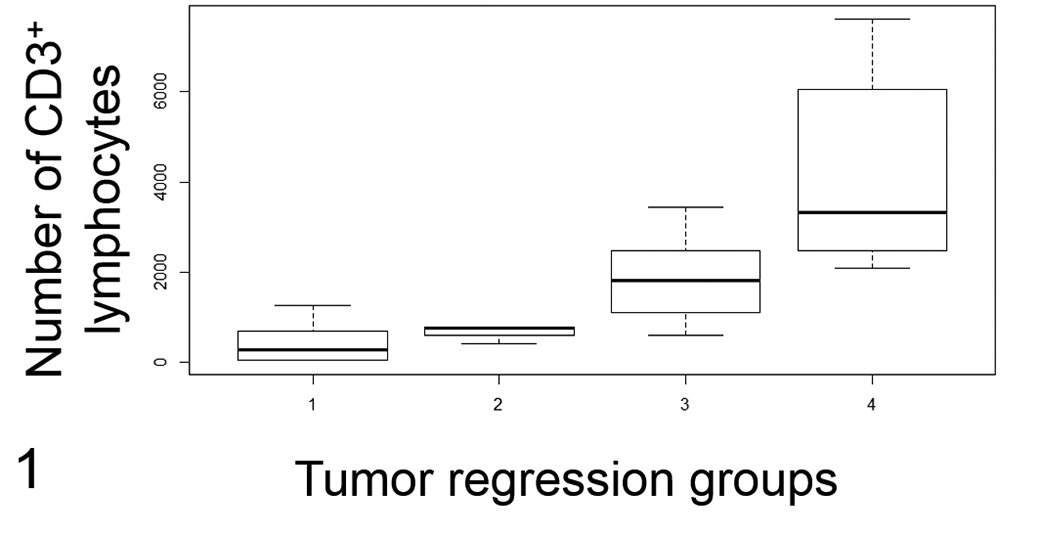
Intra-tumoral CD3+ lymphocyte infiltration (Fig. 1) and mitotic count (Fig. 2) in canine cutaneous histiocytomas grouped by 4 stages of tumor regression. Group 1, scant peripheral lymphocytic infiltrate; group 2, peripheral lymphocytic nodules; group 3, peripheral and intratumoral lymphocytic nodules; group 4, diffuse lymphocytic infiltrate. There are more intratumoral CD3+ lymphocytes in the more advanced regressing tumors (group 3 and group 4; Fig. 1; P = .04). The mitotic count is lower in more advanced regressing tumors (group 4) compared to the other tumors (groups 1, 2, and 3; Fig. 2; P = .01). The lines represent the median, the boxes enclose the 25th to the 75th percentiles, and the whiskers represent the range.
In groups 1 and 2, the mitotic count was increased compared to groups 3 and 4 (Fig. 2, Table 1). Group and mitotic count were inversely correlated (P = 0.014, τ: −0.35), as were intratumoral CD3 lymphocytic infiltration and mitotic count (P = 0.047; ρ: −0.36). No other correlations were found between mitotic count and the other parameters.
Comparison of Mitotic Count, Overall CD208+ Cells, and FoxP3+ Cells Among Canine Cutaneous Histiocytomas Grouped by 4 Stages of Tumor Regressiona.
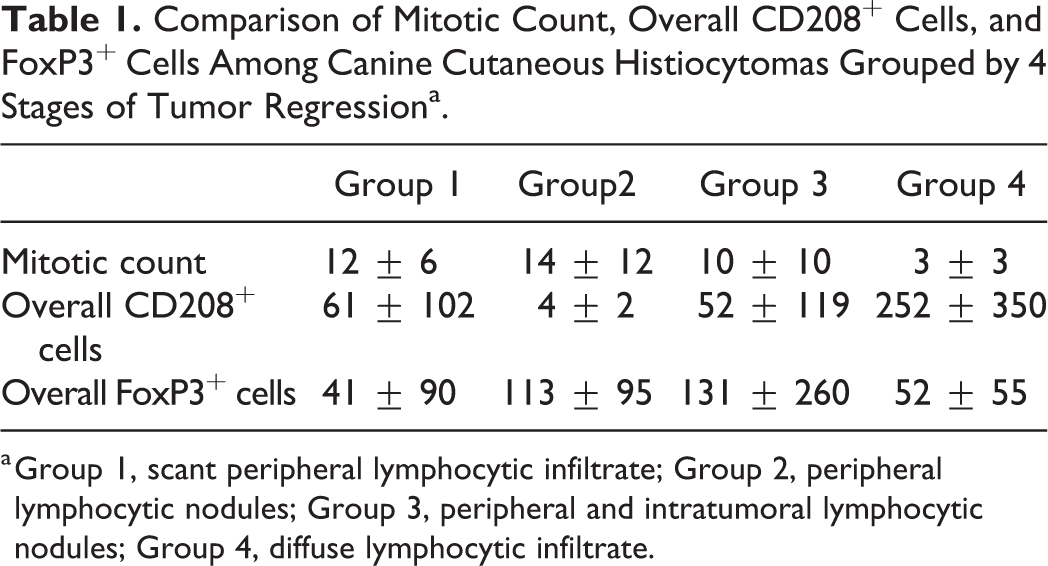
a Group 1, scant peripheral lymphocytic infiltrate; Group 2, peripheral lymphocytic nodules; Group 3, peripheral and intratumoral lymphocytic nodules; Group 4, diffuse lymphocytic infiltrate.
CD208 Labeling
The anti-CD208 antibody labelled stromal cells, but not tumor cells. Few stromal cells had cytoplasmic and finely granular labeling, with evidence of immunolabeled granules arranged in rows, consistent with dendrites (Fig. 3). The majority of CD208+ cells were round, approximately 20 to 30 µm in size, and had coarse, granular, cytoplasmic labeling (Fig. 4). These round cells were mainly located within the lymphoid follicles and around necrotic areas, although they were also observed within the solid parts of the tumors. In the sample double-labeled for CD208 and CD204, some cells expressed both markers and this was independent of the cell shape (Figs. 5, 6). These results suggested that CD208 could be a nonspecific marker for canine dendritic cells, also labeling some macrophages.
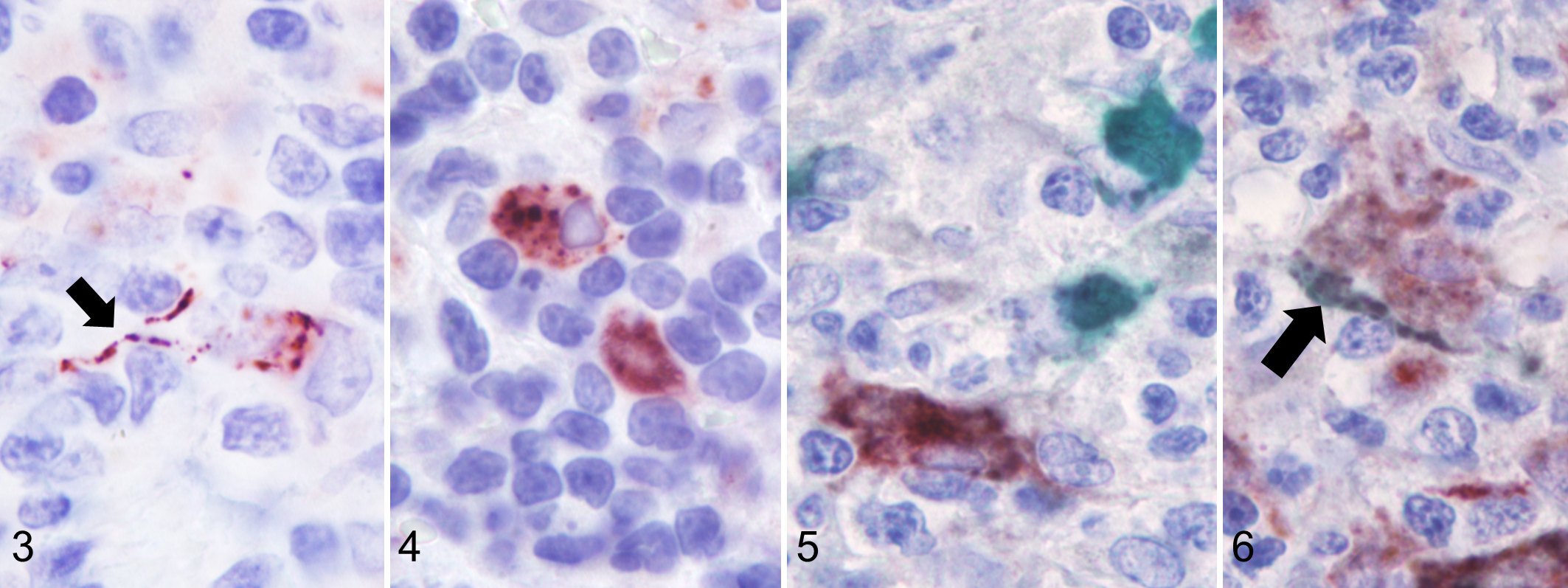
Canine cutaneous histiocytoma (CCH), skin, dog.
Eight of 31 cases were discarded from the CD208 evaluation because they did not have any CD208 labeling in the section, including in the healthy skin and, when present, in and around lymphoid follicles. These cases corresponded to old paraffin blocks and belonged to different regression phases (3, 2, 2, and 1 samples belonged to groups 1, 2, 3, and 4, respectively). In 4 cases, no CD208+ cells were detected within the tumor, although positive cells were present on the section. In the remaining 19 cases, the overall amount of CD208+ cells ranged from 1 to 907, and it was higher in group 4 (average of 251.8) compared to groups 1, 2, and 3 (average of 6, 3.5, and 51.9, respectively; Fig. 7, Table 1), but the difference was not statistically significant (Suppl. Fig. S7). The amounts of intratumoral (superficial and center tiers) CD208+ cells and intratumoral CD3+ T lymphocytes were positively correlated (P = .001; ρ: 0.6; Suppl. Fig. S8). Topographically, the number of CD208+ cells in the 3 tumor areas (deep, central, and superficial) decreased from the deep part of the tumor (where the lymphoid follicles were primarily located) to the epidermis. This distribution pattern was observed in all groups except in group 4, where increased CD208+ cells were found in the center of the tumor. No statistical correlations were found between the amount of CD208+ cells (overall, superficial, central and deep) and the other parameters.
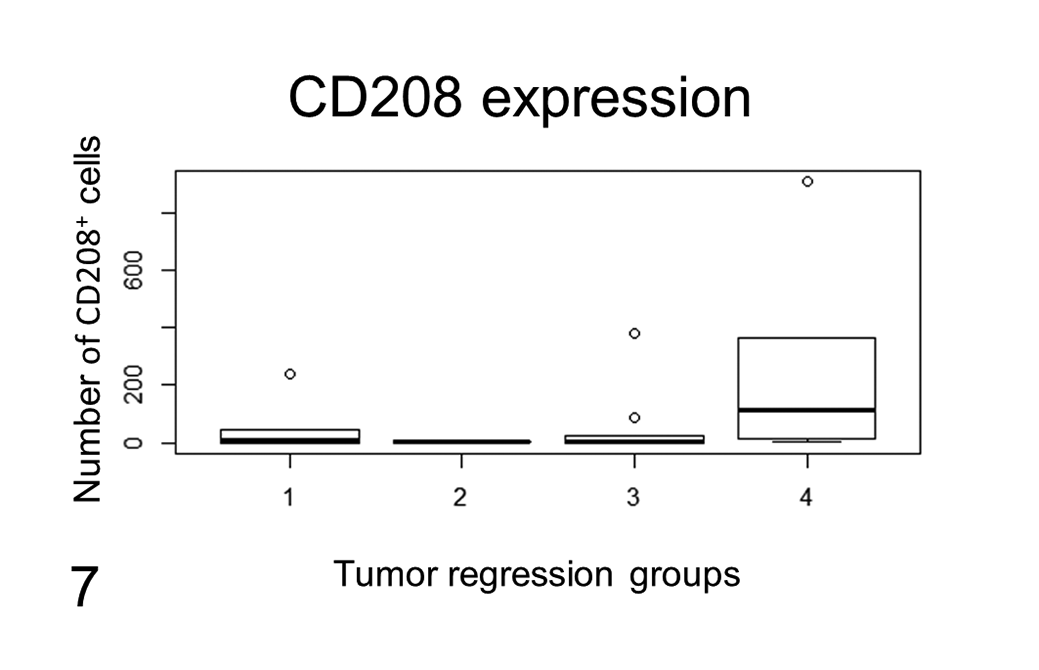
CD208 labeling related to tumor regression groups. CD208 labeling is reported as the overall number of positive cells per sample related to tumor regression group. The amount of CD208+ cells was increased in tumors in a more advanced regression phase (group 4) when compared to tumors in earlier regression phases (groups 1, 2, and 3). The difference was not statistically significant. The lines represent the median, while the boxes enclose the 25th to the 75th percentiles. The whiskers represent the range (visible only in group 4). The circles show the data outside the 25th to the 75th percentiles.
FoxP3+ Labeling
FoxP3+ cells were observed in 24 samples and were sparsely distributed within the tumors. The overall number of positive cells ranged from 2 to 911 with no evident distribution or group-related pattern. Statistically, no correlation was found between FoxP3+ cells and the other parameters.
CD206 Labeling
Western blot analysis performed on the protein extract from a canine lymph node and a mouse liver (positive control) recognized a 190 kDa protein that corresponded to the molecular weight of CD206 (Fig. 8). CD206+ labeling was present in 30 out of 31 samples. Immunolabeling observed in neoplastic cells was cytoplasmic and granular with a variable, low to moderate intensity (Figs. 9, 10). Strong cytoplasmic labeling was also observed in round to elongated cells, present primarily at the deep aspect as well as peripheral to the tumor, that were consistent with macrophages (Fig. 11). In 4 cases, tumor cells were completely negative. Labeling was observed only in the deep part of the tumor in 4 cases, in the deep part and in the center in 7 cases, or extending from the deep margin of the tumor to the epidermis in 15 samples. The topographic distribution of the labeling was strongly correlated to the tumor group, being absent or present only in the deep aspect in groups 1 and 2, and present in the deep aspect as well as in the center and near the epidermis in groups 3 and 4 (Fig. 12). The correlations between the spatial CD206 labeling and the tumor group as well as between the spatial CD206 labeling and the number of intratumoral CD3+ T lymphocytes were statistically significant (P = 6.81 × 10− 5 , τ: 0.63; P = .0002; τ: 0.54; Fig. 13).
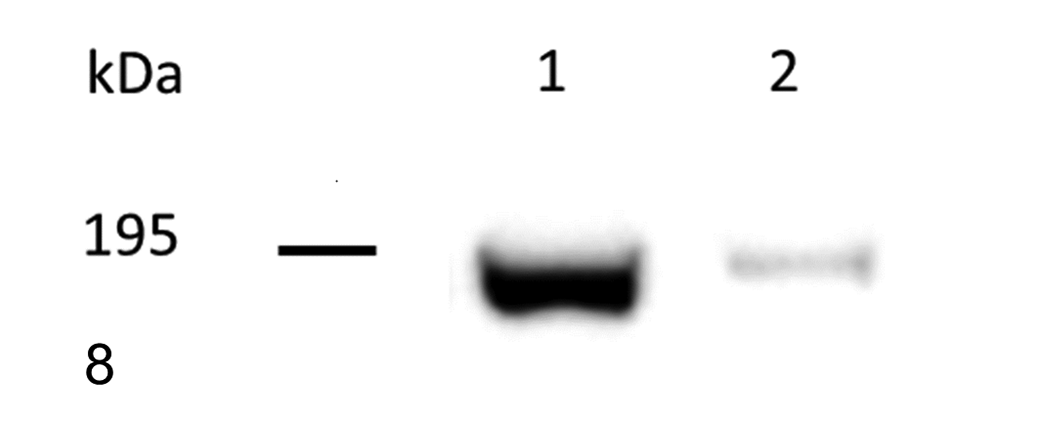
Western blot analysis of protein lysates from canine lymph node (sample 1) and mouse liver (sample 2). CD206 was detected as a 190 kDa protein.
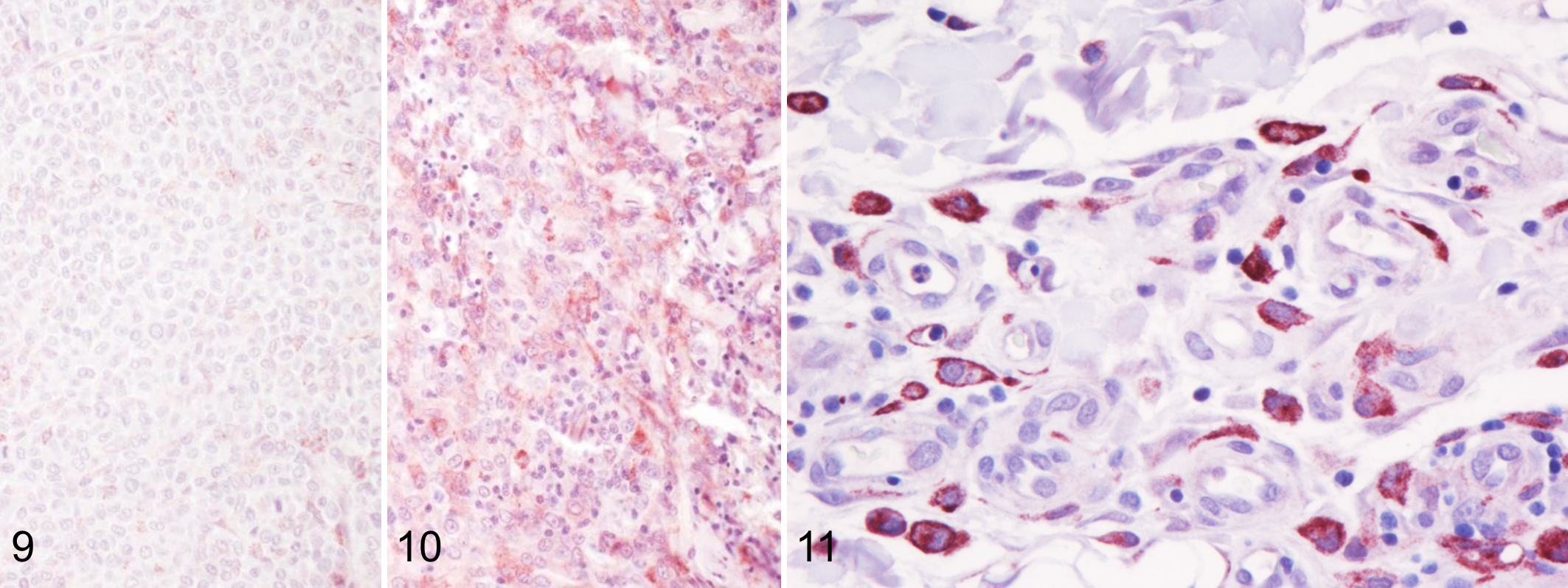
Canine cutaneous histiocytoma (CCH), skin, dog. The distribution of CD206 immunolabeling of neoplastic Langerhans cells varies within the tumor and is correlated to the regression phase of the tumor.
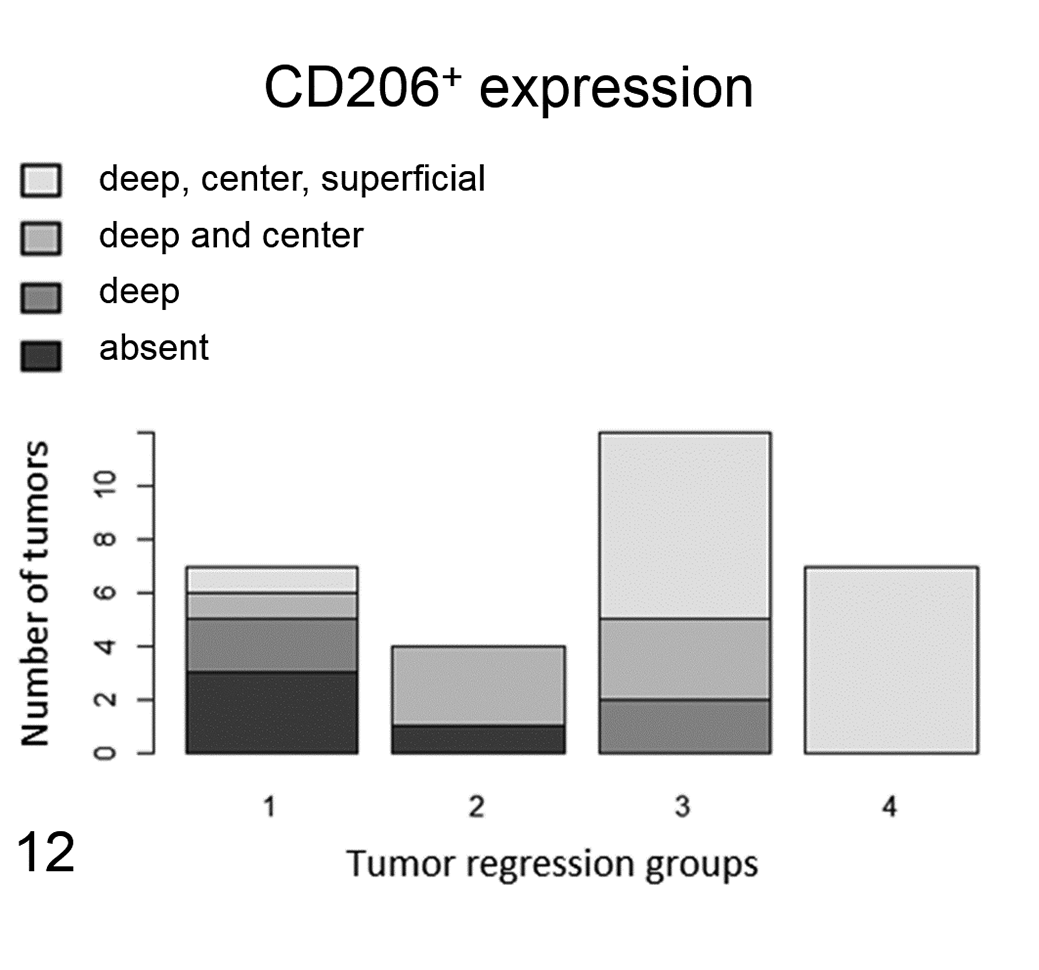
Location of CD206+ immunolabeling within tumors. CD206 expression by tumor cells varies by location within the tumor and by the tumor regression group. In the first phases of tumor regression (groups 1 and 2), the majority of neoplastic Langerhans cells are either negative or express CD206 only at the base of the tumor. In the more advanced phases of tumor regression (groups 3 and 4), neoplastic Langerhans cells also express CD206 in the center and superficial parts of the tumor. The difference was statistically significant. CD206 expression is localized in the 3 areas of the tumor as follows: black, absent (no CD206 expression); dark gray, only in the deep part of the tumor; gray, in the deep and center parts of the tumor; light gray, in the deep, center, and superficial parts of the tumor.
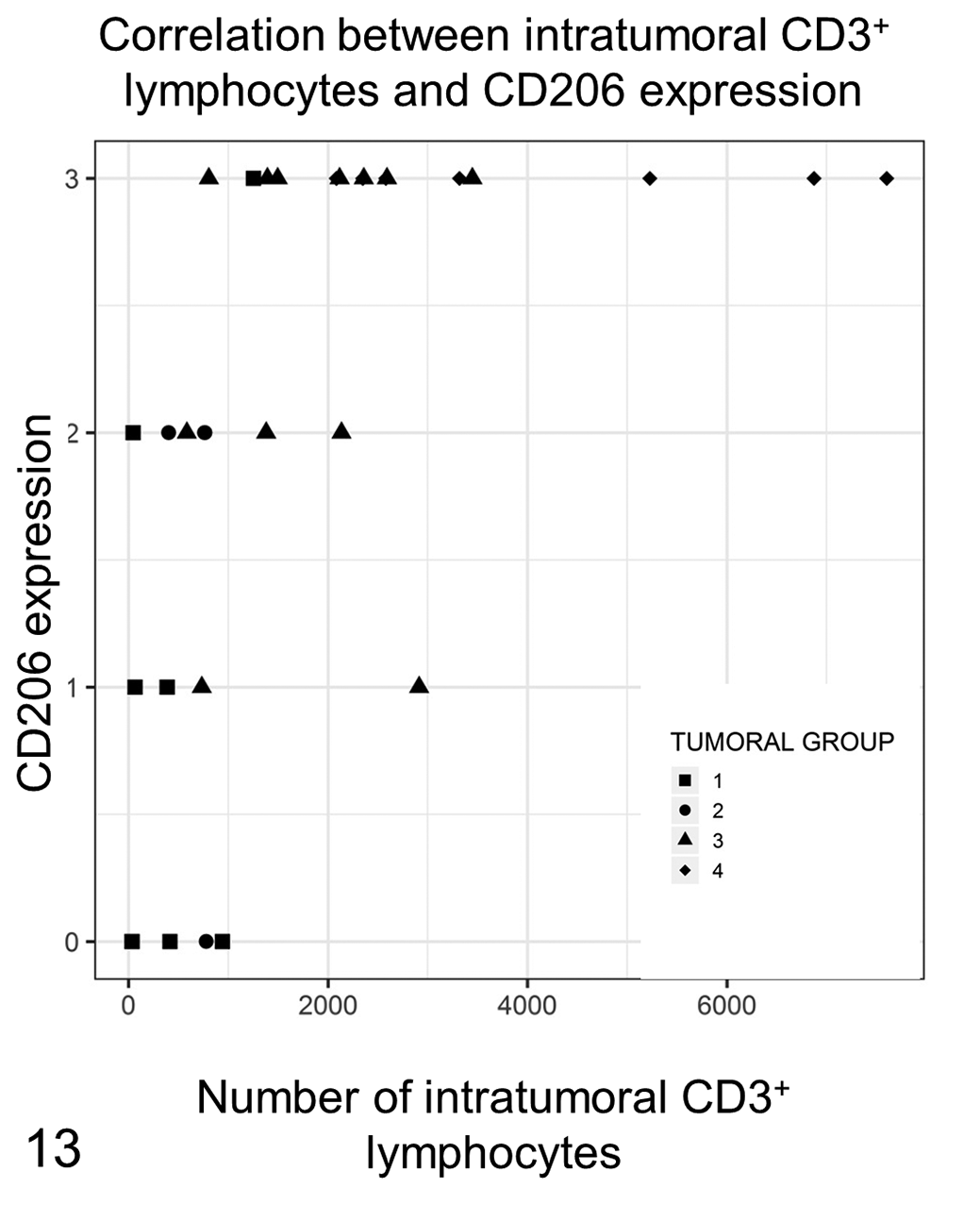
The amount of intratumoral CD3+ T lymphocytes is positively correlated with the pattern of CD206 expression in neoplastic Langerhans cells. In the CD206 pattern 0, consisting of no CD206+ tumor cell, few lymphocytes are detected, and the samples belong to the first phases of tumor development (groups 1 and 2). In CD206 pattern 2 (CD206 expression in tumor cells located at the bottom and in the center of the tumor), the amount of CD3+ T lymphocytes increases and is greatest in samples with CD206+ neoplastic Langerhans cells located in both the deep and superficial areas of the tumor. Most samples with CD206 pattern 3 have a high amount of CD3+ T lymphocytes and are in the more advanced tumor regression phases (groups 3 and 4).
Data about each sample are given in Supplementary Table S2.
Discussion
CCH is a singular tumor, characterized by spontaneous regression due to a specific host immune reaction without any known pathogens involved in tumor development and immune stimulation. Tumor regression is thought to be due to the immune response, but the stimulus for lymphocyte infiltration has not been determined. In the present study, the immune infiltrates in 32 CCH samples were evaluated and correlated with the different phases of tumor development and regression as previously defined on the basis of lymphocyte infiltration. 6 Similar to a previous study, 23 the mitotic count was higher in groups 1 and 2, corresponding to the developing and initial regression phases, than in groups 3 and 4, where the inflammatory infiltrate dramatically increased and tumor cell numbers decreased.
Notably, a prior study found no differences in tumor cell proliferation between different regression phases. 25 The discrepancy between our results and the previous study may be attributable to the different methods employed to evaluate cell proliferation (Ki67 proliferation index vs mitotic count). Mitotic count was performed by counting the number of mitotic figures on an HE-stained section, which is an easy and inexpensive solution because there is no need for immunohistochemistry. Mitotic count can be influenced by tumor cell density, which may vary among tumors. The proliferation index was calculated by counting the Ki-67 fraction of positive cells out of the total number of tumor cells. Regressing CCH (groups 3 and 4) have fewer tumor cells compared to groups 1 and 2 because of the massive lymphocytic infiltration and the presence of necrotic areas. Hence, in CCH, mitotic count could be helpful in defining the tumor group, but may not be an objective method to evaluate tumor cell proliferation.
Tumor cells were Iba-1 positive in all cases except one. The tumor that did not display Iba-1 labeling corresponded morphologically to a group 1 tumor without any lymphocytic infiltration visible on the HE-stained slide. Iba-1, also called allograft inflammatory factor-1, is a calcium binding protein expressed in microglial cells and cells from the monocyte/macrophage lineage. 9,22 This protein is involved in cellular activation, phagocytosis, and oxidative burst, 35,51 and its intensity can change depending on the cell status, being enhanced in activated cells. 9,35 It is speculated that the Iba-1 negative sample might correspond to an initial form of CCH, in which no tumor cells are mature and activated yet.
Tumor cells were positive for CD206. CD206, a C-type mannose receptor 1, has been described in canine dermal DCs in inflammatory conditions, but not in healthy skin, indicating that it is expressed in activated DCs. 30 In that report, authors could not distinguish whether CD206+ cells were Langerhans cells or intra-epidermal DCs. Our study confirms that neoplastic Langerhans cells express CD206. The spatial pattern of CD206 expression changed in a group-related manner. In groups 1 and 2, Iba-1 was expressed only in the deep/center parts of the tumor, while in groups 3 and 4, the entire tumor was labeled (deep, center, and adjacent to the epidermis). A spatial pattern of expression, related to the tumor group, was also previously observed for MHC II and E-cadherin. 15,26,27 Thus, our results regarding Langerhans cell CD206 expression support the hypothesis that Langerhans cells acquire a mature phenotype during tumor regression. This was also supported by the correlation between the location of CD206 expression and the intratumoral T lymphocyte infiltration.
CD208 is used as a specific marker for human DCs. 32 CD208 is a lysosome-associated membrane glycoprotein that is expressed in mature interdigitating DCs. 32 This glycoprotein has been described and used as a marker of DCs in other species, like camelids, cattle, chicken, dog, ferret, rainbow trout, and swine. 10,11,13,31,39 –41,44 Though generally considered specific for animal DCs, 2 authors reported the expression of this marker in cells other than DCs. In cattle, CD208 was described as being expressed in cells within B follicles in cells considered to be tingible-body macrophages. 31 In rainbow trout, CD208 was also expressed by macrophages. 11 In the present study, most CD208+ cells were round, large in size, and presented coarse intracytoplasmic labeling, consistent with intracytoplasmic vacuoles. When dual immunolabeling was performed, some CD208+ cells also expressed CD204. CD204 is a class A macrophage scavenger receptor that labels many different canine macrophage populations throughout the body. 14,42 To the best of our knowledge, CD204 expression has never been described in canine DCs and it is considered specific for canine macrophages. Thus, from the location, morphology, and immune phenotype of CD208+ cells, it seems that CD208 is not a specific marker for canine DCs but can also be expressed by some macrophages.
CD208+ cells were observed in many tumors. The amount of these cells was increased in group 4 CCH, and the amount of intratumoral CD208+ cells was correlated to the density of intratumoral T lymphocytes. The elevated presence of CD208+ cells in some regressive tumors could be due to the presence of a high number of macrophages in regressing tumors, as previously reported in literature, 12 and of DCs, stimulated by the areas of necrosis. Nevertheless, due to the lack of specificity of CD208 for canine DCs, the results need to be confirmed with more specific markers.
FoxP3+ cells were observed in most of the analyzed CCH. The number of Tregs was highly variable, but in some samples it was notably high. Tregs are correlated with a poor prognosis in canine cancers. 5,28 Considering that CCH is a benign tumor, our hypothesis was that Treg numbers were either low in all tumor groups, indicating a lack of Treg immunosuppressive role in tumor development, or were inversely correlated to the tumor groups. The latter finding may suggest that in the early phases, tumor development could be facilitated by an immunosuppressive microenvironment.
Thus, the observed presence and density of Tregs in our study were unexpected. In human oncology, the presence and role of Tregs in cancer has not been fully elucidated, and their density could be associated with a poorer prognosis. 8 Nevertheless, in colorectal cancer, lymphoma, and head and neck squamous cell carcinoma, Treg density is associated with a better prognosis. 2,43,50 Several hypotheses have been proposed to explain these results. In intense inflammatory responses, such as in cancer and chronic inflammation, there is an increased need for immune response modulation to bring the inflammation under control, and Tregs are recruited to perform this function. 8,46 Thus, in tumors with a good prognosis, such as CCH, Tregs could be present only to carry out their role of immune modulation, without having a strong impact on tumor cells. Another explanation is that the immunosuppressive role of Tregs may not be dependent only on their overall number but also on other factors, such as stimulation by an infectious agent, as observed in human colorectal cancer, 46 or anatomic compartmentalization, as described in human stomach cancer. 46 We evaluated the location of FoxP3+ cells within the tumor as well as correlations between Treg number and ulceration, but no statistically significant results were found. We recorded FoxP3 cells in CCH, but their presence was not linked to CCH progression, and their role in CCH remains to be elucidated.
In conclusion, we observed that CCH is characterized by an increase in CD206 expression by tumor cells in a spatial-group-related pattern, supporting the hypothesis that neoplastic Langerhans cells acquire a mature phenotype with tumor regression. Although the functional role of the Tregs and DCs in CCH regression remains unknown, we observed the presence of an unexpected amount of Tregs in CCH, whose role needs to be explored in further studies. Finally, according to our results, the anti-CD208 antibody seems to be a nonspecific marker of canine DCs.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Belluco_et_al - FOXP3, CD208, and CD206 Expression in Canine Cutaneous Histiocytoma
Supplemental Material, Combined_supplemental_materials-Belluco_et_al for FOXP3, CD208, and CD206 Expression in Canine Cutaneous Histiocytoma by Sara Belluco, Alessandro Sammarco, Pierrick Sapin, Thibaut Lurier and Thierry Marchal in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
