Abstract
Cutaneous mast cell tumor (MCT) is one of the most frequent cutaneous neoplasms of dogs and may vary from well-differentiated to aggressive tumors with metastasis. The authors retrospectively described the gross and histologic aspects of metastatic MCT in 49 dogs. Primary MCT was most commonly identified in the inguinal region (14/35; 40%), and at necropsy multiple, cutaneous nodules were frequently reported (23/49; 47%). All primary MCT were classified as high-grade neoplasms, and metastases involved the lymph nodes (47/49; 96%), spleen (33/49; 67%), liver (29/49; 59%), bone marrow (20/49; 41%), kidneys (16/49; 33%), and heart (14/49; 29%), while the lungs were less commonly affected (9/49; 18%). The main gross findings included lymphadenomegaly in 47 cases; splenomegaly in 28 cases, with splenic nodules in 13 dogs; hepatomegaly in 28 cases, with white pinpoint foci in 9 cases; nodules on the capsular surface of the kidneys in 9 dogs; and epicardial nodules in 6 cases. Histologically, the lymph nodes were largely obliterated by neoplastic mast cells, while in the spleen, neoplastic cells were multifocally scattered (16/33; 48%), arranged in nodules (10/33; 30%), or obliterated the parenchyma (9/33; 27%). In the liver, the neoplastic cells mainly infiltrated the sinusoids (24/29; 83%), but were also arranged in random nodules (10/29; 34%). Interstitial and nodular metastases were observed in the kidneys and the heart. Grossly unapparent metastases were common in the heart (6/14; 43%), kidneys (4/16; 25%), and lungs (6/9). KIT III and KIT II staining patterns were observed in 29 and 20 cases, respectively.
Cutaneous mast cell tumor (MCT) is one of the most frequently diagnosed cutaneous neoplasm in dogs, accounting for 16% to 21% of all skin tumors diagnosed in this species.2,9,16 MCT has a heterogeneous biological behavior, ranging from solitary, well-differentiated tumors to aggressive tumors with metastatic disease. Given its broad spectrum of clinical presentation, prognostic factors are often used in the diagnosis and classification of MCT,2,9,16 including histologic grading. There is a 3-tier grading system, with grades I, II, and III (well-differentiated to poorly differentiated tumors, respectively), 21 and a more recent 2-tier system that classifies MCTs as low or high grade. 11 Other prognostic indicators include immunohistochemistry (IHC) for KIT protein, proliferation markers, such as mitotic count (MC), Ki67, and AgNOR, and molecular detection of c-kit mutation via polymerase chain reaction.2,12,31 Nonetheless, no factor is entirely and solely predictive of clinical outcome, and therefore clinical features, such as history of tumor recurrence, staging, breed, lymph node status, and anatomic site, must also be considered. 5
Metastatic MCT may carry a poor prognosis,14,18,23 especially for high-grade MCTs, which are more prone to metastasize than low-grade tumors.11,26 More than 80% of grade III MCTs will evolve to metastatic disease, while grade I MCTs rarely metastasize.2,26 MCT metastases are reported to frequently involve the regional lymph nodes (LNs) and less often the liver, spleen, and bone marrow.2,23,26,30 Few investigations have described the pathological features of metastases detected during necropsy, 9 and most studies focused on the clinical23,30 or imaging aspects of metastatic disease,3,22,27 or solely on nodal metastasis.14,33 To our knowledge, there is only one study that investigated metastatic disease associated with MCT using necropsy data, 6 but that study lacks further investigation of the gross and histologic features of the metastases. The current work aimed to describe the pathological features of MCTs with metastases detected in dogs submitted to necropsy in a 12-year period and to define the main metastatic sites, as well as the gross and histologic features of each metastasis.
Materials and Methods
Case Selection and Analysis
A retrospective study of the necropsy database (book records and digital search) from the Veterinary Pathology Laboratory was conducted to search for cases of cutaneous MCTs with metastases in necropsied dogs from January 2008 to March 2020. Information regarding age, breed, sex, and clinical history was retrieved from the necropsy reports, and cases were grouped according to age range, sex, and breed. Cases in which a MCT was diagnosed via biopsy with complete surgical excision or limb amputation and had no cutaneous tumor detected during necropsy were also included in this study when histologic evaluation of necropsy samples revealed metastasis from the previously excised cutaneous tumor. In addition, when a diagnosis of MCT was established via biopsy or cytology, the report was retrieved, and overall survival time (OST) was defined as the time from diagnosis to death. Exclusion criteria included MCTs located at mucocutaneous junctions and oral MCTs.
Gross Evaluation
A complete necropsy was performed in all cases and all organs were grossly evaluated to detect lesions. The descriptions of the gross lesions were obtained from the necropsy reports and from available photographs taken at necropsy. When the cutaneous site of the primary tumor was known, cases were grouped according to location. Moreover, the distribution of the cutaneous neoplasm at necropsy was evaluated and characterized as a single nodule, multiple nodules, or focally extensive nodules (with diffuse enlargement of the area around the neoplasm). MCT-related progressive disease, aside from metastasis, was defined as recurrence at the primary site and/or development of MCT at a different cutaneous site. All cases of recurrence were confirmed histologically at necropsy.
The grossly identified metastases were assessed to determine the main features in each site. Metastases were classified as diffuse enlargement; pale discoloration; multifocal, white, pinpoint foci; and/or focal to multifocal nodules in the affected organs. LNs were also evaluated for loss of corticomedullary architecture. Metastasis to muscle was only diagnosed if the affected muscular tissue was distant from the cutaneous neoplasm.
The presence or absence of gastrointestinal (GI) ulcers was assessed during gross necropsy and later confirmed by histology. The location and distribution of the ulcers were investigated and divided as affecting the stomach and/or small intestine.
Histologic Evaluation and Grading
Routine samples were collected at each necropsy, including samples of the cutaneous tumor(s) when present, LNs when these were enlarged and/or were the regional LNs draining the tumor, and skeletal muscle, when gross lesions or possible metastatic nodules were observed. Samples of spleen, liver, kidneys, bone marrow, lungs, heart, adrenal glands, thyroid, pancreas, stomach, small and large intestines, and urinary bladder were also collected. Cerebrum, cerebellum, and brainstem were routinely collected, while spinal cord was only assessed when the clinical history and gross findings indicated possible lesions. Tissues were fixed in 10% formalin, routinely processed for histology, and stained with hematoxylin and eosin. The necropsy slides were reviewed by 3 pathologists (PRR, MVB, SPP) to confirm the diagnosis of MCT and provide a detailed histologic description of the tumor, and all the tissues were histologically reevaluated to search for microscopic metastases. Cases with MCTs that originated in the subcutaneous tissue and, occasionally, infiltrated the superficial and deep dermis were excluded from subsequent analysis at this point. Pre-necropsy biopsy slides were reviewed in cases in which the MCTs were surgically removed or were absent at necropsy (i.e., limb amputation). The primary tumors were classified according to the Patnaik et al 21 and Kiupel et al 11 grading systems using the necropsy samples or the pre-necropsy biopsy slides when the cutaneous MCT was absent at necropsy. The Patnaik et al 21 grading system was applied, and tumors were classified based on cellular morphology, tumor cellularity and mitotic count, as well as extent of tissue involvement and stromal reaction. Tumors were classified as high-grade according to the Kiupel et al 11 grading system by identification of any of the following criteria: at least 7 mitoses in 10 high-power fields (HPF) (2.37 mm²); at least 3 multinucleated cells in 10 HPF; at least 3 bizarre nuclei in 10 HPF; or karyomegaly in at least 10% of the neoplastic cells. In cases with multiple cutaneous MCTs at necropsy, the available histologic slides were used for the histologic grading; even though in such cases each tumor should be graded, 9 this could not be performed in all cases due to the retrospective nature of this study.
The histologic features of the metastases, such as the distribution pattern of the neoplastic mast cells, were also assessed in all metastatic sites. The organs were considered metastatic sites when the neoplastic mast cells were arranged in extensive (diffuse) cords or sheets effacing the parenchyma, as multifocal scattered clusters, or in nodules. Metastases to the LNs were histologically classified according to the HN0-HN3 classification system. 33
Immunohistochemistry (IHC)
IHC for KIT protein was performed in all cases using sections of formalin-fixed paraffin-embedded MCTs. The sections were boiled in citrate buffer (pH 6.0) for 40 minutes at 96°C and incubated with polyclonal anti-KIT antibody (code A04502, Dako) at a 1:200 dilution. The amplification signal was obtained with the peroxidase-labeled antibody method (MACH4, Universal HRP polymer, Biocare Medical, Pacheco, CA, USA), and the reactions were revealed with 3-amino-9-ethylcarbazole chromogen kit (AEC, Biocare Medical). A confirmed case of MCT was used as positive control, and negative control consisted of incubating the tissue sections with irrelevant polyclonal antibody, in addition to a case of cutaneous lymphoma.
Cases were classified according to the KIT-immunolabeling pattern as pattern I (predominantly membrane-associated staining), pattern II (focal to stippled cytoplasmic staining), and pattern III (diffuse cytoplasmic staining), as previously described. 12
Statement of Ethics
This study was conducted according to relevant institutional guidelines for retrospective studies involving necropsy data and embedded paraffin tissue samples, and it was approved by the Research Committee under the number 37897.
The data analyzed in this study are available upon request to the author.
Results
Case Evaluation
Forty-nine cases of canine cutaneous MCT with metastases to other organs were identified in the evaluated period. Affected dogs ranged in age from 2 to 16 years (median age = 10 years; mean age = 10.3 years), and the frequency was slightly higher in males (27/49; 55%). A wide range of breeds were reported, mostly mixed-breed dogs (28/49), followed by Boxer (3/49), Labrador Retriever (3/49), German Shepherd (3/49), Pit Bull Terrier (3/49), Cocker Spaniel (2/49), Bulldog, Dachshund, Belgian Malinois, Pekingese, Shar-Pei, Brazilian Mastiff, and Tosa (1 case each). Further detailed information of each case is available in the Supplemental Table 1.
Assessment of MCT progressive disease, aside from metastasis, showed that 20/49 dogs (41%) developed new MCT(s) at a different cutaneous site after excision (15/20) and/or had local recurrence of the excised tumor (10/20). In another 20/49 cases (41%), the dogs were not considered to have either local recurrence or MCT at a different cutaneous site according to the provided clinical history. In the remaining 9/49 cases (18%), the presence or absence of local recurrence and/or MCT at a different cutaneous site could not be determined due to an absence of relevant clinical history.
A previous diagnosis of MCT was available in 25/49 cases (51%), either via incisional or excisional biopsy or cytology. In these cases, OST ranged from 5 to 1324 days (mean OST = 150.6 days; median OST = 69 days) (Supplemental Fig. S1). Chemotherapy was employed in 12 cases, in which the chemotherapeutic drugs were known in 4 cases and included vinblastine, lomustine, and prednisone.
The primary cutaneous location of the MCT was determined in 35/49 cases (71%). The most common location was the inguinal region (14/35; 40%) (Fig. 1), which included the scrotum and prepuce in 9/14 dogs, followed by the limbs in 11/35 dogs (31%), similarly affecting the pelvic (6/11) and thoracic limbs (5/11). Other locations included the thorax in 7/35 cases (20%), and the head and neck in 3/35 dogs (9%). The remaining 14/49 cases (29%) had either multiple nodules or lacked adequate clinical history or a previous diagnosis, which prevented the identification of the primary location. Nonetheless, in all of these 14 cases, no primary subcutaneous, visceral, or mucosal MCT was detected.
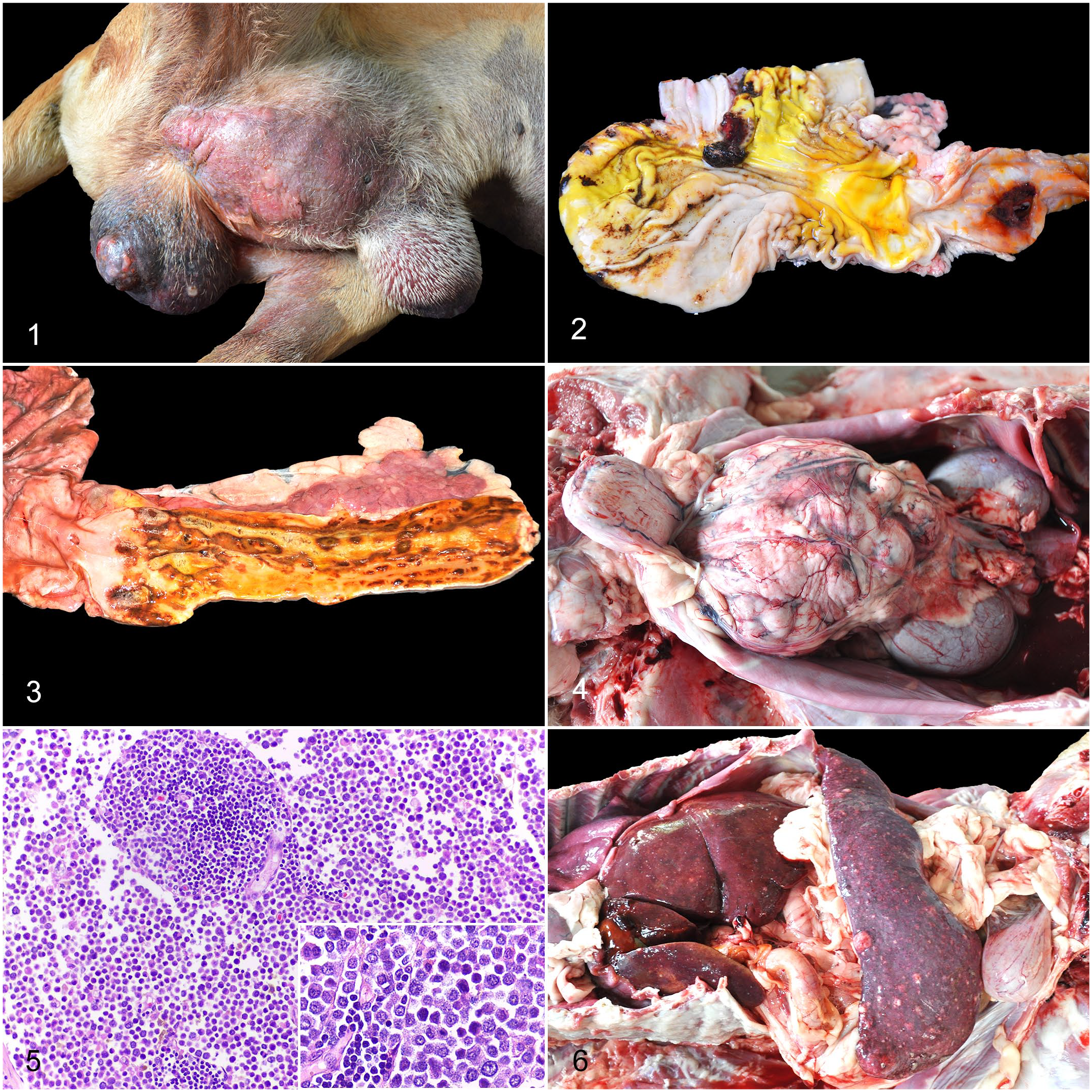
Metastatic cutaneous mast cell tumor (MCT), dog.
The skin distribution of the MCTs was assessed during necropsy, and the most common presentation was multiple cutaneous nodules in 23/49 cases (47%), followed by focally extensive nodules with diffuse enlargement of the region surrounding the neoplasm in 13/49 dogs (26.5%), and single nodules in the remaining 13/49 cases (26.5%).
Gastrointestinal ulcers, considered a paraneoplastic condition, were identified in 16/49 dogs (33%), with perforation in 3 cases (Fig. 2) and focal (1/3) or diffuse peritonitis (2/3). The stomach was affected in all 16 cases, and the duodenum in 11/16 dogs (69%) (Fig. 3). Regarding the distribution of the ulcers, the pylorus along with the duodenum (gastroduodenal region) was the most commonly affected region (11/16; 69%), with ulcers in the fundic and cardiac regions in 2/11 of these cases. The pylorus or antral-pyloric region was affected in 5/16 dogs (31%).
Histologic analysis of the MCTs revealed that, according to the Patnaik et al 21 grading system, 35/49 tumors (71%) were grade III and 14/49 cases (29%) were grade II MCTs. According to the Kiupel et al 11 grading system, all tumors were high-grade MCTs. A MC higher than 7 in an area of 2.37 mm² was observed in all cases, varying from 7 to 150 (mean MC = 41.8; median MC = 30), and it was the only histological criterion of high-grade MCTs in 23 cases.
Metastases (Gross and Histologic Analysis)
Metastasis to the LN was observed in 47/49 cases (96%). Lymphadenomegaly was noted in all of these dogs, and the nodal parenchyma had loss of corticomedullary architecture in 32/47 cases (68%) (Fig. 4). Histologically, nodal metastasis was characterized by extensive replacement of the parenchyma by neoplastic mast cells in 43/47 cases (91%; HN3) (Fig. 5). Partial replacement and/or neoplastic mast cells effacing the medullary sinuses were detected in 4/47 dogs (9%; HN2). Although, in most cases, the metastatic lesions had similar morphology to the primary cutaneous tumor, the neoplastic mast cells were more pleomorphic and had increased mitotic count in the metastatic sites when compared with the cutaneous tumor in 4 cases of grade II MCT.
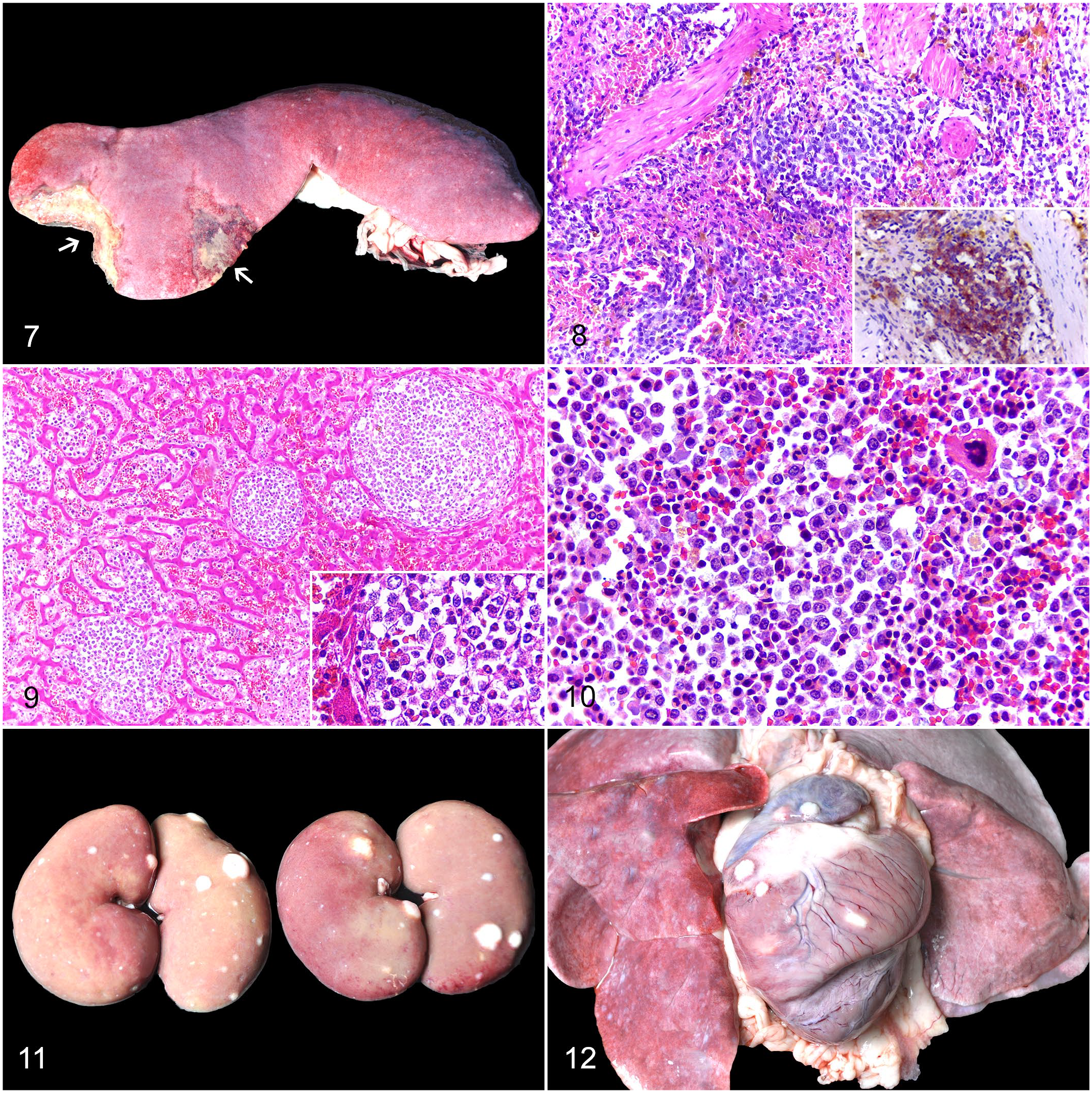
MCT metastasis to the spleen was detected in 33/49 cases (67%). Diffuse enlargement of the splenic parenchyma (splenomegaly) was noted in 28/33 cases (85%), but no gross abnormalities were observed in the 5 remaining cases. In addition, multiple (12/13) or single (1/13), white to red, capsular nodules that extended into the parenchyma were noted in 13/33 dogs (39%) (Fig. 6). Uncommon lesions included randomly distributed, white, pinpoint foci on the capsule in 4/33 cases (12%) and splenic infarcts in 3/33 cases (9%) (Fig. 7). Histologic evaluation revealed scattered, large clusters of neoplastic cells in the red pulp and along the edges of the trabeculae in 16/33 dogs (48%) (Fig. 8), nodular areas in 10/33 cases (30%), and diffuse distribution of neoplastic cells with extensive replacement of the parenchyma in 9/33 dogs (27%), with 2 cases having mixed histologic patterns.
Metastatic cutaneous mast cell tumor (MCT), dog.
Hepatic metastasis was reported in 29/49 dogs (59%). Diffuse enlargement of the hepatic parenchyma (hepatomegaly) was noted in 28/29 cases (97%). Multifocal, white, pinpoint foci, up to 0.5 cm in diameter, were observed in 9/29 dogs (31%) (Fig. 6), which were associated with a few nodules up to 1.0 cm in diameter in 2 of these cases. Less common lesions included multifocal, white to yellow nodules up to 6 cm in diameter in 2/29 cases (7%). Histologically, neoplastic mast cells were within the sinusoids in 24/29 cases (83%) and arranged in nodules in 10/29 cases (34%), wherein both patterns occurred concomitantly in 8 dogs (Fig. 9). In addition, these cells were also observed expanding the portal tracts in 5/29 dogs (17%).
The bone marrow was affected in 20/49 cases (41%). Histologically, there were moderate to large numbers of neoplastic mast cells multifocally scattered or arranged in cords within the marrow in 17/20 cases (85%) (Fig. 10). However, the neoplastic mast cells were arranged in large, cohesive groups that were visible at low magnification in 3/20 cases (15%).
MCT metastasis to the kidneys was detected in 16/49 dogs (33%). Randomly distributed, single or multiple, capsular and parenchymal, white nodules were noted in 9/16 cases (56%), and involved one (6/9) or both kidneys (3/9) (Fig. 11). Less common changes included white, pinpoint areas on the capsular surface in 3/16 dogs (19%), and gross lesions were absent in 4/16 dogs (25%). Renal metastases were histologically composed of nodular areas within the parenchyma in 10/16 cases (63%), which involved both cortical and medullary regions, and of neoplastic cells occupying the renal interstitium in the remaining 6/16 cases (37%).
Cardiac MCT metastasis was observed in 14/49 cases (29%), of which 6/14 (43%) had only histologic changes. Grossly, the metastases were characterized by single or multiple, white nodules in the epicardium in 6/14 dogs (43%) (Fig. 12), of which diffuse, pale discoloration was also present in 1 case. In 1/14 cases (7%), the heart had multifocal to coalescent, white areas in the myocardium, and in the remaining case (7%), the myocardium was diffusely and mildly pale. Histologically, neoplastic mast cells were arranged in nodules in the myocardium, which extended into the epicardium in 7/14 dogs (50%), and dissected through the myocardium in the remaining 7/14 cases (50%).
MCT metastasis to the lungs was detected in 9/49 cases (18%), but grossly visible changes were only observed in 3/9 cases. Gross lesions were characterized by focal to multifocal, white nodules extending into the pulmonary parenchyma. Histologically, in 6/9 cases, the neoplastic cells were within the blood vessels and mildly thickened the alveolar septa via expansion of the vascular lumina. Colonization of the parenchyma was observed only in 3/9 cases, with nodular areas in 2 cases and invasion of alveoli by neoplastic cells in the other dog.
Other organs with metastasis included the adrenal glands in 4/49 cases (8%), which was mainly diagnosed by the histologic identification of neoplastic mast cells within the parenchyma, although one of these cases had a grossly apparent, unilateral, white nodule. Metastasis to muscle was observed in 4/49 dogs (8%), and it was characterized by single to multiple, white nodules effacing the muscle fibers. In 3 dogs, metastasis affected the skeletal muscles ventral to the vertebral body, and 2 involved the intercostal muscles (Supplemental Fig. S2). The neoplasm also affected the thoracic vertebrae in 2 of these cases and the lumbar vertebrae in the other dog, with nodular lesions and bone lysis (Supplemental Figs. S3 and S4). In 2 cases, the mass extended into the spinal canal; histologically, there were occasional axonal spheroids in both cases and myelomalacia with accumulation of gitter cells in 1 case. There were metastases to the GI tract in 5/49 cases (10%), which affected the esophagus and the ileum in 2 cases each, and the soft palate in 1 case. Nodules were grossly noted in both cases of esophageal metastasis, which primarily affected the submucosa in 1 case (Supplemental Fig. S5) and the serosa in the other. Intestinal metastasis was characterized by a focal nodule in the serosa in 1 dog, and was confirmed by histologic identification of neoplastic mast cells expanding the lamina propria of the intestinal mucosa in the other. The metastatic lesion in the soft palate was characterized by a nodule expanding the submucosa. There were metastases to the prostate gland in 2/27 cases (7%), which were only detected histologically and were characterized by small clusters of pleomorphic neoplastic mast cells expanding the prostatic interstitium. The urinary bladder was affected in 2/49 dogs (4%), in which transmural hemorrhage was observed in 1 case. Histologically, both cases had neoplastic mast cells interspersed in the muscular layer.
Immunohistochemistry
All MCTs had positive immunolabeling for KIT protein. KIT-staining pattern III was observed in 29/49 cases (59%), and KIT-staining pattern II was observed in the remaining 20/49 cases (41%).
Discussion
MCTs have a wide biological behavior and may occur as aggressive neoplasms with potential to metastasize.9,16 The histologic grade is one of the most important and commonly used prognostic factors for MCTs,2,10 of which grade III and high-grade MCTs are associated with a higher metastatic potential and MCT-related mortality.5,11,14,21,26,30 In accordance with this, all the metastatic MCTs in the present study were classified as high-grade tumors. Proliferation markers also have prognostic value,2,9 and high MC has been associated with MCT metastasis and higher lethality rates. 5 Although a recent study did not find an effect of MC on the development of LN metastasis, dogs with MCTs presenting a MC higher than 12 had a reduced median survival time, 18 which is in accordance with our work, in which dogs had a median MC of 30 mitoses and a short median OST (69 days). This finding, in addition to the fact that a MC higher than 7 in an area of 2.37 mm² was the only histologic criterion of high-grade MCT present in all of our cases, reinforces the relevance of MC in detecting MCTs with more aggressive behavior and the impact of increasing MC on survival time, as previously described. 18 MC may be as helpful as histologic grading for prognostic assessment, acting as an independent negative prognostic factor.18,24 A MC greater than 5 in 10 HPF has been associated with a poor clinical outcome 8 and increased potential for metastatic disease. 24
The most common anatomic locations of the primary MCTs in the current investigation were the inguinal region and the limbs, comprising half of the cases. Although previous studies have not established an association between tumor location and clinical outcome,5,18,23 MCTs in the inguinal area have been regarded as more aggressive tumors.2,9 A worse prognosis may be explained in these cases by the difficulty of adequate surgical excision in this region, 2 as locoregional control improves the outcome in grade III 8 and metastatic low-grade MCT. 1 Moreover, in the current study, MCTs in the inguinal region frequently had diffuse enlargement of the affected area with local invasion, which is similar to that observed in another study. 6 Multiple cutaneous nodules were the most common gross feature observed at necropsy in this study, comprising almost half of the cases. Although solitary nodules are the most common clinical presentation of MCTs,9,10 and the prognostic value of multiple nodules is considered uncertain,13,19,30 there was a higher percentage of multiple nodules in our study (47%) compared with the prevalence of 10% to 21% in other investigations.2,19 Chemotherapy was employed in only 12 cases in the current investigation, in which 9/12 had histories of either local recurrence and/or MCT(s) at distant cutaneous site. While systemic treatment may have a more favorable outcome than surgery alone in dogs with grade III MCTs, 32 more studies are needed to evaluate therapy efficiency in delaying progression of existing metastatic disease. 2
The LN was the most common metastatic site of MCTs in this investigation, as previously described.6,23,26,30 This finding emphasizes the relevance of LN evaluation during clinical staging of MCTs, as this is most likely the first organ to be affected by metastatic disease. MCTs involving the inguinal region and pelvic limbs in our study often metastasized to the medial iliac and hypogastric LNs, which is a pattern that has also been described in dogs with MCTs of the caudal portion of the body.6,20 This feature reinforces the relevance of assessing this chain of LNs during clinical staging of MCTs, especially those in the caudal portion of the body. According to the classification system of nodal metastasis, all cases in this study were classified as either HN2 or HN3, which are considered to have a worse outcome regarding disease-free interval and survival time when compared with HN0/1. 33 In the current work, most cases (43 dogs) were classified as overt metastasis (HN3), and many of these (39/43 cases) had concomitant distant metastases, which may explain the advanced progression of the nodal metastases.
Distant metastases of MCT are less common than nodal metastases, carry a worse prognosis, 1 and are more difficult to detect clinically, 9 with reported frequencies ranging from 4.1% to 19.2%.8,26,27,30 The most common distant metastatic sites for canine MCTs are the spleen, liver, bone marrow, and non-regional LNs,2,6,17,23,26,30 similar to that observed in the current study. Furthermore, renal and cardiac metastases were also detected in our investigation, which have been rarely reported for MCTs.6,20 Distant metastases are usually preceded by local LN metastasis,6,30 which is in accordance with our findings, since 96% of the cases with distant metastases also had nodal involvement.
Splenic metastasis was frequently observed (67%), as previously described by other authors in necropsied dogs (75%–79%),6,20 and it may be as high as 86.7% in dogs with stage IV MCT. 23 Splenic nodules, a common gross finding in this investigation, may represent nodular hyperplasia, abscesses, granulomas, or neoplasms. 29 As old dogs frequently have hyperplastic nodules,6,29 and this age category is overrepresented in this study, the differential diagnosis should be considered, and histologic confirmation is essential.
The liver was the third most common metastatic site, as previously observed in necropsied dogs with a similar incidence (62%); 6 however, a study of dogs with stage IV MCTs reported a higher frequency of 84.4% assessed by fine-needle aspiration. 23 Hepatomegaly was the most common gross finding in our investigation, even though it was not a constant lesion in another study of necropsied dogs; 6 nevertheless, subjective increase in liver size has been described during ultrasound investigations of MCT metastases.25,27 Nodular formation was uncommon in the affected livers, and this might explain why nodules are not frequently noted on ultrasound of dogs with hepatic metastasis of MCT.22,27
Bone marrow MCT involvement is considered rare and carries a poor prognosis.17,23 The detection of metastatic mast cells in the marrow through cytologic evaluation ranges from 4.5% to 20%;4,23 however, this percentage increases when the evaluation is histologically performed in samples from necropsied dogs (48%–58%),6,20 similar to the current investigation. Metastasis to the bone marrow is usually accompanied by multi-organ involvement,17,23 as in our study, in which all dogs with bone marrow involvement had metastases to other organs. Our results reinforce the hypothesis that bone marrow involvement may indicate multi-organ involvement and advanced disease with poor prognosis.
Although detection of MCT metastasis to the kidney via ultrasound has been reported occasionally (2.2–8.3%),23,25 the kidneys were the fifth most common metastatic site in this investigation, with 16/49 cases (33%), similar to that described in other necropsied dogs.6,20 Although in our study pinpoint, white areas were only occasionally seen (3/16; 19%), this was the only gross renal lesion described in another investigation. 6 Metastasis should not be ruled out in cases of subtle gross lesions, such as pale discoloration, or in the absence of gross renal lesions; histologic evaluation is essential to establish a definitive diagnosis.
Cardiac metastasis of MCT is infrequently described (8.3%–21%), and the reports comprise only cases diagnosed during necropsy.6,20 Although epicardial nodules were more frequently observed, some cases may not have gross lesions, 6 as seen in 43% of our cases, indicating that micrometastasis may occur. In the authors’ opinion, the lack of clinical studies with documented MCT metastasis to the heart may be explained by the frequent absence of gross cardiac lesions or only subtle changes in the heart color.
Differing from other neoplasms, such as hemangiosarcoma, osteosarcoma, histiocytic sarcoma, and carcinoma, 15 the lung is not a common metastatic site of MCT.15,16,17,20,23,30 Previous reports have indicated low frequencies of pulmonary involvement,17,20,23 similar to our findings, or even absence of detection. 30 Pulmonary metastasis of MCT is mostly present in advanced MCT disease with multi-organ involvement, which was the main pathological presentation in the current investigation. Moreover, in the present work, gross lesions were rare, which reinforces the fact that metastatic nodules in the lungs are not frequently observed, 30 in addition to emphasizing the relevance of histologic confirmation, as demonstrated by other authors. 17
Although involvement of the GI tract has been described, the incidence in the current investigation was low, and other reports of necropsied dogs with MCT did not find evidence of metastases to the GI tract;6,20 consequently, metastatic spread to these sites should be considered particularly uncommon. We did not consider these cases visceral MCTs because, in all cases, there was a diagnosis of cutaneous MCT with metastases to other organs, which suggests the MCTs of the GI tract were part of multi-organ involvement. 9
GI ulcers may occur as a paraneoplastic condition with MCTs and may be especially prevalent in recurrent and metastatic MCTs,7,28 as the ones of the present research, which reinforces the recommendation to search for these lesions during necropsy. 28 In the current investigation, the frequency of GI ulcers (33%) was similar to that observed in necropsied dogs with MCT of another study, 20 but it may be up to 83.3%. 7 The cause of GI ulcers should be interpreted with caution, as these studies investigated populations of dogs affected by MCT at necropsy, suggesting that advanced disease and long-term treatment could also contribute to the pathogenesis of these ulcers.
The etiology of MCT is unknown, but stem cell factor, KIT protein, and c-kit have been reported to play a major role in the oncogenesis of these tumors. 9 In the current study, all MCTs exhibited KIT II and KIT III IHC staining pattern. Similarly, other studies have revealed that increased KIT cytoplasmic staining is associated with poor prognosis 12 and increased risk of metastatic disease and MCT-related death. 5
MCT is one of the most common cutaneous tumors in dogs. The wide variation in biological behavior presents a challenge in evaluating the prognosis, since aggressive metastatic disease may occur, as observed in this study. The most common metastatic sites of MCT included the LN, spleen, liver, and bone marrow, although the kidneys and the heart were also affected. Pulmonary metastasis was uncommon, and generally lacked gross lesions. The most common gross changes included lymphadenomegaly, splenomegaly with white to red, capsular nodules, and hepatomegaly with white, pinpoint foci. The confirmation of MCT metastases through histologic evaluation is essential, especially for some organs that may only have histologic lesions, such as the lungs, the heart, and the kidneys.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221114468 – Supplemental material for Pathological aspects of cutaneous mast cell tumors with metastases in 49 dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858221114468 for Pathological aspects of cutaneous mast cell tumors with metastases in 49 dogs by Paula Reis Ribeiro, Matheus Viezzer Bianchi, Marcele Bettim Bandinelli, Rafael Biondo Rosa, Joanna Vargas Zillig Echenique, Alanna Serpa Stolf, David Driemeier, Luciana Sonne and Saulo Petinatti Pavarini in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858221114468 – Supplemental material for Pathological aspects of cutaneous mast cell tumors with metastases in 49 dogs
Supplemental material, sj-xlsx-1-vet-10.1177_03009858221114468 for Pathological aspects of cutaneous mast cell tumors with metastases in 49 dogs by Paula Reis Ribeiro, Matheus Viezzer Bianchi, Marcele Bettim Bandinelli, Rafael Biondo Rosa, Joanna Vargas Zillig Echenique, Alanna Serpa Stolf, David Driemeier, Luciana Sonne and Saulo Petinatti Pavarini in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and to the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for financing this study.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
