Abstract
Failure of the cartilage canal blood supply to epiphyseal growth cartilage has been implicated in the pathogenesis of articular osteochondrosis in horses and other animal species. In a previous study of the developmental pattern of the blood supply in the tarsus of foals, early lesions of osteochondrosis were consistently found in regions where the cartilage canal vessels traversed the chondro-osseous junction. The developmental pattern of blood vessels has also been described in the distal femoral epiphysis; however, the group of foals examined in that study did not have lesions of osteochondrosis in this location. Therefore, the relationship between the occurrence of early lesions of osteochondrosis and the developmental pattern of the blood supply to epiphyseal growth cartilage in this site in foals has not been examined. Distal femora were collected from 30 fetuses and foals (up to 11 months old) submitted for postmortem examination. Sections from the lateral trochlear ridge and medial femoral condyle of both hind limbs were examined histologically. Sixteen cartilage lesions were found in 7 of the 30 fetuses and foals. All lesions contained evidence of cartilage canal necrosis and ischemic chondronecrosis. The lesions were located in regions where cartilage canal vessels traversed the chondro-osseous junction, as previously observed in the tarsus. The location and morphology of lesions indicated that a subclinical stage of ischemic chondronecrosis existed that preceded and predisposed to the development of osteochondrosis dissecans and subchondral bone cysts.
Osteochondrosis (OC) is a developmental orthopaedic disease that has been reported in human patients and several animal species.4,18,37 In all animal species in which it has been described, OC is defined as a focal disturbance in the process of enchondral ossification.4,18,37 Enchondral ossification takes place primarily at the metaphyseal growth plate, or physis, and the subarticular epiphyseal growth cartilage. The physis is responsible for longitudinal bone growth, whereas the epiphyseal growth cartilage is responsible for shaping the proximal and distal articular ends of long bones. The integrity of the articular cartilage can become disrupted superficial to lesions of OC in the epiphyseal growth cartilage, leading to the formation of hinged flaps or loose fragments within the adjacent joint cavity. These may consist of cartilage alone or cartilage and bone, and this stage of the disease process is known as osteochondrosis dissecans (OCD).418,33,37 The current report focuses on OC of the epiphyseal growth cartilage (articular OC).
In human patients, OC is considered to originate in either growth cartilage or subchondral bone.29,30 In animal species, however, in which there is an opportunity to study preclinical disease, OC is considered to arise solely within the growth cartilage.4,18,37 Proposed causes of such primary disease of growth cartilage in horses include cartilage ischemia,1,25 aberrant accessory centers of ossification, 6 dyschondroplasia8,28 and changes to the composition of the extracellular matrix.8,13–15
The hypothesis that OC arises secondary to cartilage ischemia was proposed in pigs in 1982. 11 In subsequent studies in horses, Pool 25 observed blood vessels that coursed from subchondral bone into what later became known as the cartilage canals of the epiphyseal growth cartilage. The vessels traversed the junction between subchondral bone and growth cartilage, and Pool hypothesized that shear forces could cause the vessels to fail at the osteochondral junction, resulting in ischemic necrosis of the chondrocytes they supplied. 25 This suggestion was later corroborated by observation of necrotic cartilage canal vessels and areas of ischemic chondronecrosis at predilection sites for OC in different equine joints. 1 Some of these areas of chondronecrosis were associated with a focal disturbance in enchondral ossification. 1
Further studies done to better characterize the pathogenesis of OC in horses determined that the cartilage canal blood supply was present only during the early phase of enchondral ossification in this species.1,23 The developmental pattern of the blood supply was studied in relation to early, subclinical lesions of OC in the tarsus of predisposed foals. 23 During growth, cartilage canal vessels became incorporated in the advancing ossification front and, in some cases, formed anastomoses with vessels of the subchondral bone. 23 Early lesions of ischemic chondronecrosis were consistently found in the region where the process of incorporation into the ossification front and the formation of vascular anastomoses occurred. 23 The process of incorporation therefore appeared to render the vessels particularly vulnerable to failure, 23 which was identical to observations previously made in piglets. 39
The developmental pattern of the blood supply to the epiphyseal growth cartilage of the distal femur was described in a group of Standardbred foals; 21 however, early lesions of OC were not identified in this site. Thus, the relationship between the developmental pattern of the blood supply and early lesions of OC could not be examined. 21 The aim of the current study is to describe early lesions of articular OC in the distal femur of foals and to correlate these findings with previously published data on the cartilage canal blood supply in this region.
Materials and Methods
Animals
Thirty individual horses, comprising 4 aborted equine fetuses and 26 foals, were selected from those submitted for postmortem examination at the Swedish University of Agricultural Sciences between 1994 and 1996 (n = 23) and the Norwegian School of Veterinary Science between 2003 and 2008 (n = 7).
Foals of breeds with a high prevalence of OC were selected in preference of those of low prevalence. Foals of both sexes were collected, and there was no minimum age for inclusion. The age of fetuses was recorded as gestational days, estimated on the basis of information from the owner and measured crown–rump length. 24 The youngest fetus was 210 gestational days, and the oldest foal, 336 days (11 months).
Postmortem Examination
The reason for euthanasia was noted and evidence of systemic or infectious disease recorded. The femoropatellar and femorotibial joints were examined for macroscopically visible lesions. The synovial fluid of joints with macroscopic changes was collected for bacteriologic examination.
Collection Protocol
Each femur (ie, left and right) was dissected free of soft tissues, and the distal femoral epiphysis was sawed into transverse slabs (5 mm thick) from the articular surface up to and including the proximal margin of the trochlear ridges, with the plane of section perpendicular to the weight-bearing surface of the distal femur. The total number of slabs ranged from 7 to 8, depending on the size of the individual femur.
The slab located midway between the most distal and the most proximal, as well as any slabs containing macroscopically visible changes (defined as a surface defect or focal irregularity at the cartilage–bone interface), were selected from each femur for histologic examination.
The selected slabs were fixed in 4% phosphate buffered formaldehyde for 48 hours and decalcified in 10% formic acid. After decalcification, each lateral trochlear ridge, medial femoral condyle, and any region with macroscopically visible changes (focal failure of enchondral ossification identified by thickened epiphyseal cartilage) were separated from the selected slabs and trimmed to fit standard embedding cassettes measuring 32 × 25 × 5 mm. The trimmed samples were processed and embedded before being cut into sections (6 μm thick) and stained with hematoxylin and eosin for histologic examination. A subset of the trimmed samples, including all those with macroscopic lesions, were serial sectioned and stained with toluidine blue as well as hematoxylin and eosin.
Histologic Examination
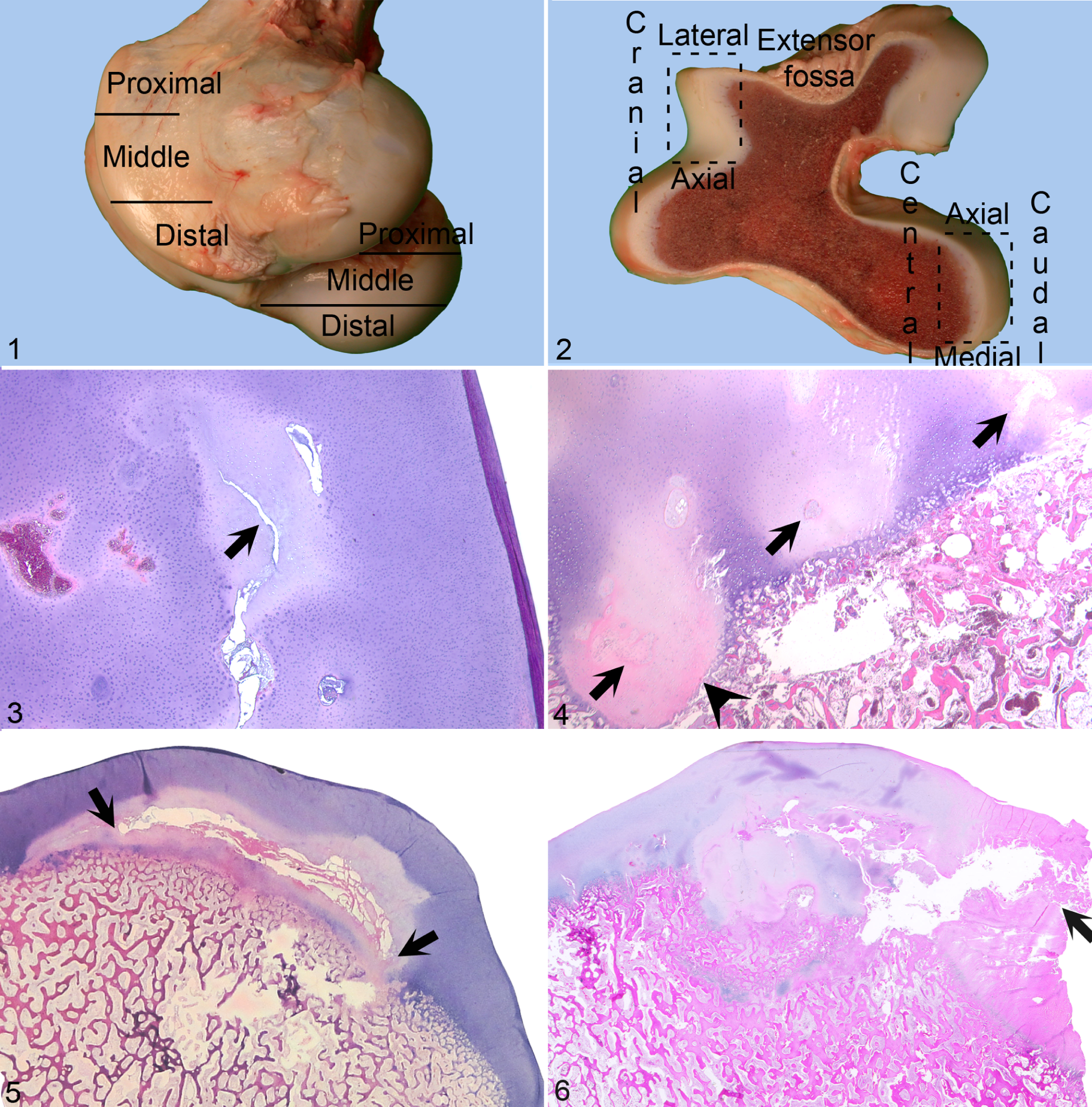
Histologic sections were evaluated with respect to the same parameters as previously observed. 20 The location of lesions was described by subdividing the trochlear ridges and medial femoral condyle into proximal, middle, and distal portions (Fig. 1). In slabs, the cranial margin of each trochlear ridge was subdivided into lateral, cranial, and axial aspects (Fig. 2). The medial femoral condyle was subdivided into axial, central, medial, and caudal aspects (Fig. 2).
Retrospective Acquisition of Clinical Data
Clinical records were available for 6 of the 7 foals with lesions. In 1 foal (case No. 6), the only clinical information available was that provided on the postmortem request form. The available records were reviewed for any history of systemic disease, infectious disease, or disease specifically involving the femoropatellar and femorotibial joints. Reported joint effusion and lameness during clinical examination were noted.
The lesion was subclinical in 1 foal, and there had been no indication for in vivo radiography (case No. 2). The remaining 6 foals with lesions had been examined radiographically in vivo, and the radiographs were available for review for 5 of these animals (case Nos. 1, 3, 4, 5, and 7). The location, size, and character of radiographic changes were recorded. In 1 foal with a clinically evident lesion, the radiographic diagnosis was available only as provided on the postmortem request form (case No. 6).
Results
The results of macroscopic and histologic postmortem examination of the distal femora were considered normal in 23 of the 30 horses; lesions were found in the remaining 7 (23.3%), which were assigned case Nos. 1 through 7. The order of numbering was determined first by location, with lesions of the trochlear ridges preceding lesions of the medial femoral condyle, and thereafter by ascending order of lesion severity. Table 1 summarizes the clinical findings of case Nos. 1–7.
Clinical Summary of Foals with Lesions a
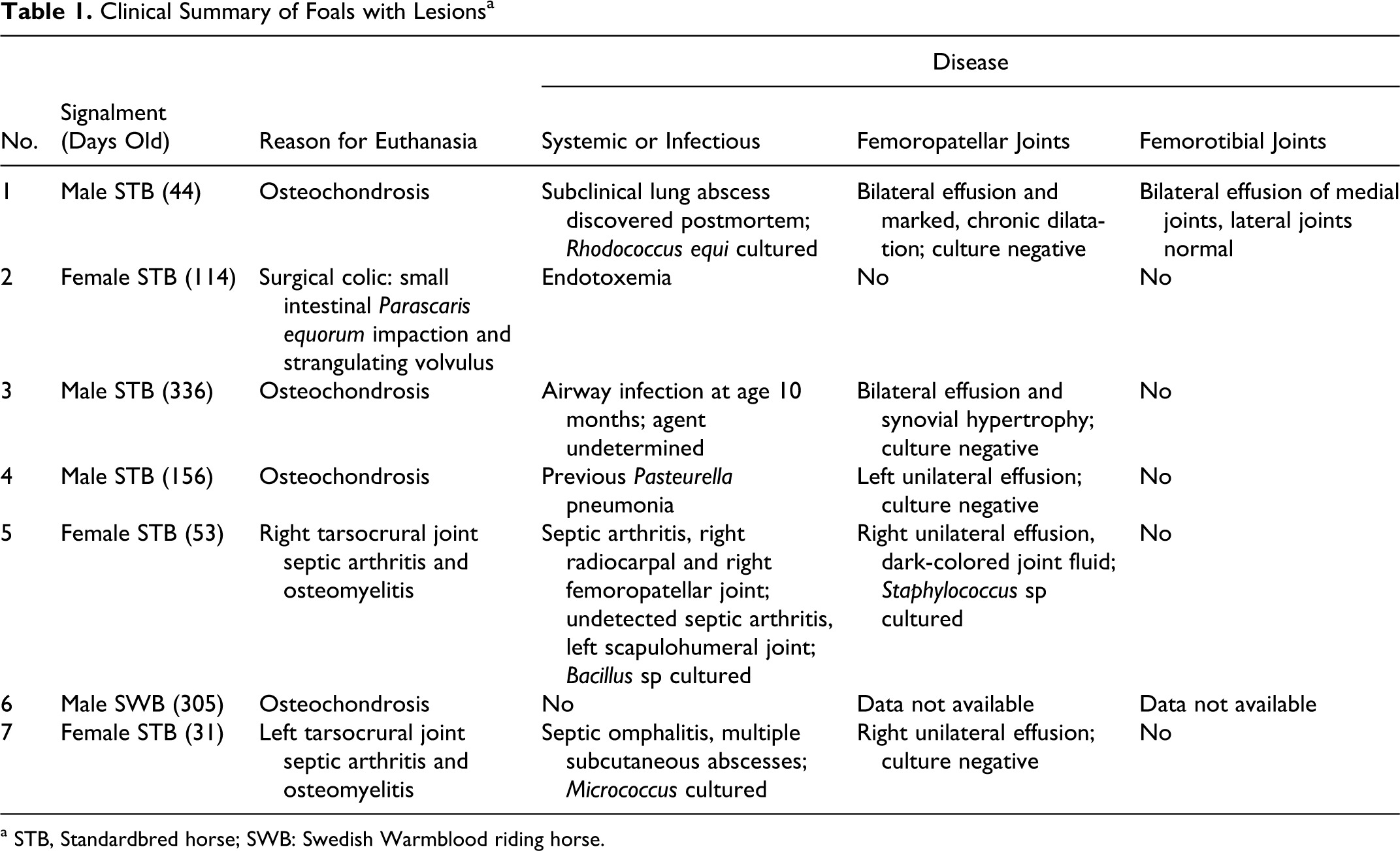
a STB, Standardbred horse; SWB: Swedish Warmblood riding horse.
A total of 16 distinct lesions were observed in case Nos. 1–7, equivalent to 13.3% of sampled sites (16 of 120). Case Nos. 1 and 3 were bilaterally affected and together had 9 of the 16 lesions (56.25%). Table 2 summarizes the location of lesions, histopathologic diagnoses, radiographic changes, and clinical observations of case Nos. 1–7; Table 3 summarizes the pathologic findings.
Histopathologic Diagnoses, Radiographic Changes, and Clinical Signs a
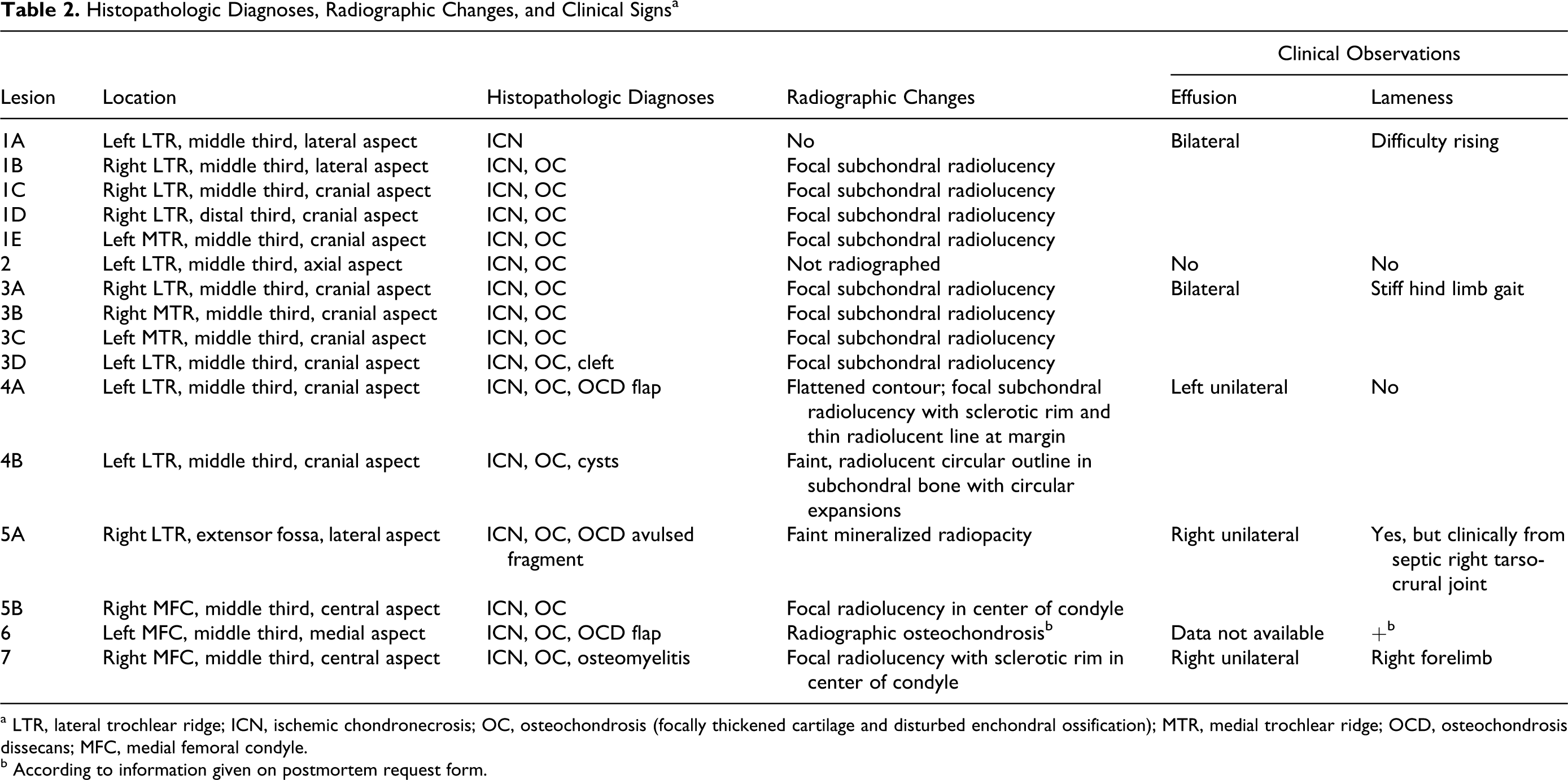
a LTR, lateral trochlear ridge; ICN, ischemic chondronecrosis; OC, osteochondrosis (focally thickened cartilage and disturbed enchondral ossification); MTR, medial trochlear ridge; OCD, osteochondrosis dissecans; MFC, medial femoral condyle.
b According to information given on postmortem request form.
Osteochondrosis Lesion Characteristics a
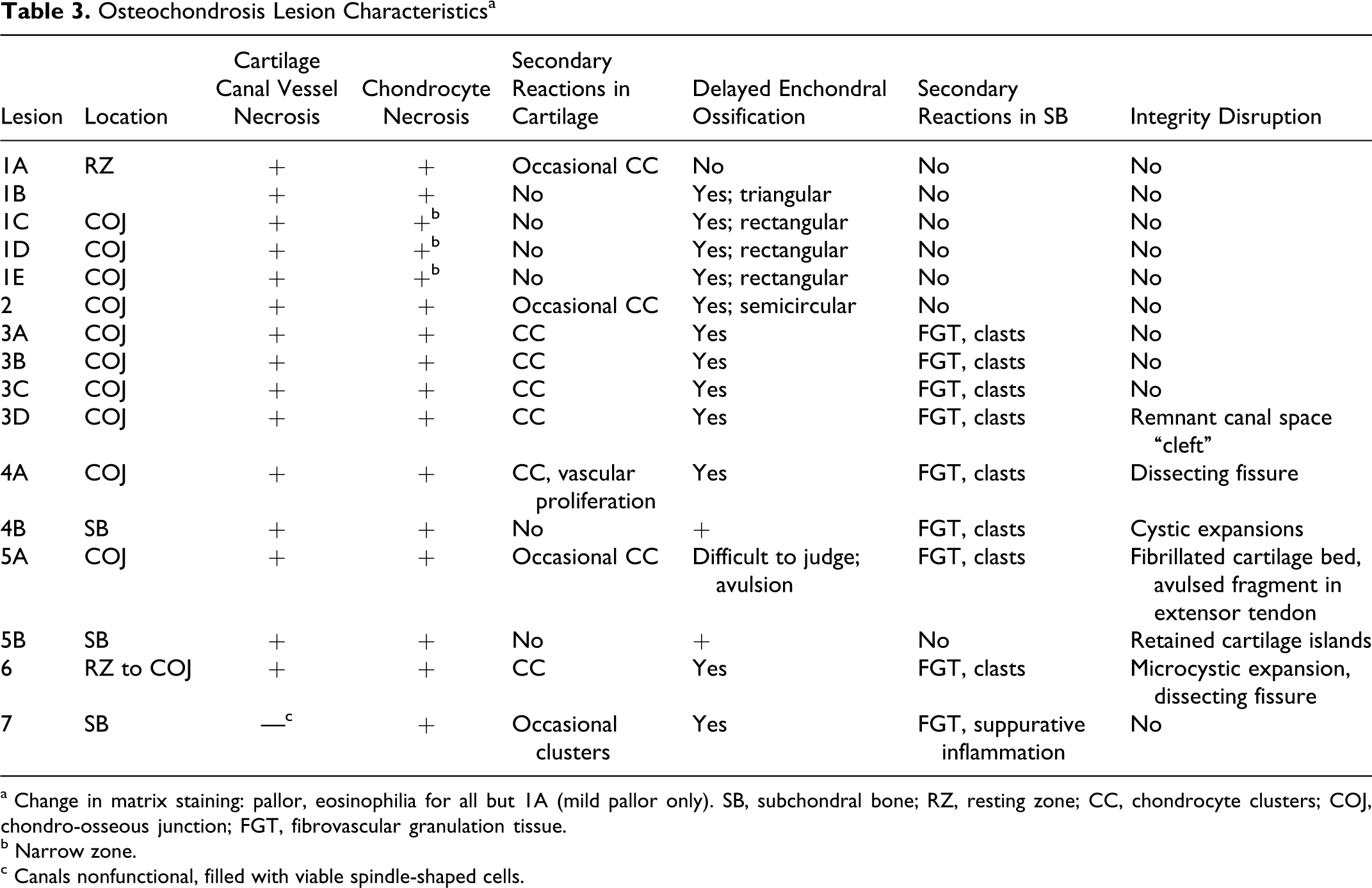
a Change in matrix staining: pallor, eosinophilia for all but 1A (mild pallor only). SB, subchondral bone; RZ, resting zone; CC, chondrocyte clusters; COJ, chondro-osseous junction; FGT, fibrovascular granulation tissue.
b Narrow zone.
c Canals nonfunctional, filled with viable spindle-shaped cells.
Case No. 1: Male Standardbred (44 Days Old)
A total of 5 distinct lesions (1A–1E) were identified in the left and right distal femora. On macroscopic postmortem inspection, the left and right femoropatellar joint compartments were grossly enlarged medially and contained approximately 350 to 400 ml of normal synovial fluid each. Lesion 1A was identified in the routinely sampled middle slab from the left femur of case No. 1. The area of ischemic chondronecrosis was visible in histologic sections only (Fig. 3). Lesions 1B–1E were macroscopically visible in slabs. Lesion 1B was found in the lateral abaxial aspect of the right lateral trochlear ridge. It consisted of a triangular area of ischemic chondronecrosis (10 mm wide × 10 mm deep) that extended from the epiphyseal growth cartilage into the subchondral bone. Lesions 1C–1E were located on the cranial aspect within the cartilage of the trochlear ridges. Lesion 1C was located in the same slab as lesion 1B. The lesions consisted of rectangular areas of growth cartilage up to 3 mm wide and 8 mm deep that extended from the epiphyseal growth cartilage into subchondral bone. There was a narrow zone of ischemic chondronecrosis at the deep margin of each rectangular area. The areas of cartilage within the subchondral bone in lesions 1B–1E corresponded in location and size to focal subchondral radiolucent areas identified radiographically in vivo.
Case No. 2: Female Standardbred (114 Days Old)
The left and right distal femora of case No. 2 were normal on postmortem inspection of the intact bones and sawed slabs. A single lesion designated lesion 2 was identified on histologic examination of the routinely sampled middle slab from the left femur, and it was located on the axial aspect of the lateral trochlear ridge (Fig. 4). The most axially located area of ischemic chondronecrosis was associated with a microscopic focal delay in enchondral ossification.
Case No. 3: Male Standardbred (336 Days Old)
Four lesions (3A–3D) were detected on postmortem inspection of the intact left and right distal femora, and they consisted of circular areas (20- to 25-mm diameter) of focally increased cartilage opacity. These areas were on the cranial aspect of the middle third of the left and right medial and lateral trochlear ridges. In the lesion 3D, the proximal half of the circular opaque area was outlined by a thin groove in the cartilage, and it yielded to pressure. In slabs, lesions 3A–3D corresponded to focal areas of mildly thickened cartilage. There was an area (5–10 mm deep) composed of soft–red or firm–pale tissue within the subchondral bone subjacent to each focus of thickened cartilage.
On histologic examination, the thickened cartilage in lesions 3A–3D contained areas of ischemic chondronecrosis. In lesion 3D, a remnant canal space (1 mm wide × 11 mm long and oriented parallel to the underlying ossification front) was present in the center of the area of chondronecrosis (Fig. 5). On radiographic examination, lesions 3A–3D corresponded in location and size to focal subchondral radiolucent areas.
Case No. 4: Male Standardbred (156 Days Old)
Two lesions (4A and 4B) were detected in the left distal femur. On surface inspection, a full-thickness dissecting fissure extended from the lateral abaxial aspect to the cranial aspect in the middle third of the lateral trochlear ridge. In slabs, the epiphyseal growth cartilage was markedly thickened in the 4 middle slabs, and the dissecting fissure extended into the area of thickened cartilage in the 2 most proximally located slabs of the 4. The thickened cartilage and dissecting fissure were designated lesion 4A. On histologic examination, the thickened cartilage consisted of an area of ischemic chondronecrosis into which the dissecting fissure directly extended (Fig. 6).
A second lesion (designated 4B) was identified in the same 4 slabs as lesion 4A, as well as 1 slab proximal and 1 slab distal to these slabs (total of 6 slabs). Lesion 4B was located deep to lesion 4A and extended up to 30 mm from the cartilage surface into the subchondral bone. Between lesions 4A and 4B, the subchondral bone was mildly disorganized. Lesion 4B consisted of a zone of firm white tissue (2–3 mm wide) within the subchondral bone in the 5 most distal slabs. In the proximally located sixth slab, lesion 4B expanded into 1 large (7-mm diameter) and 2 to 3 smaller (3-mm diameter) cystic lesions.
Histologically, lesion 4B contained occasional areas of necrotic cartilage but consisted predominantly of fibrovascular granulation tissue. The cystic lesions in the most proximal slabs were surrounded by a collagenous cyst wall and contained necrotic material that resembled vascular structures and connective tissue.
On radiographic examination, lesion 4A corresponded to a contour defect and a radiolucent area within the subchondral bone in the middle third of the lateral trochlear ridge. The radiolucent defect was surrounded by a halo (5–10 mm thick) of heterogeneously radiodense subchondral bone, which was in turn outlined by a narrow radiolucent line at the transition to normal subchondral bone. The radiolucent line expanded into 2 distinct small circular radiolucencies toward its proximal margin, corresponding in location and size to the lesion described as 4B.
Case No. 5: Female Standardbred (53 Days Old)
Two lesions (5A and 5B) were identified. On macroscopic postmortem inspection of the intact bones, there was a surface defect (10 mm deep with a 15-mm diameter) at the extensor fossa of the right distal femur, which was designated lesion 5A. A similar-sized cartilage-covered bony fragment was present within the fibers of the long digital extensor tendon adjacent to the defect, but it was not examined histologically.
When the site was sawed into slabs, areas of cartilage became centrally visible within the subchondral bone of the 2 most distal slabs from the medial condyle of the right femur and were designated lesion 5B (Fig. 7). The areas were confluent with the epiphyseal growth cartilage of the condyle, as well as with one another. The largest area was irregularly oval and located within the most distal slab; it measured 5 mm wide by 10 mm long. Together, the areas implied a conical three-dimensional volume of cartilage within the subchondral bone.
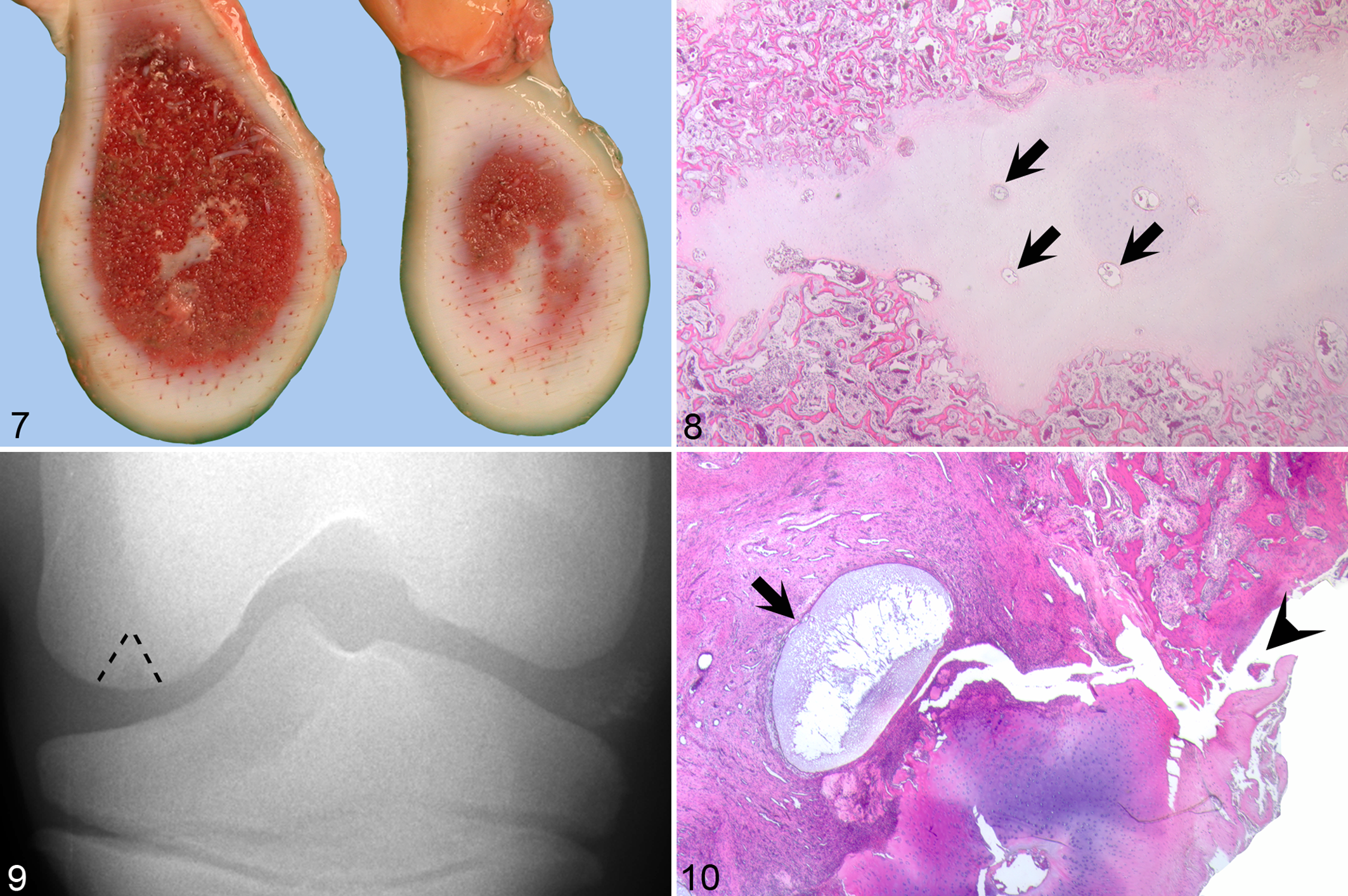
On histologic examination, the surface of the defect in lesion 5A consisted of fibrillated cartilage. Deep to this was a thin zone of ischemic chondronecrosis, accompanied by a thick zone of fibrovascular granulation tissue at the transition to normal subchondral bone. The cartilage islands in lesion 5B contained numerous necrotic vessels and consisted almost entirely of necrotic cartilage (Fig. 8).
On radiographic examination, the cartilage islands in lesion 5B corresponded to a triangular radiolucent defect in the subchondral bone of the right medial femoral condyle (Fig. 9). Upon close scrutiny of the radiographs, a faint mineralized radiopacity was visible that corresponded to the cartilage-covered bony fragment within the long digital extensor tendon (not shown).
Case No. 6: Male Swedish Warmblood Riding Horse (305 Days Old)
On macroscopic postmortem examination of the intact femora, a full-thickness dissecting fissure was visible on the medial aspect in the middle third of the medial femoral condyle of the left distal femur. The fissure was designated lesion 6.
In slabs, the fissure extended into an area of moderately thickened cartilage. Histologically, the thickened cartilage consisted of an area of ischemic chondronecrosis. Remnants of necrotic vessels at the level of the transition between the area of chondronecrosis and the adjacent granulation tissue appeared dilated compared to vessel remnants in more superficial zones of the necrotic cartilage. The most medially abaxial canal vessel remnant expanded into an oval space 1 mm wide by 2 mm long (microscopic cyst; Fig. 10). The dissecting fissure extended into the granulation tissue immediately adjacent to the cyst (Fig. 10).
Case No. 7: Female Standardbred (31 Days Old)
The distal femora were normal on postmortem inspection of the intact bones. When sectioned into slabs, roughly circular areas of abnormal tissue became visible in the center of the right medial femoral condyle in the 4 middle slabs. The circular areas were confluent, corresponding to a spherical volume of an approximately 25-mm diameter, and were designated lesion 7. The abnormal tissue was composed of a mixture of firm pale tissue, soft red tissue, and clear red fluid.
Histologically, the cartilage canals within the epiphyseal growth cartilage superficial to lesion 7 contained no viable or necrotic vessels but, rather, vascular spaces and viable spindle-shaped cells (Fig. 11). The cartilage surrounding canals with this morphology was necrotic (Fig. 11). Lesion 7 also consisted of relatively distinct areas of 2 tissue types: necrotic epiphyseal cartilage; moderately cellular granulation tissue; and tissue composed of large numbers of degenerate inflammatory cells associated with a variable but generally low volume of basophilic intercellular material compatible with suppurative inflammation.
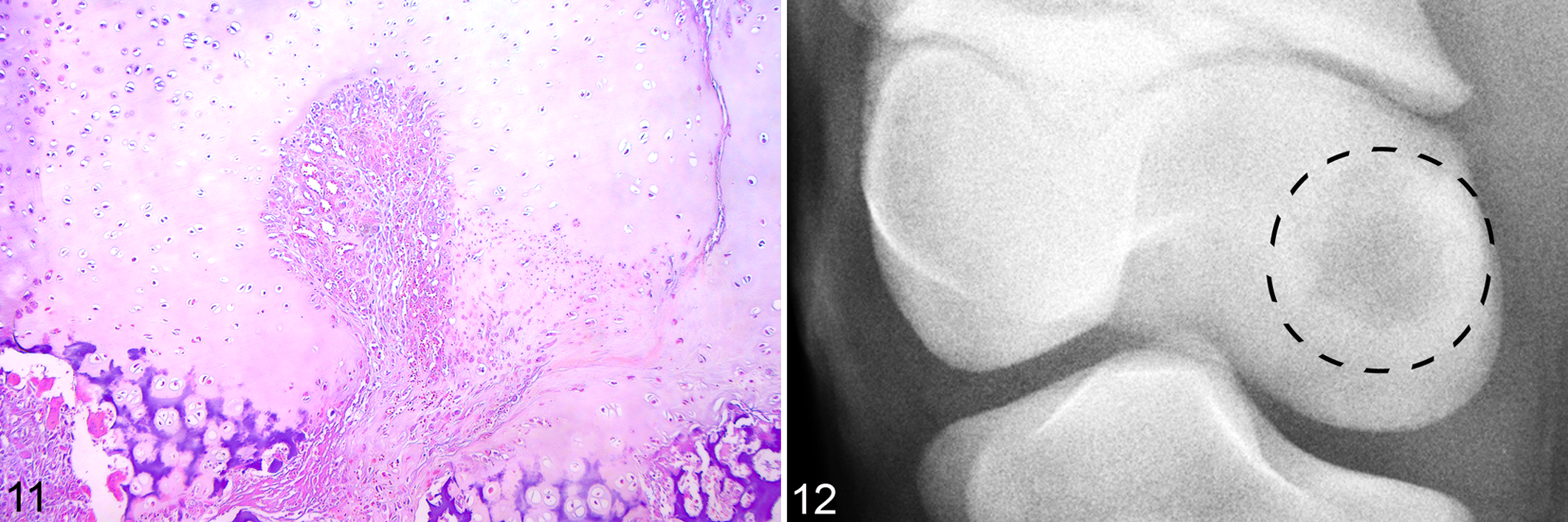
On radiographic examination, lesion 7 corresponded to an irregularly circular area of subchondral radiolucency surrounded by a thick sclerotic rim and separated from the joint space by radiographically normal bone (Fig. 12).
Discussion
Ideally, the relationship between the developmental pattern of the blood supply and OC should be studied in one group of individuals. However, such a unique collection of specimens is not available, and the prevalence of OC in horses is low in comparison to that of other domestic animals. Therefore, an absence of OC lesions was not unexpected in the small group of Standardbred foals used to describe the developmental pattern of the blood supply to the distal femur. 21 An alternative approach therefore had to be taken in the current study for comparing the location and morphology of lesions (n = 16) in one sample of foals to the pattern of blood supply described in a different group of foals of similar age.
Previous studies have suggested that cartilage canal vessels are particularly vulnerable to failure in the region where they become incorporated into the ossification front during enchondral ossification.23,39 This vulnerability may in part stem from the requirement for the vessel to traverse a junction between 2 tissues having different material properties. In the developing epiphyses, there are at least 2 such junctions to consider: that between growth cartilage and bone23,25,39 and that between growth cartilage and perichondrial connective tissue, including sites of ligament and tendon attachment.22,27
In a previous study of the developmental pattern of the blood supply, 2 configurations of vessels were described: vessels that coursed predominantly parallel to the underlying ossification front and vessels that coursed predominantly perpendicular. 21 Perpendicular vessels originating in bone traversed the chondro-osseous junction at a single location to enter cartilage canals of the epiphyseal growth cartilage. 21 Parallel vessels originated from the perichondrium and traversed the junction between connective tissue and cartilage to enter the epiphyseal cartilage canals. 21 The midsection of these parallel vessels was incorporated into the ossification front during growth, forcing them to traverse a junction between 2 tissues (chondro-osseous junction) for a second time.21,39
In the present study, all but lesions 5B and 7 were located on the cranial, craniolateral abaxial, and caudomedial abaxial aspects of the distal femur (Table 2), sites in which parallel vessels predominate. 21 Lesions 5B and 7 were located in the center of the medial femoral condyles, where vessels during the age interval examined coursed predominantly perpendicular to the ossification front. 21 The ratio of lesions in regions supplied by parallel versus perpendicular vessels was 14:2, supporting the suggestion that parallel vessels that are incorporated into the ossification front are vulnerable to failure.21,39
Chronic lesion 5A was located at the extensor fossa, where traction from the extensor tendon may have contributed both to disruption of the vessel at the point at which it traversed the junction between perichondrial connective tissue and epiphyseal growth cartilage and to the subsequent avulsion of the osteochondral fragment found within fibers of the long digital extensor tendon. An appreciation of vessel configuration and its relationship to biomechanical forces such as traction may refine our understanding of vascular failure to epiphyseal cartilage in horses. We believe, however, that the main factor that renders vessels vulnerable to failure is the requirement that they traverse junctions between tissues of different properties—the chondro-osseous junction in particular. This is in agreement with Pool’s proposed pathogenesis for OC in the lateral trochlear ridge of the distal femur in horses 25 and with previous observations made in a larger number of distal femora from piglets. 39
All identified lesions contained necrotic cartilage canal vessels associated with variable areas of ischemic chondronecrosis. The most subtle lesion, 1A, was confined to the resting zone of growth cartilage (Fig. 3). This early stage has been observed in piglets and designated osteochondrosis latens. 38
The most axially located area in lesion 2 represented an area of ischemic chondronecrosis that arose in the intermediate layers of growth cartilage but that had become partially relocated to the subchondral bone by continued advancement of the ossification front. This stage has also been recognized in piglets and subsequently designated osteochondrosis manifesta. 38
Advancement of the ossification front to surround lesions 1B–1E gave a more direct visualization of their shape. The laterally located lesion 1B was triangular, and it may have corresponded to a conical volume in 3 dimensions. The cranially located lesions 1C–1E were rectangular, and only a narrow zone of cartilage at the deep margin of the rectangle was necrotic. A rectangular shape in histologic sections could correspond to a cylindrical volume in 3 dimensions. It seems reasonable to suggest that the growing epiphysis may be organized into different subunits that produce volumes of cartilage suited to attain the eventual adult shape. Cylindrical volumes have a uniformly shorter diffusion distance to alternative, collateral blood supply compared to conical volumes, potentially explaining why many of the chondrocytes within the rectangular ossification-resistant areas of cartilage were morphologically viable.
The areas of chondronecrosis in lesions 3A–3D were associated with a large amount of subchondral granulation tissue, which may precede the production of repair bone by intramembranous ossification. 19 Evidence for this is provided in descriptions of the spontaneous resolution of radiographic lesions.2,3 Areas of ischemic chondronecrosis that arise during development therefore have the potential to heal and are not necessarily of consequence in older animals.
The remnants of a necrotic cartilage canal vessel formed a cleft through the center of the area of chondronecrosis at the ossification front in lesion 3D, and the intact overlying cartilage was palpably unstable in the gross specimen (Fig. 5). From the stage when lesions are located at and below the ossification front, the area of chondronecrosis replaces the mineralized zone of growth cartilage normally present in this site. The mineralized zone is considered to form a bond between the epiphyseal cartilage and bone. 36 Thus, necrosis of the vessels in cartilage canals or areas of chondronecrosis replacing the mineralized zone may be responsible for dissecting lesions at a stage when the delay in enchondral ossification is minimal, which could explain why clinical dissecting lesions are frequently observed in the absence of grossly visible thickening of the cartilage (ie, delayed enchondral ossification). 16
OCD lesions 4A, 5A, and 6 of the current study were all associated with focal delays in ossification. With the exception of the avulsed fragment in lesion 5A, dissection was present in only a subset of the total area of the delay. The presence of delayed ossification in all 3 lesions—only a portion of which had undergone dissection in lesions 4A and 6—suggests that the disease-associated delay had preceded and predisposed to the development of clinical OCD lesions in these foals.
Lesions 4B, 5B, 6, and 7 appear to provide insights into the relationship among ischemic chondronecrosis, OC, and cystic lesions. Several mechanisms for the formation of developmental subchondral bone cysts have been proposed, including infolding of cartilage, 32 retention and liquefactive necrosis of cartilage,17,34 and trauma with microfracture of the subchondral bone. 9 In lesion 4B, cysts were present within the subchondral granulation tissue associated with chronic ischemic chondronecrosis. The cysts apparently arose because the granulation tissue became walled off by an aggregation of collagenous connective tissue into a cystlike lining.
The ischemic chondronecrosis in lesion 5B (Figs. 7, 8) was radiographically compatible with diagnosis of a subchondral bone cyst, even though a histologic “cyst” was not present (Fig. 9). Most cases of subchondral bone cysts in horses are diagnosed on the basis of radiographic findings alone; histopathological validation is rarely feasible in live patients. Thus, the confusion over the relationship between equine OC and subchondral bone cysts is understandable. The relationship between OC and the clinical appearance of a subchondral bone cyst in case No. 5 was clear in that histopathologic examination validated that the cystic lesion within the medial femoral condyle consisted of a volume of necrotic cartilage associated with delayed enchondral ossification.
A microscopic cyst was found at the deep margin of an OCD fissure in lesion 6 (Fig. 10). The necrotic cartilage canal vessels in this foal appeared dilated, and the microscopic cyst may have represented the remnant of a necrotic vessel. Interestingly, various categories of experimentally created defects into the articular cartilage of ponies and horses 1 to 2 years old have been shown to result in the formation of subchondral cystic lesions.12,26 Thus, it is possible that a full-thickness OCD fissure, as seen in lesion 6, can similarly lead to the formation of a developmental subchondral bone cyst.12,26
The microscopic cyst in lesion 6 was peripherally lined by acellular material (Fig. 10). Fibrous tissue explant cultures from equine subchondral bone cysts have demonstrated bone resorptive enzyme activity in vitro; 35 thus, the microscopic cyst in lesion 6 could have increased in size over time through connective tissue enzyme activity, perhaps representing a precursor to a macroscopically visible and radiographically detectable subchondral bone cyst.
The cystic lesion in case No. 7 comprised elements of ischemic chondronecrosis (Fig. 11), chronic inflammation, and suppurative inflammation. Thus, this lesion may represent a manifestation of more than one disease process, with the relationship among the processes being unclear. The morphology of the cartilage canal vessels in lesion 7 was not compatible with any previously described cartilage canal morphology in horses. In juvenile equine osteomyelitis, subchondral lesions are believed to result from bacterial colonization of vascular sinusoids located immediately deep to the epiphyseal or metaphyseal growth cartilage and to the immune response to such colonization. 5 Thus, the morphology of lesion 7 could represent local spread of a subchondral infection into superficially adjacent cartilage canal spaces. Experimental infection in other species has demonstrated a primary bacterial glycocalyx binding to cartilage matrix components, as accessed through endothelial fenestrations in capillaries within cartilage canals and as followed by vascular occlusion and spread of infection to subchondral bone. 31 Another possibility is that the morphologic changes seen in lesion 7 represent the sequel to bacterial occlusion of cartilage canal vessels, with spread of infection to adjacent subchondral bone. The lesion observed in the medial femoral condyle of case No. 7 appears highly compatible with those previously described on the caudal aspect of the condyle in 20 foals. 7
In all animals except those of case Nos. 2 and 6, joints that were histologically diagnosed with ischemic chondronecrosis were clinically diagnosed with joint effusion. Joint effusion has been believed to occur secondary to surface-disrupting OCD lesions. Thus, it is interesting to note that the surface integrity of the cartilage was intact in the effused joints of case Nos. 1, 3, and 7. The mechanism behind the effusion in these foals is unknown, but note that Rhodococcus equi (as isolated from a subclinical lung lesion in case No. 1) has been associated with reactive arthritis in foals. 10
The joint effusion in case Nos. 4, 5, and 7 was not associated with any apparent lameness. Pain from sterile effusion may have been clinically masked by septic arthritis in a different joint (in case No. 5) or in a different limb (in case No. 7).
In the current study, lesions of ischemic chondronecrosis were found in regions of the epiphysis where vessels are obliged to traverse junctions between tissues—most commonly, the chondro-osseous junction. It appears that the process of traversing tissue junctions renders the canal vessels vulnerable to failure in the distal femur of foals, as previously observed in the tarsus of foals 23 and the distal femur of piglets. 39
Footnotes
Acknowledgements
We are indebted to Dr Henrik Uhlhorn for permission to include material from 2 foals and to Dr Randi Sørby, Dr Arild Espenes, and Prof Mona Aleksandersen for permission to include material from case Nos. 1, 3, and 4, respectively. The generous assistance of the staff at the departments of pathology at the Swedish University of Agricultural Sciences and the Norwegian School of Veterinary Science is gratefully acknowledged.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The current study was funded by grant No. 41514 from the Research Council of Norway.
