Abstract
Failure of the cartilage canal blood supply leads to ischemic chondronecrosis which causes osteochondrosis, and osteochondral lesions. Osteochondrosis is a disease with a heritable component and usually occurs under aseptic conditions. Because bacteria can bind to growth cartilage and disrupt the blood supply in pigs and chickens, we considered whether this might play a role in development of equine osteochondrosis. The aim of this study was to examine whether bacteria are present in canals in the growth cartilage of foals with septic arthritis/osteomyelitis, and whether this is associated with osteochondrosis. The material consisted of 7 foals aged 9-117 days euthanized because of septic arthritis/osteomyelitis. The 7 cases had 16 lesions in growth cartilage that were evaluated histologically. Bacteria were present in cartilage canals in foals with septic arthritis/osteomyelitis. Portions of necrotic canals adjacent to bacteria frequently contained neutrophils, termed acute septic canals; or granulation tissue with neutrophils, termed chronic septic canals. Acute and chronic septic canals were associated with ischemic chondronecrosis in the articular-epiphyseal cartilage complex (AECC) of 5 cases and in the physis of 2 cases, and ossification was focally delayed in 5 of those 7 cases. Lesions occurred with and without adjacent osteomyelitis. Bacteria were present in cartilage canals and were associated with focal chondronecrosis in both the AECC and the physis. This establishes sepsis as a plausible cause of some osteochondral lesions in horses. It is recommended that horses with sepsis-related osteochondral lesions may be used for breeding without increasing the prevalence of OCD-predisposing genes in the population.
The long bones of mammals grow by endochondral ossification. 29 The growth cartilage has a temporary blood supply that runs in cartilage canals, which usually contain an arteriole, a capillary network and one or more venules organized as anatomic end arteries, and perivascular mesenchymal cells. 5,26 This renders the blood supply vulnerable in case of damage. Cartilage canals regress by 2 mechanisms; chondrification, that is, becoming filled with cartilage, 4 and incorporation into the advancing ossification front. 37,39 When predilection sites for osteochondrosis in the articular epiphyseal cartilage complex (AECC) were examined in pigs and horses, necrotic cartilage canals surrounded by necrotic chondrocytes were found at intermediate depth. 39,52 Both spontaneous and experimental vascular failure in the AECC result in ischemic chondronecrosis and delayed enchondral ossification, in pigs termed osteochondrosis latens and osteochondrosis manifesta, respectively. 34,48,51,53 Ischemic chondronecrosis and delayed enchondral ossification have the potential to resolve, 7 or develop into osteochondrosis dissecans (OCD). 33 Secondary responses in the growth cartilage including proliferation of morphologically viable chondrocytes and canals have been observed adjacent to areas of ischemic chondronecrosis. 35 After areas of ischemic chondronecrosis are surrounded by the ossification front, repopulation of necrotic canals by cells from subchondral bone marrow has been observed. 35
Osteochondrosis is most often a multifocal disease and occurs at specific predilection sites in the joints of various species, including pigs, 41 horses, 43 dogs, 31 and humans. 8 A heritable predisposition for osteochondrosis has been documented in horses 16,28,49 and pigs. 1,42 Most lesions of osteochondrosis occur under aseptic conditions. 10,21 Cartilage canals have, however, been experimentally established as a target for bacterial infection in pigs 6 and chickens. 46 On transmission electron microscopy, bacteria were observed in the distal end of ingrowing cartilage canals in chickens, where the endothelium is discontinuous. 23,46 Necrosis of cartilage canals followed. 6,9,46 This represents a mechanism where bacteria cause ischemic chondronecrosis, with the potential to develop into OCD. 34 Septic arthritis has been associated with osteochondral lesions in the femoral condyles of foals. 18,20 Higher numbers of osteochondral lesions were also found in the hock and fetlock joints of horses treated for bacterial infections in their first 6 months of life compared to a control group with unknown infection history.(E.H.S. Hendrickson, personal communication)
As osteochondrosis has a heritable component, individuals with the condition are subject to a variety of breeding restrictions. Differentiating individuals with sepsis-related osteochondrosis from the ones with lesions due to heritable predisposition might allow a larger population to be used for breeding. For this to become reality, it is first necessary to investigate whether bacteria colonize cartilage canals in horses. The aim of the current study was to examine whether bacteria are present in canals in the growth cartilage of foals with septic arthritis/osteomyelitis, as previously documented in pig and chickens, and whether this is associated with ischemic chondronecrosis.
Materials and Methods
Cases
The current study contained 7 euthanized foals, 6 from the Equine Hospital of the Norwegian University of Life Sciences and one from the Swedish University of Agricultural Sciences. Inclusion criteria were treatment for septic arthritis/osteomyelitis, or necropsy diagnosis of septic arthritis/osteomyelitis. The upper age limit was 6 months. Both sexes and any breed of horse or pony were included. Histologic sections that included growth cartilage, with or without bone, had to be available. Cases where necropsy was done more than 3 days after euthanasia and where pathologic changes had resulted in complete loss of anatomical structures were excluded. The 7 cases were given ascending numbers by age, summarized in Supplemental Table 1. The femoral lesions in cases 3 and 6 had been previously described. 36
Sample Collection
The third metacarpal bone (2), ilium (1), femur (6), tibia (5), talus (2), and third metatarsal bone (2) were sampled and sawed into approximately 5 mm thick slabs. The tali and ilium were sawed in a sagittal plane, the tibia was sawed in a slightly oblique sagittal plane parallel to the intermediate ridge and other bones were sawed in a transverse plane. All samples of the AECC included bone, whereas for physes, epiphyseal bone was always included but inclusion of metaphyseal bone varied between samples. All samples were fixed in 4% phosphate-buffered formaldehyde for 24-48 hours and decalcified in 10% ethylenediaminetetraacetic acid (EDTA), except for case 3 which was decalcified in formic acid. Sampled regions were chosen based on macroscopically visible changes or taken from standardized slabs, usually the middle slab. The samples were paraffin-embedded, before at least 2 approximately 5 µm-thick sections were cut from each block and stained with hematoxylin and eosin. Based on initial screening, sections with suspicion of bacteria were subjected to Gram staining (Supplemental Methods 1 and 2), provided that there was tissue left to section in the block.
Parameters Observed
Individual cartilage canals were categorized according to criteria published earlier for patent, 5 chondrifying 3 and necrotic canals. 37 Chondrocytes were interpreted as necrotic when they were shrunken, had cytoplasmic eosinophilia and nuclear pyknosis or karyolysis. 37 When chondrocyte necrosis and matrix change (pallor and relative eosinophilia) were present together, this was referred to as chondronecrosis, and when areas of chondronecrosis were centered on necrotic cartilage canals, they were referred to as areas of ischemic chondronecrosis. 37
Results
Details of the 7 foals with septic arthritis/osteomyelitis included in the study are presented in Supplemental Table 1.
The 7 cases included 30 joints with a diagnosis of septic arthritis/osteomyelitis, and one case had osteomyelitis in the growth cartilage of the tuber coxa of the ilium, that is, there were 31 infected sites (Supplemental Table 1). Histologic material was available from 14 of the 31 infected sites. From these sites, a total of 49 blocks from 19 smaller regions were examined and lesions were found in 41 blocks (16 regions) (Supplemental Table 2).
Bacteria were suspected based on examination of hematoxylin and eosin-stained histologic sections in 6 locations from cases 1, 2, 5, and 7 (Supplemental Table 2). Gram staining confirmed presence of gram-positive (cases 1, 5, and 7) or gram-negative (case 2) bacilli or coccobacilli in the 5 locations with adequate available (Supplemental Table 2).
The most striking feature in the AECC of case 1 was that the most superficial (Fig. 1) portions of the cartilage canals contained only intensely eosinophilic, granular material (Fig. 2) comprising remnants of degenerated erythrocytes, fibrin, canal matrix, and a low number of gram-positive bacilli (Fig. 3). In the physis of cases 1 and 2, large numbers of bacteria and granular material were consistently found within portions of the deep (Fig. 4) cartilage canals (ie, those closer to the metaphyseal than the epiphyseal side of the physis). Between the bacteria-containing portions and the diaphysis, canal lumina were reduced to eosinophilic streaks (Fig. 4). In the physis of case 2, bacteria were located within ghost-like remnants of vascular lumina (Fig. 5), as well as in the perivascular connective tissue where they were apparently bound to cartilage matrix at the canal boundary (Fig. 6) and, occasionally within chondrocyte lacunae (Fig. 6). Bacteria and granular material therefore followed the pattern of being distributed superficially within the AECC (Fig. 1) and deep within the physis (Fig. 4).
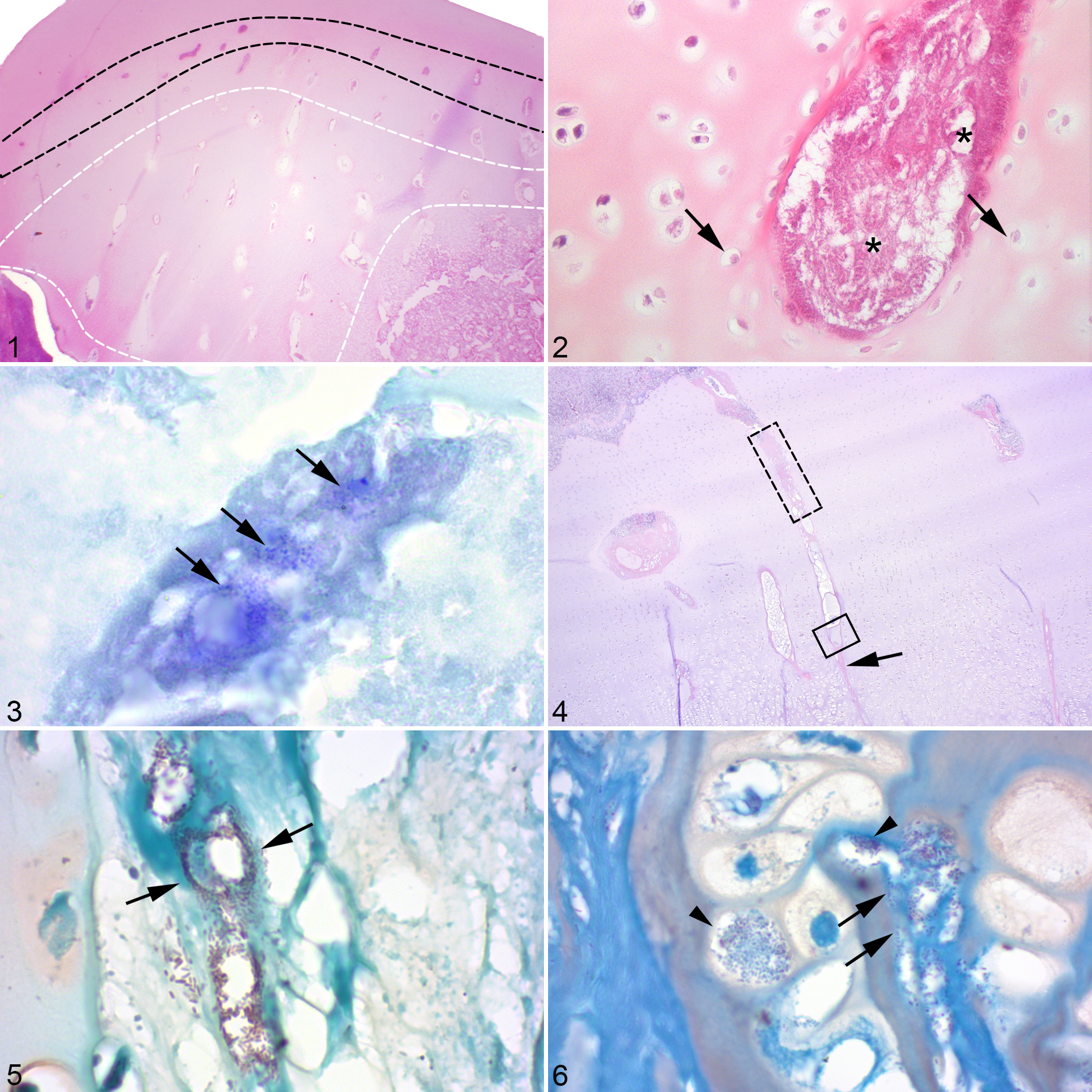
Relationship of Bacteria, Cartilage Canal Lesions, and Chondrocyte Necrosis
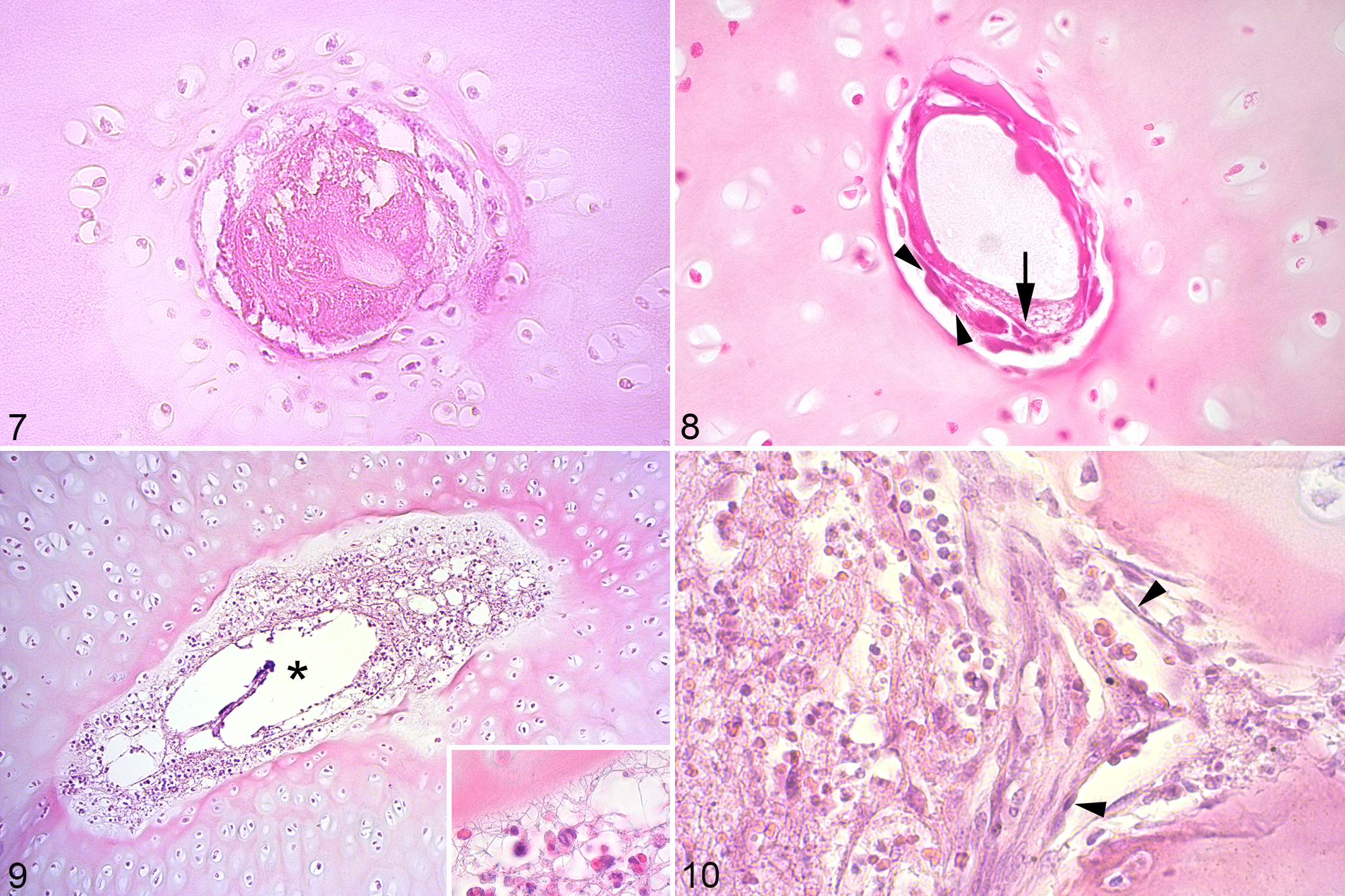
Adjacent to the bacteria and granular material, there was fibrin (Fig. 7). Next to the fibrin, there were necrotic canals, the minority of which were indistinguishable from necrotic canals earlier described in osteochondral lesions, hereafter called aseptic necrotic canals (Fig. 8). The majority of necrotic canals contained remnants of degenerated neutrophils (Fig. 9) in addition to necrotic endothelial and perivascular mesenchymal cells, and were referred to as acute septic canals (Table 1). Adjacent to the septic canals, and also immediately adjacent to and sometimes visibly bridging the ossification front, there were canal portions that contained morphologically viable cells comprising fibroblast-like cells (Fig. 10), capillaries and neutrophils, interpreted as repopulation of necrotic canals with septic fibrovascular granulation tissue from bone marrow and referred to as chronic septic canals (Table 1). The sequence of changes from canal portions containing bacteria/granular material to the secondary, epiphyseal center of ossification was therefore fibrin (Fig. 7), acute septic canals (Fig. 9) at intermediate depth, and chronic septic canals (Fig. 10) immediately adjacent to the ossification front, both for canals in the AECC and in the physis.
Definitions for Lesions of Cartilage Canals in Foals with Septic Arthritis/Osteomyelitis.
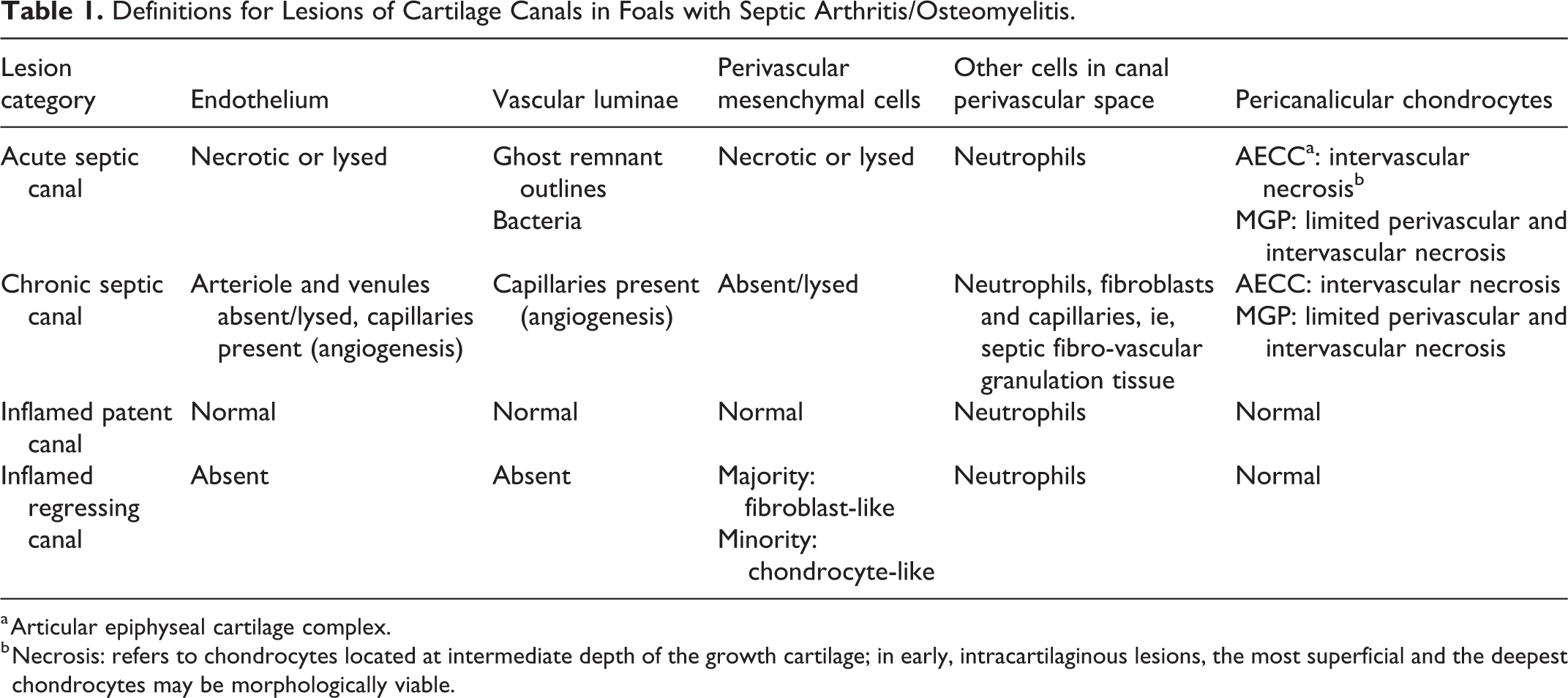
a Articular epiphyseal cartilage complex.
b Necrosis: refers to chondrocytes located at intermediate depth of the growth cartilage; in early, intracartilaginous lesions, the most superficial and the deepest chondrocytes may be morphologically viable.
Chondrocytes around granular portions superficially within the AECC of case 1 tended to be morphologically viable on the superficial side, and necrotic on the deep side of canals (Fig. 2). All chondrocytes around acute septic canals at mid-depth of the AECC were necrotic, similar smaller areas were detected around mid-depth septic canals in the AECC of cases 3, 4 and 6, respectively (Supplemental Table 2). Chondrocytes immediately adjacent to the secondary center of ossification tended to be viable irrespective of canal status. In the physis of case 1, chondrocyte necrosis was limited to occasional cells or a narrow zone surrounding septic canals. Examples of chondrocyte necrosis being extensive within the AECC and limited within the physis of the same foal were also found in cases 1, 4 and 6 (Supplemental Table 2). The only exception to limited chondrocyte necrosis within the physis was that larger areas of necrosis were observed between canals within the resting zone of the physis in one block from the left metatarsus, and at the AECC/physeal junction in another block from the right distal femur of case 2, also at the level of the resting zone.
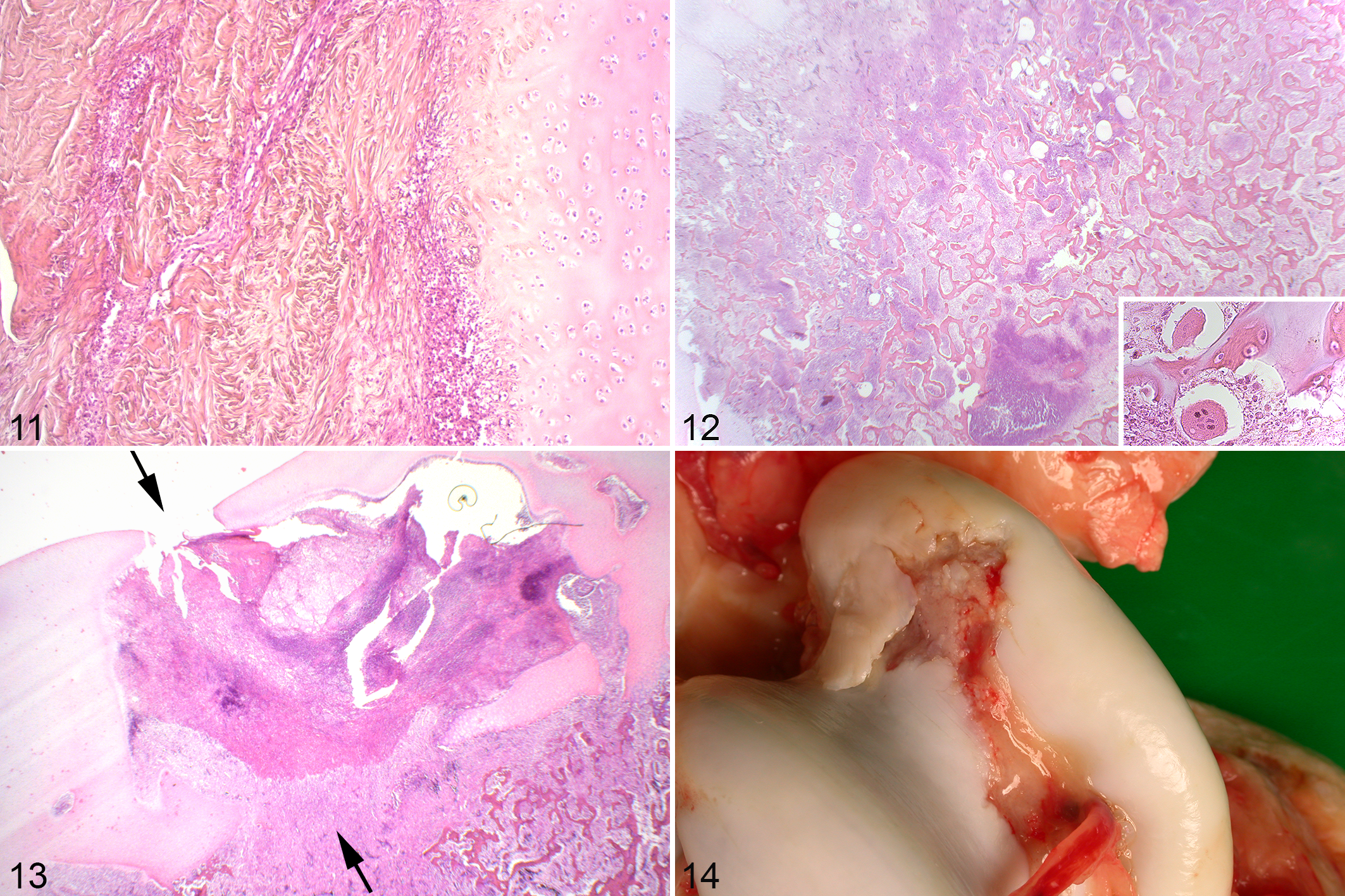
The perichondrium of the majority of cases was normal (Supplemental Table 2), but in cases 1 and 5, it contained linear tracts of strongly eosinophilic material (Fig. 11) consisting mainly of degenerated neutrophils, interpreted as inflammation and destruction of perichondral blood vessels.
Delayed Ossification, Septic Osteochondrosis, and Osteomyelitis
The area of chondronecrosis containing septic necrotic canals within the AECC of case 1 was associated with marked focal delay in endochondral ossification (Fig. 1), hereafter termed septic osteochondrosis. Delayed ossification was also detected in cases 3, 4, 5, and 6, and described macroscopically for case 7 but not represented in the available histological sections (Supplemental Table 2). In 2 sites (the tuber coxa of case 5 and the medial femoral condyle of case 6), the area of delayed ossification consisted predominantly of necrotic vessels and chondronecrosis. In the majority of cases, however, the area of delayed ossification comprised a mixture of necrotic growth cartilage and granulation tissue containing degenerated neutrophils (Supplemental Table 2).
Immediately deep and axially adjacent to the septic osteochondrosis lesion in case 1, there was evidence of neutrophil invasion, necrosis of primary spongiosa and formation of granulation tissue with multinucleated giant cells interpreted as osteoclasts (Fig. 12), that is, suppurative, necrotizing osteomyelitis. Among all cases, some AECC and physeal lesions had no adjacent osteomyelitis, some had adjacent osteomyelitis in some blocks, and some adjacent osteomyelitis in all blocks (Supplemental Table 2). Within the physis, osteomyelitis occurred within the adjacent epiphyseal center of ossification, within the diaphyseal center of ossification, or both (Supplemental Table 2).
Macroscopic Cyst- and OCD-Like Changes
Part of the lesion in case 3 was encapsulated by fibrous tissue, compatible with formation of a Brodie’s abscess (Supplemental Table 2). Superficial to this, there were vertical linear areas of septic granulation tissue within the AECC that were compatible with, but considerably larger than, most chronic septic canals. In 2/4 blocks, these extended through the entire thickness of the AECC as nondisplaced pathologic fractures (Fig. 13).
Case 6 had a radiographically visible lesion in the distal femur previously diagnosed as a subchondral bone pseudocyst, in which the chondronecrosis was confirmed to be centered on septic canals (Supplemental Table 2). In the caudal distal tibia/hamate process and talus (Fig. 14) of case 6, areas of chondronecrosis were also associated with macroscopically visible wear lines, cartilage loss, protruding bands of fibrous tissue with neutrophils and undermined cartilage margins (tibia) or a hinged cartilage flap (talus). Finally, case 6 had complete pathologic avulsion of the long digital extensor tendon through an area of septic osteochondrosis.
Canal Infection, Inflammation, and Regression Without Necrosis
A low number of patent canals some distance from lesions contained neutrophils outside the circulation, mixed with the perivascular mesenchymal cells, and were surrounded by normal chondrocytes (Fig. 15). Canals with extra-vascular neutrophils were referred to as inflamed patent canals (Table 1). Similarly, in cases 2-7 there were some inflamed canals without evidence of endothelium or vascular luminae that were surrounded by normal chondrocytes and therefore appeared to be regressing (Table 1). Some of the inflamed regressing canals contained chondrocyte-like cells located in lacunar-like structures, and were therefore regressing by chondrification. The majority, however, contained spindle-shaped, fibroblast-like cells (Fig. 16) and were regressing by fibrosis.
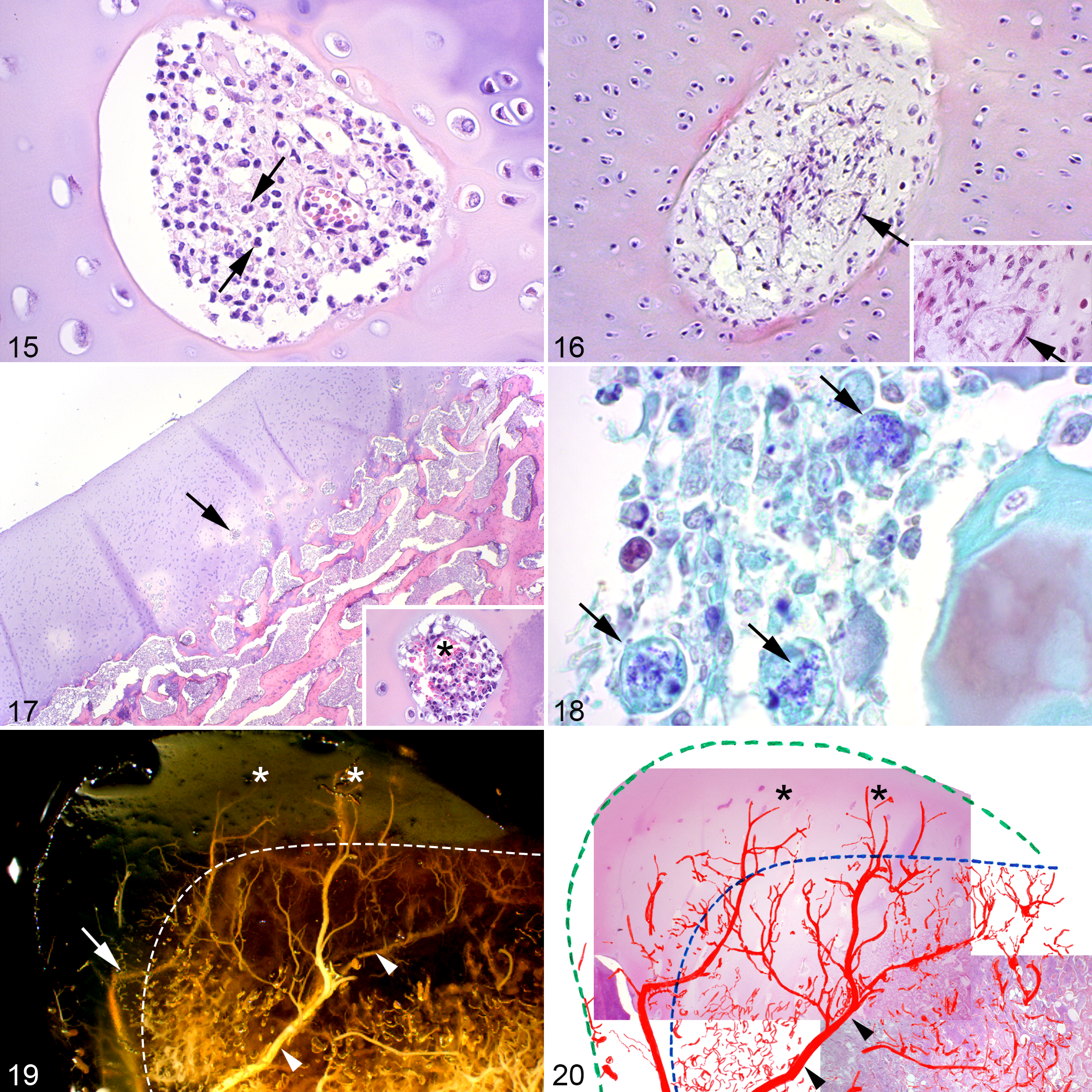
In case 7, the physis of the hamate process was virtually completely replaced by granulation tissue containing degenerated neutrophils and there was delayed ossification, osteomyelitis and a subcutaneous closed abscess. The AECC contained only patent or regressing canals located deep in the cartilage (Fig. 17), several of which were inflamed. Numerous gram-positive coccobacilli compatible with the Rhodococcus equi cultured from the abscess in case 7 (Supplemental Table 1) were also observed within the osteomyelitic area and the patent or regressing cartilage canals (Fig. 18). The coccobacilli were, however, located within macrophages (Fig. 18) and the surrounding chondrocytes were normal.
Discussion
In this study, bacteria were present in canals in the growth cartilage and bone of the AECC and physis of foals with septic arthritis/osteomyelitis. The typical location of bacteria in cartilage canals was superficially in the AECC and deep in the physis. Anatomical studies have confirmed that a major part of blood supply to the epiphyses in the forelimb of foals originates from perichondral arteries. 13,14 Branches of these vessels enter both the epiphyseal cartilage and the physis. 14,15 A similar arrangement has been identified in the AECC of hind limb joints (Fig. 19), although the physis remains to be investigated. 38,39 Vessels superficial in the AECC and deep in the physis thus are the most distal part of this vasculature (Fig. 20). The location of bacteria here is in accordance with experimental studies in chickens and pigs, where bacteria were first found in the distal tips of metaphyseal vessels. 2,9,24,46
Discontinuities in the endothelium of epiphyseal chondrifying cartilage canals of foals aged 1-122 days were recently described. 22 Intravenously injected carbon particles accumulated outside growing distal ends of vessels in cartilage canals in noninfected rats. 19 Preexisting fenestrations and gaps in the endothelium could allow small numbers of bacteria to adhere and trigger an inflammation-mediated disruption of endothelium, leading to more widespread colonization and infection. 40 In this cross-sectional study, it was not possible to determine if fenestrations or gaps were present before bacterial adhesion as the endothelium of the cartilage canals was for the most part absent in the lesions examined.
Bacteremia and the accompanying inflammatory response have an impact on the integrity of the endothelium and exacerbate the permeability to bacteria. 40,50 An inflammation-related increase in the permeability of vessel walls has been suggested to allow bacteria to cross where they otherwise would not be able to cross. 6 A more recent study of intravenous inoculation of Staphylococcus aureus in 8-week-old pigs found that destruction of endothelium in the vasculature of the physis did not occur until influx of neutrophils at 48 hours. 24 In the same study, micro abscesses in the lungs were resolved within 48 hours, while in the cartilage they persisted and increased in size. The combination of the special vascular anatomy in growth cartilage and a systemic inflammation-induced impact because of bacteremia is a likely reason for the persistence of infections in the physis. 15,38 –40
Neutrophils are not a feature of necrotic canals in studies of osteochondral lesions in heritably predisposed breeds (Fig. 8) or in surgically induced lesions. 3,35,37 Increased numbers of neutrophils in and around cartilage canals were demonstrated in experimental studies 24 hours after inoculation of bacteria in pigs and 12 hours after inoculation in chickens. 9,24 Due to their larger size and more common presence, neutrophils are easier to detect in histologic sections than bacteria. 47 In the absence of bacteria, neutrophils were therefore used as a criterion for identification of septic canals.
Chondronecrosis around septic canals was observed in 6 out of 7 foals in this study, both in the AECC and the physis. The areas of chondronecrosis were consistently more extensive in the AECC than in the physis. Osteochondral lesions have been suggested to be a consequence of septic processes in earlier case reports. 18,20 Hendrickson et al found up to 18% higher frequency of osteochondral lesions in yearlings treated for bacterial infections before 6 months of age than controls with unknown infection history.(E.H.S. Hendrickson, personal communication) Bacterial infection and destruction of cartilage canals may therefore account for a small portion of all developmental osteochondral lesions. Causes of delayed ossification other than failure of incorporation of canals were recently described. 11 It therefore makes sense to add a prefix to osteochondrosis in lesions where the cause of delayed ossification is known. Because bacterial infection can lead to ischemic chondronecrosis and focally delayed ossification—the very definition of osteochondrosis 32,33 —we propose that this condition is termed septic osteochondrosis.
The lesions in cases 3 and 6 (Figs. 13, 14) contained macroscopically visible physical disruptions of the articular cartilage. Whether the disruptions should be termed OCD is debatable. The pathologic fractures observed extended through septic granulation tissue rather than areas of ischemic chondronecrosis, which is the defining criterion for OCD. 33 There is evidence that sepsis-survivors have more OCD lesions than animals with unknown status.(E.H.S. Hendrickson, personal communication) The lesions in these cases were in the caudal part of the distal intermediate of the tibia (hamate process), which is not a recognized predilection site for OCD. None of the sepsis-survivors had lesions at the hamate process, (E.H.S. Hendrickson, personal communication) and this may be because lesions resolved or because they were so extensive that the horses did not survive to OCD-screening age (12 months). 27 Both sepsis-survivors (E.H.S. Hendrickson, personal communication) and many of the present cases had lesions at recognized predilection sites for OCD, making it likely that some lesions do persist to screening age. This could be determined through frequent radiography of survivors from initial sepsis, until screening age.
Cases 1 and 2 had areas in the AECC and physis, or just the physis, respectively, with both early chondronecrosis and early osteomyelitis in the same lesion. Osteomyelitis in foals is usually found in the junction between bone and cartilage. 12 Various mechanisms for localization of bacteria in this anatomic site have been in different species: sluggish blood flow in sinusoidal loops in the bone marrow, 12 transphyseal vessels allowing migration of bacteria from the diaphysis to the epiphysis, 2,12,30 or endothelial gaps in growing vessels which bacteria may migrate through. 46 In a previous study, it was considered whether infection spread to the growth cartilage from subchondral bone, or the other way around. 36 Given the common observation of chondronecrosis and osteomyelitis in this material harvested from foals, especially in the very early epiphyseal and metaphyseal lesions in the young cases 1 and 2, it is likely that these processes can take place simultaneously. A significant portion of the blood supply to the epiphysis in foals originates from the perichondrium in the form of transverse vessels with perpendicular branches toward the articular surface and the physis. 13,38,39 The branches enter bone, cartilage or both, depending on location in the epiphysis (Fig. 19). 13,38,39 Whether bacterial destruction of these vessels results in chondronecrosis/osteochondrosis or osteomyelitis could be related to the location of the vessel affected at the time of bacteremia (Figs. 19, 20). The distribution of epiphyseal vessels originating in the perichondrium varies depending on bone and age, 13 which could explain the age-related variations of locations of septic arthritis/osteomyelitis in foals. 12 This potentially also explains why osteomyelitis and hematogenous septic arthritis are such rare events in adult horses, as they do not have perichondral or cartilage canal vessels.
The canals categorized as inflamed patent canals (Fig. 15) had morphologically functional vasculature and were surrounded by normal cartilage, and regressed by chondrification. This illustrates that lesser grades of inflammation can be resolved without lasting damage. The majority of inflamed regressing canals (Fig. 16) seemed to regress by fibrosis instead of chondrification. Whether bacteremia results in septic cartilage lesions likely depend on factors like immuno-competence of the foal 44 combined with infective dose 17,25 and pathogenicity 45 of the bacteria involved.
In this study, bacteria were present in cartilage canals in foals and were associated with ischemic chondronecrosis in both the AECC and the physis. Together with evidence from experimental studies of osteochondrosis and higher incidence of OCD in sepsis-survivors, this establishes sepsis as a plausible cause of some osteochondral lesions in horses. It is recommended that horses with sepsis-related osteochondral lesions may be used for breeding without increasing the prevalence of heritable OCD in the population.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818777786 - Septic Arthritis/Osteomyelitis May Lead to Osteochondrosis-Like Lesions in Foals
Supplemental Material, DS1_VET_10.1177_0300985818777786 for Septic Arthritis/Osteomyelitis May Lead to Osteochondrosis-Like Lesions in Foals by Bjørn Wormstrand, Liv Østevik, Stina Ekman, and Kristin Olstad in Veterinary Pathology
Footnotes
Authors’ Note
The manuscript has been prepared in the Uniform Requirements format.
Acknowledgements
The authors are grateful to the following pathologists for permission to include material from 2 foals as follows: Dr Gjermund Gunnes (case 5) and Prof Arild Espenes (case 7). The authors also are grateful to the Imaging Centre at NMBU for their assistance with the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was funded by grant H1147117/218962 from the Swedish-Norwegian Foundation for Equine Research/Research Council of Norway, with contributions from Norsk Rikstoto and Jordbruksavtalen.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
