Abstract
Osteochondrosis arises as a result of focal failure of the blood supply to growth cartilage. The current aim was to examine the pathogenesis of pseudocysts and true cysts in subchondral bone following failure of the blood supply to the articular-epiphyseal cartilage complex in horses. Cases were recruited based on identification of lesions (n = 17) that were considered likely to progress to or to represent pseudocysts or true cysts in epiphyseal bone in histological sections and included 10 horses ranging in age from 48 days to 5 years old. Cases comprised 3 warmbloods, 3 Standardbreds, 1 Quarter horse and 1 Arabian with spontaneous lesions and 2 Fjord ponies with experimentally induced lesions. Seven lesions consisted of areas of ischemic chondronecrosis and were compatible with pseudocysts. Two lesions were located at intermediate depth in epiphyseal growth cartilage, 2 lesions were located in the ossification front, 2 lesions were located in epiphyseal bone and 1 lesion was located in the metaphyseal growth plate (physis). Ten lesions contained dilated blood vessels and were compatible with true cysts. In 2 lesions the dilated blood vessels were located within the lumina of failed cartilage canals. In the 8 remaining lesions areas of ischemic chondronecrosis were associated with granulation tissue in the subjacent bone and dilated vessels were located within this granulation tissue. Failure of the blood supply and ischemic chondronecrosis can lead to formation of pseudocysts or dilatation of blood vessels and formation of true cysts in the epiphyseal bone of horses.
Keywords
“Subchondral bone cysts” 34,41 or “osseous cyst-like lesions” 37 are an important cause of lameness in horses. 18,46 The diagnosis is most often based on identification of a focal, radiolucent defect in radiographs where the phrase “subchondral bone” is used to indicate that the defect is located near an articular surface. 17,28,37,46 In histologic sections, most cysts are located within epiphyseal trabecular bone, rather than the thin layer of compact bone that is readily identifiable in mature individuals and known as the subchondral bone plate. 21 It might be less ambiguous to refer to the cysts as epiphyseal bone cysts, but the term that is most commonly used in the literature and therefore most universally recognized is subchondral bone cysts. Subchondral bone cysts can arise through a number of different mechanisms 51 and have been observed following septic arthritis, 14,15 osteomyelitis, 15 osteoarthritis, 44 and spontaneously occurring 42,48,52 and experimentally induced trauma. 22,36 They have also been observed in association with osteochondrosis. 41,43 Osteochondrosis is the most common 24,47 of the developmental orthopedic disorders, which are united by the fact that they are initiated in skeletally immature individuals (but may be discovered after skeletal maturity has been reached). 1,17,46 In the case of osteochondrosis in horses, lesions that become persistently present are radiographically detectable by the age of 8 months. 10,11 The remaining developmental orthopedic disorders are not associated with the formation of cysts. 1 The term developmental bone cysts is therefore synonymous with cysts that arise in conjunction with osteochondrosis.
Osteochondrosis is defined as a focal disturbance in the process of endochondral ossification. 40,54 Endochondral ossification occurs within the specialized growth cartilage that is typically found at the metaphyseal growth plate (physis) and the articular-epiphyseal cartilage complex at either end of appendicular long bones. 3,54 The cartilage superficial to an osteochondrosis lesion in epiphyseal growth cartilage can fracture or fissure and give rise to an intra-articular hinged flap or free-floating fragment, known as osteochondrosis dissecans (OCD). 40,54 Alternatively, bone cysts have been observed in association with osteochondrosis lesions in central, weight-bearing regions of the epiphyses in large animal species such as the horse 40,43 and ox. 20,39 In horses, there are 2 main hypotheses for the pathogenesis of bone cysts following osteochondrosis. First, it has been proposed that an OCD fissure may permit influx of pressurized synovial fluid into epiphyseal bone. 34,41 Second, it has been proposed that an osteochondrosis lesion in a weight-bearing region of the epiphysis can give way to infolding of the articular cartilage when loaded. 40,43
In the time since these hypotheses were proposed, the pathogenesis of osteochondrosis in horses has been reexamined in light of evidence for the pathogenesis of osteochondrosis in pigs. 9,55 The results indicated that osteochondrosis arose as a result of focal failure of the blood supply to the epiphyseal growth cartilage, which runs within cartilage canals that are present temporarily during the early phases of growth. 8,32,33 Failure of the blood supply leads to necrosis of chondrocytes; the area of ischemic chondronecrosis later resists replacement by bone and causes the focal delay in endochondral ossification that is characteristic of osteochondrosis. 40,54 This proposed pathogenesis has been reproduced experimentally through surgical transection of blood vessels and monitored until a focal delay in ossification was present in piglets 9,53 and foals. 30 An OCD lesion also occurred in one of the experimental foals. 30 Whether vascular failure can lead to the formation of an epiphyseal bone cyst has, however, not been the subject of any systematic investigation to date.
It is difficult to acquire material for histologic examination that is appropriate for discovering the pathogenesis of developmental bone cysts. Only 7 of 75 publications on equine bone cysts from 1968 to 2013 contain histologic data. 6,19,28,34,35,44,50 It is even more difficult to acquire material representing the early stages that are more likely than the chronic lesions to shed light on the disease process. The horses in 4 of the 7 publications containing histologic data 6,19,34,35 and in 2 experimental studies 22,36 were >1 year old at the time of sample collection. Some of the structures that were present when the lesion was initiated, such as cartilage canal vessels, are no longer present in samples collected at this age. 8 When material from 65 fetuses and foals up to 18 months old was screened in 2 observational studies of osteochondrosis, changes that were compatible with cyst formation were identified in 3 foals (5 %). 8,32 In histologic sections, it is possible to distinguish between lesions that are truly cystic and lesions that are not. A true cyst may be defined as “an abnormal closed cavity in the body, lined by epithelium and containing a liquid or semi-solid material.” 2,4 Epithelium is not a component of normal bone. In the rare cases that epidermoid cysts have been diagnosed in horses, the cyst was located in the distal phalanx, and epithelial cells lining the cyst were suspected of being the result of embryonal misplacement or implantation via a penetrating foot injury. 13,16 Cysts in bone may therefore have to be defined differently from cysts in other tissues. 2 Aneurysmal bone cysts contain blood and may have a bony or fibrous rather than an epithelial lining. 2,45 If a true cyst is defined as an abnormal closed cavity that is separated from the surrounding tissue by a distinct but not necessarily epithelial lining, then 2 of the lesions in the observational studies were compatible with true cysts. 8,32 Pseudocysts have been defined in the same way as true cysts, with the exception that they are not separated from the surrounding tissue by a distinct lining. 2 Provided that an area of ischemic chondronecrosis can be characterized as “semi-solid material,” the third lesion from the observational studies 32 was compatible with a pseudocyst.
After the observational studies were published, lesions that were compatible with a true cyst and pseudocysts were identified in 2 of the 10 foals that were part of the aforementioned experimental study to induce osteochondrosis through transection of vessels. 30 An additional 5 cases have since been identified with lesions that are compatible with pseudocysts or true cysts following vascular failure. Thus, the 10 cases identified to date are presented with the aim of examining the pathogenesis of pseudocysts and true cysts in epiphyseal bone following failure of the blood supply to the articular-epiphyseal cartilage complex in horses.
Materials and Methods
Cases
Cases (n = 10) comprised a combination of previously described
8,32
and newly recruited cases from the following: horses submitted for postmortem examination at the Norwegian School of Veterinary Science, Swedish University of Agricultural Sciences, or the University of Minnesota and foals that were purpose-bred for an approved experimental study to induce osteochondrosis through surgical transection of cartilage canal vessels.
30
To be included, a case had to display lesions that were considered likely to progress to or to represent pseudocysts or true cysts in epiphyseal bone in histologic sections. Horses that were more than 5 years old were excluded to minimize overlap with acquired mechanisms for cyst formation. Included cases were assigned ascending numbers from 1 to 10 based on the age of the animal at the time of sampling for histologic examination (Table 1).
Cases.
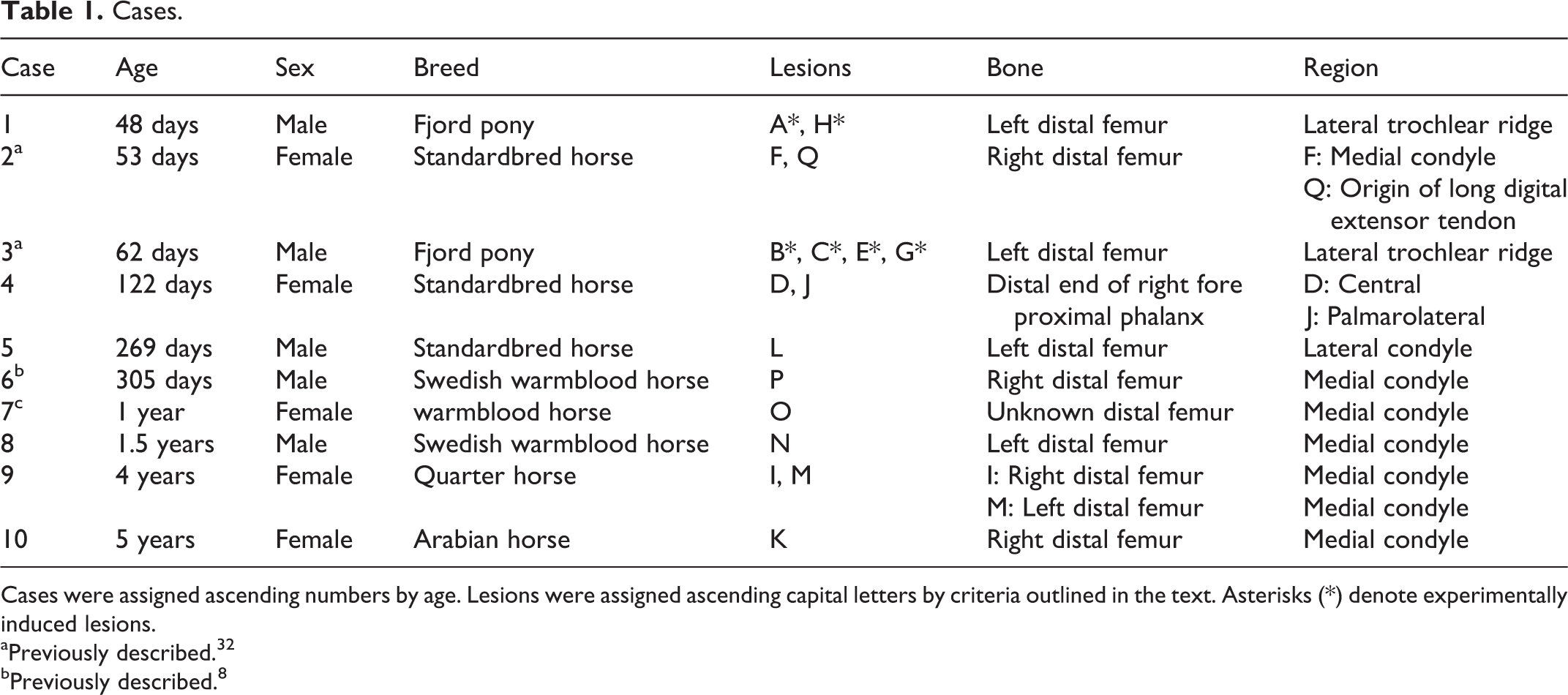
Cases were assigned ascending numbers by age. Lesions were assigned ascending capital letters by criteria outlined in the text. Asterisks (*) denote experimentally induced lesions.
aPreviously described. 32
bPreviously described. 8
Sample Collection
Lesions were identified in the distal femur and distal end of the proximal phalanx. In experimental cases 1 and 3, the operated lateral trochlear ridge was removed from the distal femur as a block of cartilage and bone by sawing with a band saw. In all other cases, the epiphyseal bone was sawed into approximately 5-mm-thick slabs. The distal femur of case 8 was sawed in an oblique plane halfway between a transverse and a frontal plane of section. The remaining distal femora were sawed in a transverse plane of section, and the distal end of the proximal phalanx was sawed in a sagittal plane of section.
The samples were fixed in 4 % phosphate-buffered formaldehyde for 48 hours and decalcified in 10% EDTA. After decalcification, the cartilage/bone blocks from experimental cases 1 and 3 were cut into 5-mm-thick slabs in a transverse plane using a razor blade. In all other cases, any slab region displaying macroscopically visible changes was sampled and trimmed to fit into cassettes measuring 32 × 25 × 5 mm. The samples were paraffin-embedded, sectioned, and stained with hematoxylin and eosin (HE) and toluidine blue for histologic examination.
Immunohistochemical Staining
All slabs from true cyst lesions that had sufficient tissue remaining in the paraffin block were immunostained with a polyclonal rabbit antibody for the endothelial marker von Willebrand factor (vWF, code A0082; Dako, Glostrup, Denmark). A 1:300 dilution was used. The full staining protocol is provided in Supplemental Methods 1.
Case Information
All available clinical and diagnostic imaging records and postmortem requisition forms were collected retrospectively for each case. Experimental cases 1 and 3 had undergone a terminal arterial perfusion procedure to create permanent barium angiograms within the operated hind limb, followed by conventional computed tomographic (CECT) and micro-CT (CEμCT) scanning. 29 Case 7 had been examined ex vivo using micro-radiography of sawed slabs. 8 All remaining cases had been radiographed. Case 9 was CT scanned after euthanasia but before postmortem examination specifically for the purpose of this report. The hip joints were disarticulated, the soft tissues were left intact, and the stifle joints were scanned using a 4-slice helical CT (slice thickness, 0.625 mm; GE LightSpeed 4 Pro, GE Healthcare, Munich, Germany). More detailed information is available in Supplemental Methods 2.
Results
Cases 1 to 10 had a total of 17 lesions that were considered likely to progress to or to represent pseudocysts or true cysts in epiphyseal bone. Lesions were assigned ascending capital letters from A to Q by criteria given below. Asterisks (*) were used to denote experimentally induced lesions.
From Vascular Failure to Pseudocysts in Epiphyseal Bone
Lesions in the pseudocyst group (n = 7, letters A* to G*) were arranged by location in order of increasing relative depth within the sampled tissue.
Lesions A* and B* (Fig. 1) consisted of an area of ischemic chondronecrosis located at intermediate depth in the epiphyseal growth cartilage. Cartilage canals were considered necrotic when vessels were lined by necrotic endothelium and the canal contained necrotic perivascular cells and debris. Chondrocytes were considered necrotic when they displayed cellular shrinkage, cytoplasmic eosinophilia, and karyolysis. The matrix surrounding necrotic chondrocytes stained pale and/or eosinophilic compared with the basophilic matrix surrounding normal chondrocytes. Chondronecrosis centered on necrotic cartilage canals was designated ischemic chondronecrosis.
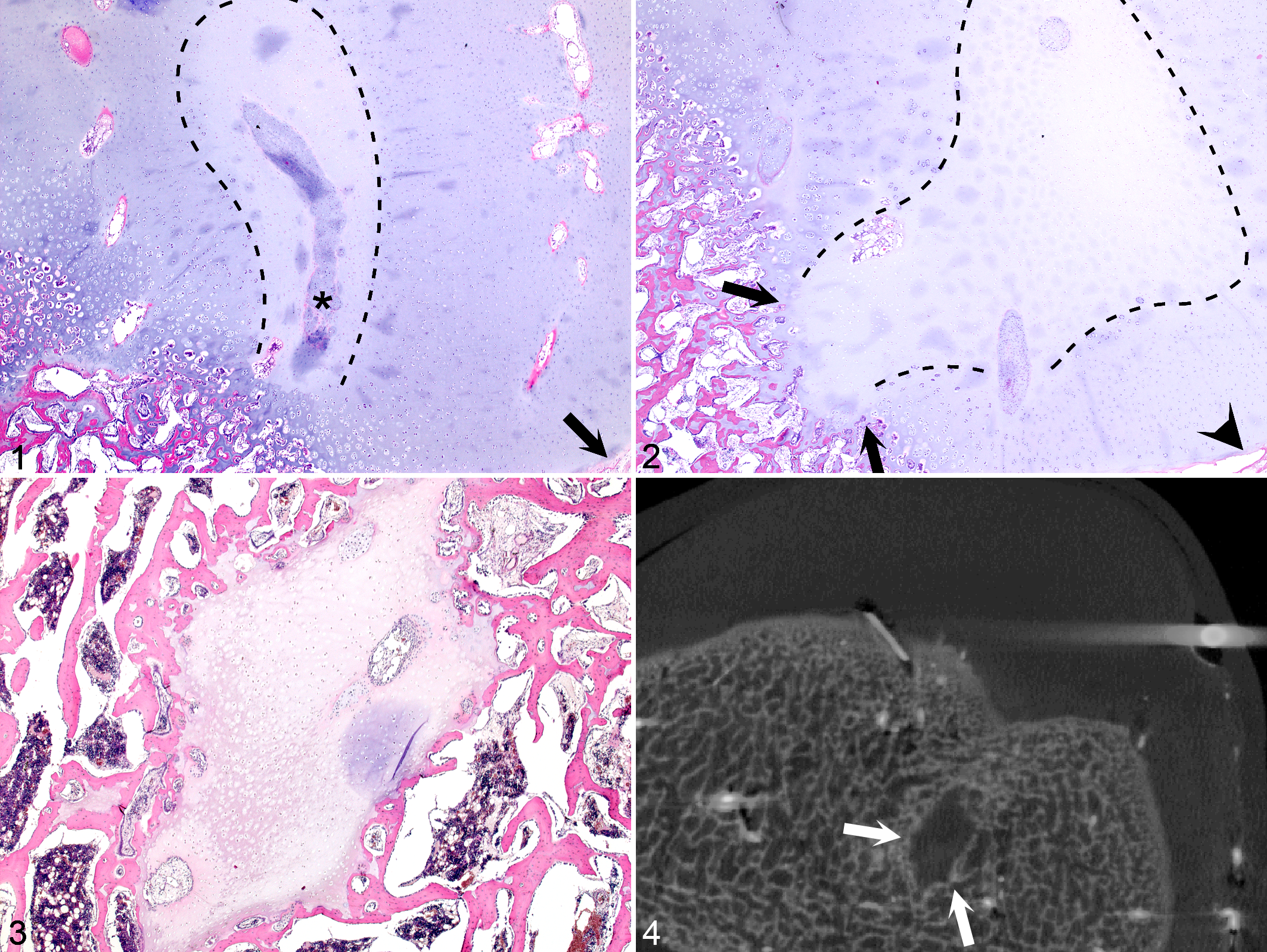
Left distal femur, horse. Orientation: cranial is to the top and left side of the animal is to the right side of each image.
Lesions A* and B* were associated with changes that are recognized secondary responses to ischemic chondronecrosis, including proliferation of adjacent viable chondrocytes and cartilage canal vessels. In addition, some canals within lesions were devoid of vascular lumina and instead contained viable cells displaying chondrocyte-like characteristics. These canals were interpreted to have undergone vessel necrosis, followed by repopulation of the canal with cells from an adjacent viable source, either from adjacent proliferating and invading cartilage canals or from marrow cavities of the advancing ossification front (Fig. 1). The lesions were, however, located superficial to the ossification front, and endochondral ossification was unaffected (Fig. 1).
Lesions C* (Fig. 2) and D consisted of a small and a large area of ischemic chondronecrosis, respectively, both of which were located within the ossification front. Endochondral ossification had advanced in the viable tissue on the lateral but not the deep margins of each lesion. Lesions C* and D therefore caused a focal delay in endochondral ossification.
Lesions E* (Fig. 3) and F consisted of small and large areas of ischemic chondronecrosis, respectively, that were located deeper within the sampled tissue than lesions C* and D. Toward the superficial margin, each lesion remained connected with the overlying cartilage, but in sections that did not include the connection, the area appeared as an “island” of necrotic cartilage completely surrounded by epiphyseal trabecular bone (Figs. 3, 4). One or 2 normal-sized marrow spaces adjacent to lesions E* and F contained fibrous tissue rather than bone marrow. A low number of multinucleated giant cells interpreted as chondroclasts were present along the margins of lesions E* and F. The bone was otherwise normal.
Lesion G* consisted of an area of ischemic chondronecrosis located toward the lateral abaxial aspect of the lateral trochlear ridge within the metaphyseal growth plate (physis). The area disrupted the columnar organization of the proliferative zone of the growth plate, but the majority was located within the resting zone, and the ossification front of the primary, diaphyseal center of ossification was normal.
From Vascular Failure to True Cysts in Epiphyseal Bone
Lesions in the true cyst group (n = 10, letters H* to Q) were arranged in order of increasing complexity and total lesion area, calculated by measuring the width and depth of the lesion in the section where it appeared largest and multiplying width by depth (Table 2).
True Cyst Lesions.
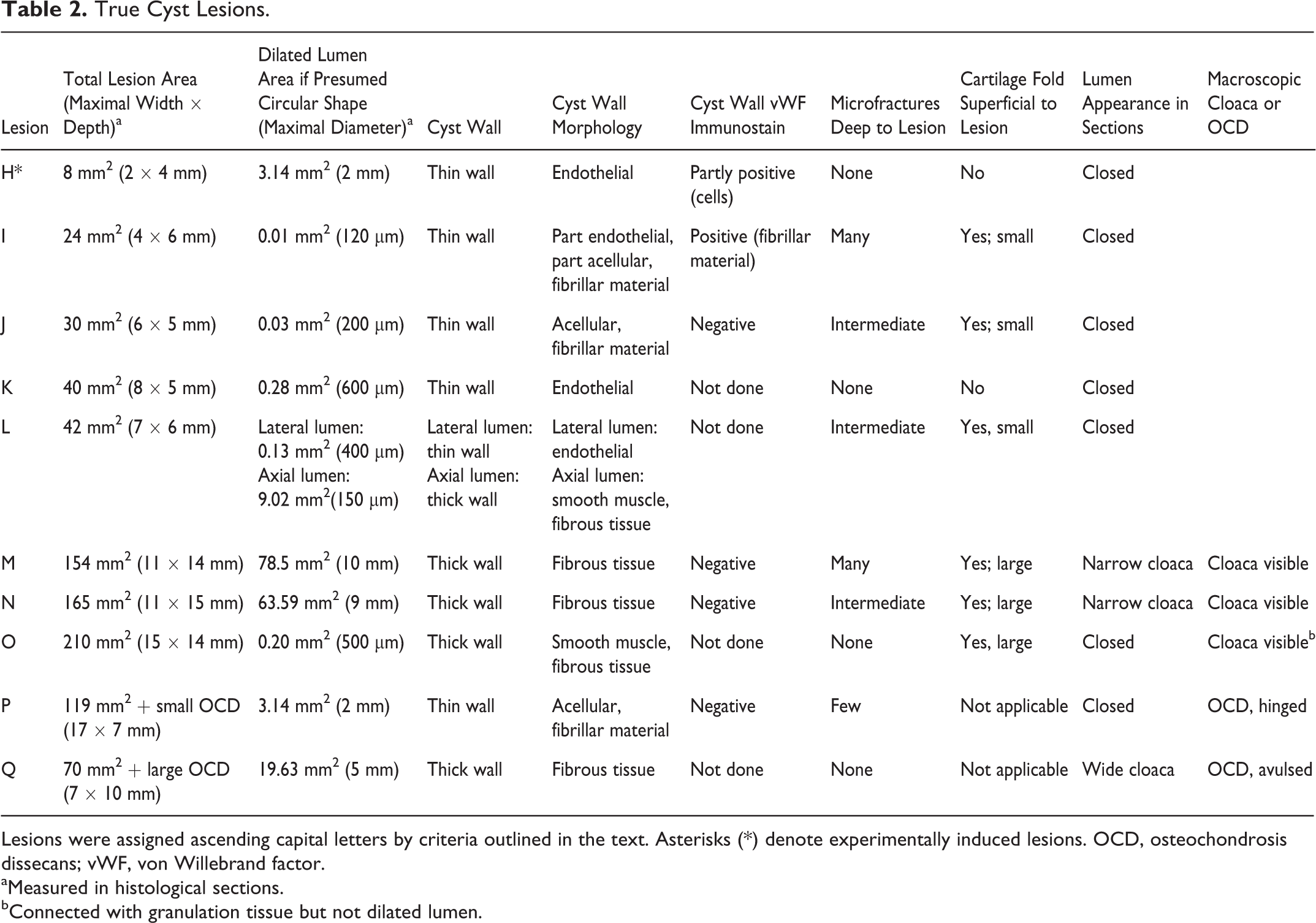
Lesions were assigned ascending capital letters by criteria outlined in the text. Asterisks (*) denote experimentally induced lesions. OCD, osteochondrosis dissecans; vWF, von Willebrand factor.
aMeasured in histological sections.
bConnected with granulation tissue but not dilated lumen.
The hypothesis for the pathogenesis of true cysts was prompted by lesion H*, which consisted of a dilated structure that was morphologically compatible with a vascular lumen within a cartilage canal, deep to the surgical incision that transected the canal (Fig. 5). The cytoplasm of some of the cells that lined the dilated lumen was immunopositive for vWF (Fig. 6). The lesion was of 35 days’ duration, and the portion of the cartilage canal containing the dilated lumen was surrounded by epiphyseal bone (Fig. 5).
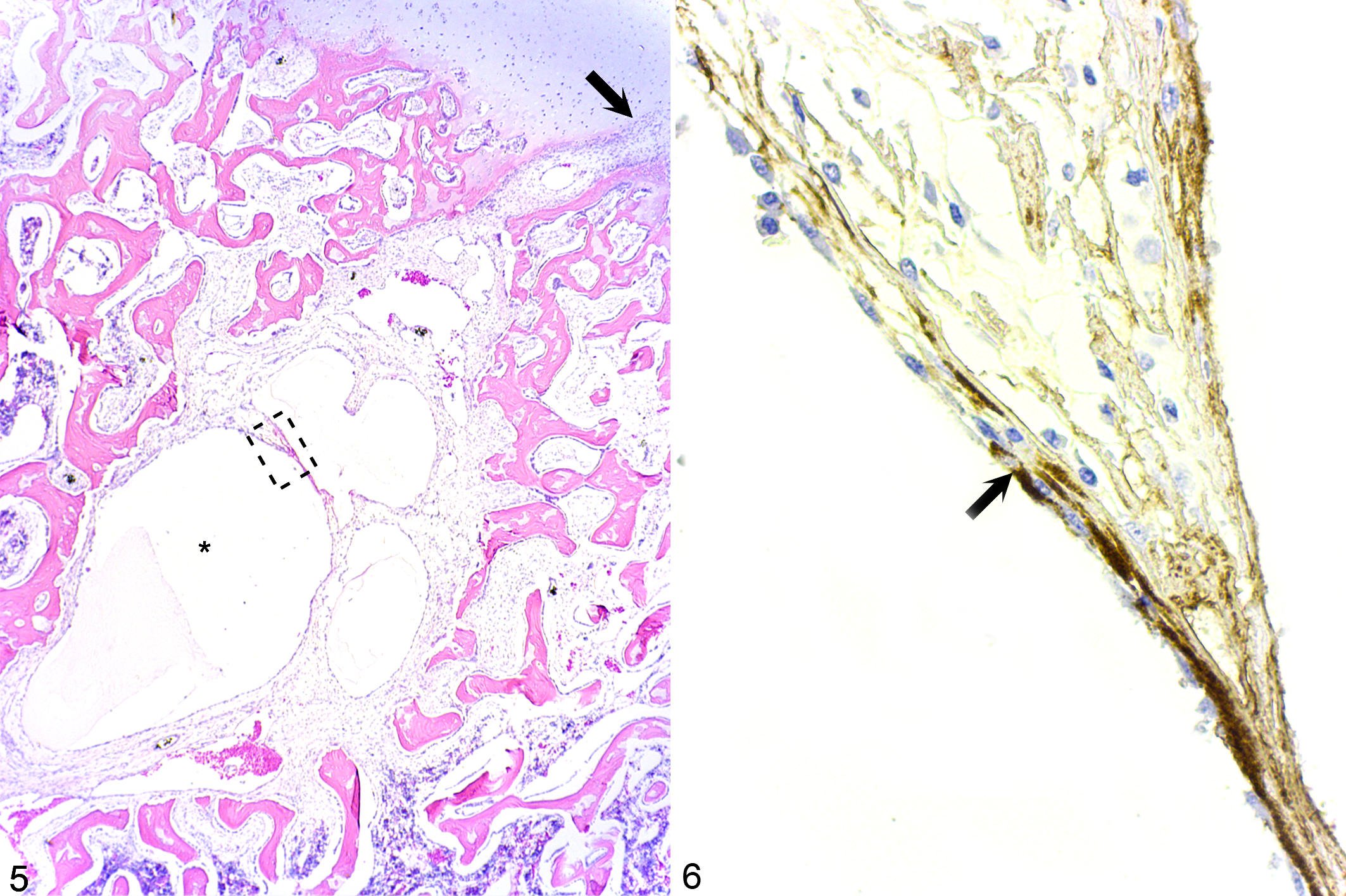
Left distal femur, horse. Orientation: cranial is to the top and left side of the animal is to the right side of each image.
All remaining true cyst lesions from I to Q contained small areas of ischemic chondronecrosis located within the ossification front and associated with variably sized areas of fibrovascular granulation tissue in the subjacent bone (Fig. 7). Superficially within the granulation tissue, there were additional small areas of necrotic cartilage. All granulation tissue contained numerous capillaries. In some areas, 3 to 5 larger vessels were grouped together in vascular bundles. In one such bundle located deep within the granulation tissue of lesion I and away from any central nutrient artery, 2 to 3 vascular lumina subjectively appeared dilated compared with any other vascular lumen within the section. The maximal diameter of the largest lumen was 120 μm. Within the granulation tissue of lesions J to O, there were similar dilated lumina with a maximal diameter of 200 μm (Fig. 7) to 10 mm (Table 2).
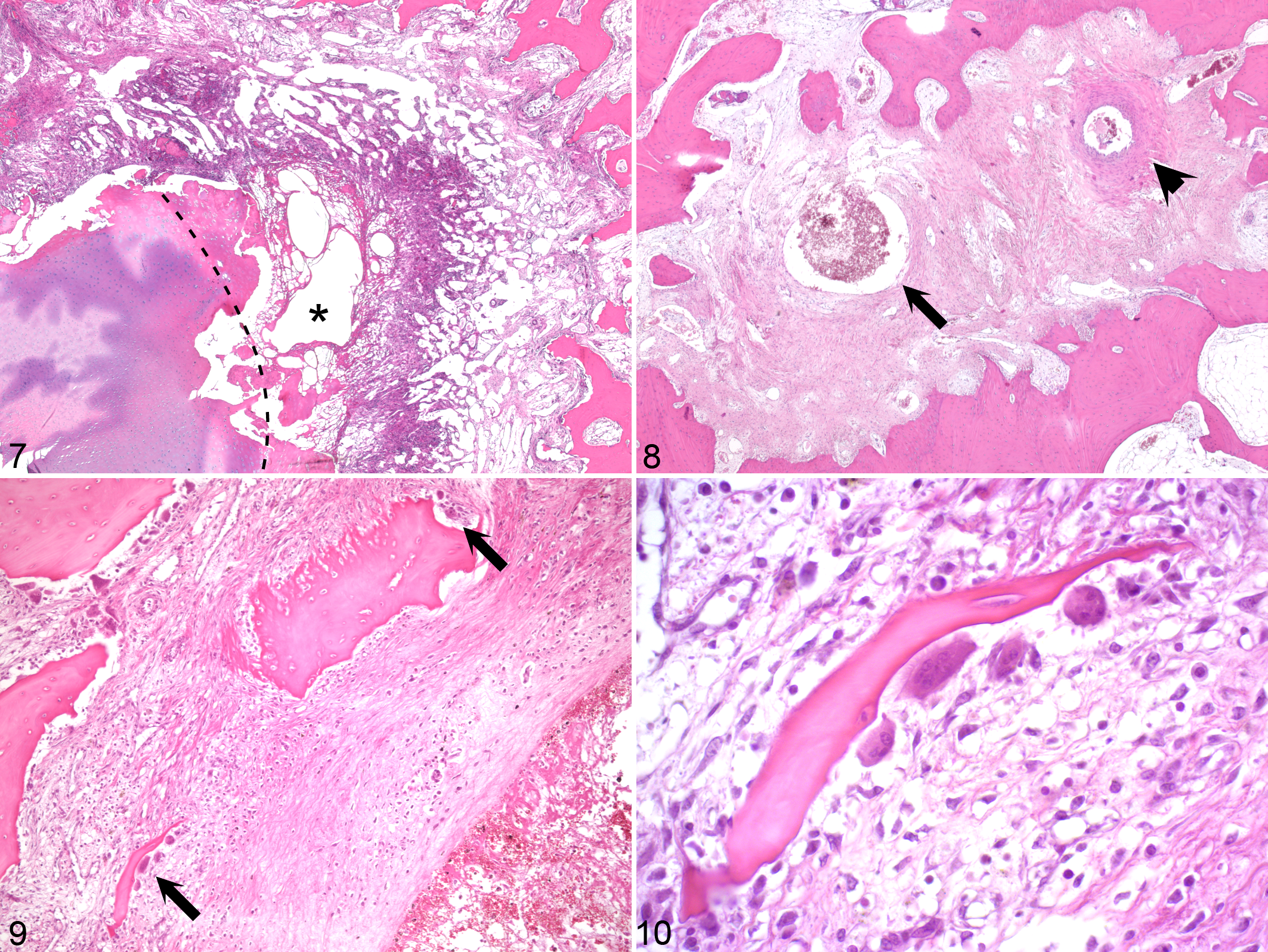
Dilated lumina could be characterized as having either a thin or a thick wall (Fig. 8, Table 2). Thin walls were composed entirely of flattened, endothelial-like cells, entirely of acellular, fibrillar material or a combination of both, and were morphologically compatible with being of venous origin. Thick walls consisted of concentrically arranged, spindle-shaped cells. Cells in the innermost layers of the thick-walled dilated lumina in lesions L (Fig. 8) and O were slender and resembled smooth muscle cells. All cells in the thick walls of lesions M and N were plump and resembled fibroblasts (Table 2).
In sections cut for immunostaining, the 120-μm diameter lumen in lesion I contained only scant shrunken, dark-staining endothelial cells presumed to be degenerate. The fibrillar material that otherwise demarcated the lumen stained similar to vWF immunopositive cells in other locations within the section. The walls of the larger dilated lumina in lesions J, M–N, and P were immunonegative for vWF (Table 2).
In addition to dilated lumina, granulation tissue commonly contained accumulations of chondrocyte-like cells surrounded by small amounts of pale basophilic, amorphous material interpreted as newly formed cartilage. In some cases, granulation tissue also contained osteoblast-like cells surrounded by intensely eosinophilic, amorphous, osteoid-like material interpreted as intramembranous ossification of granulation tissue. In lesion I, the dilated lumen and part of the granulation tissue were enclosed within an outer, mature fibrous tissue capsule. In lesion M, the dilated lumen and all granulation tissue were enclosed within an outer rim consisting of newly formed cartilage.
A variable number of osteoclasts were present on the margins of all true cyst lesions. In lesions I–J and L–N, there were multiple discontinuous trabeculae and small fragments of necrotic bone, interpreted as microfractures (Table 2). The fracture fragments were smooth or had scalloped edges in areas where they were in contact with osteoclasts (Figs. 9, 10).
Chondrocytes within the thickened cartilage superficial to areas of ischemic chondronecrosis in lesions I–J and L–O were flattened in the lateral direction and elongated in the superficial-to-deep direction of the sample, suggestive of folding of the cartilage into the subjacent granulation tissue. Cartilage folds were small when located superficial to the 24- to 42-mm2 area lesions I–J and L and considerably larger when located superficial to the >150- mm2 area lesions M–O (Table 2).
In the smallest lesions I–L, dilated lumina formed closed circles in histologic sections, and the overlying cartilage surface was intact. In the larger lesions M and N, the interior of the dilated lumen communicated with the adjacent joint space through a narrow channel or cloaca. The macroscopically visible cloaca in lesions O and P communicated with the granulation tissue but was not visibly connected with the dilated lumen in histologic sections (Table 2).
Lesions P and Q were located within the granulation tissue bed of a small, hinged OCD flap and a large, avulsed OCD fragment, respectively. The dilated lumen in lesion P had a thin, fibrillar wall and was morphologically compatible with remnants of a necrotic cartilage canal vessel (Fig. 10 in Olstad et al 32 ). The dilated lumen in lesion Q consisted of thick, mature fibrous tissue and communicated with the joint space through a wide cloaca.
Were Lesions Detectable With Diagnostic Imaging?
The experimental lesions and lesion O were detectable with the techniques that had been applied ex vivo (Fig. 4, Suppl. Table S1). The remaining 10 spontaneous lesions were detectable as variably radiolucent defects with planar radiography (Suppl. Table S1). Plain CT of the true cyst lesions I and M in case 9 enabled visualization of internal subdivision of each lesion by septa of cortical bone opacity. In lesion I, the contents of 1 subdivision was of an intermediate bone opacity that was denser than trabecular bone but not as dense as cortical bone (Fig. 11) and corresponded to intramembranous ossification of granulation tissue in histologic sections.
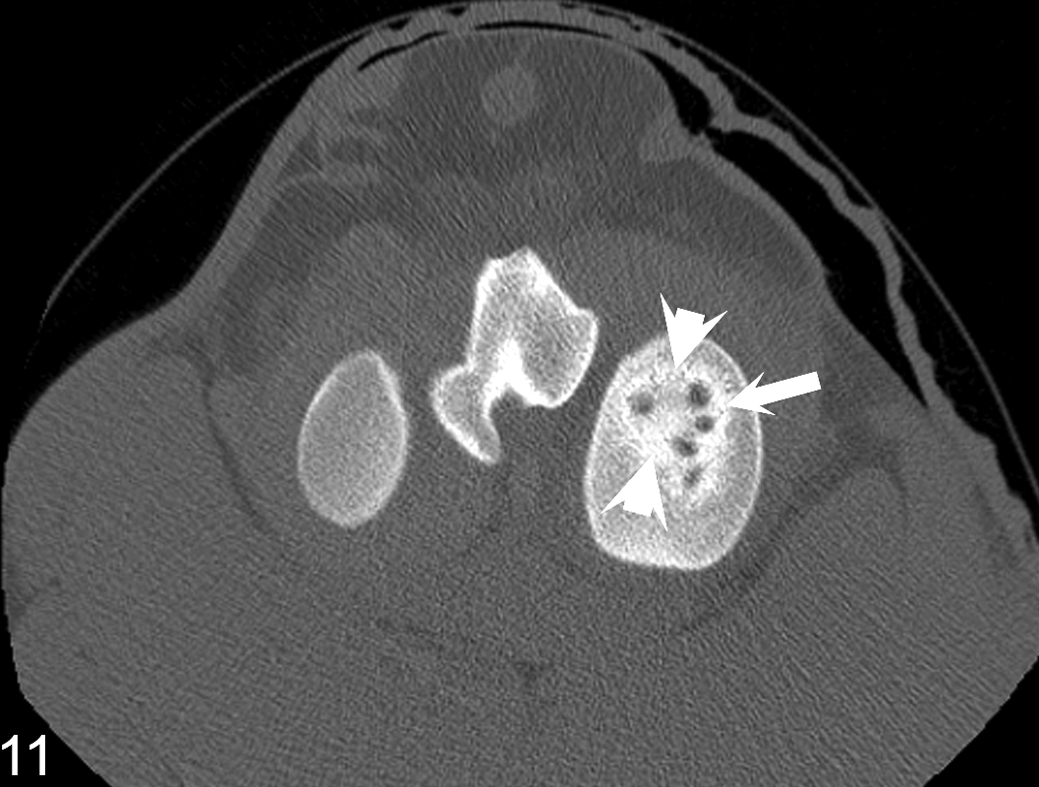
Right distal femur, horse. Orientation: cranial is to the top and left side of the animal is to the right side of each image. Medial femoral condyle, case 9, lesion I. True cyst lesion I is subdivided by internal septa of cortical bone opacity (arrow). The contents of most subdivisions are uniformly radiolucent. The contents of 1 subdivision are of an intermediate bone opacity that is denser than trabecular bone but not as dense as cortical bone (between arrowheads) and corresponded to intramembranous ossification of granulation tissue in histologic sections. Transverse conventional computed tomography slice image.
Were Lesions Associated With Lameness?
Lameness had been noted in 7 of the 10 cases (Suppl. Table S2). In 2 cases, lameness had been localized to the relevant joint by intra-articular anesthesia. Case 4 responded to anesthesia of the pastern joint that contained both pseudocyst lesion D and true cyst lesion J. Case 9 responded to anesthesia of the left hind stifle joint that contained the larger true cyst lesion M and did not show lameness on the right hindlimb with the smaller true cyst lesion I during the time that the left stifle joint was anesthetized. Lameness in cases 6, 8, and 10, which contained true cyst lesions P, M, and K, respectively, had not been localized with anesthesia, but the cysts were the only lesions detected at postmortem examination.
Discussion
The current aim was to examine the pathogenesis of pseudocysts and true cysts in epiphyseal bone following failure of the blood supply to epiphyseal growth cartilage in horses. The main findings are summarized in Fig. 12.
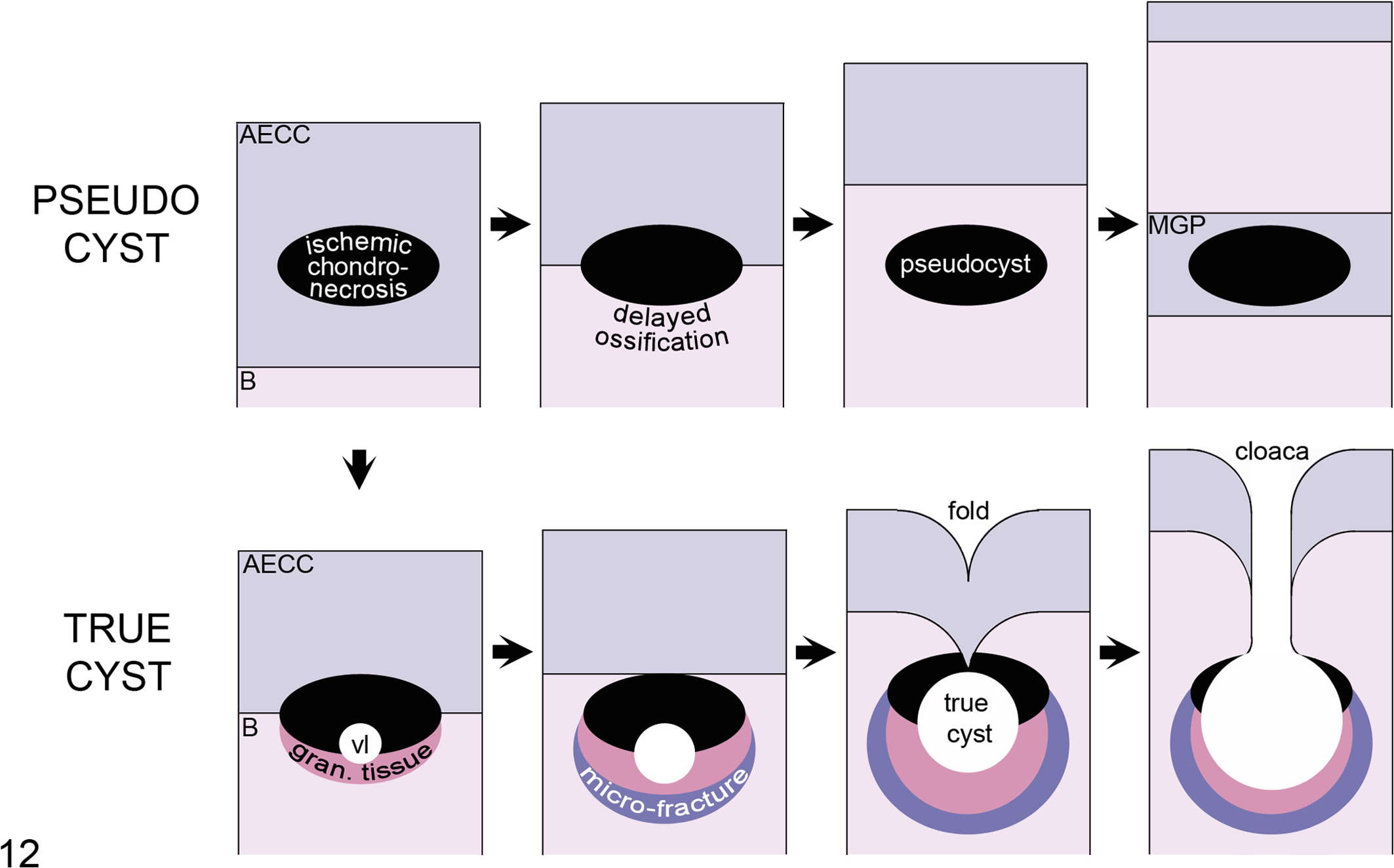
Schematic illustration of the proposed pathogeneses for pseudocysts and true cysts in epiphyseal trabecular bone (B) following vascular failure in the articular-epiphyseal cartilage complex (AECC). PSEUDOCYST: The earliest lesion following vascular failure is an area of ischemic chondronecrosis at intermediate depth of the AECC. Due to continued ossification within the viable tissue on the margins, the lesion becomes surrounded by the ossification front, where it causes a focal delay in endochondral ossification or osteochondrosis. When it has become completely surrounded, the area forms a pseudocyst in the epiphyseal trabecular bone. In very young individuals, the area of ischemic chondronecrosis can become surrounded by the metaphyseal growth plate (MGP). TRUE CYST: The first step in the formation of a true cyst is dilatation of a blood vessel (vl) within the area of ischemic chondronecrosis or in the associated granulation tissue (gran. tissue). Trabecular microfractures occur on the margins of the lesion when loaded. The lesion increases in size and gives way to infolding of cartilage. The folded cartilage can eventually break and permit communication between the cyst interior and joint space through a cloaca.
Pseudocysts
Five of the 7 pseudocyst lesions represented surgically induced rather than spontaneously occurring lesions. The experimental lesions were included because of the added information they provide by virtue of being of known history and duration. The lesions were identical to previously described spontaneously occurring lesions of osteochondrosis in foals. 8,32
Experimental studies in both foals 30 and piglets 9,53 confirm that the earliest lesion following surgical transection of the blood supply is an area of ischemic chondronecrosis located at intermediate depth of the epiphyseal growth cartilage (lesions A* and B*; Fig. 12). Chondrocytes in the most superficial and deep layers survive, presumably due to diffusion from adjacent synovial fluid or intact vessels in subjacent bone. 9,30 With increasing postoperative time, lesions are found progressively deeper within the sampled tissue (lesions C* and D). This is considered the result of continued endochondral ossification within the viable tissues on the margins of the lesion. 9,27,30,53
Cases 1 and 3 had multiple lesions that were of the same duration and yet were located at different depths of the sampled tissue. This was a result of the chosen experimental technique (fully accounted for in Olstad et al 30 ). Vessels were transected in a total of 4 separate locations. Lesions B* and C* were located superficial to cranial incisions, whereas lesions E* and G* were located between cranial and caudolateral incisions. Lesions A* and B* originated from the middle third and lesions H* and C* from the proximal third of the lateral trochlear ridge, where the rate of advancement of the ossification front was faster. 30 Spontaneous lesions sometimes consist of multiple adjacent areas or defects in sections 28,32 or radiographs. 17 Adjacent areas were located at different depths in the transverse plane in 1 foal, indicating that the rate of ossification also was faster toward the axial compared with the cranial aspect of the lateral trochlear ridge. 32 Indeed, multilobulated lesions are so common (83%) in the distal femur of piglets that they may be pathognomonic for osteochondrosis in this site. 31
Once it has been overtaken by the ossification front, the area of ischemic chondronecrosis can constitute a pseudocyst in epiphyseal bone (lesions E* and F). 19,46 Vascular failure results in lesions that are morphologically distinct in epiphyseal and metaphyseal (physeal) growth cartilage. 54 Lesion G* represented ischemic necrosis of epiphyseal growth cartilage and provided important new information because it has never been suggested that lesions in epiphyseal growth cartilage can be overtaken by the metaphyseal growth plate (physis), although cysts have been noted clinically in association with metaphyseal growth plates (physes). 5,7,46 The morphology of lesion G* suggests that physeal cysts may be the result of vascular failure arising on the abaxial aspect of an epiphysis in foals in which there is sufficient remaining growth for the metaphyseal growth plate to advance beyond the lesion.
True Cysts
Our interpretation of the current observed changes is that the first step in the pathogenesis of true cysts following vascular failure is abnormal dilatation of 1 or more blood vessels (Fig. 11). The interpretation was prompted by the compelling morphologies of lesions H* and P (Fig. 10 in Olstad et al 32 ), which were compatible with dilated blood vessels within the lumina of previously patent cartilage canals. Identical dilatation of a blood vessel has since been observed in a spontaneous lesion without experimental incision (as in lesion H*) or OCD (as in lesion P) in the distal femur of a piglet (Fig. 5 in Olstad et al 31 ). Cells lining the dilated lumen in lesion H* were immunopositive for vWF, as was some of the fibrillar material lining the lumen in lesion I. The fibrillar positivity could represent artifact or vWF bound to subendothelial connective tissue in a damaged, denuded vessel. 26,38 The fact that the lining of the dilated lumina in larger, presumably more chronic lesions was immunonegative for vWF need not rule out the possibility of the lumina being of vascular origin as with time, endothelial cells could de-differentiate, 23 cease production of vWF, or be lost.
While the appearance of lesions H* and P was compatible with dilatation of a vessel directly within a failed cartilage canal, the morphology of the 8 remaining true cyst lesions was compatible with dilatation of vessels within lesion-associated granulation tissue. The risk that a vessel will become dilated therefore appears increased in ischemic chondronecrosis that is associated with granulation tissue compared with ischemic chondronecrosis alone. The factors that govern the amount of granulation tissue that is formed in response to a particular lesion are not known. Pseudocyst lesion F was located in a weight-bearing region and was larger than 4 of the true cyst lesions (I–L), and thus the amount of granulation tissue that forms may be independent of lesion size and loading. An alternative suggestion is that granulation tissue formation may be the result of individual predisposition in terms of animal age/maturity, immune status, or genetic characteristics. 28
In the current lesions, trabecular microfractures, cartilage infolding, and cloacae were more common in large than in small lesions and were therefore interpreted to occur after vessel dilatation and formation of granulation tissue. Material testing studies of cartilage/bone cylinders from healthy, adult horse joints demonstrate that cartilage will absorb 7 times more energy than bone before breaking. 25 When a lesion is exposed to load, we therefore believe that trabecular microfractures will occur before cartilage infolding. Trabecular microfractures and osteoclastic resorption of necrotic fracture fragments will result in loss of bone from the periphery of lesions such as current lesion I. In addition, in vitro studies have shown that fibrous tissue from equine bone cysts can produce enzymes and cytokines that are associated with bone resorption. 49,50 Both microfractures and enzyme production may result in progressive enlargement of the lesion until it can no longer support the overlying articular cartilage and gives way to infolding. The small cartilage folds superficial to the current lesions I–J and L confirm that cartilage will withstand some deformation 25 but may eventually break because the interior of lesions M–O communicated with the joint space by way of cloacae running through the large cartilage folds superficial to each of these lesions.
The current hypothesis for the pathogenesis of true cysts may appear to disagree with the hypotheses presented at the beginning of this article that include pressurized fluid influx 34,41 or infolding of cartilage. 40,43 In fact, there is broad agreement on the character of observed changes 19,28,34 and contrast only in the interpretation of the sequence in which they occur. According to the current hypothesis, true cyst formation relies on the presence of blood vessels and is independent of cartilage infolding or influx of pressurized fluid. This explains how cysts may be found a distance of up to 2 cm away from 37 and with no evidence of ever having communicated with the joint space. 34 In addition, the suggestion that the cloaca forms late rather than early in the pathogenesis explains why many cyst cases present with sudden-onset lameness when the causal lesion is chronic. 17,28
In the current study, 2 cases had both a cyst and an OCD lesion. In lesion P, the OCD did not communicate with the cyst, and fluid could neither have been trapped nor pressurized through the wide cloaca in lesion Q. Despite the fact that the current study was cross-sectional, it is highly unlikely that the OCD was the cause of the cysts in these 2 cases. Based on lesions P and Q, we tentatively suggest that it may be the underlying osteochondrosis disease process that leads to cyst formation rather than the OCD. 40,54 The integrity of the tissue may be more compromised in an underlying cystic compared with a noncystic osteochondrosis lesion; thus, the cyst may predispose for the OCD lesion rather than the reverse.
Several of the lesions were detectable in radiographs, and some were associated with lameness. The relationship between pseudocysts and lameness localized with anesthesia could not be studied independently from true cysts in the current cases, but difficulty in rising and a stiff hindlimb gait were previously noted in foals in which ischemic chondronecrosis was the only joint lesion detected postmortem. 32 Treatment may therefore be indicated for both pseudocyst and true cyst lesions. In pseudocyst lesions, there was no lining to debride or cavity in which to inject into, so pseudocysts may have to be treated differently from true cysts. 12 Different treatment is possible only if one can distinguish between pseudocysts and true cysts in a clinical setting. The observation that a tomographic modality such as CT 14,16 revealed more information about lesion interior than planar radiography in case 9 is promising in this respect and could be explored further in the future.
Failure of the blood supply to epiphyseal growth cartilage and ischemic chondronecrosis can lead to the formation of pseudocysts in epiphyseal bone that are radiographically detectable. Vascular failure and ischemic chondronecrosis can lead to dilatation of blood vessels and the formation of true cysts in epiphyseal bone that are radiographically detectable and can be associated with lameness.
Footnotes
Acknowledgements
We thank Beate Hillman for preparation of sections, Paula Ohern for immunostaining, Bert Masschaele for μCT scanning, and Jørgen Kongsro and Lena Stenhaug for CT scanning.
Author Contribution
Conception or design: KO, LØ, CSC, SE. Data acquisition, analysis, or interpretation: KO, LØ, CSC, SE. Drafting the manuscript: KO, LØ. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
