Abstract
Lyme neuroborreliosis—characterized as chronic, necrosuppurative to nonsuppurative, perivascular to diffuse meningoradiculoneuritis—was diagnosed in 2 horses with progressive neurologic disease. In 1 horse, Borrelia burgdorferi sensu stricto was identified by polymerase chain reaction amplification of B burgdorferi sensu stricto–specific gene targets (ospA, ospC, flaB, dbpA, arp). Highest spirochetal burdens were in tissues with inflammation, including spinal cord, muscle, and joint capsule. Sequence analysis of ospA, ospC, and flaB revealed 99.9% sequence identity to the respective genes in B burgdorferi strain 297, an isolate from a human case of neuroborreliosis. In both horses, spirochetes were visualized in affected tissues with Steiner silver impregnation and by immunohistochemistry, predominantly within the dense collagenous tissue of the dura mater and leptomeninges.
Lyme disease is a systemic disorder caused by spirochetes belonging to the broad group Borrelia burgdorferi sensu lato, including B burgdorferi sensu stricto (ss), B garinii, and B afzelii. 8,16 The natural disease exists wherever permissive reservoir hosts and appropriate tick vectors (Ixodes sp) coexist and intermingle with susceptible hosts of wide species diversity, including humans, dogs, and horses.3,4,8,9,11,12,15,16 In humans and in experimental animal models, persistent infection progresses from early local infection to randomly disseminated infection, resulting in variable clinical manifestations.2,8 Early infection, associated with the erythema migrans rash, is best recognized. Late-stage infection can be associated with oligoarthritis, carditis, and neurologic disease.
Lyme neuroborreliosis is the neurologic manifestation of Lyme disease potentially caused by any of the 3 aforementioned B burgdorferi sensu lato genospecies.15,16 At least 2 geographically distinct syndromes have been defined. 15 In the United States, Lyme neuroborreliosis is uncommon, clinically characterized by vague neurologic symptoms, and described as subacute meningitis. 15 The disease develops within weeks to months of the initial erythema migrans rash and may be accompanied by arthritis. Nerve root or secondary parenchymal and spinal cord involvement is rare. In Europe, neuroborreliosis is more common, and it usually presents as a painful radiculitis, referred to as Bannwarth syndrome. Few cases are associated with an erythema migrans rash or arthritis, and more cases progress to chronic encephalomyeloradiculitis.11,15 These considerable clinicopathologic differences have been speculatively attributed to the causative B burgdorferi genospecies. In the United States, almost all Lyme disease cases are caused by B burgdorferi ss. In Europe, cases are caused by B burgdorferi ss, B garinii, or B afzelii.
In horses, Lyme borreliosis is similarly systemic, manifesting as arthritis, uveitis, and, less commonly, encephalitis.3,4,9 The few cases of equine central nervous system disease associated with B burgdorferi have been documented by microbiological techniques (culture and direct immunofluorescence), seropositivity, and polymerase chain reaction (PCR) assays that target Borrelia-specific and nonspecific genes.4,9 Only one previously published case included a description of the neuropathology, 9 and in no case have spirochetes been identified in tissue sections. Thus, the current understanding of the host inflammatory reaction, localization of lesions, and distribution of the causative agent B burgdorferi in equine Lyme neuroborreliosis is limited. This article reports the histopathology, lesion distribution, tissue burden of spirochetes, strain of B burgdorferi involved, and distribution of spirochetes within the central nervous system.
Case No. 1 was a 9-year-old thoroughbred gelding with a 1-year history of progressive neurologic disease. Clinical signs began when the horse was stabled in Pennington, New Jersey, and they continued after a move to Livermore, California. Signs were initially described as pain along the neck and back, with lumbar hyperesthesia, lethargy, inappetence, and weight loss. At that time, another horse pastured with the gelding developed shifting lameness. Based on the pasture mate’s signs, both horses were tested for Lyme disease by serology, 3 times over 9 months at 2 laboratories. Case No. 1 was consistently seronegative, whereas the pasture mate was seropositive with a falling convalescent titer and resolution of clinical signs after 6 weeks' treatment with doxycycline. Five months after the onset of clinical signs, case No. 1 developed unilateral conjunctivitis and uveitis. Ten months later, there was progression to ataxia, facial paralysis, front limb hypermetria, proprioceptive deficits (circumduction, hind limb interference), and continued lumbar pain. Based on the neurologic signs and serologic evidence of exposure to Sarcocystis neurona, treatment for equine protozoal myeloencephalitis with successive rounds of ponazuril (Marquis, Bayer Animal Health, Shawnee Mission, Kansas) and sulfamethoxazole–trimethoprim was initiated, with flunixin meglumine (Banamine, Schering-Plough Animal Health Corp, Union, New Jersey) as needed. The response was equivocal. Clinical signs continued to worsen with development of abnormal mentation (staring into space, spooking without cause, severe depression) and a head and neck tremor. Case No. 1 was euthanized after being found down and unable to rise.
Case No. 2 was an 18-year-old quarter horse mare with a 4-year history of episodic muscle atrophy that waxed and waned in various muscles with mechanical gait deficits and mild hyperesthesia. The mare was stabled in Rancho Santa Fe, California; it was trail ridden in a heavily wooded area; and it had never left California. Based on the clinical signs and equivocal cerebrospinal fluid seropositivity to S neurona (Neogen Corp, Lexington, Kentucky), the horse was treated for equine protozoal myeloencephalitis with a course of sulfadiazine–pyrimethamine for 120 days, 2 successive courses of toltrazuril (Baycox for poultry, Bayer Animal Health, Toronto, Ontario, Canada) for 30 days, and one course of toltrazuril for 45 days. No response to therapy was observed. Subsequently, in the third year of clinical disease, the horse was reevaluated and diagnosed with a peripheral neuropathy, unlikely to be equine protozoal myeloencephalitis. At that evaluation, muscle atrophy was evident over the left thoracic and lumbar spine and in the left gluteal (Fig. 1), right pectoral, and right semitendinosus muscles. A left facial peripheral neuropathy was also observed. Cytology of cerebrospinal fluid was recommended but not performed, because of the mare’s violent response to the initially performed procedure. Oral dexamethasone (20 mg) was instituted with initial muscle mass recovery after 3 weeks of treatment and complete resolution over multiple months. After 6 months of therapy, dexamethasone was withdrawn because of laminitis development. The muscle atrophy returned, and in the fourth year, clinical disease progressed to ataxia, incoordination, and abnormal mentation (obtundation and failure to recognize the owner), at which time euthanasia was elected.
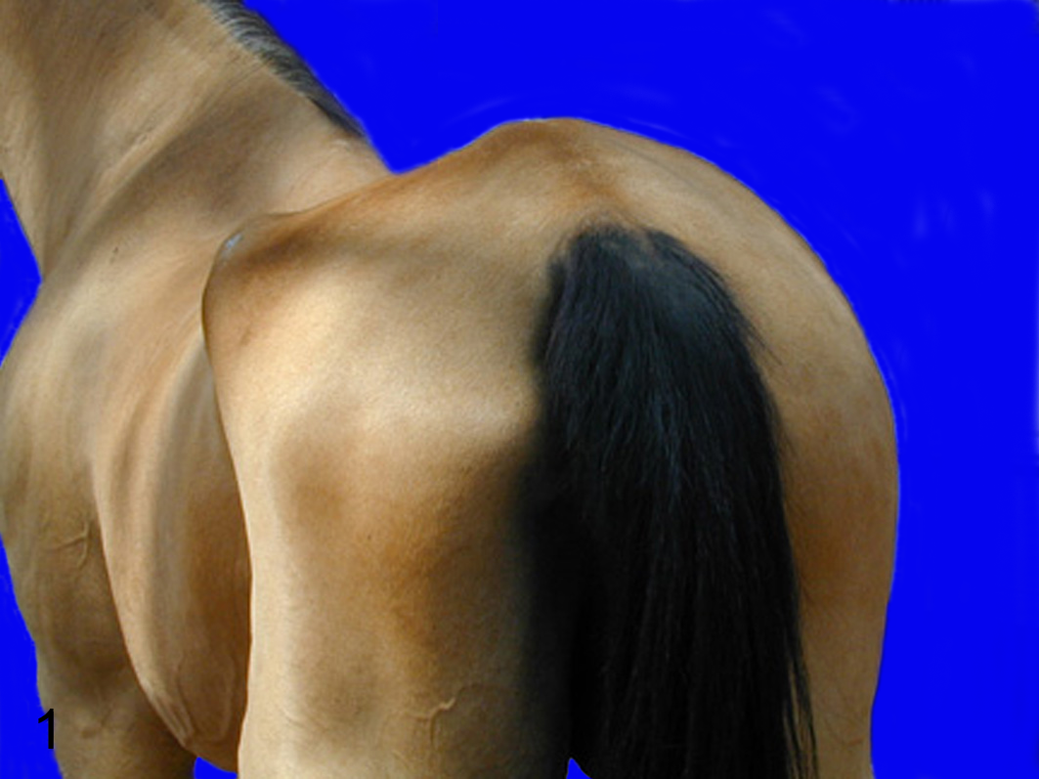
Horse No. 2. Atrophy of the left gluteal and flank musculature.
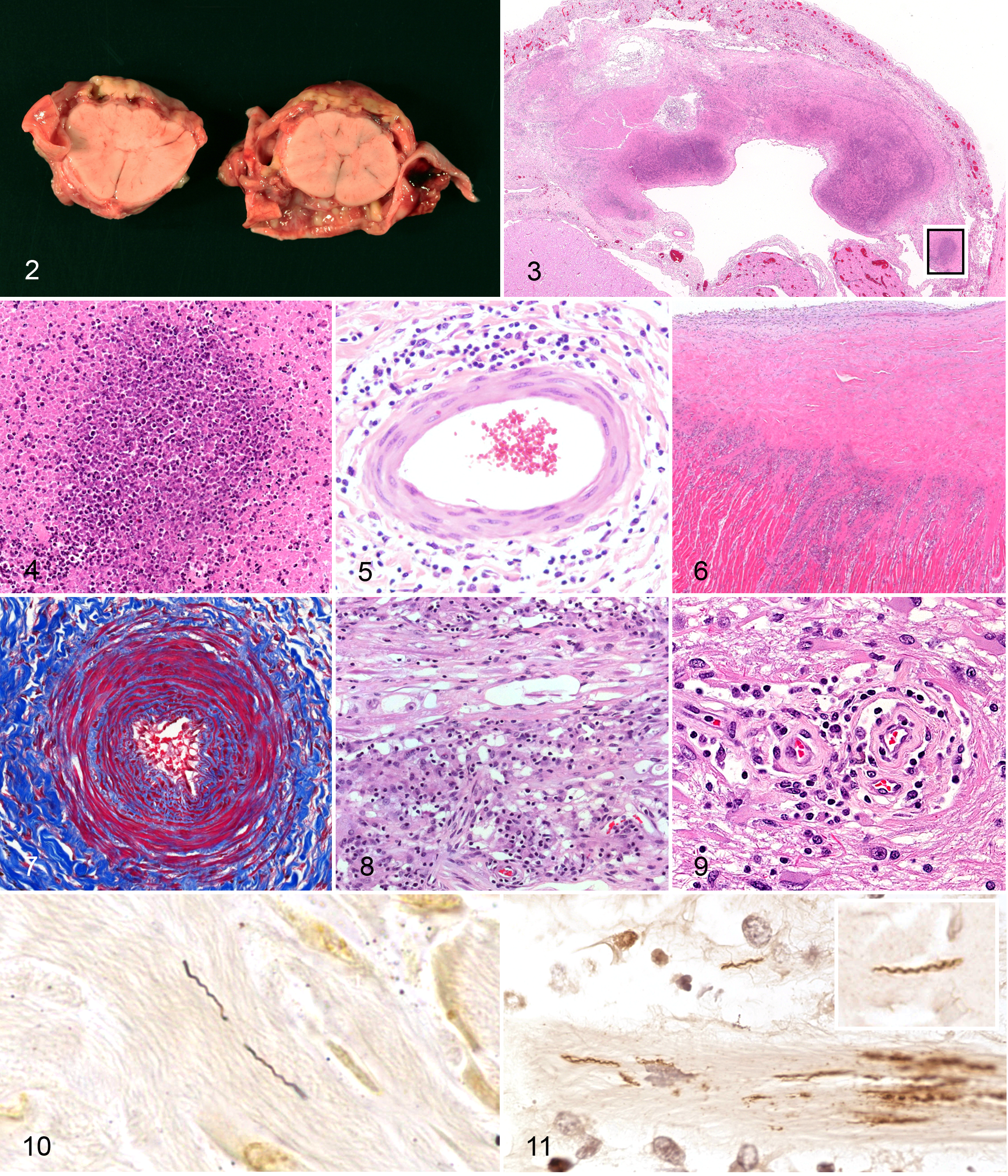
Both horses were submitted to the California Animal Health and Food Safety Laboratory System for necropsy at the Davis and San Bernardino branches, respectively. At necropsy, fibrous adhesions between the leptomeninges and dura mater prevented easy removal of the brain in both horses. The meninges were 1 to 2 mm thick and opaque, with widespread hemorrhages and subdural pale yellow to tan purulent deposits. Similar changes were in the cervical spinal cord (Fig. 2). Case No. 1 had extensive subcutaneous hemorrhage and edema in the distal forelimbs and right hind limb, as well as moderate serous pleural effusion. Case No. 2 had subcutaneous fibrosis that thickened the circumference of the distal aspect of the left hind limb, focal pleural fibrosis in the dorsal aspect of the caudal lung lobes, and bilateral asymmetric hindquarter muscle atrophy.
Tissue Samples
Tissue samples were immersion-fixed in 10% neutral-buffered formalin, routinely processed, and embedded in paraffin. Fresh tissues from case No. 1 were stored at 4°C, including lung, pleura, pleural fluid, brain, spinal cord, skeletal muscle, and joint capsule.
Histopathology, Histochemistry, and Immunohistochemistry
Tissues were sectioned 4 to 5 μm thick and stained with hematoxylin and eosin. Select sections of brain, spinal cord, heart, and skeletal muscle were evaluated with Gram stain, periodic acid–Schiff, acid–fast, Gimenez, Gomori methenamine silver, and Steiner silver stain. Similar sections were evaluated by indirect immunohistochemistry (IHC) for the presence of B burgdorferi, as described 2 with the following modifications. Sections were incubated with the primary rabbit B burgdorferi ss antiserum for 12 hours at 4°C, and the secondary antibody was goat anti-rabbit EnVision-HRP (DAKO, Carpinteria, California).
Quantitative PCR Assays
DNA was extracted from fresh tissues from case No. 1 and from 25-μm scrolls of formalin-fixed, paraffin-embedded (FFPE) spinal cord from case No. 2, with a DNeasy tissue kit (Qiagen, Valencia, California) according to the manufacturer’s instructions, and eluted into 200 μl of sterile deionized water. Tissues from case No. 1 were weighed before DNA extraction.
Quantitative PCR (qPCR) assays for multiple B burgdorferi ss–specific gene targets—including outer surface protein A (ospA), outer surface protein C (ospC), flagellin (flaB), decorin-binding protein A (dbpA), and arthritis-related protein (arp)—were standardized and optimized. 10 All gene targets were less than 150 base pairs (bp) in length, ranging from 95 to 142. Multiple nontemplate and negative control reactions with sterile deionized water and fresh and FFPE tissues from unaffected horses were included within each plate. Reactions were accomplished using a 7900HT Fast Real-Time PCR System (Applied Biosystems, Foster City, California). Copy numbers were calculated with a reference standard curve and expressed per milligram of tissue or microliter of fluid from which DNA was extracted.
Sequence Analysis
Target genes that were positive by qPCR, including ospA, ospC, and flaB, were amplified by conventional PCR for sequence analysis. Table 1 lists the manually designed primers for the full-length ospA gene, the full-length ospC gene, and a fragment of the flaB gene. Amplification reactions, based on HotStar Taq Master Mix (Qiagen), were accomplished with a Dyad-Peltier Thermocycler (Bio-Rad Laboratories, Hercules, California). Cycling parameters included denaturation at 95°C for 10 minutes, followed by 30 cycles of 95°C for 1 minute, 53°C for 1 minute, and 72°C for 1 minute, with postamplification extension at 72°C for 10 minutes. PCR products were separated by agarose gel electrophoresis, visualized with SYBR Green I (Invitrogen, Eugene, Oregon), and purified with a Rapid PCR Purification Kit (Marlingen Biosciences, Inc, Rockville, Maryland). Sequencing was performed by Davis Sequencing (Davis, California), using the respective forward primers. Sequences were manually corrected and compared with the BLAST database in the National Center for Biotechnology Information.
Primer sequences used to amplify select Borrelia burgdorferi genes for sequence analysis.
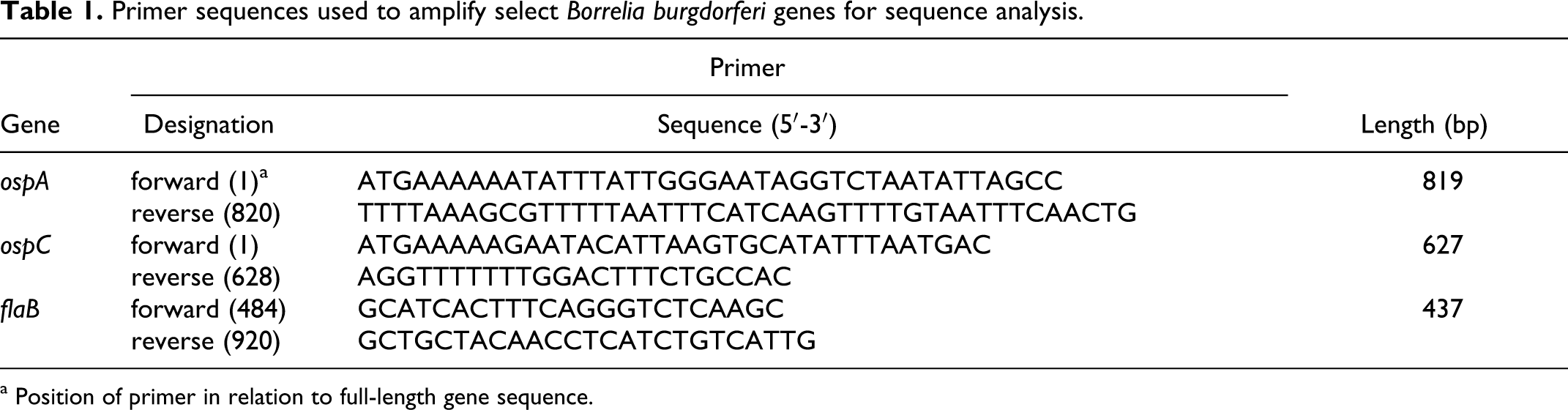
a Position of primer in relation to full-length gene sequence.
Ancillary Diagnostics
From case No. 1, swabs of the subdural deposits were submitted for bacterial culture (including cold enrichment for Listeria sp) and fungal culture. FFPE tissue sections were submitted for Leptospira sp, Listeria sp, Neospora sp, and Sarcocystis neurona IHC. Postmortem serum samples were submitted for Leptospira sp and West Nile virus serology. Fresh liver tissue was submitted for a toxicological screen, including heavy metals and vitamin E. Fresh lung and brain tissues were submitted for equine herpesvirus 1 and rabies virus direct fluorescent antibody assays. Fresh brain tissue was submitted to the National Veterinary Services Laboratories (Ames, Iowa) for PCR assays specific West Nile virus, Eastern equine encephalomyelitis virus, and Western equine encephalomyelitis virus, as well as virus isolation specific to Eastern equine encephalomyelitis, Western equine encephalomyelitis, and Venezuelan equine encephalomyelitis. From case No. 2, swabs of the subdural deposits were submitted for bacterial culture. FFPE tissue sections were submitted for IHC and electron microscopy for equine herpes virus 1 and 4.
Results
In both cases, histopathologic findings were most severe in the central nervous system, extending from the rostral cerebrum to the thoracolumbar spinal cord. Although both cases shared tissue distribution (meninges, ganglia, cranial and spinal nerve roots) and pattern of inflammation (perivascular to diffuse), they varied distinctly in the amount of necrosis, fibrosis, and secondary parenchymal involvement.
In case No. 1, necrosuppurative meningitis of the cervical spinal cord (Figs. 3, 4 ) corresponded to the grossly evident purulent subdural exudate. Extending from the rostral cerebrum to the lumbosacral cord, there was nonsuppurative perivascular meningitis (Fig. 5) and radiculoneuritis. In the anterior brainstem, there was minimal focal lymphoplasmacytic perivascular encephalitis with gemistocytic astrogliosis and fibrinoid vasculitis and endarterial fibrous proliferation in the leptomeninges. At the base of the heart, there was nonsuppurative myocarditis (Fig. 6). Additional lesions included perivascular to interstitial nonsuppurative myositis, chronic proliferative arthritis, interstitial pneumonia, and distal limb cellulitis with hemorrhage. No evidence of conjunctivitis or uveitis was identified.
In case No. 2, lesions included neutrophilic and histiocytic meningitis in the cervical and thoracic cord and diffuse lymphoplasmacytic perivascular meningoencephaloradiculoneuritis extending from the cerebrum to lumbar cord. There was marked dural and perivascular fibrosis with variable medial sclerosis and intimal proliferation (Fig. 7). In cranial and spinal nerve roots, there was axonal dropout and interstitial fibrosis (Fig. 8). Within the rostral cerebrum, the perivascular encephalitis was associated with neuropil rarefaction, spheroids, digestion chambers, and gemistocytic astrogliosis (Fig. 9). In atrophied muscles, interfascicular nerve fibers had similar axonal dropout and fibrosis; scattered myofibers were small and angular (neurogenic muscle atrophy). In both atrophied and unaffected muscles, there was nonsuppurative perivascular to interstitial myositis. Incidental findings included chronic hepatic granulomas and a focally extensive chronic pneumonia with alveolar histiocytosis, type II pneumocyte hyperplasia, and interstitial and pleural fibrosis.
Spirochetes were identified by Steiner silver impregnation in both cases, predominantly in the affected dura mater of brain and spinal cord (Fig. 10) and less commonly in the necrosuppurative debris (case No. 1). Numbers of spirochetes observed were similar in both cases. No spirochetes were identified within the neural parenchyma or within heart or muscle. No other etiologic agents were histochemically identified. In both cases, a similar distribution and tissue burden of spirochetes was demonstrated by IHC, also confirming expression of B burgdorferi ss–specific antigen (or antigens; Fig. 11).
In case No. 1, at least 2 B burgdorferi ss gene targets were amplified by qPCR from one or more samples of each tissue type (Table 2). The highest copy numbers were generated in the assays for ospC and flaB. The highest copy numbers of ospC and flaB were amplified from spinal cord, muscle, and joint capsule, respectively. None of the gene targets were amplified from samples of FFPE spinal cord from case No. 2.
Tissue Burdens of Borrelia burgdorferi in a Horse With Chronic Neuroborreliosis by Quantitative Polymerase Chain Reaction for 5 Select Gene Targets a
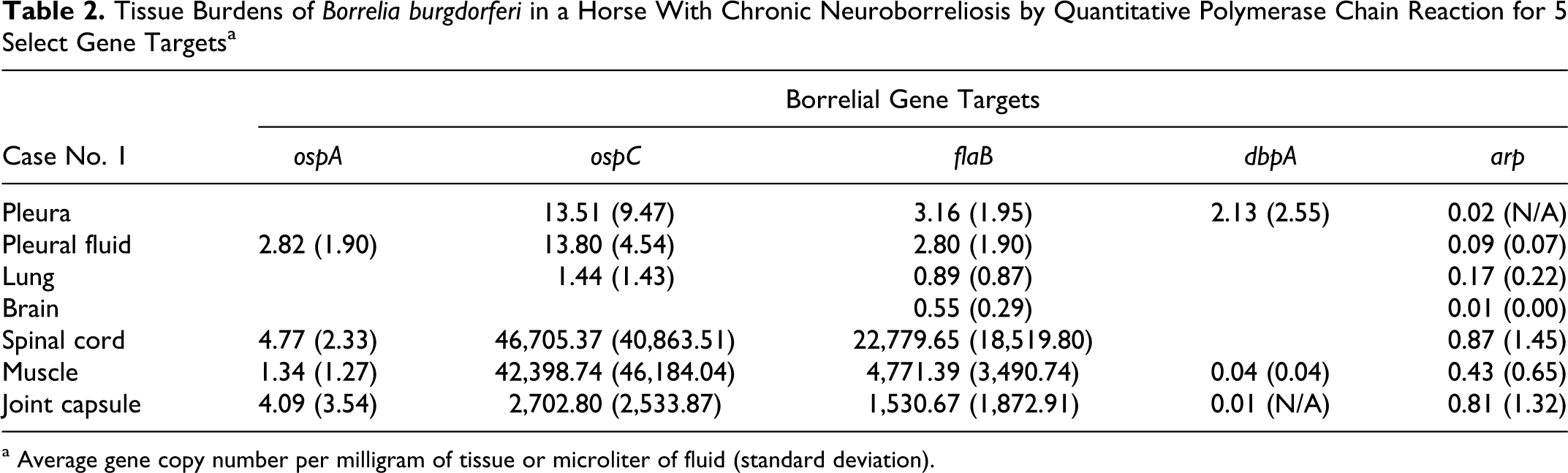
a Average gene copy number per milligram of tissue or microliter of fluid (standard deviation).
From spinal cord DNA in case No. 1, a partial segment of the flaB gene was sequenced with closest identity (380 of 381 bp) to the B burgdorferi ss flaB gene. The full-length ospA and ospC genes, also derived from the spinal cord DNA sample from case No. 1, were sequenced with closest identity (766 of 768 bp and 588 of 589 bp, respectively) to the B burgdorferi ss ospA and ospC genes. Sequence analysis of the ospC gene identified the borrelial strain as being closely related to B burgdorferi strain 297. In case No. 2, amplification of target genes for sequencing was unsuccessful. All other ancillary diagnostic assays were negative or within normal limits.
Discussion
Amplification of B burgdorferi ss–specific gene targets, sequence analysis of multiple genes, positive immunoreactivity to B burgdorferi ss–specific immune serum, and demonstration of spirochetes in affected tissue sections unequivocally identified the presence of B burgdorferi ss in association with the meningoradiculoneuritis in case No. 1. The arthritis and myositis in case No. 1 was also attributed to B burgdorferi ss on the basis of qPCR for multiple B burgdorferi ss–specific gene targets in the respective tissues, whereas the association of Borrelia with the myocarditis could not be confirmed, because no spirochetes were demonstrated in tissue section by silver impregnation or IHC and fresh tissue was not available for DNA extraction. In case No. 2, the presence of B burgdorferi was confirmed by IHC and demonstration of spirochetes in affected tissue sections.
In case No. 1, Lyme borreliosis was considered in the differential diagnosis because the region in which the horse was stabled was Lyme endemic and because the other horse stabled on the property was seropositive and had developed typical clinical signs. No explanation is available for the lack of seroconversion to borrelial antigens at any of the 3 time points or 2 independent laboratories. Whereas Lyme disease is considered endemic in New Jersey, borreliosis is not considered common in Southern California, where the horse of case No. 2 was stabled. Based on canine serosurveillance in northern San Diego county (which includes Rancho Santa Fe), exposure to B burgdorferi does occur in the region. 12 Though not definitive, the neurogenic muscle atrophy in case No. 2 was attributed to Lyme borreliosis. The infection could have persisted during the 4-year duration of clinical disease, possibly becoming exacerbated in the final stages by prolonged steroid therapy.
A causal relationship is considered likely, but Koch postulates have yet to be fulfilled for equine neuroborreliosis. Chang et al 6 established B burgdorferi infections in transiently immunosuppressed ponies but were unable to induce clinical disease or histologic lesions in any system except for dermatitis at the site of tick attachment. In that study, dura mater from 1 of 7 ponies was qPCR positive for ospA and 23S rRNA gene targets. In the nonhuman primate model of neuroborreliosis, meningoradiculitis has been induced but only with infection by the European borrelial subspecies B garinii. 1 With B burgdorferi ss strain N40, subarachnoid infection could be established in nonhuman primates, and although cerebrospinal fluid pleocytosis was observed, inflammation could be measured only by elevated cytokine expression.13,14 These observations reflect the clinicopathologic differences in human Lyme neuroborreliosis that have been attributed to the causative subspecies. 15
In case No. 1, sequence analysis of 3 borrelial genes, ospA, ospC, and flaB, identified the subspecies and strain involved as being closely related (99.9%) to B burgdorferi ss strain 297. This high level of DNA sequence identity of multiple genes to the 297 strain suggests potential strain-specific tissue tropism and pathogenicity because strain 297 was initially cultured from a human patient with neuroborreliosis. 7 Two gene targets, ospA and flaB, are relatively conserved across borrelial subspecies; however, ospC is highly variable even within the subspecies B burgdorferi ss, permitting strain identification. 17 Our data also suggest that qPCR was more sensitive than conventional PCR and that dbpA and arp gene targets were the least sensitive of the 5 targets. Wherever the copy numbers of particular genes were low, the targets could not be amplified by conventional PCR and were therefore not sequenced (eg, dbpA and arp). In case No. 2, qPCR analysis (even for short gene targets flaB and dbpA) and sequencing were unsuccessful and thus attributed to suboptimal DNA quality due to formalin fixation.
Tissue localization of spirochetes within the central nervous system appears to be consistent across host species. In both cases, spirochetes were found predominantly within the leptomeninges and dura mater, with few present within the suppurative reaction in case No. 1 and with none within the parenchyma in either. In the rare autopsy cases of human neuroborreliosis, spirochetes have been demonstrated in the leptomeninges and around the subependymal vessels. 11 In the nonhuman primate model, a similar distribution has been observed within the dura mater and leptomeninges.1,5 In the equine model, dura mater in one pony was the only tissue from the nervous system that was qPCR positive for B burgdorferi. 6 Given these observations and the recognized preference of B burgdorferi for connective tissue, we hypothesize that dissemination of spirochetes to the brain may be direct, centripetally along connective tissue planes, such as the epineurium or tunica adventitia, rather than by a hematogenous route.
These 2 equine cases share the meningeal distribution, perivascular inflammation, obliterative vascular sclerosis, dural fibrosis, and progression to parenchymal involvement in association with tissue-invasive spirochetes. In 1996, Hahn et al 9 recognized these same lesions in an equine case from the United Kingdom. That case and case No. 1 probably reflect an earlier stage of neuroborreliosis (necrosuppurative meningitis), whereas case No. 2 probably represents a later stage (nonsuppurative meningoradiculoneuritis, dural fibrosis, endarteritis obliterans, parenchymal involvement). These cases emphasize the need to consider neuroborreliosis in the differential diagnosis for equine neurologic disease (chronic progressive polyneuropathy or central nervous system disease), particularly when this unique histologic pattern of meningitis, radiculoneuritis, and myositis/myocarditis is recognized.
Footnotes
Acknowledgment
We thank Drs Brian A. Summers and Robert J. Higgins for their expertise and advice and Dr Christine Ross, Dr Cory Soltau, and Mrs Nicole Keightley and the histopathology staff at California Animal Health and Food Safety, Davis and San Bernardino, and University of California, Davis, School of Veterinary Medical Teaching Hospital for excellent technical support. This study was supported in part by National Institute of Health grant Nos. T32 AI 06055 (D.M.I.), T35 RR 07067 (J.M.J.), and R01 AI 26815 (S.W.B., S.F., E.H., K.J.O.).
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
