Abstract
A direct relationship has been firmly established between cyclooxygenase-2 (COX-2) expression and malignant behavior in human melanoma. This report examines the relationship between COX-2 expression and tumor location, mitotic and proliferative indices, degree of T CD3+ lymphocyte infiltration, overall survival, and frequency of recurrence and metastasis of 57 melanocytic tumors (25 oral and 32 cutaneous). COX-2 was highly or moderately expressed in 88% of oral neoplasms (22 of 25), whereas for their cutaneous counterparts, COX-2 expression was low or insignificant in 75% of cases (24 of 32). High and moderate COX-2 expression levels were observed in 73% of melanocytic tumors with a mitotic index ≥ 3 per 10 high-power fields (26 of 36), whereas in 81% of tumors with a mitotic index < 3 (17 of 21), expression was mild or absent. There were 41 cases with known clinical outcomes; of those showing high, moderate, and mild COX-2 expression, 83.3% (10 of 12), 37.5% (3 of 8), and 25% (2 of 8) died, respectively, whereas 100% of animals showing no COX-2 expression (13 of 13) were still alive at the last follow-up. COX-2 expression was statistically correlated with tumor location, mitotic and percentage Ki-67 proliferative indices, and overall survival, frequency of neoplastic recurrence and metastasis. Regression analysis also showed disease-specific predictive value for COX-2 expression for subjects with melanocytic neoplasms. Additionally, only high COX-2 expression showed significant differences in overall survival, in comparison with moderate, mild, or absent expression. These results suggest that high COX-2 expression may be considered a prognostic biomarker and potentially as a target for therapeutic and preventive strategies in canine melanocytic neoplasms.
Canine melanocytic neoplasms occur most frequently in the skin and oral cavity. Classically, body location is one criterion used to establish a prognosis of these tumors.17,10,36 In the skin, melanocytic neoplasms account for 4 to 20% of all cutaneous neoplasms, of which fewer than 5% are malignant.16,32 Yet canine oral melanomas account for 30 to 40% of all neoplasms in the head and are considered malignant, 36 with poorer prognosis and lower survival rates than their cutaneous melanocytic counterparts. The reasons for this more aggressive behavior are not clear. Recent data suggest that this malignancy may be due to a higher atypia and proliferation rate observed in oral melanomas.27,37 However, several reports suggest a lack of correlation between tumor location and clinical course.6,20 Of the extensive number of factors used in human pathology to establish an accurate prognosis of melanocytic neoplasms, 40 the mitotic index and Ki-67 proliferation index are the factors that have shown the best correlation with clinical outcome of canine melanocytic neoplasms.27,34–37 Although the mitotic index is considered the most reliable feature for establishing the biological behavior of cutaneous and ocular melanomas,4,42 some reports have found no statistical relationship between mitotic index and survival rate in canine oral melanomas. 33 On the basis of these results, new markers should be studied that offer a more reliable canine melanocytic neoplasm behavior. Cyclooxigenases are a group of inducible enzymes involved in catalyzing the conversion of arachidonic acid to prostaglandin H2, the precursor of prostanoids during inflammatory processes. Cyclooxygenase-2 (COX-2) is expressed in other pathologic conditions, such as cancer, where it is responsible for a range of effects, including angiogenesis, metastasis, antiapoptosis, immunosuppression, and neoplastic cell proliferation. 41 In human pathology, COX-2 expression has been proposed as a new immunohistochemical marker to distinguish early human cutaneous melanomas from benign melanocytic skin tumors 9 and a prognostic factor that might be considered a reasonable therapeutic target. 3 In veterinary pathology, COX-2 expression has been described in several canine carcinomas512,21,24 and in canine oral, ocular, and cutaneous melanocytic neoplasms.28,30,31 Although high COX-2 expression has been reported in histologic malignant melanomas, the relation with the clinical outcome of these neoplasms has not been analyzed. The aim of the present study was therefore to study differential COX-2 expression in oral and cutaneous melanocytic neoplasms and to determine any statistical correlation between COX-2 expression and other prognostic parameters (grade of tumor malignancy according to mitotic index and Ki-67 proliferation index) and clinical outcome.
Material and Methods
Retrospective Case Material
Sixty-seven cases in which a melanocytic neoplasm was histologically diagnosed were obtained from the archives of the veterinary pathology laboratories of the School of Veterinary Medicine of the University of Murcia and from the private laboratory Histovet (Barcelona). Ten cases were excluded because of small size or excessive melanin pigmentation, both of which preclude immunocytochemical evaluation. Thus, 57 cases were available for analysis. Twenty-five tumors were located in the oral cavity (gingiva and labial mucosa), and 32 were located in the skin (head, abdomen, and extremities). Tumors were classified according to their histologic pattern: 19 epithelioid, 8 fusiform, and 30 mixed (epithelioid + fusiform). Thirty-three affected dogs were male and 24 were female. The median age was 9.8 ± 3.4 years (range, 1–17 years). No breed predisposition was observed in oral neoplasm, but Rottweiler was the most affected breed with regard to cutaneous melanocytic neoplasm (15.6%, 5 of 32).
Immunocytochemical Confirmation of Histologic Diagnosis
The primary diagnosis of melanocytic neoplasm was confirmed by melan-A and S-100 protein immunostaining (Dako, Carpinteria, CA) in hydrogen peroxide–bleached sections. All immunostainings were performed with an Envision System kit (Dako). After the antigen was demasked (EDTA solution, pH 8.0, at 121°C in melan-A immunostaining), the endogenous peroxidase was blocked by a solution of phosphate buffered saline (PBS) and 0.3% hydrogen peroxide for 5 minutes, followed by a wash in PBS and Tween-20. Sections were then incubated with the primary antibody overnight at 4°C (at 1:100 dilution for melan-A and 1:500 for S-100 protein) and, after a brief wash in PBS–Tween, incubated for 30 minutes with the labeled polymer at room temperature. Sections were then revealed with 3-aminoethylcarbazole (Dako), which stains positive cells red. Sections were then finally counterstained with Harry’s hematoxylin. All stains were performed twice to assess the specificity of positive staining.
Establishment of Mitotic and Percentage Ki-67 Proliferation Indices
After immunocytochemical confirmation, the mitotic index was established according to the parameters defined by the World Health Organization, 15 following the methodology described by Spangler and Kaas, 37 in bleached hematoxylin and eosin–stained sections. Thus, the mitotic index was calculated as the number of mitoses in 10 high-power fields (40×). Percentage cellular proliferation was revealed by immunocytochemical detection with an anti-Ki-67 monoclonal antibody (clone MIB-1, dilution 1:100, Dako) in bleached and citrate-treated sections (citrate buffer, pH 6.0, 121°C). The percentage Ki-67 proliferation index was then estimated by measuring the average expression of positive nuclear Ki-67 immunostaining in 500 nuclei, as previously described. 23
Analysis of T CD3+ Lymphocyte Infiltrate
The degree of specific T CD3+ inflammatory infiltrate was evaluated with an anti-CD3 antibody (dilution 1:50, Dako) in nonbleached and citrate-treated sections. A subjective scale was then used to establish the degree of T-cell infiltration: absent (0), mild (1), moderate (2), and high (3).
Analysis of COX-2 Expression
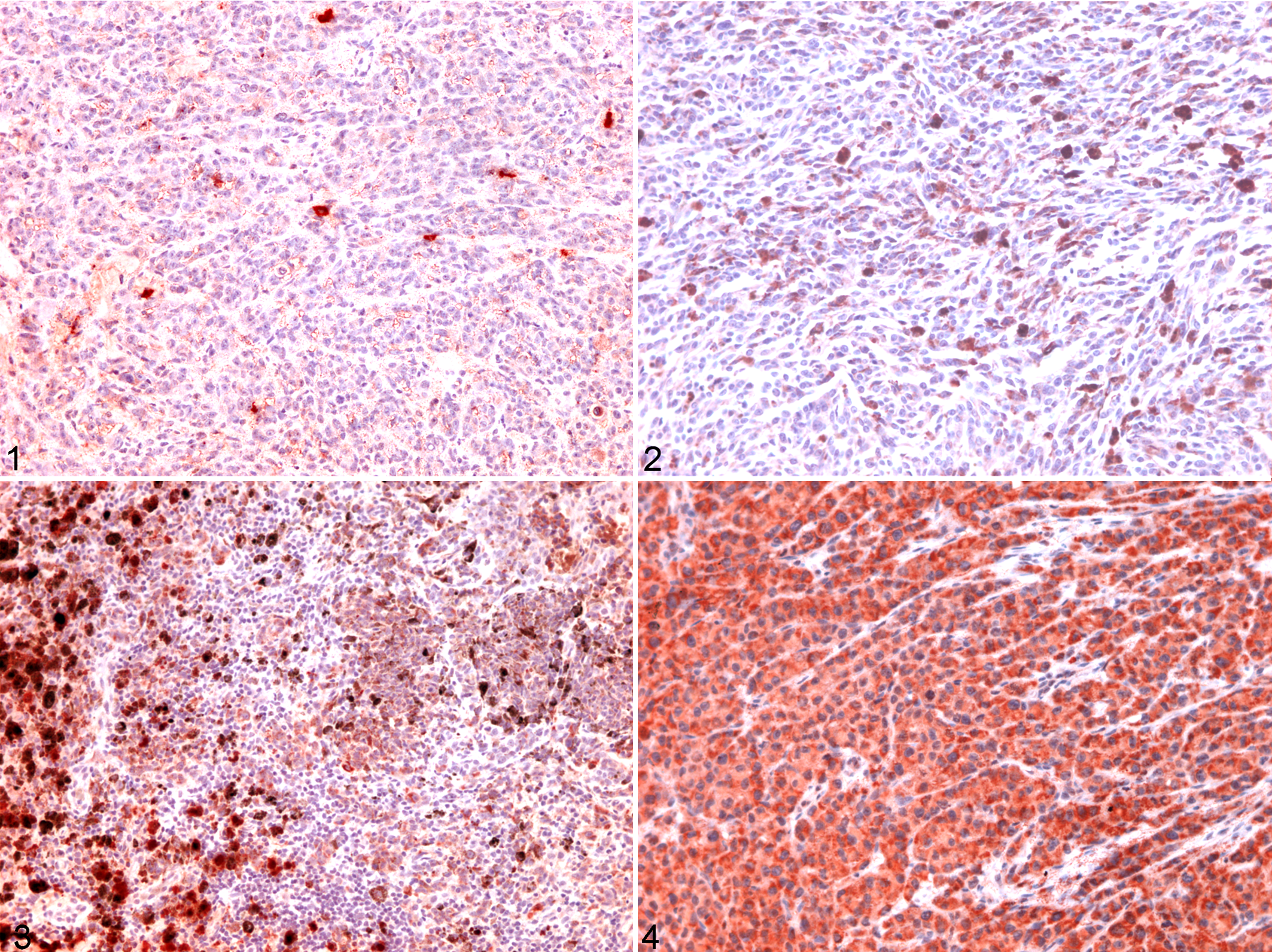
COX-2 immunostaining (dilution 1:50, Dako) was performed in nonbleached and pH 9.0 solution–treated sections (Dako) to avoid loss of immunocytochemical antigenicity. 29 A subjective scale was used to evaluate the number of positive cells (Figs. 1–4): 0 (absent, < 5% positive cells), 1 (mild, 5–25%), 2 (moderate, 25–50%), and 3 (high, > 50%). All evaluations were performed by at least 2 independent pathologists to validate all observations.
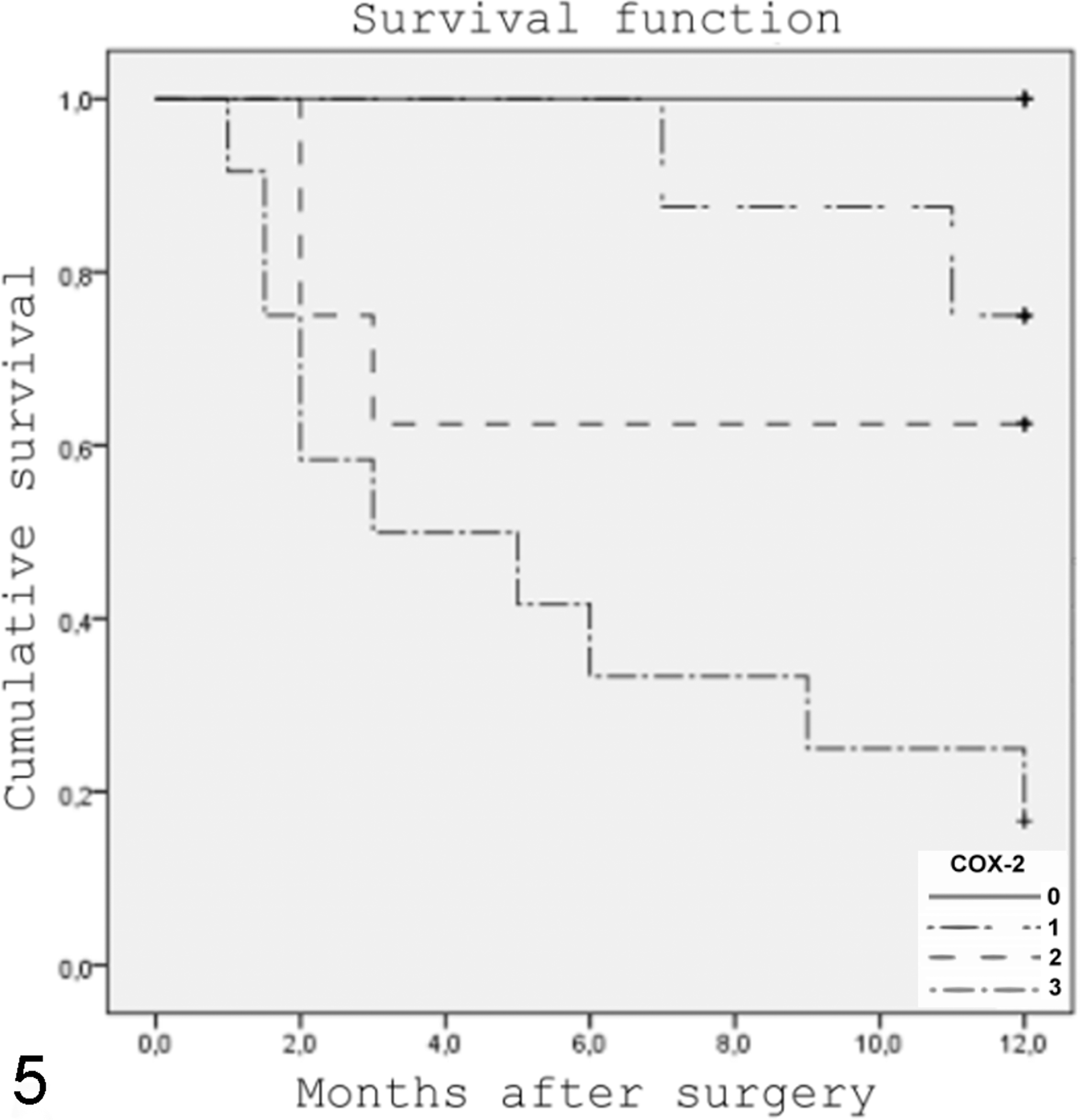
Survival curves for dogs in the 12 months following a diagnosis of melanocytic neoplasm (n = 41). The curves reflect the deaths related with neoplastic progression. High cyclooxygenase-2 immunoexpression is related to poorer survival.
Survival Study
The clinical course of the disease in animals from which surgical biopsies were obtained was ascertained in a telephone survey of the referring veterinarians. Twelve-month postsurgical follow-up was available in 46 cases, but 5 had to be excluded from the analysis because animals died for reasons unrelated with the neoplasia. The overall survival of the 41 cases with known clinical outcome (Table 1) was defined as the time elapsing from the day of diagnosis to the day of death or last follow-up.
Neoplastic Characteristics and Clinical Outcomes of the Animals Used in the Survival Study
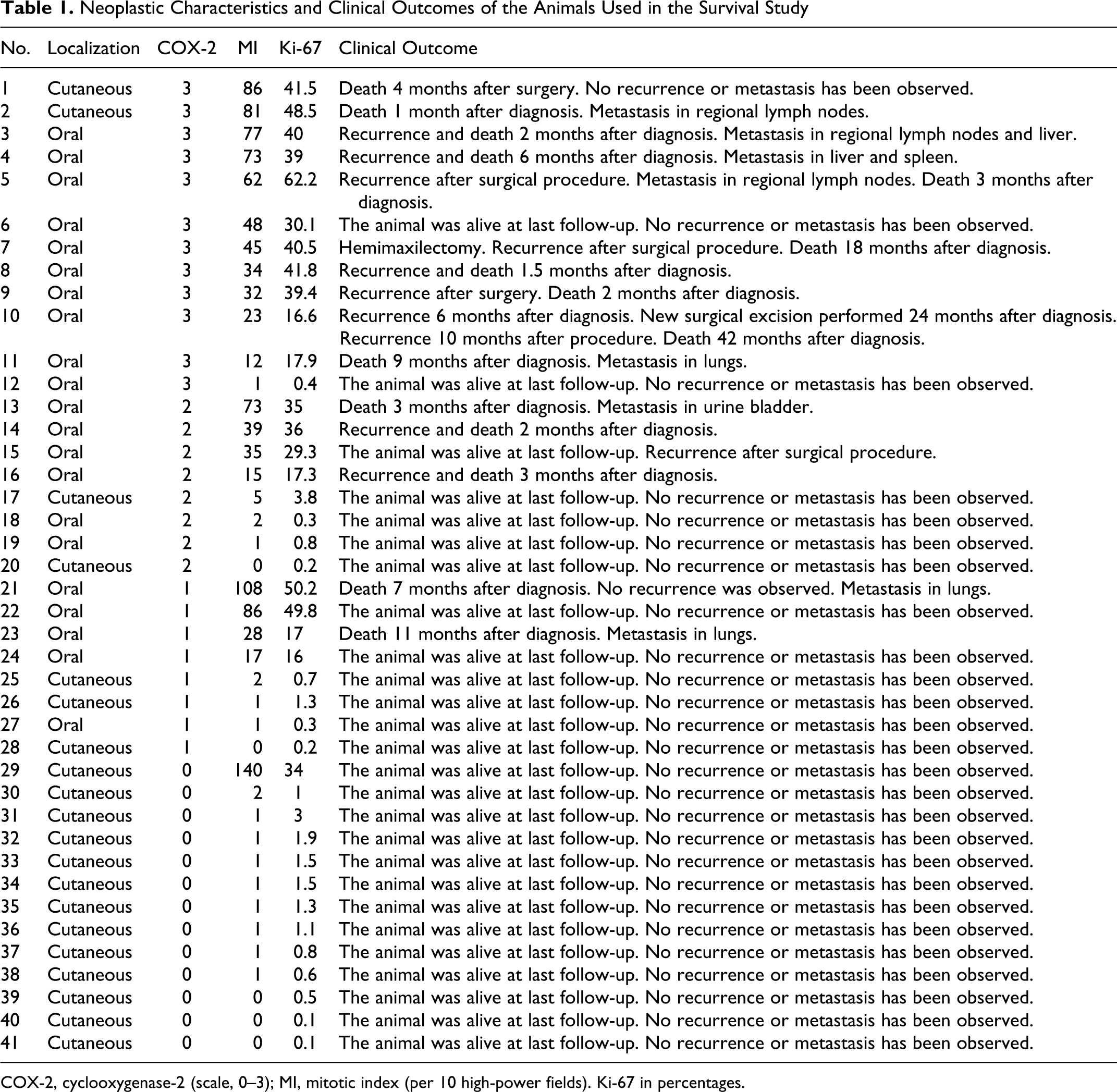
COX-2, cyclooxygenase-2 (scale, 0–3); MI, mitotic index (per 10 high-power fields). Ki-67 in percentages.
Statistical Analysis
Statistical analysis was performed with the PASW Statistics 18 (SPSS, Chicago, IL). Analysis of variance was used to compare COX-2 expression and values for the location of the neoplasia (oral or cutaneous), tumor cell type, mitotic and percentage Ki-67 proliferation indices, T CD3+ lymphocyte infiltrate, overall survival, and neoplastic recurrence and metastasis. Correlation between variables was determined with the Pearson correlation test (r). Survival curves for 12 months of clinical follow-up were generated by the Kaplan-Meier method, and differences in survival times were compared using the log-rank test. Statistical differences between survival rates according to the level of COX-2 expression (grade 0 vs grades 1, 2, and 3) were analyzed by an unpaired 2-tailed t test. To study the relationship of mitotic index, percentage Ki-67 proliferation index, tumor location, and COX-2 expression with clinical outcome, an univariate logistic regression analysis was performed. To establish the predictive value of COX-2 expression in canine melanocytic neoplasms, a multivariate regression analysis with reported hazard ratio (Cox) was performed to simultaneously evaluate the predictive effect of all factors (mitotic and percentage Ki-67 proliferation indices, tumor location, COX-2 expression, and clinical outcome) to determine the independence of COX-2 expression in predicting disease-specific survival for patients with melanocytic neoplasms. In multivariate analysis, the parameter was considered related with outcome for a hazard ratio value > 1. All values were considered statistically significant when P < .05.
Results
Immunocytochemical Diagnosis of Melanocytic Neoplasms
Of the 57 cases histopathologically diagnosed as melanocytic neoplasm, 100% were S-100 protein positive and 96.5% (55 of 57) were positive for melan-A. These results agree with previous reports that suggest that both antigens are sensitive and specific markers for the diagnosis of melanocytic neoplasms. 33
Mitotic Index and Ki-67 Expression
Regarding the mitotic index, 63.2% of the cases (36 of 57) had a mitotic index ≥ 3, whereas 36.8% (21 of 57) had a mitotic index < 3. For location, mitotic index was ≥ 3 in 96% of oral melanocytic neoplasms (24 of 25) and 40.7% of cutaneous neoplasms (13 of 32), whereas a mitotic index < 3 was observed in 4% of oral neoplasms (1 of 25) and 59.3% of cutaneous melanocytic neoplasms (19 of 32).
Overall, mean Ki-67 expression was 18.9 ± 17.4%, with a median value of 17.2% (range, 0.1–50.2%). For location, mean Ki-67 expression for oral neoplasms was 26.5 ± 14.2%, with a median value of 32.2% (range, 0.8–41.8%), whereas for its cutaneous counterpart, it was 12.3 ± 17.7%, with a median of 1.3% (range, 0.1–50.2%).
T CD3+ lymphocyte infiltration
Overall, T CD3+ cell infiltration showed 3 distribution patterns: few dispersed through the neoplastic cells, those organized into stromal perivascular cuffs throughout the neoplasm, and a peripheral distribution. In 17.6% of the cases (10 of 57), the T CD3+ lymphocyte infiltration was high or moderate, whereas in the 82.4% (47 of 57) the infiltration was mild or absent.
Immunocytochemical COX-2 expression
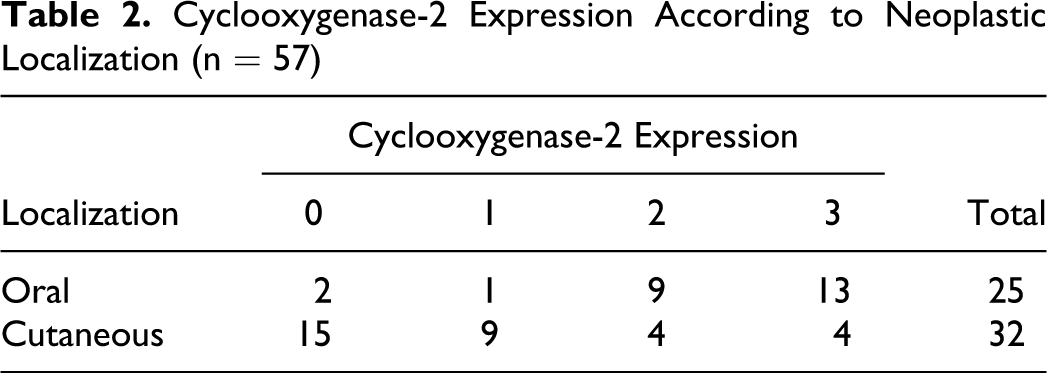
In all cases, positive COX-2 neoplastic cells showed an intracytoplasmatic and granular pattern, and no special immunostaining pattern was observed. Depending on the location of the neoplasms (Table 2), high or moderate expression was generally observed in oral melanocytic tumors (88%, 22 of 25), whereas mild or absent scores were generally observed in the cutaneous counterpart (75%, 24 of 32). Univariate statistical analysis showed that COX-2 expression was correlated with neoplasm location (P < .001, r = .671) but not with tumor cell type (P = .151). This statistical correlation was also observed between COX-2 expression and mitotic index (P < .001, r = .295; Table 3) where absent or mild expression was observed in melanocytic neoplasms with a mitotic index < 3 (81%, 17 of 21) and where moderate or high expression was most frequent in those neoplasms with a mitotic index ≥ 3 (73%, 26 of 36). Table 4 shows the relationship between percentage Ki-67 proliferation index after quartilization and COX-2 expression, where a statistically significant relationship was observed (P = .04, r = .489). Cases with a low-percentage Ki-67 proliferation index expressed an absent or mild amount of COX-2, whereas those cases with a high-percentage Ki-67 proliferation index, a higher COX-2 expression was evidenced. Finally, no statistical relationship between T CD3+ lymphocyte infiltrate and COX-2 expression was found (P = .109).
Cyclooxygenase-2 Expression According to Neoplastic Localization (n = 57)
Cyclooxygenase-2 Expression According to Mitotic Index (n = 57)
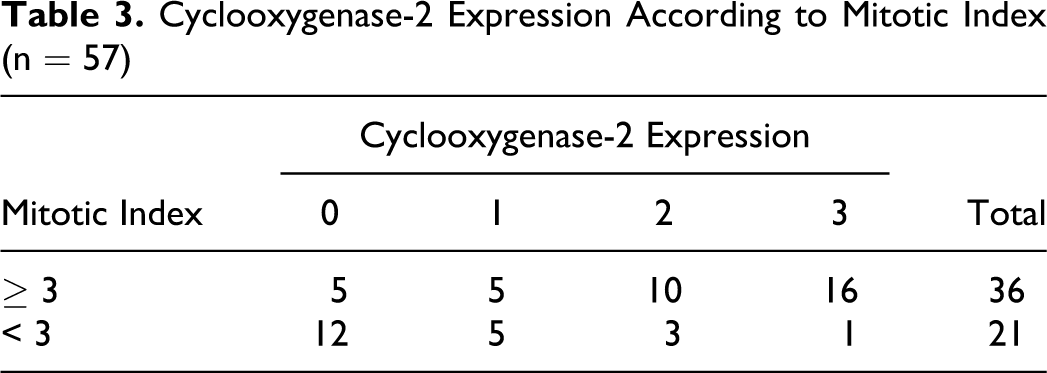
Quartiles of Ki-67 Values According to Cyclooxygenase-2 Expression (n = 57)
Survival Analysis
Overall, 83.3% of animals with high COX-2 expression died (10 of 12; Table 1), 66.7% (8 of 12) after 12 months of surgical follow-up and 16.6% (2 of 12) at 18 and 42 months of follow-up; 16.6% (2 of 12) were alive at the last follow-up. Yet 35.7% of animals showing moderate COX-2 expression (3 of 8) and 25% showing low COX-2 expression (2 of 8) died during the 12 months of surgical follow-up. These results contrast with those of animals showing no COX-2 expression, where 100% (13 of 13) were alive at 12 months of follow-up. Survival curves (Fig. 5) reflect the deaths due to neoplastic progression during the 12 months of surgical follow-up. Survival analysis showed that dogs with high neoplastic COX-2 expression had a significantly lower life expectancy (P < .001) than those with absent neoplastic COX-2 expression. Additionally, the statistical relationship between COX-2 expression and life expectancy was evidenced by analysis of variance (P < .001, r = –.567). In analyzing the statistical differences between survival rates according to level of COX-2 expression, the only statistically significant differences were between levels 0 and 3 of COX-2 expression (P = .005).
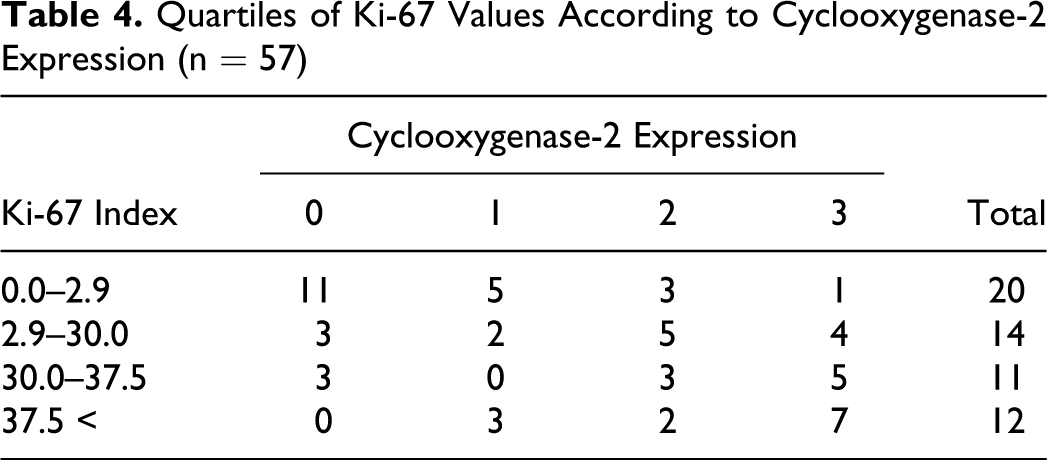
The univariate logistic regression analysis comparing individual parameters (mitotic index, percentage Ki-67 proliferation index, tumor location, and COX-2 expression) with clinical outcome showed that COX-2 expression was more related to clinical outcome than the rest of parameters were (odds ratio = 5.362, P = .001; Table 5). Finally, the multivariate regression analysis showed that only COX-2 and Ki-67 expression had an independent predictive value related with survival, but the hazard ratio for COX-2 expression (hazard ratio = 2.762, P = .004; Table 6) was higher than that for Ki-67 (hazard ratio = 1.082, P = .001). Yet in 50% animals showing high or moderate COX-2 expression (10 of 20) and in 5% (1 of 21) with mild or absent COX-2 expression, tumoral recurrence was observed. Statistically, there was a significant correlation between tumor recurrence and COX-2 expression (P < .001, r = .574). Metastasis was observed in 35% cases with high or moderate COX-2 expression (7 of 20) and in 5% (1 of 21) with mild or absent COX-2 immunolabeling. Statistical significant differences were found when COX-2 expression and metastasis were considered (P = .009, r = .421).
Univariate Logistic Regression Model Comparing Clinical Outcome With Individual Parameters (n = 41)
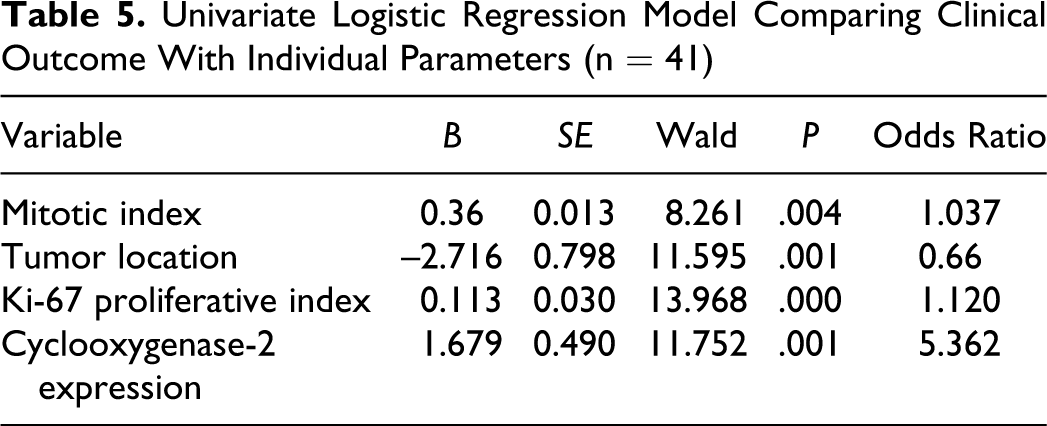
Cyclooxygenase-2 Expression in a Multivariate Cox Regression Analysis Comparing Individual Parameters With Clinical Outcome (n = 41)
Discussion
In this study, we examined the association between COX-2 expression and prognostic factors in melanocytic tumors, such as location, mitotic and proliferation indices, and survival time after diagnosis. The univariate and multivariate regression analyses showed that COX-2 expression has an independent disease-specific survival predictive value for subjects with melanocytic neoplasms. These findings lead to considering COX-2 expression as a suitable tool in the prognosis of this kind of tumor. Mohammed et al 28 provided the first report on COX-2 expression in canine melanomas; they observed that COX-2 was expressed by 60% of oral malignant melanoma, but they did not study the relationship between COX-2 expression and other prognostic parameters used to establish melanoma malignancy. Pires et al 31 more recently studied COX-2 in cutaneous, oral, and ocular melanocytic neoplasms. They found COX-2 expression in 11 of 29 cutaneous melanocytic neoplasms (30%; 9 in a high or moderate expression) and 9 of 9 oral melanomas (89%; 8 in a high or moderate expression), with a higher proportion of COX-2 expression in oral neoplasias than its cutaneous counterpart; as such, a relationship between the high expression of COX-2 and the histopathologic grade of malignancy was established. The results of these authors and our own findings on the relation of COX-2 with percentage Ki-67 proliferation index and survival rate suggest that COX-2 may be associated with the acquisition of malignancy during the tumorogenesis of melanocytic neoplasms. The mechanisms through which the enzyme favors tumor progression vary. 19 For example, the expression of COX-2 has been linked with a limitless replicative potential through the production of prostaglandin E2 involving a Gs–axin–β catenin signaling axis. 8 It has also been established that the overexpression of COX-2 increases matrix metalloproteinase activity to promote the invasion that favors tumor recidivism and metastasis. 39 Finally, the overexpression of COX-2 in some types of tumors induces the production of angiogenic factors, such as vascular endothelial growth factor and basic fibroblast growth factor, which are instrumental in stimulating the formation of new blood vessels, a crucial requirement for tumor progression. 38
In the case of melanocytic tumors, previous reports in human medicine suggest that COX-2 expression depends on both the stage and the histopathologic type of human melanoma.9,22 Additionally, we have observed not only a statistically significant higher COX-2 expression in oral versus cutaneous malignant melanomas but also a correlation with COX-2 expression and neoplastic recurrence and metastasis. Classically, canine oral melanomas have been considered malignant on the basis of their infiltrative growth, recurrency rate, and capacity to induce local and distant metastasis due to an incomplete surgical excision.6,14,36 However, this premise has recently become the subject of discussion given the new studies suggesting that well-differentiated oral melanocytic neoplasms have a better prognosis with local excision and no adjunct therapy. 13 These recent results suggest that factors other than anatomic location determine neoplastic progression. Some authors have proposed a functional role for COX-2 in human melanoma invasion 11 and metastases. 17 In agreement with these results, our observations suggest that the malignant behavior classically observed in canine oral melanomas might be related with COX-2 expression and might act as a promoter of its infiltrative growth and metastatic potential.
We found no relationship between COX-2 expression and T CD3+ lymphocyte infiltration. The prognostic significance of tumor lymphocyte infiltration is controversial. In human pathology, some studies have established that lymphocyte infiltration is only prognostic for T2–T4 primary cutaneous melanoma (tumor–nodes–metastasis system). 18 However, other studies were unable to demonstrate a relationship between tumor-infiltrating lymphocytes and survival rate. 2 Although COX-2 is an inducible enzyme involved in inflammatory processes, our data point to the lack of an effective adaptive inflammatory response against neoplastic cells, although in many malignant neoplasias—especially, those located in the oral cavity—a strong COX-2 signal was observed. Similarly, L’Eplattenier et al 26 observed that COX-2 expression in canine prostatic carcinoma cells is independent of the presence of an inflammatory infiltrate. Thus, these results and our observations suggest that COX-2 could develop alternative functions in melanoma progression to its naturally proinflammatory activity.
Our findings suggest that COX-2 expression differs between biologically aggressive and nonaggressive melanocytic tumors, an observation that could help to explain why oral canine melanomas have poorer prognosis than those of their cutaneous counterparts. On the basis of these observations, we suggest that COX-2 expression might act as a potential prognostic biomarker in canine melanocytic neoplasias. Although more studies are necessary to establish the exact role of COX-2 in the progression of canine malignant melanoma, this molecule could represent a potential target for therapeutic and preventive strategies in cases of canine melanocytic tumors. In human oncology, cases of complete tumoral regression have been observed in metastatic cutaneous melanomas25,43 in treatments involving COX-2 inhibitors. These reports should be taken into consideration when evaluating anticancer targeting strategies in cases of canine melanocytic neoplasias.
Footnotes
Acknowledgments
We thank all the submitting veterinarians and owners for providing us with all necessary data to perform this study.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
