Abstract
Molecular assays are widely used to prognosticate canine cutaneous mast cell tumors (MCT). There is limited information about these prognostic assays used on MCT that arise in the subcutis. The aims of this study were to evaluate the utility of KIT immunohistochemical labeling pattern, c-KIT mutational status (presence of internal tandem duplications in exon 11), and proliferation markers—including mitotic index, Ki67, and argyrophilic nucleolar organizing regions (AgNOR)—as independent prognostic markers for local recurrence and/or metastasis in canine subcutaneous MCT. A case–control design was used to analyze 60 subcutaneous MCT from 60 dogs, consisting of 24 dogs with subsequent local recurrence and 12 dogs with metastasis, as compared to dogs matched by breed, age, and sex with subcutaneous MCT that did not experience these events. Mitotic index, Ki67, the combination of Ki67 and AgNOR, and KIT cellular localization pattern were significantly associated with local recurrence and metastasis, thereby demonstrating their prognostic value for subcutaneous MCT. No internal tandem duplication mutations were detected in exon 11 of c-KIT in any tumors. Because c-KIT mutations have been demonstrated in only 20 to 30% of cutaneous MCT and primarily in tumors of higher grade, the number of subcutaneous MCT analyzed in this study may be insufficient to draw conclusions on the role c-KIT mutations in these tumors.
Subcutaneous mast cell tumors (MCT) are located in the subcutis, usually surrounded by fat. Until recently, 24,35 this type was not investigated separately from its cutaneous counterpart. Subcutaneous MCT are classified by many as grade II MCT, based on current grading schemes. 10,27 Because intermediate (grade II) tumors have a highly variable prognosis 10,25,26 and low interobserver agreement among pathologists, 25,26 accurate prognostic data for the subcutaneous variant of MCT are needed. In a recent retrospective survival analysis of subcutaneous MCT from 306 dogs, 35 we demonstrated that the majority of subcutaneous MCT do not behave aggressively, thus confirming the conclusion of a prior smaller study of subcutaneous MCT. 24 Furthermore, our results showed that a strong predictor of survival time, time to local recurrence, and metastasis was mitotic index (MI), assessed as the number of mitotic figures per 10 high-power fields. 35
MI is a valuable prognostic test 7,31 and an integral part of grading schemes for cutaneous MCT. 3,10,27 Despite this, there are drawbacks to using MI as a sole determinant for assessment of cellular proliferation. Accurate counting of mitotic figures may be influenced by field selection, plane of section, field diameter, intensity of cytoplasmic granularity (or presence of crush artifact), necrosis, and/or apoptotic cells. All of these variables can contribute to interobserver disagreement. In addition, MI detects only cells in the mitotic phase rather than the entire growth fraction (ie, all active cells within the cell cycle). To address these issues, many studies have assessed the use of adjuvant cellular proliferation assays, including histochemical staining for argyrophilic nucleolar organizing regions (AgNOR) 4,13,23,32,37 and immunohistochemistry for Ki67. 1,23,32,37 The nuclear protein Ki67 is expressed in all cell cycle stages but is not present in noncycling cells; thus, Ki67 expression can be used to assess the tumor growth fraction. 37 AgNOR are nucleolar subunits involved in RNA transcription, and the number of AgNOR per cell is associated with the rate of cellular proliferation. 37 Together, these markers assesses both the number of cycling cells and the rate of cell cycle progression, thus providing a more comprehensive assessment of tumor growth.
Mutations of c-KIT, the gene encoding the tyrosine kinase receptor KIT, are further responsible for the progression of some canine cutaneous MCT. 6,9,11,14–22,29,36 –38 Studies show that up to 30% of dogs with MCT have mutations in the juxtamembrane domain, 6,9,20,22,29,37,38 the majority of which are internal tandem duplications (ITD) in exon 11. The presence of these mutations is associated with higher-grade tumors. 20,29,38 Mutations result in constitutive activation of KIT, initiating signaling cascades leading to proliferation, survival, and invasion. 9,15 –18,21,22,29 One group reported that the presence of ITD mutations of c-KIT or aberrant cytoplasmic KIT protein localization was significantly associated with higher values of Ki67. 37
It is not clear from any study of cutaneous MCT if subcutaneous MCT were included; if so, the data could be compromised. Only one group has separately evaluated the use of immunohistochemical proliferation markers in subcutaneous MCT. 24 Its study found that mean counts of Ki67, proliferating cell nuclear antigen, and AgNOR were similar to grade I cutaneous tumors, 24 in comparison to values reported by other groups, 1,4 but these markers, as well as KIT expression pattern, were not demonstrated to have significant prognostic value, likely due to the small number of behaviorally aggressive cases. 24
The aim of the current study was to evaluate the prognostic utility of MI, Ki67, AgNOR, cellular localization of KIT receptor, and c-KIT mutational status for subcutaneous MCT. To ensure sufficient numbers of biologically aggressive tumors (because the majority of subcutaneous MCT have a benign clinical course), 24,35 we selected the most biologically aggressive cases (local recurrences and metastases) from our previous retrospective investigation, to compare these to nonaggressive subcutaneous MCT, using a case–control study design. As a further aim, we matched cases and controls for breed and, as possible, age and sex to eliminate the potential confounding effects of these variables.
Materials and Methods
Case Selection
A total of 60 subcutaneous MCT diagnosed between 2002 and 2008 were used to compare cases and controls for 2 outcomes: local recurrence and metastasis. Tumors were selected from a larger subset (n = 306) of subcutaneous MCT that we previously investigated for histologic prognostic indices. 35 Details of inclusion and exclusion criteria for that retrospective investigation are as previously reported. 35 In brief, cases were included if they met the following criteria: First, the tumor was a primary occurrence; second, all were histologically diagnosed as subcutaneous MCT based on adequate representation of the tumor; third, adequate follow-up data were obtained from veterinary clinics in the form of a questionnaire or telephone interview. Follow-up information included signalment, tumor location, dates of additional tumor development, metastasis, death or last examination, history of prior MCT, cause of death, and status at last examination. Additional information obtained for dogs with recurrence or metastasis included details on adjuvant treatment (surgery, chemotherapy, or radiation) and further diagnostic testing. Cases were excluded if there was immediate loss of follow-up, if there was a history of a previous MCT, if the sample was an incisional biopsy or was cytoreductive, or if the patient had concurrent MCT that were cutaneous.
Case–Control Design
There were 2 separate case–control analyses performed in this study. The first consisted of 24 dogs that developed local recurrence following tumor removal (cases) and 24 dogs that did not (controls). The second study consisted of 12 dogs that developed metastasis following tumor removal (cases) and 12 dogs that did not (controls). Six cases of metastasis had prior local recurrence, and these cases and their controls were used in both analyses. Six additional dogs with metastasis (without prior recurrence) and their controls were used for the metastatic outcome study. None of the control dogs for either outcome developed local recurrence or metastasis. Two control dogs (one for each outcome) developed an additional tumor distant to the initial surgical site 46 and 1,078 days after the initial surgery. The first dog had additional surgery for the second occurrence and was healthy at the end of the study (1,546 days later); the second dog was euthanized at 1,078 days because of the new MCT. All cases were matched by breed; 25 of 30 (83%) were matched by sex; and 22 of 30 (73%) were matched within 1 year of age at diagnosis. For local recurrence analysis, 12 pairs were matched for all 3 variables (breed, age, and sex); for metastasis, 8 pairs were matched.
The date of surgical excision was defined as the date of diagnosis. Follow-up time was defined as the date of diagnosis to the date of last follow-up or death. Local recurrence was defined as regrowth at the surgical site, and distant occurrence was defined as occurrence of a subsequent cutaneous or subcutaneous MCT at a anatomical location different from the initial surgery. Metastasis was defined as spread to the local lymph node or as disseminated MCT disease (ie, spread to internal organs). Metastasis was confirmed by at least one of the following: cytology of fine-needle aspirates of lymph nodes (n = 4), abdominal and thoracic radiographs (n = 4), histology of lymph node biopsies (n = 3), buffy coat analysis (n = 3), exploratory laparotomy (n = 1), abdominal ultrasound (n = 1), and whole body magnetic resonance imaging (n = 1). Six dogs had histologic or cytologic confirmation of metastasis; no postmortem examinations were performed on any dogs. We chose to treat cases unconfirmed by histology as metastases because we did not want to bias our study by including only tumors with favorable outcomes.
Evaluation of Histologic Variables
Histologic features were evaluated in a blinded fashion and included confirmation of subcutaneous location. MCT were determined to be subcutaneous based on a location within the subcutaneous tissue and no invasion of the dermis (Fig. 1 ). Two or more separate histologic sections of each tumor were examined to ensure this determination. In some cases, there was apparent multifocal extension of low numbers of mast cells around the base of hair follicles; however, the bulk of the tumors were in the subcutaneous tissue, and they were classified as subcutaneous MCT. In cases where the overlying epithelium was not present, the tumor was classified as subcutaneous because sections were completely surrounded by adipose tissue with no follicular or epidermal involvement. The original pathology report was available for each tumor.
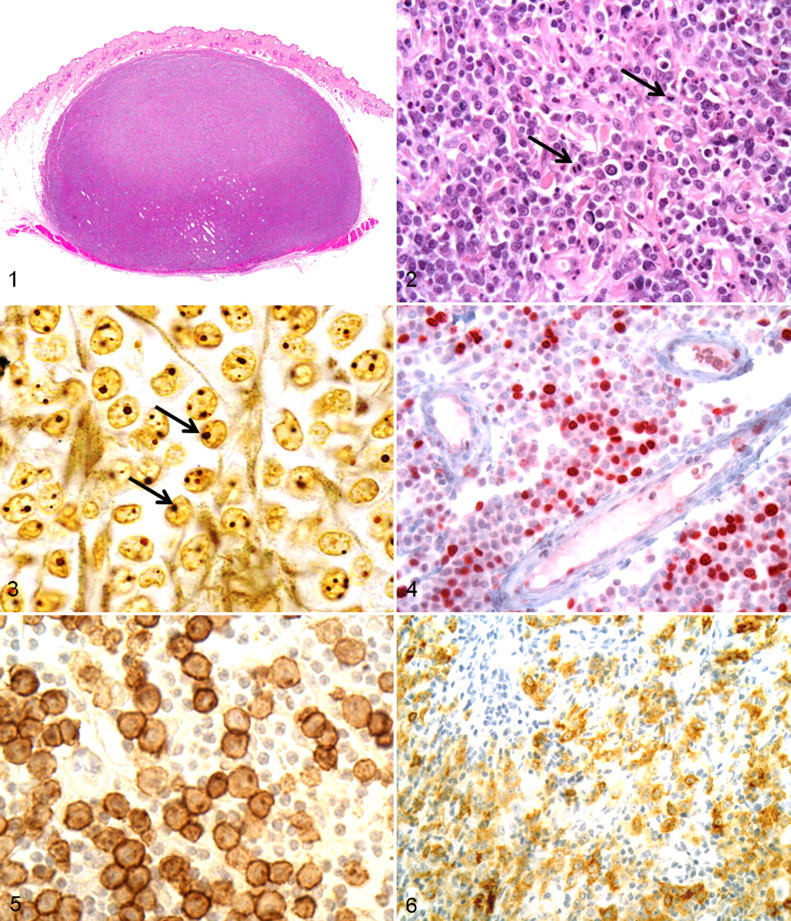
MI—defined as the number of mitotic figures per 10 high-power fields, using a 40× objective and a field diameter of 550 μm—was recorded for each tumor using the method described by Romansik et al 31 The area with the highest mitotic activity was chosen for evaluation (Fig. 2).
AgNOR and Immunohistochemistry
Histochemical and immunohistochemical labeling was conducted by the Michigan State Diagnostic Laboratory, using techniques previously described. 28,37 Slides were prepared using 5-μm sections of formalin-fixed paraffin-embedded tissue, which were cut, deparaffinized in xylene, rehydrated in graded ethanol, and rinsed in distilled water. For AgNOR staining, slides were incubated for 30 minutes at room temperature in the dark with freshly made AgNOR staining solution consisting of 0.02 g of gelatin in 1 ml of 1% formic acid and 1 g of silver nitrate in 2 ml of distilled water. Following AgNOR staining, slides were rinsed with distilled water, dehydrated with graded ethanol and xylene, and coverslipped (Fig. 3).
Ki67 immunolabeling was performed on the Benchmark Automated Staining system (Ventana Medical Systems, Inc, Tucson, AZ) following heat-induced epitope retrieval. Briefly, a mouse monoclonal anti-Ki67 antibody (MIB-1, Dako Cytomation, Carpinteria, CA) at dilution 1:50 was applied for 32 minutes and detected using the Enhanced V-Red Detection System (Ventana), which utilized an alkaline phosphatase and the chromogen Fast Red/Naphthol. For KIT, a rabbit polyclonal anti-KIT antibody was used (Dako Cytomation) at a 1:100 dilution for 30 minutes. Deparaffinization, antigen retrieval, immunohistochemical staining, and counterstaining were performed on the Bond maX Automated Staining System (Vision BioSystems, Leica, Bannockburn, IL) using the Bond Polymer Detection System (Vision BioSystems, Leica) with 3,3-diaminobenzidine substrate chromogen and hematoxylin counterstain (Fig. 4). Negative controls were included in each run, and they consisted of canine cutaneous MCT treated identically to the other tissue sections except that buffer was used in place of primary antibody. Known sections of canine cutaneous MCT were included in each run as a positive control for KIT. The basal layer of the epidermis served as an internal positive control for Ki67.
Evaluation of AgNOR Histochemical Staining
Staining for AgNOR was evaluated as previously described 37 by a single pathologist (J.J.T.). AgNOR were counted in 100 randomly selected neoplastic mast cells at 1,000× magnification and an average count per cell recorded. Individual AgNOR were resolved by focusing up and down while counting within nuclei (Fig. 3).
Evaluation of Ki67 and KIT Immunolabeling
KIT and Ki67 immunohistochemical labeling was evaluated in a blind fashion by a single pathologist (M.K.) using methods previously described. 12,37 For Ki67, all cell counting was performed manually. Areas with the highest proportion of immunopositive neoplastic mast cells were identified at 100× magnification using an light microscope (American Optical Instruments, Buffalo, NY). Upon identification of highly proliferative areas, the number of immunopositive cells present in a 10- × 10-mm grid area was counted using a 1-cm2 10 × 10 grid reticle at 400× magnification (Fig. 4). The number of immunopositive cells per grid area was evaluated over 5 high-power fields and subsequently averaged to obtain an average growth fraction.
All slides were assigned one of 3 patterns of KIT protein localization as previously described. 37 KIT pattern 1 consisted of a predominately perimembranous pattern of KIT protein localization with minimal cytoplasmic KIT protein localization (Fig. 5). KIT pattern 2 consisted of focal to stippled cytoplasmic KIT protein localization, and KIT pattern 3 consisted of diffuse cytoplasmic KIT protein localization (Fig. 6). Cells on the margins of the tissue sections were not considered, owing to possible artifactual staining.
c-KIT Mutational Analysis
Mutational analysis was performed as previously described. 37 In brief, DNA was extracted from 10-μm shavings of neoplastic tissue and subsequent polymerase chain reaction (PCR) amplification of c-KIT exon 11 and intron 11 to identify ITD c-KIT mutations. Formalin-fixed paraffin-embedded shavings were deparaffinized and incubated overnight in 50 μl of DNA extraction buffer—10 mM, Tris; pH, 8.0; 1 mM, ethylenediaminetetraacetic acid; 1% Tween (United States Biochemical Corp, Cleveland, OH)—and 1.5 μl of 15-mg/ml proteinase K (Roche, Indianapolis, IN) at 37°C. Samples were centrifuged at 1,306 × g for 5 minutes, and proteinase K was inactivated by heating at 95°C for 8 minutes. PCR amplification of c-KIT exon 11 and intron 11 was performed using a previously described 11 primer pair that flanks exon 11 and the 5′ end of intron 11, which includes the ITD region of the c-KIT proto-oncogene in canine MCT. The primers used for PCR amplification of the c-KIT juxtamembrane domain were based on the 5′ end of exon 11 (PE1: 5′-CCATGTATGAAGTACAGTGGAAG-3′ sense, base pairs 1,657–1,680 of exon 11) and the 5′ end of intron 11 (PE2: 5′-GTTCCCTAAAGTCATTGTTACACG-3′ antisense, nucleotides 43–66 of intron 11). PCRs were prepared in a 25-μl total reaction volume, with 5 μl of extracted DNA, 5 pmol of each primer, 0.5 U of Taq polymerase (Dinitrogen, Carlsbad, CA), and final concentrations of 80 μM of deoxynucleotide triphosphate, 2 mM of magnesium chloride, 20 mM of Tris–hydrogen chloride, and 50 μl of potassium chloride. Cycling conditions were as follows: 94°C for 4 minutes; 35 to 45 cycles at 94°C for 1 minute, 55°C for 1 minute, and 72°C for 1 minute; and 72°C for 5 minutes. Amplified products and ITD mutations were visualized by agarose gel electrophoresis on a 2% agarose gel after ethidium bromide staining.
Statistical Analysis
All statistics were performed using STATA 10 (StataCorp, College Station, TX). Univariable conditional logistic regression using exact methods was used to compare matched groups. Because not all cases and controls were matched for age and sex, subanalyses for each outcome were performed, including only dogs that matched for all 3 variables. Subanalyses were performed excluding dogs that had received chemotherapy. The results of the subanalyses showed similar results (increased odds ratios [ORs]); however, there was often not enough power to show statistical differences based on the small number of cases. Multivariable models were attempted, but these could not be performed; that is, because of the small sample size, they did not often converge. Additionally, interpretation of multivariable statistics was difficult because of the high degree of correlation of the variables (up to 80%). Results for conditional logistic regression are reported as ORs.
Analyses of the continuous variables MI, Ki67, AgNOR, and the product of Ki67 and AgNOR (Ag67), were performed using exact conditional logistic regression (EXLOGISTIC command in STATA 10). Each variable was assessed for a linear association for each outcome by graphically assessing the standardized residues against the predictor, as well as by performing conditional logistic regression (CLOGIT command in STATA 10) for the quadratic transform of each variable. Because our sample size was small and we had concerns about linearity, we categorized our independent variables and used exact methods for analyses. Because information can be lost with categorization, both continuous and categorical statistics are presented. Categorization of risk factor variables was done as follows: For MI, groups were stratified into MI ≤ 4 and MI > 4, based on the results of our previous retrospective investigation of subcutaneous MCT. 35 Counts of Ki67, AgNOR, and Ag67 were categorized on the basis of examination of quantiles that provided adequate sample size for subsequent analyses. For Ki67, the 25%, 50% and 75% quantiles were, respectively 3.8, 7.6, and 21.8 positive cells per grid area; for AgNOR, 1.75, 2.10, and 2.71 per cell; and for Ag67, 6.3, 17.1, and 55.0. The data were initially analyzed using the 50% and 75% quantiles to form 3 strata; however, there was no statistically significant difference between the 50% and 75% strata for any of these risk factors. Strata were therefore combined into 2 groups based on the 75% quantiles for risk factor analyses (Ki67 ≤ 21.8 and Ki67 > 21.8, AgNOR ≤ 2.7 and AgNOR > 2.7, and Ag67 ≤ 55 and Ag67 > 55).
For comparison of our results with published studies of cutaneous MCT, data were reanalyzed using previous published cutoff values for MI, 31 AgNOR, 37 Ki67, 37 and Ag67, 37 which were found to be prognostic markers for local recurrence and survival for dogs with cutaneous MCT. For MI, groups were stratified into MI ≤ 5 and MI > 5; for Ki67, Ki67 ≤ 23 and Ki67 > 23; for AgNOR, AgNOR ≤ 2.3 and AgNOR > 2.3; and for Ag67, Ag67 ≤ 53 and Ag67 > 53.
Agreement between tests for all matched pairs was evaluated using the Cohen κ statistic, and the interpretation of the magnitude of κ was based on Dohoo et al. 5 To assess whether test bias was present, the exact McNemar test was used to determine any significant differences in the proportions positive to the tests being compared. A significant P value (P < .05) for the McNemar χ2 suggests serious disagreement between tests, and assessment of agreement is unreliable. 5
Results
There were 12 breeds in the study, consisting of Labrador Retrievers (n = 24), mixed-breed dogs (n = 12), Siberian Huskies (n = 4), and 2 each of other purebred dog breeds. The median and mean age of all dogs was 7 years 11 months and 8 years 2 months (range 4 years 10 months to 12 years 7 months). Tumors were located on the abdomen (n = 7), thorax (n = 20), extremities (n = 19), head/neck (n = 12), and inguinal/perineal area (n = 1). Location was not known for one case. Twenty-two dogs were male and 38 were female.
Four dogs received chemotherapy during the study period. Three of these dogs developed widespread metastasis and were subsequently treated with chemotherapy for 1, 76, and 155 days, before death. The last dog was diagnosed with an additional, distant MCT and disseminated metastasis 1,184 days after the initial surgery; it began chemotherapy at that time and was healthy at the last date of examination, 21 days later. Because no dogs received chemotherapy before a diagnosis of local recurrence or metastasis, the effect of chemotherapy on statistical data for these outcomes should not be relevant. Despite this, we reanalyzed the data excluding these cases, with similar overall conclusions (data not shown).
Tables 1 and 2 present the proliferation indices for cases (reoccurrence/metastasis) and matched controls (without metastasis/reoccurrence). MI counts for all dogs ranged from 0 to 29, with a mean and median of 3.9 and 1.0, respectively. Ki67 counts ranged from 1.6 to 68.8, with a mean and median of 13.7 and 7.6 positive cells per grid area. AgNOR counts for all dogs ranged from 1.2 to 4.1 per cell, with a mean and median of 2.2 and 2.1. Ag67 counts ranged from 2.6 to 239.8, with a mean and median of 36.9 and 17.1. KIT pattern distribution for all dogs was as follows: diffuse cytoplasmic (n = 15), focal/stippled cytoplasmic (n = 24), and perimembranous (n = 20). One tumor (a control for local recurrence) had large areas of necrosis; thus, Ki67, AgNOR, and KIT pattern could not be evaluated. AgNOR staining was not demonstrated in a different control (also for local recurrence outcome). Case–control pairs for these tumors were excluded from matched analyses for these markers.
Proliferation Markers for 48 Dogs With Subcutaneous Mast Cell Tumors a
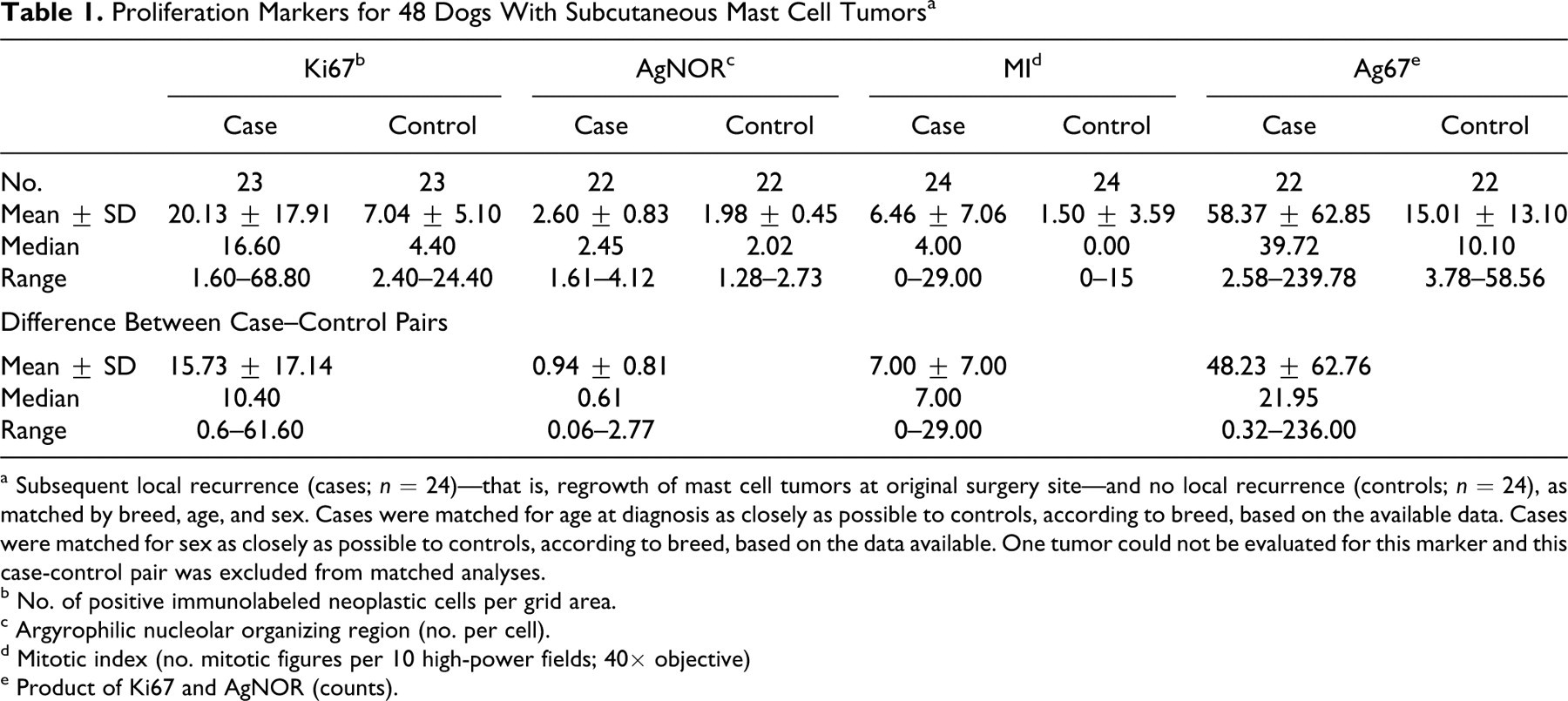
a Subsequent local recurrence (cases; n = 24)—that is, regrowth of mast cell tumors at original surgery site—and no local recurrence (controls; n = 24), as matched by breed, age, and sex. Cases were matched for age at diagnosis as closely as possible to controls, according to breed, based on the available data. Cases were matched for sex as closely as possible to controls, according to breed, based on the data available. One tumor could not be evaluated for this marker and this case-control pair was excluded from matched analyses.
b No. of positive immunolabeled neoplastic cells per grid area.
c Argyrophilic nucleolar organizing region (no. per cell).
d Mitotic index (no. mitotic figures per 10 high-power fields; 40× objective)
e Product of Ki67 and AgNOR (counts).
Proliferation Markers and KIT Pattern Expression for 24 Dogs With Subcutaneous Mast Cell Tumors a
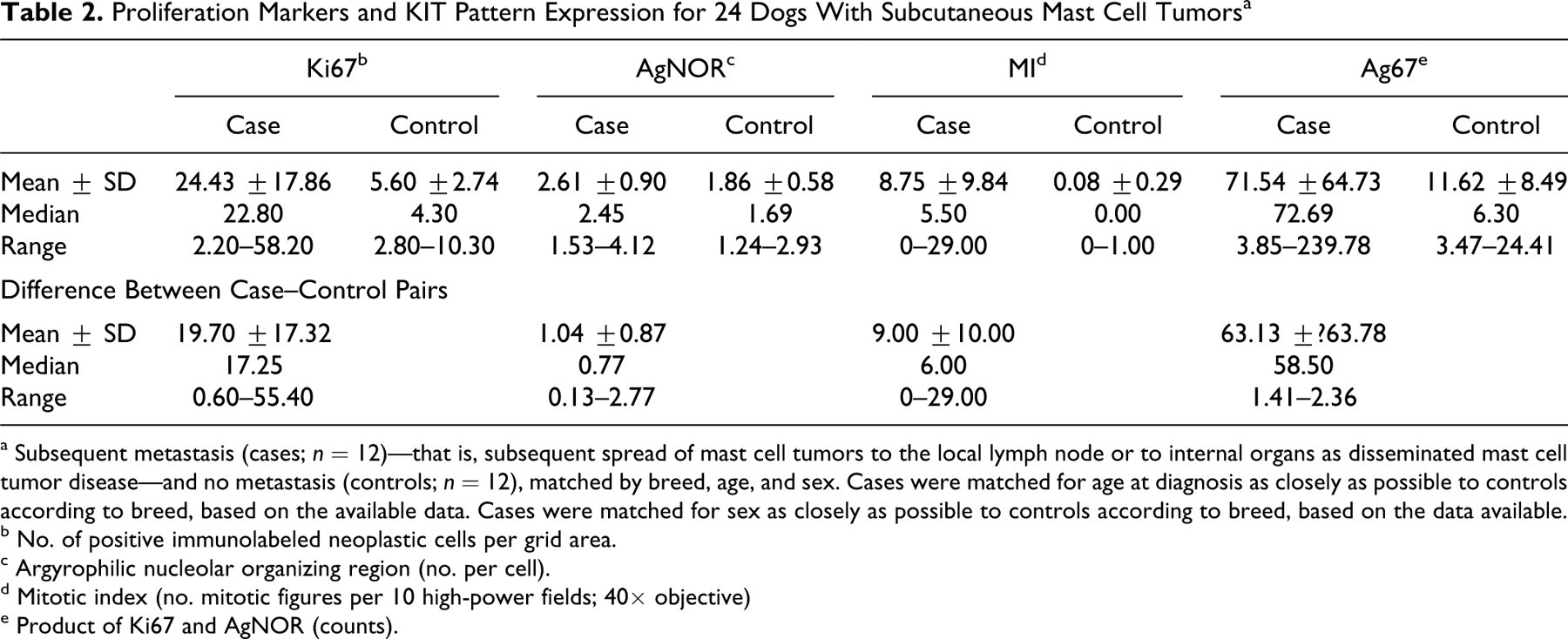
a Subsequent metastasis (cases; n = 12)—that is, subsequent spread of mast cell tumors to the local lymph node or to internal organs as disseminated mast cell tumor disease—and no metastasis (controls; n = 12), matched by breed, age, and sex. Cases were matched for age at diagnosis as closely as possible to controls according to breed, based on the available data. Cases were matched for sex as closely as possible to controls according to breed, based on the data available.
b No. of positive immunolabeled neoplastic cells per grid area.
c Argyrophilic nucleolar organizing region (no. per cell).
d Mitotic index (no. mitotic figures per 10 high-power fields; 40× objective)
e Product of Ki67 and AgNOR (counts).
c-KIT Mutational Analysis
No c-KIT mutations (presence of ITD in exon 11) were demonstrated in any of the 60 subcutaneous MCT.
Clinical Outcomes
Thirty-six dogs died during the study: 20 as a result of MCT-related disease, 4 of unknown causes, and 12 to diseases unrelated to MCT. MCT-related deaths were due to local recurrence (n = 9), distant occurrence (n = 1), and metastasis (n = 10). Of the remaining 24 dogs, 8 were lost to follow-up and 16 were alive at the end of the study.
Local Recurrence
The mean and median age for dogs with local recurrence was 8.3 and 7.5 years and for controls, 7.8 and 8.0 years, respectively. The mean and median difference in age between matched pairs was 1.4 and 7.0 months (range, 6 days to 7 years 2 months) with 17 of 24 pairs matched within 1 year of age. Nineteen pairs were matched by sex. The median time to local recurrence was 198 days (18 to 1,023) days, and median follow-up time for dogs with local recurrence was 213 days (48 to 1,354). Six dogs with local recurrence developed metastasis consisting of lymph node (n = 4) and disseminated disease (n = 2), whereas 15 of 24 dogs died from either recurrence or metastasis. The mean and median follow-up time for controls was 1,078 and 1,082 days, and 20 of 24 had follow-up times that were greater than their matched cases.
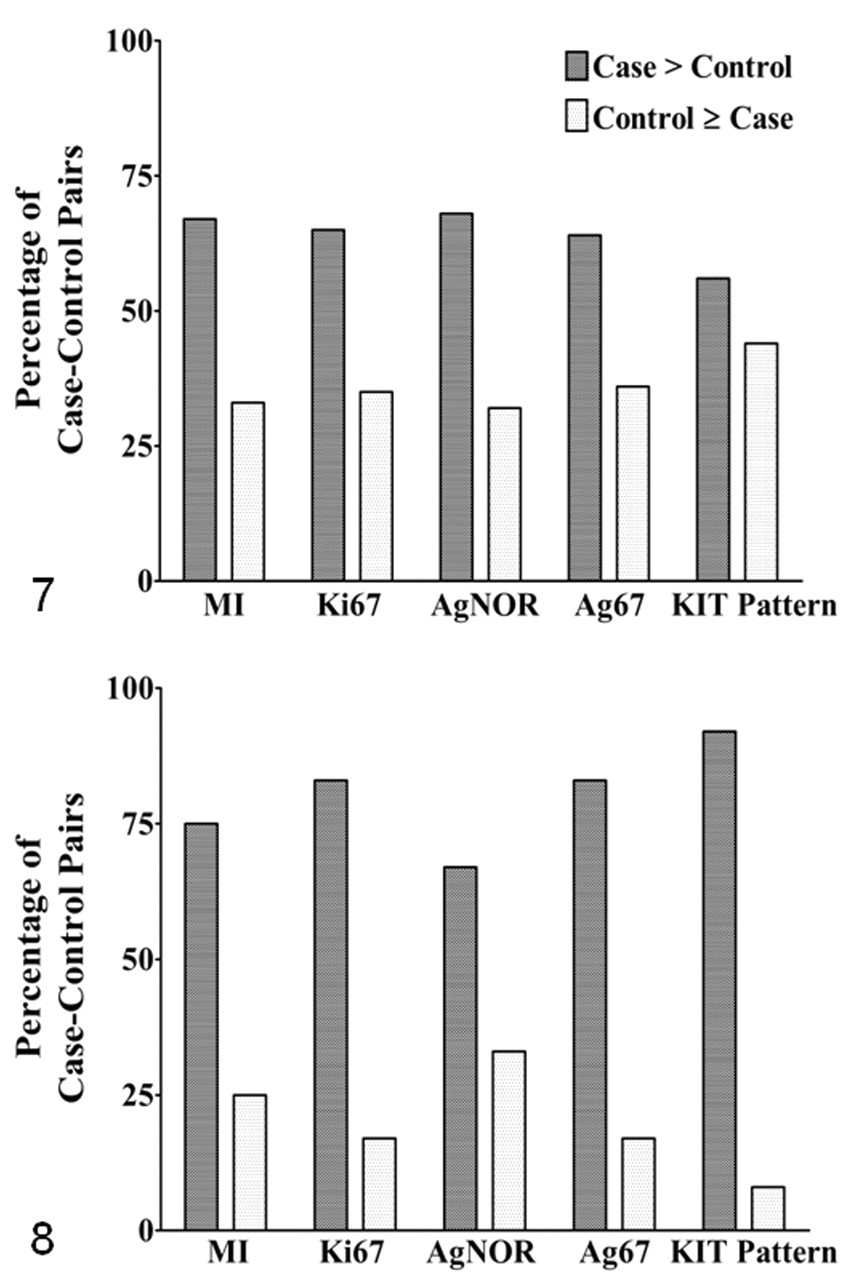
Mean and median values for MI, Ki67, AgNOR, and Ag67 for cases of local recurrence were consistently higher than those of matched controls (Table 1), and diffuse cytoplasmic KIT labeling was more frequently observed in reoccurring cases compared to controls. Perimembranous KIT labeling was observed in 13 MCT (3 cases and 10 controls); focal/stippled KIT labeling was present in 21 (10 cases and 11 controls); and diffuse cytoplasmic KIT labeling was found in 13 (11 cases and 3 controls). The percentage of matched pairs that had values greater than controls were consistently higher than the percentage of pairs for which the control had greater or equal values for all proliferation markers as well as cellular localization of KIT labeling (diffuse cytoplasmic > focal/stippled > perimembranous; Fig. 7).
Univariable exact logistic regression analysis showed MI, Ki67, AgNOR, and Ag67 counts to be significantly associated with increased odds of developing local recurrence, using either continuous or categorical data (Tables 3 and 4 ). For comparison purposes, the data were reanalyzed using previously reported cutoff counts for MI, 31 Ki67, 37 and Ag67. 37 Univariable statistics showed that dogs with MCT having MI > 5 and Ag67 > 53 had significantly increased odds of developing local recurrence than those with lower counts (OR = 5.5, P = .02; OR = 12.49, P < .01, respectively); however, the OR for Ki67 (Ki67 > 23 vs Ki67 ≤ 23) was not statistically significant (OR = 7.0, P = .07).
Univariable Analyses of Predictive Markers for Recurrence or Metastasis in Dogs With Subcutaneous Mast Cell Tumors a
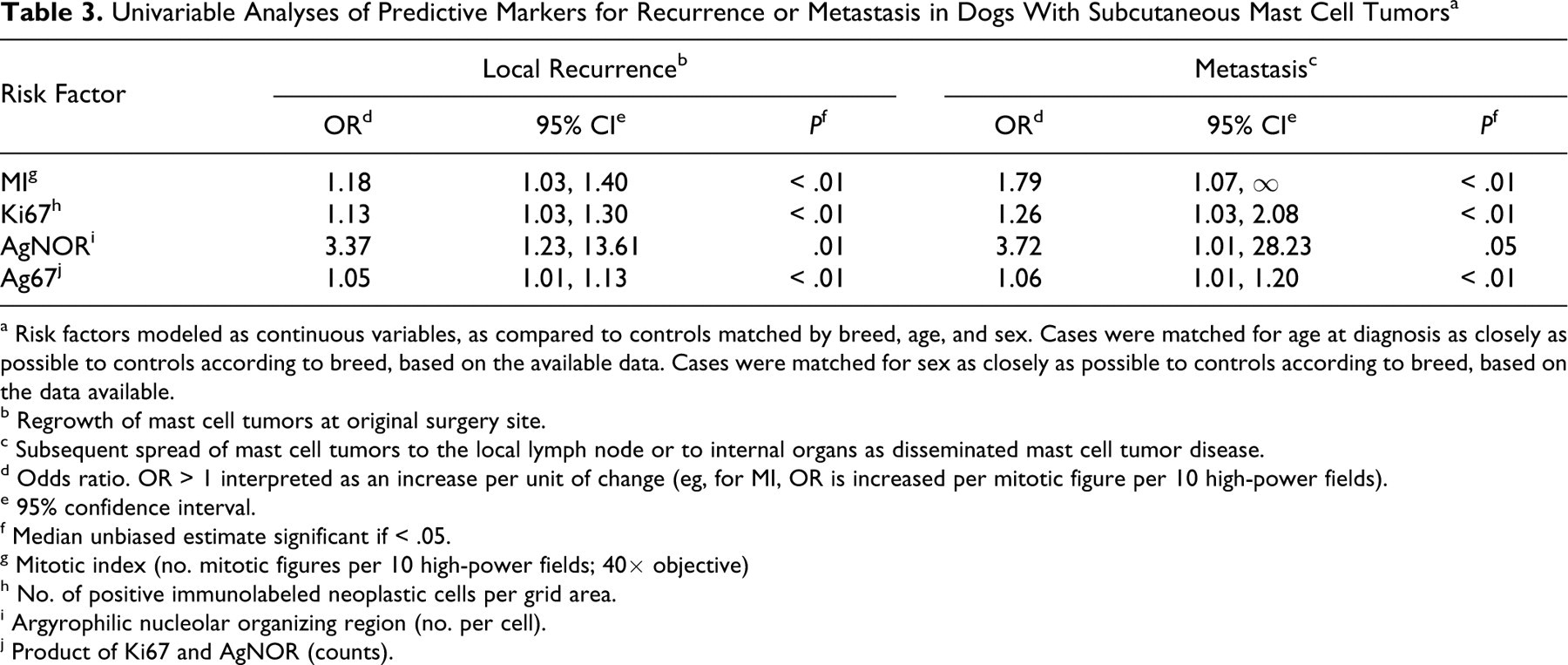
a Risk factors modeled as continuous variables, as compared to controls matched by breed, age, and sex. Cases were matched for age at diagnosis as closely as possible to controls according to breed, based on the available data. Cases were matched for sex as closely as possible to controls according to breed, based on the data available.
b Regrowth of mast cell tumors at original surgery site.
c Subsequent spread of mast cell tumors to the local lymph node or to internal organs as disseminated mast cell tumor disease.
d Odds ratio. OR > 1 interpreted as an increase per unit of change (eg, for MI, OR is increased per mitotic figure per 10 high-power fields).
e 95% confidence interval.
f Median unbiased estimate significant if < .05.
g Mitotic index (no. mitotic figures per 10 high-power fields; 40× objective)
h No. of positive immunolabeled neoplastic cells per grid area.
i Argyrophilic nucleolar organizing region (no. per cell).
j Product of Ki67 and AgNOR (counts).
Univariable Analyses of Categorized Risk Factors for Recurrence or Metastasis in Dogs with Subcutaneous Mast Cell Tumors a
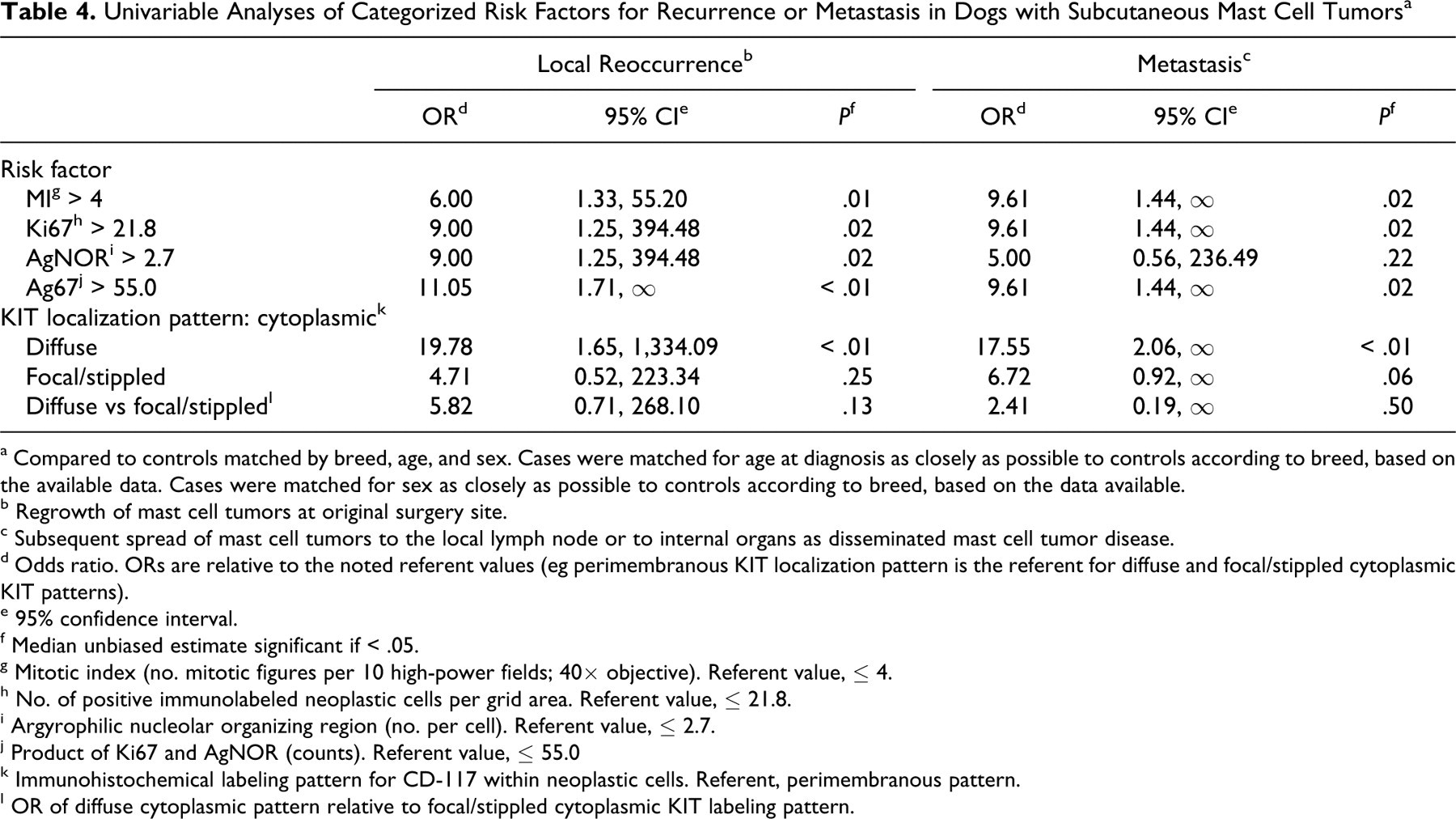
a Compared to controls matched by breed, age, and sex. Cases were matched for age at diagnosis as closely as possible to controls according to breed, based on the available data. Cases were matched for sex as closely as possible to controls according to breed, based on the data available.
b Regrowth of mast cell tumors at original surgery site.
c Subsequent spread of mast cell tumors to the local lymph node or to internal organs as disseminated mast cell tumor disease.
d Odds ratio. ORs are relative to the noted referent values (eg perimembranous KIT localization pattern is the referent for diffuse and focal/stippled cytoplasmic KIT patterns).
e 95% confidence interval.
f Median unbiased estimate significant if < .05.
g Mitotic index (no. mitotic figures per 10 high-power fields; 40× objective). Referent value, ≤ 4.
h No. of positive immunolabeled neoplastic cells per grid area. Referent value, ≤ 21.8.
i Argyrophilic nucleolar organizing region (no. per cell). Referent value, ≤ 2.7.
j Product of Ki67 and AgNOR (counts). Referent value, ≤ 55.0
k Immunohistochemical labeling pattern for CD-117 within neoplastic cells. Referent, perimembranous pattern.
l OR of diffuse cytoplasmic pattern relative to focal/stippled cytoplasmic KIT labeling pattern.
Diffuse cytoplasmic localization (pattern 3) of KIT receptor protein was significantly associated (P < .01) with increased odds of local recurrence compared to the perimembranous pattern (detailed later), but there was no significant difference between cytoplasmic and focal/stippled pattern (P = .13) or between focal/stippled and perimembranous patterns (P = .25). When the focal/stippled pattern was combined with cytoplasmic patterns (nonperimembranous pattern), the odds of local recurrence were increased compared to those with a perimembranous pattern (OR = 8.0, P = .04).
Metastasis
The mean and median age for dogs with metastasis was 8.77 and 8.23 years and for controls, 8.38 and 7.99 years, respectively. The mean and median difference between matched cases and controls were 1.2 years 9 months, with 9 of 12 dogs matched within 1 year of age. Eleven pairs were matched for sex. The mean and median times to metastasis were 255.67 and 171.50 days, and total follow-up times for cases were 395.67 and 205.50 days, respectively. For controls, the mean and median follow-up times were 1,078 and 1,082 days, and of these 12 dogs, 9 had follow-up times that were greater than their matched cases that had metastasized.
In all but one case, metastasis occurred subsequent to the original surgery. The one exception was a Cocker Spaniel that had metastasis confirmed histologically at the time of the original surgery, at which time both node and tumor were excised. Six dogs had local recurrences diagnosed either before or in conjunction with metastatic disease. Three dogs with metastasis had distant occurrences of MCT following the original surgery, and it could be determined whether metastasis resulted from the original MCT or subsequent, distant MCT. Ten dogs with metastasis died from MCT disease; 1 dog died of unrelated causes; and 1 dog was alive at the end of the study.
Table 2 presents results for MI, Ki67, AgNOR, and Ag67 for matched case–control pairs. Mean and median values for all proliferation markers for cases of metastasis were consistently higher than controls. As for the local recurrence outcome, diffuse cytoplasmic KIT labeling was more frequently observed in cases compared to controls. Perimembranous KIT labeling was observed in 11 MCT (1 case and 10 controls); focal/stippled KIT labeling was present in 7 (5 cases and 2 controls); and diffuse cytoplasmic KIT labeling was found in 6 MCT (all in cases that metastasized). The percentage of matched pairs that had values greater than controls were consistently higher than the percentage of pairs where the control had greater or equal values for all proliferation markers or cellular localization of KIT labeling (diffuse cytoplasmic > focal/stippled > perimembranous; Fig. 8).
Univariable logistic regression (Tables 3 and 4) showed MI, Ki67, and Ag67 values to be significantly associated with increased odds of developing metastasis. Dogs with MCT having a MI > 4, Ki67 > 21.8, or Ag67 > 55 were significantly more likely to develop metastasis than those with lower values. AgNOR counts were not significantly associated with increased odds of metastasis. To assess the influence of the 3 dogs that had distant occurrences, the data were reanalyzed excluding these cases with similar overall conclusions (data not shown).
Based on previously published higher cutoff counts, univariable analysis showed that dogs with MCT having MI > 5 had significantly increased odds of developing metastasis compared to those with MI ≤ 5 (OR = 8.16, P = .03) and those with Ag67 > 53 had significantly increased odds compared to those with ≤ 53 (OR = 11.05, P < .01); however, there was no statistically significant difference between those with Ki67 > 23 vs Ki67 ≤ 23 (OR = 6.72, P = .06).
Diffuse cytoplasmic immunohistochemical labeling for KIT protein was significantly associated (Table 4) with increased odds of developing metastasis compared to the perimembranous pattern (OR = 17.55, P < .01); however, there was no significant difference between cytoplasmic and focal/stippled patterns (OR = 6.72, P = .06) or between focal/stippled and perimembranous patterns (OR = 2.41, P = .50). When the focal/stippled pattern was combined with cytoplasmic patterns (nonperimembranous pattern), the odds of metastasis were increased compared to those with perimembranous localization (OR = 12.49, P < .01).
Statistical Associations Between Tests
According to exact McNemar values, there were no significant differences in the proportions positive for MI, Ki67, and AgNOR; therefore, Cohen κ statistic could be used to assess agreement between these tests (Table 5 ). The level of agreement between proliferation markers was moderate (between MI and AgNOR) to substantial (between MI and Ki67) and statistically significantly better than that expected due to chance (P < .01). In addition, κ statistics between proliferation markers and KIT pattern were found to be significant; however, exact McNemar χ2 values were also statistically significant (Table 5), indicating that agreement between these tests is unreliable. 5 Table 6 presents the distribution of proliferation marker counts and KIT localization pattern for cases and controls for both clinical outcomes, as stratified by MI. For both MI ≤ 4 and MI > 4 groups, Ki67, AgNOR, and Ag67 stratification illustrates a high degree of correlation between MI and other tests for cases and controls (Tables 5 and 6). The presence of nonperimembranous KIT localization pattern (combined diffuse and focal cytoplasmic) was a relatively more sensitive test than MI for clinical outcome. Of those tumors that recurred (n = 24) or metastasized (n = 12), 21 (88%) and 11 (92%) had nonperimembranous KIT localization, respectively, but only 12 recurrent (50%) and 7 metastatic (58%) tumors had a MI > 4 (Table 6). KIT localization appeared to be relatively less specific for local recurrence (43%) and metastasis (83%) compared to MI (specificity, 58% and 100%, respectively).
Statistical Associations Between Tests Used to Predict Clinical Outcome—Local Recurrence and Metastasis—for Dogs With Subcutaneous Mast Cell Tumors
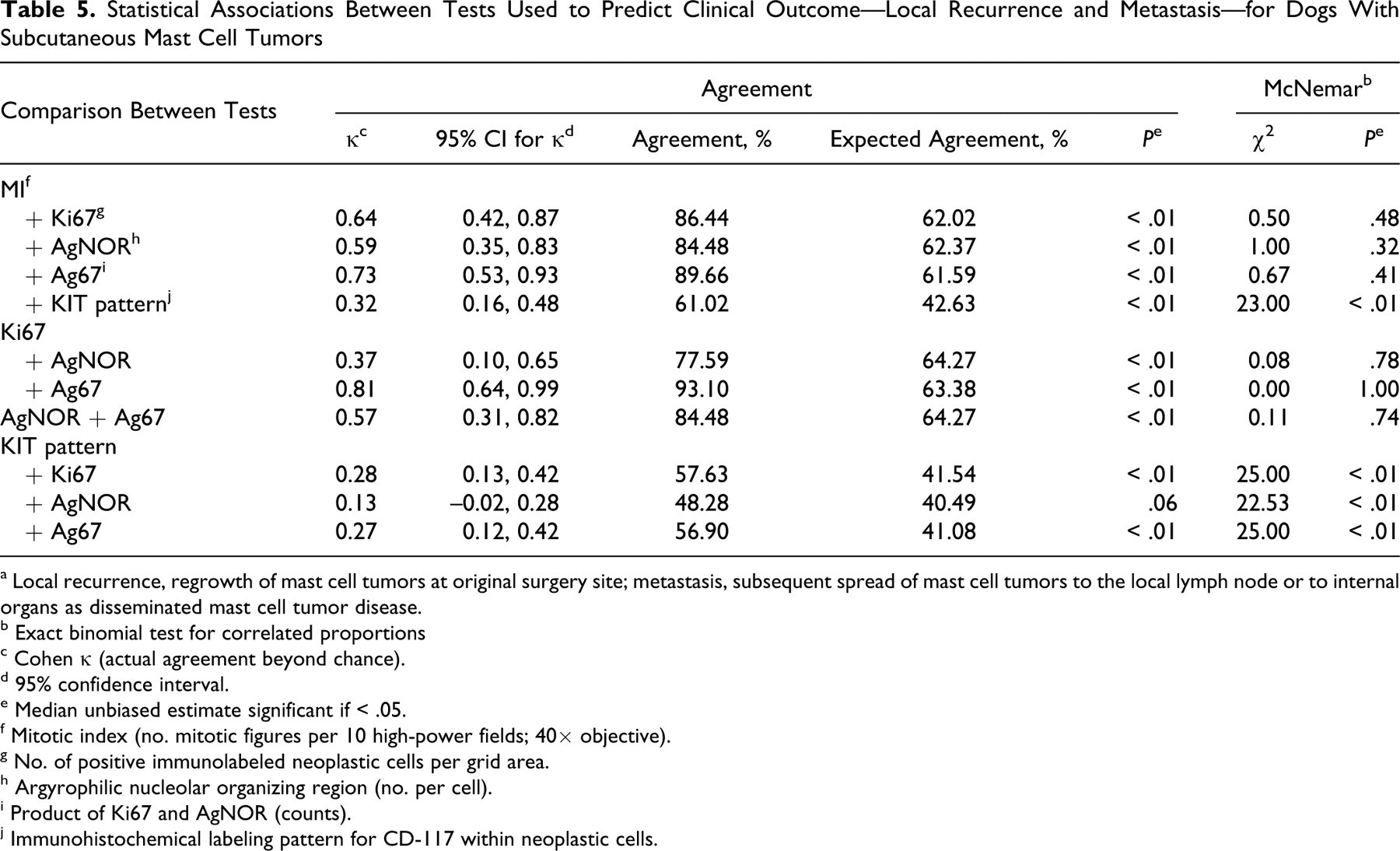
a Local recurrence, regrowth of mast cell tumors at original surgery site; metastasis, subsequent spread of mast cell tumors to the local lymph node or to internal organs as disseminated mast cell tumor disease.
b Exact binomial test for correlated proportions
c Cohen κ (actual agreement beyond chance).
d 95% confidence interval.
e Median unbiased estimate significant if < .05.
f Mitotic index (no. mitotic figures per 10 high-power fields; 40× objective).
g No. of positive immunolabeled neoplastic cells per grid area.
h Argyrophilic nucleolar organizing region (no. per cell).
i Product of Ki67 and AgNOR (counts).
j Immunohistochemical labeling pattern for CD-117 within neoplastic cells.
Count Distribution for Dogs With Subcutaneous Mast Cell Tumors for Local Recurrence and Metastasis Outcomes, Stratified by Mitotic Index a
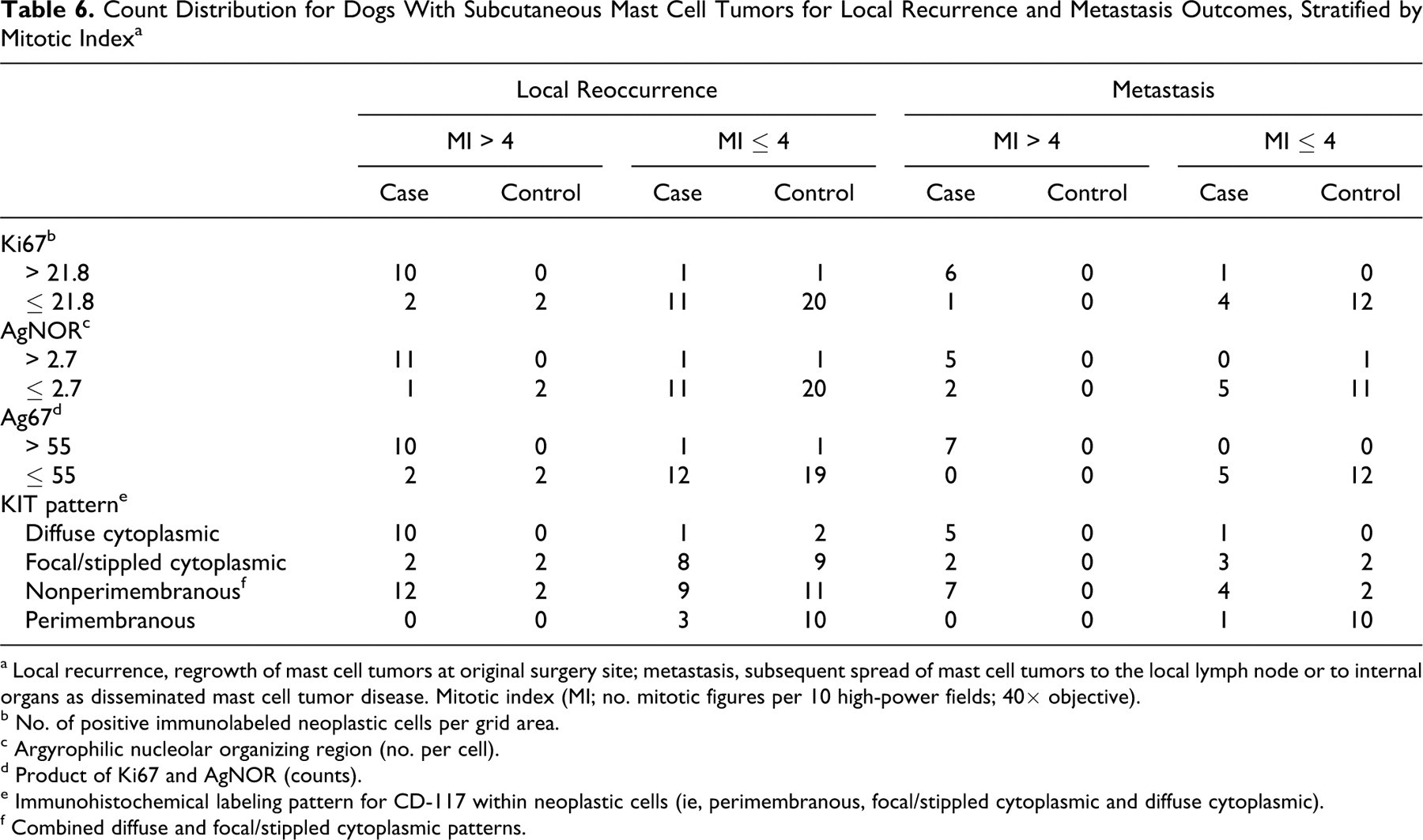
a Local recurrence, regrowth of mast cell tumors at original surgery site; metastasis, subsequent spread of mast cell tumors to the local lymph node or to internal organs as disseminated mast cell tumor disease. Mitotic index (MI; no. mitotic figures per 10 high-power fields; 40× objective).
b No. of positive immunolabeled neoplastic cells per grid area.
c Argyrophilic nucleolar organizing region (no. per cell).
d Product of Ki67 and AgNOR (counts).
e Immunohistochemical labeling pattern for CD-117 within neoplastic cells (ie, perimembranous, focal/stippled cytoplasmic and diffuse cytoplasmic).
f Combined diffuse and focal/stippled cytoplasmic patterns.
Discussion
MI, Ki67, AgNOR, and immunohistochemical KIT expression pattern are useful prognostic tests for subcutaneous MCT, as they are for cutaneous MCT. Dogs that developed postsurgical local recurrence and metastasis had tumors with consistently higher counts of MI, Ki67, AgNOR, and Ag67 and a cytoplasmic KIT localization pattern (Tables 1 and 2; Figs. 7, 8). Subcutaneous MCT with MI > 4, Ki67 > 21.8, or Ag67 > 55 were significantly more likely to locally reoccur and metastasize than those with lower counts (Table 4). Tumors with AgNOR > 2.71 were significantly more likely to locally reoccur than those with ≤ 2.7. Values for AgNOR were not found to be statistically associated with metastatic disease.
We used a matched case–control study to ensure inclusion of all aggressive tumors. Certain breeds are predisposed to MCT and the development of multiple tumors, 3,10,21 so we minimized these spurious effects by matching for breed, followed by sex and age. Multivariable analyses of an even larger population would have allowed us to control for these factors; however, because the majority of subcutaneous MCT are not aggressive, our sample size was small. Analyses of the data matching for all 3 variables showed similar ORs, but there was not always adequate power to show significant differences (data not shown).
The results of our study illustrate that no single test appears to be definitive in identifying aggressive disease for subcutaneous MCT. Measures of agreement between proliferation markers show that these are highly correlated (Table 5); however, confidence intervals are wide, indicating considerable uncertainty about the estimate. MI has been shown to be a valuable prognosis predictor for canine cutaneous 7,10,27,31 and subcutaneous 35 MCT; however, uniform methods of evaluation and reporting have not been established, which makes comparisons among studies difficult. Ki67 and AgNOR are more accurate measures of cell proliferation than MI because these tests evaluate the growth fraction and rate. 37 Furthermore, if standardization of molecular assays and count evaluation (eg, a grid reticle) can be achieved, these tests will be potentially less subject to interobserver disagreement. In a diagnostic setting, however, they are complex and expensive.
Ki67 and AgNOR counts are predictive for aggressive behavior of canine cutaneous MCT. 4,23,32,33,37 One group 37 showed that tumors having Ki67 counts > 23 positive cells per grid area and Ag67 values > 54 had a greater incidence and increased rate of local recurrence than did those with lower counts. We reanalyzed our data using these cutoffs and found that ORs were increased but statistical associations did not remain significant (although they were very close for Ki67). This disparity is likely due to our small sample size (predicated by the study design) and the low number of tumors with high proliferation indices.
Ki67 and AgNOR counts are reported to be lower for subcutaneous MCT than for grade II cutaneous MCT. 24 Ki67 counts were relatively lower (Tables 1 and 2) than those reported for cutaneous MCT in a previous study 37 using the same grid quantification method that we used—this despite having 50% of our data composed of the most aggressive MCT (by design) from our previous retrospective study (n = 306). Ki67 counts for cutaneous tumors 37 ranged from 3 to 97 positive cells per grid area, with a mean and median of 25 and 17, which were similar to our results for cases that locally recurred and metastasized (Tables 1 and 2). We cannot, however, conclude that all subcutaneous MCT have lower proliferation indices, as has been previously suggested. 24 A parallel study comparing cutaneous and subcutaneous variants would be needed to investigate this.
We found AgNOR counts for both cases and controls to be higher (Tables 1 and 2) than previously determined for subcutaneous MCT. Mean and median AgNOR counts for subcutaneous MCT were reported by one group to be 1.3 and 1.3 per cell. 24 Those researchers used a computerized counting system for AgNOR, whereas we manually counted cells to be able to focus up and down while counting. This may partially explain the difference. That study was also much smaller and had few behaviorally aggressive tumors. Our results for AgNOR were similar to cutaneous MCT, which reportedly vary from 2.3 37 to 1.9, 32 2.2 (for nonrecurring MCT), 2.9 (for recurring), 33 and 3.2 per cell (grade II MCT). 4
This study demonstrates that KIT diffuse cytoplasmic localization is a statistically significant risk factor for aggressive subcutaneous MCT disease. Those dogs with diffuse cytoplasmic localization of KIT receptor protein had significantly increased odds of developing local recurrence and metastasis than did those with a perimembranous pattern (Table 4). The presence of cytoplasmic KIT protein may be associated with downstream events responsible for increased proliferation, 37 but the molecular evidence to further elucidate this for canine MCT is lacking. One possible explanation for diffuse cytoplasmic accumulation in neoplastic mast cells is genetic mutations of c-KIT, resulting in abnormal maturation and trafficking, as found in human gastrointestinal stromal tumors. 34 Autophosphorylated (and thus constitutively activated) immature KIT receptor proteins accumulate in the endoplasmic reticulum and Golgi of mutant c-KIT gastrointestinal stromal tumor cells but not in cells with wild-type c-KIT, where perimembranous localization occurs. 34 Interestingly, treatment with imatinib, a tyrosine kinase receptor inhibitor, restores KIT to the cell surface (perimembranous) in a mature form. 34
None of our 60 subcutaneous MCT demonstrated ITD mutations in exon 11 of c-KIT, despite many of them having aberrant KIT localization and high values for proliferation markers. Given that up to 30% of high-grade cutaneous MCT have these mutations, 6,20 we expected to detect some in our subcutaneous MCT, particularly because many demonstrated biologically aggressive behavior. It is possible that other genetic or epigenetic mechanisms are more important for the neoplastic progression of subcutaneous MCT. Possibilities include undiscovered c-KIT mutations, spontaneous dimerization of receptor tyrosine kinases, or failure of ubiquitinization. 2,18 –20,29,30,39 These mechanisms may explain the diffuse cytoplasmic expression of KIT protein observed in many higher-grade canine cutaneous MCT, and molecular studies investigating this could prove invaluable. Another factor that may play an important role in tumor progression is the local microenvironment, such as angiogenic or growth factor stimuli. 18 –20,30 Because the development of nonneoplastic mast cells is known to be influenced by the surrounding environment, 8 neoplastic mast cells that differentiated within adipose tissue may be phenotypically different from those that arose in the dermis, owing to local growth factor variability, which may result in variability of protein expression profiles or responses to local signals.
There are limitations to retrospective analyses and long-term follow-up studies such as this. Because cases and follow-up information were obtained from primary care veterinary clinics instead of a research center, metastasis and additional tumor occurrence may have been underestimated (if there were silent metastases) or overestimated (by the presence of non-MCT-related disease), given that histologic confirmation was not routinely performed. A prospective analysis or larger retrospective study would be needed to ensure adequate numbers for stricter inclusion criteria.
Proliferation markers including MI and Ki67 are important indices in predicting aggressive behavior of subcutaneous MCT. Aberrant cellular localization of KIT may prove to be an even more valuable prognostic marker if molecular studies are able to identify further genetic causes (eg, additional KIT mutations) or epigenetic causes for this phenomenon. The presence of diffuse cytoplasmic KIT labeling is suggestive of dysregulation, and further testing may indicate that these dogs are amenable to targeted therapy. Based on our results, diffuse cytoplasmic KIT localization may be a highly sensitive but less specific test; however, larger cross-sectional studies (where the prevalence is not predetermined) are needed to verify this and to establish better cutoff values for proliferative indices for comparison. Multivariable analyses of a larger subset of tumors would be helpful in determining how best to use these tests in combination in a diagnostic setting. Finally, c-KIT mutational status (ITD) is unlikely to be a sensitive test for subcutaneous MCT, but it may prove to be a specific marker of aggressive disease.
Footnotes
Acknowledgment
This article is dedicated to the memory of Dr Susan Best, our colleague and friend and a superb veterinary pathologist. We thank the veterinary clinics who participated in this study for providing follow-up information for dogs. We would also like to acknowledge both the Ontario Veterinary College Pet Trust Organization and the Rouse Family Foundation for funding this study.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded by the Ontario Veterinary College Pet Trust Organization and the Rouse Family Foundation.
