Abstract
Histologic grading schemes for canine cutaneous mast cell tumors (MCTs) were not developed for subcutaneous MCTs. Despite this, subcutaneous MCTs are currently categorized by many as grade II or higher. The aim of this investigation was to assess the pathology and clinical outcome for subcutaneous MCTs to provide a more accurate prognosis. Information on clinical outcome for 306 dogs was obtained from veterinarians and correlated with histologic features. Mean and median follow-up was 842 and 891 days, respectively (range, 3–2,305 days). Only 27 (9%) were confirmed as mast cell–related deaths. Metastasis occurred in 13 (4%), and 24 (8%) had local reoccurrence, even though 171 (56%) cases had incomplete surgical margins. Median survival time was not reached, and the estimated 6-month, 1-, 2-, and 5-year survival probabilities were 95%, 93%, 92%, and 86%, respectively. Dogs were euthanized or died as a result of local tumor reoccurrence, additional MCT development distant to the surgical site, or metastasis. Decreased survival time was linked to mitotic index (number of mitotic figures per 10 high-power fields), infiltrative growth pattern, and presence of multinucleation. Both univariable and multivariable analysis showed mitotic index to be strongly predictive of survival, local reoccurrence, and metastasis. The results of the study indicate that the majority of subcutaneous MCTs have a favorable prognosis, with extended survival times and low rates of reoccurrence and metastasis.
The majority of canine mast cell tumors (MCTs) occur in the skin and subcutis. Many MCTs originate in the dermis and extend into the subcutis, but there is a subset that is restricted to the subcutaneous fat. Only one publication has described this type. 33 Survival times were prolonged, and there was a low rate of reoccurrence and metastasis, but the number of cases was too small to provide accurate prognostic data. The seminal work by Patnaik et al, 37 which formed the basis of the commonly used grading scheme, did not include subcutaneous MCTs. Many pathologists include subcutaneous MCTs with cutaneous MCTs and ascribe them as being grade II because of their subcutaneous location. 18,37 Intermediate (grade II) tumors have a highly variable prognosis, and low interobserver agreement among pathologists has been reported. 34,35
Many studies of cutaneous MCTs attempt to establish better prognostic schemes; however, there is still no uniform way to prognosticate the majority of these tumors. Studies have examined numerous predictors—including location, 18,48 surgical margins, 11,30,36,46,47,50 mitotic activity, 10,43 nuclear morphometry, 28,51 and vascular density, 40 as well as molecular markers, including immunohistochemical assays for proliferation markers (Ki-67, 1,27,36,44,45,52 PCNA, 1,27,45,49,52 and AgNOR 5,19,27,45,49,52 ), KIT expression, 15 KIT immunohistochemical staining pattern, 17,52 and vascular endothelial growth factor expression. 41 Genetic mutations of c-KIT, a gene encoding the tyrosine kinase receptor KIT, may be responsible for the progression of some of these tumors. 9,20 –22,24 –26,42,52,55 Up to 30% of dogs with cutaneous MCTs have mutations in the juxtamembrane domain of c-KIT, 9,21,22,55 and their presence is associated with higher-grade tumors. 55 Mutations result in constitutive activation of KIT, initiating signaling cascades leading to cancer cell proliferation, survival, and invasion. 9,21,22,25,26 Because more than 70% of dogs do not have identified mutations, this marker has limited predictive value.
Some publications combine subcutaneous MCTs with cutaneous MCTs. 18,39 If subcutaneous MCTs have a different prognosis, the data generated from such studies could be inaccurate. The aim of this investigation is to separately evaluate a large number of subcutaneous MCTs to characterize their behavior, histology, and clinical prognosis.
Materials and Methods
Case Selection
In sum, 550 subcutaneous MCTs from 354 veterinary clinics diagnosed from 2002 to 2008 were obtained from the pathology archives of Yager-Best Histovet, Histologic and Cytological Services, Guelph, Ontario, Canada.
Cases were included if they met the following criteria: First, the tumor was a primary occurrence; second, all were histologically diagnosed as subcutaneous MCTs on the basis of adequate representation of the tumor; and third, adequate follow-up data were obtained from veterinary clinics in the form of a questionnaire or telephone interview. MCTs were determined to be subcutaneous on the basis of a location within the subcutaneous tissue and no invasion of the dermis. Two or more separate histologic sections of each tumor were examined to ensure this. In some cases, there was apparent, multifocal extension of low numbers of mast cells around the base of hair follicles, and in other cases, mast cells infiltrated the underlying panniculus musculature; however, the bulk of the tumor was in the subcutaneous tissue. In cases where the overlying epithelium was not present, tumor sections were completely surrounded by adipose tissue with no follicular or epidermal involvement. In addition, the pathology report diagnosing each tumor as a subcutaneous MCT was available for each tumor.
Follow-up information included signalment, tumor location, dates of additional tumor development, metastasis, death or last examination, history of prior MCT, cause of death, and status at last examination. Table 1 presents summary statistics for clinical outcomes. Additional information included details on adjunctive surgery (if applicable) and further diagnostic testing. Cases were excluded if there was immediate loss to follow-up, if there was history of a previous MCT, if the sample was an incisional biopsy or cytoreductive, or if the patient had concurrent MCT that was cutaneous.
Dogs With Various Clinical Outcomes and Stage of Subcutaneous Mast Cell Tumors (N = 306)
a Mast cell tumor regrowth at the original surgical site.
b Subsequent mast cell tumor development at a site distant to the original surgery site.
The date of surgical excision was defined as the date of diagnosis. Follow-up time was defined as the time from date of diagnosis to date of last follow-up or death. Local reoccurrence was defined as regrowth at the surgical site, and distant occurrence was defined as occurrence of a subsequent cutaneous or subcutaneous MCT at an anatomic location different from that of the initial surgery. Metastasis was defined as spread to the local lymph node or as disseminated disease (ie, to internal organs). Metastasis was determined by physical examination and at least one of the following: cytology of fine-needle aspirates, histology, surgery, ultrasound, magnetic resonance imaging, and buffy coat analysis. Only 3 dogs had histologic confirmation of metastasis, and postmortem examination was not performed on any dogs. We chose to treat these cases not confirmed by histology as metastases because we did not want to bias our cases to include only tumors with favorable outcomes.
Disease-free interval (DFI) was defined as the time between date of diagnosis to the date of local or distant MCT development or metastasis, whichever occurred first. Dogs lost to follow-up, healthy dogs, and dogs that died from causes unrelated to mast cell disease were right-censored and included for survival analysis. Dogs that died or were euthanized because of unknown causes were right-censored for analysis of risk factors; however, sensitivity analysis was performed, treating these as events to assess a worst-case scenario (ie, assumed to have died from MCT). Median survival time (MST) was defined as the time at which 50% of dogs were alive, and median DFI was defined as the time at which 50% of dogs had no MCT-related disease.
Evaluation of Histologic Variables
Histologic features were evaluated in a blinded fashion and included confirmation of subcutaneous location and examination of surgical margins. The completeness of surgical margins was described as “incomplete” if neoplastic cells were at or within 1 mm of the margin. For larger tumors, at least four peripheral sections of marginal tissue were available. All tumors were nonencapsulated, composed of neoplastic mast cells on a background of variably dense collagenous tissue stroma and preexisting adipose tissue, admixed with variable numbers of eosinophils.
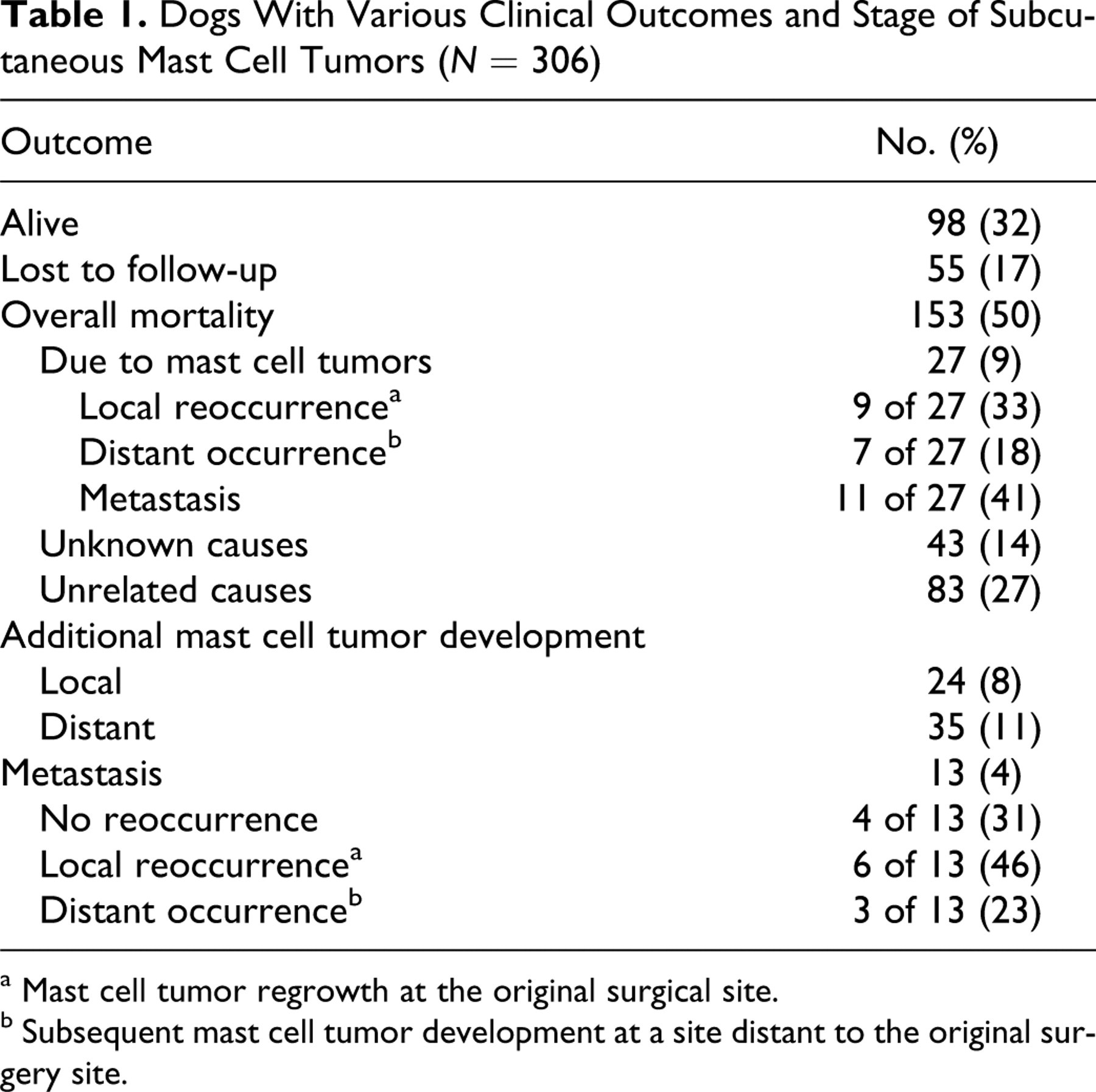
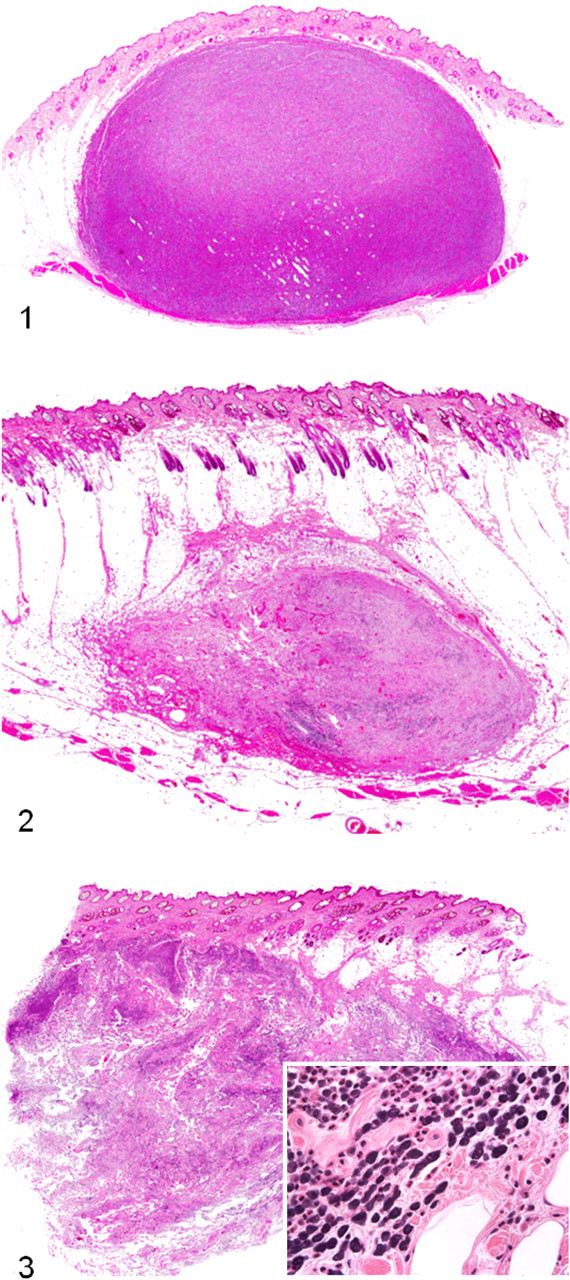
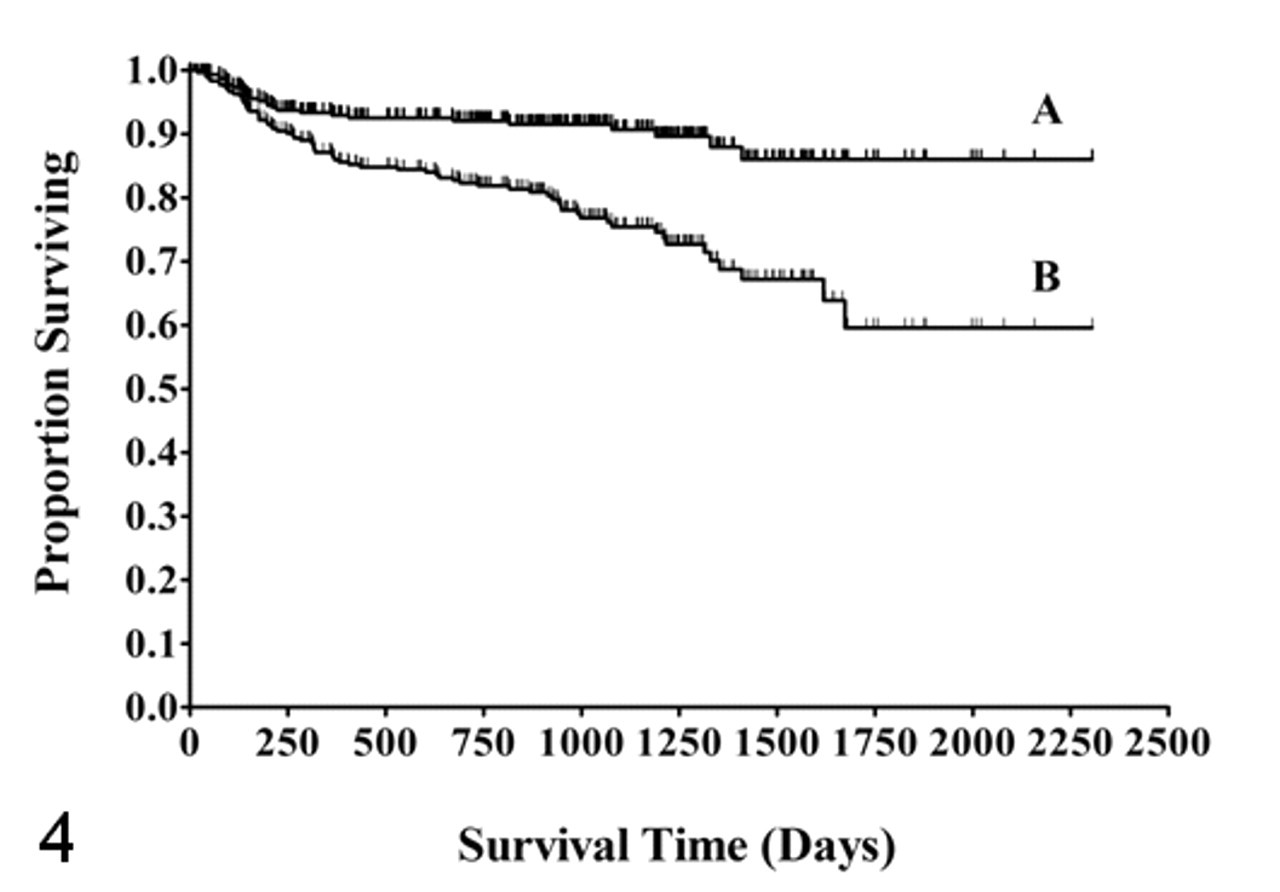
Histologic appearance was recorded as one of three growth patterns: circumscribed, combined, or infiltrative, as assessed by subgross (4× objective) histologic examination (Figs. 1–3). Circumscribed tumors (n = 53) were grossly well demarcated, densely cellular, and expansile, elevating the overlying dermis and epidermis; in some cases, they extended to the underlying panniculus musculature (Fig. 1). Combined (infiltrative–demarcated) tumors (n = 90) consisted of solitary nodules formed by neoplastic cells arranged in variably dense aggregates and rows and as single cells demarcated from the surrounding normal tissue by abundant collagenous fibrous connective tissue (Fig. 2). Infiltrative tumors (n = 163) were composed of neoplastic cells arranged as in combined tumors, but they lacked demarcation (Fig. 3). Table 2 summarizes quantitative histologic features and qualitative (recorded as present or absent). Slides were assessed on the basis of areas of greatest variability of anisocytosis and anisokaryosis and greatest degree of multinucleation. Anisocytosis and anisokaryosis were recorded as present if there was more than a 2-fold change in at least 10% of 100 cells. Multinucleation (more than 1 nuclei) was recorded as present if there was at least 1 multinucleated cell in 10 high-power fields (400×). Cytoplasmic granules were recorded as absent if there were no discernible granules within neoplastic cells in the entire section. Except for mitotic index (MI), quantification was not performed, because many attributes often varied widely by region and were influenced by cellular integrity or plane of section. The MI was defined as the number of mitotic figures per 10 high-power fields and recorded for each tumor using the method described by Romansik et al. 43 The area with the highest mitotic activity was chosen for evaluation. For the cases that had multiple subcutaneous tumors (n = 16), the tumor with the highest MI was included for evaluation of risk factors.
Dogs With Tumors Having Various Histologic Features of Subcutaneous Mast Cell Tumors (N = 306)

a No. of mitotic figures per 10 high-power fields.
Statistical Analysis
All statistical analyses were performed with SAS 9.1. Risk analyses were performed with Cox proportional hazards models. MST and median DFI were calculated with the Kaplan-Meier product limit survival method. Cox proportional hazards models were generated for the outcomes of survival, DFI, and time to local and distant tumor occurrence and metastasis. Only those risk factors having a P value of < .20 in univariable analysis were included in multivariable model building; breed, sex, age at diagnosis, and tumor location were initially forced into all models because they were believed to be important confounding variables. Backward selection methods were used to create a fixed effects model, retaining only those values that had a P value of < .05. Each model was then tested in a stepwise fashion for pairwise interactions of significant risk factors and to assess for confounding variables. Results are reported as hazard ratios: Values for categorical risk factors are interpreted as the ratio of the predicted hazard of one group relative to a referent group (ie, presence vs absence of risk factor). Hazard ratios for continuous predictors (eg, age at diagnosis) represent the effect of a unit of change in the risk factor on the hazard of the outcome. Predicted survival curves were generated on the basis of multivariable models using the baseline option in PROC TPHREG, using referent covariates.
MI was categorized on the basis of quantiles that provided adequate sample size for subsequent analyses. Risk factors were tested for validity of the Cox proportional hazards assumption by graphically assessing log-cumulative hazard plots and examination of Schoenfeld residuals. Evaluation of the functional form of the relationship between continuous predictors (age and MI) and survival was assessed using Martingale residuals generated from the null model and then verified with the final model and plotted against the predictor, using a smoothing function. We assessed the correlation among risk factors using Spearman-Rank coefficients to make certain that no variables in the models were highly correlated (ie, > 60%).
Results
Sixty-two percent (221 of 354) of veterinary clinics returned questionnaires for 367 of 550 (67%) dogs. Sixty-one cases (17%) were excluded from the study because of immediate loss of follow-up (n = 20), because of history of prior MCT (n = 26), because samples were incisional (n = 6) or cytoreductive (n = 1) biopsies, or because dogs had concurrent cutaneous MCTs (n = 7). A total of 306 cases fulfilled the inclusion criteria, and for all these, dates of last follow-up, subsequent MCT development, and metastasis were available. The median and mean age at time of diagnosis was 8 years and 8 years, 2 months, respectively, with a range of 1 year and 5 months to 18 years. Age was not recorded in one case. There were 54 breeds consisting of mixed breeds (n = 72), Labrador Retrievers (n = 63), Boxers (n = 25), and other purebred dogs (n = 145), which included Golden Retrievers (n = 15), Cocker Spaniels (n = 14), Jack Russell Terriers (n = 9), Pugs (n = 8), Lhasa Apsos (n = 7), Beagles (n = 6), and Shetland Sheepdogs (n = 5). Breeds were consolidated into 4 categories based on the largest groups (Labrador Retrievers, Boxers, mixed breed, and other purebred) to create categories of an adequate size for statistical analyses. The breed of one animal was unknown. There were 7 intact females, 7 intact males, 173 spayed females, and 116 neutered males. Sex was unknown for 3 dogs. Because there were few intact dogs, sex was dichotomized into female and male. Tumor location included extremities (n = 104), thorax (which included back; n = 80), abdomen (n = 45), head/neck (n = 37), and inguinal/perineal (n = 20). Location was not known for 20 cases. Sixteen dogs had multiple subcutaneous MCTs (up to 3). Five dogs received chemotherapy during the study period, 3 of which had widespread metastasis and were treated with chemotherapy for 1, 76, and 155 days before death. One dog was diagnosed with an additional (distant) MCT and disseminated metastasis 1,184 days following the initial surgery and was healthy at the last date of examination, 21 days later. The last dog was diagnosed with a distant MCT 1,001 days after the initial surgery and was treated with chemotherapy up until the date of last examination (427 days later), despite there being no evidence of metastasis. Because there were too few dogs, multivariable analyses could not be performed to account for the effect of chemotherapy. However, models were reanalyzed excluding these cases, and this did not change the significance of the outcomes of any models.
Clinical Outcomes
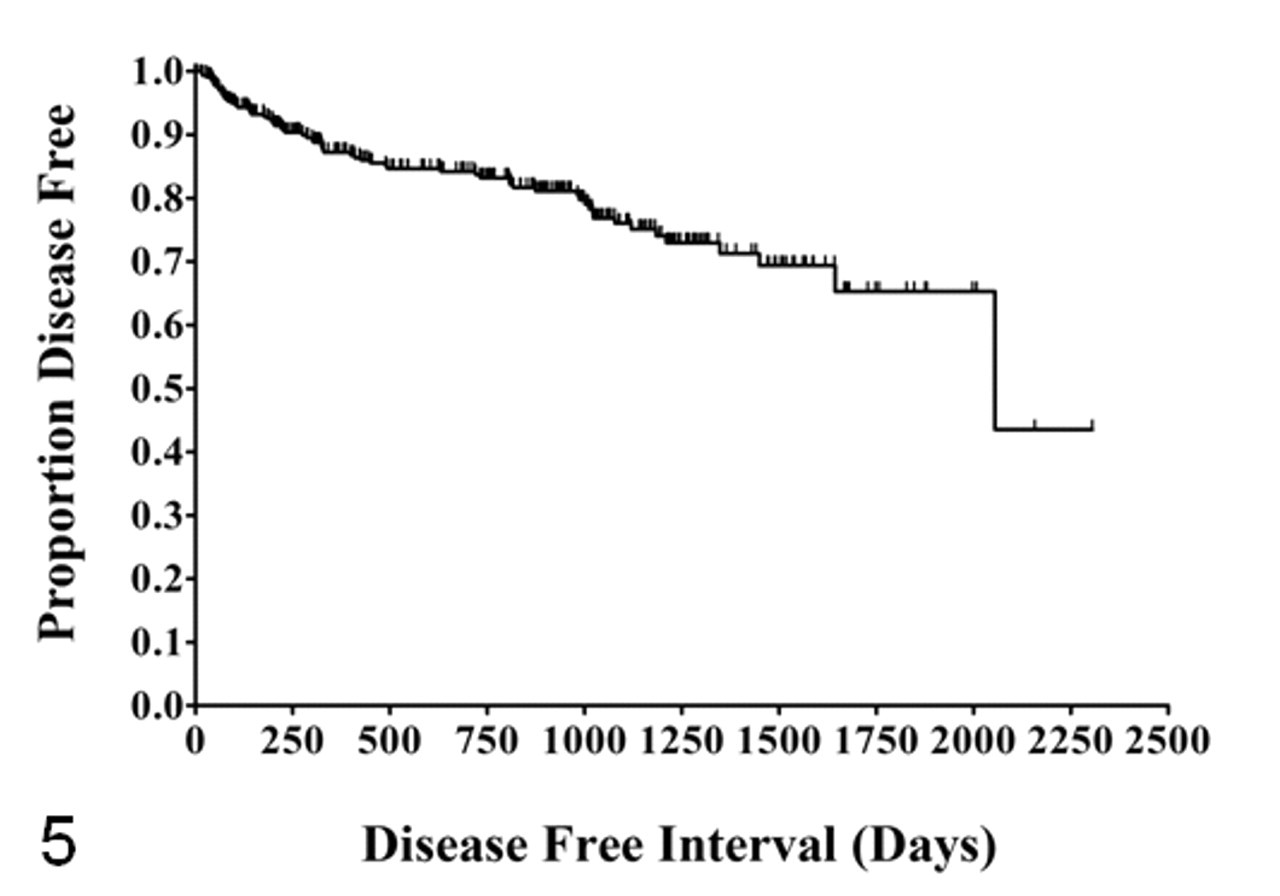
Table 1 summarizes the outcomes for dogs. Follow-up time ranged from 3 to 2,305 days, with a median and mean of 891 and 848 days, respectively. Sixty-one percent of dogs (186 of 306) had follow-up times greater than 2 years. The shortest follow-up time (3 days) was for a dog euthanized for an unrelated cause (ruptured cruciate ligament) following surgery for MCT. The MST for all dogs was not reached, and the 6-month, 1-, 2-, and 5-year estimated survival probabilities were 95%, 93%, 92%, and 86% (Fig. 4, curve A) with 84% of dogs estimated as surviving at 1,500 days. Kaplan-Meier survival curves were also generated ascribing deaths due to unknown causes as mast cell disease (ie, were not censored). The 6-month, 1-, 2-, and 5-year estimated survival probabilities were 92%, 87%, 82%, and 59%, respectively, with 67% predicted survival at 1,500 days (Fig. 4, curve B).
Over the study period, 153 of 306 dogs died (50%). Mortality due to confirmed mast cell disease was low (9%), consisting of 27 of 306 dogs. Dogs were euthanized or died because of metastasis and additional MCT development. Local occurrence and distant occurrence were 8% (n = 24) and 11% (n = 35), respectively, and metastasis occurred in 4% of dogs (n = 13) and involved the local lymph node (n = 5) or dissemination (n = 8). The median estimated DFI for all 306 dogs was 2,055 days, and the 6-month, 1-, 2-, and 5-year estimated disease-free probabilities were 92%, 87%, 84%, and 65%, respectively (Fig. 5 ).
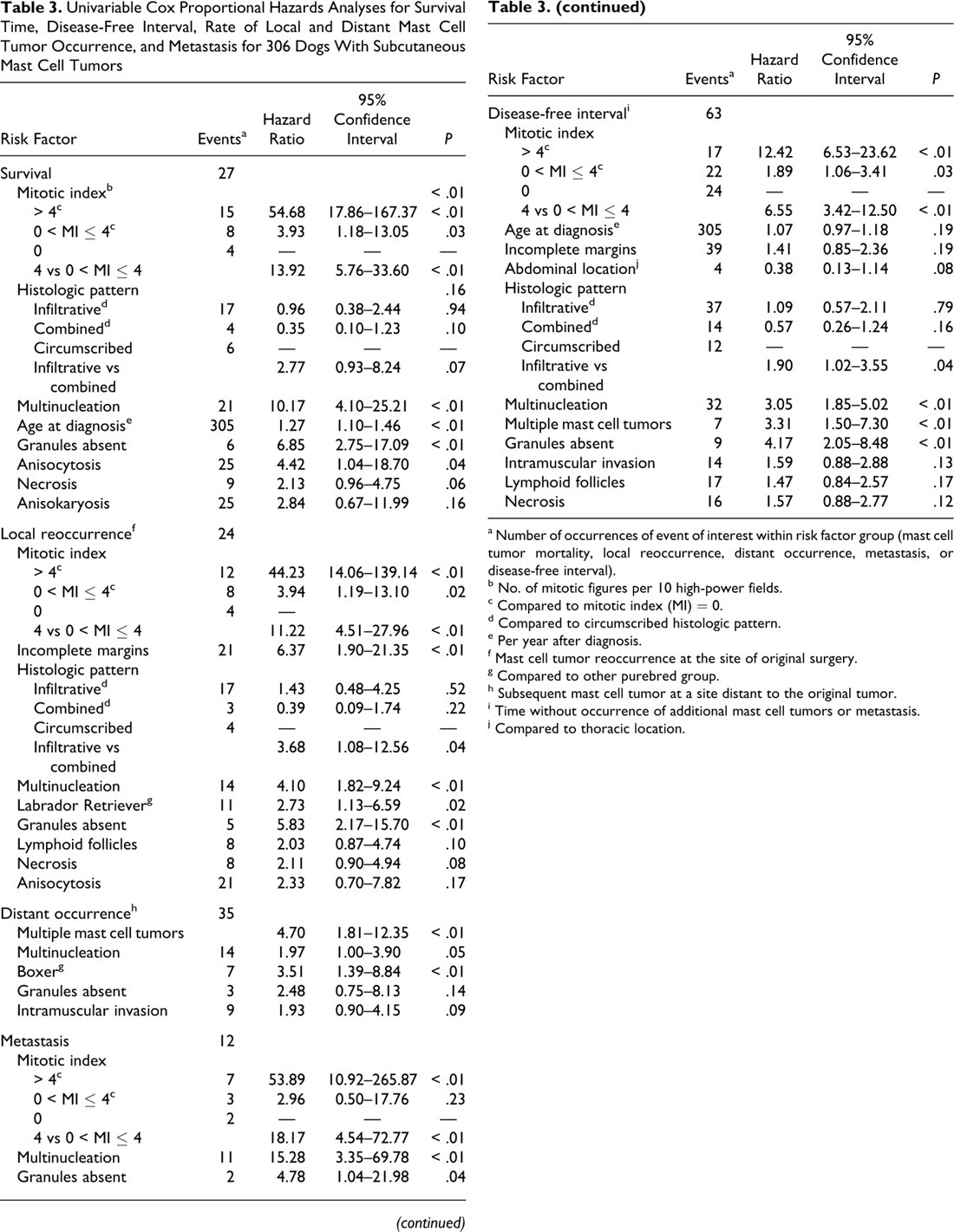
Tables 3 and 4 present the univariable and multivariable Cox proportional hazard analyses for time to outcome. MI had the largest coefficient among significant risk factors associated with decreased DFI and increased rate of MCT-related mortality, local reoccurrence, and metastasis. The distribution of MI was highly skewed, with 60% (185 cases) having MI = 0 (Table 2). The median MI (50% quantile) and 75% and 90% quantiles were 0, 1, and 4, respectively; thus, fewer than 10% (28 of 306) had MI > 4, and yet these cases represented 50% (n = 12), 54% (n = 7), and 56% (n = 15) of local reoccurrences, metastases, and MCT deaths (Tables 3 and 4). Cases were stratified into groups based on MI quantiles for risk factor analyses: Group 1 (n = 185) had tumors with MI = 0; group 2 (n = 93), MI = 1–4 (0 < MI ≤ 4); and group 3 (n = 28), MI > 4.
Univariable Cox Proportional Hazards Analyses for Survival Time, Disease-Free Interval, Rate of Local and Distant Mast Cell Tumor Occurrence, and Metastasis for 306 Dogs With Subcutaneous Mast Cell Tumors
a Number of occurrences of event of interest within risk factor group (mast cell tumor mortality, local reoccurrence, distant occurrence, metastasis, or disease-free interval).
b No. of mitotic figures per 10 high-power fields.
c Compared to mitotic index (MI) = 0.
d Compared to circumscribed histologic pattern.
e Per year after diagnosis.
f Mast cell tumor reoccurrence at the site of original surgery.
g Compared to other purebred group.
h Subsequent mast cell tumor at a site distant to the original tumor.
i Time without occurrence of additional mast cell tumors or metastasis.
j Compared to thoracic location.
Multivariable Cox Proportional Hazards Models for Survival Time, Disease-Free Interval, Rate of Local and Distant Mast Cell Tumor Occurrence, and Metastasis for 306 Dogs With Subcutaneous Mast Cell Tumors

a Number of occurrences of event of interest within risk factor group (mast cell tumor mortality, local reoccurrence, distant occurrence, metastasis, or disease-free interval).
b No. of mitotic figures per 10 high-power fields.
c Compared to mitotic index (MI) = 0.
d Compared to circumscribed histologic pattern.
e Per year after diagnosis.
f Mast cell tumor reoccurrence at the site of original surgery.
g Subsequent mast cell tumor at a site distant to the original tumor.
h Time without occurrence of additional mast cell tumors or metastasis.
Survival
Significant risk factors for decreased survival time, based on multivariable analysis, were MI, histologic pattern (infiltrative vs well circumscribed), the presence of multinucleation, and age at diagnosis (Table 4). The largest coefficient in the model was MI. Increased hazard of MCT-related mortality was associated with increasing MI; rates of MCT-related death for dogs with tumors having MI > 4 and 0 < MI ≤ 4 were 36.05 (P < .01) and 3.72 (P = .03) times higher than for those with MI = 0, and rates for the group MI > 4 were 9.70 (P < .01) times higher than for the 0 < MI ≤ 4 group (Table 4).
Infiltrative tumors had 3.18 times (P = .02) higher rates of MCT mortality than well-circumscribed tumors, according to multivariable analysis (Table 4); however, there was no significant difference in survival between infiltrative and combined patterns (P = .18) or between combined and circumscribed tumors (P = .58). Multinucleation was also significantly associated with decreased survival times (hazard ratio = 3.40; P = .02), and age at diagnosis increased the MCT-related mortality rate by 1.20 per year.
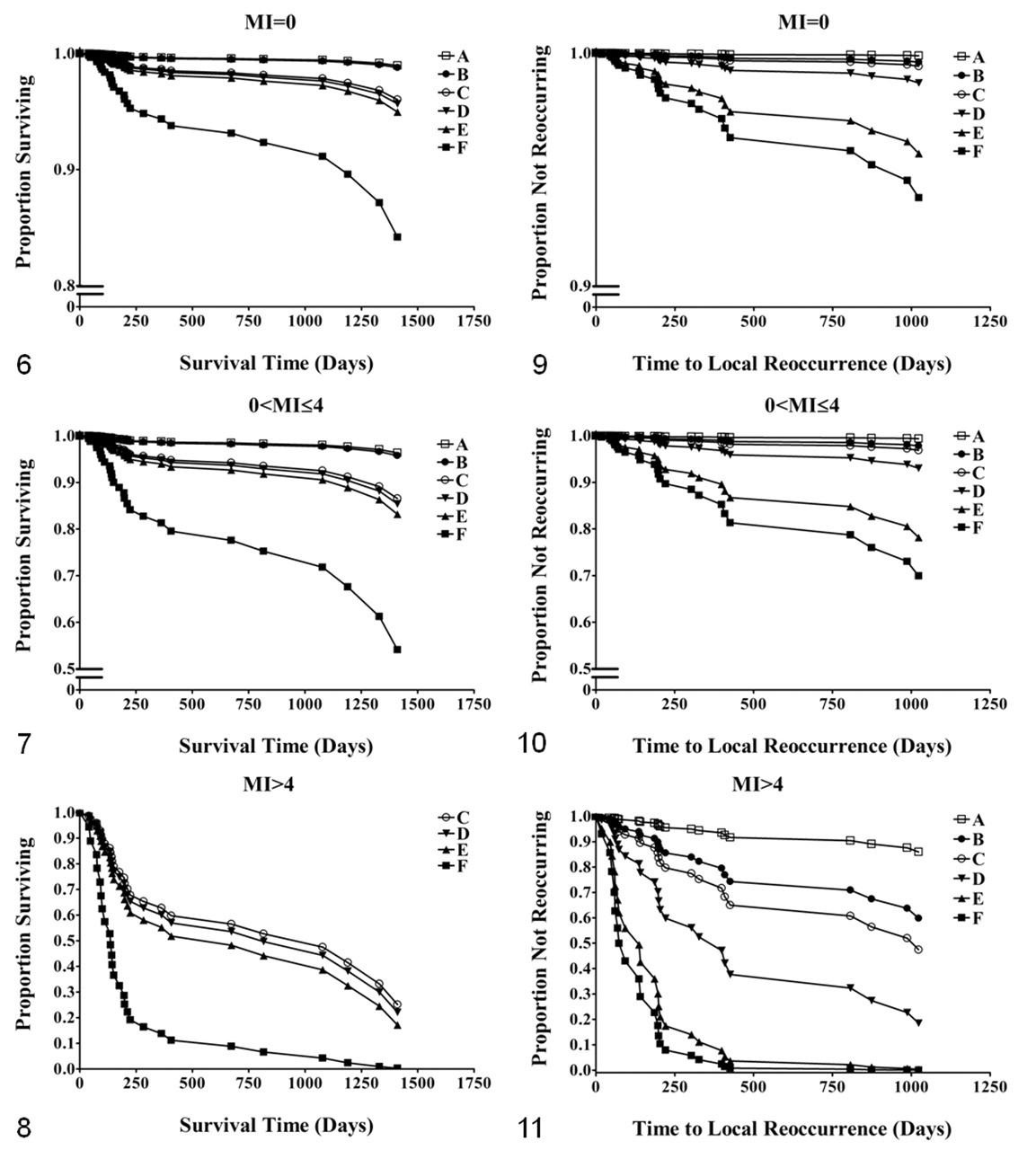
Risk factor–adjusted predicted survival curves were generated on the basis of multivariable analysis (Figs. 6–8), as stratified by MI. Predicted survival times are decreased for increasing MI, controlling for the presence of multinucleation and histologic pattern. Having multiple risk factors resulted in the worst predicted survival times (Fig. 8). For cases having MI > 4, the largest decreases in survival are seen regardless of the other combinations of covariates. The predicted MST for dogs with infiltrative tumors having MI > 4 and multinucleation is 140 days (Fig. 8, curve F), compared to 950 days for those having MI > 4, no multinucleation and infiltrative pattern (Fig. 8, curve C).
Local Reoccurrence
Tumors locally reoccurred in Labrador Retrievers (n = 11), mixed breeds (n = 4), and other purebred dogs (n = 9) and were located on the extremities (n = 11), trunk/thorax (n = 9), and head/neck area (n = 4). Boxers (n = 25) did not experience any local reoccurrence. Location was not a significant risk factor for rate of local reoccurrence. Median time to local reoccurrence for these dogs (n = 24) was 198 days (95% confidence interval [CI], 93–327) with a range of 18 to 1,023 days; the MST of these dogs was 283 days (142–open-ended). Six dogs with local reoccurrence developed metastasis consisting of lymph node (n = 4) or disseminated metastasis (n = 2), and 15 died from their disease. Two dogs were euthanized at the time of reoccurrence; 9 dogs had additional surgery; and 13 continued to live with the tumor (no surgery). Kaplan-Meier survival curves were generated to compare the effect of additional surgery on survival for these dogs. Excluding those that died at the time of reoccurrence, log-rank statistical analysis showed no significant difference in survival between those that had additional surgery and those that did not (P = .31).
Tables 3 and 4 present univariable and multivariable statistics for risk factors associated with time to local reoccurrence. Significant risk factors based on multivariable analysis were MI > 0, incomplete surgical margins, and infiltrative histologic pattern (Table 4). The majority (n = 20) of tumors that reoccurred had MI > 0, consisting of MI > 4 (n = 12) and 0 < MI ≤ 4 (n = 8). Dogs having tumors with MI > 4 and 0 < MI ≤ 4 had rates of local reoccurrence that were 130.21 times (P < .01) and 5.59 times (P < .01) higher than dogs with MI = 0; the rate was 23.31 times higher for MI > 4 compared to 0 < MI ≤ 4 (P < .01).
Tumors with incomplete surgical margins (n = 171) reoccurred sooner than those with complete margins (hazard ratio = 11.19; P < .01). Tumors with incomplete margins reoccurred in 12% of tumors (21 of 171). Infiltrative tumors (n = 163) had 4.94 times higher rates of reoccurrence (P < .01) than did circumscribed ones (n = 53); however, similar to the survival outcome, there was no difference in local reoccurrence rate between infiltrative and combined patterns (P = .58) or between combined and circumscribed (P = .14). This suggests that the combined growth pattern has intermediate behavior between circumscribed and infiltrative patterns, as supported by predicted time to reoccurrence curves (Figs. 9–11). Predicted time to reoccurrence curves show that the relative effects of incomplete margins and histologic pattern are similar for each strata of MI but that the prognosis is much poorer for those with MI > 4 (Fig. 11). The predicted time to local reoccurrence for cases having incomplete margins and infiltrative pattern (Fig. 11, curve F) is 70 days, compared to 1,000 days for completely excised infiltrative tumors (Fig. 11, curve C) and 365 days for incompletely excised circumscribed tumors (Fig. 11, curve D).
Distant MCT Occurrence
Additional tumor development distant to the surgical site occurred in mixed-breed dogs (n = 8), Boxers (n = 7), Labrador Retrievers (n = 7), Pugs (n = 3), Cocker Spaniels (n = 3), and 7 other purebred dogs (n = 1 of each breed). Median time to distant occurrence was 450 days (95% CI, 275–808) with a range of 17 to 2,055 days; the MST for these dogs was not reached. Nine dogs with distant occurrence died or were euthanized because of metastasis (n = 2) or the occurrence itself (n = 7). Five dogs were euthanized at the time of occurrence; 21 dogs had additional surgery; and 9 continued to live with the tumor. Kaplan-Meier curves were used to compare survival of dogs that had additional surgery with those that did not, excluding cases that were euthanized for the occurrence. Additional surgery significantly improved survival (log-rank, 17.4984; P < .0001): The MST for dogs with distant occurrences without additional surgery was 68 days (95% CI, 33–not reached) compared to dogs with surgery (MST not reached). Of the dogs that did not have surgery, 4 were eventually euthanized for the occurrence; 2 metastasized; 5 died of unknown /unrelated causes; and 1 was lost to follow-up. Of those that had additional surgery, only 1 dog died from MCT (metastasis). Twelve were alive at the end of the study; 6 died of unknown/unrelated causes; and 2 were lost to follow-up.
Risk factors significantly affecting the rate of distant MCT occurrence, based on multivariable analyses, were the presence of multiple MCTs and multinucleation (Table 4). MI was not a significant risk factor. Dogs with multiple MCTs (n = 16) had rates of distant occurrence that were 5.62 times (P < .01) higher than that for dogs with a single MCT; multinucleation increased the rate by 2.24 times (P = .02) compared to those with none (Table 4). According to univariable analysis (Table 3), Boxers had significantly increased rates of distant MCT occurrence, but this was not significant in multivariable analysis.
Metastasis
Metastasis occurred in 13 of 306 dogs (4%). Metastasis was diagnosed postoperatively in 12 dogs and at the time of the initial surgery in 1 case (local lymph node). The latter dog died of unrelated causes 1,644 days after excision of both tumor and node. This case was excluded for risk factor analysis for the outcome of metastasis, but it was included for all other analyses, given that exclusion did not affect these outcomes. Metastasis occurred in 6 dogs following local reoccurrences. Lymph node (n = 4) and disseminated disease (n = 2) were the sites. Three dogs had widespread metastasis after MCTs were found at distant sites. For these 3 cases, it was not possible to determine which tumor metastasized (the initial or subsequent MCT). Three dogs had disseminated metastasis with no prior reoccurrence and were euthanized 41, 144, and 816 days after the initial surgery. The only significant risk factor for decreased time to metastasis using multivariable analysis was MI > 4 (Table 4). Multinucleation and absence of granules were other significant risk factors in univariable analysis (Table 3); however, these did not remain significant in multivariable analysis. The rate of metastasis for dogs with tumors having MI > 4 was 53.89 times greater (P < .01) than for MI = 0 and 18.17 times higher (P < .01) than for those having 0 < MI ≤ 4 (P < .01); however, there was no significant difference between the 0 < MI ≤ 4 and MI = 0 groups (P = .23). The MST of dogs with metastasis was 212 days (95% CI, 142–406), which was significantly decreased (P < .01; log-rank statistic) when compared with dogs that did not have metastasis (MST not reached).
Disease-Free Interval
Table 4 summarizes significant risk factors affecting DFI. MI had the largest coefficient for decreased DFI, and risk was correlated with increasing MI: Tumors with MI > 4 had 11.13 (P < .01) and 5.80 (P < .01) higher rates of MCT-related disease than those having M = 0 and 0 < MI ≤ 4, respectively, and the rate was 1.92 times higher for 0 < MI ≤ 4 compared to MI = 0 (P = .04). The presence of multiple MCTs at the time of diagnosis (P < .01) and multinucleation (P = .02) decreased the DFI when compared to cases that lacked these features. Infiltrative tumors had significantly higher rates of MCT disease (local reoccurrence, distant MCT occurrence, and metastasis) than both circumscribed (P = .02) and combined (P = .02) tumors, but there was no difference between combined and circumscribed ones (P = .83).
The DFI is a potentially more relevant outcome than survival because multiple confounding factors, including age and reasons for euthanasia (eg, financial constraints or perception of quality of life by owners), do not influence statistical analyses. These factors, in addition to the increased number of events (additional MCT and metastases), are reflected by the relatively narrowed CIs compared to values obtained in the other models.
Discussion
The majority of dogs with subcutaneous MCTs had extended survival times, DFI, and lower rates of local reoccurrence (8%) and metastasis (4%) than did those reported for grade II cutaneous MCTs. Estimated 6-month, 1-, 2-, and 5-year survival probabilities were 95%, 93%, 92%, and 86%, respectively, with 84% of dogs estimated to be alive at 1,500 days. Of the 306 cases, less than 10% of dogs died from confirmed mast cell disease, and when dogs that died from unknown causes were attributed to mast cell disease (ie, worst-case scenario), mortality was only 23% and survival was estimated to be 67% at 1,500 days. Our findings are similar to those reported by Newman et al, 33 who found that of 53 dogs with subcutaneous MCTs, there was a low rate of reoccurrence (9%) and metastasis (6%), and survival rates were 61% at 1,206 days, 30% at 1,392 days, and 0% at 1,780 days (last date of follow-up). Published survival rates of dogs with grade II/intermediate cutaneous MCTs (which may have included subcutaneous types) include 1,500-day survival of 44%, 37 20-month (610 days) survival of 16%, 1 and MSTs of 28 weeks (196 days). 4 Local reoccurrence rates are reported as 11%, 53 23%, 46 and 33%, 49 and mortality rates vary from 17% 3 to as high as 56%. 37
Our results indicate that subcutaneous tumors are more effectively controlled by surgery alone than their cutaneous counterparts. Surgery is curative for the majority of these tumors. Of tumors with complete margins (n = 135), only 3 (2%) locally reoccurred; thus, surgical excision alone was effective in local control of 98% of cases. This is higher than that of published studies 47,53 for completely excised grade II cutaneous MCTs, which report a local reoccurrence rate of 11% and, thus, effective control in 89% of cases. Of the incompletely excised tumors in our study (n = 171), only 21 reoccurred (12%). This is lower than that reported for incompletely excised grade II cutaneous MCTs treated by surgery alone (23%) 46 and the same as for those treated with adjuvant radiation (12%). 2
One theory regarding why some MCTs do not reoccur following incomplete removal is related to postsurgical healing. Inflammatory cells recruited to the surgical site, in conjunction with release of cytokines or disturbance of vasculature, may play a role in killing remaining neoplastic cells. 23 Alternatively, it may be related to heterogeneity of cells within the tumor. For example, there is recent evidence to suggest that hypoxic gradients within tumors provide stem cell “niches” within central areas of glioblastoma, and these central cells are more resistant to chemotherapy and radiation. 38 It may be that neoplastic cells from the periphery of the tumor may represent a more committed cell type than those located centrally, and they cannot sustain their survival.
Our study included only dogs with primary subcutaneous MCTs, and they mostly came from primary care veterinary clinics. Retrospective studies conducted at referral centers are based on higher-grade or reoccurring tumors, which require specialized surgery and treatment, so their results are potentially biased toward the more aggressive neoplasms. It is possible that by selecting only those MCTs that clearly arose in the subcutis, smaller tumors may have been overrepresented; that is, larger MCTs may have progressed to invade the overlying tissue and would have thus been indistinguishable on routine histologic sections from a cutaneous MCT with downward extension. Additionally, some tumors may have been sectioned obliquely, and despite every attempt to ensure a subcutaneous origin, cutaneous involvement could have been missed in some cases. Despite these possible biases, we show that MCTs with the criteria outlined here have a low metastatic and local reoccurrence rate.
It is possible that subcutaneous MCTs may be less aggressive than cutaneous tumors because of the surrounding fat. In recent years, adipose tissue has been intensively investigated as an endocrine organ because it is important in human disease, such as type-2 diabetes, atherosclerosis, and cancer. 6 –8 Cytokines, growth factors, and chemokines (collectively referred to as adipokines) produced by adipocytes and stromal cells (including fibroblasts and nonneoplastic mast cells) influence disease and neoplastic progression through effects on endocrine balance, inflammation, and immunity. 6,7,32 Additionally, nonneoplastic mast cell development is known to be influenced by the tissue microenvironment. 14 Mast cells are unique because they are released from the bone marrow as hematopoietic precursor cells and fully differentiate within tissues, as influenced by signals from the local microenvironment. 12 –14 MCTs that arose within adipose tissue may have acquired different phenotypic properties than did those arising within the dermis, resulting in less aggressive behavior than that of grade II cutaneous MCT with downward expansion. Molecular studies would be invaluable in elucidating these effects.
The results of this study show that the strongest effect on clinical outcome is related to mitotic activity. Dogs with MI > 4 had significantly shorter survival times and decreased time to local reoccurrence and metastasis than did those with MI < 4. This finding is consistent with numerous publications that showed cellular proliferation to be associated with poorer outcomes for dogs with cutaneous MCTs. 10,39,43 Two studies have evaluated MI in cutaneous MCTs as a sole prognostic indicator. 10,43 One of these studies reported MSTs of 5 months for dogs bearing tumors with MI > 5 (n = 19) compared to 70 months for those with MI ≤ 5 (n = 80), regardless of grade. 43 A second study, 10 consisting of 57 dogs, showed similar results using the same cutoff for MI but also found that stratification of MI into 3 groups (MI = 0, 1 ≤ MI < 7, and MI > 7), based on the cutoff points of 1 and 7 had superior prognostic potential. The most likely reason for the discrepancy between cutoff values is due to the small number of cases in these studies, which makes statistical comparison between strata difficult. In our study, individual MI values were often represented by one case, and the relationship between MI and the log hazard of the various outcomes being investigated was not linear, so MI could not be modeled as a continuous variable. Consequently, we based our cutoff values on quantiles that provided adequate sample sizes in each group for statistical comparison.
Curves for predicted survival (Figs. 6–8) and local reoccurrence (Figs. 9–11) show that although MI is a useful prognostic factor, as a sole test it should be interpreted with caution because effects of other histologic risk factors have a significant impact. When MI is a sole predictor, the estimated MST (Kaplan-Meier analysis) for dogs having tumors with MI > 4 is 212 days, compared to those with MI ≤ 4, where the MST was not reached (P < .01). Predicted survival curves differ substantially when controlling for other significant risk factors determined by multivariable analysis (age, presence of multinucleation, and histologic growth pattern). For example, for those cases having multinucleation and an infiltrative growth pattern, the MST for those having MI > 4 is 200 days (Fig. 8, curve F), compared to those having MI = 0 or 0 < MI ≤ 4 (MST not reached; Figs. 6, 7, curve F). Similarly, predicted local reoccurrence curves (Figs. 9–11) demonstrate that regardless of MI, those cases with complete margins and a well-circumscribed appearance (Figs. 9–11, curve A) are unlikely to locally reoccur (less than 10% reoccurring by 1,000 days). This information clearly demonstrates the clinical relevance of these histologic features for prognostication and therapeutic intervention.
The presence of multinucleation in MCTs is a feature described in higher-grade cutaneous MCTs, 37 and when based on univariable analyses, this was a significant risk factor in all models. It also remained significant in survival, DFI, and distant occurrence models with multivariable statistics (Table 4). Having an infiltrative tumor was significantly associated with decreased time to local reoccurrence, which affected overall survival and DFI (Table 4). Intuitively, it suggests that complete surgical margins are harder to obtain in these cases when compared to well-circumscribed tumors; however, there was no statistically significant interaction between margins and histologic pattern. Because this feature is indicative of potential aggressive disease, further staging and monitoring is warranted for these dogs, even if neoplastic cells are not seen at the surgical margins.
There were 56 breeds in this study, and many of these had too few animals to draw statistical associations. Univariable analysis showed that Labrador Retrievers had significantly increased rates of local reoccurrence and that Boxers had an increased rate of distant occurrence; however, these findings were not significant in multivariable analyses. Boxers did tend to have less aggressive tumors: None of the 25 experienced local reoccurrence or metastasis, nor did any have tumors with MI > 1. This is similar to previous studies of cutaneous MCTs reporting that Boxers frequently develop additional distant and multiple tumors 18 but have favorable outcomes. 3,4,16,25 Pugs also have less aggressive tumors, 29 which suggests that there may be a genetic component to predisposition and tumor behavior.
Subsequent distant occurrence rates were significantly increased for cases that had multiple MCTs (P < .01) based on multivariable analysis (Table 4), suggesting a propensity to develop many MCTs. Our follow-up information did not indicate whether these tumors arose sequentially or synchronously. Multiple MCTs are reportedly present in 9% 31 to 15% 25 of cases of cutaneous MCTs, and certain breeds (Boxers, Boston Terriers, and Pugs) seem to be predisposed. These breeds also develop additional distant tumors. 16,18 The number of cases in our study was too low to detect interaction effects between breed and the presence of multiple tumors. Having multiple MCTs did not significantly influence survival or metastasis, suggesting that they represent separate (de novo) events. Only one published study 54 determined a clonal origin of multiple cutaneous MCTs arising in a small number of dogs, suggesting that each tumor may be a form of metastasis.
There are limitations to retrospective analyses and long-term follow-up studies such as this. Because follow-up information was obtained from primary care veterinary clinics instead of a research center, metastasis and additional tumor occurrence may have been underestimated (if there were silent metastases) or overestimated (by the presence of non-MCT-related disease), given that postmortem examination and histologic confirmation were not performed. Rather than exempt cases, which would have biased our results toward more favorable outcomes, we chose to include these as events, despite the lack of histologic confirmation in the majority of cases. Also, risk factors are based on a relatively small number of subjects that had aggressive disease (ie, observed the outcome of interest), and there were wide confidence intervals for some hazard ratios.
The results of the study show that the majority of subcutaneous MCT are not aggressive and that although mitotic activity is a key prognostic factor, other factors should be considered; that is, they should not be automatically categorized as grade II cutaneous MCTs based on their location.
Footnotes
Acknowledgements
This article is dedicated to the memory of Dr Susan Best, our colleague and friend and a superb veterinary pathologist. We thank the veterinary clinics who participated in this study for providing follow-up information for dogs. We also acknowledge both the Ontario Veterinary College Pet Trust Organization and the Rouse Family Foundation for funding this study.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded by the Ontario Veterinary College Pet Trust Organization and the Rouse Family Foundation.
