Abstract
Myeloid neoplasms include cancers associated with both rapid (acute myeloid leukemias) and gradual (myelodysplastic syndromes and myeloproliferative neoplasms) disease progression. Percentage of blast cells in marrow is used to separate acute (rapid) from chronic (gradual) and is the most consistently applied prognostic marker in veterinary medicine. However, since there is marked variation in tumor progression within groups, there is a need for more complex schemes to stratify animals into specific risk groups. In people with acute myeloid leukemia (AML), pretreatment karyotyping and molecular genetic analysis have greater utility as prognostic markers than morphologic and immunologic phenotypes. Karyotyping is not available as a prognostic marker for AML in dogs and cats, but progress in molecular genetics has created optimism about the eventual ability of veterinarians to discern conditions potentially responsive to medical intervention. In people with myelodysplastic syndromes (MDS), detailed prognostic scoring systems have been devised that use various combinations of blast cell percentage, hematocrit, platelet counts, unilineal versus multilineal cytopenias and dysplasia, karyotype, gender, age, immunophenotype, transfusion dependence, and colony-forming assays. Predictors of outcome for animals with MDS have been limited to blast cell percentage, anemia versus multilineal cytopenias, and morphologic phenotype. Prognostic markers for myeloproliferative neoplasms (eg, polycythemia vera, essential thrombocythemia) include clinical and hematological factors and in people also include cytogenetics and molecular genetics. Validation of prognostic markers for myeloid neoplasms in animals has been thwarted by the lack of a large case series that requires cooperation across institutions and veterinary specialties. Future progress requires overcoming these barriers.
Myeloid neoplasms are cancers of hematopoietic cells and are distinct from cancers of lymphoid cells. In most instances, myeloid neoplasms arise from clonally proliferating cells in bone marrow and manifest either with a lack of normal blood cells (anemia, leukopenia, thrombocytopenia) or an accumulation of neoplastic cells in blood. Lymphoid leukemia may also predominantly affect bone marrow, but diagnosis, prognosis, and therapy for lymphoid leukemia differ from those of myeloid leukemia. Myeloid neoplasms in people are broadly divided into acute myeloid leukemia (AML), myeloproliferative neoplasms (MPNs, formerly termed chronic leukemia), and myelodysplastic syndromes (MDSs). Generally, these broad categories correspond to the natural history of the neoplasm: AML is rapidly progressive without aggressive therapy, and MPN and MDS are more gradually progressive cancers, which in some instances do not require specific therapy. In people and animals, assessment based on clinical features of the patient, routine laboratory findings, and morphology of the tumor cells allows classification of most myeloid neoplasms into AML, MPN, or MDS and often allows subclassification of MPN according to accumulation of a particular cell type. In people, progress in identification of molecular lesions associated with certain types of leukemia and their prognosis has resulted in incorporation of such information into classification schemes. The World Health Organization (WHO) developed a classification of tumors, including those involving hematopoietic and lymphoid tissues, which was an international effort to periodically generate an updated review of neoplastic entities defined by morphology, cytochemistry, immunophenotype, genetics, and clinical features in order to arrive at clinically significant disease entities. 109 The 2008 edition categorizes approximately 28 types of myeloid neoplasms into AML, MPN, and MDS by these criteria (more if specific disease types specified by genetic mutations are counted as individual subtypes), and many of these categories have strong statistical support for associated prognoses. 109 Nevertheless, classification of many myeloid neoplasms in people remains a complex, imperfect, and rapidly evolving science. Comparatively, classification of myeloid neoplasms in animals is in early development. Except for chronic myelogenous leukemia (CML), a rare tumor in animals, molecular lesions associated with morphological types of leukemia have not been characterized. Current classification of leukemia in animals largely relies on morphological features of tumor cells, assisted by immunophenotypic, electron microscopic, and/or cytochemical evaluation of undifferentiated blast cells in some cases of AML. 62 Nevertheless, myeloid neoplasms with morphologic similarity, or even identity, to entities in human oncology are diagnosed in animals, particularly in dogs, suggesting that classification schemes used for human myeloid neoplasms may be suitable for animal tumors. However, the molecular lesion, response to therapy, and prognosis for most myeloid neoplasms in animals remain unknown.
The objectives of this review are to summarize current knowledge regarding prognostic markers of myeloid neoplasms across species (people, dogs, and cats) and to define parameters and objectives for future investigation.
Acute Myeloid Leukemia
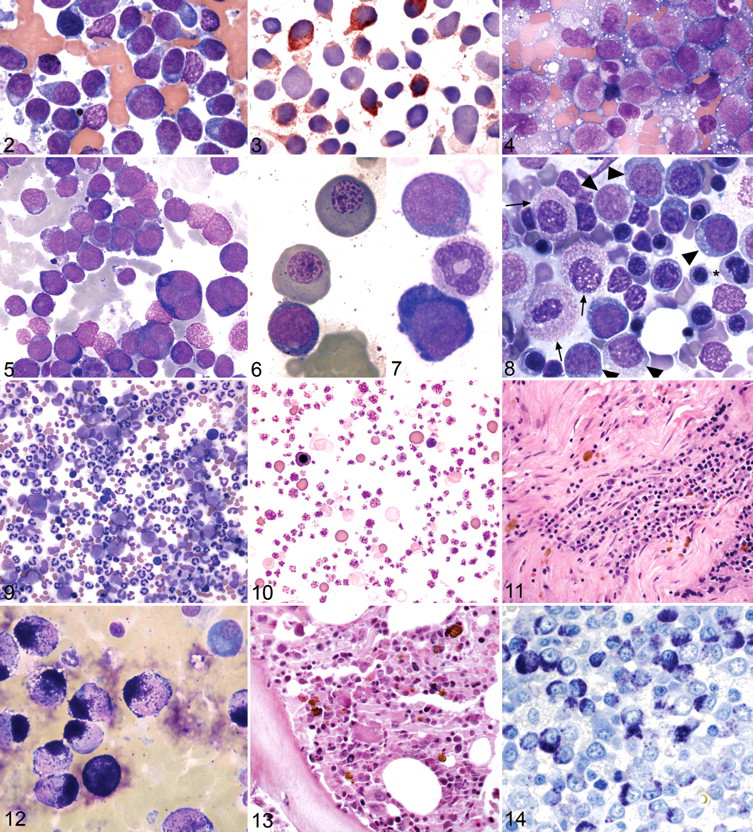
Acute myeloid leukemia (AML) is a neoplasm of myeloid blasts resulting in rapid mortality if untreated (Figs. 1-7).
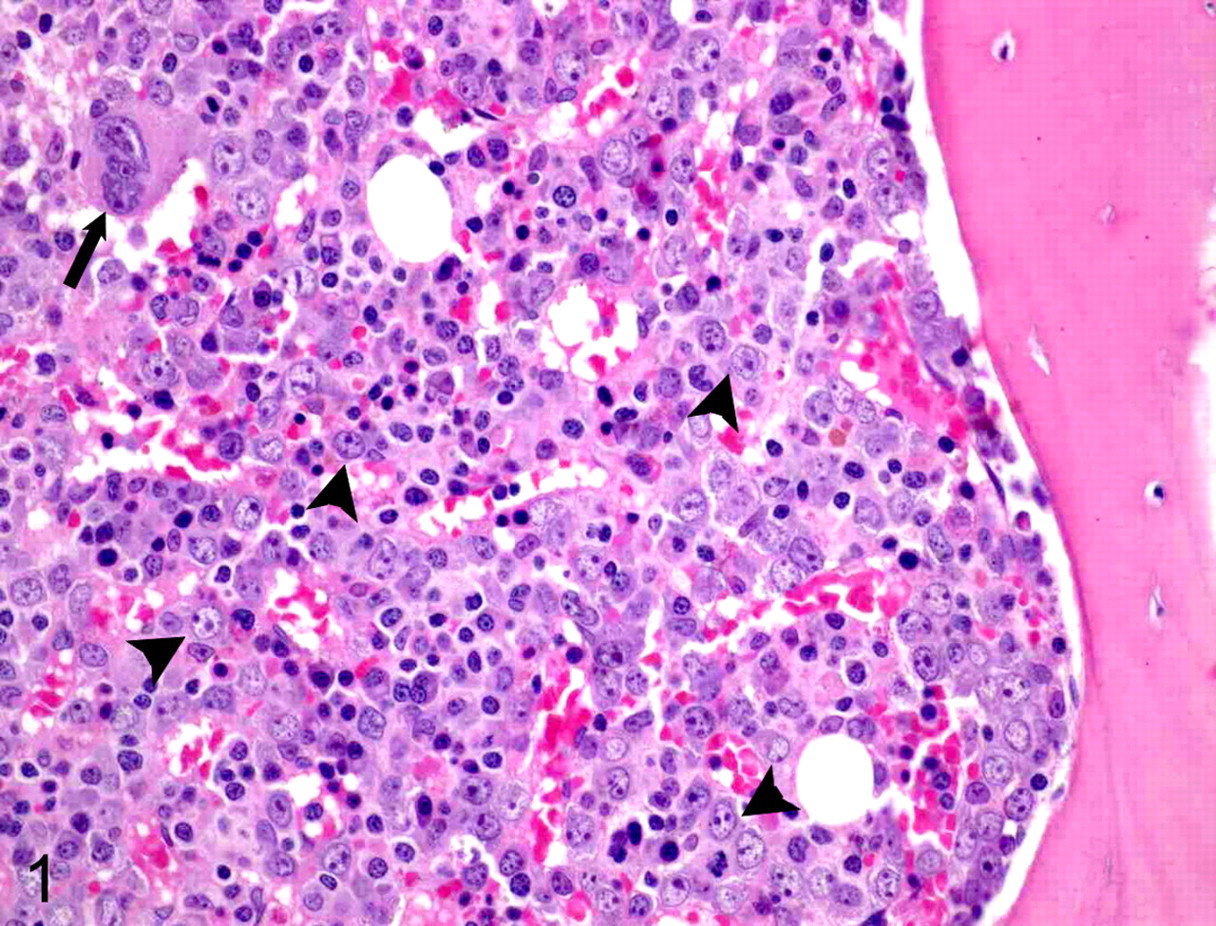
Bone marrow tissue, dog with pancytopenia and acute myeloid leukemia. The bone marrow is highly cellular. Megakaryocytes are rare (arrow), granulocytic reserve is absent, and blast cells (arrowheads) with round nuclei, 1 or 2 prominent nucleoli, and lack of differentiating features are increased to approximately 60% to 70% of all nucleated cells. Remaining cells are primarily identifiable as erythroid precursors. Designation regarding specific type of AML is best done using preparations made from aspirates of marrow and confirmation of lineage of blasts requires immunophenotyping and cytochemical stains. HE stain.
Acute Myeloid Leukemia—People
Decades of research on acute myeloid leukemia have revealed the morphologic and biological heterogeneity of this disorder in people. To assist with patient diagnosis and management, classifications, such as the French–American–British (FAB) and the WHO classification, were developed to define clinically relevant disease subtypes. The FAB system was developed in the 1970s, 7 was revised in the 1980s, 8 and was widely used both in people and animals. 51,108 The first WHO classification and the more recent revision arose from many retrospective studies performed to identify potentially useful prognostic factors for de novo AML. 24,28 Early investigations examined clinical and laboratory parameters and documented the prognostic significance of patient age 1 and magnitude of initial white blood cell concentrations on overall survival. 70,81,123 However, the highly variable clinical outcome of AML provided impetus to seek additional predictive patient and tumor-specific characteristics.
During the approximately 30 years that the FAB system was used for classifying AML in people, it was discovered that many cases of AML are associated with recurrent genetic abnormalities that affect pathways of myeloid cellular proliferation and maturation. 108 Additionally, it was found that correlation between morphology and genetics was not perfect and that genetics may predict biological behavior and prognosis of leukemia more consistently than morphology. 108 Cytogenetic analysis has therefore become an essential and routine component of the diagnostic workup for AML in people. Pretreatment karyotype is one of the strongest prognostic markers for AML and stratifies patient risk as low (25% relapse rate, 70% survival to 4 years), intermediate (50% relapse rate, 40%–50% survival to 4 years), or high (70% relapse rate, 20% survival to 4 years). 21,38,87 These advances were reflected in the 2001 WHO classification of myeloid neoplasms 50 and in the revised version in 2008. 109 In the current scheme, the largest groupings of AML include AML with recurrent genetic abnormalities; AML with myelodysplasia-related changes (formerly AML with multilineage dysplasia); therapy-related myeloid neoplasms (formerly AML, therapy related); and AML, not otherwise categorized. Entities recognized in the FAB system, and with terminology more familiar to veterinary pathologists, are subgrouped in the “AML not otherwise categorized” group. Acute promyelocytic leukemia, AML M3 in the FAB system, is a notable exception, since it is associated with recurrent genetic abnormalities in people. AML that develops in cats with feline leukemia virus is often preceded by myelodysplastic syndrome and would be most appropriately assigned to “AML with myelodysplasia-related changes.” This entity in people, regardless of morphologic or immunologic phenotype and marrow blast percentage, has a poor response to chemotherapy and is associated with multiple high-risk cytogenetic defects. Entities in the group “AML with recurrent genetic abnormalities” usually have balanced translocations, favorable prognoses, and high rates of complete remission when treated with appropriate therapeutic agents. 18 The critical role of cytogenetic analysis as a pretreatment prognostic factor was further emphasized in the 2008 revision of the WHO classification system of AML. 109 A major change was expansion of the “AML with recurrent genetic abnormalities” subgroup to include 3 additional forms of AML with newly recognized cytogenetic abnormalities. Furthermore, for 3 of the original entities, detection of the specific abnormal karyotype is considered sufficient for a diagnosis of AML regardless of blast cell count, emphasizing the crucial role of chromosomal aberrations for these specific disorders. 109
Application of molecular genetics has further advanced characterization of prognostic markers for AML. Detailed molecular analyses of tumors have uncovered several somatic gene mutations of clinical relevance, particularly in patients with karyotypically normal tumors, who represent approximately 40% to 50% of cases. 21,38 Mutations in nucleophosmin (NPM1), CCAAT/enhancer binding protein α (CEBPA), and fms-like tyrosine kinase 3 (FLT3) genes have been shown to have prognostic significance. 25,30,80 The NPM1 mutation was the most frequent molecular lesion observed in adult patients with cytogenetically normal (CN)-AML. 30,80 Studies have demonstrated that CN-AML with NPM1 mutations in the absence of FLT3-internal tandem duplications (ITD) and CN-AML with CEBPA mutations were favorable prognostic markers associated with complete remission. 80 Conversely, detection of FLT3-ITD mutation was consistently associated with poorer survival. 80 Clinical relevance of these findings was evident in the revised WHO classification system by addition of 2 provisional disease entities: “AML with mutated NPM1” and “AML with mutated CEBPA.” 109 Incorporation of these genetic mutations as distinct disease subtypes emphasized the growing importance of molecular abnormalities for AML prognosis.
The expanding arsenal of molecular biology techniques has generated innovative strategies to characterize AML subtypes and provide additional key prognostic indicators. DNA sequencing, 12 gene expression arrays, 36,104 micro-RNA profiling, 34,52 and DNA methylation assays 20,101 have been used to study AML biology. These assays allow comprehensive and systematic examination of the genome of a tumor. Although promising data resulted, such as the finding that specific single nucleotide polymorphisms can have a negative impact on overall survival in CN-AML, 111 further validation is required to determine whether integrating these costly and labor intensive assays can aid in identifying patients who require aggressive treatments (such as allogeneic stem cell transplantation) or have potential for relapse. Regardless, it is now recognized that the prognosis for people with AML is dependent on age, karyotype, and genetics of the neoplastic cells. 66
Immunophenotyping is considered important in the diagnosis of AML, but its use in predicting treatment response and survival has yielded conflicting results with little consensus. 56 CD56 expression may be associated with a worse prognosis in some cases of AML with recurrent genetic abnormalities, which otherwise have a favorable prognosis. 3,23 AML is often diagnosed or defined based on expression of 2 or more of the following myeloid cell markers: myeloperoxidase, CD13, CD15, CD33, CD65, and CD117. 19,56 Concurrent expression of these markers may be associated with a better prognosis (higher complete remission rates, disease-free survival, and overall survival). 56
Acute Myeloid Leukemia—Animals
In animals as in people, AML is a fulminant disease that is often rapidly fatal if untreated. 62,110 However, in veterinary medicine, there is a paucity of studies examining either molecular mechanisms of AML or identifying prognostic factors. Lack of accurate diagnosis and lack of uniform diagnostic criteria and nomenclature have complicated retrospective analysis of epidemiologic information, prognosis, and response to therapy. 122 In 16 dogs with confirmed AML (consistent hematologic and morphologic criteria, positive for myeloperoxidase, consistent pattern of β2 integrin expression, and absence of lymphoid antigens) for which follow-up information was available, median and mean survival after diagnosis was 7 days and 20 days, respectively, with a range of 2 to 138 days (W Vernau, unpublished data 1996–2002). However, these data are skewed because the majority of dogs in this small study were euthanatized given their severe and rapidly progressive clinical signs (anorexia, pyrexia, severe lethargy) and presumed poor prognosis. Most dogs were treated, but not uniformly. Not all AML may be rapidly fatal, as evidenced by a case report of a dog that survived 24 months while being treated with chemotherapy for acute megakaryoblastic leukemia. 121 The dog’s disease was well controlled during that time with periods of apparent remission.
Investigative approaches for determination of prognostic markers for acute myeloid leukemia in animals
Given the importance of karyotyping in human AML, it is reasonable to speculate that prognostically significant chromosomal abnormalities are also present in animals with AML. Detailed cytogenetic studies of AML in dogs and cats are lacking, partly because of the difficulty in karyotyping canine chromosomes. 91 Karyotyping of canine leukemias using conventional cytogenetic techniques revealed chromosomal aneuploidy and double minute chromosomes. 40 A single case report of trisomy 1 with an X;8 translocation in a dog with AML has also been described. 78 These initial studies were limited not only by small sample sizes but also, more importantly, by the technology available to examine and correctly identify canine chromosomes. With the advent of molecular cytogenetics and availability of chromosome painting and comparative genomic hybridization in the dog, it is now possible to evaluate canine AML tumor samples to the base pair level. 14,16
Assessment of FLT3, C-KIT, and RAS sequence in canine AML identified 15 of 21 (71%) dogs with at least 1 missense mutation, 102 indicating similarity in genetic abnormalities with human AML. Although clinical relevance of these mutations for classification or prognosis has yet to be determined, these findings have provided targets for future investigation.
There is an obvious need for basic research on the genetics of AML in dogs and cats along with meaningful clinical research. To overcome the apparent deficiency of adequate prognostic markers, it will be necessary to take cues from our human medicine counterparts and initiate multicenter trials and coordinated sample banking. Studies aimed at identifying genetic lesions in AML have the potential to reveal new disease entities and discover molecular targets that may affect how this disorder is diagnosed and treated, in both animals and people.
Myelodysplastic Syndrome
Myelodysplastic syndromes (MDS) are neoplastic disorders resulting from clonal proliferation of genetically altered hematopoietic stem cells. 98 The cause of MDS is often unknown, although genetic mutations may be caused by viral infection, radiation, or chemical carcinogens. Increased apoptosis of dysplastic cells, induced by an increase in tumor necrosis factor-α secretion by stromal cells, is thought to play a role in the pathogenesis of MDS; this is in contrast to leukemic cells, which exhibit decreased apoptosis. 49 As a result, MDS are characterized by ineffective hematopoiesis with persistent unilineal or multilineal cytopenia despite marrows that are typically hypercellular. Morphologic atypia is evident (Fig. 8) and blast cell percentages in marrow are below limits required for designation as AML (<20%). Classification schemes used for human patients are complex, with 9 subtypes currently recognized, and are ever evolving as new syndromes are identified both morphologically and genetically. 89 MDS subtypes are designed to reflect known prognostic markers: bone marrow blast cell percentage; unilineal or multilineal dysplasia; percentages of cells with dysplastic features; presence or absence of anemia; severity of thrombocytopenia; and extent of cytopenias (eg, bicytopenia vs pancytopenia). Detailed discussion reviewing various classification systems can be found elsewhere. 51,62,77,82,98,103,114
Myelodysplastic Syndromes—People
Prognostic scoring systems
In human medicine, several prognostic scoring systems based on multiple criteria have been devised to classify MDS patients into risk groups that predict overall survival and risk of progression to AML. The first to be widely used was the Düsseldorf score, named for the location of the authors' institution and published in 1992. 2 One point was assigned for each of the following abnormalities: bone marrow blasts >5%, serum lactate dehydrogenase activity >200 IU/liter, anemia, platelet count <100,000/μl. Patients were divided into 3 prognostic groups: a score of 0 is associated with longer survival and low risk for AML. A score of 1 to 2 indicates moderate risk and moderately decreased survival times, and a score of 3 to 4 indicates high risk for AML with 5-year survival times of 0%. This system does not depend on karyotyping, an assay that is not usually available for veterinary patients, and thus has potential for use in veterinary medicine, but in human studies lack of karyotyping is cited as a limitation. 67
The second system is the International Prognostic Scoring System (IPSS), which was published in 1997. 37 It was based on the FAB MDS classification scheme and is the system that is most commonly used. 89,98 Stratification into risk groups is based on percentage of blasts in bone marrow (eg, score = 0 if blasts <5%, score = 0.5 if blasts are 5%–10%), cytogenetic findings (eg, score = 0 if normal karyotype, score = 1.0 if “poor” karyotype), and number of hematopoietic lines affected by cytopenia (eg, score = 0.5 if 2 or 3). Patients are classified into 4 categories based on total score: 0 = low risk; 0.5 to 1.0 = intermediate-1 risk; 1.5 to 2.0 = intermediate-2 risk; and ≥2.5 = high risk.
A new scoring system based on revised WHO MDS classification criteria is in development. 98 It is a modification of IPSS but introduces multilineal dysplasia and transfusion dependence as additional predictors of adverse outcome. Patients who require 1 or more transfusions every 8 weeks for 4 months do poorly compared with patients who are less dependent on transfusions, partly because frequent transfusions eventually lead to iron overload. Additional proposed variables draw from the Düsseldorf system to include severity of thrombocytopenia and assessment of serum lactate dehydrogenase activity but also include β2-microglobuin concentration, gender, age, and immunophenotyping of progenitor cells. 53,98
Genetics
Genetic abnormalities play an important role in both the WHO-based scoring system and IPSS. Cytogenetic abnormalities are detected in 20% to 70% of human patients with MDS, 98 with deletions in chromosomes 5, 7, and/or 20 most common. In a population of 2,072 human patients with MDS, 684 different cytogenetic categories were identified. 42 Median survival was 53.4 months for those with normal karyotypes and 8.7 months for those with complex anomalies. Patients with abnormal karyotypes that persist after standard treatment are regarded as high-risk patients and receive intensified treatment. 5 The isolated deletion of chromosome 5q is considered a favorable prognostic marker because it is associated with longer survival. Just as molecular genetics has elucidated abnormalities that can be used as prognostic markers for AML, it seems likely that marrow cells from MDS patients will soon be subject to high-throughput “next generation” DNA sequencing for detection and identification of single nucleotide polymorphisms. 89
Flow cytometry
Immunophenotyping by flow cytometry is not always used in human MDS classification schemes given a lack of consensus on protocols. 90 However, when used, multiparameter flow cytometry (at least 3 colors) is recommended, 105 and the panel should be sufficient to determine lineage as well as an aberrant antigen profile of the neoplastic population. 90,106 A high frequency of phenotypic abnormalities has been correlated with a poor IPSS score, increased risk of progressive disease, increased risk of transfusion dependence, and reduced survival time. 53,71,106 The types of immunophenotypic abnormalities include aberrant expression, abnormal intensity of expression, and abnormal coexpression of antigens. In addition, decreased side scatter correlates with hypogranularity, which is a potential indicator of dysplasia. Recommended analysis consists of a panel for blast cells, myeloid progenitors, endothelial cells, T cells, B cells, plasma cells, monocytes, macrophages, and megakaryocytes. Immunophenotypic testing is also performed for normal and atypical mast cells and for basophils.
A recent study of 1,013 people suspected to have MDS found excellent correlation between blast counts based on cytomorphology, and blast counts determined flow cytometrically. 53 That same study reported a greater diagnostic yield when multiparameter flow cytometry was combined with cytomorphology and cytogenetics, primarily due to ability to detect aberrantly expressed antigens. Flow-cytometric scoring systems have been designed to be used in conjunction with prognostic scoring systems (FCSS) and have been validated for MDS patients as predictors of transfusion dependence 106 and as predictors of relapse following bone marrow transplants. 83 There is a high likelihood that use of flow cytometry will become a standard part of diagnostic evaluation of patients suspected to have MDS, particularly when dysplastic morphologies are subtle. This may improve the sensitivity of diagnosing MDS and enhance prognostic scoring systems but may also further complicate classification systems.
Other prognostic parameters
In people, it has been found that males classified as low risk according to the IPSS had less favorable outcomes than low-risk females. Also, among high-risk patients, women younger than 66 years were at particularly high risk. 69 In suspected MDS human cases, where only anemia was present and the diagnosis was uncertain, numbers of circulating BFU-E (burst-forming unit–erythroid) and CFU-GEMM (colony-forming unit–granulocytic, erythroid, monocytic, megakaryocytic) were determined to be below normal and lower than those in patients with other causes of anemia, such as anemia secondary to chronic disease and endocrine disorders. Patients with low numbers of circulating CFU-GM (colony-forming unit granulocytic-monocytic) have been determined to have an intermediate prognosis. 72
Myelodysplastic Syndromes—Animals
Myelodysplastic syndromes have been described in cats, 4,10,11,17,41,46,84,86,99,112 dogs, 33,45,63,65,115 –119 and 1 horse. 27 Beyond classification of MDS subtypes based on blast cell percentage and myeloid to erythroid ratio (M/E), there is only a single report on the application of the Düsseldorf system to feline MDS patients. 46 Therefore, although survival times for MDS can range from days to many months, veterinarians in clinical practice have few tools for counseling clients about likely survival times of individual animals with MDS. As with human patients, high blast percentage, multiple cytopenias, and marked morphologic atypia are generally accepted as negative prognostic markers, and reports support this assumption. 46,114
Classification schemes used for animals are far simpler than those applied to people. The Animal Leukemia Study Group in 1991 recommended 2 MDS categories: MDS (M/E >1.0) and MDS–erythroid (M/E <1.0). These were based on consensus diagnoses of 10 MDS cases out of 49 animals with potential myeloid neoplasms. 51 These categories allowed generous room for diagnosing MDS, by essentially emphasizing detection of dysplastic features, blast cell percentage, and M/E as key features. Since then there have been recommendations for 3 or more categories, 9,77,112,114 but there is inconsistent application in the literature and predictive values for each group are uncertain because survival times within each category are highly variable. Three subtypes have emerged as perhaps the easiest to identify and diagnosis requires only a history of persistent unexplained cytopenias, bone marrow aspirate, blood film, and complete blood cell count.
The first subtype, MDS with excess blasts (MDS-EB), includes blast cell percentages in marrow that are >5% but below 20% (≥20% indicates progression to AML with myelodysplasia-related changes). Affected animals in this group are expected to have the highest risk for progression to leukemia and, therefore, have the worst prognosis. Age ranges in cats have been reported to be very young to old, 0.6 to 17 years, 10,11,17,46,84 –86,112 and middle-aged to old, 4 to 13 years, in dogs. 114,119 Survival time for cats and dogs is reported as days to weeks. 112,119 Human recombinant erythropoietin and granulocyte-macrophage and granulocyte colony stimulating factors (GM-CSF and G-CSF) have been given to some patients; cytopenias improved, 13,113 but survival times appeared not to be increased. Chemotherapeutic agents have also been tried, but controlled studies have not been reported. 65
The second subtype, MDS, with refractory cytopenia (MDS-RC), includes MDS with refractory anemia. These cases have blast cell counts <5% and an indolent course, with less likelihood of progression to AML, but patients can be plagued by repeated crises secondary to their cytopenias if severe. They exhibit unilineal, bilineal, or trilineal cytopenias. Some patients with MDS-RC have very mild signs, and MDS is sometimes detected incidentally with geriatric or preanesthetic blood work. Age range in cats is reported to be 5 to 18 years. 86,112 The age in dogs is reported to be >7 years. 114,119 Animals have been reported to survive 1 to 74 months. 46,112 –114 Early death is most often associated with pancytopenia; the best prognosis is when cytopenia is confined to anemia. 112,119 One dog was reported to have been “cured” within 8 weeks after treatment with prednisone and human recombinant erythropoietin, 13 and human recombinant colony stimulating factor and erythropoietin have been reported to be occasionally used by others. 113 MDS is not considered a curable disorder; therefore, an animal that is considered to be cured by these drugs might have had secondary dysplasia.
The third subtype, MDS-ER, is identified when the M/E ratio is <1.0. This subtype carries a poor prognosis: survival times have been reported as days to weeks. 11,51,60
Düsseldorf scoring system applied to cats with MDS
A single case series applied the Düsseldorf scoring system to 16 cats with MDS and compared the score with blast cell percentage as a predictive marker. 46 Survival curves were compared using the Kaplan-Meier method with appropriate censoring of data. Although a trend was evident when survival of cats with low blast counts was compared with that of cats with high blast counts, the P value (.06) was not statistically significant. When Düsseldorf scoring was applied, there was statistical significance, at P = .01. There were too few animals that progressed to AML to determine the leukemia-free interval and too few cats to compare other variables. The study was presented as preliminary, and a need for greater numbers of animals in future studies was emphasized.
Other prognostic markers
Gender, age, and retrovirus infection status are possible prognostic markers for MDS in animals but are unsubstantiated by large studies. Gender, although recommended in the revised WHO prognostic scoring system, has not received attention in veterinary literature as either a risk factor for developing disease or a marker for outcome after diagnosis. In 2 reports from Japan, one a study of 13 cats and another of 16 cats, male cats, at 69% and 62.5%, respectively, outnumbered females, but information regarding shortened survival times specific for males, as suggested by human studies, was not included in these studies. 46,86
Although feline leukemia virus is an important risk factor for MDS in cats, with reports of infection that range from 36% to 100%, 46,86,112 its usefulness as a predictor of negative outcome is not clear. In a study that included 22 cats diagnosed with primary MDS, 13 were classified as MDS-EB and 8 as MDS-RC. 112 There were more FeLV+ cats in the MDS-EB group (6/13, 46%) than in the MDS-RC group (2/8, 25%), but the 2 FeLV+ cats in the MDS-RC group had 2 of the longest survival times—1.0 and 1.4 years. In another study of 16 cats with MDS, 15 were FeLV+, but survival times were widely distributed: from 10 days to 74 months. 46 The authors did not specify FeLV status of the cat that survived to 74 months, but 3 cats survived more than 1 year so at least 2 of these were FeLV+.
Colony-forming unit assays have been examined as potential prognostic tools for human MDS (see above). Comparable information regarding in vitro growth of hematopoietic cells as a prognostic marker is lacking in veterinary medicine, although this tool has been used to examine pathogenesis. 68 Numbers of CFU-E colonies were decreased in 4 of 7 cats with MDS; numbers of CFU-GM colonies were decreased in 4 cats; and CFU-GM colonies were small in all 7 cats.
Investigative approaches to determine prognostic markers for myelodysplastic syndromes in animals
A diagnosis of MDS carries the expectation that supportive care could be long term, costly, and ultimately not successful; therefore, guidelines are needed for making the correct diagnosis and predicting outcome. One obstacle is the similarity between primary neoplastic MDS and nonneoplastic secondary myelodysplastic disorders, which are potentially reversible, short term, and presumed to be due to immune-mediated and/or myelotoxic conditions rather than intrinsic genetic mutations. 9 To make progress in diagnosing and prognosticating MDS in animals, investigators must develop uniformly applied diagnostic criteria that incorporate cytomorphologic, hematologic, and possibly histologic determinants. Statistical analyses of data sets to identify valid prognostic markers are necessary, as are immunophenotypic and genetic analyses to confirm clonality. Survival curves should provide information on leukemia-free interval and overall survival. Veterinary publications have largely lacked sufficient numbers, consistent application of diagnostic criteria, inclusion of details to facilitate consolidation of data in a statistically meaningful manner, and correlation of outcome to clinical findings. Adequate sample size will only be achieved via large-scale, collaborative, multi-institutional investigation with contributions from both pathologists and clinicians; this will require creation of a common registry for veterinary MDS cases. The ultimate goal is construction of a classification system based on prognosis that uses diagnostic tools readily available in academic and reference laboratories.
Myeloproliferative Neoplasms
MPNs are a phenotypically and genetically heterogeneous group of clonal, neoplastic conditions of hematopoietic tissue characterized by gradual disease progression (Figs. 9-14).
Myeloproliferative Neoplasms—People
Classification schemes for human MPNs are part of the WHO classification of tumors of the hematopoietic and lymphoid tissues, developed by an international group of hematopathologists in consultation with clinicians. The most recently revised edition, published in 2008, 109 incorporates morphology, cytochemistry, immunophenotype, genetics, and clinical features of hematologic conditions to arrive at clinically significant disease entities. These include chronic myelogenous leukemia (CML), chronic neutrophilic leukemia (CNL), polycythemia vera (PV), essential thrombocythemia (ET), primary myelofibrosis (PMF), chronic eosinophilic leukemia, not otherwise specified (CEL, NOS), mastocytosis, and MPN, unclassifiable (MPN, U). The phenotypic diversity among these neoplasms is attributable to an array of genetic mutations resulting in abnormal signal transduction via tyrosine kinases or related molecules. 96 Best characterized among these mutations is consistent association of the BCR-ABL fusion gene, located in the Philadelphia chromosome, with CML. 97 Enhanced tyrosine kinase activity of the BCR-ABL protein results in constitutive activation of several signal transduction pathways. 97 Similarly, mutations in the Janus kinase 2 (JAK2) gene characterize approximately 90%, 60%, and 50% of human patients with PV, ET, and PMF, respectively, and result in constitutive phosphorylation and aberrant signal transduction via erythropoietin, thrombopoietin, and/or other cytokine receptors. 55 Differences in JAK2 gene dosage or presence of additional genetic modifications are thought to influence the phenotype of the MPN. 55
CML in people is a clonal stem cell disorder with proliferations involving several or all hematopoietic cell lineages. 96 It is characterized by a neutrophil-predominant leukocytosis accompanied by significant eosinophilia and basophilia. 44 An initial indolent chronic phase lasting several years is followed by an accelerated phase and/or a blast phase. Improved survival in patients diagnosed during the chronic phase correlates with younger age, moderate splenic enlargement, relatively normal platelet concentration, and a low proportion of blood blast cells, eosinophils, and basophils. 44 Median survival has progressively increased from 3 years without chemotherapy to 6 years with chemotherapy and/or interferon-α and more than 6 years after treatment with imatinib, a specific inhibitor of BCR-ABL tyrosine kinase overexpression in CML. 44 The prognosis in people can be stratified based on the above clinical and hematological factors, but completeness of response to tyrosine kinase inhibitors at hematologic, cytogenetic, and molecular levels is the strongest predictor of progression-free survival. 44
Chronic neutrophilic leukemia is a very rare MPN of people characterized by marked leukocytosis composed of segmented neutrophils and band forms (Fig. 9). Prognostic factors, other than the presence of myelodysplastic features that herald transformation to AML, are not established. 109
PV and ET have a relatively indolent natural course resulting in a modest reduction in life span. PV is characterized by a prodromal period, when erythrocytosis is mild or absent, followed by a long period of progressive polycythemia with gradual development of myelofibrosis and splenomegaly due to extramedullary hematopoiesis and a postpolycythemic phase with bone marrow failure from fibrosis. Large vein thromboses are characteristic for PV and may occur prior to overt polycythemia. 107 Patients succumb to eventual complications from thrombosis, hemorrhage, hematopoietic failure, and/or transformation to AML. 55,107 JAK2 mutations are present in most PV patients and impart cytokine-independent receptor signaling and cell proliferation, which translates to autonomous in vitro erythroid colony formation. 107 Risk factors for increased mortality are age greater than 50 years, history of thrombosis, presence and allelic burden of JAK2 mutations, and increased leukocyte count. 55,75
Essential thrombocythemia is clinically similar to PV, but the predominant proliferating cells are megakaryocytes resulting in sustained blood thrombocytosis of >450 × 109/liter (Fig. 10). 107 ET is typically diagnosed in older individuals, often as an incidental finding on screening tests. The clinical course of ET is gradual and characterized by microvascular thrombotic events, bleeding from mucosal surfaces, transient ischemic attacks, and peripheral neurologic deficits. As in PV, patient stratification according to age greater than 60 years, prevalence of thrombosis, JAK2 mutation status, and leukocyte count is correlated with survival. 107 Therapy is directed largely at reducing thrombotic events, although specific JAK2-targeted inhibitors are under development. 55
Primary myelofibrosis is an MPN with a worse prognosis than ET or PV (Fig. 11). 64 The proliferating cells are megakaryocytes and bone marrow granulocytic precursors, and affected individuals typically present with anemia and splenomegaly. 64 Progressive myelofibrosis, dacryocytosis, thrombocytosis, increased CD34+ cells in circulation, and eventual blast crisis characterize the disease course. An accumulation of morphologically abnormal cells in marrow distinguishes PMF from PV and ET. Median survival is 5 years but is highly dependent on stage of PMF at the time of diagnosis. 55 Risk of death may be stratified similarly as in PV and ET according to age at onset, degree of anemia, abnormal leukocyte and platelet morphology and concentration, and presence of JAK2 mutations. Therapy for advanced disease is currently limited to hematopoietic stem cell transplantation. 55
CEL, NOS is a clonal proliferation of eosinophil precursors resulting in persistent blood, bone marrow, and tissue eosinophilia. 95 It is distinguished from hypereosinophilia (HES), which lacks evidence of clonality, and from CEL, which has either BCR-ABL fusion genes or rearrangement of platelet-derived growth factor (PDGF) or fibroblast growth factor (FGF) genes. 96 Although CEL, NOS by definition has a clonal genetic abnormality, rearrangement is likely subtle or novel, which may account for overdiagnosis of HES and underrecognition of CEL, NOS. 95 Given the heterogeneous definition of CEL, NOS, prognosis is also highly variable. Unfavorable prognostic factors were degree of splenomegaly, presence of blasts in blood, dysplastic features of other myeloid cell lineages, and karyotypic abnormalities. 95
Mastocytosis in people consists of a group of heterogeneous neoplasms of clonal mast cells. 74 Tumors are broadly divided into localized or diffuse cutaneous mastocytosis and systemic mastocytosis with variable involvement of bone marrow and other tissues. Cutaneous mastocytosis is most commonly diagnosed in children, has an indolent disease course, and may spontaneously regress. 74 Systemic mastocytosis (SM) has 3 manifestations: accumulations of morphologically unremarkable mast cells in lymph nodes, spleen, liver, and/or gastrointestinal tract with an indolent disease course (Figs. 12-14); mast cell leukemia with a rapid clinical course; and mast cell sarcoma with rapid distant spread and development of mast cell leukemia. The latter 2 neoplasms are rare. Within the SM group, anemia, extensive bone marrow mastocytosis, abnormal mast cell cytomorphology, and hepatosplenomegaly are associated with a worse prognosis. 74 Activating mutations in KIT tyrosine kinase is a feature of most cases of mastocytosis but does not distinguish between indolent and aggressive SM. 74,107
The label MPN, U is reserved for neoplasms that do not meet the criteria for specific MPN. Most of these patients are in either early PV, ET, or PMF stages without full development of the diagnostic features; have advanced-stage MPN where extensive myelofibrosis and/or blast accumulation obscures characteristic features of other MPN; or have MPN with concurrent inflammation or another neoplasm masking diagnostic features. 95,96 Presence of the BCR-ABL fusion gene, or PDGF or FGF gene rearrangement, excludes the diagnosis of MPN, U. The prognosis for this group of MPN may be similar to the disease that MPN, U evolves into or may be very poor if diagnosed at an advanced stage of myelofibrosis and blast presence. 107
Myeloproliferative Neoplasms—Animals
Many MPNs identified in people have also been reported in animals. In most cases, the diagnosis was based on clinical and morphological criteria. MPNs usually consist of relatively normal or mild-to-moderately dysplastic appearing cells; therefore, morphologic categorization of such tumors is rarely challenging. However, since they mimic hyperplasia, diagnoses of MPN are often made after extensive efforts to rule out reactive causes for increases in white blood cells, red blood cells, and/or platelets.
Chronic leukemia with neutrophil predominance has been reported in 6 dogs and 5 cats. In each case vague illness was present for a prolonged period of time. 32,57,94 Neutrophil concentrations in blood were typically 5 to 10 times increased and consisted predominantly of differentiated neutrophils with modest increases in band neutrophils, metamyelocytes, and myelocytes. 32 Dysgranulopoiesis may be present and may include ring-shaped or hypersegmented nuclei, giant band neutrophils, giant metamyelocytes and myelocytes, prominent azurophilic granulation in metamyelocytes and myelocytes, and increased cytoplasmic basophilia. 32 Cytoplasmic basophilia of immature neutrophils may be profound, making them difficult to differentiate from monocytes. Bone marrow of affected animals was hypercellular, M/E was increased, and hepatosplenomegaly was common. 32,57,94 However, these cases of chronic leukemia with neutrophil predominance morphologically more closely resemble CNL than CML of people given the absence of eosinophilia and basophilia. The human morphologic equivalent of CML has not been described in detail in animals. However, molecular genetic investigation identified that regions on human chromosomes 9 and 22 are evolutionarily related to regions on canine chromosomes 9 and 26, and a canine equivalent to the BCR-ABL mutation was identified in 3 spontaneous cases of canine CML. 16 In 1 dog described as having CML in blast crisis, a fusion protein was produced by tumor cells. 16
Chronic eosinophilic leukemia, which is by definition clonal, can be difficult if not impossible to distinguish from HES. 95 Chronic eosinophilic leukemia has been reported in 12 cats and was often associated with FeLV infection. 35,48,58 Hypereosinophilia, attributed to either inflammation or paraneoplastic interleukin-5 production by lymphoma, was more common than CEL. 48 CEL, but not hypereosinophilia, was associated with immature or abnormal eosinophils in circulation, M/E ratios exceeding 10:1, and anemia. 48 Hypereosinophilia attributed to conditions other than leukemia was reported in 13 dogs, most commonly in Rottweilers. 92 CEL has not been reported in dogs. It is conceivable that some feline or canine cases of HES may represent indolent eosinophilic leukemia; however, without detecting clonality it is difficult to confirm them as neoplastic.
Chronic basophilic leukemia is a neoplasm rarely reported in dogs and cats. 61 A similar entity is not described in the WHO classification, and the veterinary entity therefore should be considered as MPN, U. Similar to CNL and CEL, there was pronounced blood basophilia including immature forms, and there may be neutrophilia, eosinophilia, and thrombocytosis. 61
Polycythemia vera has been described in dogs, cats, and cattle. 54,76 Presenting manifestations usually relate to increased red blood cell mass resulting in poor tissue perfusion, microvascular hemorrhage, or thrombosis. Hematocrit and hemoglobin concentration often exceed 65% and 200 g/liter, respectively. Diagnosis of PV was based on assessment of erythrocyte mass relative to plasma volume and erythropoietin (EPO) concentration. PV is due to EPO-independent cell proliferation; therefore, serum EPO is normal or decreased, whereas secondary polycythemia results from increased EPO production, (eg, with systemic chronic hypoxemia or localized hypoxemia due to a primary renal lesion) or elaboration of an aberrant EPO-like product by a nonmyeloid neoplasm. 54,76
Essential thrombocythemia has been reported in several dogs 6,22,26,29,31,47,88,93 and cats. 29,43 In most cases ET was characterized by marked and persistent peripheral thrombocytosis and megakaryocytic hyperplasia in marrow. Thrombocythemia typically exceeded 1 million/µl and consisted of platelets with marked variation in size. Atypical megakaryocytes with abnormal lobation were noted on bone marrow examination. Causes of reactive thrombocytosis such infection, iron deficiency, inflammation, or other malignancies must be excluded. Increased circulating platelet volume (plateletcrit) may result in microvascular thrombosis or hemorrhage in animals as in people. 6,93
Myelofibrosis in dogs is most often secondary to chronic systemic inflammation, drug therapy, or nonhematopoietic neoplasia. 115 Primary myelofibrosis has only been reported in 1 dog and may therefore be a rare condition. 115 In cats, PMF has not been reported, but myelofibrosis is a well-recognized sequel of FeLV infection. 10
Mast cell neoplasms are more common in dogs than in people and can be classified within the WHO mastocytosis group. Benign (grade 1) canine cutaneous mast cell tumors are most similar to human skin mastocytoma, with both having a good prognosis. 120 Malignant (grade 3) mast cell tumors in dogs are most similar to human mast cell sarcoma, and both have a rapidly progressive clinical course. However, the majority of canine cutaneous mast cell tumors are grade 2, and prognosis within this group has been described to vary with cytomorphology, mitotic rate, and KIT mutations. 120 Hence, significant prognostic indicators of survival for dogs with grade 2 mast cell tumors remain to be determined. In cats, mast cell proliferative diseases are fairly common and include a condition similar to SM in people as well as solitary mast cell tumors. 79 Clonality has not been determined for either condition, but in most cases of SM in cats the disease course is similar to the indolent type of SM in people. 79
Investigative approaches to determine prognostic markers for MPNs in animals
Identifying signature genetic changes in chronic leukemia of animals would likely be valuable to better understand pathogenesis and for refined diagnosis and prognosis, as demonstrated by studies in people.
Assessment of metaphase spreads identified numerical chromosomal abnormalities in leukemias of dogs and cats more than 2 decades ago. 39,40 Subsequent development of molecular cytogenetics and fluorescence in situ hybridization (FISH) technology allowed greater refinement in detection of chromosomal aberrations. 100 Availability of an accessible and annotated genome assembly for the domestic dog in 2005, followed rapidly by sequencing of the genome of the cat and other animals, has moved veterinary oncology research into the era of species-specific genomics. 15,59 Technology incorporating high throughput chip- or bead-based approaches is now applied to canine cancer tissue to assess tumor features such as DNA copy number variation, presence of single nucleotide polymorphisms, and other mutations. 15 High-resolution gene profiling has identified the BCR-ABL rearrangement and other areas of genetic variation associated with cancers. 15,16 Greater genetic homogeneity in dogs relative to people has guided identification of analogous genetic regions and lesions of importance in human disease; hence, future molecular genetic investigation of cancer in dogs also promises to yield instructive information for leukemia and other cancers in noncanine species. 15
Since MPNs are uncommonly diagnosed neoplasms in animals, precise descriptions of historical, clinical, and laboratory findings, treatment, quality of life, and survival for cases from multiple geographical locations will be necessary to obtain a sufficiently large sample and to identify meaningful prognostic factors.
Summary
Chronic MPNs are typically diagnosed at a relatively advanced stage in animals with a history of long-standing vague illness. As such, it is difficult to assess the natural history of these diseases, which may be substantially longer than the period between diagnosis and death. Epidemiological data on animal populations most affected by MPN, risk factors, and response to therapy are largely lacking. However, considerable similarity in clinical, laboratory, and morphological features of MPN in animals and people suggests that common signal transduction pathways may be involved. Hence, future effort should be focused on precise classification of MPN with available diagnostic modalities (as outlined in Table 1 ), large-scale studies with clear hypotheses, and generation of a tissue repository to facilitate correlation of genetic abnormalities with phenotype.
Approach to Diagnosis of Myeloid Neoplasms
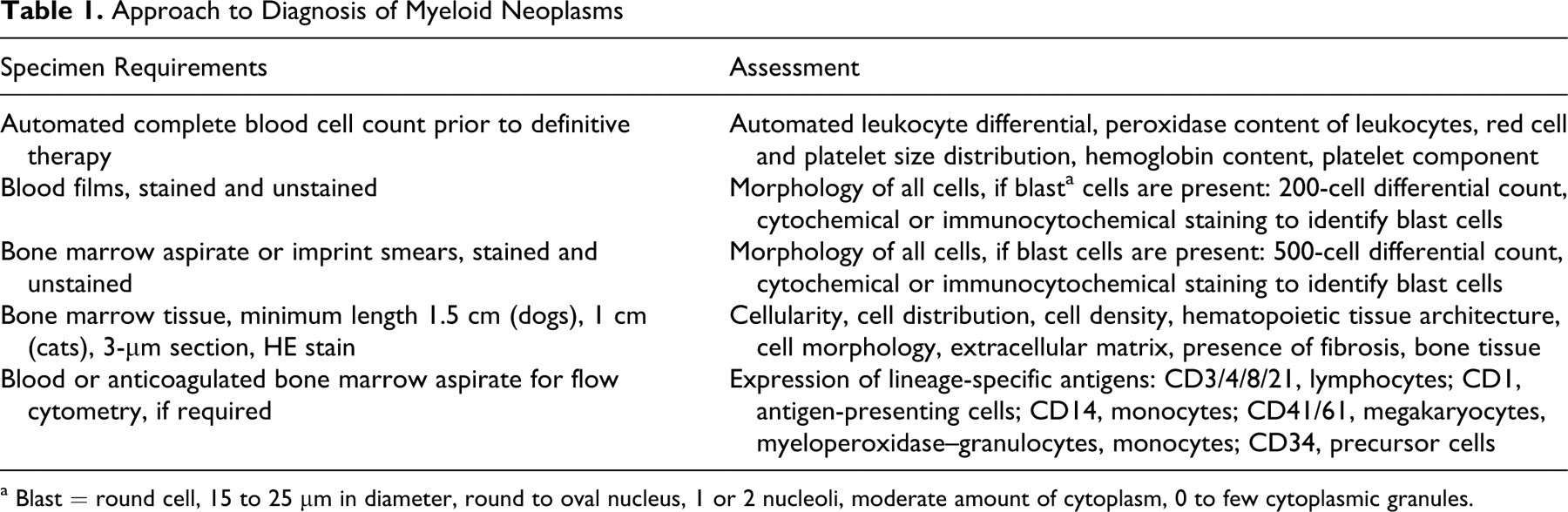
a Blast = round cell, 15 to 25 μm in diameter, round to oval nucleus, 1 or 2 nucleoli, moderate amount of cytoplasm, 0 to few cytoplasmic granules.
Conclusions
Tables 2 and 3 summarize prognostic markers for the major categories of myeloid neoplasms. Notice that the heading in Table 3 for prognostic markers for animals includes the word possible. To most veterinary pathologists concerned with interpretation of hemato-oncology specimens, it is apparent that there are many different types of myeloid neoplasms and that classification and prognostication are difficult or impossible based on morphology alone. Incorporation of immunophenotypic or cytochemical characteristics of blast cells aids with classification of some types of AML, but statistically meaningful data on response to therapy and survival are lacking for all types of myeloid neoplasms in veterinary medicine. Hence, as a first step to improved understanding of these neoplasms, it is necessary to link patient data, laboratory features, and tumor morphology with response to therapy and survival. Since myeloid neoplasms may be rapidly progressive resulting in euthanasia without attempting therapy (AML) or may be diagnosed late during the disease course after elimination of nonneoplastic causes (MDS and MPN), establishing a myeloid neoplasia registry for compilation of data on a large number of individuals would be a useful starting point. It is readily conceivable to have a Web-based repository for myeloid neoplasms in animals, which would necessarily include patient, clinical, laboratory, morphological, and ancillary diagnostic data as well as outcomes. Table 1 summarizes the diagnostic approach to assessment of animals suspected to have a myeloid neoplasm; results of these assays should be included in the database. Such a database would not only provide epidemiological information but also facilitate data mining and potentially allow recruitment of more than 1,000 patients. This would identify statistically meaningful variables at least concerning the main categories of myeloid neoplasms. Furthermore, such a data set would identify frequencies of different categories of myeloid neoplasia in animals and their similarity to molecularly characterized human counterparts, which in turn would be instructive for future molecular genetic investigation. Anticipation of these future studies suggests a need for tissue banking of marrow and blood; therefore, contact information for protocols to be followed and institutions willing to handle these tissues could be included in the registry. Veterinary pathologists are in a unique position to contribute to this field by recommending and encouraging routine acquisition of peripheral blood and bone marrow specimens for further diagnostic and molecular testing.
Prognostic Markers of Myeloid Neoplasms in People

BFU, burst-forming unit–erythroid; CFU-GEMM, colony-forming unit–granulocytic, erythroid, monocytic, megakaryocytic; CFU-GM, colony-forming unit granulocytic-monocytic
Prognostic Markers of Myeloid Neoplasms in Dogs and Cats

The greater genetic homogeneity of dogs relative to people has proven useful for identifying genetic changes associated with morphologically similar cancers, such as osteosarcoma, which are common to both species. 73 Might genetic assessment of myeloid neoplasms in dogs and cats also provide insight into these diseases in people and other species?
Footnotes
Acknowledgements
This study represents an initiative of the American College of Veterinary Pathologists' Oncology Committee. The study has also been reviewed and endorsed by the World Small Animal Veterinary Association. We thank both organizations for their support and guidance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
