Abstract
Tissues from sequential-kill time course studies of bovine spongiform encephalopathy (BSE) were examined to define PrP immunohistochemical labeling forms and map disease-specific labeling over the disease course after oral exposure to the BSE agent at two dose levels. Study was confined to brainstem, spinal cord, and certain peripheral nervous system ganglia—tissues implicated in pathogenesis and diagnosis or disease control strategies. Disease-specific labeling in the brainstem in 39 of 220 test animals showed the forms and patterns observed in natural disease and invariably preceded spongiform changes. A precise temporal pattern of increase in labeling was not apparent, but labeling was generally most widespread in clinical cases, and it always involved neuroanatomic locations in the medulla oblongata. In two cases, sparse labeling was confined to one or more neuroanatomic nuclei of the medulla oblongata. When involved, the spinal cord was affected at all levels, providing no indication of temporal spread within the cord axis or relative to the brainstem. Where minimal PrP labeling occurred in the thoracic spinal cord, it was consistent with initial involvement of general visceral efferent neurons. Labeling of ganglia involved only sensory ganglia and only when PrP was present in the brainstem and spinal cord. These experimental transmissions mimicked the neuropathologic findings in BSE-C field cases, independent of dose of agent or stage of disease. The model supports current diagnostic sampling approaches and control measures for the removal and destruction of nervous system tissues in slaughtered cattle.
Keywords
Prion diseases are fatal neurodegenerative disorders characterized by tissue accumulations of an abnormal isoform (PrPSc) of a host protein, PrPC. 22 The aberrant protein, generally assumed to be the pathogen, is transmissible, infectious, and, in some forms of prion diseases, contagious, but it does not evoke measurable immune responses like those that form the basis of diagnosis of diseases caused by conventional pathogens. Consequently, critical to the characterization and diagnosis of prion diseases is the definition of the pathologic phenotype. Initial characterization of the pathology of each disease necessarily comes from the end stage or clinical phase of the disease obtained through passive surveillance. In naturally occurring bovine spongiform encephalopathy (BSE), a zoonotic prion disease of domestic cattle,21,67,73 the predominant disease phenotype, designated classical BSE (BSE-C), has been defined in terms of morphologic changes and demonstration of tissue accumulations of PrPSc in the central nervous system (CNS) by immunohistochemical (IHC) and immunochemical methods.3,6,7,15,30,33,36,42,50,55,58,60,63,68,70 Based initially on spongiform changes and examination of large case numbers, the invariable predominance of lesions in the brainstem established this and, specifically, the medulla oblongata as the predilection brain region for diagnostic sampling.18,30,53,63,66,68,70 Correlation of the highest concentrations of PrPSc in the brainstem with the greatest degree of spongiform change30,43,51,68 is considered suggestive of the initial brain pathologic alternations in BSE occurring in this region, consistent with the proposed pathogenesis of transmissible spongiform encephalopathy (TSE) in ruminants and laboratory rodent models after oral exposure.4,5,13,20,26,29,39,40,61 This is supported by observations in a subset of clinical cases with restricted PrPSc accumulation, which was invariably confined to the brainstem. 57 The medulla oblongata currently remains the anatomic predilection site for application of diagnostic tests in passive and active surveillance.18,38,63,68 Where active surveillance is employed for BSE, it is important to establish if and to what extent this defined phenotype is expressed preclinically and therefore whether diagnostic approaches used in passive surveillance, including sampling and test method, are applicable during the incubation period.
Studies of the pathogenesis of BSE in cattle have been based on an experimental design in which calves are dosed orally with 100 g1,2,20,37,63,69,71 or 1 g2,14,37 of affected brainstem and sequentially killed at intervals throughout the incubation and clinical periods. The detection of PrPSc in this experimental model utilizing a range of diagnostic approaches has been used to estimate the temporal relationship between initial detection of PrPSc by diagnostic tests and incubation period or age of cattle. 2 In this model, the distribution of tissue infectivity or PrPSc during the incubation period suggests initial involvement of brainstem and spinal cord.1,20,71 Information from this model has also been used in the assessment of the likelihood of infectivity in the neuraxis and those associated peripheral nervous system (PNS) ganglia in relation to the removal of specified tissues (specified risk materials; SRM) at slaughter for the control of BSE.12,37
The present study utilizes tissues analyzed in a previous oral exposure study in cattle 2 —first, to define PrP IHC labeling forms observed in the brainstem and certain PNS ganglia in the model and, second, to map the topography and temporal progression of disease-specific labeling in the brainstem and ganglia over the time course of the disease. Progression of PrP deposition was also correlated with the onset of vacuolar changes in the brainstem and the onset of clinical signs. Knowledge of this progression of changes in this experimental model of BSE-C in the natural host thus consolidates the interpretational criteria for confirmatory IHC of preclinical natural cases identified through active surveillance and informs on risk assessment and risk management with regard to SRM removal.
Materials and Methods
All the materials for this study were sourced from archived tissues of two previous experiments of the oral exposure of cattle to the BSE agent.2,35,38,43,64,69,71
Source Animals and Inoculum
All animal experimentation was conducted according to the Animals (Scientific Procedures) Act 1986 under license from the UK government’s Home Office. Details of the design of the first experimental challenge study of BSE (experiment 1) have been reported.64,69,71 In brief, 40 Friesian/Holstein calves, born in Great Britain (GB) in 1991, were sourced from farms with no history of BSE. The inoculum consisted of a homogenate of a pool of brainstems from 75 cases of BSE (inoculum code BBP12/91) sourced in GB in 1991. End point titration of the homogenate pool in RIII mice gave a titer of 103.5 mouse intracerebral and intraperitoneal ID50 (median infective dose) units per gram of tissue and murine strain–type characteristics consistent with BSE-C. At 3 to 4 months of age, 30 calves were orally dosed with 100 g of the pooled brainstems, applied by syringe to the base of the tongue at the entrance to the pharynx. 35 Ten calves received no treatment and served as controls.
Clinical monitoring of cattle was maintained throughout the study.64,71,72 Starting at 2 months postexposure (mpe) and then at 4-month intervals until 22 mpe, 3 challenged calves and 1 control calf were randomly selected and killed for postmortem examinations. Thereafter, challenged and control cattle were killed at discretionary intervals, with the final kill at 40 mpe. 64
In the second experiment (experiment 2), two groups of 100 calves, born in 1998 and also sourced from GB farms with no prior history of BSE, were orally dosed at 4 to 6 months of age, one group with 100-g and the other with 1-g of a pool of BSE-affected brainstem tissue derived from 254 cases of BSE sourced in GB in 1996 and 1997. End point titration of the brain pool homogenate (inoculum code SE1736:BBP1) in RIII mice gave a titer of 103.1 mouse intracerebral and intraperitoneal ID50 units per gram of tissue and, again, strain-typing characteristics of BSE-C. A further 100 calves, similarly sourced, served as undosed controls. Clinical monitoring of cattle throughout the study was conducted as before. 72 Six challenged and three age-matched unexposed control cattle were randomly selected and killed at 3-month intervals after exposure, increasing to 6-month intervals after the first year postexposure, in the case of the 1-g dose group. As previously described, 72 four categories of the clinical TSE status were defined: no apparent signs, possible signs, probable signs, and definite signs of BSE. The last status was assigned to animals displaying evident neurologic signs consistent with BSE, comparable with a stage when the animal would have been reported as a BSE suspect field case by a farmer, and, in this study, only animals with definite signs were considered to be in the clinical stage of disease.
Polymorphisms of the prion protein gene (PRNP) are important determinants of susceptibility and incubation period in most species affected by TSE. Although not associated with any susceptibility traits to BSE infection in GB,23,49 an octapeptide repeat polymorphism of the coding region of PRNP in cattle occurs as either five, six, or seven copies, with genotypes expressed as 5:5, 6:5, 6:6, or 6:7.16,49 Cattle homozygous for six-octapeptide repeats predominate in the GB population. As for some previous transmission studies of BSE in cattle, this polymorphism was determined from DNA extracted directly from EDTA blood samples for all calves entering the two experiments. 65 A 400–base pair (400-bp) fragment of the PRNP, containing the octapeptide repeat region, was amplified by polymerase chain reaction. The octapeptide repeat allelism was then determined by gel electrophoresis. Different genotypes were allocated across test and control groups to provide, as far as possible, an even mix in the populations.
Tissues
Tissues sampled at each sequential kill of cattle in both experiments were those described for a previous study. 2 The brainstem and spinal cord, dorsal root ganglia (DRG), trigeminal ganglion, stellate ganglion, and cranial cervical ganglion, collected into 10% formol saline, were subject to histopathologic examinations. After primary fixation, the brainstem was cut to give 3 coronal blocks52,70 selected on the basis of previous knowledge of the stereotypic distribution pattern of changes in BSE in GB:10,53,63 medulla oblongata at the obex, medulla at the level of the rostral cerebellar peduncles, and mesencephalon at the level of the rostral colliculi and the medial geniculate nucleus. Transversely oriented blocks of spinal cord were cut at C5 (experiment 1) or C6 (experiment 2), T6, and L5-6. DRG were selected from cervical and thoracic spinal cord segments. The majority of selected DRG were C6 and T6 (experiment 1) and C7 and T4 (experiment 2). Where these DRG were not available, the next adjacent segment ganglion was taken (range: C3–C8, T2–T11). Tissues were processed routinely and embedded in paraffin wax. Table 1 presents the total number of each tissue available to the study, as examined according to experiment and dose group. Previously published results for the biochemical examination of corresponding frozen tissue samples for PrPSc, where available, provided alternative diagnostic criteria for assessment of findings. 2 The biochemical techniques applied were a commercially available ELISA test approved for the diagnosis of BSE (Bio-Rad TeSeE, Bio-Rad, Hercules, CA) and a Western blot including a PrPSc enhancement step using precipitation with sodium phosphotungstate dibasic hydrate (WBNaPTA). 2 The WBNaPTA was not conducted on ganglia.
Total Number of Each Tissue Examined According to Experiment and Treatment Groups a
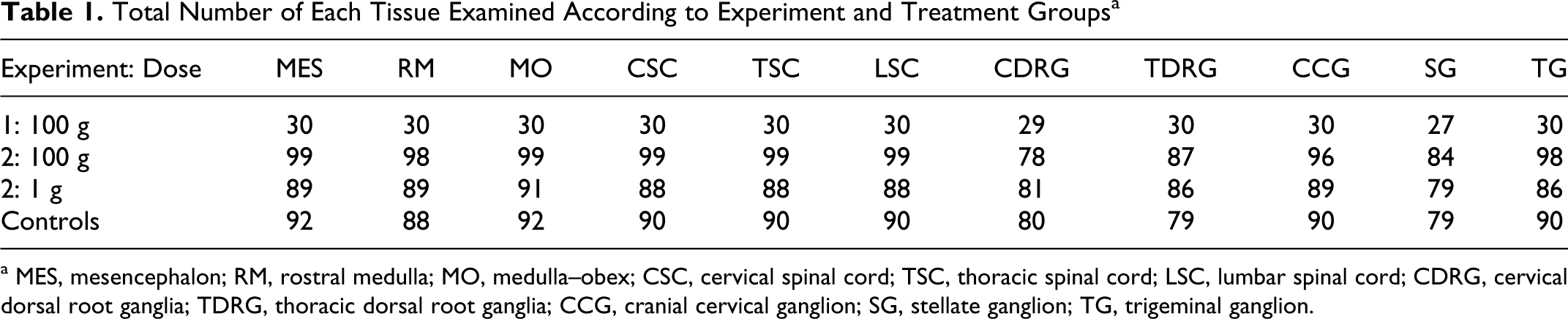
a MES, mesencephalon; RM, rostral medulla; MO, medulla–obex; CSC, cervical spinal cord; TSC, thoracic spinal cord; LSC, lumbar spinal cord; CDRG, cervical dorsal root ganglia; TDRG, thoracic dorsal root ganglia; CCG, cranial cervical ganglion; SG, stellate ganglion; TG, trigeminal ganglion.
IHC and Histologic Preparations
IHC detection of PrPSc was carried out as previously described 11 using two anti-PrP monoclonal antibodies (mAb) employed for confirmatory diagnosis of BSE: R145 (Veterinary Laboratories Agency, Weybridge, UK) 59 and F99 (VMRD Inc, Pullman, WA). 41 All the tissues from a single case were immunolabeled on the same technical run. For examination of vacuolar changes, 5-μm sections were cut and stained with hematoxylin and eosin.
Recording of Observations and Distribution Mapping of Pathologic Changes
Twenty cases were initially analyzed unblinded, as selected by review of the original data from experiment 2 and comprising 4 BSE-affected animals, 2 from the 1-g dose group, 2 from the 100-g dose group, and 16 undosed control animals. 2 These were reviewed, and a schema was devised for the recording of immunolabeling forms. Immunolabeling deemed incidental or artifactual and recorded in undosed controls was defined as part of this exercise to establish baseline interpretational criteria for the experimental model, to enable the forms of labeling to be subsequently disregarded in the main study.
In subsequent evaluations, investigators were blinded to dose grouping or disease status. Observations of the occurrence of spongiform changes and/or disease-specific immunolabeling by CNS region were recorded. Immunolabeling in peripheral nerve ganglia was similarly recorded. As a result of the initial assessment (unblinded series) of ganglia, in which occasional vacuoles within neuronal perikarya were a feature of some test animals and some control animals, vacuolar changes were not recorded in ganglia. The forms and topographic distributions of immunolabeling in the brain were mapped using diagrams based on a stereotaxic atlas of the Friesian cow brain 34 as previously used for mapping vacuolar changes. 70 The configurations and neuroanatomic locations of immunolabeling and, where observed, their cellular associations, were diagrammatically recorded for all positive brain and spinal cord sections. For the spinal cord levels, labeling was mapped according to the main neuronal groups, drawn from appropriate levels of bovine spinal cord and approximated, as far as possible, to Rexed laminae. 44 The diagrammatic representations of the forms and extent of immunolabeling for each neuraxis level were ordered from the least to the greatest extent of labeling.
Results
Immunolabeling Definitions and Interpretation
Both antibodies revealed the same forms and patterns of immunolabeling in the CNS, with non-disease-specific labeling generally more marked and widespread with the mAb F99. Distinctions between non-disease-specific (Figs. 1–6) and disease-specific (Figs. 7–12) labeling forms in the initial, unblinded series were necessarily based on the nature and configuration of the immunolabeling, presence of cellular associations indicative of pathologic change, and correlation with disease status as determined by alternative diagnostic methods (WBNaPTA and Bio-Rad TeSeE). 2
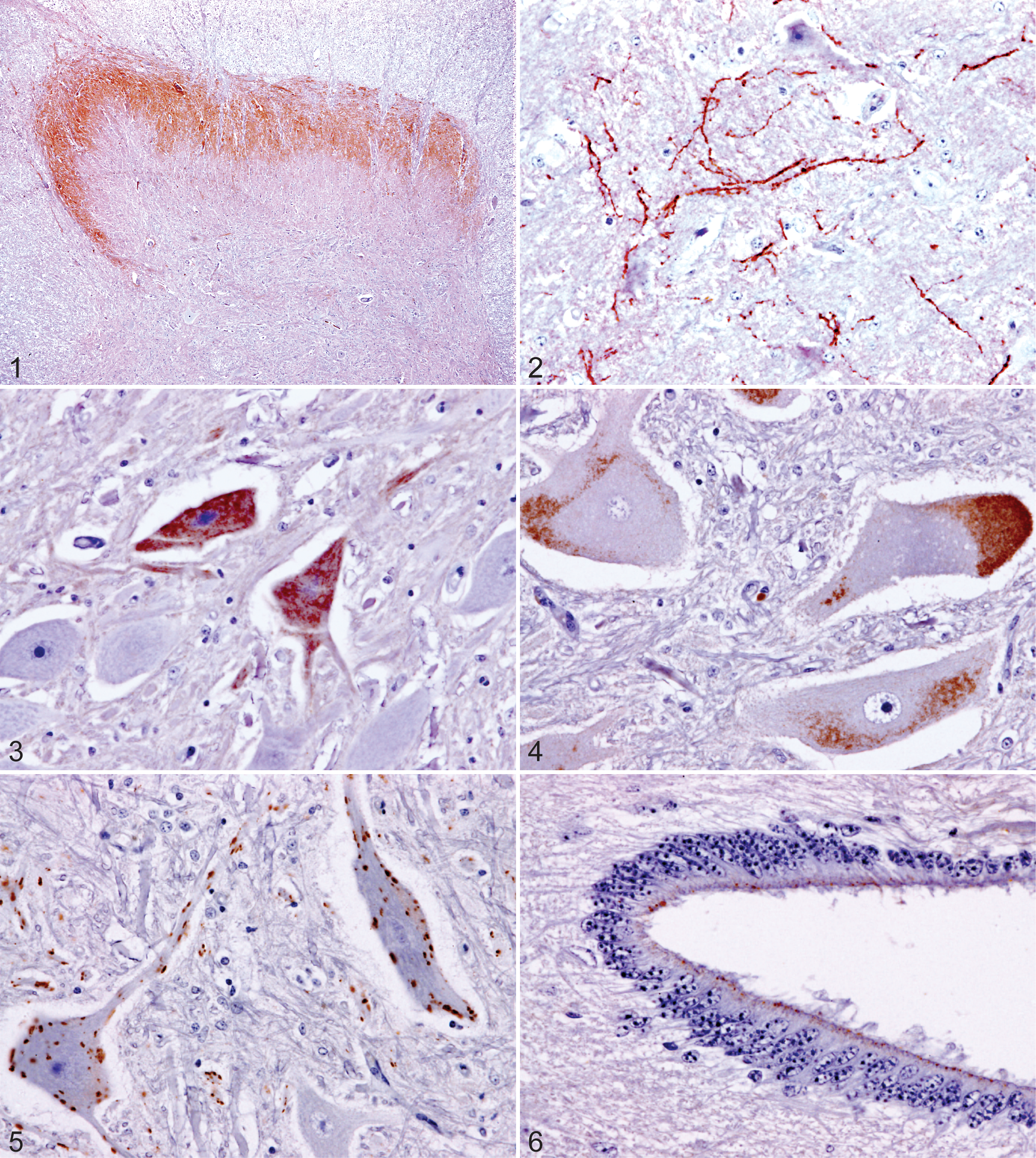
Nondisease-specific immunohistochemical labeling in the central nervous system with anti-PrPSc monoclonal antibody R145 or F99.
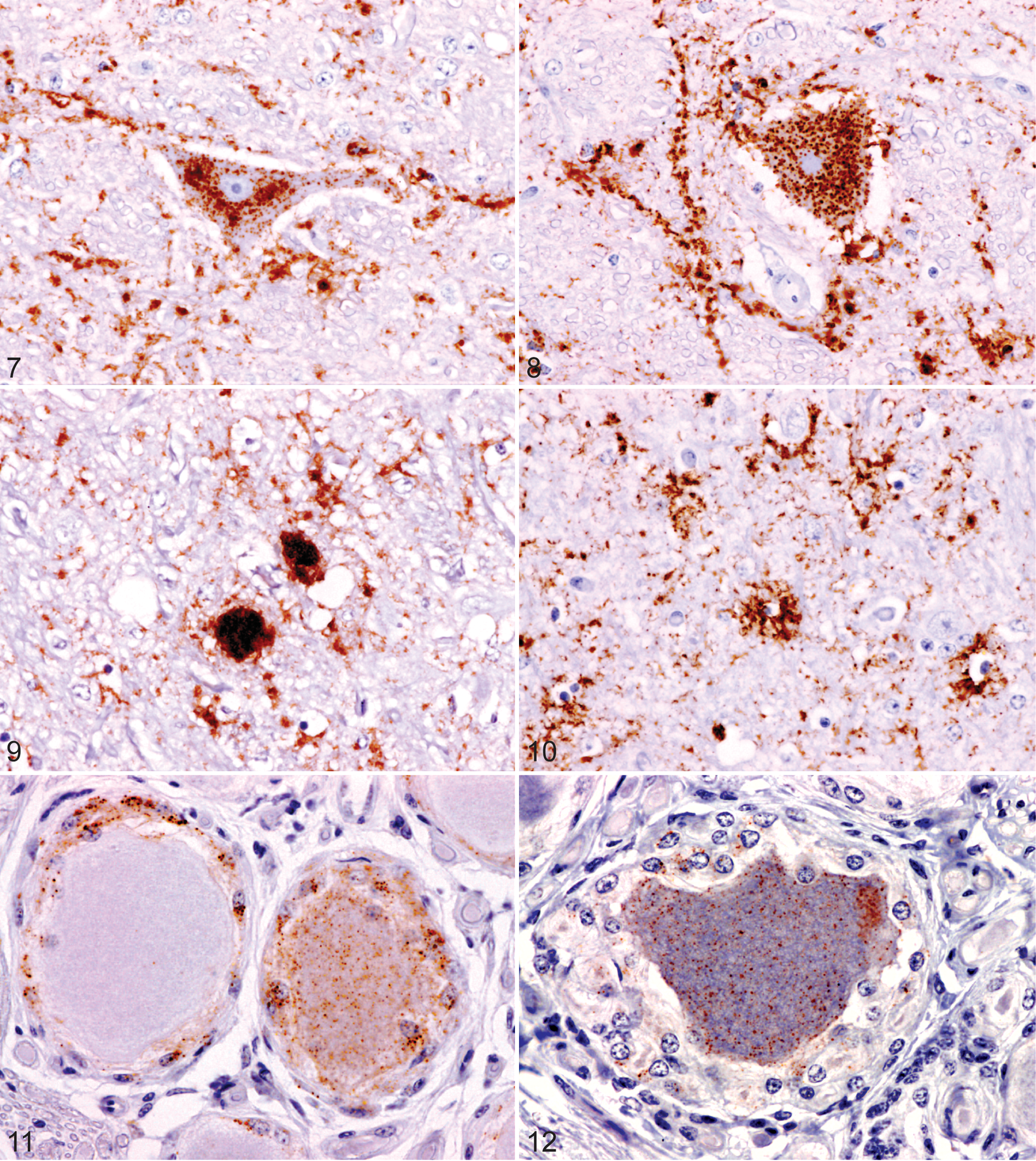
Disease-specific immunohistochemical labeling patterns in the central nervous system and peripheral nerve ganglia with anti-PrPSc monoclonal antibody R145 or F99 in cases of experimentally induced bovine spongiform encephalopathy.
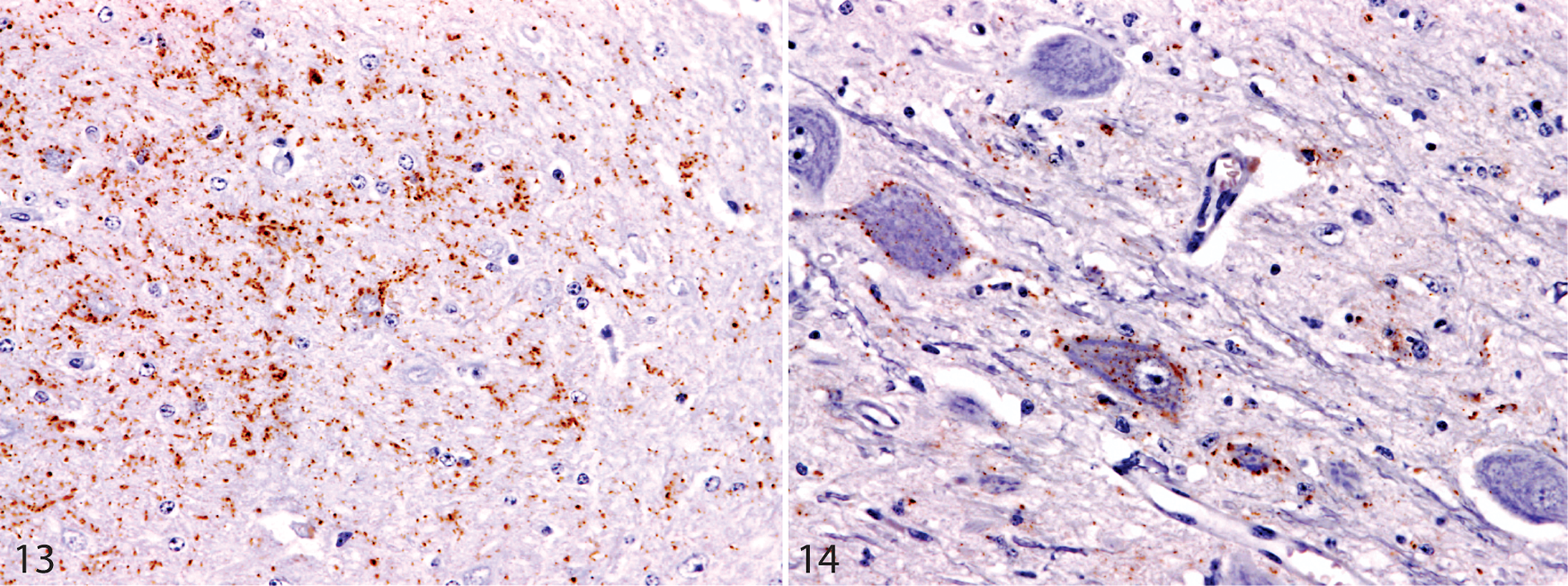
Medulla–obex; nucleus of the solitary tract, case No. 10. PrPSc labeling (particulate) confined to this single nucleus. R145.
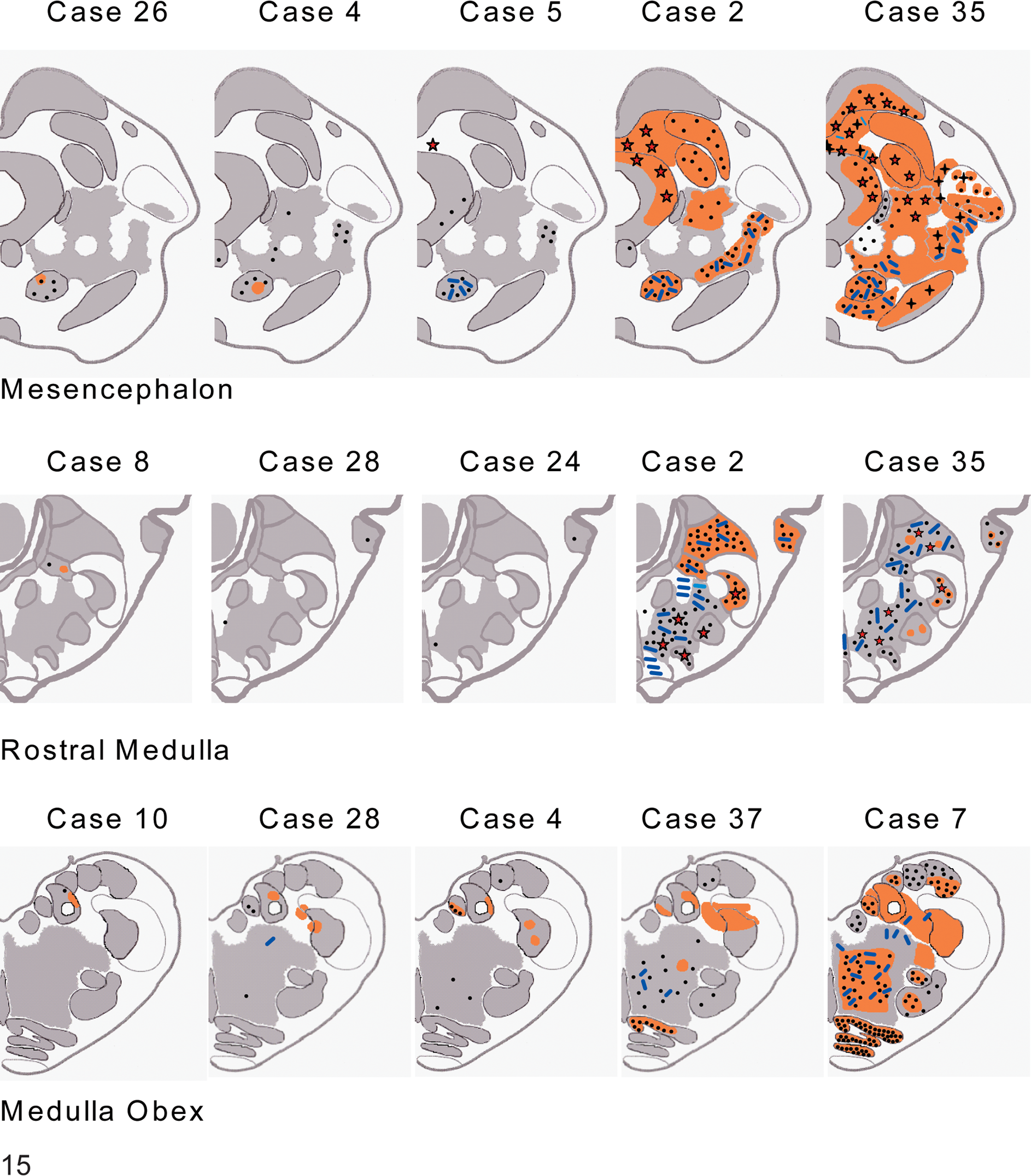
Disease-specific immunolabeling distribution patterns in brainstem with examples of labeling patterns for each brain level examined: minimal (first three images of each row), moderate (fourth image of each row), and maximal (final image of each row). Refer to Table 4 for experiment number and months postexposure of cases cited. Symbols: •, granular cytoplasmic (gc); —, linear (lin); orange shading, particulate (part) ☆, stellate (stell);
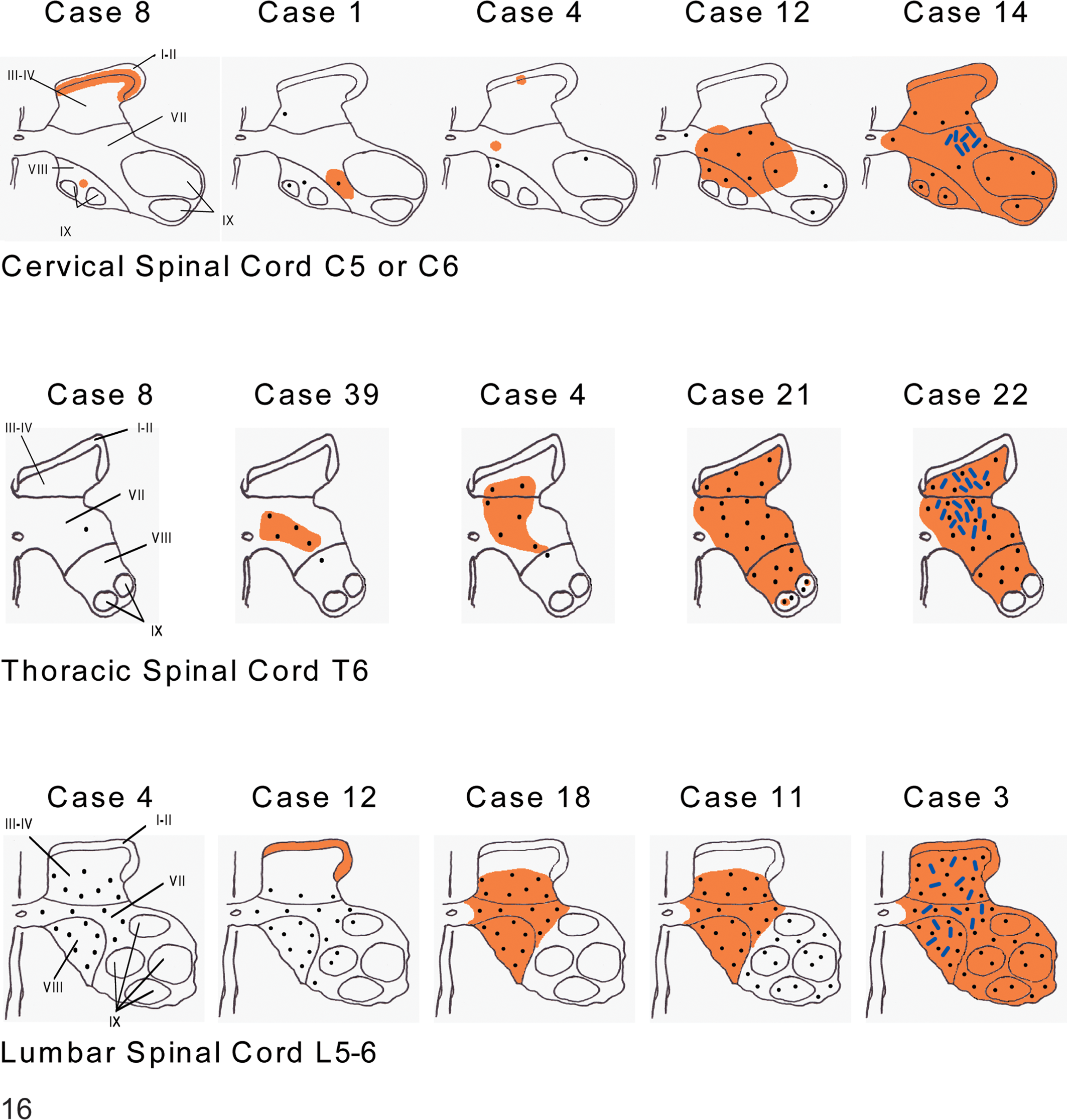
Disease-specific immunolabeling distribution patterns in the spinal cord with examples of labeling patterns for each level examined: minimal (first three images of each row), moderate (fourth image of each row), and maximal (final image of each row). Spinal cord gray matter neuronal groups at cervical, thoracic, and lumbar levels were mapped approximating to Rexed laminae I–IX, 28 as indicated on the first diagram for each spinal cord level. Refer to Table 4 for experiment number and months postexposure of cases cited. Symbols: •, granular cytoplasmic (gc); —, linear (lin); orange shading, particulate (part). Spinal cord–cervical. The least labeling presented as part in laminae I–III and VIII (eg, case No. 8), as gc in occasional neurons in VI and VIII with sparse part in lamina VII (eg, case No. 1), or as occasional neurons with gc in VIII and IX and focal part in I–II and VII (eg, case No. 4). In some more extensively labeled cases, the ventral and intermediate zones were involved without labeling in the dorsal horn (eg, case No. 12). The most extensive pattern in clinical cases was part and gc labeling throughout the gray matter with lin in the intermediate zone (lamina VII) (eg, case No. 14). Spinal cord–thoracic. The least immunolabeling was in a single neuron, in lamina VII (case No. 8). Additional labeling was seen as sparse gc and part in laminae VII and VIII (eg, case No. 39) extending to laminae III (eg, case No. 4).
Nonspecific labeling forms in CNS encompassed the spectrum of technical artifact and epitope-specific labeling related to cross-reactivity and probable PrPc locations and were observed in control and exposed animals (Figs. 1–6). Incidental immunolabeling forms present in the PNS ganglia of control animals were distinct from those in the CNS and comprised diffuse faint labeling throughout the cytoplasm of occasional satellite cells, focal wisps of labeling in the interneuronal interstitium, and fine granular cytoplasmic labeling associated with the cell membrane.
In the brainstem and spinal cord the most prevalent disease-specific labeling comprised particulate deposits distributed widely in the neuropil. In addition to scattered particles (Fig. 7), these deposits were sometimes aggregated or arranged to present perineuronal (Fig. 7), linear (Fig. 8), plaque-like (Fig. 9), and periglial stellate (Fig. 10) forms. Labeling within neuronal perikarya was granular cytoplasmic in form (Figs. 7, 8).
Disease-specific labeling in peripheral ganglia was limited to a diffuse fine granular form throughout the cytoplasm of occasional neurons in the trigeminal ganglion and DRG with a marked variation in granule size (Figs. 11, 12). This labeling was also seen in some associated satellite cells (Fig. 11). Detection of the labeling required systematic searches of fields at 200× magnification, which contrasts with the similar but more uniformly coarse granular neuronal perikarya labeling (granular cytoplasmic) in the CNS, which in most instances was readily detected at lower magnifications.
Temporal Occurrence of PrPSc Labeling and Spongiform Changes
The occurrence of disease-specific immunolabeling and spongiform changes by CNS region were expressed according to experiment, dose, and time after exposure (Table 2). Immunolabeling in peripheral nerve ganglia was similarly recorded (Table 3). Disease-specific labeling in CNS was detected in a total of 39 cases. In general, granular cytoplasmic and particulate forms were observed from an early stage, with stellate, linear, and plaque-like not occurring until immunolabeling was relatively widespread. Individual animal results for immunolabeling and spongiform changes (Table 4) showed that, with 5 exceptions, in all 100-g dose group cases, PrPSc was detected at all levels of brainstem and spinal cord examined. In 2 of 4 cases (case Nos. 24, 28) where PrPSc was not detected at any level of the spinal cord, it was present in both levels of medulla. In the remaining 2 cases (Nos. 10, 20), PrPSc was confined to medulla–obex, although the neuroanatomic targeting was different in each case (Figs. 13, 14). In these 4 cases, additional examinations made of brain regions rostral to the brainstem did not reveal further immunolabeling. In the fifth animal (case No. 8), immunolabeling was detected focally throughout the brainstem and spinal cord but involved only 1 or 2 neurons at each level examined. These cases with restricted labeling did not exhibit definite clinical signs and were killed 38 to 45 mpe.
Data on the Central Nervous System of All Animals Killed at and Subsequent to the Sequential-Kill Time Point Postexposure When PrPSc Was First Detected by Immunohistochemistry With Monoclonal Antibody R145 or F99 a
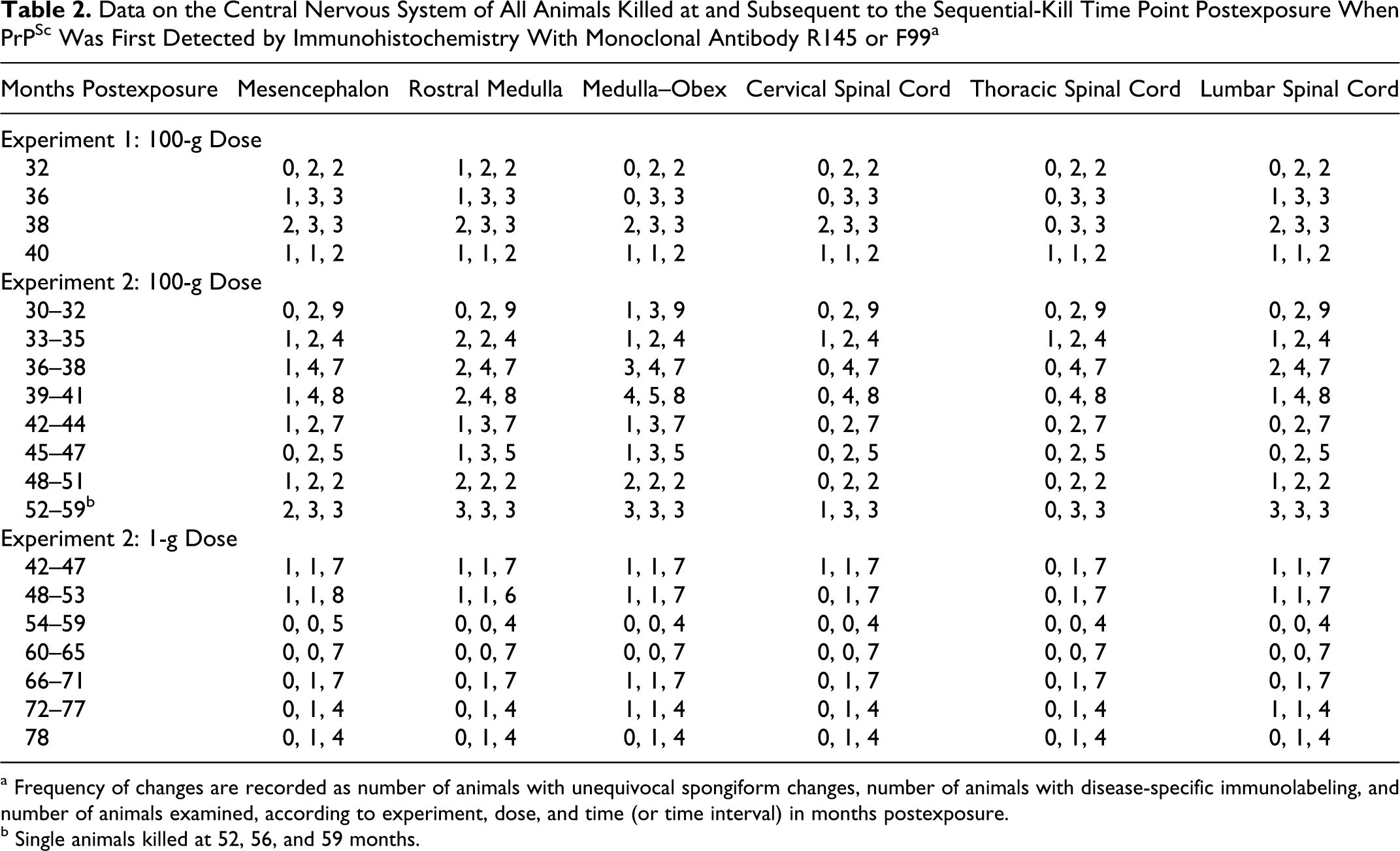
a Frequency of changes are recorded as number of animals with unequivocal spongiform changes, number of animals with disease-specific immunolabeling, and number of animals examined, according to experiment, dose, and time (or time interval) in months postexposure.
b Single animals killed at 52, 56, and 59 months.
Data on Peripheral Nerve Ganglia From All Animals Killed at and Subsequent to the Sequential-Kill Time Point Postexposure When PrPSc Was First Detected in Ganglion Tissues by Immunohistochemistry With Monoclonal Antibody R145 or F99 a
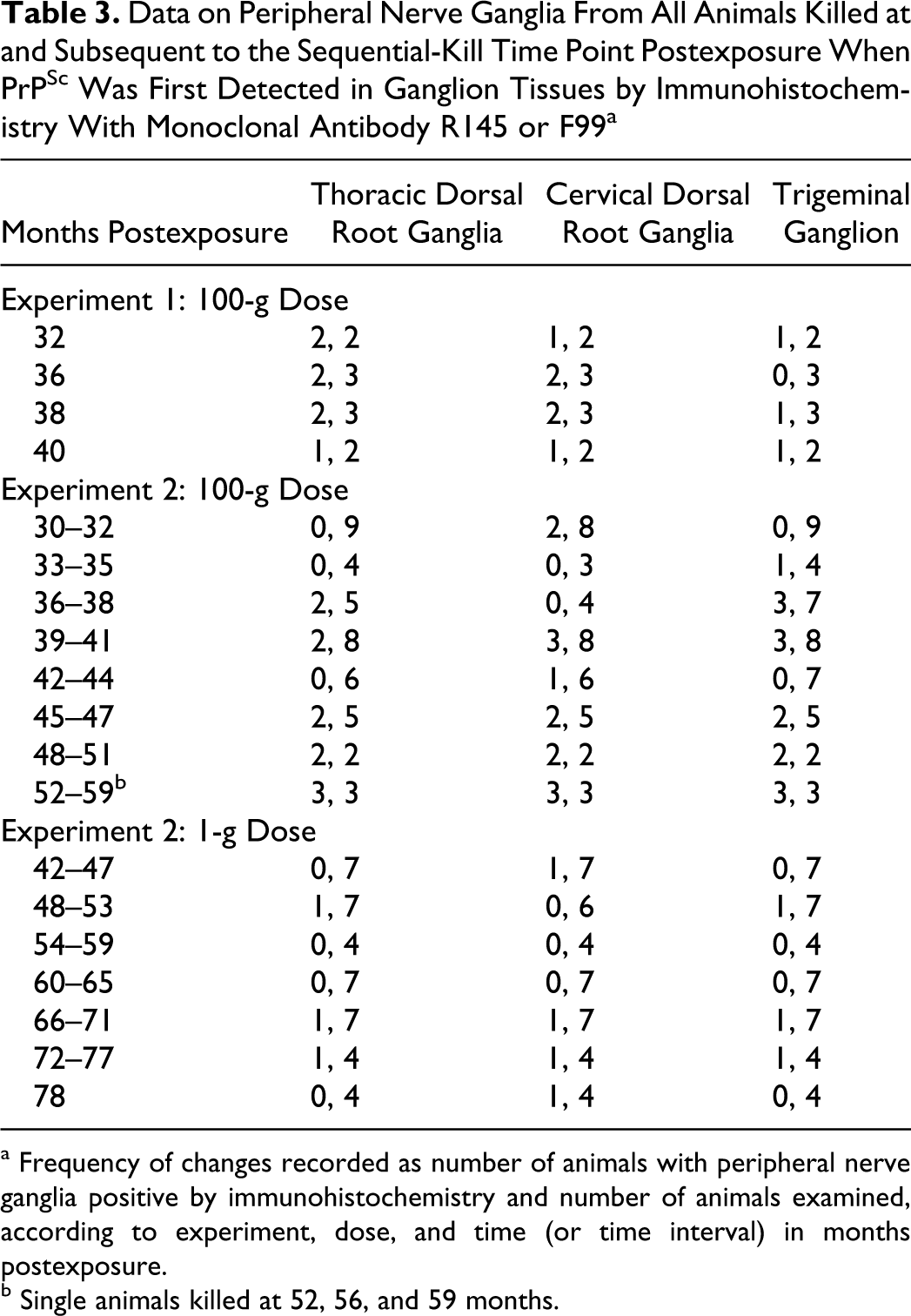
a Frequency of changes recorded as number of animals with peripheral nerve ganglia positive by immunohistochemistry and number of animals examined, according to experiment, dose, and time (or time interval) in months postexposure.
b Single animals killed at 52, 56, and 59 months.
Individual Animal Data: Clinical Status, Disease-Specific Immunolabeling, and Central Nervous System Spongiform Change in All Animals That Gave Positive Disease-Specific Immunolabeling in at Least One Brainstem Region (n = 39) a
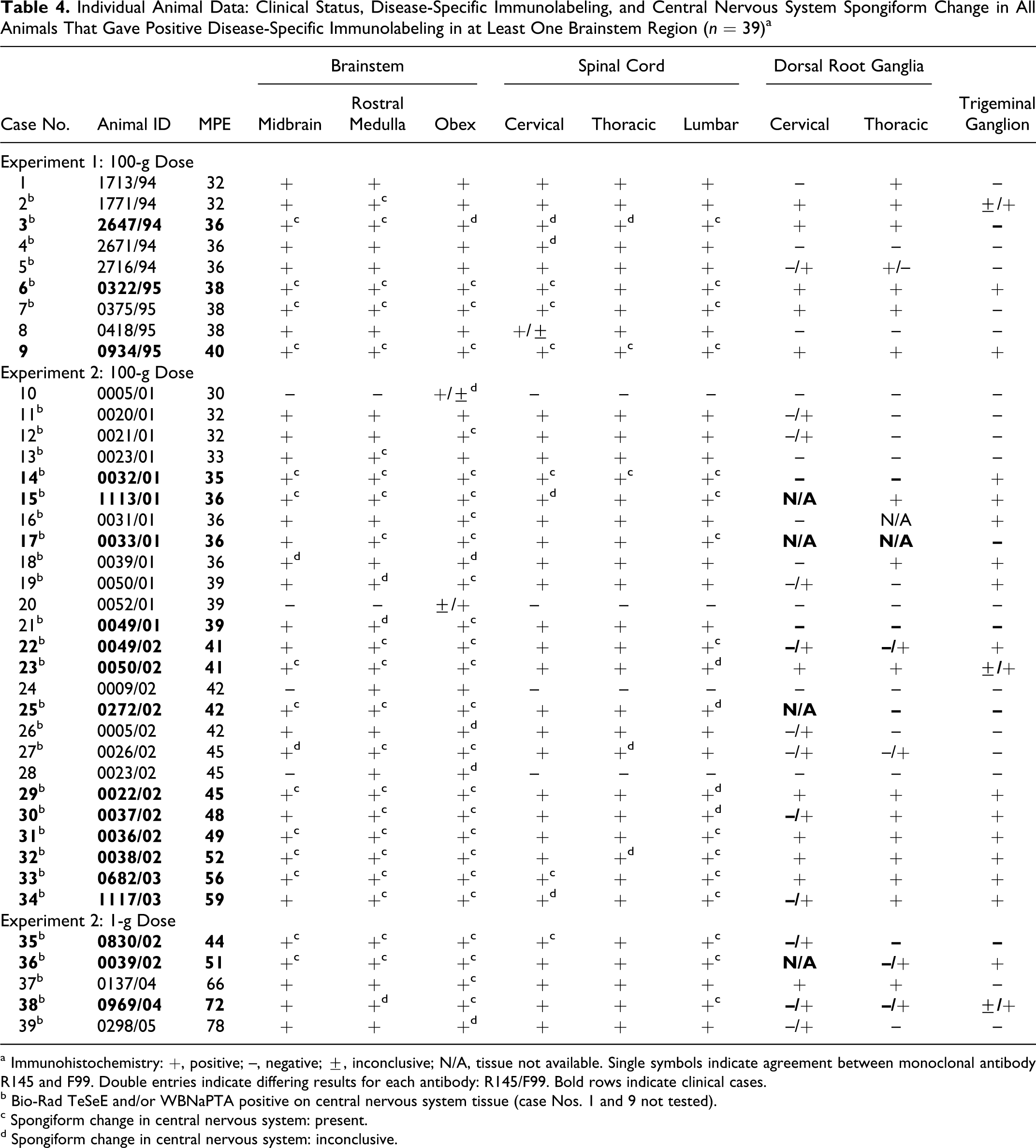
a Immunohistochemistry: +, positive; –, negative; ±, inconclusive; N/A, tissue not available. Single symbols indicate agreement between monoclonal antibody R145 and F99. Double entries indicate differing results for each antibody: R145/F99. Bold rows indicate clinical cases.
b Bio-Rad TeSeE and/or WBNaPTA positive on central nervous system tissue (case Nos. 1 and 9 not tested).
c Spongiform change in central nervous system: present.
d Spongiform change in central nervous system: inconclusive.
Labeling in DRG and trigeminal ganglion was not observed without labeling in the brain and spinal cord and, in general, was recorded more consistently in later-onset clinical cases. Whereas differences in detection of PrPSc and the extent of labeling between the antibodies were not apparent in the majority of cases and tissues, F99 appeared to result in marginally increased sensitivity for disease-specific labeling in many ganglia and in 1 medulla–obex (case No. 20). Occasionally, the converse was true (in 1 thoracic DRG and 2 medulla–obex) (Table 4).
Vacuolar changes diagnostic of a spongiform encephalopathy were present in the brainstem, but not exclusively at the obex, in 27 of 39 cases. In the initial cases in which vacuolar changes were detected at 32 mpe in the 100-g dose groups of experiments 1 and 2, the changes were confined mainly to neuronal vacuolation in the vestibular nuclear complex in the rostral medulla (case No. 2) or spongiform change in the medulla–obex (case 12), respectively. This was compared with initial detection of PrPSc, at 32 mpe (case Nos. 1, 2) in experiment 1 and at 30 mpe (case No. 10) in Experiment 2. In 14 of the 39 positive cases, PrPSc immunolabeling preceded diagnostically significant spongiform changes in the obex (13 in the 100-g dose groups, 32 to 45 mpe; 1 in the 1-g dose group, 78 mpe). In no cases did spongiform encephalopathy precede PrPSc labeling. As distributed throughout the time course from initial PrPSc detection, 24 of the 39 cases (Table 4) had unequivocal vacuolation in the medulla–obex, and 18 of these showed definite clinical signs. As noted previously in the natural disease, 63 there was 1 clinical case (case No. 3) in which conclusive vacuolar changes could be confirmed in the brainstem only rostral to the obex. In 2 preclinical cases (case Nos. 2 and 13), spongiform changes were also detected in the rostral medulla but not at the obex. Unequivocal spongiform changes were present in the medulla oblongata (rostral or at the obex) in all clinical cases.
Vacuolar changes in 8 cases (case Nos. 2, 7, 12, 13, 16, 19, and 27 in the 100-g dose groups, 32 to 45 mpe; and case No. 37 in the 1-g dose group, 66 mpe) occurred without definite clinical signs (Table 4). Furthermore, according to the criteria used in the study for the definition of clinical status, 68 none of these showed evidence of probable clinical signs, whereas case Nos. 10, 24, 26, and 39 all showed probable signs but lacked unequivocal spongiform changes. Two animals (case Nos. 37 and 39, 1-g dose group) had widespread immunolabeling and, in case 37, unequivocal vacuolation but were not considered clinical cases. Whereas unequivocal spongiform changes were present in the lumbar spinal cord in 14 of 19 clinical cases, only 5 clinical cases had such changes in the cervical cord and only 2 in the thoracic cord (Table 4). No disease-specific immunolabeling or vacuolar changes were detected in any tissues from the unexposed animals. There was also no specific labeling in any tissues of exposed animals prior to 32 mpe in experiment 1 or 30 mpe and 44 mpe (100-g and 1-g dose groups, respectively) in experiment 2 (Tables 2–4).
Topographic Distribution of Disease-Specific Immunolabeling
The extent of disease-specific immunolabeling at each level of the neuraxis ranged from sparse focal to widespread (Figs. 15, 16). An increasing extent of labeling and, therefore, an apparent progressive accumulation did not have a clear temporal relationship with the period postexposure; but, in general, the least labeling was associated with cattle late in the incubation period, and the greatest extent of immunolabeling was associated with the presence of clinical disease. Neuroanatomically restricted or minimal labeling was confined to the 100-g dose group. The small number of cases resulting from the 1-g dose prevented any detailed comparisons of distribution patterns between the dose groups, but no obvious differences were observed.
The least immunolabeling in medulla–obex—the specific brainstem area sampled for the statutory diagnosis of BSE—was seen in 5 animals (case Nos. 4, 8, 10, 20, 28) (Table 4), 4 of which showed some neurologic signs but were not considered to be definite clinical cases of BSE.
In the trigeminal ganglion and DRG, disease-specific labeling was infrequent, and it involved only a small proportion of the neurons represented in any one section. There was little difference in the extent of disease-specific labeling, although this was often more readily identified in sections labeled with the F99 mAb but against a background of more nonspecific labeling than that with the R145 mAb. Satellite cell labeling was variable; not all labeled neurons had satellite cell involvement, and vice versa. In the trigeminal ganglion of 6 animals from the 100-g dose group—all killed during the clinical stages of disease—there were relatively larger numbers of affected neurons (from about 20 to more than 60; case Nos. 15, 22, 29, 30, 33, 34), but this still represented less than 5% of the visible neuronal population.
Only 2 animals from the 1-g dose group had disease-specific labeling in the trigeminal ganglion, and in both cases only in 1 or 2 neurons of the more than 100 examined. In one case, this labeling was confined to neurons (case No. 38); in the other, it involved the associated satellite cells. No immunolabeling was detected in stellate or cranial cervical ganglia.
PrP Sequencing
Genotyping of all challenged and control animals with respect to the octapeptide repeat polymorphism of the PRNP resulted, as expected, in the 6:6 genotype predominating but with the 6:5 genotype occurring in 40 cattle (experiment 2) and the 5:5 genotype in 1 (experiment 2). Confirmed cases were mainly of the 6:6 genotype, but 4 were 6:5 (case Nos. 14, 21, 24, 34) (Table 4). The frequency of the 6:5 genotype was similar in the remaining experimental population of control and unaffected challenged cattle.
Discussion
The morphologic forms and CNS distribution patterns of PrPSc immunolabeling described here, resulting from the experimental oral exposure of cattle, are entirely consistent with what has been described for natural BSE in GB.10,20,70 The CNS changes, both vacuolar lesions and PrPSc immunolabeling, in all clinically affected animals were similar, suggesting that the spatial progression of changes and, therefore, the pathogenesis are similar irrespective of dose. 2 The phenotype is consistent with BSE-C, characterized on the basis of pathology and biochemical properties of PrPSc and differentiated from variant forms, denoted BSE-H and BSE-L (referring, respectively, to either a high or low molecular weight of the unglycosylated band in a Western blot). 25
The greater attack rate and shorter incubation periods in the 100-g dose group compared with the 1-g dose group are entirely consistent with attack rate data 65 and with previous modeling of the temporal relationship between PrPSc detection and incubation period in animals of this study. 2 These experimental challenges provide a host-specific model in which the inferred pathogenesis of BSE-C can be studied by the immunochemical methods that are an important component of current BSE surveillance programs. The model can also be deployed to inform on the appropriateness of current controls to prevent human exposure to the BSE agent.
Reported polymorphisms in the promoter region of the PRNP have been implicated in susceptibility to occurrence of BSE.9,27,28,48 These were not determined for animals in the present study, but PRNP open reading frame sequencing and the 23-bp and 12-bp insertion/deletion polymorphisms—considered to have a substantial genetic effect on BSE susceptibility 28 —were determined for cattle in a related experimental transmission study, 61 without significant differences in polymorphisms between exposed cattle that developed or did not develop disease (G. Saunders, personal communication). The relatively small numbers of cattle examined in the experimental study 65 were insufficient to draw valid conclusions. Furthermore, because the allele combinations in question do not confer complete resistance to BSE and because the largest risk factor for developing BSE remains exposure to the agent, significant genetic effects on susceptibility are likely to be apparent from studies of representative numbers from large cattle populations. 28 Additional genetic analysis extended to cattle in the present study would substantially increase the data available from experimental transmission studies 35 and might provide insights to variability in susceptibility and incubation period.
Neither of the antibodies chosen for the study (or other available antibodies) exhibit exclusive binding to PrPSc epitopes, so the first goal was to define disease-related versus non-disease-related (specific vs nonspecific) and immunolabeling forms, which could result from binding of the antibody to the specific epitope against which it was raised, which might be present in proteins other than PrPSc. Modulation of the immunolabeling signal by dilution of the primary antibody diminishes disease-specific and unwanted immunolabeling, which could reduce the sensitivity of the method. Attention to the detail of form, topography, and cellular association of PrP immunolabeling is a reliable approach to distinction of nonspecific and disease-specific reactivity. In the diagnosis of field cases, these descriptive elements play a pivotal role in confirming equivocal results. F99 appeared to have better sensitivity in the ganglia of a small number of minimally immunolabeled cases, but the levels of nonspecific labeling seen with this mAb were greater than that obtained with R145.
Non-disease-specific labeling forms in relation to certain PrP antibodies have been described in pigs and mice8,46 but are not well documented in cattle. One form, filamentous, notably in the medial geniculate nucleus, appeared to be consistent in anatomic location and cellular association and, although variable in both extent and density, consistently presented in controls and negative and positive test animals. One possible explanation may be that there are higher local levels of PrPC expression that are above the modulated threshold of the IHC method for detection of disease-specific labeling. This form of labeling has been recorded in cattle and mice unaffected by TSE (G.A.H.W., unpublished observations) and reported as “thread-like” in otherwise normal sheep. 47 Similarly, it has been noted at restricted brain locations in pigs inoculated with BSE but in which confirmatory pathologic evidence of prion disease was lacking. 31 It has also, in the absence of adequate control material, been interpreted as an atypical pattern of scrapie-specific labeling in goats. 56 Discrete, finely granular labeling colocalizing with lipofuscin pigment and restricted or eccentrically localized in the perikaryon has been reported 26 and probably represents lysosomal PrPc. Other reproducible non-disease-specific labeling that has not been previously described was, in general, antibody associated: Diffuse nonparticulate labeling affecting some gray and white matter, diffuse cytoplasmic labeling of neurons, and subapical granular labeling in ependymal cells were features of mAb F99, and rod or seedlike (lenticular) intracytoplasmic labeling of neuronal perikarya was seen only with mAb R145 and tended to be restricted to certain neuronal groups. Disease-specific labeling forms were consistent with those previously described in naturally occurring terminal BSE with regard to morphology and anatomic distribution, although there are inconsistencies of nomenclature throughout the literature.10,36,55,68,70
In this study, in the 100-g dose groups from both challenge experiments, PrPSc could be detected bilaterally at multiple levels of CNS from 32 mpe, as previously reported. 64 Of particular interest in terms of diagnosis are those cases in which PrPSc labeling is focal or restricted. Labeling, confined to medulla–obex, occurred unilaterally in a single animal at 30 mpe (case No. 10) and bilaterally in a further animal at 39 mpe (case No. 20). Another case at 38 mpe (case No. 8) had labeling of only a single neuron per CNS region, suggesting that dissemination of PrPSc can be widespread at very low levels, without evidence of spread from restricted specific primary target areas. The overall timing of initial detection of PrPSc in the model is in keeping with the earliest detection of infectivity (by assay in bovinized transgenic mice) in brainstem samples from selected animals of experiment 2 at 27 mpe, with a marked rise in titer by 33 mpe. 14 The extent of labeling in relation to incubation period or time postexposure and dose was highly variable, although the greater extent of labeling generally occurred in clinical cases.
The presence of PrPSc was variable in the DMNV, the solitary tract nucleus, and/or the spinal tract nucleus of the trigeminal nerve (case Nos. 10 and 20); these cases also had labeling in the medulla–obex. Similar findings have been observed in 7 naturally occurring cases detected in GB through active surveillance (M.M.S., unpublished data): 1 showed labeling restricted to the DMNV, and 4 had labeling in the solitary tract nucleus in the absence of DMNV involvement; the remaining 2 had no involvement of either area but had labeling restricted to the nucleus of the spinal tract of the trigeminal nerve. Although little can be concluded from this variation with respect to pathogenesis, the apparent initial manifestation of PrPSc in medulla–obex supports the efficacy of sampling this area for diagnosis. Because of small necessary differences in sampling locations for histologic techniques using fixed material and for biochemical methods using frozen material, studies such as this do not allow true comparison of the sensitivities of different diagnostic approaches. Nevertheless, biochemical methods gave negative results in some cases in which minimal but unequivocal disease-specific IHC labeling occurred (case Nos. 8, 10, 20, 24, 28) (Table 4), whereas the opposite was never observed. This suggests that IHC may have greater diagnostic sensitivity in terms of an ability to detect focal PrPSc at a cellular level, and it reinforces the need for sampling accuracy for active surveillance, when PrP distribution is likely to be restricted. At a practical level, using IHC also enables histologic confirmation of the anatomic accuracy of the sample being examined. In contrast, an erroneous tissue sample could be presented to a biochemical test and remain undetected, giving rise to false-negative results.
That initial immunolabeling in both spinal cord and brainstem in the majority of cases is consistent with the theory that after oral exposure, the pathogenesis of BSE follows the splanchnic (sympathetic) route via the spinal cord or the vagal route (parasympathetic) directly to the brainstem 20 or, most likely, both. Minimal PrPSc labeling in the thoracic spinal cord is in keeping with initial involvement of general visceral efferent neurons. This is also consistent with the pathogenesis reported for other TSE, such as scrapie or BSE in sheep, implicating the autonomic nervous system as the major route of neuroinvasion.13,45,61,62 An alternative hypothesis for neuroinvasion suggests that the agent can spread into the brain through the circumventricular organs, where the blood–brain barrier is absent. 54 However, the ability to detect PrPSc in the spinal cord, where the blood–brain barrier remains intact, as soon as it is detected in the medulla suggests that the neuronal route is the only significant way in which the agent spreads into the CNS in this model. Conclusions with respect to neural pathogenesis based on detection of PrPSc are subject to an important caveat—namely, that the sensitivity of the PrPSc detection reflects a constant relationship between concentrations of PrPSc and infectivity, which is broadly true with respect to studies of BSE pathogenesis in the natural host.1,2,60
PrPSc IHC labeling associated with DRG neurons has been reported in clinical cases of experimental transmission of BSE-C and BASE (for bovine amyloid spongiform encephalopathy, or BSE-L). 35 Labeling in sensory ganglia neurons (including trigeminal and DRG) and sympathetic ganglia neurons has also been described in human prion disease cases, including variant Creutzfeldt-Jakob disease.17,19,32 Western blotting examinations of selected PNS tissues, including DRG from the present study (100-g and 1-g dose groups), failed to detect PrPSc before detection in the brainstem and spinal cord. 37 This is significant in relation to SRM rules for the timing, relative to age, of the removal of the vertebral column (inclusive of DRG) at slaughter for the prevention of potentially infected tissues entering the food chain, 12 and it is consistent with the previous conclusion that there is retrograde involvement of the DRG from the CNS.2,37 That PrPSc could not be detected in the sympathetic ganglia (stellate ganglion and cranial cervical ganglion) examined in the present study might indicate that there is no generalized involvement of sympathetic nervous system components in BSE, as evidenced by detectable accumulation of PrPSc and despite being implicated in initial pathogenetic pathways. However, Western blot examinations of stellate ganglion from the same animals used in the present study have shown PrPSc in a proportion of cases but only in concert with a positive diagnosis on brainstem. 37 The Bio-Rad TeSeE failed to detect PrPSc in any ganglia. 2 Vacuolar changes were invariably accompanied by PrPSc deposition, and PrPSc accumulation occurred in the absence of vacuolation, as previously observed in natural disease and experimental models.24,65
Consistent with the identification of natural disease cases through active surveillance of the healthy slaughter population, cases can occur with both PrPSc accumulation and spongiform change evident in the apparent absence of clinical signs.10,55 Vacuolation in the medulla was a constant feature of definite clinical cases, as historically established for the diagnosis of BSE. 59 The invariable occurrence of PrPSc at the obex and the high frequency of detection of vacuolation at the obex in these experimental cases further support the statutory diagnostic approach of targeting this region of the brainstem for disease confirmation.
This study is unarguably the largest of its kind in the natural host, but variation among cases relative to the period postexposure prevents the establishment of a clear sequence of involvement of specific anatomic nuclei in the CNS. It might be speculated that improvements in experimental design with greater numbers of animals and frequency of timed culls over the disease course would provide better insights to the process, but given the resources required to conduct such studies and the diminishing prevalence of BSE, future studies on such a scale are unattainable. The absence of early minimal changes in the 1-g dose group in the study may well be a feature of such deficiencies. Nevertheless, the experimental disease phenotype closely resembles that of field cases, including the variation seen in animals detected through active surveillance, which gives confidence that the progress of PrPSc accumulation in this model is relevant for the formulation of robust surveillance strategies for natural disease. Confidence in the results of the temporal sequence of involvement of tissues in the natural host disease model also underpins use of these data for risk assessment and modulation of public health control measures.2,12,37
Footnotes
Acknowledgements
We thank the staff of the neuropathology and histopathology workgroups, VLA Weybridge, for their consistently excellent technical expertise and support. We are also grateful to Mr A. R. Austin for the clinical assessment of animals in one of the studies.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded by Health Canada (contract 4500095344) utilizing tissues from two experimental studies previously funded by the UK MAFF/Defra/FSA (project codes SE 1901/MO3011 and SE 1736).
