Abstract
Small intestinal T-cell lymphoma and mucinous adenocarcinoma are rarely reported in the pig, with most lymphomas being of B-cell origin and only a single report of mucinous adenocarcinoma. Two aged Vietnamese potbellied pigs had concurrent T-cell lymphoma and mucinous adenocarcinoma of the small intestine. The lymphomas formed polypoid masses that projected into the intestinal lumen, whereas the mucinous adenocarcinomas were mural masses that bulged from the serosal surface. Immunohistochemically, neoplastic cells within the lymphomas were positive for CD3 and negative for CD79a. Mucicarmine stain highlighted the abundant cytoplasmic and extracellular mucin in the adenocarcinomas.
Small intestinal carcinomas are rare in pigs, in part because most domestic pigs, excluding breeding stock, are slaughtered before reaching an age when these neoplasms are likely to occur. The few reported cases of intestinal carcinoma have been in mature or aged pigs.1,8
Mucinous adenocarcinoma (MA) of the small intestine represents a subtype of adenocarcinoma in which greater than 50% of the tumor consists of extracellular mucin that accumulates in “lakes” or epithelium-lined cysts. MAs have been reported in many domestic species including the dog,11,13 cat, 12 ox, 17 nonhuman primate,7,14,16 and ferret. 2 In the pig, there is a single report of 5 cases of jejunal adenocarcinoma that contained mucus-producing glandular to cyst-like structures, but these were not specifically classified as MA. 17 The same study included 1 MA of the cecum. In contrast to its low prevalence in pigs, MA is far more common in human colonic biopsies, where it comprises 10–20% of colorectal tumors.3,10
Overall, lymphoma is the most commonly diagnosed porcine tumor. 4 Lymphomas affecting the small intestine may be of the alimentary or multicentric form, most of which have been shown to be of B-cell origin.6,15 In one report of ileal B-cell lymphomas, neoplastic cells originated in Peyer’s patches and spread only to regional lymph nodes. 15 A single case of multicentric lymphoma of T-cell origin with involvement of the small intestine has been described. 18 Evidence of hereditary transmission of multicentric lymphoma in domestic pigs via an autosomal recessive gene has been documented, although the only changes noted in the small intestine of those cases were increased prominence of ileal Peyer’s patches. 5
Vietnamese potbellied pigs, first brought to the United States from Canada in 1986, are widely kept as pets and have an average life span of 12–18 years, with anecdotal evidence of pigs living as long as 21 years. The relatively short time they have been in North America and the increasing presence of aged pigs have resulted in a number of reports of newly described diseases in this breed. A report of alimentary-associated carcinomas in Vietnamese potbellied pigs included 1 pig with gastric carcinoma and 2 with intestinal carcinomas (jejunum and spiral colon). 9 In the present study, 2 aged Vietnamese potbellied pigs had concurrent MA and alimentary T-cell lymphoma of the small intestine. The coexistence of these 2 malignant neoplasms in the small intestine has not, to our knowledge, been reported in any breed of pig.
Clinical Histories
Pig No. 1 was a 33-kg, 17-year-old, castrated, male, Vietnamese potbellied pig with a 3- to 4-month history of weight loss and periodic vomiting. During the month prior to necropsy, it developed liquid, yellow to brown feces. A spiral colon bypass surgery, performed when the pig was 10 years old, had resulted in multiple abdominal adhesions, but the healed anastomosis site was patent. Abnormal serum biochemistry values at presentation were attributed to vomiting and dehydration. Hematology results were typical of a stress leukogram and likely concurrent inflammation. Gas-filled loops of small intestine were evident in radiographs; a fecal occult blood test was positive. Given the advanced age and a poor response to treatment, the pig was euthanatized and submitted for cosmetic necropsy.
Pig No. 2 was an unrelated 18-year-old, castrated, male, Vietnamese potbellied pig with a history of vomiting, anorexia, melena, and severe weight loss. Treatment with gastrointestinal protectants was unrewarding, and the pig was euthanatized because of its poor condition.
Gross and Histologic Findings
At necropsy, pig No. 1 had numerous nodular masses, 0.5–3.5 cm in diameter, in the wall of the small intestine that protruded from its serosal surface (Fig. 1). The firm mural thickening associated with these masses resulted in circumferential small intestinal strictures. Additional masses in the intestinal mucosa were raised, firm, flat-surfaced to polypoid and extended into the intestinal lumen beneath a coating of fibrin and blood (Fig. 2). Slightly raised, oval, red patches covered with fibrin, were overlying Peyer’s patches. A soft, yellow–tan mass was in the spleen.
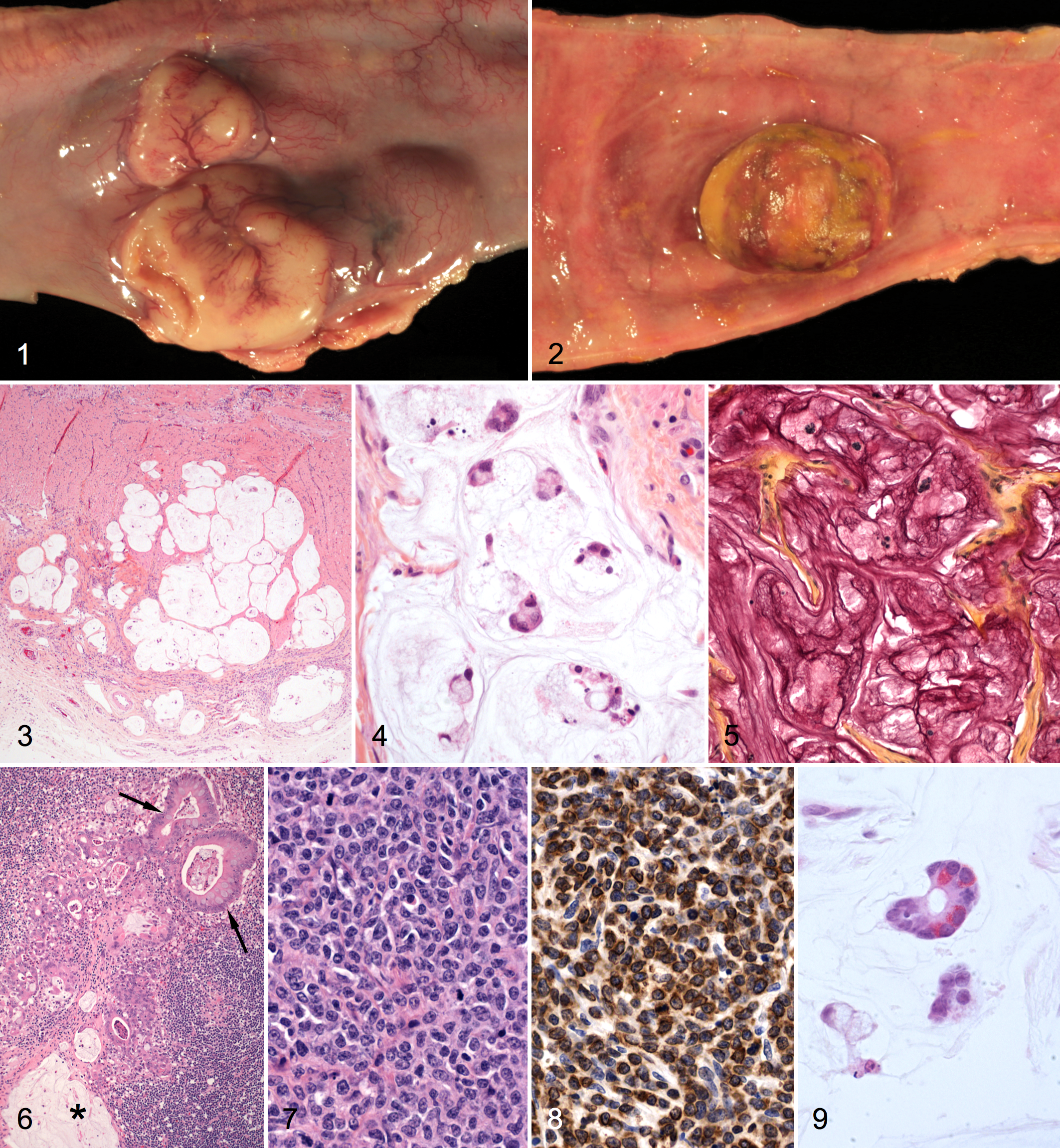
Histologically, pig No. 1 had 2 different neoplasms (adenocarcinoma and lymphoma) in multiple small intestinal sites. The adenocarcinoma consisted of well-circumscribed, multilobulated masses that expanded the tunica muscularis and extended focally into the serosa and submucosa. Each mass was divided into lobules by a fine collagenous stroma (Fig. 3). Individual lobules contained abundant mucin with scattered islands and clusters of epithelial cells. The neoplastic epithelial cells had round to oval, lightly basophilic nuclei with finely stippled chromatin and moderate to abundant amounts of foamy, vacuolated cytoplasm that displaced the nuclei to the periphery in many cells (Fig. 4). Anisocytosis and anisokaryosis were mild; mitotic figures were fewer than 1 per 400× field. Small clusters of tumor cells and abundant mucinous material were found within dilated submucosal and serosal lymphatic vessels. The cytoplasm of neoplastic cells and the extracellular mucinous material reacted strongly with mucicarmine stain (Fig. 5). Similar neoplastic masses were found in a splenic lymph node and the spleen. Within the lymph node, in addition to lakes of mucin, numerous glandular structures were lined by tall columnar epithelial cells with basal nuclei and an apical microvillus brush border and by goblet cells with large cytoplasmic mucin vacuoles (Fig. 6). According to the World Health Organization classification, this tumor was diagnosed as MA.
The lymphoma was confined almost exclusively to the mucosa and formed ulcerated, polypoid masses that projected into the intestinal lumen (Fig. 2). Histologically, the lymphoma consisted of sheets of densely packed round cells with a large round to oval nucleus, finely stippled chromatin, 1 or 2 prominent nucleoli, 3–4 mitotic figures per 400× field, and small to moderate amounts of pale basophilic cytoplasm with distinct cell borders (Fig. 7). Small lymphocytes and rare eosinophils were interspersed among the neoplastic cells, particularly at the base of the neoplasm near the submucosa. The diagnosis was intestinal lymphoma.
Immunohistochemical phenotyping of the lymphoma was performed on formalin-fixed, paraffin-embedded tissue sections with antibodies for CD3 and CD79a. Briefly, antigen retrieval was performed using a Biocare Medical Decloaking Chamber and Reveal antigen-retrieval solution (Biocare, Concord, CA). Primary polyclonal rabbit anti-human CD3 (1:500) or monoclonal mouse anti-human CD79α (1:500) antibodies (Dako, Carpinteria, CA) were applied for 30 minutes. MACH 2 Rabbit HRP Polymer (Biocare) or ImPRESS anti-mouse Ig (peroxidase) (Vector Laboratories, Burlingame, CA), respectively, was applied for 30 minutes. Slides were then developed using diaminobendizine (Dako) as chromogen and were counterstained with hematoxylin (Fisher Scientific, Pittsburgh, PA). Positive and negative canine (lymph node) and porcine (spleen) control tissues were used for both antibodies. Universal Negative Control Serum (Biocare) was substituted for the primary antibody on the negative control tissues. The neoplasms were strongly positive for CD3 (Fig. 8) and negative for CD79a, indicating a T-cell origin.
Upon necropsy of pig No. 2, a 10-cm long × 3-cm thick intramural mass that bulged from the serosal and mucosal surfaces was found in the proximal jejunum. Four additional, 5-cm-long × 3-cm-thick intramural masses were in the mid- and distal jejunum. There were multiple omental adhesions, and several mesenteric lymph nodes were enlarged.
Histologic evaluation of small intestine from pig No. 2 confirmed the presence of both MA and T-cell lymphoma. The MA consisted of pools of mucin with islands and clusters of cuboidal to columnar epithelial cells that included many goblet cells with highly vacuolated cytoplasm. The cells also formed glandular structures lined by 1 or 2 layers of cells. A few epithelial cells, typical of Paneth cells, contained numerous, small, brightly eosinophilic, cytoplasmic granules oriented toward the glandular lumen (Fig. 9). The abundant, faintly amphophilic mucinous material that surrounded the neoplastic cells in the submucosa and tunica muscularis was positive with mucicarmine stain. Less differentiated clusters of neoplastic epithelial cells in desmoplastic stroma were found both in the adjacent mucosal lamina propria and inner aspect of the serosa.
The small intestinal lymphoma in pig No. 2 was separate from the MA and consisted of sheets of neoplastic lymphocytes, with 6–8 mitotic figures per 400× field, that diffusely infiltrated all intestinal layers. As in pig No. 1, a few eosinophils were scattered throughout the neoplasm, and neoplastic cells were immunohistochemically positive for CD3 and negative for CD79a.
Discussion
The coexistence of mucinous adenocarcinoma and T-cell lymphoma in the small intestine of 2 unrelated Vietnamese potbellied pigs is unusual. Because most reported cases of porcine alimentary lymphoma have been of B-cell origin, the presence of T-cell lymphoma combined with mucinous adenocarcinoma in both of the present cases could indicate a predilection for multiple forms of intestinal neoplasia in aged Vietnamese potbellied pigs.
The aging of the North American population of potbellied pigs is likely a major contributing factor to the development of intestinal neoplasia, which is underscored by the advanced age of the 2 pigs in this report. In a previous report of alimentary tumors in 5 Vietnamese potbellied pigs, 2 of which had intestinal carcinomas, ages ranged from 11 to 16 years. 9 Other dietary, environmental, or genetic factors could also explain more prevalent intestinal neoplasia in this breed. With their closer association with people, compared with that of most domestic pigs, prolonged consumption of human food and a tendency toward obesity may play a role. In addition, a higher level of veterinary care and an increase in the number of necropsies of potbellied pigs may increase the detection of intestinal neoplasia.
Last, a genetic predisposition for mutational events in this breed could promote the development of intestinal carcinomas. Human germ-line mutations in the tumor suppressor gene adenomatous polyposis coli (APC) are associated with familial adenomatous polyposis and increased development of colorectal carcinoma later in life. Similarly, mutations in the murine APC gene lead to development of intestinal adenomas throughout the intestinal tract in mice (multiple intestinal neoplasia). Given the high prevalence of colorectal carcinoma in people and the importance of the pig in biomedical studies, efforts to document intestinal carcinoma or multiple intestinal neoplasia in aged potbellied pigs and any relationship with alterations in porcine homologs of tumor suppressor genes are warranted.
Footnotes
Acknowledgements
We thank Dr. James H. Maciulla of Continental Animal Wellness Center, Flagstaff, Arizona, for necropsy and submission of tissues from pig No. 2.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
