Abstract
A 12-year-old Appaloosa gelding was referred to the Texas Veterinary Medical Center with a history of chronic diarrhea and weight loss. At necropsy, numerous oval, craterlike ulcers were observed throughout the small intestine. Histologically, these lesions were composed of a neoplastic proliferation of round cells with intracytoplasmic phosphotungstic acid-hematoxylin-positive granules. The tumor cells stained positively for the CD3 antigen and negatively for a B-cell marker. A diagnosis of large granular lymphoma was based on the morphologic and immunohistochemical characteristics of the neoplasm. The postmortem presentation of this case depicted unusual multifocal, ulcerative lymphomatous lesions throughout the small intestine without involvement of the regional lymph nodes. The histologic and ultrastructural morphology of the neoplastic lymphocytes was similar to that in previously reported cases of abdominal equine large granular lymphomas, but in this case the neoplasm was restricted to the small intestine.
Alimentary tract lymphomas, arising from the lymphocytes of the lamina propria or lymph nodes of the intestinal tract, have been identified in many species and may be present as primary intestinal neoplasia or as part of a more disseminated disease. 2 In a majority of cases, a segmental thickening of the intestine is present, with extension to regional lymph nodes. In horses, these neoplasms are usually associated with a malabsorption syndrome, with protein loss and inconsistent diarrhea. Many of these tumors are of B-cell origin and may be associated with hypergammaglobulinemia. 2
Intestinal neoplasms of round granulated cells include globule leukocyte tumor (GLT), 7 granulated round cell tumor (GRCT), 12 and large granular lymphoma (LGL). 13 15 These neoplasms are characterized by a proliferation of cells with a lymphocyte-like nucleus and eosinophilic cytoplasmic granules. The origin of the neoplastic granular cells is still controversial. 7 8 Recently, several groups have demonstrated that globule leukocytes from different species (cat, chicken, and goat) have characteristics of large granular lymphocytes (perforin-like immunoreactivity and/or lymphocytic surface antigens). 8 10 In humans, immunophenotypic results indicated that the cellular phenotype of granular lymphocytes includes natural killer (NK) lymphocytes and several subsets of T lymphocytes. 16
Here, we present an equine case of a primary intestinal lymphoma that consisted of cells resembling previously defined GLT or GRCT cells. By immunohistochemistry, these cells were identified as lymphocytes, emphasizing the importance of immunophenotyping in the characterization of granular round cell tumors.
A 12-year-old Appaloosa gelding with a clinical history of chronic weight loss and diarrhea was presented at the Texas Veterinary Medical Center. The animal had dependent edema, and laboratory analysis showed hypoproteinemia (2.5 g/dl) with hypoalbuminemia (1.1 g/dl), neutropenia, and mild electrolyte imbalance. A modified transudate was present in the abdominal cavity. The horse was treated with fluids, antibiotics, and a nonsteroidal antiinflammatory drug. Improvement was transient. A change in the treatment was recommended, but the owner elected euthanasia, and a complete necropsy was performed.
Gross examination revealed numerous, oval, craterlike ulcers of various sizes and with slightly raised margins throughout the length of the small intestine (Fig. 1). The mesenteric lymph nodes were slightly enlarged and edematous. The only other gross lesions present were dependent edema and abdominal effusion.
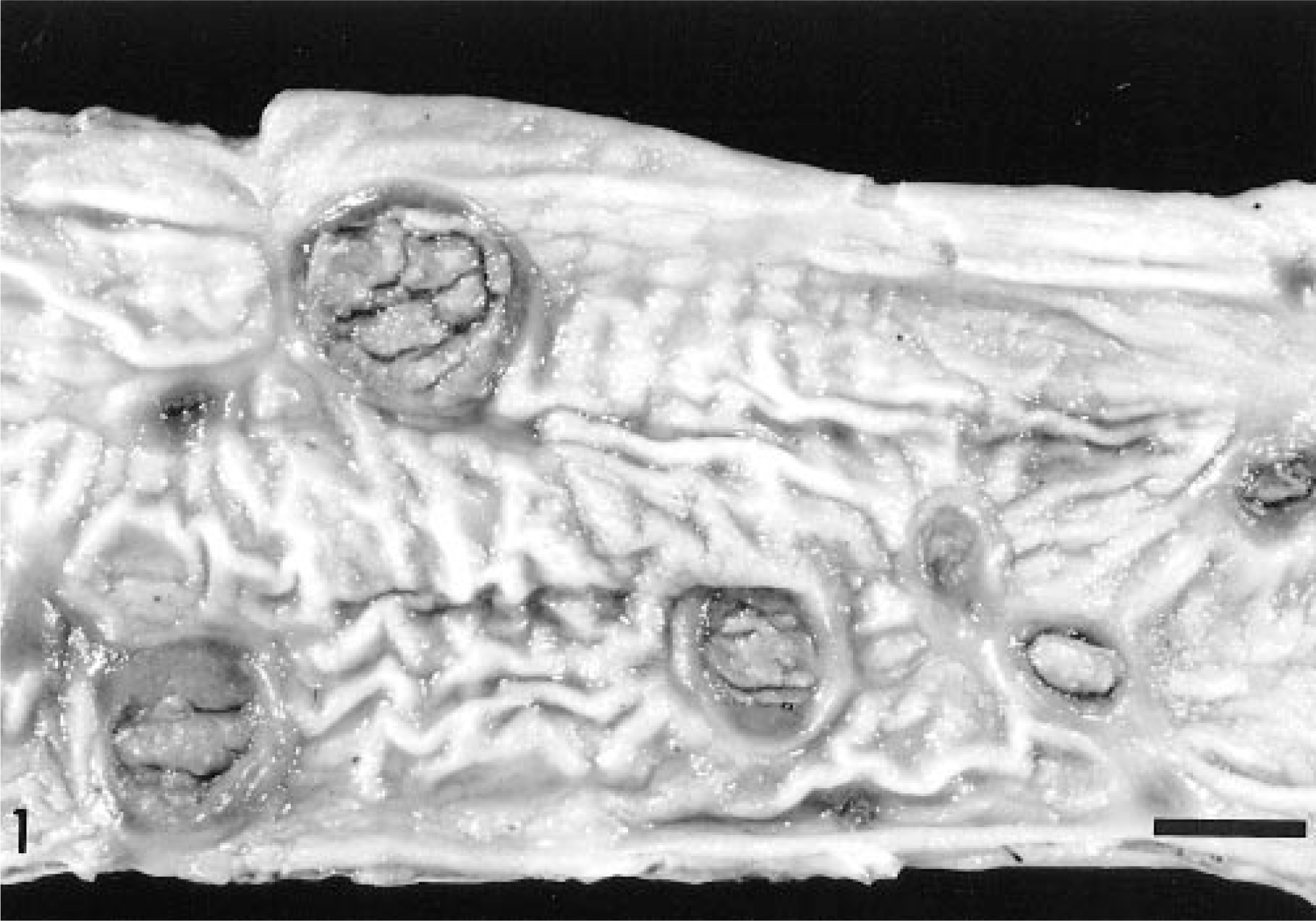
Small intestine; horse. Ulcerative lesions with necrotic center are scattered throughout the mucosa. The neoplastic proliferation is limited to the ulcers. Bar = 0.8 cm.
Selected tissue samples were fixed in 10% neutral buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Giemsa, toluidine blue, and phosphotungstic acid–hematoxylin (PTAH) techniques. Intestinal samples were postfixed in 2.5% glutaraldehyde and osmium tetroxide, dehydrated in a graded series of ethanol, cleared in propylene oxide and embedded in Epon araldite for transmission electron microscopy processing. Thin sections were stained with saturated uranyl acetate and lead citrate and examined with a Zeiss EM10 transmission electron microscope. To characterize the immunophenotype of the neoplastic cells, two different lymphocyte markers were applied: a polyclonal antibody to T lymphocytes (CD3, Dako Corporation, Carpinteria, CA) 9 and a monoclonal antibody to surface glycoprotein BLA.36 as a B-lymphocyte marker (Dako). 9 Also, a monoclonal antibody to proliferating cell nuclear antigen (PCNA, Dako) was used to analyze the proliferative status of the cells. 9
The histopathologic study revealed intestinal lesions characterized by a multifocal, nonencapsulated, noncircumscribed, densely packed, neoplastic proliferation at the lamina propria and extending to the submucosa. The neoplasm was composed of slightly pleomorphic, 12–20-µm, round to oval mononuclear cells with round to irregularly shaped nuclei, coarsely stippled chromatin, and an inconspicuous nucleolus. The cytoplasm was slightly basophilic and contained 2–12 eosinophilic granules (diameter, 0.5–1.2 µm) usually at or close to nuclear infolds (Fig. 2). There was a prominent fibrovascular stroma. The mitotic index was 1–3 mitoses/high-power field (400×). The granules did not stain metachromatic with toluidine blue or Giemsa. PAS staining varied from negative to intensely positive. In all cases, the granules were best visualized with PTAH, staining as deeply dark blue granules (Fig. 3).
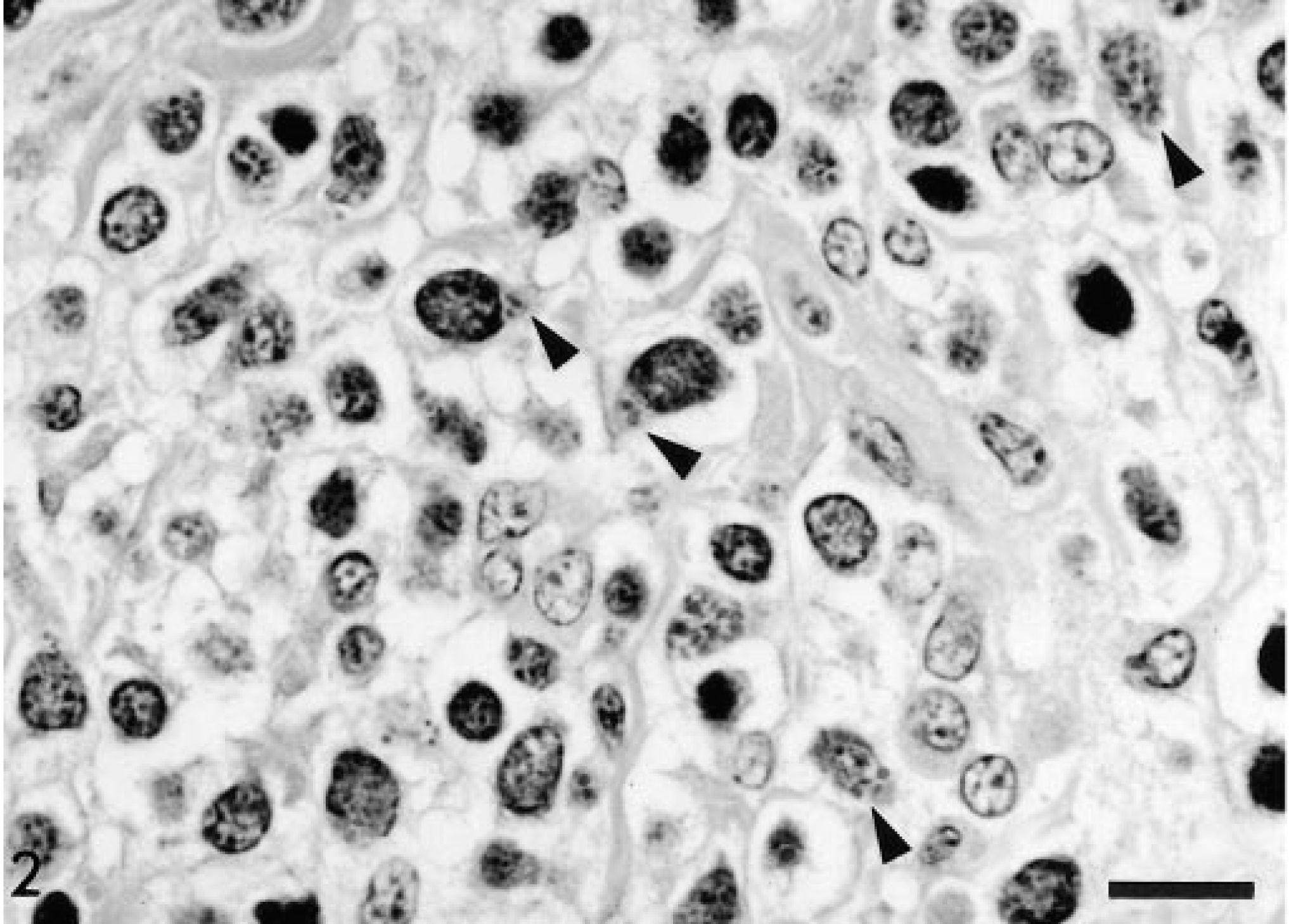
Intestinal mucosa; horse. Neoplastic lymphocytes have intracytoplasmic granules weakly stained by HE (arrowheads). Bar = 22 µm.
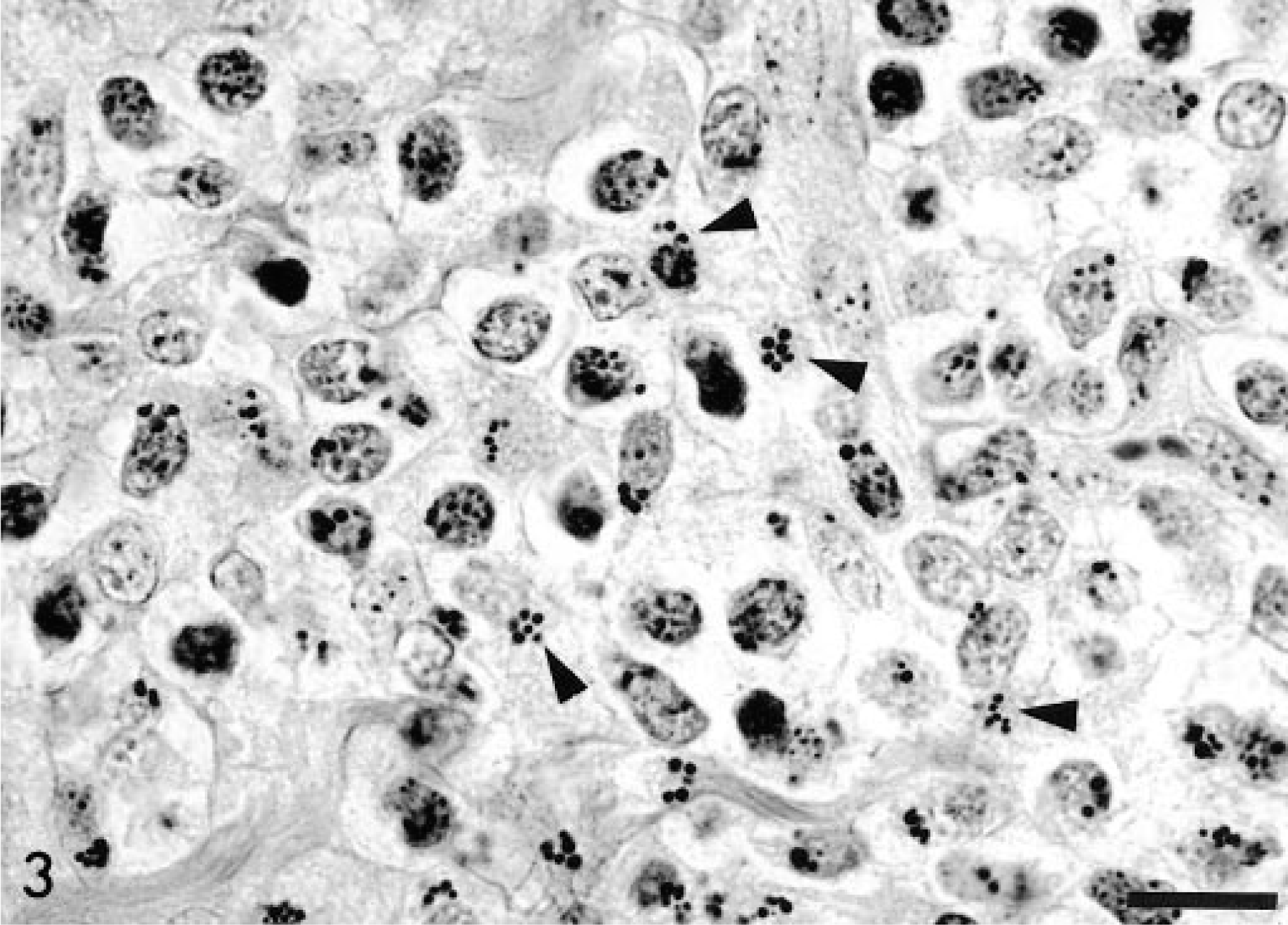
Intestinal mucosa; horse. Cytoplasmic granules in neoplastic cells stain intensely with PTAH (arrowheads). Bar = 22 µm.
Immunohistochemically, the majority of the neoplastic cells labeled positively with the anti-CD3 antibody (pan T cell marker) (Fig. 4). Very rare cells stained positively with the anti-BLA.36 antibody (B cell marker). The proliferative rate of the tumor cells, as characterized by PCNA immunostaining, was approximately 15–20%, which is in the low range as compared with other horse lymphomas. 9
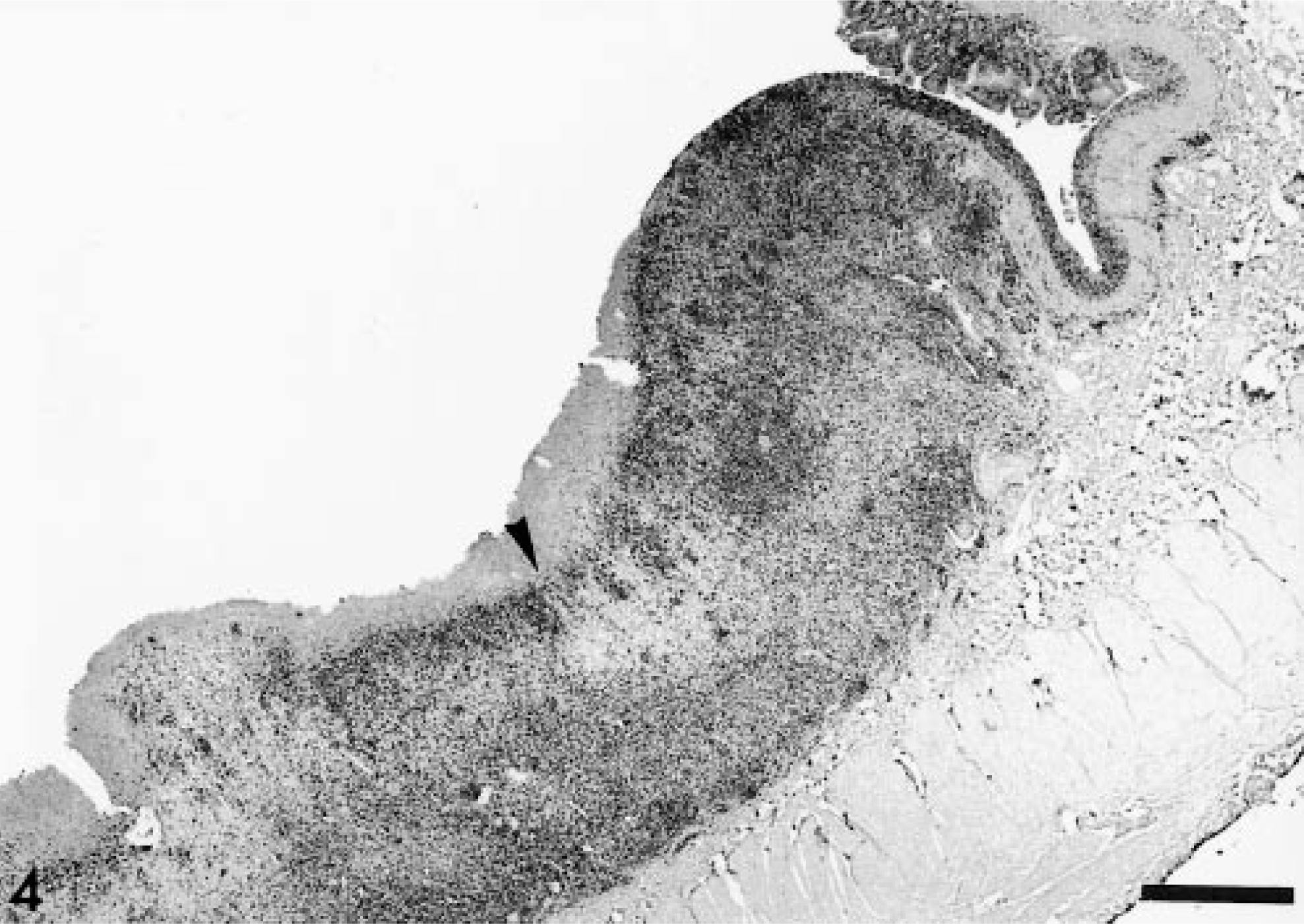
Intestinal mucosa; horse. The ulcerative lesions are deeply infiltrated with neoplastic cells, which express the CD3 antigen (dark area). The arrowhead points to superficial necrosis. Avidin–biotin–peroxidase immunohistochemistry. Bar = 1.7 mm.
The neoplastic cells were concentrated at the grossly identified lesions, with some cells infiltrating to the adjacent intestinal mucosa and submucosa (Fig. 4). Neoplastic cells were not found in lymph nodes or in other organs, indicating a primary intestinal origin for this tumor.
Ultrastructurally, the neoplastic cells were round to elongated, moderately pleomorphic, and organelle poor. The nuclei were oval and often indented, with peripherally clumped heterochromatin and one or two small nucleoli. The cytoplasm contained variable numbers of granules that were usually clustered in a perinuclear location. Granules were round to oval and 0.5–1.2 µm in size and contained amorphous, electron-dense material and were limited by a single unit membrane (Figs. 5, 6), similar to others previously described. 5 7 12 13
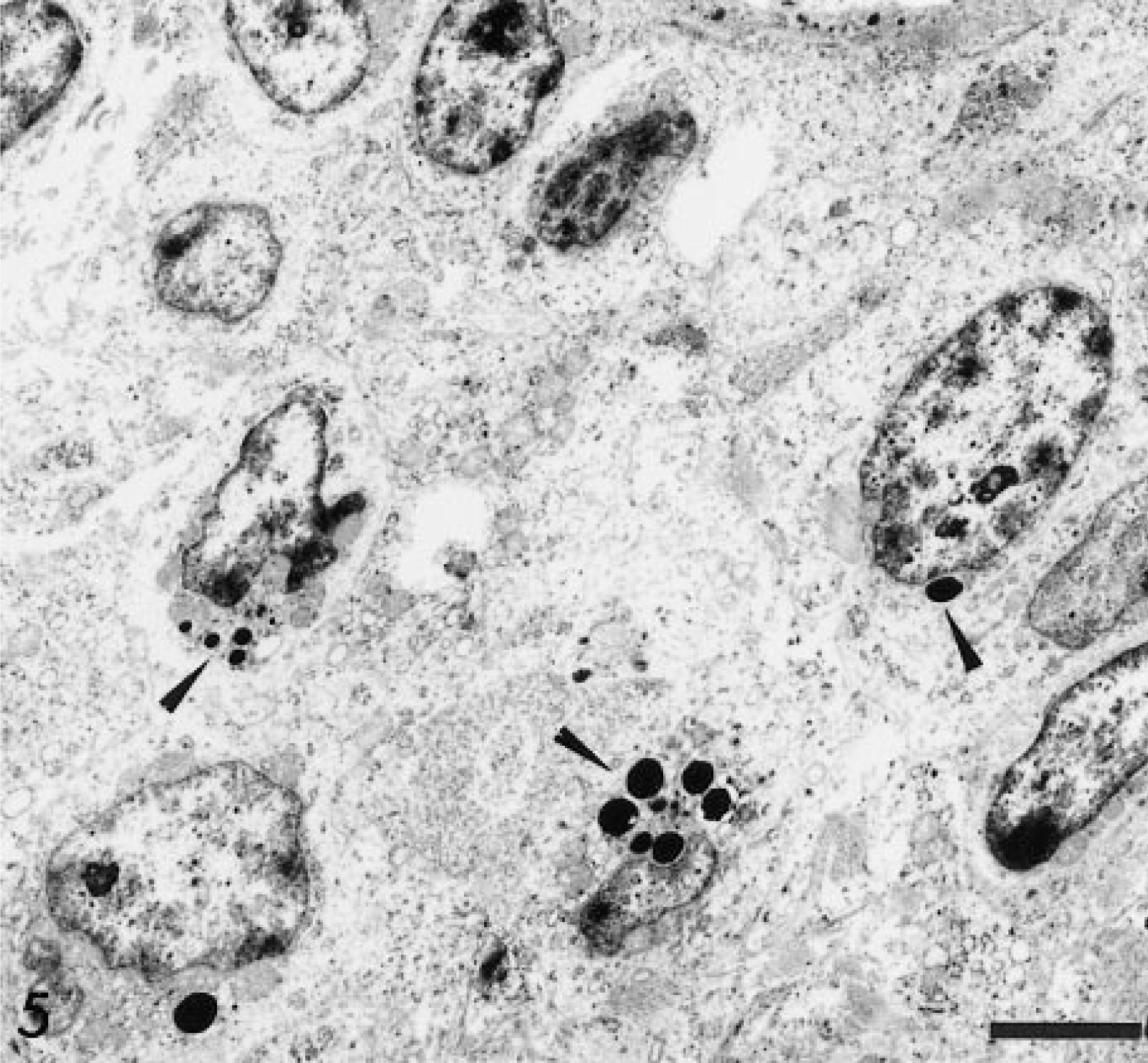
Transmission electron micrograph. Intestine; horse. The granules are round, electron dense (arrowheads), and generally located adjacent to the nucleus or in areas of nuclear indentations. Uranyl acetate and lead citrate. Bar = 4 µm.
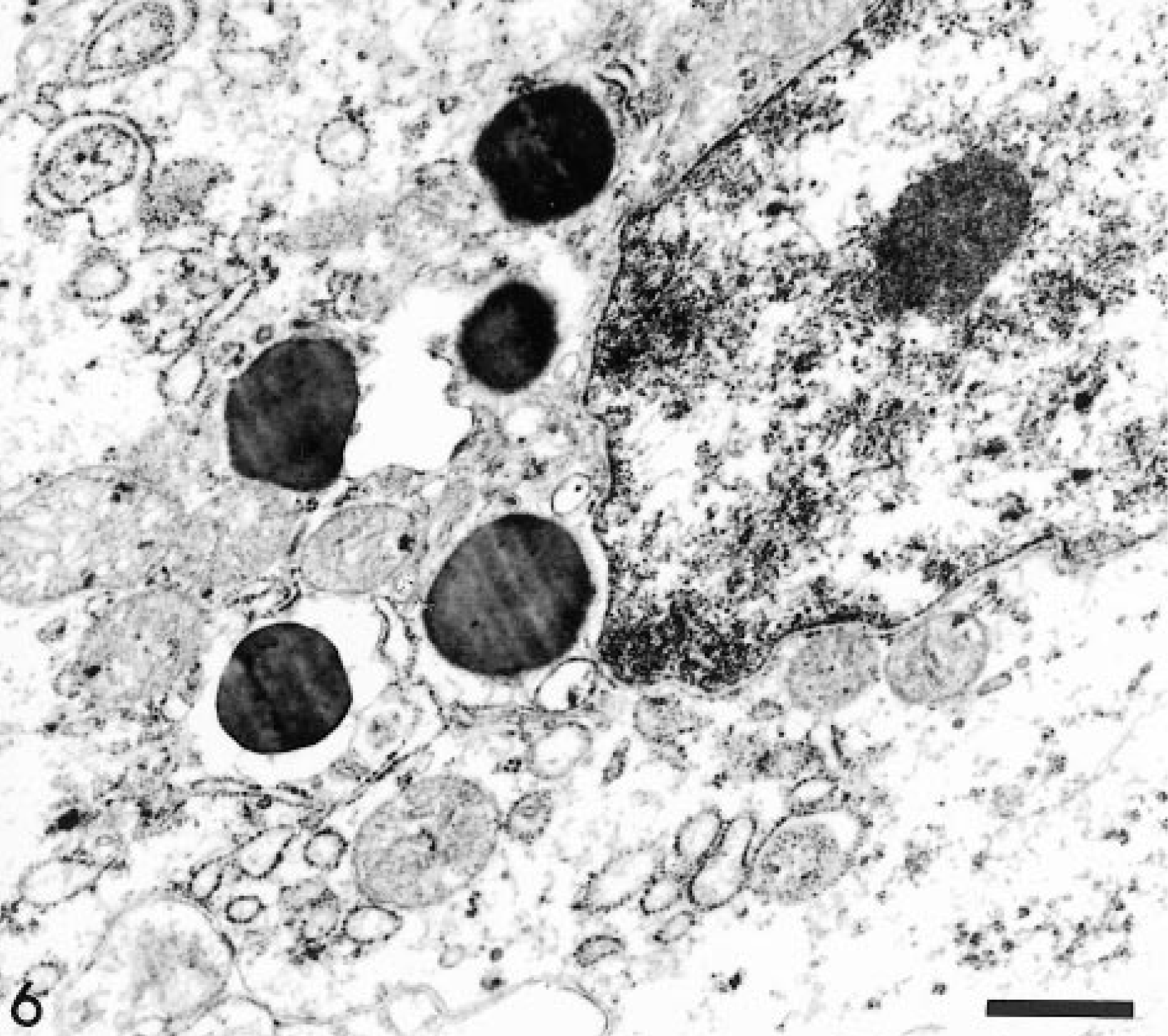
Transmission electron micrograph. Intestine; horse. At higher magnification the granules appear membrane-bound. The nucleus has a central nucleolus (top right corner). Uranyl acetate and lead citrate. Bar = 0.5 µm.
According to the morphology, the presence of intracytoplasmic granules and the immunocharacterization of the neoplastic cells, a diagnosis of primary intestinal lymphoma composed of large granular lymphocytes was made.
Large granular lymphocytes are normally found in the intestinal mucosa (intraepithelial lymphocytes) of many species 8 and are classified into two categories: cells of NK origin and cells of T-lymphocyte origin, i.e. cytotoxic T lymphocytes (CTLs) and γδ-T cells. 8 10 14 T lymphocytes express CD3 antigen in their membranes, and a subgroup of NK cells also expresses the CD3∊ chain in humans and mice, but more intensely after activation. 11 16 To differentiate between these cell populations, the use of different surface markers is required. Contrary to CTLs, NK cells do not express the T-cell receptor (TCR) proteins and have a germline configuration of TCR genes. 4 11 16 They are usually CD5 negative (or weakly positive) 14 and CD56 positive in humans, 4 11 16 but the presence of the CD56 antigen is variable in animal species. 3 The murine NK1.1, Ly-49, and 2B4 molecules, the human killer inhibitory receptors, and CD94 and NKG2 are selectively expressed by NK cells, 16 but NK cell marker use in other species still remains to be studied. The CTLs are primarily CD8-positive cells, and specific markers for the variable chains of the TCR can be used to identify them. More specifically, peptide specific major histocompatability class (MHC)-I tetramers have been used to quantify and differentiate antigen-specific CTLs. 1 The γδ-T cells are cytotoxic cells not restricted by classical MHC molecules, are CD8α positive, and have been studied by the use of the antibody TCR1 N-24. 10 The CTLs and γδ-T cells are CD5 positive. 4 16 Only the anti-CD3 antibody (cytoplasmic chain) was used for the present study. This antibody does not allow precise definition of the origin of the cells but did provide enough information to classify this proliferative lesion as a true large granular lymphocyte lymphoma. The use of the anti-CD5 antibody 4 and other antibodies, such as an specific NK marker (anti-FAM), 6 would be useful in further differentiating the origin of granular cell tumors.
In previous studies, histochemical characteristics of the intracytoplasmic granules have been used to differentiate LGLs from other granulated cell neoplasms. 12 15 The cytoplasmic granules in the present case and in two previously reported LGLs in horses 5 13 stained red-orange with HE, did not stain with toluidine blue and Giemsa, and stained with PAS and PTAH. Other reports have indicated that granules of LGL in cats and rats are negative for PTAH, 12 15 and in one of the reports, the term GRCT was proposed for neoplasms composed of round, mononuclear cells containing distinct blue, PTAH-stained cytoplasmic granules. 12
GLT is a rare neoplasm reported primarily in cats in the terminal small intestine. 7 The histochemical characteristics of cytoplasmic granules of globule leukocytes are similar in all species 7 and similar to those described in equine LGL. 5 13 Although the origin of globule leukocytes has been a matter of great discussion, some authors have suggested that globule leukocytes in cats are intraepithelial large granular lymphocytes because they stain positively for a pore-forming protein (perforin) specific to CTLs. 8 The present and previous results 3 8 10 indicate that histochemical characteristics are not reliable for classifying granular cell neoplasms and that immunohistochemistry with selected antibodies is a more reliable technique.
LGLs with phenotypes similar to that presented here have been previously described in horses. 5 13 The distribution of lesions in those horses was more disseminated throughout different organs, including liver, spleen, lymph nodes, intestine, kidney, and lungs, indicating a more aggressive behavior. 5 13 In the present case, infiltrative neoplastic cells were found only in the intestinal lesions in the lamina propria, which indicates behavioral differences in intestinal LGLs in horses. The low PCNA index observed may be an indicator of the low replicative capacity the tumor described here, however, similar determinations were not reported for other cases. Different phenotypic characteristics of the lymphocytes (surface antigens, enzymes) may account for a different infiltrative capacity, but further immunohistochemical studies in similar cases will be necessary to explore this possibility.
This horse was diagnosed with a primary intestinal, multifocal, CD3 positive LGL with PTAH-positive granules. The tumor appeared grossly similar to some lesions associated with chronic intestinal infections of horses, such as salmonellosis, mycobacteriosis, or rhodococcosis. Microscopically, the differential diagnosis included other granulated round cell tumors. The histochemical characteristics of intracytoplasmic granules in LGL vary among species and do not define the origin of granulated round cells neoplasms. The use of an expanded range of lymphoid surface markers is important in the differentiation of round cell neoplasms and may allow more specific therapy.
