Abstract
California horses incur a bone fragility syndrome manifested by pathologic fractures. This study investigated gross, radiographic, and histologic features of the disorder as well as relationships with silicosis and levels of heavy metals and trace minerals through a postmortem study of 9 affected and 3 unaffected horses. Bones and soft tissues were evaluated grossly and histologically. Bones, lymph nodes, and lung tissue were evaluated radiographically. Tissues were evaluated for silicon levels, intracytoplasmic crystals, heavy metals, and trace minerals. All 9 affected horses had osteoporosis and clinical or subclinical pulmonary disease due to silicosis (8/9) or pneumoconiosis (1/9). All affected horses had radiographic findings consistent with osteopenia and histologic evidence of osteoporosis characterized by osteopenia, numerous resorption cavities, cement lines, and a mosaic lamellar pattern indicative of multiple remodeling events. Silicosis was characterized by widespread pulmonary granuloma formation with fibrosis; variable tracheobronchiolar and mediastinal granulomatous lymphadenitis; intracellular crystals within lung and lymph node macrophages; and pronounced lymph node fibrosis, focal necrosis, and dystrophic calcification. Crystals in lung (6/9) and lymph node (8/9) tissues were identified as cytotoxic silica dioxide polymorphs. Lung and liver tissue from affected horses had elevated levels of elemental silicon. Osteoporosis was highly correlated (r = 0.8, P < .01) with silicosis. No abnormalities in heavy metal or trace minerals were detected. This evaluation indicated that horses with bone fragility disorder have systemic osteoporosis associated with fibrosing pulmonary silicosis. The etiopathogenesis of the bone fragility syndrome is unknown; however, this study provides circumstantial evidence for a silicate associated osteoporosis.
Keywords
Clinical features of a bone fragility syndrome that affects horses have recently been described. 2,17 Affected horses have chronic lameness of vague origin; skeletal deformities that include lateral bowing of 1 or both scapulae, lateral bowing of the rib cage, and lordosis; and decreased range of motion of the cervical vertebrae. Some affected horses also have loss of appetite and severe weight loss. The disorder appears to affect multiple foci in multiple bones of the axial skeleton and proximal portion of the appendicular skeleton. Known antemortem radiographic abnormalities, limited to bones that can be assessed radiographically, include cervical vertebral body lucencies and periarticular facet exostoses, as well as segmental rib thickening consistent with bone remodeling. 2,17 Routine complete blood counts, serum chemistry, and urinalysis results have been unremarkable. 2 Bone scintigraphy has been the most useful diagnostic tool and reveals multiple areas of increased radiopharmaceutical uptake primarily of the scapulae, ribs, vertebrae, and pelvis. 2 All reported animals either died due to catastrophic bone fracture or were humanely euthanatized. 2,17
The cause of this bone fragility syndrome is unknown; however, many affected horses also have clinical signs and history consistent with pulmonary silicosis, 17 and thus this disease has been suggested to be a silicate-associated osteoporosis (SAO). 16 Pulmonary silicosis, defined here as silicate pneumoconiosis with accompanying pulmonary fibrosis, can be caused by inhalation of cytotoxic silica dioxide (SiO2) crystal polymorphs including quartz, cristobalite, and tridymite. These cytotoxic SiO2 polymorphs can be found in certain soil types such as Miocene Monterey Shale and some volcanic geologic formations. Historically, equine pulmonary silicosis has been attributed to inhalation of cristobalite, 45 and many horses affected with SAO have lived in geographic areas with soil containing cristobalite. Consequently, cristobalite is a candidate for consideration as either an etiologic agent directly responsible for the bone disorder or one of possibly several insults that indirectly initiate a sequence of biological events resulting in the bone disease. Direct evidence for a cause and effect association with silicates is lacking.
The goal of this study was to gain insight into the etiopathogenesis of equine bone fragility syndrome by determining the distribution and nature of lesions found in affected horses. The objectives were to (1) characterize pathologic, histopathologic, and radiographic postmortem findings and (2) assess relationships among bone abnormalities, pulmonary silicosis, and levels of heavy metals and trace minerals.
Methods and Materials
Animals
Nine horses diagnosed with the equine bone fragility syndrome (Nos. 1–9) and 3 unaffected (Nos. 11 and 12) horses were studied. All affected horses were presented to veterinarians because of lameness with (Nos. 1, 2, 4, 6, 7, and 9) or without (Nos. 3, 5, and 8) skeletal deformities. Diagnosis was confirmed initially in 7 affected horses (Nos. 2–6, 8, 9) by bone scintigraphy and in 2 affected horses (presented dead) by necropsy. Three clinically unaffected horses (Nos. 10–12) were donated to the University of California, Davis, Center for Equine Health. All 3 unaffected horses had navicular syndrome and were euthanatized for reasons unrelated to the current study. These horses were considered controls since navicular syndrome is not a recognized hallmark of the bone fragility syndrome. All horses presented alive were euthanatized by intravenous injection of 200 mg of xylazine and 78,000 mg of pentobarbital sodium (as Euthasol).
Clinicopathology
Eight of the 9 (Nos. 1–6, 8, 9) affected horses had blood collected via the external jugular vein before euthanasia. Complete blood count and serum chemistry analyses were performed on 8 horses including, for some horses, total and ionized calcium (Nos. 2–6, 9), phosphate (Nos. 1–6, 8, 9), alkaline phosphatase (Nos.1, 2, 4–6, 8, 9), parathyroid hormone (PTH) (Nos. 2–6, 9), and vitamin D (Nos. 2–4, 6, 9) levels.
Necropsy Examination and Soft Tissue Sampling
Necropsies performed at the California Animal Health and Food Safety Laboratory, in Davis, California, included examination of thoracic and abdominal viscera, brain, and spinal cord and an extensive gross examination of the skeletal system. Tissue samples were immersion fixed in 10% neutral buffered formalin for routine histologic examination. Sampled organs included liver, kidney, heart, lung (4–14 sections), spleen, gastrointestinal tract (multiple), lymph nodes (tracheobronchial, cranial and caudal mediastinal, mesenteric, cervical, and, where visible, renal, gastrohepatic, and cranial lumbar), pancreas, adrenal gland, thyroid, parathyroid (when found), pituitary gland, gonad (if present), urinary bladder, skeletal muscle (diaphragm, quadriceps femoris, triceps), peripheral nerve (sciatic and 1 nerve from the brachial plexus), brain, spinal cord, and bone (primarily rib or vertebral body). Adrenal and pituitary glands were weighed.
Additional tissue samples were collected for toxicology (liver, kidney, lung, thoracic lymph node, eye, and bone), serology (whole blood), virology (lung), and bacteriology (lung, liver, lymph node, feces) tests. Tests performed included fluorescent antibody on lung for Equine Herpesvirus I; polymerase chain reaction (PCR) on lung for Equine Influenza; acid-fast stains on impression smears of tracheobronchial and/or mediastinal lymph node or lung; modified Ziehl-Neelson acid-fast stains and Gomori methenamine silver stain on tissue sections of thoracic lymph node and lung; aerobic culture of lung, bronchiolar lymph node, and liver; Salmonella culture and fecal flotation on feces; and serology on postmortem serum for Equine Influenza, Equine Viral Arteritis, Equine Herpesvirus I, and Corynebacterium pseudotuberculosis.
Submandibular, thoracic, mesenteric, and cranial abdominal lymph nodes (renal, cranial lumbar) that were large enough for identification were isolated and grossly examined, and the largest from each anatomical location was measured and weighed. Hepatic, gastric, and splenic lymph nodes were grossly abnormal in 2 horses and so were removed and assessed. Additional tissue samples of lung, liver, lymph node, and kidney were stored at –20°C. In addition, frozen thoracic lymph node tissues from any horse (Nos. 2, 3, 9) with suspect acid-fast bacteria detected on histologic examination of acid-fast stains of thoracic lymph nodes were tested by PCR 15 for Mycobacteria (Washington Animal Disease Diagnostic Laboratory, Pullman, WA).
Bone Sampling
The entire skeleton was harvested from all horses in the study. All bones were dissected free of soft tissues and examined grossly with a focused examination performed on the scapula, ribs, vertebrae, pelvis, humerus, femur, and all appendicular joint surfaces. The vertebral column was isolated and cut in a parasagittal plane through the spinal canal to examine the spinal cord, canal, and vertebrae. Following gross dissection of soft tissues from bones, the remaining soft tissues were removed by lye dissolution from the entire skeleton of 1 clinically, moderately affected horse (No. 3). Bone specimens (transverse sections, 3- to 5-mm thick) from cervical (C4) and lumbar (L4) vertebrae, right 7th rib, right iliac wing, right scapula, right humerus, right third metacarpal bone, and right third carpal bone were collected for radiographic and/or histologic evaluation from all horses. Fractures were categorized as either acute (unhealed) or chronic (healed), depending on the absence or presence of visually evident bone fracture calluses. Intact bones were stored at –20°C.
Tissue Processing for Histology
Formalin-fixed soft tissues were trimmed, routinely processed, paraffin embedded, cut (3- to 5-μm thick sections), mounted on glass slides, and stained (hematoxylin eosin) for histologic examination.
Rib specimens were chosen for histologic evaluation because ribs are commonly affected on bone scintigraphy and contain compact and trabecular bone tissues. Rib specimens were decalcified by suspension in a solution containing 170 ml of 88% formic acid and 34 g of sodium formate diluted to 1 liter with deionized water. Decalcification was monitored using radiography in a cabinet X-ray unit (Lincolnshire, IL, Faxitron cabinet radiography unit; model 43805) on high-detail oncology film (Rochester, NY, Kodak Portal Pack for Localization Imaging [PPL2]); (30 kVp, 1.5 mA for 1 minute). Decalcified specimens were rinsed in running water for 2 hours, placed in 70% ethanol for at least 2 days, and then infiltrated and embedded with paraffin. Five-micrometer-thick sections were cut, deparaffinized, and stained with hematoxylin and eosin.
Histological Scoring
After review of bone, lung, and thoracic lymph node histopathologic findings, the severity of the salient patterns of histopathologic changes was graded using a subjective ordinal scale ranging from 0 to 3. Histopathologic evaluation of the bone was performed by A. Arens and histopathologic evaluation of the remaining soft tissues was performed by B. Barr.
Radiographic Survey
The entire skeleton from 1 moderately affected horse (No. 3) that was processed with lye dissolution, all of the 3- to 5-mm-thick bone specimens, the lymph nodes, a 4-cm-thick transverse section of the cranial third of the right caudal lung lobe, and a slab of liver taken across 1 major liver lobe from each horse were radiographed and assessed for lesions, density, and mineralization when appropriate. Intact bones, lymph nodes, lung specimens, and liver specimens were radiographed with a Bennett HFQ300 (Chicago, IL, Bennett) X-ray unit or with a digital radiography system (Carlsbad, CA, Sound-Eklin digital radiography unit; model EDR3) using a range of bone, lymph node, lung, and liver-specific techniques (60–94 kVp, 0.20–0.90 mA; 40 kVp, 1.3 mA; 40 kVp, 1.0 mA; 40 kVp, 1.0 mA; respectively). The 3- to 5-mm-thick bone specimens were radiographed (30 kVp, 1.5 mA for 1 minute) in a cabinet X-ray unit (Lincolnshire, IL, Faxitron cabinet radiography unit; model 43805) on high-detail oncology film (PPL2).
Silicon Assay
A silicon assay was performed on lung, tracheobronchial lymph node, liver, and bone tissue samples. One gram (or less if necessary) of tissue was digested using 0.2 ml of concentrated hydrofluoric acid and 3 ml of concentrated nitric acid in Teflon bombs using a MARS-Xpress microwave digestion apparatus (Matthews, NC, CEM Corp.). Silicon overspikes of lung (100 ppm Si) and liver (50 ppm Si) were included in the process for evaluation of recovery. Spiking levels varied based on tissue type and expected concentrations of silicon (no tissue standard reference materials are available for silicon). After digestion, the samples were diluted to an appropriate final volume (depending on expected silicon concentration) and analyzed on the Agilent 7500 ICP-MS equipped with a PFTE (hydrofluoric acid resistant) sample introduction system (Wilmington, DE, Agilent Technologies, Inc.).
The assays were semiquantitative and post hoc categories were created based on discrete clustering of results such that the lung silicon levels were considered negligible at ≤50 ppm, low at 51–150 ppm, medium at 151–350 ppm, and high at >350 ppm; the tracheobronchial lymph node results were considered negligible at ≤5 ppm, low at 6–100 ppm, medium at 101–200 ppm, and high at > 200 ppm; the liver results were considered negligible at ≤10 ppm, low at 11–100 ppm, medium at 101–200 ppm, and high >200 ppm; and the bone results were considered negligible at ≤5 ppm, low at 6–100 ppm, medium at 101–200 ppm, and high at >200 ppm.
Metal and Trace Mineral Assays
A heavy metal and trace mineral analysis was performed on tissue samples of lung, liver, tracheobronchial lymph node, scapula, and vitreous humor. Standard reference materials (TORT-2 [lobster hepatopancreas]) and DOLT-3 ([dogfish liver]; Ottawa, Canada, National Research Council of Canada) were used for additional control samples. One gram (or less if necessary) of tissue was digested in 3 ml of concentrated nitric acid at 190°C using a programmable block digestion system (Saxonburg, PA.AIM600 Block Digestion System). The digestate was mixed thoroughly with 2 ml of concentrated hydrochloric acid and then diluted to 10 ml with reagent grade deionized water (ASTM type 1). The levels of 16 elements (Pb, Mn, Fe, Hg, As, Mo, Zn, Cu, Cd, Ba, Be, Co, Cr, Ni, V, Tl) were analyzed using a Fisons AccurisICP-OES (inductively coupled plasma–optical emission spectrophotometry; Valencia, CA, Accuris, Fisons Instruments, Inc.).
Crystal Identification
Crystal Collection
Lung, lymph node, and bone tissue samples were processed for crystal presence and identification. Approximately 3 g (when available) of sample was digested in 9 mL of trace element grade nitric acid at 190°C for approximately 2 hours in a 50-ml Pyrex tube. The sample was centrifuged at 2000 rpm for 5 minutes and the supernatant acid was removed by pipette leaving any undissolved crystalline matrix in the bottom of the tube. Ten milliliters of water was added to the tube, it was mixed and recentrifuged, and the aqueous supernatant was removed. This washing process was repeated 4 times total. The remaining undissolved crystalline matrix was washed 3 times using acetone in the same manner as it was with the water. Last, the undissolved crystalline matrix was transferred to a small 2-mL plastic vial using three 0.5-mL volumes of acetone. The small vials were then centrifuged for 3 minutes at 3500 rpm, and the acetone supernatant was removed carefully using a pipette. Each vial containing the undissolved crystalline matrix was then allowed to dry overnight at room temperature. The undissolved crystalline matrix was then ready for further analysis.
Soil samples were collected from 9 locations: 3 endemic farms (Monterey, Napa, and Sonoma counties in California), a known Miocene Monterey Shale outcropping in an endemic region (Monterey County, California), and 5 locations with no known affected horses (Monterey, Yolo, Sacramento, and San Bernadino counties in California and Washoe County in Nevada).
Crystal and Element Analyses
Element analyses were performed on soil samples and on crystalline material remaining after acid treatment of tissue samples using a Phillips XL20 Tungsten filament scanning electron microscope (Eindhoven, Netherlands, Philips Electron Optics) fitted with an Oxford 5175 electron dispersion spectroscopy (EDS) detector system. A small quantity of soil or crystalline powder positioned on a conducting carbon pad was analyzed directly using the EDS detector. A few miniscule samples were transferred to the carbon pad by suspension in a drop of 95% ethyl alcohol which evaporated before imaging. The EDS system was calibrated using a copper standard before imaging samples.
Subsequently, the structure of crystals in remaining samples of tissue and soil was analyzed using X-ray diffraction (XRD; Rigaku Rotaflex; Tokyo, Japan, Rigaku Denki Co., Ltd.). Powdered tissue samples were spread on a textured amorphous slide and analyzed at 40 kV, 120 mA at 10° to 60° diffraction angles, in 0.2° increments at 1.5 seconds per increment. Hard soil and rock samples were broken up into fragments with a 4-cm2 surface, mounted in Struers Multifast phenolic hot mounting resin, and polished to a flat surface using FEPA P #2400 (US grit 800) silicon carbide paper. These mounts were then placed directly in the X-ray diffraction instrument. Loose soil samples were hand ground to a fine consistency and packed onto a powder sample holder for X-ray diffraction. Soil samples were analyzed at 40 kV and 100 mA at 20°–65° diffraction angles in 0.2° increments at 1.5 seconds per increment.
Correlations
Spearman correlation coefficients (r) were calculated to assess the relationships between measured variables and syndrome state (affected, unaffected); among the presence or absence of histological grades, gross pathology, radiographic opacity, and cytotoxic crystals; and among horse age and silicon (Si) levels. Because of the small sample size, r ≥ 0.6 and P ≤ .10 were considered to indicate a significant correlation.
Results
Study Population
The 9 affected horses (5 castrated male, 4 female) were 4 to 22 years old (9 years median age). The 3 unaffected horses (2 castrated male, 1 female) were 8, 19, and 20 years old. Affected horses came from 3 distinct geographic areas in California: the Monterey and Salinas valleys (Monterey County) (Nos. 1, 4, 7 and 9); the Marin, Sonoma, and Napa Counties (Nos. 2, 3, 5, and 6); and Yolo County (No. 8). The horse from Yolo County had also lived in the Sierra Mountain foothills, with possible exposure to mine tailings, and in unknown locations. The 3 unaffected horses were most recently from Yolo County; 2 for at least 4 years (Nos. 11 and 12) and 1 for at least 4 months (No. 10) with unknown previous location histories.
All horses had antemortem history, clinical signs, deformations, or abnormal diagnostic test results associated with problems of the skeleton. All 7 affected horses that presented antemortem (Nos. 2–6, 8, 9) and all 3 unaffected horses were lame at presentation; the 2 affected horses that presented postmortem had histories of progressively worsening stiffness (No. 1) and chronic lameness (No. 7). One affected horse (No. 3) had a noticeably enlarged left mandible. Seven affected horses (Nos. 2–5, 7–9) had clinically visible scapular bowing; 5 affected horses (Nos. 2–4, 6, and 9) had cervical vertebral facet remodeling and/or lytic lesions on radiographs; and 4 (Nos. 2, 5, 6, 9) of the 6 (Nos. 2, 5–9) affected horses that had scapular ultrasound examination had irregular margination and thickening of the scapular spine. All 7 affected horses that had bone scintigraphy had increased radiopharmaceutical uptake predominantly in the scapula and ribs but also commonly in the cervical vertebrae and pelvis.
Seven of the 9 affected horses had antemortem clinical signs or abnormal diagnostic test results indicative of pulmonary disease. Four affected horses (Nos. 1, 4, 7, 9) had flared nostrils and elevated respiration rate. Five (Nos. 1, 2, 4, 6, 9) of the 7 (Nos. 1–6, 9) affected horses that had bronchoalveolar lavage had cytology consistent with pulmonary silicosis; the sample from 1 horse (No. 3) was acellular and undiagnostic. Five (Nos. 1–4, 9) had radiographic evidence of chronic airway disease.
Clinicopathological, Metal, and Trace Mineral Findings
Complete blood counts were normal for all horses. Some affected horses had phosphate, vitamin D, and PTH abnormalities. Six (Nos. 2–4, 6, 8, 9) of 8 affected horses had slightly elevated serum phosphate levels. Three (Nos. 2, 3, 6) of 5 affected horses had low serum vitamin D levels. Six of 6 affected horses (Nos. 2–6, 9) had elevated PTH levels. Total serum calcium was normal in all affected horses except No. 3, where it was low; ionized calcium was normal in all affected horses measured except No. 5, where it was slightly elevated. Serum alkaline phosphatase was normal for 7 affected horses.
Heavy metal levels in lung, tracheobronchiolar lymph node, liver, kidney, and bone and trace mineral levels in bone and vitreous humor of the eye were normal for all horses.
All horses were negative for the infectious diseases tested. No significant findings noted on serology for Equine Herpesvirus 1, Equine Influenza, Equine Viral Arteritis, and C. pseudotuberculosis. All Salmonella cultures on feces were negative. Acid-fast stains of impression smears of lung and/or lymph node sections were negative. Acid-fast stains of tissue sections of lung and lymph node were problematic because of nonspecific staining of random crystalline particulates within granulomatous lesions. No discrete granulomatous lesions containing central acid-fast bacteria were found, but possible single isolated, acid-fast positive-rod bacteria, considered possible Mycobacteria, were detected histologically in thoracic lymph nodes from 3 horses (Nos. 2, 3, and 9). Frozen thoracic lymph node samples from these horses were then examined by PCR for Mycobacterium. 15 One lymph node from horse No. 9 was positive for Mycobacterium celatum, a nontuberculous mycobacterial species. Additional sections of lymph node from this horse were examined with acid-fast stains, but no foci of mycobacterial granulomatous inflammation were detected.
Bone
Gross Lesions
Only affected horses had visible bone abnormalities other than osteoarthrosis at necropsy. Mild to severe abnormalities were seen predominantly in the axial skeleton and proximal portion of the appendicular skeleton. Acute fractures or fracture calluses were observed most commonly in the ribs (8 horses) or pelvis (3 horses). One horse (No. 7) with a pelvic fracture also had a crushed second cervical vertebra. Two horses (Nos. 3 and 8) had bone nodules on the middle of the horizontal ramus of the mandible.
Scapular abnormalities were apparent in all affected horses. Abnormalities included diffuse thickening of the spine, neck, and cranial and caudal borders of the body; roughening of the spine; and lateral convex curvature (Fig. 1 ). Some horses had focal hemorrhage or muscle pallor adjacent to the scapular spines, which was consistent with muscle atrophy and fibrosis of the infraspinatus and/or supraspinatus muscles.
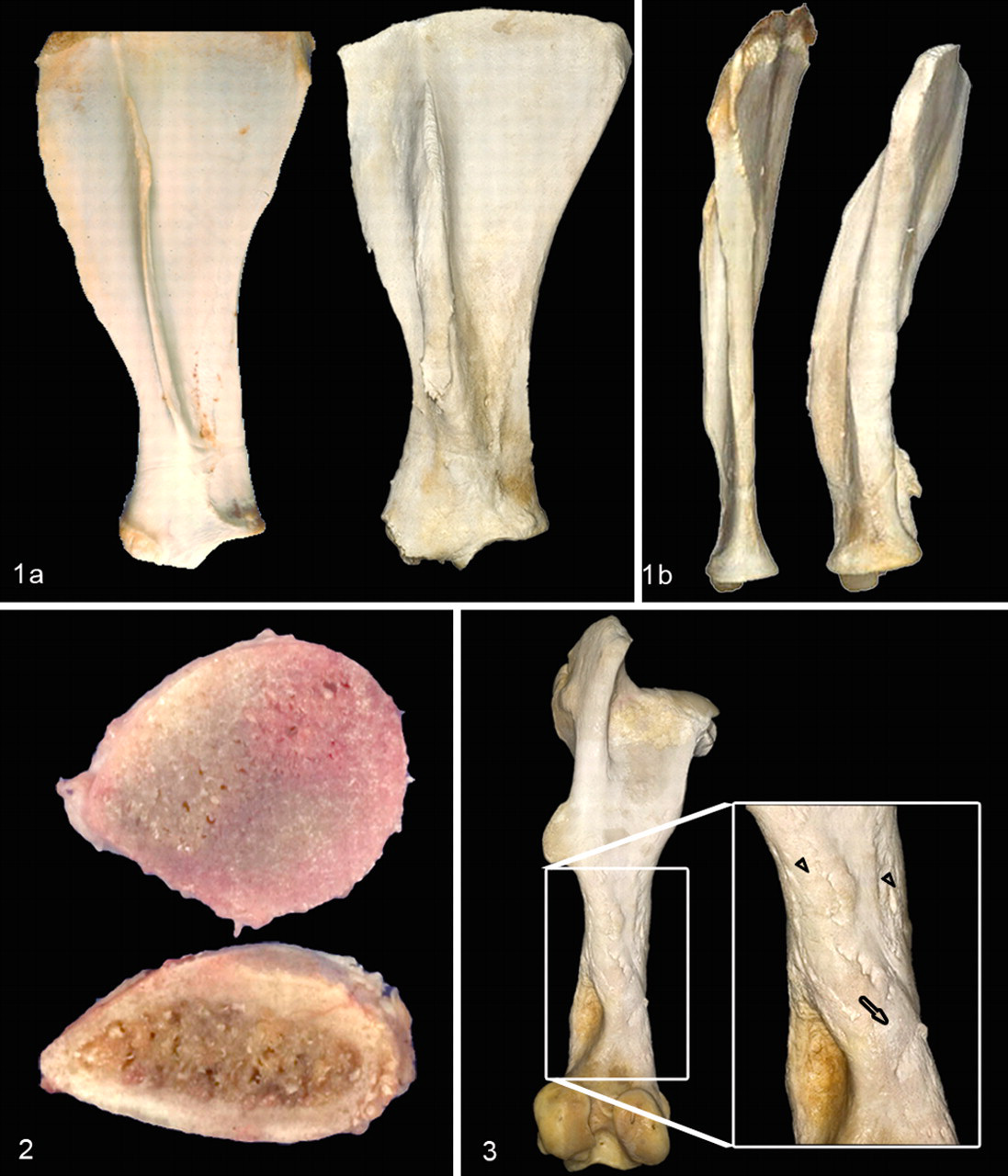
Ribs either were diffusely enlarged or had focal enlargements consistent with fracture calluses. As ribs became enlarged, transverse sections became more rounded. The cut surface of the cortex either appeared thick (most affected horses) compared with unaffected horses or was indiscernible from the medullary trabecular bone tissue (Fig. 2 ). Ribs with these gross abnormalities were easily transected with rib cutters, compatible with osteopenia. Evidence of acute (eg, hemorrhage with sharp fracture edges) or chronic healed fractures (eg, domed smooth bone calluses) was present in 7 affected horses. Two horses (Nos. 7 and 8) had gross evidence of >10 fractured ribs, 5 horses (Nos. 1–4, 6) had evidence of 1 to 5 fractured ribs, and 2 horses (Nos. 5 and 9) had no observed rib gross pathology.
Lesions noted on the parasagittal section of the cervical vertebrae included articular facet exostoses and vertebral body protrusion into the spinal canal.
Pelvic abnormalities included deformations and/or acute or healed fractures. Four affected horses (Nos. 2, 3, 6, 9) had pelvic deformities, 2 (Nos. 7 and 8) had comminuted fractures, and 1 (No. 2) had a unilateral iliac wing simple fracture. The most common deformity was bilateral ventral deviation of the iliac wings.
Osteoarthrosis was apparent in 1 or more limbs of 5 affected and all 3 unaffected horses. Affected horses had osteoarthrosis of the scapulohumeral (Nos. 2, 3, 8), coxofemoral (No. 7), and the distal interphalangeal (Nos. 7, 9) joints. All 3 unaffected horses (Nos. 10–12) had osteoarthrosis of interphalangeal joints; 1 unaffected horse (No. 11) also had osteoarthrosis of the coxofemoral and femorotibial joints.
Periosteal surface pathology was present on all bones of the lye digested skeleton of horse No. 3. Gross lesions ranged from discrete, focal areas of rough textured woven bone to larger regions of rough surfaced reactive woven bone overlying smoother surfaced, consolidated remodeled bone tissue (Fig. 3 ). The surfaces of vertebrae, ribs, pelvis, and scapulae were completely affected by these lesions and had little normal parent cortical bone surface visible. These same periosteal abnormalities were present but less frequent in the remaining appendicular skeleton and, when present, were less prominent or smaller in size on distal bones compared with proximal bones.
Radiographic Lesions
Lye-digested skeleton
Radiographs of the bones of the moderately affected horse (No. 3) illustrated extensive distribution of abnormalities among bones. Lesions were apparent in most bones; however, the more severe changes were in bones of the axial skeleton and the proximal portion of the appendicular skeleton. The scapulae, ribs, proximal aspect of the femur, proximal aspect of the humerus, cervical and thoracic vertebrae, pelvis, and mandible were most severely affected. The only bones without radiographically detectable lesions were the carpal bones and the third metatarsal bones.
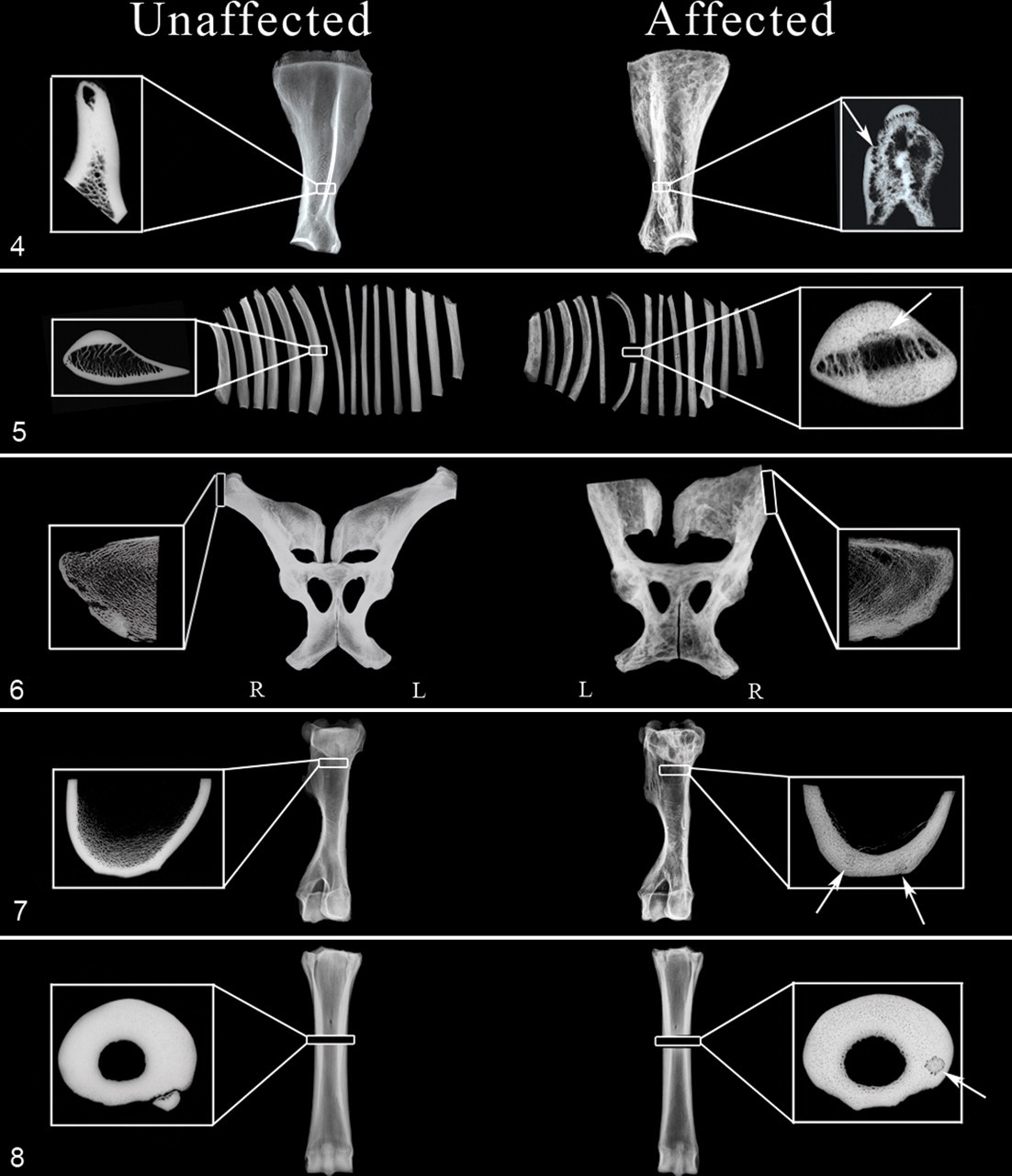
Osteolytic foci were apparent in trabecular and cortical bone tissue of scapulae, ribs, vertebrae, and affected long bones. Coalescing foci gave the appearance of loculated lesions. Lesions ranged in size from punctuate to 4 × 2 cm and were often surrounded by sclerosis. A diffuse, coarse trabecular pattern was apparent in the vertebral bodies, pelvis, and medullary cavities of affected ribs, long bones, and phalanges. Scapulae had multiple osteolytic coalescing foci (lesions averaging 1.5 × 1.5 cm) that created a multilocular appearance throughout the infraspinous and supraspinous fossae, neck, and supraglenoid tubercle (Fig. 4 ). Irregular bone production thickened the scapular spine, and some scapulae had calluses that were consistent with fracture repair. Vertebral bodies had a diffuse, coarse trabecular pattern and multilocular osteolysis with coalescing focal lytic lesions averaging 2 × 2 cm in size. Ribs had a coarse trabecular pattern with multilocular osteolysis as well as discrete osteolytic foci up to 4.1 × 2.3 cm in size (Fig. 5 ). The pelvis was diffusely affected with multilocular osteolysis throughout the ilium, ischium, and pubis (Fig. 6 ). The proximal aspects of bilateral femora and humeri had large osteolytic foci up to 2.9 × 2.5 cm in size (Fig. 7 ).
Three- to 5-mm-thick transverse bone sections
All affected horses had radiographic abnormalities in at least 4 of the 7 bones assessed. Abnormalities were apparent in sections of scapula (Nos. 1, 3–9; Fig. 4), rib (Nos. 1–5, 7–9; Fig. 5), iliac wing (Nos. 1, 3, 5–9; Fig. 6), humerus (Nos. 1, 3, 5, 8, 9; Fig. 7), third metacarpal bone (Nos. 1–4, 6–9; Fig. 8 ), vertebrae (Nos. 2–5, 7, 8), and third carpal bone (Nos. 1–3, 6–9). Mild to severe cortical and trabecular osteopenia and large locules (scapulae, Fig. 4) and osteolytic foci (ribs, Fig. 5) were apparent. Cortical thickness was increased in the rib, third metacarpal bone, and humeral sections. The cortical–trabecular boundary was indistinct in the scapular and rib sections (Figs. 4, 5). Two horses (Nos. 1, 3) had discrete, focal circular lesions surrounded by large porosities within the cortex of the humerus (Fig. 7), and 1 horse (No. 3) had a similar lesion in the third metacarpal bone (Fig. 8). This same pattern of an osteopenic region surrounded by a lucent rim could also be appreciated at a larger scale in the scapular cross sections (Fig. 4) from 3 horses (Nos. 3, 5, 8). In some instances, most notably in the scapula, the lucent rim was surrounded by a shell of bone tissue. Diffuse osteopenia, thickened cortex, and narrow medullary cavity were evident throughout the length of a serially transected rib from 1 horse.
Bone Histology: 7th Rib
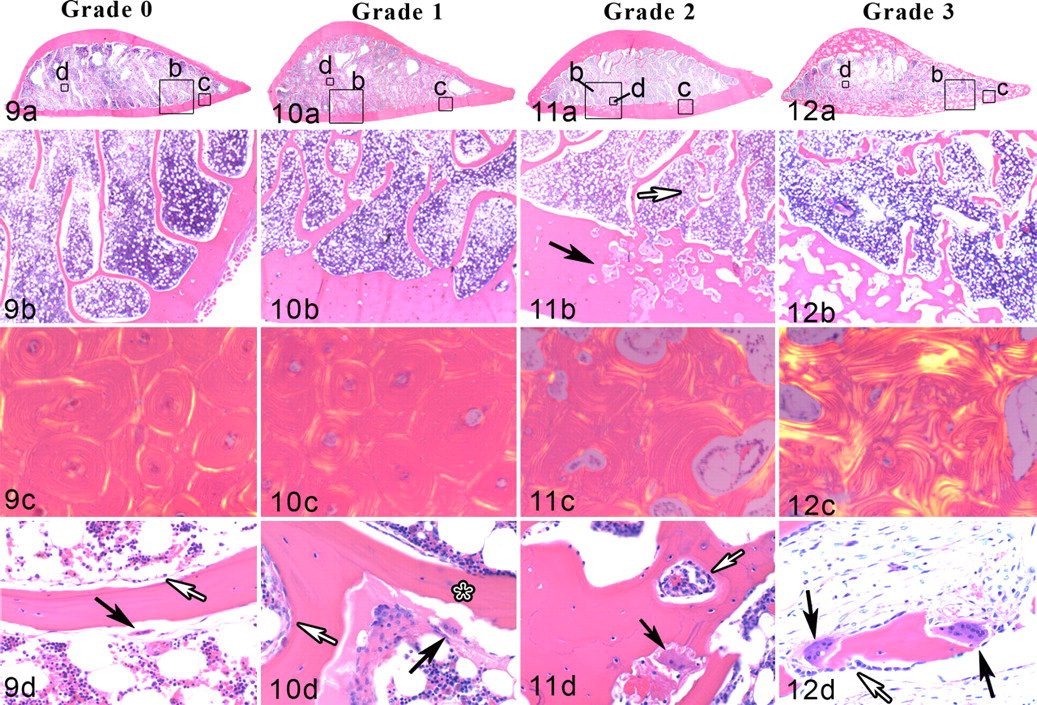
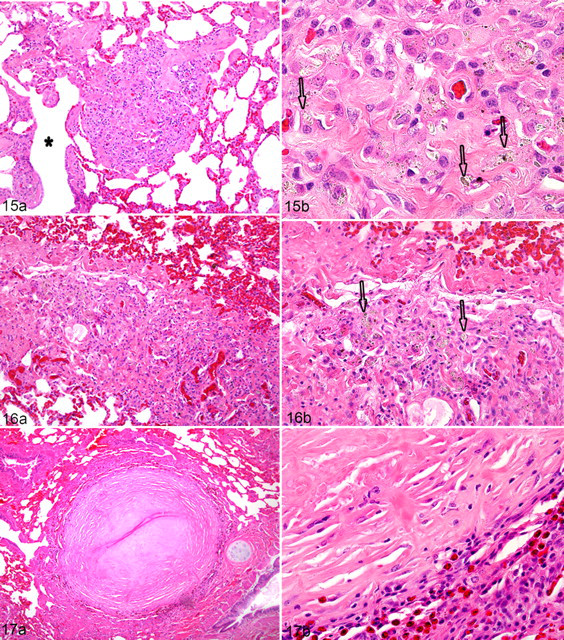
A pattern of histologic abnormalities was observed with disease progression. Based on these observations and irrespective of overall disease status, a histologic grading score was created. Trabecular changes were observed in all affected horses, whereas cortical changes were only observed in moderately and severely affected bones (Figs. 14–17 ). Mildly affected bones (grade 1) had an apparent increase in the number and size of resorption cavities along trabecular surfaces (Fig. 10 ). With disease progression, osteoclastic bone resorption appeared unrestricted, resulting in trabecular transection and fragmentation and occasionally lengthwise dissection (dissecting osteitis) (Figs. 11–13 ). Moderately and severely affected bones also had an increase in number and size of resorption cavities in the cortex (Figs. 11, 12). Cortical findings in affected horses ranged from normal osteonal architecture (mildly affected bones) to focal, isolated regions of large resorption cavities (moderately affected bones) and to diffuse, coalescing resorption cavities that obliterated normal osteonal architecture and obscured the cortical–medullary cavity boundary (Figs. 9–12 ). Severely affected horses had random arrangements of collagen bundles indicative of multiple and disorganized remodeling events (Fig. 12c). Cortical width increased with disease severity with the greatest change appreciated on the medial cortex (Figs. 9–12).
Remodeling exacerbated with disease progression. Osteoclasts in trabecular and cortical bone tissue became more numerous and larger and acquired small intracytoplasmic vacuoles and supernumerary nuclei (Fig. 12d). Osteoblasts that were flat in normal or mildly affected bones (Figs. 9d, 10d) were cuboidal shaped in more severely affected bones (Figs. 11d, 12d). Cement lines were numerous in affected bones and formed a patchwork (mosaic) pattern in moderate and severely affected bones (Fig. 14).
All 9 affected horses (Nos. 1–9) had a histologic grade ≥1, whereas the 3 unaffected horses had a histologic grade = 0 (Table 1 ).
Histopathological Patterns and Grades a for Bone and Lung Specimens and Radiographic Opacity for Lung Specimens From 9 Horses With and 3 Horses Without Bone Fragility Syndrome

Rows are ordered by bone disorder severity and then by severity of lung histopathology pattern 1, 2, and 3 within a bone syndrome severity level.
a See text for pattern and grade descriptions.
b –, grade 0; +, grade 1; ++, grade 2; +++, grade 3.
c –, no pattern present; +, miliary pattern; ++, foci of opacity; +++, both patterns present.
Lung
Gross Lesions
The lungs in 5 affected horses (Nos. 1–4, 9) had gross abnormalities. These lungs had infrequent pinpoint firm to gritty foci palpable throughout that, when visible, appeared as tiny 1- to 2-mm diameter tan-white foci. The lungs of 3 horses (Nos. 1, 4, and 9) had additional large random firm foci. In horse No. 4 there were 3 larger (3–4 cm in size across their longest axis) pale, tan-white, fibrotic subpleural foci in the right middorsal and caudal lung lobes. The lungs from horses 1 and 9 had numerous small gritty foci and larger (the largest measured 6 cm across its longest axis), random tan-white fleshy or firm foci. Additionally, horses 1 and 9 had single large, solitary, firm tan-white foci with a hard mineralized central region in a caudal lung lobe. The lungs of 4 affected horses (Nos. 5–8) and all unaffected horses (Nos. 10–12) were grossly unremarkable. They had a uniform soft texture, variable congestion, edema, and a red-pink to plum red color.
Radiographic Lesions
Two distinct pulmonary radiographic patterns were observed in the lung sections. The first pattern was characterized by a diffuse, miliary pattern comprised of small mineral opacities (1–3 mm); the second pattern consisted of larger foci of mineral opacity (20–30 mm). Two of the unaffected horses and 2 of the affected horses (Nos. 3, 5, 10, 11) had the diffuse, miliary pattern; affected horses 1 and 9 had the second pattern with at least 1 focal area of increased opacity; and affected horse No. 4 had both patterns. The remaining horses (Nos. 2, 6–8, 12) had no abnormal opacities visible on radiographs (Table 1).
Lung Histology
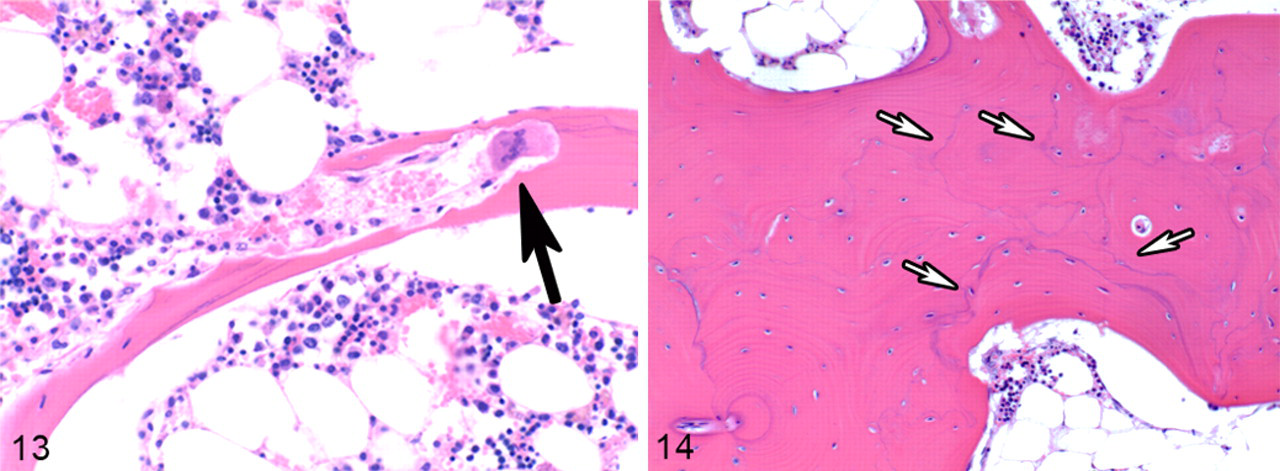
One or more of 3 distinct histologic patterns of pulmonary inflammation of variable severity were seen in the lungs of affected horses but not in the lungs of unaffected horses. Widespread granulomatous fibrosing bronchiolitis with macrophage particulates was the most common pattern, observed in 8 of 9 affected horses (Nos. 1–7, 9). Discrete nodules adjacent to small airways and alveolar ducts consisted predominantly of macrophage aggregates surrounded by reactive fibrous stroma and small numbers of lymphocytes (Fig. 15a). Macrophages contained heterogeneous, pigmented to clear crystals (Fig. 15b). This pattern of peribronchiolar granulomatous inflammation was generally disseminated with the size and density of the granulomatous foci varying regionally throughout the lungs. With increasing size, these foci expanded into the adjacent alveolar parenchyma, surrounded adjacent distal airways, and were accompanied by airway smooth muscle hypertrophy. Enlargement of the foci led to coalescence of contiguous fibrotic parenchymal foci causing compression of terminal airways, entrapment of alveoli, and loss of normal parenchyma. Entrapped alveoli often were lined by hyperplastic epithelium and occasionally contained small numbers of free macrophages and, in 1 horse (No. 9), neutrophils.
A second pattern of pulmonary inflammation, dissecting interlobular fibrosis with macrophage particulates, was seen in 5 affected horses (Nos. 1–4, 9) and only in horses that had moderate to marked widespread granulomatous fibrosing bronchiolitis. Variable-sized, random foci of interlobular and/or pleural fibrosis were associated with scattered aggregates of macrophages that contained heterogeneous, pigmented to clear, crystals (Fig. 16). These macrophage infiltrates were accompanied by variable numbers of lymphocytes. Prominent focal eosinophil infiltrates were also seen infrequently within foci of interlobular/pleural fibrosis in 2 horses (Nos. 2, 4).
The third pattern of pulmonary inflammation, random focal nodular fibrosis with necrosis, was seen in 2 affected horses (Nos. 3, 4) that also had pronounced widespread granulomatous fibrosing bronchiolitis and dissecting interlobular fibrosis. Infrequent, randomly located, discrete circular nodules were composed of dense reactive fibrous connective tissue with variable amounts of central necrosis of collagenous stroma. These nodules often contained small numbers of necrotic macrophages (Fig. 17). In many of these nodules, the necrotic central region had undergone dystrophic mineralization. The periphery of these fibrotic nodules was invariably surrounded by eosinophils, lymphocytes, macrophages, and plasma cells (Fig. 17b). Small aggregates of particulate-laden macrophages were often seen in the immediately surrounding compressed parenchyma. No bacteria or fungi were found on special stains within these nodules, and crystalline particulate could not be found in the vast majority of these nodules. Rare individual crystals were visualized in only 3 of 9 nodules from horse No. 3 and 2 of 25 nodules from horse No. 4.
Extensive focal interstitial eosinophil and lymphocyte infiltrates were seen in a few of the lung sections from 1 horse (No. 4) and were associated with several of the random foci of nodular fibrosis with necrosis. These infiltrates were visualized as dissecting through alveolar septa, around larger vessels, and into interlobular septa as well as around the foci of nodular fibrosis with necrosis. A single small, discrete eosinophilic granuloma consistent with possible previous pulmonary larval parasite migration was also present. A similar focus of pronounced interstitial eosinophilic inflammation, lacking any fibrotic nodules or eosinophilic granuloma formation, was seen in the lung of horse No. 2.
In horse No. 1, there was a single large focus of pulmonary parenchymal effacement and loss with replacement by dense mineralized reactive fibrous connective tissue. This large focus was composed of intersecting foci of nodular fibrosis and was identical to the thoracic lymph node pattern of silicotic fibronodular lymphadenitis with necrosis described below.
A histologic grading scheme (1–3) was developed to document the relative extent of each of these 3 lung inflammatory patterns in the affected horses.
Intracytoplasmic crystals were heterogeneous in color (black, brown, or clear) and were consistent with mixed silicates and/or inhaled dust particles (Fig. 15b). A variable number of crystals were birefringent under polarized light, but the relative density and number of birefringent crystals varied markedly among horses. There were no apparent correlations between the size, shape, and tinctorial qualities of the crystals or the density or number of birefringent crystals seen under polarized light. In a majority of the horses, relatively large numbers of easily visible particulate matter were present within the macrophages. However, in 3 of the affected horses (Nos. 1, 2, 9), this crystalline particulate matter was very low in density, often nonpigmented, and submicron in size and thus very difficult to detect. Detection required manipulation of the microscope light by either closing the condenser diaphragm to increase the refractive index of light or examining the sections under polarized light. The particulates were often more visible using acid-fast stains where individual particulate matter stained either eosinophilic or basophilic.
The presence of pulmonary fibrosis associated with granulomatous foci of inflammation and phagocytized heterogeneous, partially birefringent (under polarized light) crystalline material compatible with silicates, was the minimum requirement for a histologic diagnosis of pulmonary silicosis. 3,47 In this study, this equated with the presence of lung pattern 1, widespread granulomatous fibrosing bronchiolitis, which appeared to be the initial fibrotic pattern noted within affected horse lungs. In contrast, pneumoconiosis was diagnosed when there was no reactive fibrosis associated with the presence of these same crystalline-laden macrophages. Eight affected horses (Nos. 1–7, 9) had pathologic findings consistent with pulmonary silicosis. The remaining affected horse (No. 8) had very mild multifocal pleural, interlobular, and peribronchiolar fibrosis within the dorsal aspect of the lung lobes but no appreciable particulate-laden macrophages were observed. One unaffected horse (No. 12) had infrequent small aggregates of crystal-laden macrophages adjacent to some terminal airways consistent with pneumoconiosis but no associated fibrosis.
Lymph Node
Gross Lesions
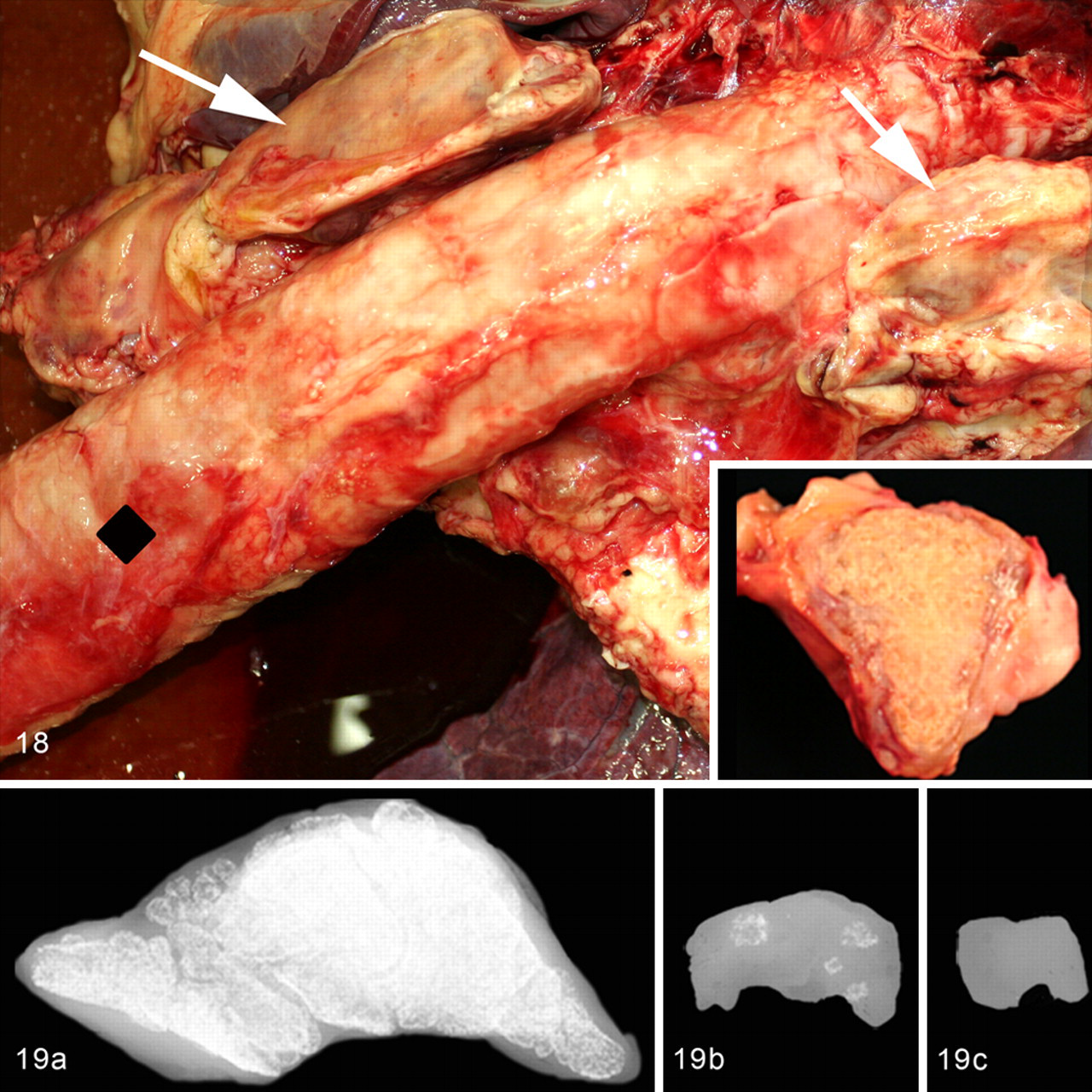
Lymph nodes from affected horses that had gross lesions included thoracic (which included tracheobronchiolar and mediastinal), cranial abdominal, and, in only 1 horse (No. 9), mesenteric. Thoracic lymph nodes were enlarged and firm in 8 of 9 affected horses (Nos. 1–5, 7–9). Of these, tracheobronchiolar lymph nodes were usually the most prominently enlarged (3–16 cm in diameter, 1–262 g), fibrotic, tan-white, and variably mineralized with a firm, gritty, or hard texture in 7 affected horses (Nos. 1–5, 7, 9) (Fig. 18 ). Cranial and caudal mediastinal lymph nodes were enlarged to a lesser extent (1–5.6 cm, 1–42 g), and although they often appeared fibrosed and mineralized, these features were usually less pronounced. These nodes were hard or gritty in 7 affected horses (Nos. 1, 3–6, 8, 9). Cranial abdominal (gastric, hepatic, renal) lymph nodes were also enlarged (1.9–5.5 cm, 2–22 g) in 5 affected horses (Nos. 1–4, 9) with focal pale tan-white firm to gritty foci. The mesenteric lymph nodes were grossly hard and gritty in 1 affected horse (No. 9) (2.9 cm, 6.25 g).
All thoracic and cranial abdominal lymph nodes in unaffected horses were soft and tan to red-brown in color and ranged from 3.3 to 5 cm, 7 to 7.5 g (bronchiolar lymph nodes); 0.8 to 1.1 cm, 0.12 to 0.41 g (thoracic lymph nodes); and 0.44 to 4.2 cm, 0.44 to 2.5 g (cranial abdominal lymph nodes).
The submandibular and cervical lymph nodes were grossly unremarkable with a soft texture in all horses.
Radiographic Lesions
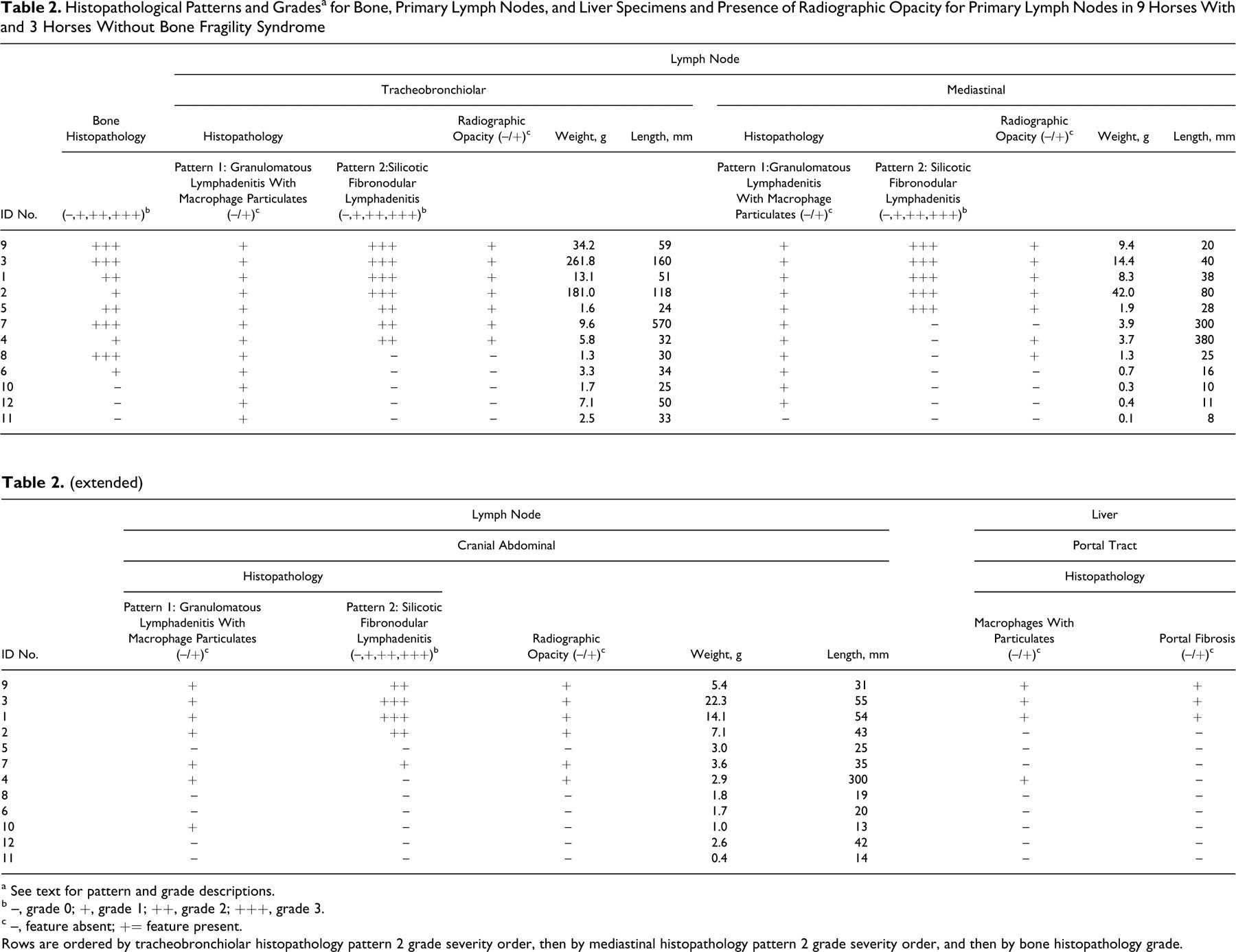
Radiographic mineral opacity was present in grossly enlarged, gritty, and hard lymph nodes. Mineralization ranged from complete mineralization of the node (Fig. 19a ) to a few to localized foci (Fig. 19b). Seven of 9 affected horses (Nos. 1–5, 7, 9) had mineralization in bronchiolar lymph nodes, and 7 of 9 affected horses (Nos. 1–5, 8, 9) had mineralization in mediastinal and cranial abdominal lymph nodes (Table 2 ). The caudal (mesenteric) abdominal lymph nodes had mineralization in 2 affected horses (Nos. 3 and 9)
Histopathological Patterns and Grades a for Bone, Primary Lymph Nodes, and Liver Specimens and Presence of Radiographic Opacity for Primary Lymph Nodes in 9 Horses With and 3 Horses Without Bone Fragility Syndrome

a See text for pattern and grade descriptions.
b –, grade 0; +, grade 1; ++, grade 2; +++, grade 3.
c –, feature absent; += feature present.
Rows are ordered by tracheobronchiolar histopathology pattern 2 grade severity order, then by mediastinal histopathology pattern 2 grade severity order, and then by bone histopathology grade.
None of the unaffected horses had lymph node mineralization (Fig. 19c).
Lymph Node Histology
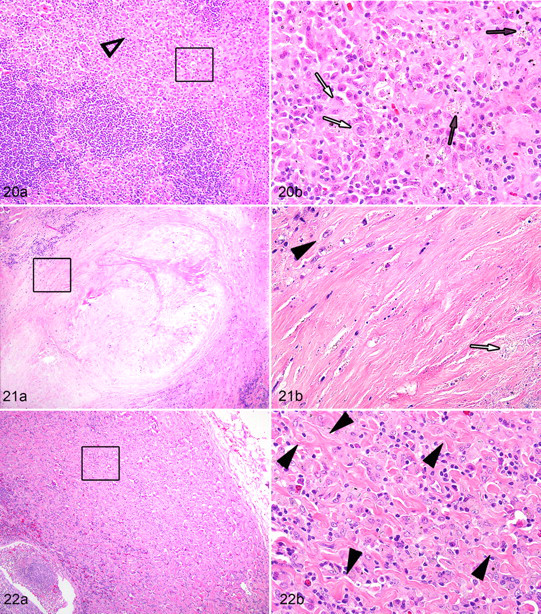
Three histologic patterns were seen in lymph nodes, with 2 of the patterns only observed in affected horses. Granulomatous lymphadenitis with macrophage particulates was found in all 12 affected and unaffected horses. This pattern was more pronounced and widely distributed in the lymph nodes of the affected horses and consisted of variable-sized infiltrates of plump macrophages that contained rare to abundant heterogeneous, pigmented to clear, crystalline particulate matter (Fig. 20 ). Macrophage infiltrates were present within medullary cords and, where more prominent, extended from the medullary cords into the adjacent cortex (Fig. 20). Random multinucleate giant cells were also present. This pattern was seen in tracheobronchiolar lymph nodes of all 12 horses, in the mediastinal lymph nodes of all affected and 2 unaffected horses (Nos. 1–9, 11, 12), and in cranial abdominal lymph nodes (renal, hepatic, gastric, splenic) of 6 affected horses (Nos. 1–4, 7, 9) (Table 2).
The term silicotic fibronodular lymphadenitis was adopted to describe the second and dominant histopathological pattern found only in the affected horse group. This pattern was present in 7 horses (Nos. 1–5, 7, 9) and consisted of random coalescing dense nodular foci of reactive fibrosis in which there were loosely scattered individual macrophages containing heterogeneous crystalline particulate matter. The centers of these nodular fibrotic foci often had undergone coagulation necrosis, and this was often accompanied by dystrophic calcium mineral deposition (Fig. 21 ). Necrotic foci contained crystalline particulates in either necrotic macrophages or free within the central necrotic stroma. In most affected horses, the crystalline material in macrophages within these fibrotic foci was difficult to detect as it was nonpigmented and rather sparse (Fig. 21). In severely inflamed lymph nodes (Nos. 1–5, 7, 9), this dense coalescing multinodular pattern of fibrosis expanded the parenchyma resulting in pronounced lymph node enlargement with effacement of the majority or the entire lymph node parenchyma. In these same severely fibrosed lymph nodes there often was accompanying fibrosis of the lymph node capsule and immediately surrounding connective tissue stroma, resulting in firm adhesions of the lymph nodes to adjacent structures including other lymph nodes, the pulmonary pleura, adjacent large vessels, and bronchi. As the lymph node parenchyma was progressively replaced by this fibrotic reaction, the infiltrates of plump particulate-laden macrophages (granulomatous lymphadenitis with macrophage particulate) peripheral to these nodular fibrotic foci correspondingly diminished in extent. This pattern was the dominant histologic finding in thoracic and in select abdominal lymph nodes of only affected horses and in a mesenteric lymph node of 1 affected horse (Table 2). In general, the pattern was most severe in the tracheobronchiolar lymph nodes, less severe in the mediastinal lymph nodes, and least severe in the cranial abdominal lymph nodes; although in a few horses the cranial abdominal lymph nodes were greatly enlarged with severe silicotic fibronodular lymphadenitis. Affected nodes were firm and usually grossly enlarged (Table 1).
The final histologic pattern, dissecting fibrosing lymphadenitis, was infrequently observed only in affected horses and then usually was present in lymph nodes from horses that had concurrent silicotic fibronodular lymphadenitis in thoracic lymph nodes. This last histologic pattern consisted of fine bands of reactive fibrosis that dissected around and between individual particulate-laden macrophages within the lymph nodes (Fig. 22 ). The pattern was only seen as foci in a tracheobronchial, mediastinal, and cranial abdominal lymph node of horse No. 4; in a tracheobronchial lymph node of horse No. 6; and in a mediastinal and a cranial abdominal lymph node of horse No. 9.
With very few exceptions, the mesenteric lymph nodes were relatively unremarkable. Very mild granulomatous lymphadenitis with macrophage particulates was found in only 4 affected horses (Nos. 1, 3, 5, 9), and mild silicotic fibronodular lymphadenitis was found in only 1 horse (No. 9).
Based on these observations, a histologic grading scale was created for the dominant histologic pattern of silicotic fibronodular lymphadenitis. The histopathological grade was based on the percentage of total node parenchyma affected on histologic examination and was not related to the size of the total node.
Other organs with particulate containing macrophage infiltrates
Four of the 9 affected horses (Nos. 1, 3, 4, 9) had very mild hepatic portal infiltrates of macrophages containing crystalline particulates with mild to moderate accompanying fibrosis in 3 horses (Nos. 1, 3, 9). Rare macrophage infiltrates containing crystalline particulates were present in splenic cords of horse No. 1, in the interstitium of the renal cortex in horse No. 9, and lying adjacent to Peyer patches in the ileal or colonic submucosa of horses Nos. 1–4 and 6.
Other Organ Systems—Gross and Histologic Lesions
At least 1 parathyroid gland was located and examined histologically from all horses but unaffected horse No. 12. No significant differences were noted in either the gross or histologic appearance of the parathyroid glands from the affected horses compared with the unaffected horses. There were occasional foci of mild chief cell hypertrophy in 4 affected horses (Nos. 2, 3, 5, 8), but similar mild focal hypertrophy was also detected in unaffected horse No.10.
No additional significant pathologic findings were noted in the viscera of the affected horses.
The 3 unaffected horses had additional, unrelated findings. Horse No. 10 was diagnosed clinically with bilateral front limb navicular disease and lower limb tendonitis. Horse No. 11 had postmortem findings that included a moderate to marked chronic multifocal fibrosing tubulointerstitial nephritis (healed infarcts) and lower limb abnormalities including a distal limb keratoma and focal subcutaneous fibrosis at the level of the distal aspect of the third metatarsal bone. Horse No. 12 had a thyroid adenoma, thyroid adenocarcinoma, and 2 small pituitary adenomas with accompanying mild adrenal cortical hypertrophy consistent with mild subclinical Cushing disease.
Silicon Assay
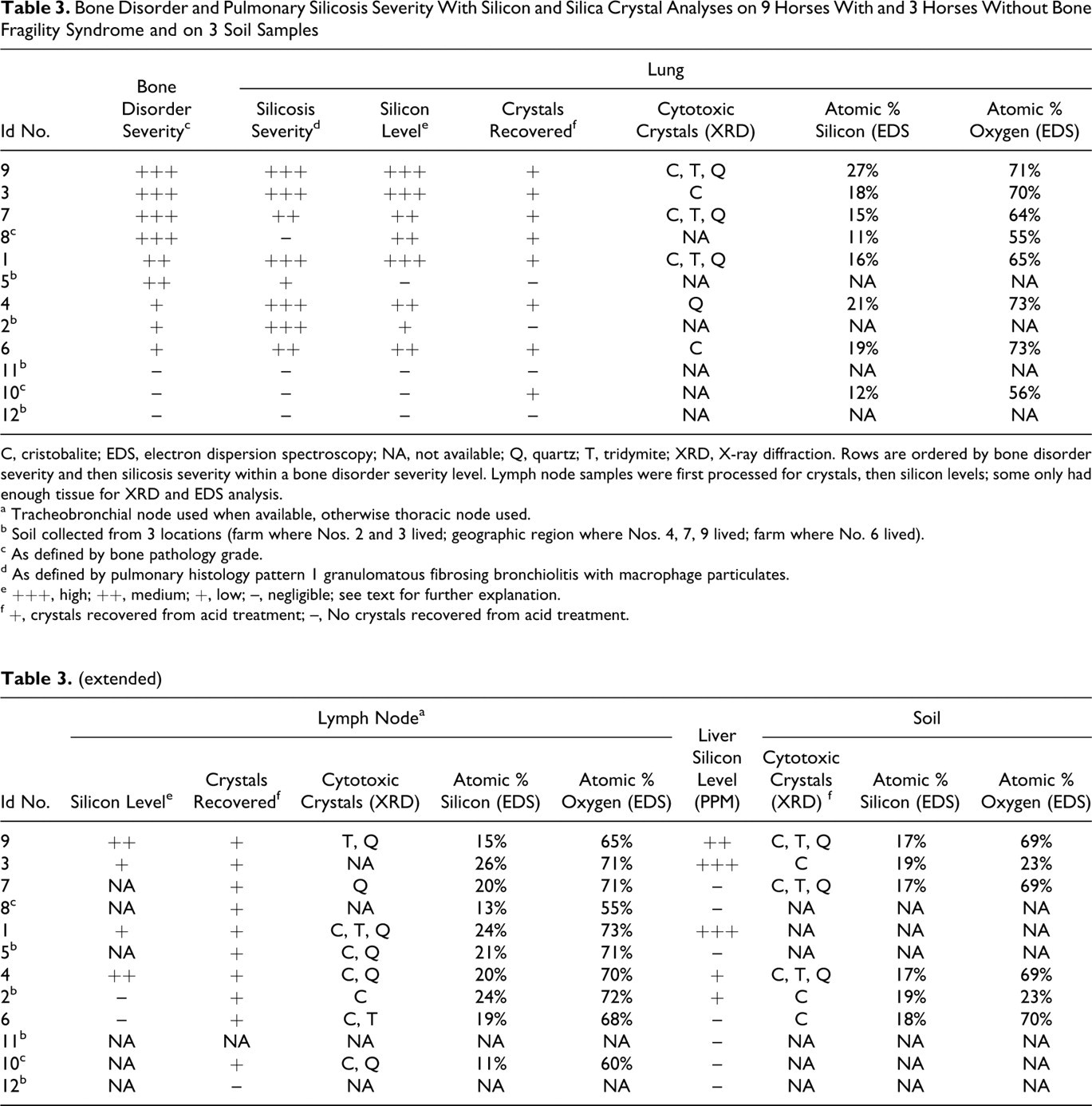
Eight of the affected horses (Nos. 1–4, 6–9) had above negligible lung levels of elemental silicon (Si), 5 of the affected horses (Nos. 1–4, 12) had above negligible liver Si levels, and 5 of the affected horses (Nos. 1–4, 9) had above negligible tracheobronchial lymph node Si levels. Unaffected horses had negligible Si amounts in all 3 tissues when measured (Table 3 ). No silicon levels were detected in bone tissue.
Bone Disorder and Pulmonary Silicosis Severity With Silicon and Silica Crystal Analyses on 9 Horses With and 3 Horses Without Bone Fragility Syndrome and on 3 Soil Samples

C, cristobalite; EDS, electron dispersion spectroscopy; NA, not available; Q, quartz; T, tridymite; XRD, X-ray diffraction. Rows are ordered by bone disorder severity and then silicosis severity within a bone disorder severity level. Lymph node samples were first processed for crystals, then silicon levels; some only had enough tissue for XRD and EDS analysis.
a Tracheobronchial node used when available, otherwise thoracic node used.
b Soil collected from 3 locations (farm where Nos. 2 and 3 lived; geographic region where Nos. 4, 7, 9 lived; farm where No. 6 lived).
c As defined by bone pathology grade.
d As defined by pulmonary histology pattern 1 granulomatous fibrosing bronchiolitis with macrophage particulates.
e +++, high; ++, medium; +, low; –, negligible; see text for further explanation.
f +, crystals recovered from acid treatment; –, No crystals recovered from acid treatment.
Crystal Identification
All 12 horses had samples of lung and lymph node tissue processed by acid dissolution for recovery of crystalline material. Material was recovered from lung tissue from 8 horses (Nos. 1, 3, 4, 6–10) and from lymph node tissue from 11 horses (Nos. 1–10, 12). The EDS from all recovered samples indicated that the largest component (by percentage) of the crystalline material was oxygen (>50%), followed by silicon (11%–27%) with close to a 2:1 relationship indicating the predominant presence of SiOx structures. The remaining elements, which are often found in mixed silicates, were found in only trace amounts (<2% average) (Table 3).
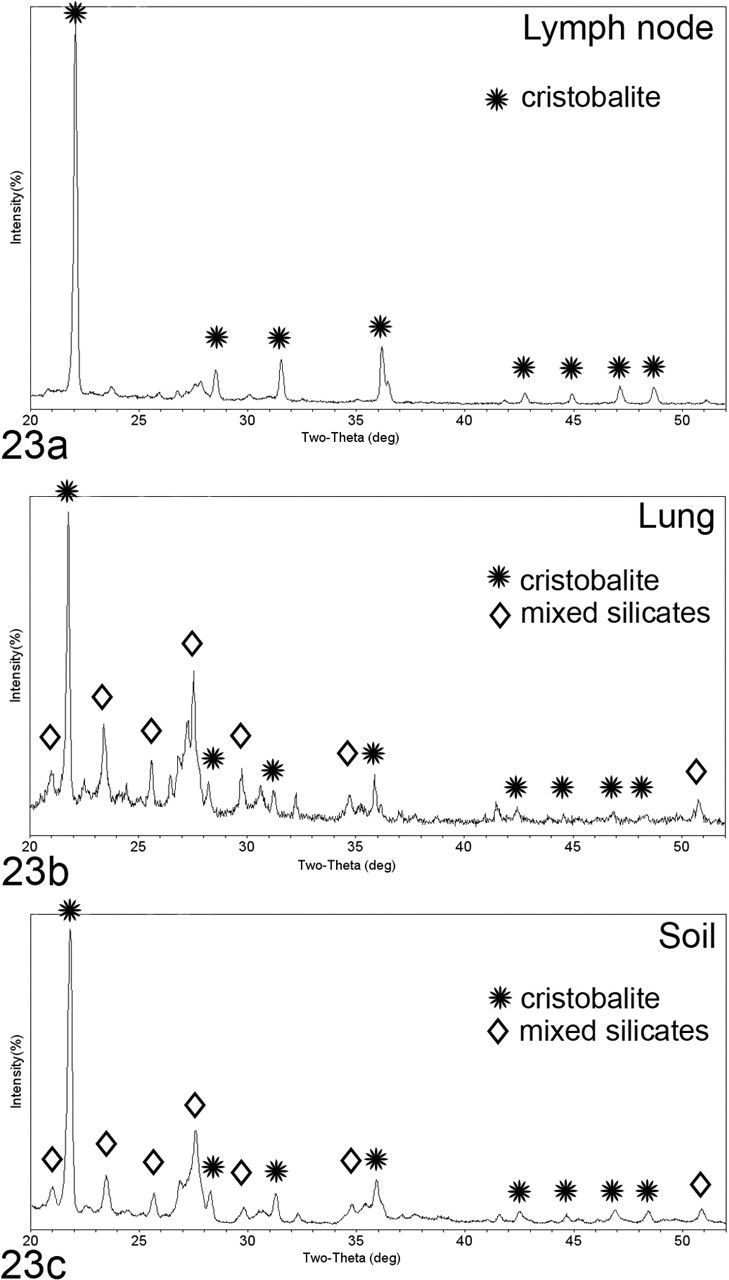
X-ray diffraction (XRD) was performed on recovered crystals from tissue samples from the lung from 6 affected horses (Nos. 1, 3, 4, 6, 7, 9) and the lymph node from 8 affected horses (Nos. 1–7, 9) and 1 unaffected horse (No. 10). All analyzed material contained cytotoxic polymorphs of SiO2 (cristobalite, quartz, and/or tridymite). The crystalline material in some samples was pure SiO2 with the sample from the lymph node of horse No. 3 having only cristobalite (Fig. 23 ), whereas other lung and lymph node samples (Nos. 1, 4, 7, 9, 10) had a mixture of cytotoxic polymorphs. The remaining lung and lymph node samples (Nos. 2–6) had a combination of cytotoxic SiO2 polymorphs, mixed silicates (opal, SiO2 H2O); orthoclase, microcline, sanidine (KAlSi3O8); aluminum silicate (AlSiO); mullite (Al6Si2O13); albite (NaAlSiO3); sodium aluminum silicate (NaAlSiO4); and other soil particles (sodium titanium oxide (NaTiO3) and akaganeite (FeO(OH)). In all samples, the cytotoxic crystals were the polymorphs with the greatest peak intensity on the XRD scan and thus the dominant crystalline structure within the sample (Table 3).
No crystals were recovered from the bone samples via the acid dissolution procedure, and thus XRD was not performed on bone.
Silica dioxide polymorphs were confirmed in all 4 of the soil samples from endemic regions. The outcropping of Miocene Monterey Shale from Monterey county consisted of 5 types of SiO2 (quartz, cristobalite, tridymite, silicon oxide, and moganite), and the sample from an endemic farm in Monterey county had 3 types of SiO2 (quartz, cristobalite, and silicon oxide). The 2 endemic properties in Sonoma and Napa counties had cristobalite, other silica oxide polymorphs (orthoclase, microcline, albite), magnetite (Fe3O4), hematite (Fe2O3), calcite (CaCO3), and calcium oxide (CaO2). When the lymph node, lung, and soil XRD scans are compared for a given horse housed on a particular property or soil type, the dominant crystal on the scan is consistent across all 3 samples (Fig. 23).
The unaffected properties in California also had silica oxide polymorph dominant peaks, whereas the property from Nevada did not have any silica in the soil. The unaffected property from Monterey had quartz and silica oxide polymorphs (microcline, albite, and magnesium and iron oxide). The control sample from Sacramento county had 3 types of SiO2 (quartz, cristobalite, tridymite) and anorthite (CaAl2Si2O8). The other 2 control samples from California counties had quartz as the only SiO2 polymorph and included Yolo with quartz, anorthite, and muscovite (KMgAlSi4O10(OH)2) and San Bernadino with quartz, halloysite, albite, anorthite, and magnesium iron oxide. The sample from Washoe Valley in Nevada did not have any SiO2 polymorphs (microcline, albite, anorthite, sanidine).
Correlations
The presence of osteoporosis was correlated with the presence of silicosis (r = 0.8, P < .01). Bone pathology severity grade was correlated with the lung silicon levels (r = 0.7, P < .01), lung gross pathology (r = 0.8, P < .05), tracheobronchiolar lymph node gross pathology (r = 0.7, P < .05), histology pattern 2 (r = 0.7, P < .05), and radiographic opacity (r = 0.7, P < .05) and with the presence of cytotoxic crystals in tracheobronchial lymph nodes (r = 0.6, P < .05) but not with the lung histopathology findings. The presence of cytotoxic crystals found in lung tissue was only correlated with the presence of cytotoxic crystals in the lymph nodes (r = 0.6, P = .03) and with lung silicon levels (r = 0.7, P < .01). The presence of cytotoxic crystals in lymph node tissue was correlated with bone pathology grade (r = 0.6, P = .04), the presence of osteoporosis (r = 0.8, P < .01), the presence of gross lung pathology (r = 0.6, P = .03), and the presence of lung histology pattern 1 (r = 0.6, P = .03). Liver silicon levels were correlated with the presence of macrophages with particulate matter and with portal fibrosis (r = 0.9, P < .01; and r = 0.8, P < .01, respectively). Age was not correlated with any of the variables.
Discussion
Findings
Through postmortem examination and ancillary tests on 9 horses with a bone fragility syndrome and on 3 unaffected horses, this study aimed to (1) characterize the pathologic, histopathologic, and radiographic postmortem findings associated with the bone fragility syndrome and (2) to assess associations between bone disease and pulmonary silicosis and levels of heavy metals and trace minerals. Eight of the 9 affected horses had gross pathological, histopathological, and radiographic evidence of both systemic osteoporosis and pulmonary silicosis. Silicon and oxygen were the predominant elements found in crystals from lung tissue of 7 affected horses and tracheobronchial lymph node tissue from 9 affected horses; cytotoxic silica dioxide (SiO2) crystals (including quartz, cristobalite, and tridymite) were identified in both lung (6 affected horses) and tracheobronchial lymph nodes (8 affected horses). Elemental silicon levels were quantifiably elevated in lung tissue from 8 affected horses. The presence of cytotoxic crystals in lymph node tissue was correlated with severity of bone pathology and the presence of histologic findings consistent with pulmonary silicosis, that is, granulomatous fibrosing bronchiolitis with macrophage particulates. Severity of bone pathology was also correlated with the level of elemental silicon detected in lung tissue. Despite the presence of bone, lung, and lymph node disease in all affected horses, correlations between the severities of pathological changes among the respective organs were not observed. PTH levels, measured in two thirds of the affected horses, were elevated. Serum phosphate was high and vitamin D was low in several affected horses. Heavy metal and trace mineral levels were within normal limits in all horses. No infectious causes of either the pulmonary or the bone diseases were found, although an incidental M. celatum infection was detected in the bronchiolar lymph node of 1 horse. Because 8 of the 9 affected horses had osteoporosis, pulmonary silicosis, and silicate crystals or elemental silicon in lung and thoracic lymph node tissues, this study provides supportive evidence for SAO. The presence of cytotoxic crystals and inflammation in the tracheobronchiolar lymph nodes may be a key factor in the development of pulmonary disease related to osteoporosis.
Salient features of SAO included gross, radiographic, and histologic bone abnormalities. With disease progression, bones of the skeleton developed visible deformations, exostoses, and periosteal callus. The wide distribution of loculated radiographic lesions supported systemic disease with focal areas of intense remodeling activity. Histologically, trabecular and cortical bone resorption was mild to marked, with random resorption in severely affected bones causing osteoporosis and loss of normal architecture. The moderately and severely affected SAO bones had large osteoclasts with supernumerary nuclei (>12 nuclei) and a foamy cytoplasm, associated with excessive bone resorption and multiple remodeling events. Plump osteoblasts concurrent increased bone formation resulted in a patchwork (mosaic) of lamellar bone packets.
SAO and Hyperparathyroidism
Horses in the current study had some features consistent and other features inconsistent with horses with hyperparathyroidism. Common clinical features included stiffness, shifting leg lameness, thickened and curved scapulae, lordosis or kyphosis, ligamentous avulsions, and pathologic fractures; 35 however, only 1 horse in the current study had bilateral enlargement of the skull (fibrous osteodystrophy) typical of equine hyperparathyroidism. Similar to horses with hyperparathyroidism, many SAO horses had elevated serum PTH levels, increased third metacarpal cortical porosity, 31 and dissecting resorption of trabeculae on histology. 38 However, SAO horses had increased cortical width and loss of trabecular connectivity in contrast to the decreased cortical width and maintenance of trabecular architecture observed in horses with hyperparathyroidism. 32 Parathyroid gland hyperplasia was not apparent in SAO horses; however, the lack of visible parathyroid gland hyperplasia may be due to the difficulty in locating all the parathyroid gland tissue in the horse to assess for abnormalities. 32
SAO and Paget Disease of Bone
The systemic, multifocal lytic aspects of the SAO bone disease are similar to those of Paget disease of bone (PDB) in people. 42 PDB is characterized by skeletal deformations, pathologic fractures, multifocal lytic lesions, and a histological pathognomonic lamellar patchwork mosaic indicative of multiple remodeling events. 46 The distribution of skeletal lesions, scintigraphic findings, and the location of affected individuals in geographic clusters are common to SAO and PDB. Both diseases have fragmented trabeculae, osteoclasts with supernumerary nuclei (>12 and up to 100 nuclei) along the trabecular surfaces, deep bone resorption bays, numerous active cuboidal osteoblasts, random arrangement of collagen fibers, and the mosaic pattern of lamellar bone. 46 Both diseases are also characterized by an increase in cortical thickness presumably due to increased bone formation.
SAO and PDB differ in age of disease onset, disease progression, and the persistence of osteoclast activity. Adult PDB (polyostotic disease) typically affects people older than 60 years. Pagetic lesions initiate with an increase in osteoclastic activity and resorption; subsequently, osteoblastic activity increases; and finally both osteoblastic and osteoclastic activity decrease. 13 Once Pagetic lesions develop, they rarely progress to new locations. 46 In contrast, equine SAO appears to affect horses of all age 2,17 and is progressive, 2 and osteoclastic hyperactivity persists late in the course of the disease.
SAO and Pulmonary Silicosis
The other salient features of SAO, which included lung and tracheobronchiolar lymph node histopathological changes, were consistent with pulmonary silicosis. Although the pathology of pulmonary silicosis has been well documented for humans, 24,47,48 there are only limited descriptions of equine silicosis 4,45 and the similarities to and differences from the human condition. The results of the current study reveal that the pathology of equine silicosis is strikingly similar to that of human silicosis with a few prominent differences. Briefly, silicosis in humans has many clinical and pathological forms that have been described including acute silicosis (silicoproteinosis), chronic silicosis with formation of simple nodules, and conglomerate nodular formation that occasionally results in progressive massive fibrosis. Based on exposure time and duration, the disease can also be considered chronic (low dose over decades), accelerated (medium dose over years), or acute (high dose over months) in cause. 24,44
The pulmonary silicosis seen in SAO horses most closely resembles the early changes associated with chronic, or simple, silicosis in humans. In humans, chronic silicosis is the most common form and is a progressive disease characterized initially by the formation and dissemination of small reactive fibrotic nodules (microscopic lesions) containing silica-laden macrophages within the pulmonary parenchyma. These lesions are somewhat similar to the dominant equine lung histopathological lesion widespread granulomatous fibrosing bronchitis with macrophage particulates (pattern 1) seen in affected horses. In humans, these microscopic nodular foci progress to the formation of the “classic silicotic nodule” (2–6 mm in diameter, 685) with “a central zone composed of whorls of dense, hyalinized fibrous tissue and a midzone made up of concentrically arranged collagen fibers, often exhibiting a feature known as onion skinning” (685) surrounded by peripheral mixed dust-laden macrophages and lymphocytes. On rare occasions, these nodules undergo necrosis and dystrophic mineralization. These classic silicotic nodules are considered “a specific tissue response to crystalline silica” (685). 47 In the current study, the lung histopathological lesion focal nodular fibrosis with necrosis (pattern 3) appeared somewhat similar to classic human silicotic nodules; however, this pattern was rarely encountered in only 2 horses. Established chronic pulmonary silicosis in humans can include the presence of focal pleural and subpleural fibrosis with silicotic nodule formation. In our affected horses, interlobular/pleural fibrosis was also detected (pattern 2, dissecting interlobular fibrosis with macrophage particulates) but silicotic nodule formation as noted in humans 47 was not a feature of this interlobular fibrotic lesion.
Progressive massive fibrosis occurs when simple silicotic nodules coalesce resulting in a nodule >2 cm in diameter, cause extensive damage to and effacement of the lung parenchyma and compromise lung function. 24,44 A few affected horses (Nos. 1, 4, 9) did have large foci (>2 cm in diameter) of fibrosis with mineralization, although it was difficult to determine in horses 4 and 9 whether these originated in parenchyma or interlobular septa. In horse No.1 there was a massive focus of parenchymal fibrosis with mineralization. Histologically, this focus appeared very similar to the description of progressive massive fibrosis in humans consisting of coalescing necrotic fibrotic nodules identical to the dominant multinodular conglomerate lesion described in humans and identical to the silicotic fibronodular lymphadenitis found in thoracic lymph nodes from the affected horses in this study.
We did not find evidence of pulmonary alveolar proteinosis, which is a feature of early-onset pulmonary silicosis in humans and referred to as “acute silicosis” or “silicotic alveolar proteinosis” (680). 47
The unusual discrete pulmonary fibrotic nodules (pattern 3, focal nodular fibrosis with necrosis) found in 2 affected horses are of unknown origin. These nodules were found only in affected horses with pulmonary lesion pattern 1 and pattern 2 and have been detected in additional horses with pulmonary silicosis subsequent to this study (personal observation, B. Barr). Although crystalline particulate could not be identified within most of these pulmonary nodules, their unique morphology is very similar to some of the descriptions of well-developed human pulmonary and extrapulmonary silicotic nodules. 47,48 Shared features include the presence of an expanded central zone of collagen fibers arranged in concentric configurations with variable central necrosis, variable numbers of macrophages within the nodules, and peripheral lymphocytic infiltrates. In addition, this pulmonary pattern of nodular fibrosis with central necrosis resembled the pattern of silicotic fibronodular lymphadenitis seen in the thoracic lymph nodes of the affected horses in which intralesional crystalline particulates were consistently found (Figs. 17, 21). In contrast, the consistent presence of eosinophilic infiltrates admixed with the peripheral lymphocytic infiltrates leaves concern that these nodules may be a result of either previous migrating parasites or a hypersensitivity reaction. In particular, in 1 of the 2 horses (No. 4) with these nodules, there were intense focal parenchymal and interlobular eosinophilic infiltrates including a single eosinophilic granuloma adjacent to some of the nodules that could be due to previous larval parasite migration.
Pulmonary fibrosis occurs in horses with other diseases or subsequent to focal tissue injury. Multinodular pulmonary fibrosis has been reported in association with equine herpes-5 (EHV5) infection, where the salient features consist of alveolar septal fibrosis with preservation of an alveolar-like architecture, epithelial hyperplasia, airway/alveolar infiltrates of neutrophils and macrophages, and rare macrophage intranuclear inclusions. 54 None of these features were the primary feature of the 3 pulmonary lesion patterns in the current study. Only alveolar septal fibrosis, with epithelial hyperplasia and alveolar luminal macrophage/neutrophil infiltrates, was observed focally in more severely affected horse lungs when the multifocal fibrosing granulomatous inflammation expanded from around bronchioles into the parenchyma. Although there appeared to be a direct association between the presence of particulate-laden macrophages and foci of alveolar fibrosis in the current study, it is also possible that EHV5 could be associated with the expanded foci of alveolar inflammation and fibrosis. Interlobular fibrosis in the absence of crystals has been noted in racehorses with a history of exercised-induced pulmonary hemorrhage. 53 None of the horses in the current study were racehorses or had known histories of epistaxis. Furthermore, in the current study, fibrosis was colocalized with crystals.
In contrast to the relative absence of classic silicotic nodule formation in the equine lung, the dominant lesion pattern seen in the thoracic lymph nodes (silicotic fibronodular lymphadenitis) was very similar in appearance to the description of classic silicotic nodules 47 or silicosclerohyaline nodules as described for extrapulmonary silicosis in humans. 48 Furthermore, small numbers of clear crystalline particulates compatible with silicates were consistently seen in the centers of these nodules. Collectively, lymph nodes with this fibronodular change appeared to progressively become more enlarged, fibrotic, and calcified and were ultimately effaced by the coalescing nodules. The progressive parenchymal lymph node effacement appeared consistent with the proposed, self-perpetuating, stepwise development of silicosclerohyaline nodules in people. 48 These nodules have occasionally been found in thoracic lymph nodes of humans with no detectable clinical pulmonary silicosis. Similarly, we found that in some affected horses, the degree of lymph node destruction and enlargement seemed out of proportion to the severity of the lung lesions.
It is hypothesized that lymph node silicosis with fibrosis may be an important factor in, and may precede the development of, pulmonary silicosis because lymph node fibrosis impairs clearance of crystals from lung parenchyma. 12,36 This hypothesis of impaired clearance might explain the development of focal interlobular pulmonary fibrosis in only some of the affected horses. Even though the correlations were not statistically significant, 4 of the 5 horses with severe (grade 3) silicotic fibronodular lymphadenitis in tracheobronchial and mediastinal lymph nodes also had severe (grade 3) dissecting interlobular fibrosis with macrophage particulates in the lung. Lesion distribution may be associated with efferent lymphatic obstruction by silicate-laden macrophages trafficking towards tracheobronchiolar lymph nodes. 37 An extension of this concept might also explain the extrapulmonary lesions in cranial abdominal lymph nodes and liver of some affected horses. Similar extrapulmonary silicotic lesions in humans have been attributed to retrograde migration of silica-laden macrophages from the tracheobronchial lymph nodes to the lymph nodes below the diaphragm. The extrathoracic lesions in this study were similarly only seen in horses with the silicotic fibronodular lymphadenitis formation in tracheobronchial lymph nodes. 48
The mechanism of disease in human chronic, simple silicosis is well documented. Disseminated, airway-oriented, granulomatous inflammation is accompanied by variable fibrosis in the lung, and/or thoracic lymph nodes. 1,12 Inhalation of fibrogenic SiO2 or silicate (SiO2 mixed with other cations) crystals initiates inflammation. Crystals that reach the terminal airways are either cleared by mucociliary transport through the bronchial tree or phagocytized by alveolar macrophages to remain resident in the lung or be transported through lymphatic channels that follow the airway tree or arteries and veins to regional lymph nodes. 26,37 Crystal-laden macrophages apoptose 7,44 and release the crystals back into the adjacent (eg, lung, lymph node) tissue to be engulfed by other macrophages. Once activated through crystal phagocytosis, the macrophages release inflammatory mediators (eg, interleukin [IL]-1, tumor necrosis factor [TNF]-α), reactive oxygen species, and arachidonic acid metabolites. 25,44 Mediators recruit additional macrophages and B and T lymphocytes to the region. 33 Chronic T-lymphocyte activation preferentially shifts toward a T-helper 1 response that leads to an upregulation of interferon-γ and thus continued macrophage stimulation as well as stimulation of fibroblasts to secrete collagen. 21,22 Activated macrophages and lymphocytes are amplified in the alveolar and interstitial spaces stimulating chronic inflammation and fibrosis. Occasionally, extrapulmonary dissemination occurs resulting in silicotic nodules in the liver (periportal), spleen, and bone marrow due to the migration of silica-laden macrophages 18,47,48 as well as occasional glomerulonephritis thought to be indirectly related to silicosis. 48
Development and severity of silicosis in humans depend on the type and amount of dust retained, the duration of exposure, individual susceptibility, and the nature and property of the crystals. 3,20 High exposure to cytotoxic crystals causes more rapid and progressive disease, low exposure over a long time causes chronic disease, and low exposure over short time causes reversible inflammation. 52 Individual susceptibility to disease appears to be mostly related to alveolar retention and dust clearance. 3 Horses with SAO appear to be similar to people with silicosis in that not all exposed individuals develop clinical or pathological disease.
Many types of silica particles, including pure SiO2 crystals and mixed silicates, can cause silicosis. Seven polymorphs of SiO2 including quartz, cristobalite, and tridymite, have been shown to be pathogenic and are associated with pulmonary silicosis in people 9,39,44 and animals. 45 The different silica polymorphs have different pulmonary clearance rates with cristobalite more difficult to clear than quartz, 27 and different fibrogenic potentials, with tridymite greater than cristobalite, cristobalite greater than quartz, and quartz greater than mixed silicate crystals. 9,30,55 Pathogenicity of the crystals may be greater in newly exposed silica (ie, silicon recently exposed to air) because of a higher redox potential of the newly exposed crystals. 3,51 Anecdotal reports from practitioners that development of clinical disease has followed excavation of horse corrals in endemic regions supports newly exposed silica as a possible factor in SAO.
Geographic clustering of affected horses in regions containing soil with cytotoxic crystals is consistent with pneumoconiosis. Cristobalite, tridymite, and quartz were recovered from soils from endemic farms and from lung and/or lymph node tissues from all but 1 affected horse. Quartz was also recovered from the lymph node of 1 unaffected horse and from soil samples from unaffected horse farms in California but not in the soil sample from Nevada. This was not surprising since quartz is the most common free silica polymorph found in the environment. 39
Given the small sample size of the study and the smaller sample size of recovered crystals from tissues, it was not possible to assess statistical associations between crystal type and severity of pathology. However, the dominant crystals in endemic farm soils were also the dominant crystals in lung and lymph node tissues of horses living on those farms. Consequently, environmental exposure is likely linked to lung and lymph node pathology. The actual cellular mechanisms that cause pulmonary silicosis subsequent to inhalation of silica crystals are still unclear. People respond differently to crystal exposure, even under the same environmental conditions. 14 Similarly, not all horses living on an endemic property may develop clinical disease. Dust-laden macrophages in thoracic lymph nodes are a common incidental finding, particularly in animals living in a dusty environment, and are compatible with incidental pneumoconiosis. The pneumoconiosis in the unaffected horse (No. 10) was likely incidental because of the lack of reactive fibrosis in the lungs and regional lymph nodes. The reasons for the development of the reactive fibrosis in certain individuals are unknown, but given that the pathogenesis of pulmonary disease in horses with SAO parallels that of human pulmonary silicosis, the severity of equine pulmonary disease is likely related to exposure duration, dust retention, individual susceptibility, and type of crystal inhaled. Given a similar inhalation exposure, cristobalite and tridymite could be more likely to induce disease than quartz because of higher fibrogenic potentials and lower clearance rates. Because of a disparity in the development or degree of clinical disease among cohorts of horses on the same property (personal observation), it is likely that unknown idiosyncratic factors may predispose some horses to the development of clinical pulmonary silicosis.
Potential Mechanisms of Pathogenesis
The pathogenesis of the osteoporosis in SAO is still unknown. Potential mechanisms include hyperparathyroidism, an animal form of PDB, an inflammatory-mediated osteoporosis secondary to chronic silicate-induced inflammation in the lung and lymph nodes, and even possibly a direct effect of silicate-laden macrophages disseminated to the bone marrow. The first 3 of these mechanisms have been reported as causes of osteoporosis in other diseases by affecting the bone resorption–formation balance through the nuclear factor-κB ligand (RANKL), the RANKL receptor (RANK), and the decoy receptor, osteoprotegrin (OPG) system. 5,8,10,28,41 –43,48,50
Primary hyperparathyroidism is rare in horses. More commonly, low serum calcium or high serum phosphate induces secretion of PTH and secondary hyperparathyroidism. Hyperphosphatemia was observed in some SAO-affected horses, but there was no physical evidence of hyperparathyroidism based on size or histomorphology of the parathyroids, and thus the cause for the hyperphosphatemia is unknown. The elevated phosphate and/or the hypovitaminosis D 19 seen in some SAO affected horses may induce elevation of PTH. 27 Chronic elevations in PTH lead to coupled bone resorption and formation with a net bone loss. Circulating PTH induces the release of RANKL from stromal cells and osteoblasts while simultaneously decreasing the release of the decoy OPG receptor by osteoblasts, stromal cells, and bone lining cells. The RANKL binds to the RANK receptor on osteoclast precursors and on osteoclasts, causing osteoclastogenesis and increased osteoclastic activity. Continuous elevated PTH levels also lead to an increase in the number of active, bone forming osteoblasts. 5 Hyperparathyroidism occurs in 12%–18% of PDB patients. Hyperparathyroidism is believed to be secondary to the increased calcium requirement generated by the increased bone formation in PDB. 23,40 Thus, the hyperparathyroidism in equine SAO could also be secondary to increased bone formation, occurring later in the disease process.
Although SAO and PDB appear to have some fundamental differences (age of onset, persistence of osteoclastic activity, and disease progression), the histological appearance and lesion distribution within the skeleton indicate potential shared mechanisms of disease. The etiopathogenesis of PDB is unknown; however, PDB is considered a disease of osteoclastic regulation and is characterized by an increase in both osteoclastic number and size. A proposed mechanism for Paget disease includes altered regulation of the nuclear factor-κB activation through IL-1 and TNFα, causing upregulation of RANKL signaling and leading to increased osteoclastogenesis. 42 The bone formation response is disorganized, producing bone of poor quality that has a tendency to fracture or deform. Proposed causes for these mechanisms include genetic mutation, virus, or toxic environmental exposure. 13,34
Pulmonary inflammation and osteoporosis are both features of equine SAO. Osteoporosis is recognized as a comorbidity of chronic pulmonary inflammatory diseases in people, including chronic obstructive pulmonary disease 6,29 and cystic fibrosis. 11,49 Although the cause of osteoporosis in these human patients is multifactorial (ie, long-term corticosteroid use, decreased exercise, hypoxia, decreased calcium absorption, altered lifestyle decreasing exposure to sunlight and therefore compromising vitamin D synthesis), the chronic pulmonary inflammatory disease itself appears to have an important role in the development of systemic osteoporosis. As a chronic inflammatory disease, pulmonary silicosis is known to release inflammatory cytokines including IL-1, IL-6, and TNFα as well as activate T lymphocytes 22,44 that may be releasing soluble RANKL, all of which might initiate osteoclastogenesis and osteoclastic activity. Thus, the osteoporosis in SAO may be secondary to the pulmonary and/or lymph node inflammation. This possible etiopathogenesis of SAO is supported by the statistical correlations between the tracheobronchial lymph node nodular histology pattern, the presence of cytotoxic crystals in the tracheobronchial lymph nodes, the lung silicon levels, and the severity of bone disease or presence of osteoporosis. In particular, the presence of the silicosclerohyaline nodule formation in tracheobronchial lymph nodes was significantly correlated with osteoporosis.
It is also possible that undetected silicate particles or silicate-laden macrophages disseminated to bone tissues and served as nidi for bone lesions. Particulate-laden macrophages were observed in hepatic, renal, splenic, ileal, and colonic tissues of some affected horses; indicating that silicates are capable of accessing extrathoracic sites. Silicotic lesions have been seen in the bone marrow of humans. 48 In the current study, the loculated pattern of osteolytic foci observed on bone radiographs is consistent with the concept of multiple coalescing lesions, each emanating from a central nidus. Silicates or particulate-laden macrophages were not detected in bone tissues in the current study, although detection of silicates in bone tissue is inherently difficult because of bone’s mineralized matrix. We were unable to recover any crystalline material from bone tissue through acid dissolution for either the silicon level assay or X-ray diffraction.
Conclusion
Pulmonary inflammation and osteoporosis coexisted in horses with systemic bone fragility. It is unknown whether all horses with silicosis develop osteoporosis. Osteoporosis was characterized by extensive bone remodeling. Bone resorption by large, supernumerary nucleated osteoclasts was excessive, randomly located, and inconsistent with the expected load environment. The severity of osteoporosis most closely correlated with the presence of cytotoxic crystals in the tracheobronchial lymph nodes. Although the features of the disease share some similarities with human hyperparathyroidism and PDB, the etiopathogenetic mechanism is unknown. Circumstantial evidence supports a cytotoxic silicate inflammatory pneumoconiosis-induced osteoporosis, possibly through the generation of inflammatory cytokines that stimulate osteoclastogenesis. Not all horses on endemic farms are severely affected. Genetic predisposition, immunologic competency, behavioral patterns, and exposure circumstances likely affect the development of both silicosis and SAO. Further investigation of SAO may elucidate pathogenetic mechanisms that play a role in other comorbid pulmonary–bone syndromes.
Additional tables and figures can be found online as supplemental files and include Table 4, signalment, antemortem clinical signs and diagnostic tests; Table 5, histopathological grading schemes; Table 6, Spearman correlations; and Figure 24, XRD of soil, lung, and lymph node sample from a second horse.
Footnotes
Acknowledgements
We thank Drs. Mathew Durham and Coral Armstrong for their contributions and Tanya Garcia-Nolan, Dr. Anja Welihozkiy, DVM, Melissa Garrick, Larry Melton, and Ian Holser for their technical assistance and Professor Kenneth Vecchio for the use of his laboratory at UC San Diego.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
This project was supported by the National Institute of Health Services, Ruth L Kirschstein National Service Research Award (5-T32-ES0755-33), and the Center for Equine Health (05-06) with funds provided by the State of California parimutuel fund and contributions by private donors.
