Abstract
A 6-y-old female dromedary camel (Camelus dromedarius L.) was presented for assessment of firm, bilateral swellings rostral and ventral to the eyes. Serum biochemistry revealed hyperglycemia (28.5 mmol/L), hypocalcemia (1.27 mmol/L), hyperphosphatemia (3.39 mmol/L), hypoproteinemia (total protein 50 g/L), and hypoalbuminemia (20 g/L). Based on the poor prognosis associated with the presumptive diagnosis of fibrous osteodystrophy, the camel was euthanized. Gross postmortem findings revealed expanded fibrous tissue replacing the maxilla and mandible, and bilaterally prominent parathyroid glands. Histology of the maxilla revealed proliferative loose fibrous tissue with widely scattered, regularly spaced, small spicules of mineralized bone. The parathyroid glands were prominent bilaterally; the internal and external parathyroid glands were composed of plump cells with abundant pale basophilic cytoplasm and open nuclei. The pathologic findings were consistent with the antemortem diagnosis of fibrous osteodystrophy. The camel’s diet, which was not specifically balanced for a camel, included grass hay, sweet feed, and alfalfa pellets. The camel’s caregivers reported feeding her treats of cookies. A feed analysis was not available. The biochemistry abnormalities and clinical and postmortem findings, along with a diet that was not balanced for a camel, are consistent with a diagnosis of nutritional secondary hyperparathyroidism.
Parathyroid hormone (PTH) plays a critical role in maintaining calcium, phosphate, and vitamin D homeostasis in the healthy animal. In response to low serum calcium, PTH is released and subsequently raises serum calcium concentration by increasing reabsorption of calcium from bone, the renal system, and the small intestine through the actions of vitamin D.2–4,11,12,15–17,19 This absorption and reabsorption of calcium raises serum calcium to within a normal range. Prolonged dysregulation of PTH concentration results in serious, potentially fatal problems, and can be caused by secretory primary tumors of the parathyroid glands, insufficient dietary calcium, or other metabolic derangements of the calcium–PTH–vitamin D axis, such as vitamin D deficiency.1–4,10–12,15–17,19
Nutritional secondary hyperparathyroidism leading to fibrous osteodystrophy is a well-documented condition in domestic species, including small ruminants and horses. It is characterized by increased PTH and active vitamin D in response to an imbalanced dietary calcium-to-phosphorus ratio or excessive consumption of calcium-binding oxalates in feed.1,4,17 In response to increased PTH and vitamin D, calcium and phosphorus are reabsorbed from the gastrointestinal tract and bones. Mineralized bone is replaced with proliferative fibrous tissue, a process called fibrous osteodystrophy.1,12,17 Clinicopathologic abnormalities may include increased serum phosphorus, PTH, and alkaline phosphatase, decreased serum calcium, and reduced serum Ca:P ratio.12,17 Nutritional secondary hyperparathyroidism is primarily differentiated from primary disease through dietary history and blood calcium and phosphorus concentrations.7,13
In renal secondary hyperparathyroidism, decreased renal function leads to the retention of phosphate and reduced calcitriol synthesis.13,15 Worsening hypocalcemia and glomerular filtration rate subsequently lead to PTH elevation.13,15 Skeletal lesions in renal secondary hyperparathyroidism range from mild to severe fibrous osteodystrophy and have a similar distribution to the lesions of nutritional secondary hyperparathyroidism, with cancellous bones of the skull more readily affected, whereas limb bones are generally spared.13,15 Differentiation from nutritional secondary hyperparathyroidism relies largely on nutritional history and laboratory findings of renal insufficiency. Renal secondary hyperparathyroidism is seen most commonly in dogs, less commonly in cats, and is considered rare in other species. 15 It has not been described in horses, to our knowledge.1,13
In horses and small ruminants, nutritional secondary hyperparathyroidism causes gross swelling of the high-turnover facial bones that result in a “big head” appearance.1,12,17 “Big head disease” or “bran disease” in horses has been associated with feedstuffs high in phosphorus and low in calcium, such as bran and sweet feed.7,13 Diets with phosphorus concentrations at least double that of calcium predispose horses to this disease.13,14 It is important that the basis of the equine diet be good quality hay; exclusively grain diets lead to improper calcium intake, with reported Ca:P ratios as low as 1:2. 7 The recommended Ca:P ratio for horses is 2:1. 7
Although fibrous osteodystrophy has not been documented extensively in dromedary camels specifically, metabolic derangements of the calcium–phosphorus–PTH–vitamin D axis have been well-studied in camelids in the Middle East given their cultural and economic significance.2–4,6,8–11,16 Rickets and osteomalacia are notable concerns in llamas, alpacas, and Bactrian camels (Camelus bactrianus L.) when kept in temperate climates with low levels of solar radiation or during periods of drought.10,16 Compared to domestic ruminants and women, the physiology and evolutionary history of the dromedary camel has led to innately higher basal concentrations of circulating calcitriol, PTH, PTH-related peptide (PTHrp), and vitamin D.4,6,9 During times of drought, average circulating PTH levels in dromedary camels increase dramatically.6,9 Growing camels have a calcium intake requirement of 10–30 g/d, depending on body size. 18 These species-specific endocrine idiosyncrasies have led to the suggestion that dromedary camels kept in regions of the world with relatively little sunlight might benefit from dietary vitamin D supplementation, particularly during the winter when they experience seasonal decreases in serum vitamin D concentration.3,4,6 It has been proposed by some authors that many bone disorders in camelids are a result of vitamin D deficiency and can be remedied with proper nutritional balance of calcium, phosphorus, and vitamin D. 4
Despite relatively abundant literature on phosphocalcic metabolism in dromedary camels, there are only 2 published case series on fibrous osteodystrophy in dromedary camels. Neither report provides detailed histologic findings or assesses the parathyroid glands.2,11 All 6 camels in these reports were female, 2–12 y old.2,11 Four of 6 camels were euthanized because of poor prognosis.2,11 It is unclear whether the all-female representation of cases is the result of sex-related differences in phosphocalcic metabolism, the possibility of which is represented by significantly higher concentrations of calcitonin and PTH in female dromedary camels. 6 We report herein the gross and histopathologic findings of this case to contribute to the limited literature on this subject, and to document the associated lesions and histology.
A 6-y-old female dromedary camel was presented to the Veterinary Teaching Hospital (VTH) at the Virginia-Maryland College of Veterinary Medicine (Blacksburg, VA) for evaluation of rapidly enlarging bilateral facial swellings of 3–4-wk duration. This camel was born on the farm and was reported to be diminutive in stature from birth. She was housed separately on a grass lot with a stream and received special treatment. In addition to her regular diet of grass hay, alfalfa pellets, and sweet feed formulated for horses, she also received treats of cookies from the grocery store. In association with the facial swellings, she had a history of increased respiratory effort, anorexia, weight loss, lethargy, and a change to a less friendly personality. When the swellings were first noticed, they were small and focal, and the referring veterinarian prescribed dexamethasone to reduce swelling. When the swellings increased rapidly in size, skull radiographs were taken, and a fine-needle aspirate performed. The referring veterinarian noted malalignment of the cheek teeth radiographically and possible mitotic figures on the fine-needle aspirate. The camel was administered oxytetracycline and penicillin because one differential diagnosis was an abscess, and she was referred to the VTH for further evaluation.
On presentation at the VTH, the camel was alert but dyspneic with increased effort and inspiratory wheezing appreciated on tracheal auscultation. Firm, bilaterally symmetrical facial swellings in the maxillary region that were painful on palpation were noted. Other findings included bilateral ocular and nasal discharge, evidence of loose, watery stool on her hindquarters, and abrasions on the dorsal aspect of her carpi and stifles, consistent with increased recumbency.
Skull radiography revealed severe swelling of the maxilla with soft tissue opacity present bilaterally at the level of all maxillary premolar teeth and approximately the first molar tooth. The swelling protruded dorsally into the nasal canal and laterally to the external aspect of the maxilla. Thinning of the maxillary cortices was present around the swelling. All maxillary teeth were displaced and abnormally positioned throughout the expansion, creating the appearance of “floating” teeth. Mild bilateral expansion of the mandible with soft tissue opacity was also noted just rostral to the mandibular premolars, with cortical thinning of the mandible. The rostral mandibular premolars were also displaced. No significant abnormalities were identified on the complete blood count. Serum chemistry revealed hyperglycemia (28.5 mmol/L), hypocalcemia (1.27 mmol/L), hyperphosphatemia (3.39 mmol/L), hypoproteinemia (total protein 50 g/L), and hypoalbuminemia (20 g/L; Table 1). 5
Serum biochemistry results for a dromedary camel patient with clinical signs of fibrous osteodystrophy. 5
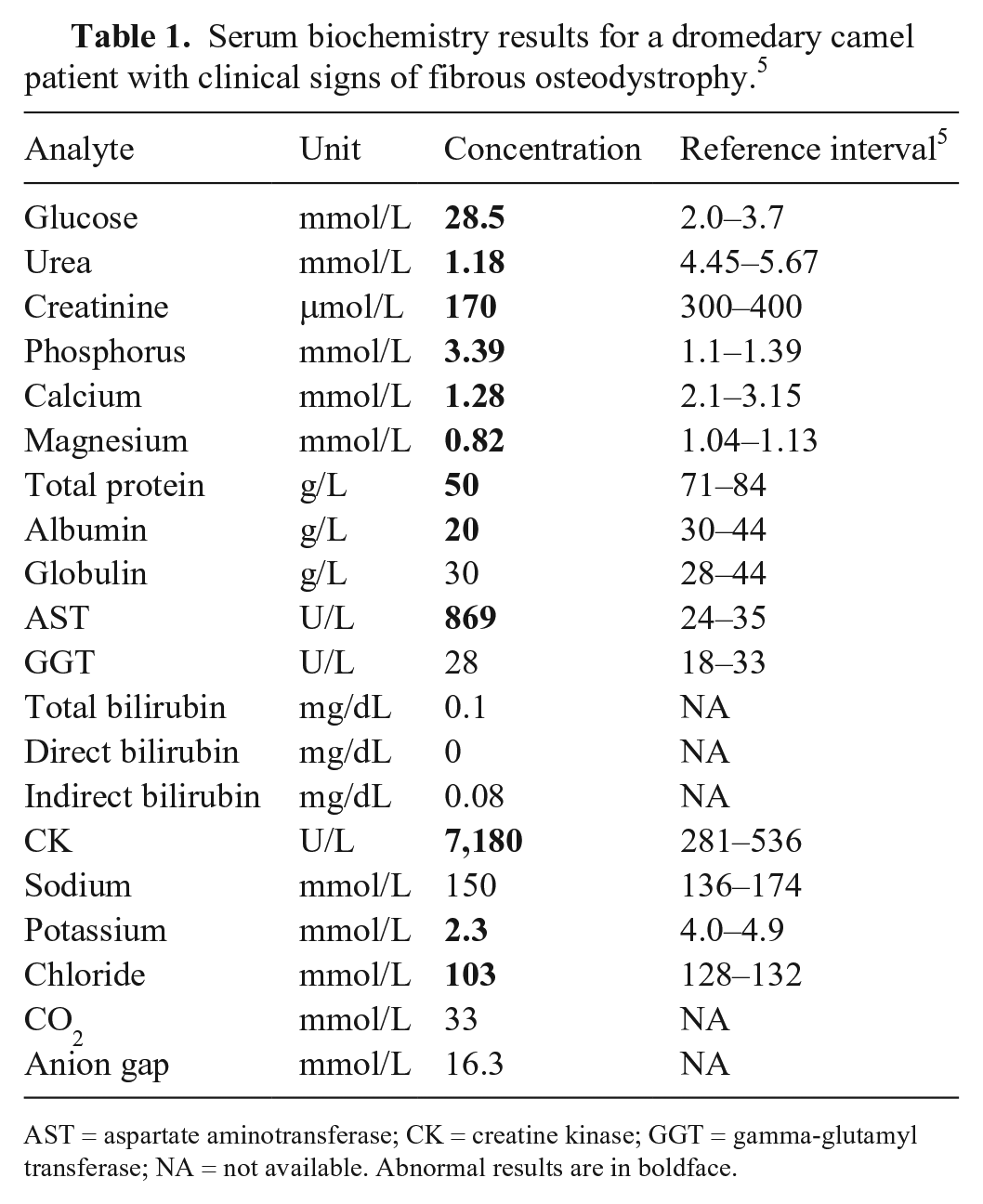
AST = aspartate aminotransferase; CK = creatine kinase; GGT = gamma-glutamyl transferase; NA = not available. Abnormal results are in boldface.
Differential diagnoses based on the history prior to presentation included neoplasia, given the referring veterinarian’s report of mitotic figures, and bacterial or fungal abscess. However, based on the physical examination findings of symmetrical maxillary enlargement and serum chemistry abnormalities, a tentative antemortem diagnosis of fibrous osteodystrophy was made. The prognosis for successful treatment was estimated to be grave given the advanced nature of the problem, and based on discussion with the owner, euthanasia was elected, and a postmortem examination performed.
Grossly, upon sectioning, the maxilla and mandible were largely replaced by symmetrical proliferative fibrous tissue, and the teeth were loose (Fig. 1). A solitary 3-cm firm tan mass was identified in the wall of the distal jejunum, with ulceration of the luminal surface. The parathyroid glands were relatively prominent (8 mm diameter) and estimated to be approximately double the expected size. The jejunal mass, parathyroid glands, and samples of the maxilla were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin.
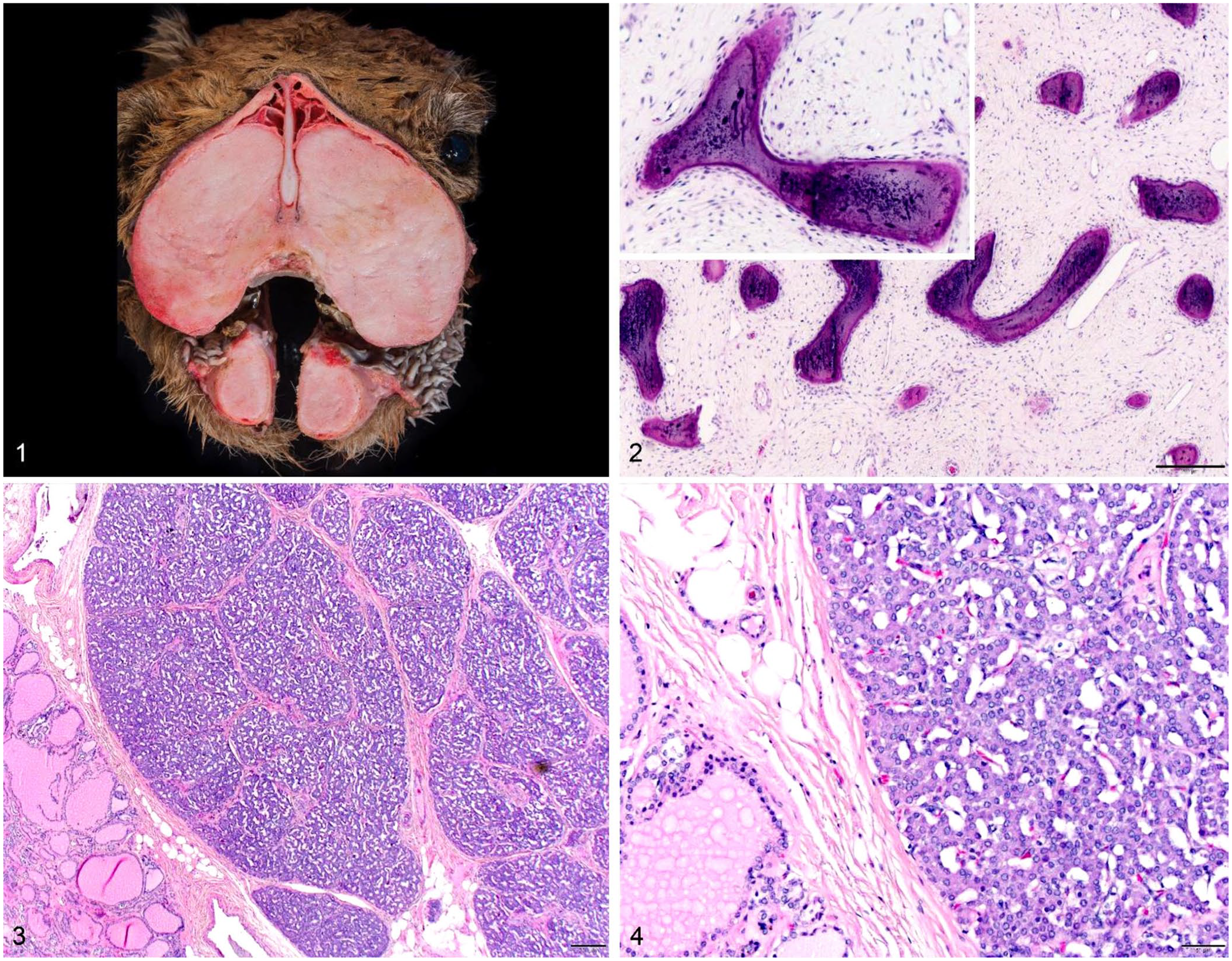
Fibrous osteodystrophy in a dromedary camel.
Microscopically, loose proliferative fibrous tissue with widely scattered, regularly spaced, small spicules of mineralized bone were present in the maxilla (Fig. 2). Osteoclasts were rare, indicating minimal ongoing bone resorption in the bones of the head. A diagnosis of chronic fibrous osteodystrophy was made. The cells of the internal parathyroid glands were diffusely and uniformly plump, with abundant pale basophilic cytoplasm and open nuclei (Figs. 3, 4). Parathyroid glands are normally composed of chief, oxyphil, and water-clear cells. In our case, the glands were uniformly chief cells, consistent with active production and secretion of PTH. There was no compression of adjacent tissues. The external parathyroid glands had a similar histologic appearance, with a central duct lined by cuboidal epithelium and surrounded by moderate lymphocytic infiltration. A more complex superficial duct was noted, with occasionally keratinized squamous lining and lymphocytic and macrophage infiltration. These changes are consistent with a diagnosis of chronic diffuse parathyroid hyperplasia.
The jejunal mass extended to the mucosa, which was compressed over it but with only a few areas of ulceration. The mass was composed of fusiform cells in a scant stroma, consistent with a diagnosis of gastrointestinal stromal tumor. Portal areas of the liver had minimal fibrosis; periportal areas were obscured by dense, mature fibrous tissue. The periphery of central veins was also densely fibrotic, extending minimally into surrounding tissue. Hepatocytes were swollen and vacuolated. These changes were nonspecific, and a diagnosis of chronic hepatic fibrosis secondary to degenerative hepatopathy was made. The lack of inflammation is most consistent with a non-infectious etiology. The lungs had delicate fibrous tags along the pleural surfaces adjacent to the body wall, most concentrated on the diaphragmatic aspect and also involving the parietal pleura of the diaphragm. The kidneys were normal on gross examination; autolysis unfortunately interfered with microscopic evaluation; however, no obvious or severe changes were observed. Sections of the thyroid gland, lymph nodes, and heart were unremarkable.
The gross and microscopic findings confirmed the clinical diagnosis of nutritional secondary hyperparathyroidism leading to severe fibrous osteodystrophy. This is the second time this condition has been documented in this species in North America. 2 Unfortunately, a feed analysis was not available for the diet being fed this camel. However, a dietary cause for the development of fibrous osteodystrophy must be considered in this case given the high ratio of sweet feed to hay in the diet, the history of feeding human cookie treats, and the fact that the diet was not formulated for this species. A diagnosis of renal secondary hyperparathyroidism can be ruled out by the lack of clinicopathologic or postmortem evidence of renal disease; the microscopic findings are inconsistent with primary hyperparathyroidism. The clinical presentation and postmortem findings support a diagnosis of nutritional secondary hyperparathyroidism leading to fibrous osteodystrophy.
Footnotes
Acknowledgements
We thank Robert Kresslein for his help in creating Figures 1–4.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
