Abstract
A 2-year-old Red Holstein cow was presented with uterine torsion at 235 days of pregnancy. The fetus extracted by cesarean section had weak vital signs and marked abdominal distention. An edematous pouch that contained tubular structures with peristaltic activity was associated with the umbilical cord. Because of poor prognosis, both dam and fetus were euthanized. At necropsy, the fetus had severe distention of the forestomachs, abomasum, and proximal small intestine; absence of distal small intestine, cecum, and proximal colon; atresia of the 2 blind ends of the intestine; and atrophy of distal colon and rectum. The tubular structures associated with the umbilical cord were identified as the segments of intestine that were absent in the fetus. Intestinal atresia combined with ectopia may be caused by local ischemia during temporary herniation and rotation of the fetal gut into the extraembryonic coelom. The close connection between ectopic intestine and amniotic sheath of the umbilical cord in this case may have facilitated vascularization and allowed development and viability of the ectopic intestine.
Segmental anomalies of the bovine intestine may be categorized as stenosis (partial occlusion of the intestinal lumen) or atresia (complete occlusion of the intestinal lumen). Atresia can be subdivided into types I through IV. In membrane atresia (type I), the intestinal lumen is obstructed only by a thin membrane. In cord atresia (type II), a segment of the intestine is reduced to a cord of muscle or connective tissue. An intestinal section is completely missing in blind-end atresia (type IIIa), leaving 2 blind ends and a V-shaped mesenteric defect, whereas in Christmas tree or apple peel atresia (type IIIb), the distal end of the intestine is wrapped around a single perfusing vessel. 16 Several of the defects mentioned above are combined in type IV atresia (multiple atresias). 4 The most common segmental anomaly in the bovine intestine is atresia coli, followed in decreasing order of frequency by atresia jejuni and atresia ilei. 16 Duodenal atresia is thought to be caused by the failure of luminal recanalization after normal deposition of multiple layers of epithelium. 6,16 Atresia of other parts of the small and large intestine is thought to be caused by local ischemia during embryonic development. Vascular compromise during the extracorporeal rotation of the intestine around the third to fourth week of gestation may impair blood supply to a segment. 6,16 Interruption of the blood supply during embryogenesis has been shown to cause intestinal atresia in lambs 3 and chickens. 15 Ness and coworkers 12 postulated that transrectal palpation of the amniotic sac before day 40 of gestation may result in intestinal atresia in calves. In contrast, palpation of the allantochorion (slipping of the fetal membranes) does not appear to affect the incidence of intestinal atresia; this procedure is usually carried out after day 40 of gestation and does not involve the embryo itself. 12
Genetic factors may also be involved in the etiopathogenesis of intestinal atresia. Atresia jejuni in Jersey cattle and atresia ilei in Swedish Highland cattle are inherited as autosomal recessive traits. 2 Jubb 7 reported a possible predisposition for intestinal atresia in the Holstein-Friesian breed, and when Holstein cows and bulls that had produced affected calves were bred to each other, a higher proportion of calves had intestinal atresia compared with the general cattle population. 14 However, in another study, no familial connections among affected animals were found. 16 Intestinal atresia in Brazilian ruminants has been attributed to consumption of plants of the genus Mimosa. 13
We report a unique form of intestinal atresia in a Red Holstein fetus. To the best of our knowledge, no case of intestinal atresia with extrafetal ectopia has been reported.
Case History
A 2-year-old Red Holstein cow (first-calf heifer) was referred to the Ruminant Clinic at the Vetsuisse Faculty of the University of Berne on day 235 of pregnancy because of abdominal colic that had started the previous day. Uterine torsion had been diagnosed by the referring veterinarian. Reportedly, pregnancy had been confirmed by transrectal palpation on day 42 after artificial insemination. The animal was housed in a free stall with other pregnant heifers and fed a hay-based ration. None of the other heifers had any signs of illness or abnormal pregnancy.
Upon physical examination, the abdomen was bilaterally distended. Counterclockwise (as viewed from the caudal aspect) uterine torsion was detected by transrectal palpation. The cervix was minimally dilated (3 cm), and a dark red, thick-walled portion of fetal membranes protruded into the vagina, which contained several blood clots. Surgical correction of the uterine torsion was attempted because the incomplete dilation of the cervix precluded manual correction per vaginam, and the rolling technique was considered too stressful for the animal. At laparatomy, the gravid uterus was found to be rotated 180° counterclockwise immediately cranial to the cervix. The left uterine horn, which contained the fetus, was distended and taut, filling the ventral abdomen. The torsion could not be corrected, so the dam was euthanatized after the fetus was removed by hysterotomy. The female fetus was underdeveloped, with weak vital signs and a severely distended abdomen (Fig. 1). It was immediately euthanatized.
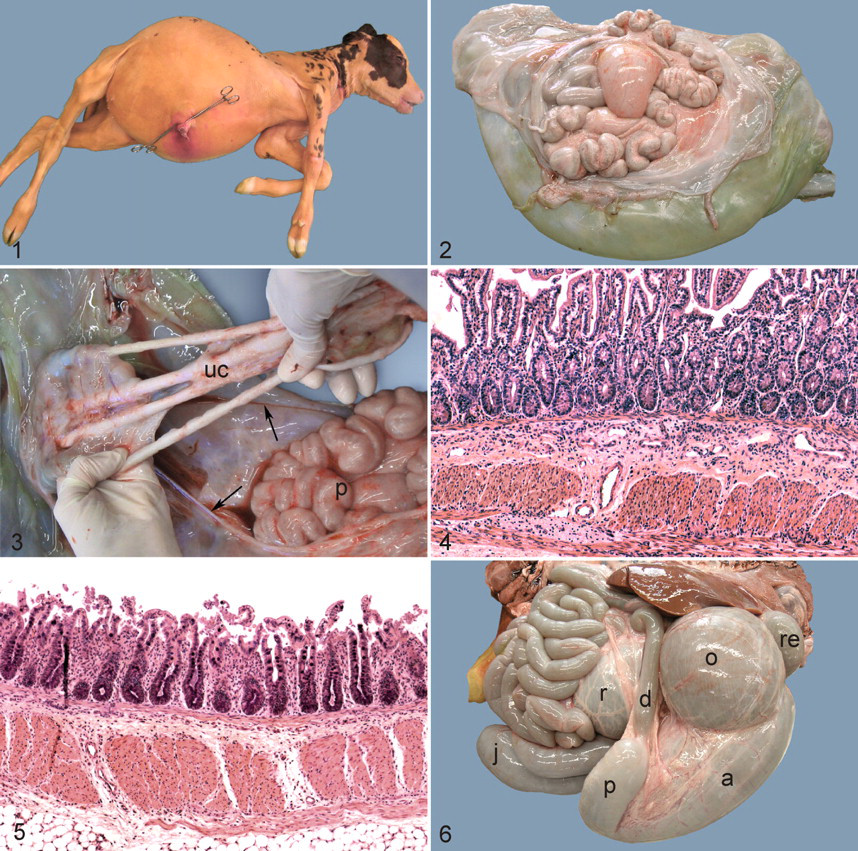
Female bovine fetus at 235 days gestation. The umbilical cord has been excised. Note marked abdominal distention and normal closure of the abdominal wall.
Necropsy Findings
The wall of the cow’s uterus, cranial to the site of torsion, was hemorrhagic. Within the uterine lumen, an edematous pouch, approximately 30 cm in diameter, was connected to the fetal membranes at the umbilical cord. The pouch contained tubular structures (Fig. 2) with peristaltic activity. These structures were extensively adhered to the lining of the pouch; the outer layer of the pouch was continuous with the amniotic sheath of the umbilical cord (Fig. 3). The tubular structures and the lining of the pouch had a smooth surface, typical of peritoneum.
Selected tissues were fixed in 10% neutral buffered formalin and processed routinely for histologic examination. Paraffin sections were stained with hematoxylin and eosin (HE). Histologically, the tubular structures within the pouch were identified as well-differentiated jejunum, ileum, cecum, and colon. All intestinal layers from the mucosa to the tunica muscularis, as well as submucosal and myenteric plexuses, were present and well-differentiated (Figs. 4 and 5).
The crown–rump length of the fetus was 80 cm. The forestomachs, abomasum, and proximal small intestine were severely distended (Fig. 6) by abundant yellowish fluid, causing pronounced abdominal enlargement. The jejunum ended blindly after approximately three quarters of its expected length. The distal jejunum, the ileum, the cecum, and the proximal colon, as well as the corresponding parts of the mesentery and mesenteric vasculature, were absent. The remaining colon and rectum were approximately 20 cm long and atrophic. All other abdominal organs, including the umbilicus and abdominal wall, were normal.
Discussion
Most cases of bovine intestinal atresia have been reported in stillborn or neonatal calves. 8,9 However, intestinal atresia with massive abdominal distension in bovine fetuses can result in dystocia. 8 The uterine torsion in this case was probably secondary to the massive fluid accumulation in the fetal forestomachs, abomasum, and small intestine orad to the site of atresia. Cases of bovine intestinal atresia of comparable severity have been reported. 8 However, to our knowledge, ectopia of the missing intestine has not been described.
The gastrointestinal tract forms during lateral body folding and involves the incorporation of the dorsal portion of the yolk sac into the embryo. The embryonic gut is divided into foregut (from the oropharyngeal membrane to the duodenal papilla), midgut (from the duodenal papilla to the middle transverse colon), and hindgut (from the middle colon to the anus). The midgut is connected to the yolk sac by the vitello-intestinal duct. In ruminants, the yolk sac does not provide nutrition for the embryo. Because a choriovitelline placenta does not develop in the bovine fetus, the yolk sac atrophies rapidly to form the vestigial umbilical vesicle. The vitello-intestinal duct becomes the omphaloenteric duct, which is located within the umbilical cord. Although nutritionally irrelevant for the embryo, the yolk sac is crucial for intestinal vascular development. The right vitelline artery gives rise to the cranial mesenteric artery, and the vitelline veins constitute the basis for the hepatic sinusoids. 10
During the third to fourth week of gestation, the loop formed by the midgut increases rapidly in length and outgrows the space available in the abdominal cavity. Consequentially, a reversible physiological umbilical herniation of the fetal gut into the extraembryonic coelom of the umbilical cord occurs; this extraembryonic space is called the umbilical sac. Simultaneously, the midgut loop rotates about an axis formed by the cranial mesenteric artery. During midgut rotation, the omphaloenteric duct atrophies, although it may persist as an umbilical fistula, a fibrous cord with or without an enclosed cyst, or a blind pouch known as Meckel diverticulum. If the loop of the midgut fails to return to the abdominal cavity after rotation and instead remains in the umbilical sac, a so-called omphalocele, also known as an umbilical cord hernia or open umbilical hernia, develops. This type of omphalocele is usually associated with abdominal wall defects. 10
Although newborn animals with an open umbilical hernia have been referred to our clinic for surgical repair (M. Meylan, personal communication), there are few published reports on this condition in cattle. 1,11 In a rare form of anomalous vitello-intestinal duct in a human infant, a small segment of normally formed large intestine was found within the umbilical cord, 1 cm from the abdominal wall. 5 With the exception of this so-called large intestinal cyst in the umbilical cord, the child was normal and did not have, as one would have expected, an abdominal wall defect. The ectopic large intestine was embedded in Wharton jelly and had a normal histologic appearance, including a vascular connection to the tissue surrounding the umbilical cord. The authors suggested that the ectopic intestine was formed during the physiological herniation and underwent apparently normal development at the ectopic site.
This case had a combined malformation with ectopia of a major part of the intestine and atresia of the blind ends of the intra-abdominal intestine. These findings are consistent with failure of the physiologically herniated portion of the intestine to return to the abdomen during development, possibly because of adhesion to the umbilical cord. As the umbilical passage narrowed, the herniated intestine would have been disconnected from the remainder of the intestinal tract. The resultant disruption of blood supply would have caused localized ischemia, which explains the atresia. The loop of midgut that undergoes physiological herniation during fetal development consists of the aborad duodenum, jejunum, ileum, cecum, transverse and ascending colon, which essentially correspond to the ectopic intestinal segments in this bovine fetus. The normal histologic structure of the ectopic intestine indicates that the blood supply was adequate for development. This assumption is corroborated by its size and strong motility. Whether the blood supply was derived from the cranial mesenteric artery was not determined, but, considering the pathogenesis of atresia and omphalocele, this seems unlikely. We suspect that the herniated loop of midgut underwent ischemia at the points of exit from and reentry into the peritoneal cavity during abdominal wall closure, with resultant atresia at the points of ischemia. Vessels originating from the wall of the umbilical cord, having gained access to the ectopic intestinal tissue through extensive adhesions, may have compensated for disruption of the blood supply from the cranial mesenteric artery. 5 The explanation for the adhesions between the ectopic intestine and the wall of the umbilical cord is unknown. Closure of the abdominal wall despite incomplete relocation of the intestine into the abdominal cavity contrasts with the classic omphalocele, in which an abdominal wall defect persists.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
