Abstract
Mycoplasmas are host-specific commensals on mucous membranes of the genital tract, but infection and clinical disease by Mycoplasma bovis in the genital tract of cattle is not well described. In the current study, 1 aborted bovine fetus and 1 neonatal calf were examined macroscopically and histologically. For the detection of M. bovis, bacterial isolation, immunohistochemistry (IHC), and in situ hybridization (ISH) were performed. For further characterization of the inflammatory infiltrates, IHC was performed using antibodies to cluster of differentiation (CD)3, CD79a, lysozyme, L1, S-100A8, S-100A9, and von Willebrand factor VIII. Gross examination revealed a lobular consolidation of the lung. Histologically, the lungs of both animals showed an interstitial pneumonia associated with suppurative bronchopneumonia, intraalveolar multinucleated giant cells, and lymphocytic aggregates. The expression of L1, S-100A8, and S-100A9 in multinucleated giant cells supports a histiocytic origin. Mycoplasma bovis antigen was detected by IHC in brain, lung, liver, and placenta of the fetus, and M. bovis DNA was detected by ISH in various organs of the fetus, including lung and placenta and within the lung of the calf.
Keywords
Many different bacterial infections cause infertility and late-term abortion in cattle. 10 Although Ureaplasma diversum, Mycoplasma bovigenitalium, and Mycoplasma bovis are part of the normal flora of the reproductive tract, they can also cause reproductive failure. 8 Transmission can occur via direct contact, environmental contamination with infected urine, and sexual transmission. 10 Mycoplasmas are host-specific commensals on mucous membranes of the respiratory, intestinal, and genital tract and the mammary gland. 7 Infection, lesions, and clinical significance of M. bovis in genital disease of cattle are less described and include experimentally induced or naturally occurring chronic endometritis, suppurative salpingitis, infertility, and abortion.5,6,8,10,12 Mycoplasma bovis has been isolated from the uterus of slaughtered cows and from aborted fetuses during and after mastitis. 17 Experimental and field studies have revealed that there is a vertical transmission of M. bovis infection from the infected cow to the fetus or horizontal from the dam to the newborn calf to young cattle.15,16 The presence of the agent in blood, amnion and cervical mucus immediately after parturition, the endometrium of mastitic cows, and organs and abomasal content of viable fetuses suggest the possibility of hematogenous dissemination and vertical transmission of M. bovis through genital organs and amnion to the fetus and, finally, to the newborn calf (Stipkovits L, Jaka J, Huszenyica G: 1996, Field studies on M. bovis infection of adult cattle. Proceedings of the 1st workshop of COST Action 826, Mycoplasmas of ruminants: pathogenicity, diagnostics, epidemiology and molecular genetics, pp. 115–118).5,15,17 There are few reports of M. bovis–induced abortions and isolation of M. bovis from aborted fetuses and joints from neonatal animals.4,8,11,18 Intrauterine infections with M. bovis in viable calves resulting in pneumonia or delayed occurrence of polyarthritis are rarely reported. 11 The current study describes an aborted bovine fetus and a neonatal weak calf with uncommon pneumonic lesions and a disseminated infection caused by M. bovis with demonstration of the agent in various organs by immunohistochemistry (IHC) and in situ hybridization (ISH).
The aborted bovine fetus was sent to the Staatliches Veterinaeruntersuchungsamt Arnsberg (Arnsberg, Germany) and the neonatal calf to the Veterinary Institute Oldenburg (Oldenburg, Germany) for necropsy. Animal no. 1 was a female Holstein–Friesian fetus, aborted in the last trimester of gestation with a crown-rump length (CRL) of 60 cm. Animal no. 2 was a female Holstein–Friesian calf (CRL: 80 cm) and was born preterm in the eighth month of gestation and died a few hours after birth with severe respiratory symptoms. The animals tested negative for antigen to Bovine viral diarrhea virus, Bovine herpesvirus 1, Bovine parainfluenza virus 3, and Bovine respiratory syncytial virus by fluorescent microscopy. In tissue samples, genome fragments of Bluetongue virus, Brucella abortus, Neospora caninum, Coxiella spp., and Chlamydia spp. were not detected by polymerase chain reaction. Abomasum, liver, pericardium, and lung of animal no. 1 were examined bacteriologically (Columbia sheep blood agar and Gassner agar) under aerobic conditions and with Brucella horse blood agar, Skirrow Campylobacter agar, and Mycoplasma agar under microaerophilic conditions. Organ tissue samples were fixed in 10% neutral buffered formalin and routinely processed in paraffin wax. Serial sections (4 μm) were prepared and stained with hematoxylin and eosin. Selected paraffin-embedded tissue samples from animal no. 1 (cerebrum, cerebellum, brain stem, lung, bronchial lymph node, heart, liver, and placenta) and the lung of animal no. 2 were investigated by IHC and ISH. Immunohistochemistry was performed using the avidin–biotin–peroxidase complex method a and biotinylated goat anti-mouse immunoglobulin (Ig)G or goat anti-rabbit IgG, respectively. b The different antibodies and details of their application are given in Table 1. As positive controls for detection of T and B lymphocytes and macrophages, sections of normal bovine lymph nodes were used. As positive control for M. bovis antigen, lung sections from a calf that had been euthanized after respiratory infection with M. bovis strain 1067 were used. Normal lung tissue and bronchial lymph nodes from 4 aborted bovine fetuses served as negative controls. For ISH, a digoxigenin-labeled c DNA probe targeting the vspA gene of M. bovis was applied. 13 Tissue sections incubated with hybridization buffer d lacking the DNA probe served as negative controls. Lung samples from healthy, slaughtered cattle injected with a concentrated suspension of M. bovis cells served as positive controls. 13
Antibodies used for immunohistochemistry on paraffin sections for the detection of Mycoplasma bovis in an aborted bovine fetus and neonatal calf.*
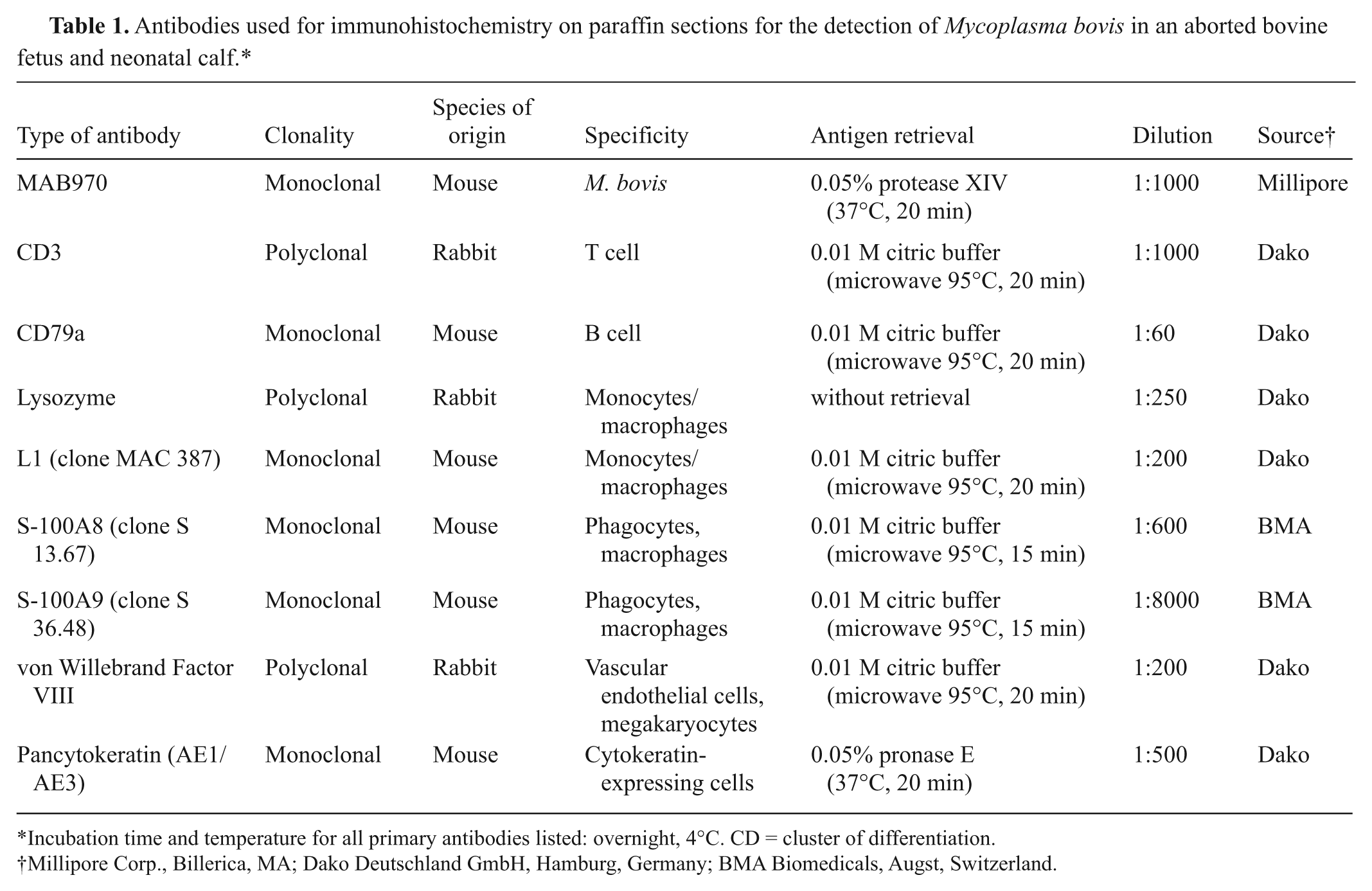
Incubation time and temperature for all primary antibodies listed: overnight, 4°C. CD = cluster of differentiation.
Millipore Corp., Billerica, MA; Dako Deutschland GmbH, Hamburg, Germany; BMA Biomedicals, Augst, Switzerland.
At necropsy, both animals showed a swelling of visceral or bronchial and mediastinal lymph nodes and a lobular consolidation of the lung parenchyma. A fibrinous pericarditis and a mild hepatomegaly were found in animal no. 1. The histological examination of the lung of both animals revealed a severe multifocal to coalescing lymphoplasmahistiocytic interstitial pneumonia (Fig. 1) associated with mild to moderate suppurative bronchopneumonia. In addition, moderate numbers of large multinucleated giant cells in alveolar spaces with up to 10 peripherally located nuclei were found (Fig. 1). These cells were negative for von Willebrand factor VIII but showed a positive labeling for lysozyme, L1, S-100A8, and S-100A9. The epithelium of the majority of small bronchioli was moderately swollen with a clear cytoplasm, suggesting hydropic degeneration. Partially around bronchi/-oli, but also multifocally distributed throughout the lung parenchyma, a moderately increased infiltration of lymphocytes in comparison to the control fetuses was recognized. Equal numbers of lymphocytes in these areas expressed cluster of differentiation (CD)3 or CD79a, confirming the presence of T and B lymphocytes. Scattered, factor VIII–positive megakaryocytes were present within the alveolar spaces in animal no. 2. In animal no. 1, mild multifocal infiltrates of neutrophils were detected within the bronchial lymph node as well as some factor VIII–expressing megakaryocytes. In animal no. 1, the following lesions in other organs were diagnosed: moderate, multifocal fibrinonecrotizing epicarditis; mild, multifocal, predominantly lymphohistiocytic myocarditis; moderate, periportal lymphohistiocytic hepatitis with mild periportal fibrosis; mild lymphohistiocytic meningitis; and mild to moderate, multifocal fibrinonecrotizing placentitis (Fig. 2). In the normal lung from aborted fetuses, mild numbers of S-100A8–, S-100A9–, L1-, and lysozyme-expressing macrophages and scattered CD3-expressing T cells were present within the pulmonary interstitium, the alveolar walls, and the lumen of blood vessels. Only few CD79a-positive B cells were present within blood vessels.
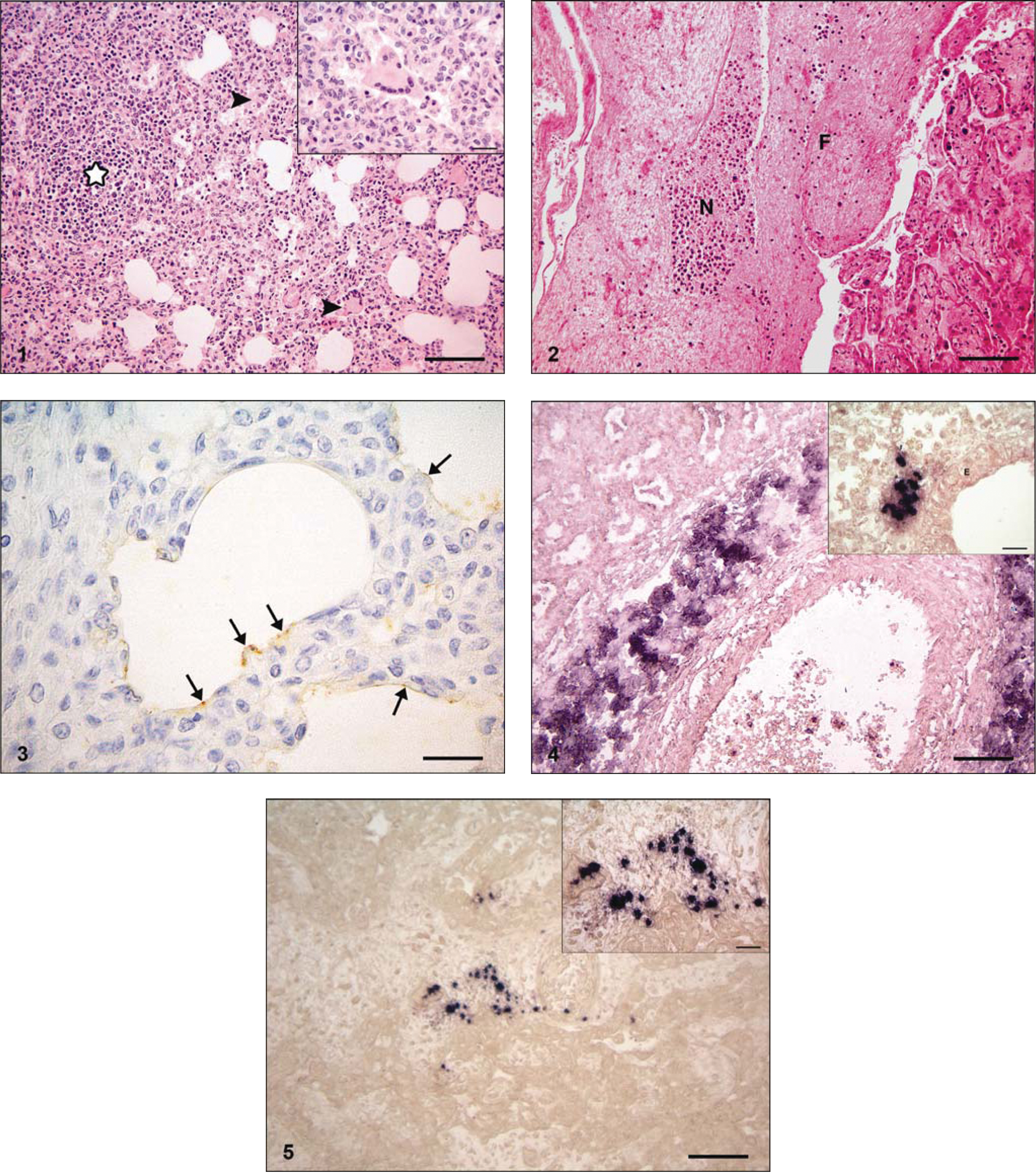
Aborted female Holstein–Friesian fetus. Lung with lymphoplasmahistiocytic interstitial pneumonia, multinucleated giant cells (arrowheads; inset) and lymphocytic aggregates (star). Hematoxylin and eosin (HE). Bar = 100 μm. Inset: HE. Bar = 20 μm.
From the pericardial sac of animal no. 1, fried egg–shaped Mycoplasma colonies were isolated. Aspergillus spp. were detected in lung and abomasal content of animal no. 2. No pathogenic bacteria were isolated from lung and liver of animal no. 1 and liver and spleen of animal no. 2. The results of IHC and ISH for M. bovis in different organ tissues of the 2 animals are given in Table 2. In situ hybridization detection of M. bovis DNA was demonstrated in more organs and with a stronger intensity than M. bovis antigen by IHC. The molecular layer of the cerebellum, the alveolar surface of the lung (Fig. 3), periportal macrophages within the liver, and a perivascular accentuated positive labeling for M. bovis antigen within the placenta were detected in animal no. 1. The lung of animal no. 2 was negative for MAB970 but showed a mild multifocal distribution of M. bovis DNA within the lung parenchyma (Fig. 4). Within the cerebrum, the molecular layer, and Purkinje cells of cerebellum, choroid plexus, brain stem, lung, bronchial lymph node, liver, heart, and placenta (Fig. 5) of animal no. 1, M. bovis DNA was multifocally and perivascularly detected. All tissues of control animals were negative by IHC and ISH to M. bovis.
Immunohistochemical labeling of Mycoplasma bovis antigen and results of in situ hybridization in different organs of an aborted bovine fetus and neonatal calf.*

− = no M. bovis antigen or DNA found by immunohistochemistry or in situ hybridization; (+) = minimal amount of M. bovis antigen or DNA; + = mild amount of M. bovis antigen or DNA.
It is postulated that there is a hematogenous dissemination from the lung to other tissues in which M. bovis can persist and be recovered 21 days after infection from the spleen, liver, and kidney of experimentally challenged calves.7,19 Mycoplasma bovis antigen has been localized in macrophages in various tissues, hepatocytes, bile ductules, renal tubules, and rarely axons in facial nerves of calves. 14 Thus, bacteremia following infection of the respiratory tract with M. bovis with subsequent arthritis has been reported in young calves but not in adult pregnant cattle.7,9,19 Mycoplasma bovigenitalium and M. bovis are commensals of the reproductive tract of cows and bulls and have been demonstrated in both healthy and sick animals, but their role is not clear in bovine reproductive disease. Isolation of M. bovis from aborted fetuses has been reported only infrequently; M. bovis has been isolated from the abomasal contents of an aborted bovine fetus.5,8 Mycoplasma bovis isolation has been reported in endocarditis, purulent meningitis, and cerebral necrosis from young calves. 3 Ureaplasma diversum, which belongs to the family Mycoplasmataceae, causes late-term abortion, stillbirth, or newborn weak calves. Ureaplasma-infected cows develop a fibrinonecrotizing histiocytic and plasmacytic inflammation in amnion and chorioallantois. Aborted fetuses showed firm lungs with a nonsuppurative alveolitis and a peri-airway lymphocytic mononuclear infiltrate. 1 In the current study, 1 aborted fetus and 1 neonatal weak calf of asymptomatic cows developed a moderate to severe suppurative bronchointerstitial pneumonia with multinucleated giant cells of macrophage origin within the alveolar spaces. In the lungs of both animals, M. bovis DNA was intralesionally detected. Neutrophilic aggregates in placenta, lung, bronchial lymph node, heart, and the leukostasis within the meninges and the brain parenchyma in animal no. 1 suggest a hematogenous spread resulting in septicemia. Mycoplasma bovis DNA and antigen was found in nearly all of the inflamed organs of animal no. 1 and intralesionally within the placenta, which is consistent with primary placental infection with M. bovis and subsequent bacteremia of the fetus. These findings, particularly the detection of M. bovis DNA in the lung lesions of both animals, lead to the conclusion that the lung lesions are directly caused by M. bovis. Because of the exclusion of other potentially infective agents that can lead to bovine abortion, the bacteriological isolation of fried egg–shaped mycoplasmal colonies from the pericardial sac of animal no. 1 and the detection of M. bovis antigen and DNA confirm M. bovis as the etiological agent of abortion and preterm birth in these 2 cases.
The multinucleated giant cells expressed lysozyme, L1, S-100A8, and S-100A9, confirming the histiocytic origin of these cells. Within the alveolar spaces and the alveolar walls of inflamed lung parenchyma of both animals, a moderately increased amount of macrophages was present in comparison to the lung of the control fetuses. These findings lead to the conclusion of the presence of an innate immune response. Around bronchi/-oli and scattered throughout the lung parenchyma, moderately increased lymphocytic aggregates consisting of T and B lymphocytes were detected in comparison to the control animals. Bronchus-associated lymphoid tissue (BALT) is absent in neonatal lungs of calves, as confirmed in the current study, and increases progressively with age. Widely scattered lymphocyte aggregates are the predominant morphological type of BALT in cattle without a follicular development. 3 These findings lead to the conclusion that the infiltration of T and B lymphocytes seem to be a specific immune reaction triggered by the presence of M. bovis within the lung tissue of these animals. The lung lesions of these 2 cases caused by M. bovis are different from the nonsuppurative alveolitis that is reported in calves infected with U. diversum. Peri-airway lymphocytic aggregates, however, are described in both M. bovis– and U. diversum–infected calves. In summary, the present report describes pneumonic lesions associated with multinucleated giant cells in an aborted bovine fetus and a neonatal calf caused by M. bovis.
Footnotes
a.
VECTASTAIN® Elite ABC Kit, Vector Laboratories Inc., Burlingame, CA.
b.
Biotinylated goat anti-mouse antibody and biotinylated goat anti-rabbit antibody, Vector Laboratories Inc., Burlingame, CA.
c.
Roche Diagnostics GmbH, Mannheim, Germany.
d.
Qbiogene, Heidelberg, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
