Abstract
Brain tissue from 12 aborted bovine fetuses submitted to the Wisconsin Veterinary Diagnostic Laboratory revealed histologic lesions that consisted of glial nodules and variable degrees of mononuclear inflammation, microhemorrhage, neuronal necrosis, and cerebral cortical cavitation. A diagnosis of
Introduction
Histopathologic lesions in 8 Angus and 4 Holstein late-term BHV-aborted fetuses, submitted to the Wisconsin Veterinary Diagnostic Laboratory (University of Wisconsin, Madison, WI) over the course of 3 years, were reviewed. The diagnosis in all of these cases was based on classic gross and histopathologic lesions, including severe multifocal hepatocellular necrosis, and BHV-1–positive indirect fluorescent antibody staining of multiple fetal tissues. Encephalitis was observed in all cases. Because encephalitis has not been previously associated with BHV-1 abortion, brain tissue was then subjected to confirmatory diagnostics, including immunohistochemical (IHC) staining, virus isolation (VI), and polymerase chain reaction (PCR), followed by sequencing to identify the herpesvirus subtype. Finally, the possibility of infection with
Summary of diagnostic tests performed to detect and/or confirm the presence of

IHC = Immunohistochemistry performed on brain sections; FA = fluorescent antibody–positive tissue (L = lung; K = kidney; Li = liver; B = Brain; Pl = placenta); VI = virus isolation; H = Holstein breed; ND = specified test was not run; A = Angus breed.
All BHV-1 polymerase chain reaction (PCR) assays were positive on brain tissue; specific cycle threshold (Ct) level is given.
Materials and methods
Histopathologic and IHC specimen preparation
Tissues from necropsied fetuses were fixed in 10% buffered neutral formalin. Multiple tissues, including one or more sections of cerebral cortex and a section of brainstem, were paraffin embedded, sectioned at 5–8 μm, and stained with hematoxylin and eosin. Brain tissue blocks from 8 cases identified on the basis of lesions were then sectioned and mounted on charged glass slides. a The slides were incubated overnight with BHV-1 monoclonal antibody F-2 directed at BHV-1 glycoprotein C. b The slides were then stained with an enhanced alkaline phosphatase red detection kit c and hematoxylin counterstain. Positive and negative control tissues were tested concurrently.
Immunofluorescent antibody staining and Neospora serology
Fresh fetal tissues (brain, liver, lung, and kidney) were obtained at necropsy and were embedded in tissue-freezing media at −20°C and were sectioned. Cryosections were incubated at 37°C with a polyclonal antisera directed to BHV-1 and were conjugated to fluorescein isothiocyanate (FITC).
b
The slides were washed in phosphate buffered saline (PBS) and then mounted with buffered glycerol. The sections were examined with epifluorescence microscopy for apple-green fluorescence. For
Indirect fluorescent antibody
An indirect fluorescent antibody technique was performed on the last 3 samples (Table 1). Chamber slides with
Virus isolation
One-gram portions of tissues collected at necropsy (Table 1) were minced to approximately 3–5-mm pieces. Samples were diluted 1:10 in Hanks' Balanced Salt Solution d and were processed on a stomacher for 60 sec. Supernatant was transferred to primary bovine testicular cells and primary bovine turbinate cells. Cultures were incubated for 7 days and observed for cytopathic effect. Cultures were frozen, and supernatant was passed to bovine turbinate cells seeded in a chamber slide, which were observed for cytopathic effect for 2–3 additional days. Cells were stained by fluorescent antibody technique for the presence of BHV-1.
Nucleic acid extraction
Nucleic acid was extracted from 25 mg of fresh tissue samples by using the Qiagen DNeasy tissue kit, e by following the protocol recommended by the manufacturer. Nucleic acid was eluted in 50 μl of the supplied elution buffer. All paraffin-embedded tissues were pretreated by using a Chelex-100 f method, 9 followed by extraction with the Qiagen DNeasy tissue kit.
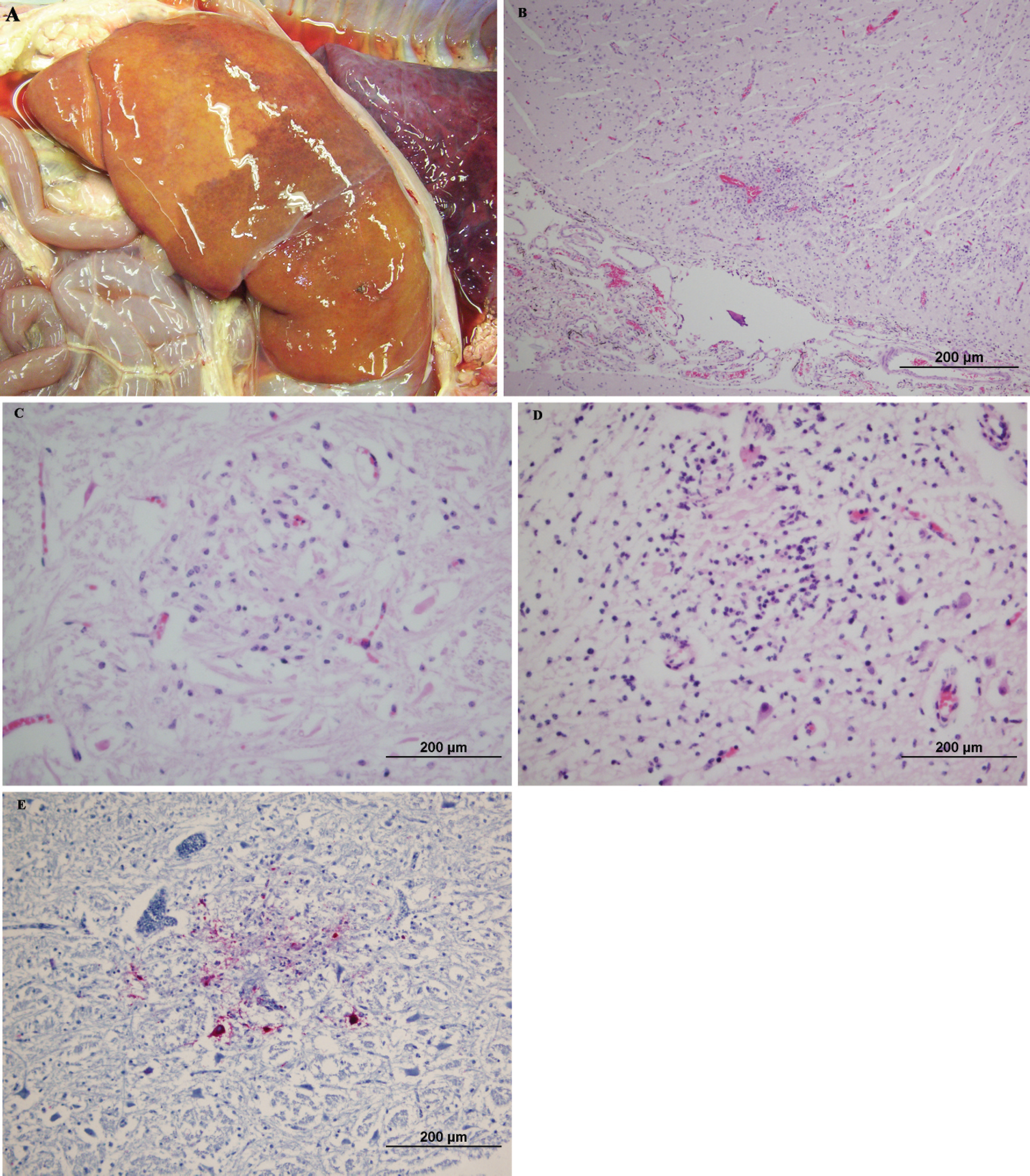
Aborted bovine fetal tissues.
Polymerase chain reaction
Polymerase chain reaction was performed on paraffin-embedded blocks for detection and confirmation of the presence of
Sequencing
To substantiate that the isolates were BHV-1, larger segments of the glycoprotein C and thymidine kinase genes were amplified and sequenced. Gene segments chosen were previously identified as having regions that differentiated BHV-1 from BHV-5. 4–6,13 Previously published primer sequences were chosen for the thymidine kinase gene: forward (TK1, 5′-TGGTACGGACGCCTTAAGTGG-3′) and reverse (TK2, 5′-GTTGATCTCGCGGAGGCAGTA-3′). 13 The following primer sequences were chosen for the glycoprotein C gene: BHV-1 gC 1F (5′-CATGGGCCCGCTGGGGCGA-3′) and reverse (BHV-1 gC 477R, 5′-CAGTGCACGTACAGCGGCT-3′). The master mix included 5.00 μl of Buffer A, 200 nM of each primer, 0.50 μl of AccuPrime GC-rich DNA polymerase, k 5.00 μl of template, and nuclease-free water up to a volume of 25 μl. Cycling parameters for the assay were as follows: a denaturation step of 95°C for 10 min, 40 cycles of 95°C for 1 min, 60°C for 1 min, 72°C for 1 min, and a final extension at 72°C for 10 min.
Polymerase chain reaction products were purified by using Qiagen QIAquick PCR purification kit, e by following the recommended protocol. The nucleic acid concentration was determined by spectrophotometry, and discrete bands were confirmed on agarose gels before submission for sequencing at the DNA Sequence Laboratory of the Genome Center of Wisconsin (University of Wisconsin, Madison, WI). All postsequence analysis of data was performed by using Vector NTI and AlignX software. k
Results
Pathology
Multifocal hepatocellular necrosis was the most severe gross and histopathologic lesion identified and was present in all of the 12 fetuses reviewed (Fig. 1A). In addition, present in all of the cases was mild-to-severe multifocal encephalitis (Fig. 1B). A postmortem change precluded a detailed description of the anatomic distribution of brain lesions, but lesions were identified in the cerebral cortex in 5 of the 12 cases and in the brainstem in 9 of the 12 cases. Glial nodules were a histologic feature present in all of the lesions. Additional findings included mononuclear inflammation with cavitation in the cerebral cortex, neuronal necrosis, and microhemorrhage. Intranuclear inclusion bodies were not found in any of the cases. All cases lacked the fetal cardiac lesions commonly associated with
Serology, virology, and molecular diagnostics
Serologic testing for

Thymidine kinase (
Discussion
It was initially unclear whether the described fetal brain lesions were an unrecognized or underappreciated manifestation of infection by BHV-1, an atypical manifestation of BHV-1, or the result of a different strain of BHV. Because of the known neurotropism and histologic description of encephalitis in BHV-5–infected calves, this strain was considered a possible etiologic agent. Fluorescent antibody and IHC results were inconsistent in the detection of BHV-1 in the brain, presumably because of the viral load, viral viability, or tissue integrity; nevertheless, in all cases, PCR with sequence analysis showed complete homology with BHV-1 and limited homology with BHV-5. The BHV-1 PCR was designed to rule out BHV-5 by selection of primer and probe sequences that define an amplicon more closely homologous to BHV-1. 13 Whereas BHV-1 and BHV-5 share 85% homology throughout most of the genome, there are regions where the 2 strains are more diverse. Among these are regions in the thymidine kinase, glycoprotein C, and glycoprotein E genes. 1,4–7,13,14 To distinguish BHV-1 from BHV-5, larger regions of the thymidine kinase gene and the amino terminus of glycoprotein C, which contain distinct species differences, were sequenced. Targeted primer regions were highly conserved between BHV-1 and BHV-5, and internal sequences allowed differentiation between the 2 species because of multiple mutations and deletions in BHV-5. The sequence results support an identification of BHV-1.
The encephalitis consistently present upon review of clinically typical BHV-1 abortions, and the described molecular investigations suggest that the observed neurologic infection and tissue damage that result in encephalitis is an underappreciated manifestation of BHV-1 fetal infection. The reason for the apparent inability of BHV-1 to cause significant neurologic lesions postpartum is unknown. Clearly, there are many host factors specific to the manifestation of intrauterine viral infections that do not exist postpartum, and, in some cases, viral factors, such as gene expression, vary with developmental stages of the fetus. 20 The severity of the lesions in other organ systems in the fetuses reviewed precluded the possibility of a live birth. It is unclear if mild in utero BHV-1 infections with live births and latent or active infections occur.
Because
The brain has a limited range of morphologic responses to injury such that multiple infectious causes of abortion can result in similar lesions. Use of a multiple-agent real-time PCR assay for bovine abortion cases can rapidly resolve the ambiguity nonspecific lesions generate and will distinguish between BHV-1 and
Acknowledgements
The authors thank Beth Gray for her excellent histopathology technical support and Chris Schatz for editorial assistance.
Footnotes
a.
Fisher Scientific, Chicago, IL.
b.
VMRD, Pullman, WA.
c.
Ventana, Tucson, AZ.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Qiagen Inc., Valencia, CA.
f.
Bio-Rad Laboratories, Hercules, CA.
g.
BioSearch, Novato, CA.
h.
Applied Biosystems, Foster City, CA.
i.
Center for Veterinary Biologics, Ames, IA.
j.
Integrated DNA Technologies (IDT), Coralville, IA.
k.
Invitrogen Corp., Carlsbad, CA.
