Abstract
Cyclooxygenase (COX; also known as prostaglandin endoperoxide synthase) is a key enzyme in the biochemical pathway leading to the synthesis of prostaglandins. A large amount of epidemiological and experimental evidence supports a role for COX-2, the inducible form of the enzyme, in human tumorigenesis, notably in colorectal cancer. COX-2 mediates this role through the production of PGE2 that acts to inhibit apoptosis, promote cell proliferation, stimulate angiogenesis, and decrease immunity. Similarly, COX-2 is believed to be involved in the oncogenesis of some cancers in domestic animals. Here, the author reviews the current knowledge on COX-2 expression and role in cancers of dogs, cats, and horses. Data indicate that COX-2 upregulation is present in many animal cancers, but there is presently not enough information to clearly define the prognostic significance of COX-2 expression. To date, only few reports document an association between COX-2 expression and survival, notably in canine mammary cancers and osteosarcomas. Some evidence suggests that COX inhibitors could be useful in the prevention and/or treatment of certain cancers in domestic animals, the best example being urinary transitional cell carcinomas in dogs. However, determination of the levels of COX-2 in a tumor does not appear to be a good prognostic factor or a good indicator for the response to nonsteroidal anti-inflammatory drug therapy. Clearly, additional research, including the development of in vitro cell systems, is needed to determine if COX-2 expression can be used as a reliable prognostic factor and as a definite therapeutic target in animal cancers.
Cyclooxygenases and Prostaglandins
Prostaglandins are short-chain lipid-derived metabolites involved in a wide variety of important physiological processes such as ovulation, maintaining renal blood flow, and contributing to the cytoprotection of the gastric mucosa. However, prostaglandins are also significant players in many pathological processes, including the development of cancers. Cyclooxygenase (COX; also known as prostaglandin endoperoxide synthase or PGHS) is the rate-limiting enzyme in the biochemical pathway leading to the biosynthesis of all prostaglandins. It mediates the conversion of arachidonic acid to prostaglandin G2 (PGG2) and, subsequently, reduces PGG2 to prostaglandin H2 (PGH2). PGH2 is then converted into different biologically active prostanoids by specific prostaglandin synthases. 109 Two distinct forms of cyclooxygenase have been recognized. The first isoform, COX-1, was initially identified in the 1970s by Sir John Vane as the target of aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs). 123 The existence of a second and distinct isoform of the enzyme, designated COX-2, was only revealed in the early 1990s. 47,59,84,106,108 It was then shown that the 2 isoforms are encoded by 2 different genes located on 2 separate chromosomes, although they share 60 to 65% identity at the amino acid level. 49,128,130
COX-1 is constitutively expressed in a wide variety of cells and tissues and is responsible for the production of prostaglandins involved in maintaining homeostasis. In contrast, COX-2 is normally absent (except in a small number of tissues such as the placenta, brain, and kidney), and its expression is induced following stimulation by different mediators of inflammation such as interleukin-1, tumor necrosis factor–1, and lipopolysaccharide. 47,71 Although COX-2 can contribute to some physiological processes (eg, ovulation), it is mostly expressed in pathological states, principally in inflammatory reactions and in oncogenesis. A third isoform of cyclooxygenase, COX-3, was initially identified in dogs, but the relevance of this isoform to pathological states in dogs as well as in other species has yet to be established. 14 Although COX-3 was first believed to be the site of action of acetaminophen, a commonly used antipyretic and analgesic drug, this has since been refuted. 54
Cyclooxygenases and Cancer
A large body of evidence suggests that COX-derived prostaglandins contribute to tumorigenesis in humans. 30 Colorectal cancer represents the first cancer where the role of COX-2 and prostaglandins was suspected after epidemiological studies had revealed that the regular intake of low doses of aspirin reduced the risk of colorectal cancer. 15,120 This initial indication of a role for COX-2 in colorectal cancer was then followed by a series of studies demonstrating that COX-2 is overexpressed in colorectal cancers. 24,51,99 Immunohistochemical analyses revealed that COX-2 was present in colonic epithelial cancer cells. 99 Moreover, the levels of PGE2, one of the principal metabolites of COX-2, are elevated in cancerous colonic tissues compared to normal tissues. 91,96
PGE2 exerts its actions via binding to 4 specific cell surface G protein-coupled receptors (designated EP1 to EP4) and can influence many cellular events that contribute to cancer development. Characterization of the precise role of each EP subtype in carcinogenesis is a relatively new and complex field of investigation. Different EP receptors appear to be involved at various stages of carcinogenesis and in different types of cancer. 27,125 For example, experimental evidence indicates that EP1 and EP4 play a role in colon cancer, whereas EP2 is involved in mouse skin tumor development. 10,116,125 The presence of EP1 has also been demonstrated in human breast cancer cells, and EP4 antagonists have been shown to inhibit breast cancer metastasis. 68,119 Moreover, PGE2 stimulates through the EP4 receptor the transcription of Id-1, a gene involved in breast cancer metastasis. 114 Via these receptors, PGE2 is able to promote cell proliferation and survival (through different mechanisms, including suppression of apoptosis), promote tumor angiogenesis (through the production of proangiogenic growth factors such as vascular endothelial growth factor [VEGF] and basic fibroblast growth factor [bFGF]), increase cell invasion and metastasis, and suppress the immune response in order for the tumor cells to escape immunosurveillance (Fig. 1 ). 30,32,39,122
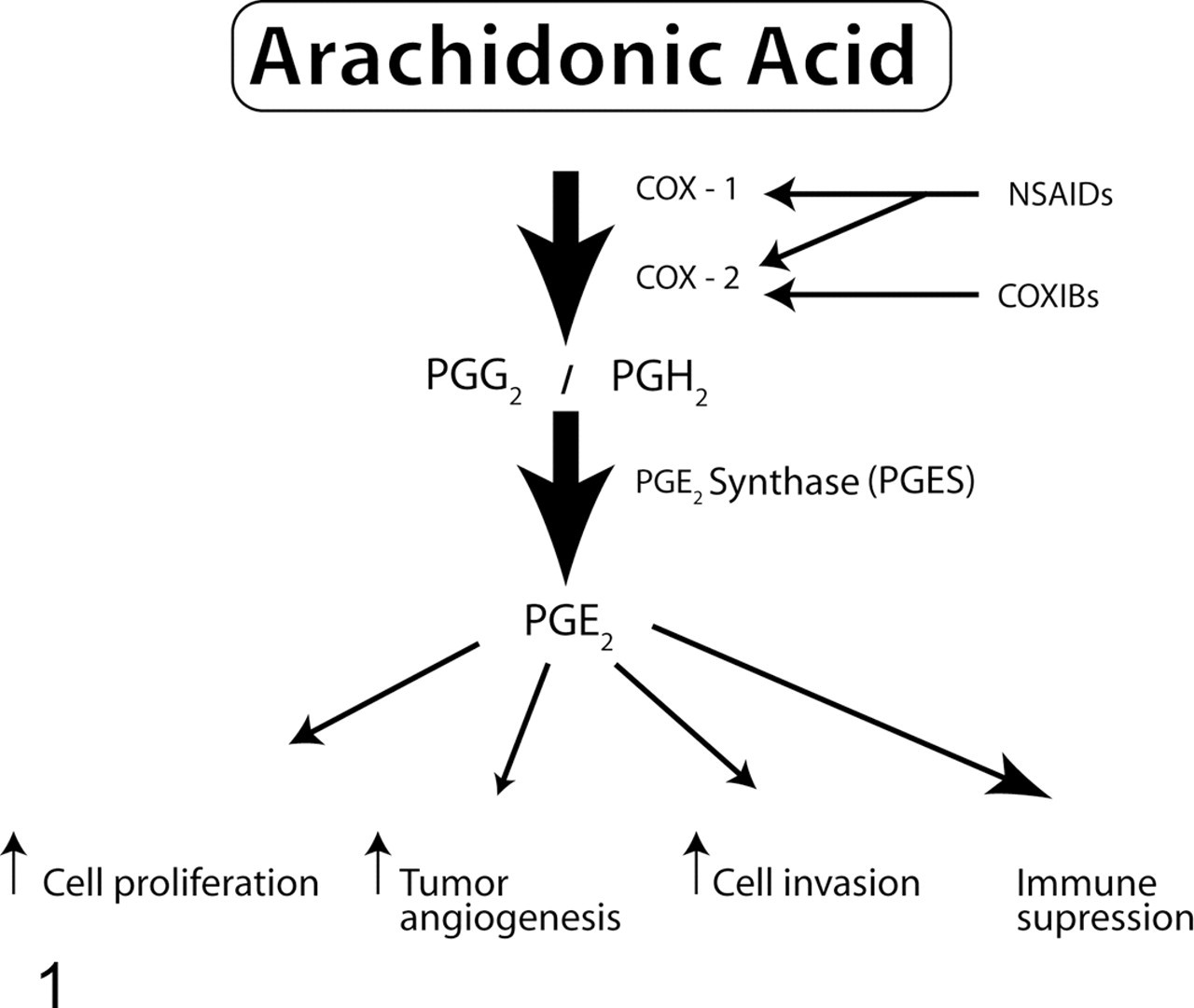
Pathway of prostaglandin E2 (PGE2) synthesis and roles of PGE2 in cancer development. Cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) mediate the conversion of arachidonic acid into PGG2 and PGH2, whereas the enzyme prostaglandin E synthase (PGES) converts PGH2 into PGE2. PGE2 contributes to oncogenesis by acting on cell proliferation, tumor angiogenesis, cell invasion and metastasis, and immune modulation. Nonsteroidal anti-inflammatory drugs (NSAIDs) block the activity of COX-1 and COX-2, whereas specific inhibitors (COXIBs) inhibit only COX-2.
In addition to colorectal cancer, many other types of human malignancies have now been shown to overexpress COX-2, such as breast, pulmonary, head and neck, pancreatic, prostatic, and gastric cancers. 17,26,29,33,36,46,48,113 Several pieces of experimental evidence also support a role of COX-2 in the tumorigenic process of these cancers (see following sections). Although some studies revealing the expression of COX-1 in some cancers suggest that COX-1 could also contribute to oncogenesis in humans, notably in ovarian cancer, 22,34 this review does not address the possible role of COX-1 in animal cancers as there is currently little information on the subject.
COX-2 in Animal Cancers
Canine Cancers
Canine Mammary Cancer
As in colorectal cancer, elevated expression of COX-2 has been reported in breast cancer in women with studies showing that approximately 40 to 50% of invasive breast carcinomas have high COX-2 protein levels. 17,36,113 COX-2 overexpression appears to be present particularly in ductular carcinoma in situ (DCIS), a lesion that is a common precursor to invasive breast cancer. 5,36,105 Expression of COX-2 in breast cancer has also been linked to overexpression of HER-2/neu, a receptor of the epidermal growth factor family. 115 And as for colorectal cancer, some epidemiological studies have revealed an inverse relationship between breast cancer incidence and the use of NSAIDs in women. 38,104 Moreover, experimental overexpression of the human COX-2 gene was able to induce tumorigenesis in multiparous transgenic mice. 67 Additional evidence for a role of COX-2 in breast oncogenesis comes from experimental models of induced mammary tumors in rodents that have demonstrated the chemopreventive efficacy of NSAIDs and COXIBs to reduce breast cancer. 37,58,62 COX-2 expression in human breast cancer has been reported in some studies to correlate with a decreased disease-free survival. 17,86,97
Similarly, COX-2 is overexpressed in canine mammary tumors, with an increased expression in malignant tumors compared to benign ones. A study showed that COX-2 was expressed in 24% of adenomas and in 56% of adenocarcinomas, with the intensity of COX-2 expression being higher in adenocarcinomas compared to adenomas. 23 As seen in breast cancer in women, COX-2 staining was localized to the neoplastic epithelial cells (Fig. 2 ). This finding was then confirmed by other studies that found a lower (42%), similar (56%), or higher (62–100%) percentage of COX-2-positive malignant mammary tumors. 19,44,63,74,78,92 In some studies, the normal canine mammary gland appeared to express no or little COX-2, 23,63,78,92 whereas a recent investigation reported that the normal canine mammary gland expresses COX-2 with a membranous apical localization in contrast to the cytoplasmic distribution of COX-2 in cancerous cells. 19
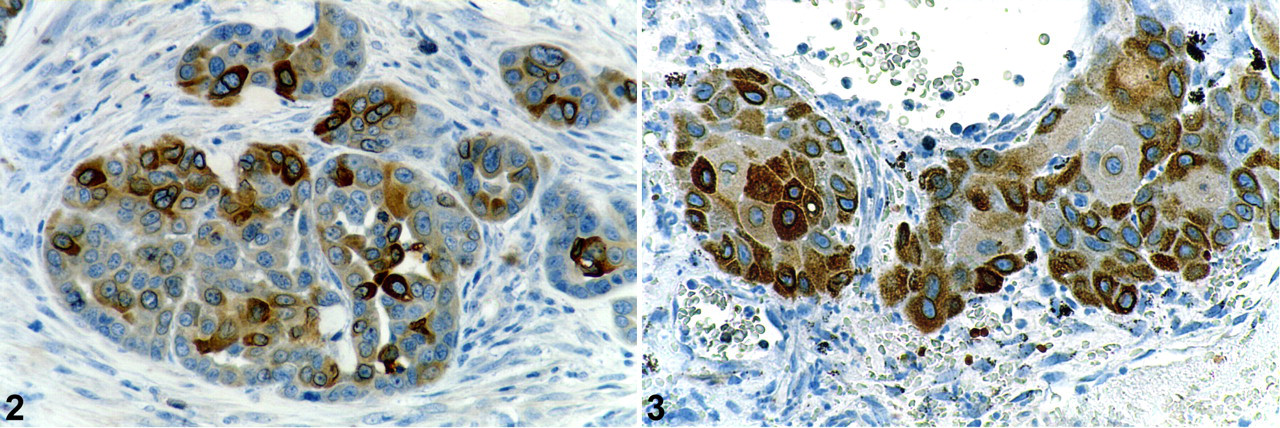
Specific types of canine mammary carcinomas have been reported to express higher levels of COX-2, including tubulopapillary, anaplastic, inflammatory, and squamous cell carcinomas as well as carcinosarcomas. 16,44,92,93 Metastatic lesions from mammary cancers also appear to overexpress COX-2, as Dias Pereira et al 19 found that the majority of metastatic sites (10/12) they evaluated had a labeling intensity of COX-2 similar to or stronger than the primary tumor. As observed in breast cancer, an association between COX-2 and HER2/neu overexpression has also been reported in canine mammary tumors. 74
There is little currently available information on the prognostic significance of COX-2 expression in canine mammary tumors. Only 4 studies have addressed this issue so far. The first one reported that 100% of canine mammary carcinomas were COX-2 positive (79% with elevated expression) and that COX-2 was significantly correlated to poorer prognosis. 74 However, no details were provided on the duration of the survival period of the subjects. In the second study, where an association between high COX-2 levels and poor prognosis was found, COX-2 levels were assessed by enzyme immunoassay, a technique not commonly used for determination of COX-2 expression. 93 The 2 most recent studies also found a significant relationship between higher COX-2 expression and the development of distant metastasis, a worse prognosis, and a shorter overall survival. 63,94 However, a multivariate analysis showed that COX-2 expression was not an independent prognostic factor. 94 Similarly, there is little published information on the use of NSAIDs for the treatment of mammary cancer in dogs. Only one recent clinical report revealed that piroxicam treatment of 12 dogs suffering from inflammatory mammary carcinomas resulted in clinical improvement compared to dogs treated with doxorubicin. 16 Although inflammatory carcinoma is not recognized as an entity in the World Health Organization (WHO) classification of mammary tumors of dogs, the name inflammatory carcinoma refers to a mammary carcinoma with a specific clinical presentation. 60,75
Canine Prostatic Carcinoma
COX-2 is believed to contribute to prostatic oncogenesis in humans. 1 Interestingly, overexpression of COX-2 in prostatic carcinomas was first reported in the canine species. 121 In 1999, our laboratory showed that COX-2 was absent from normal prostates but was expressed by epithelial tumor cells in the majority (75%) of prostatic adenocarcinomas. Western blot analysis confirmed the presence of COX-2 only in prostatic adenocarcinomas. This was soon followed by demonstration of the overexpression of COX-2 in human prostatic carcinomas. 33 Several later reports confirmed the overexpression of COX-2 in prostatic cancer and its absence in normal prostate in the canine species. 65,78,111 Experimentally, treatment of dogs with estrogen has been shown to induce squamous metaplasia and COX-2 expression in the prostate. 21 So far, only one clinical study has investigated the effect of COX inhibitors on canine prostatic carcinoma. 111 The study of 35 dogs found that treatment with nonspecific COX-2 inhibitors (piroxicam and carprofen) significantly prolonged survival time in dogs with prostatic carcinomas compared to dogs that were not given NSAIDs (6.9 vs 0.7 months). 111 However, the authors found no association between COX-2 tumor-staining intensity and survival times.
In human prostate cancer, the intensity of COX-2 staining has been found to be higher in less differentiated tumors than in well-differentiated ones 69 and to correlate with the degree of local chronic inflammation. 124 This does not appear to be the case in dogs as the presence of inflammatory infiltrates was associated with lower COX-2 expression, and the COX-2 expression did not vary with the degree of differentiation of the tumors. 65
Canine Transitional Cell Carcinoma
Urinary bladder cancer affects each year more than 67,000 people in the United States, with the most common form being invasive transitional cell carcinoma (TCC). 12 In dogs, TCC is also the most common tumor in the urinary bladder. Several pieces of evidence point to a role for COX-2 in this cancer in both humans and dogs, and the dog has been proposed as a unique naturally occurring model for this cancer. 18 Indeed, COX-2 is expressed in the neoplastic epithelium of the majority of human and canine TCCs of the urinary bladder. 52,57,64,79 Furthermore, treatment with NSAIDs (piroxicam and celecoxib) of athymic mice bearing xenografts of human TCC cell lines that overexpress COX-2 significantly delayed the progression of the xenografts. 77
Clinical trials with piroxicam have also shown promising results in dogs suffering from TCC with complete and partial remission observed in some cases (18–33%) as well as cancer stabilization in other dogs (50–53%). 56,76 The tumor response was associated with the induction of apoptosis and a reduction in the concentration of a proangiogenic factor in the urine (bFGF). 76 However, the expression of COX-2, the concentration of PGE2 in the tumor, or the combination of the two were not useful in predicting response to piroxicam treatment in canine TCC. 82 Similarly, no correlation was present between PGE2 concentration and COX-2 expression within each tumor sample. 82
Canine Squamous Cell Carcinoma
COX-2 expression has also been shown to be present in a large proportion (56–100%) of canine squamous cell carcinomas (SCCs) originating from various locations (oral, cutaneous, or digital SCCs; Fig. 3 ). 78,89 One clinical study has evaluated the effect of a nonspecific COX-2 inhibitor (piroxicam) with a reported remission (including complete or partial) rate of 18% and stable disease in 29% of dogs suffering from oral SCCs. 102 A higher remission rate (56%) was obtained when dogs suffering from oral SCCs were treated with piroxicam in combination with cisplatin. 7
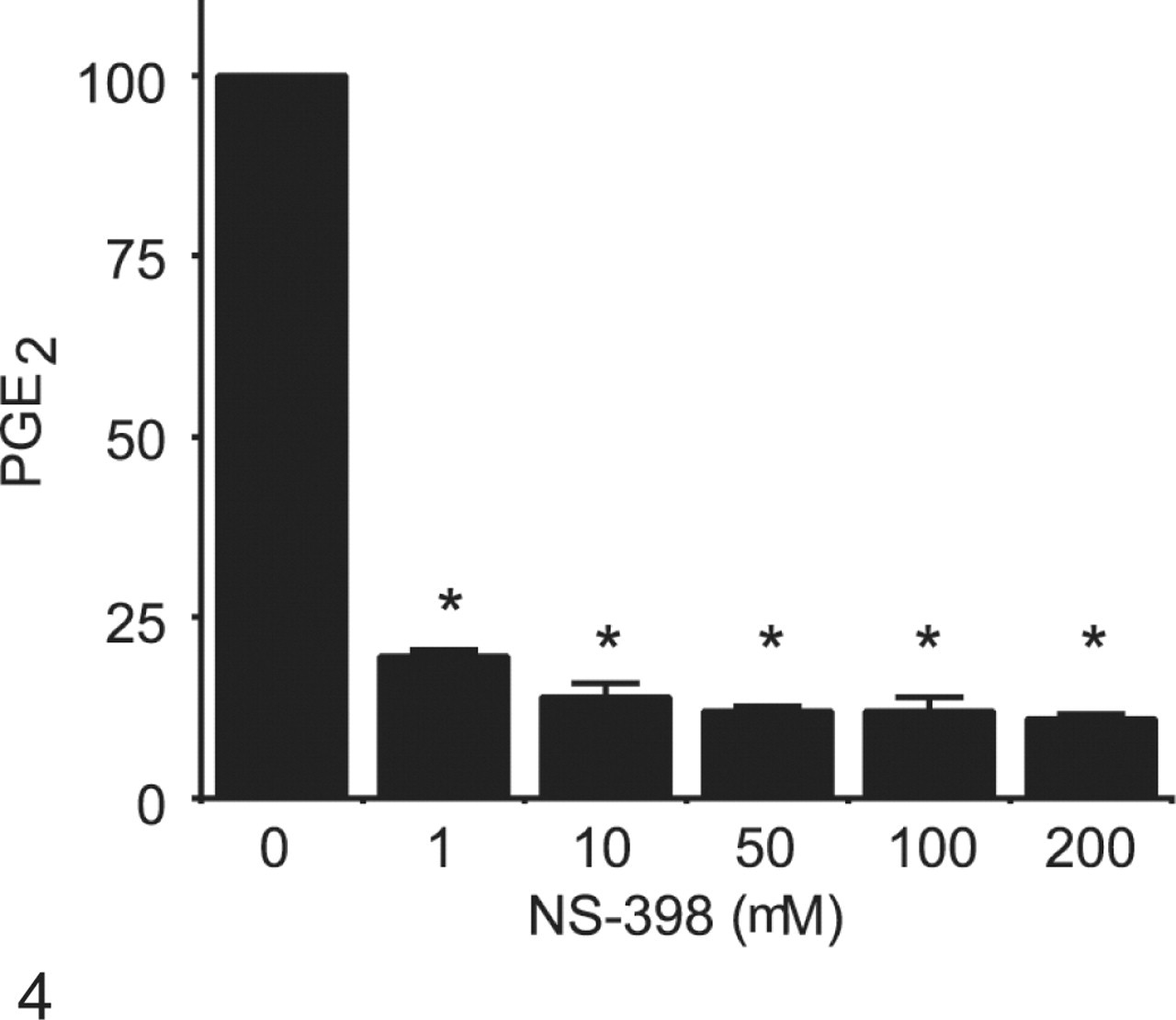
Effect of COX-2 inhibition on PGE2 synthesis by neoplastic canine mammary epithelial cells. Neoplastic canine mammary cells (CMT12) were cultured in 24-well plates in the absence (0) or presence of 1, 10, 50, 100, or 200 µg/ml NS-398 for 48 hours. Concentrations of PGE2 in culture medium were determined by radioimmunoassays. Bars marked with an asterisk are significantly different from control without inhibitor (P < .05).
Canine Intestinal Tumors
Although the role of COX-2 in intestinal cancer is well established in humans, its involvement in similar types of cancer in dogs is much less obvious. 107 Moreover, in contrast to humans, epithelial intestinal tumors in dogs are not common. 42 One report indicates that the neoplastic epithelium from 7/15 (47%) of canine intestinal adenocarcinomas (colorectal and small intestine) expressed COX-2, but most positive tumors had only a small percentage (5–10%) of stained cells. 72 COX-2 was found only in the tumor stroma of canine colorectal adenomas. 72 Similarly, only low levels of COX-2 expression were found in 38% of 21 cases of rectal adenocarcinomas with rare or occasional single cells positive for COX-2. 57
Other Canine COX-2-Positive Cancers
Expression of COX-2 has been reported in a few other types of cancers in dogs, including nasal tumors, renal cell carcinomas, ovarian carcinomas, oral melanomas, osteosarcomas, and meningiomas. 7,8,9,50,53,55,78,81,98 A high percentage (from 71–87%) of nasal epithelial tumors have been reported to overexpress COX-2, including various types of tumors (carcinomas, adenocarcinomas, squamous cell carcinomas, transitional, anaplastic). 8,50,55 A very small number (3) of renal cell carcinomas have been evaluated, with 2 cases being COX-2 positive. 53 Borzacchiello et al 9 reported that 9 out of 11 ovarian carcinomas expressed COX-2 with an intensity ranging from faint to strong. Some oral melanomas have been reported to express COX-2, but treatment with cisplatin and piroxicam of dogs with oral melanomas had very limited success, with a remission in only 2 of 9 dogs with oral melanoma. 7,78 Finally, no COX-2 was found in osteosarcomas in one study, 78 whereas another study reported that most osteosarcomas demonstrated poor to moderate COX-2 staining. 81 In the latter study, dogs with osteosarcomas displaying strong COX-2 staining had a significantly decreased survival time. 81
Canine COX-2-Negative Cancers
Oral fibrosarcomas, lymphomas, hemangiosarcomas, histioctytic sarcomas, and mast cell tumors of dogs are mostly COX-2 negative. 43,78 Definitive intraneoplastic COX-2 expression was found only in 3.5% of canine uveal melanocytic neoplasms, but the presence of COX-2 in many normal ocular structures made the exact determination difficult. 88
Equine Cancers
Equine Squamous Cell Carcinoma
Investigations on the expression of COX-2 in equine SCCs have yielded conflicting results, and the significance of COX-2 expression in equine SCCs has still to be defined. Elce et al 25 analyzed by Western blot COX-2 expression in 14 equine SCCs from different locations (conjunctival, vulvar, preputial, and penile) and found that all nonneoplastic and neoplastic tissues expressed COX-2. The only significant difference between nonneoplastic and neoplastic tissues they found was in preputial SCCs that expressed more COX-2 than normal tissues. 25 Another study evaluating 15 ocular SCCs by immunohistochemistry (IHC) found that COX-2 expression was significantly greater in corneal SCCs compared to site-matched control tissues but not in SCCs from the eyelid and third eyelid. 73 However, no details regarding the extent of COX expression, the magnitude of the difference, or the cellular distribution of positive cells were provided. Another group reported that a small percentage (<10%) of neoplastic cells from 27% of corneal SCCs were only weakly positive. 95 In agreement with this observation, Smith et al 110 recently reported that less than 1% of the neoplastic cells from SCCs originating from the corneo-scleral limbus, the third eyelid, and the eyelids were positive for COX-2. Finally, a study reported that the majority (32/37) of SCCs (eye/periocular, prepuce/penis, vulva) were positive for COX-2, with 56% of them having moderate to strong staining, but there was no mention of the localization in the eye and the number of ocular tumors that were positive. 118 The authors reported a trend of lower COX-2 expression in the metastases compared to the primary site.
There is also a paucity of clinical information about the potential benefit of treating SCCs with COX-2 inhibitors in horses. In fact, a single case report addresses the benefit of using anti-COX-2 therapy for equine SCCs. 80 A complete remission was obtained with piroxicam treatment in a horse suffering from a lip SCC that had spread to the regional lymph node. 80 The mass was reported to contain 10 to 20% neoplastic cells with moderate to intense COX-2 immunoreactivity. 80
Equine Sarcoid and Melanoma
Only one study examined COX-2 expression in other equine tumors and found that melanomas were positive for COX-2 (28% with a moderate to strong staining) while sarcoids were mostly negative. 118
Feline Cancers
Feline Squamous Cell Carcinoma
COX-2 expression in feline SCCs does not seem to be as extensive as in dogs. A first study in the cat found that only 9% of oral SCCs displayed positive immunoreactivity for COX-2, with less than 10% of neoplastic cells staining positively. 3 Hayes et al 40,41 then reported that 67% of feline oral SCCs were positive, but most cases showed labeling in <1% of cells. Similarly, DiBernardi et al 20 found that the majority of oral SCCs had weak immunoreactivity but that a small percentage of cases (18%) had high-intensity immunoreactivity. Also in contrast to dogs, feline cutaneous SCCs are reportedly COX-2 negative. 3
Feline Mammary Carcinoma
There are controversies in the literature regarding the COX-2 status of feline mammary cancers. Indeed, a first report found that 9 feline mammary adenocarcinomas were negative for COX-2. 3 On the contrary, Millanta et al 74 reported that 96% (47 cases) of feline invasive carcinomas were COX-2 positive with elevated expression of COX-2 observed in 81% of tumors. In their study, COX-2 overexpression was significantly correlated to estrogen receptor-negative status, increased progesterone receptor expression, angiogenesis, and a poorer prognosis. 74 Results from our laboratory are in agreement with the study by Millanta et al as we found COX-2 expression in 87% of 40 feline mammary carcinomas, with COX-2 levels varying from low (50%) to intermediate (32%) and high (5%). 100 A recent retrospective study of 23 cases of feline mammary adenocarcinomas found no difference in survival time or disease-free interval when using meloxicam in combination with surgery and doxorubicin-based chemotherapy. 6 However, the COX-2 status of the tumors was not determined.
Other Feline Cancers
A single study found that 7 of 19 (37%) of feline transitional cell carcinomas displayed positive immunoreactivity for COX-2. 3 Feline pulmonary adenocarcinomas, intestinal adenocarcinomas, lymphomas, and vaccine-associated sarcomas were reported COX-2 negative. 3 A study of 8 feline pancreatic adenocarcinomas reported that only 2 were COX-2 positive, with less than 10% of the cells expressing an intense staining. 85
Limitations of COX-2 Expression Studies
As mentioned previously, for some animal cancers, there exists a great variability in the results of studies looking at COX-2 expression for the same type of cancer and within a same species. This variability may be due in part to differences in techniques and methodologies used. Indeed, various techniques, including IHC, Western blot, and enzyme immunoassay, have been applied. IHC has the advantage of providing information about the cell distribution and the intensity of expression of the enzyme, although its sensitivity is less than Western blot. On the other hand, Western blot provides information about the nature of the isoform that is being recognized based on its molecular weight. In a comparative study of Western blot and IHC to determine the COX-2 expression of canine neoplastic cell lines, Heller et al 45 found that Western blot was more accurate than IHC. However, Western blot does not permit discrimination between COX-2 being located in neoplastic cells, inflammatory cells around the tumor, and stromal cells. In addition, different antibodies raised against various epitopes of COX-2 have been used, or a similar antibody was used at different concentrations (Table 1 ), and this very likely contributes also to the differences in results. Finally, and not the least, heterogeneity in the grading system used to determine the extent of COX-2 expression makes it difficult to compare results of different studies.
Antibodies Used in Studies of COX-2 Expression in Animal Tissues
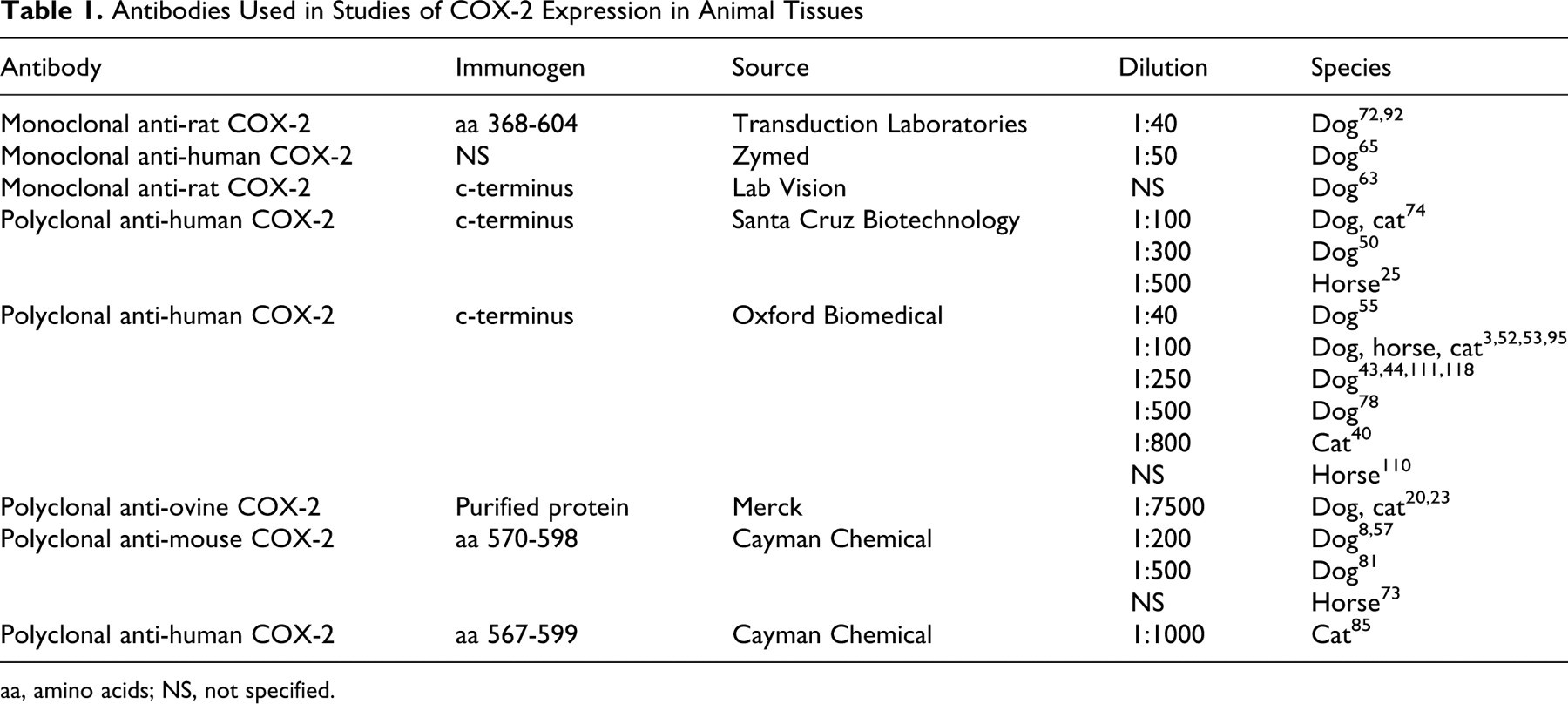
aa, amino acids; NS, not specified.
In Vitro Effect of COX-2 Inhibitors on Canine Neoplastic Cell Lines
In vitro cell systems are valuable tools to investigate the roles played by COX-2 and to evaluate possible chemotherapeutic agents. However, in vitro cell systems to investigate COX-2 and tumor cell growth in domestic animals are rare, having so far only been developed with canine cell lines. Constitutive expression of COX-2 was demonstrated in the canine mammary cell line CMT12 and in a canine neoplastic prostatic cell line. 11,65 The endogenous COX-2 expression in the neoplastic prostatic cell line was shown to be dependent on different signaling pathways (PI3K, PKC, and ERK/MAPK). 65 Our laboratory has also shown that NS398, a specific inhibitor of COX-2, significantly blocked PGE2 synthesis (Fig. 4 ) and diminished cell proliferation (Fig. 5 ) of a canine neoplastic mammary cell line and of canine neoplastic keratinocytes derived from an oral SCC. 11,90 Also, meloxicam, an NSAID that preferentially inhibits COX-2, was shown to inhibit cellular growth and to induce apoptosis in canine osteosarcoma cells. 126 These results reveal that, as observed with human cancerous cell lines, inhibition of COX-2 diminishes cell proliferation of animal neoplastic cells, suggesting that in vivo inhibition of COX-2 could help to control tumor growth. Experimental models of tumor xenografts in nude mice could represent an interesting way to test in vivo effects of COX-2 inhibition before clinical trials. By re-creating in a dynamic environment the growth of tumor cells, they offer the possibility of measuring the effect of COX inhibitors on in vivo tumor growth as well as other parameters such as angiogenesis and apoptosis. Such models have been used to study the effect of COX inhibition on some human cancers, 77,132 but no studies have yet been published looking at COX inhibition and xenografts of animal cancer cells.
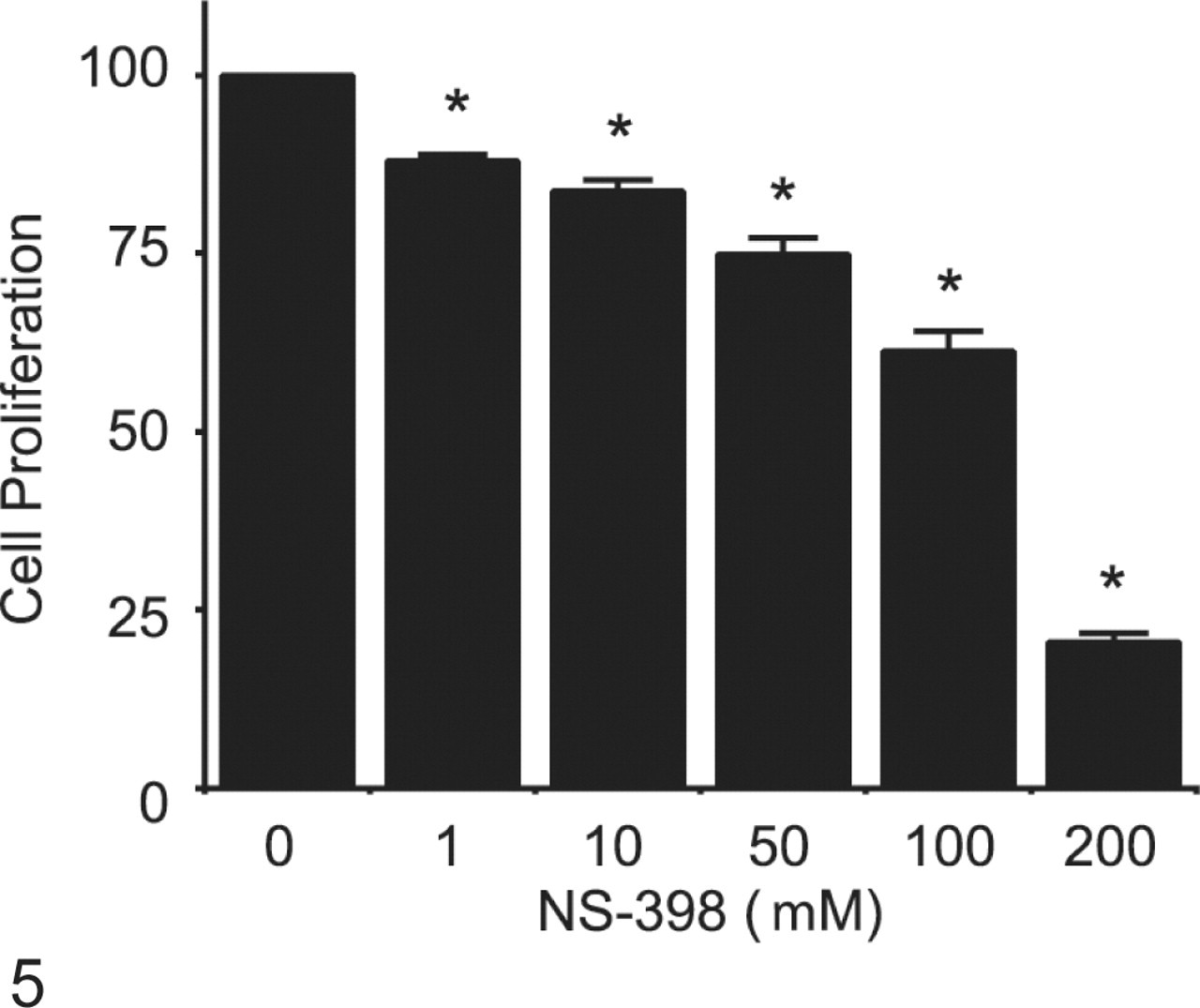
Effect of COX-2 inhibition on cell proliferation of neoplastic canine mammary cells. Neoplastic canine mammary cells (CMT12) were cultured in the absence (0) or presence of 1, 10, 50, 100, or 200 µg/ml NS-398 for 48 hours. Cell proliferation was measured using an assay based on the cleavage of the tetrazolium salt WST-1 to a soluble formazan salt by metabolic active cells (Roche Diagnostics, Basel, Switzerland). Results for cell proliferation are presented as percentages compared to control cultures without inhibitor. Bars marked with an asterisk are significantly different from control without inhibitor (P < .05).
COX-2 Inhibitors as Chemopreventive and Chemotherapeutic Tools
Considering all the evidence supporting a role for COX-2 in oncogenesis, targeted inhibition of COX-2 represents an interesting avenue for chemoprevention and/or chemotherapy. 129 As previously mentioned in the sections above, some studies have demonstrated that COX-2 inhibitors can have beneficial effects on animals affected with cancer, the most well-documented cancer to date being piroxicam treatment of TCC in dogs. Agents that can be promising tools for chemoprevention in dogs have been the subject of a recent review. 2 In humans, an international expert group recently reviewed a large number of epidemiological studies in humans and stated that, for the moment, aspirin is the only NSAID for use in chemoprevention and that additional studies will be necessary to determine the appropriate dose, best age to begin treatment, and duration of therapy. 15
NSAIDs act to reduce carcinogenesis by inhibiting COX-2 and PGE2 synthesis and their cellular effects but also through mechanisms independent of COX-2. 31 Such COX-2-independent mechanisms include induction of apoptosis and inhibition of cell cycle arrest, angiogenesis, and metastasis. 31 Moreover, there are differences in the anticarcinogenic potential between different selective COX-2 inhibitors. For example, in vitro and in vivo studies using colon carcinoma models have shown that celecoxib and methylcelecoxib, a close structural analogue of celecoxib lacking COX-2 inhibitory activity, decreased cell survival by induction of cell cycle arrest and apoptosis and reduced the growth of tumor xenografts in nude mice, whereas other selective COX-2 inhibitors demonstrated no antiproliferative effects. 101
However, determination of COX-2 expression does not appear to be helpful to predict a tumor response to COX-2 inhibitors. For example, a study on pancreatic cancer in humans reports that immunohistochemical analyses confirming COX-2 expression in the tumors were not associated with the tumor response to celecoxib treatment. 13 Interestingly, Mutsaers et al 82 demonstrated that the pretumoral levels of PGE2 and COX-2 expression of canine invasive transitional cell carcinomas of the urinary bladder were not associated with the tumor response to piroxicam. They also did not find an association between COX-2 expression and PGE2 concentration in the tumor. Moreover, although clinical response was seen in 7 dogs treated with piroxicam for inflammatory mammary carcinoma, the percentage of COX-2-positive cells in their tumors varied from 28 to 91%. 16 These results suggest that COX-2 expression as measured by IHC might not be indicative of the actual enzymatic activity and, consequently, of the levels of PGE2 produced in the tumor. In support of this conclusion, a lack of correlation between identification of COX-2 by IHC or Western blot and the enzymatic activity as measured by PGE2 production was demonstrated with neoplastic canine cell lines from various species. 45
Prognostic Significance of COX-2 Expression
Even though the role of COX-2 and its derived metabolites in tumorigenesis appears clear, the prognostic significance of COX-2 expression in cancer has not been clearly established. For example, in the case of human colon cancer, some studies are in support of COX-2 as a predictive biomarker as they found that COX-2 overexpression was associated with a worse survival. 35,87,112 However, other studies have not found such an association. 28,66,127 Similarly, the prognostic significance of COX-2 expression in other types of human cancers is controversial, with some studies supporting COX-2 as a useful predictive factor and other studies refuting such a role. 4,26,29,46,61,70,83,103,117,131 In animal cancers, there is currently not enough information to clearly define the prognostic significance of COX-2 expression. In most studies addressing COX-2 expression in animal cancers, a follow-up of the subjects is not available. To date, only few reports document an association between COX-2 expression and survival, notably in mammary cancers and in osteosarcomas. 63,74,81,92,93 However, no association was found between COX-2 in prostatic carcinoma in dogs and survival times. 111
Conclusions
As demonstrated in humans, COX-2 is overexpressed in many types of cancers in dogs, cats, and horses. There is, however, still insufficient information available on each type of cancer in the different species to determine the exact role of COX-2 in their oncogenesis. In comparison to canine cancers, feline and equine tumors appear to express less constantly COX-2. Some evidence suggests that COX inhibitors could be useful in the prevention and/or treatment of certain cancers in domestic animals, the best example being urinary TCC in dogs. However, determination of the levels of COX-2 in a tumor does not appear to be a good prognostic factor or a good indicator for the response to NSAID therapy. Clearly, additional work needs to be done to more accurately delineate the role of COX-2 in domestic animal cancers and to assess the use of NSAIDs as chemotherapeutic and chemopreventive agents in animals.
Footnotes
Acknowledgements
I thank Dr. Jean Sirois for critically reading the manuscript and Marco Langlois for his help with graphic work.
The author(s) declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
Work from my laboratory was supported by grants from the Morris Animal Foundation (grant no. D00CA-47), the American Kennel Club Canine Health Foundation (grant no. 2222), and the Natural Sciences and Engineering Council of Canada (grant no. 183964-03).
