Abstract
Incisional biopsies from the oral cavity of 2 adult cats were submitted for histological investigation. Cat No. 1 showed a solitary well-circumscribed neoplasm in the left mandible. Cat No. 2 demonstrated a diffusely infiltrating neoplasm in the left maxilla. Both tumors consisted of medium-size epithelial cells embedded in a fibrovascular stroma. The mitotic index was 0 to 1 mitosis per high-power field. The epithelial cells showed an irregular arrangement forming nests or streams in cat No. 1, whereas a palisading growth was noted in cat No. 2. Both tumors, especially that of cat No. 1, showed multifocal accumulations of amyloid as confirmed by Congo red staining and a distinct green birefringence under polarized light, which lacked cytokeratin immunoreactivity as well as and AL and AA amyloid immunoreactivity. In addition, the amyloid in cat No. 2 was positive for the odontogenic ameloblast-associated protein, formerly termed APin. In sum, both cats suffered from an amyloid-producing odontogenic tumor, but their tumors varied with respect to morphology and type of amyloid produced.
Keywords
An adult male neutered European short-hair cat (case No. 1) showed a gingival mass of about 0.5 cm in diameter in the left mandible, located near the first premolar. An infiltration into the surrounding tissue could not be detected macroscopically or radiographically. Clinically, the animal showed anorexia and severe salivation. Because the owner did not request an excision of the tumor, 4 incisional biopsies (0.1 to 0.3 cm in diameter) were taken from the middle of the mass under general anesthesia and submitted for histopathological investigation. The animal died 15 months after initial presentation, following a surgery unrelated to the tumor, as reported by the owner. A second European short-hair cat (case No. 2, female spayed), aged 4 years and 7 months, was presented to the veterinarian with epiphora and stridor nasalis. By computer tomography, a neoplasm could be detected that involved almost the whole left maxilla (Fig. 1), which, according to the owner, grew slowly during the last 2.5 years before presentation. Because surgical excision of the mass was not possible, several incisional biopsies (up to 0.3 cm in diameter) were taken about 0.5 cm deep within the mass under general anesthesia and submitted for histopathological workup. Six months after the initial presentation, the animal was still alive and in a good condition.
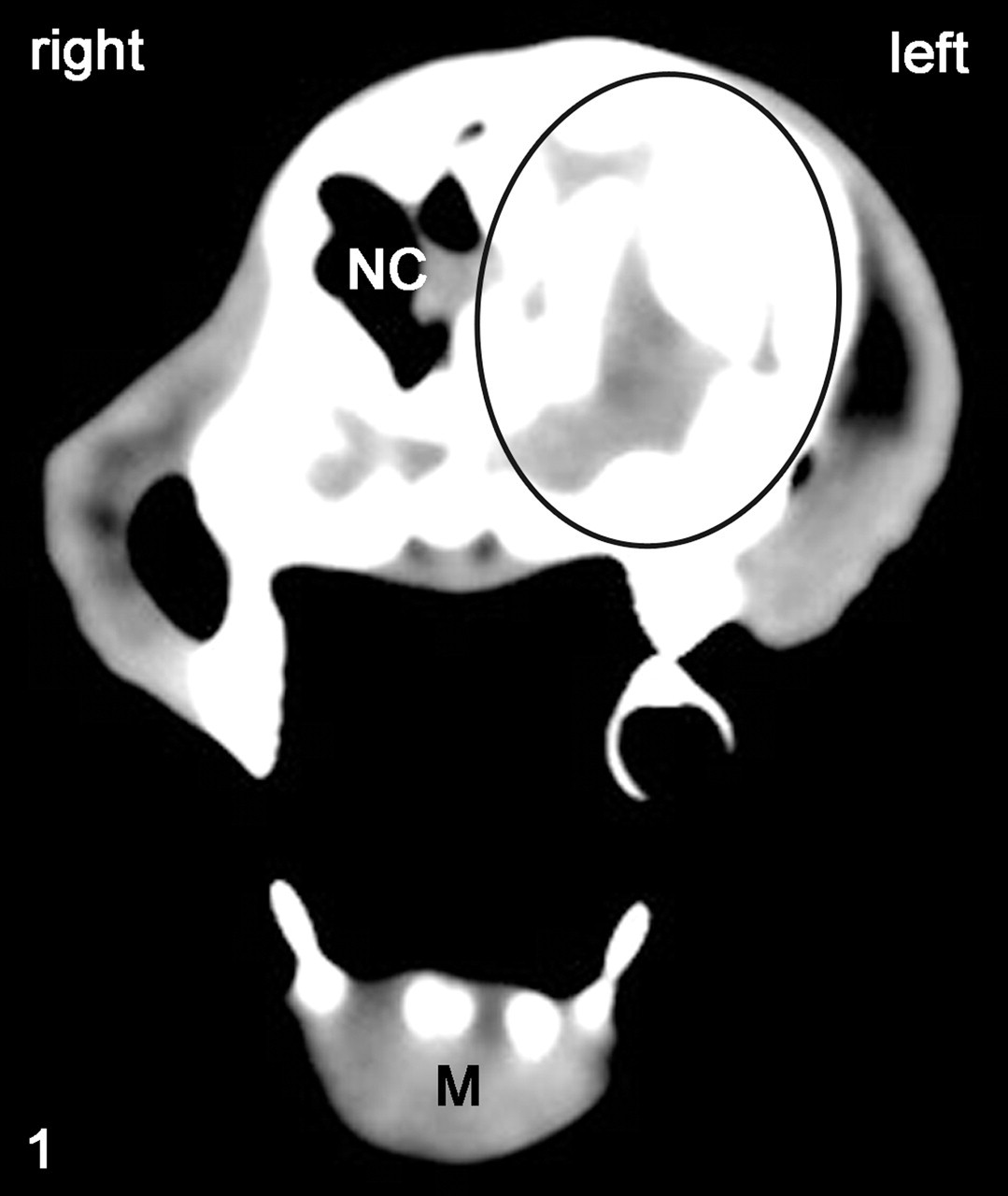
Head, cat No. 2. Tumor infiltrating most of the left maxilla (black oval). NC, nasal cavity; M, mandible. Computer tomography.
Differential Diagnoses
Based on clinical findings, differential diagnosis should include squamous cell carcinomas, fibrosarcomas and odontogenic tumors such as ameloblastomas, amyloid-producing odontogenic tumors (APOTs), and feline inductive odontogenic tumors, as well as inflammatory osteolytic processes. Note that squamous cell carcinomas and fibrosarcomas tend to progress rapidly, unlike the tumors in the present cases.
Microscopic Findings
Hematoxylin and eosin–stained sections from incisional biopsies of both masses were examined. Tumors of both animals consisted of medium-size epithelial cells with large round to ovoid nuclei containing 1 or 2 nucleoli and, occasionally, abundant eosinophilic cytoplasm with sometimes distinct cell borders. A mild anisokaryosis was detected, and the mitotic index was 0 to 1 mitosis per high-power field. Tumor cells were arranged in nests or streams in cat No. 1 (Fig. 2), whereas they showed a palisading growth pattern in cat No. 2 (Fig. 3). Additionally, the tumor of cat No. 1 contained a high amount of extracellular homogeneous eosinophilic material, only rarely noted in the tumor of cat No. 2. No stellate reticulum was present in both cases, and the neoplasm of cat No. 1 showed dentinoid formation with prominent mineralization (Fig. 2, inset). A moderate amount of fibrovascular stroma was present in both tumors (Figs. 2, 3). No inflammatory changes were noted. The homogeneous eosinophilic material found in both tumors stained positive for Congo red and displayed a distinct green birefringence under polarized light. (Figs. 4A, 4B, 5A).
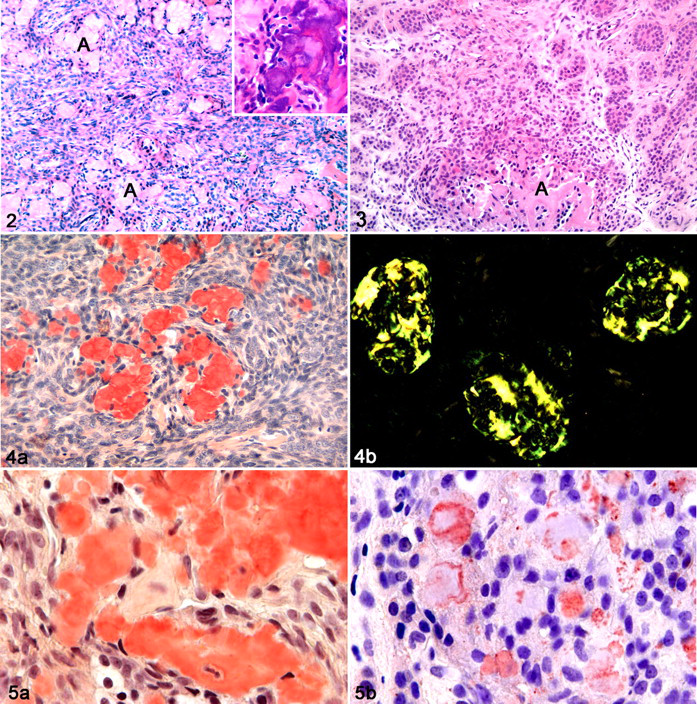
Tumor, left mandible, cat No. 1. Tumor cells are arranged in nests or streams, and a high amount of amyloid (A) is detectable. HE. Inset: Dentinoid formation with prominent mineralization. HE.
To further characterize the neoplastic cells and the Congo red positive material, immunohistochemistry was performed by the avidin-biotin-peroxidase method. Antibodies directed against the following were used: pancytokeratin (1:2500, monoclonal mouse anti-human), cytokeratin AE1/AE3 (1:500, monoclonal mouse anti-human), vimentin (1:100, monoclonal mouse anti-porcine; all DakoCytomation, Glostrup, Denmark), laminin (1:75, polyclonal rabbit anti-mouse, Quartett, Berlin, Germany), AA amyloid (1:30, monoclonal mouse anti-human), AL (λ, κ) amyloid (1:1000, polyclonal rabbit anti-human), and odontogenic ameloblast-associated protein (ODAM; formerly, Pindborg amyloid or APin; 1:1000, polyclonal rabbit anti-human; all amYmed GmbH, Martinsried, Germany). 10 Furthermore, 3,3′-diaminobenzidine was used as chromogen for pancytokeratin, cytokeratin AE1/AE3, vimentin, and laminin, and 3-amino-9-ethyl-carbazole was used for AA, ALλ, and ALκ amyloid and ODAM. 4,6 Tumor cells of both animals were positive for cytokeratin, whereas the stroma stained strongly positive for vimentin and weakly for laminin. The amyloid material was negative for cytokeratins and for AL and AA amyloid in both tumors. However, cat No. 1 demonstrated a weakly positive immunolabeling of amyloid with vimentin and laminin. Interestingly, the amyloid material of cat No. 2 stained moderately positive with antibodies against the ODAM (Fig. 5B), whereas the amyloid in cat No. 1 lacked a respective reaction (Table 1).
Immunohistochemical reactivity of tumor cells, stroma, and amyloid in the tumor of cat No. 1 and cat No. 2. a
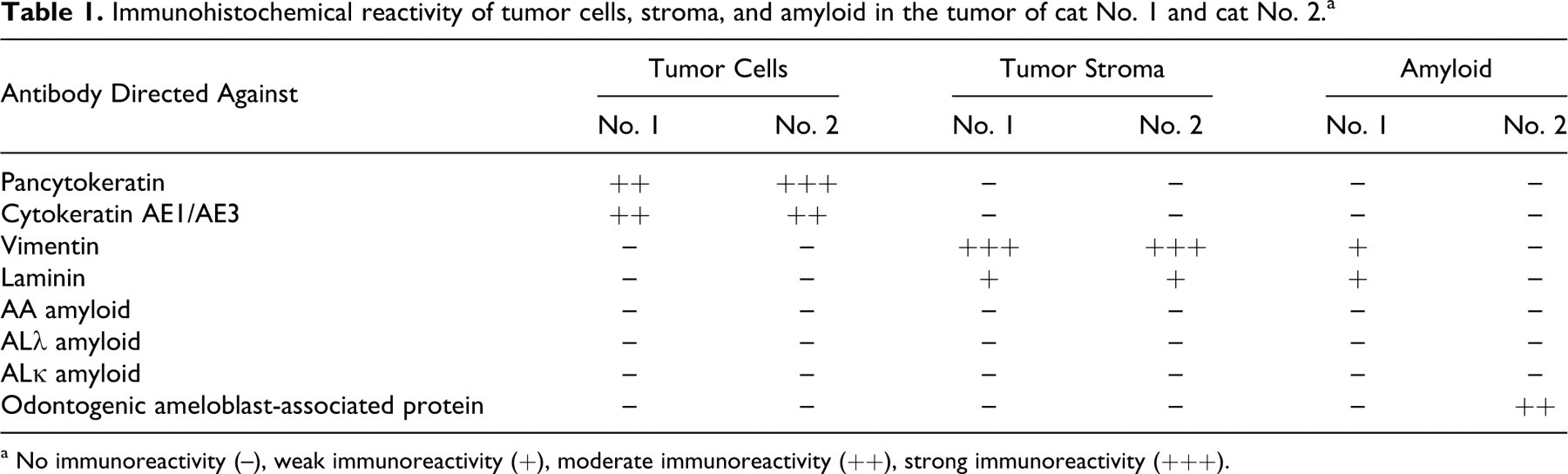
a No immunoreactivity (–), weak immunoreactivity (+), moderate immunoreactivity (++), strong immunoreactivity (+++).
Diagnosis
APOTs with and without ODAM.
Discussion
The APOT is a rare, benign, slowly progressing but often locally invasive epithelial odontogenic neoplasia, which has been reported in dogs, cats, and a bengal tiger (Panthera tigris tigris). 1,3,5,7,9,17 It is often referred to as calcifying epithelial odontogenic tumor (CEOT), according to its similarities to the human counterpart. 3,18,19 However, the human CEOT, or Pindborg tumor, is not totally equivalent to the CEOT in animals. 5,14 Animal CEOTs often show palisaded basal cells and stellate reticulum, 5 neither of which is common in human CEOTs. 5,11 Moreover, nuclear pleomorphism of tumor cells is rather typical for human CEOTs but was not prominent in the present cases. 14 Therefore, APOT was introduced as an alternative term for CEOT or Pindborg tumor in animals. 5,7 The most prominent feature to distinguish APOTs and CEOTs from ameloblastomas is the varying amounts of amyloid, which tends to calcify (as in the present cases). 7,9,15,20 In addition, stellate reticulum is found less frequently in APOTs than in ameloblastomas. 5,7
The tumors of both cats differed in size and morphology. In cat No. 1, the tumor was comparatively small and showed a rather disordered arrangement of epithelial cells in nests or streams, as well as a high amount of amyloid; the neoplasia of cat No. 2, however, extended into most of the whole left maxilla and consisted of palisading tumor cells and only a few patches of amyloid. These findings suggest that APOTs display a variable morphology in cats; yet, the different sizes of the tumors and the fact that only incisional biopsies were taken might in part account for the varied morphologies observed. The origin of the amyloid found in APOTs and CEOTs remains speculative. Researchers have suggested that it is a glycoprotein complex secreted by the neoplastic cells. 1,10 To further characterize the amyloid deposits in both feline APOTs and, in particular, to compare the feline amyloid to the amyloid deposits found in the human Pindborg tumor, immunohistochemical investigations were performed. Previous studies revealed a positive cytokeratin immunoreactivity of the amyloid in CEOTs and APOTs, which led to the assumption that the amyloid might be of epithelial origin. 2,3,7 These observations could not be confirmed in the present cases. Similarly, Solomon et al described a human Pindborg tumor containing cytokeratin-negative amyloid. 16 Using microanalytic techniques, they found that the CEOT-associated amyloid consists of a unique (till then) nondescribed protein, which they designated APin in recognition of Dr Pindborg’s initial report of congophilic deposits in CEOTs. 16 These assumptions were verified by a positive immunohistochemical reaction of the amyloid to antibodies against APin and so confirmed the unique nature of the human CEOT-related amyloid. 16 Recent studies have shown that the amyloid associated with CEOTs is composed of N-terminal fragments of the ODAM. 8,12 Therefore, this unique odontogenic amyloid, formerly designated APin, is now termed ODAM. 8 Apart from CEOTs, ODAM is highly expressed by mature ameloblasts and is present in the enamel organ as well as in ameloblastomas. 8,13 These data lead to the assumption that ODAM plays an essential role in normal and tumor-related odontogenesis. 8 To see whether this could also apply to the cat, we performed an immunohistochemistry using APin/ODAM-specific antibodies, which revealed a positive reaction of the amyloid in cat No. 2 but not in cat No. 1. This finding indicates a close similarity of the tumor in cat No. 2 to the human Pindborg tumor/CEOT, despite the described morphological differences. It also shows that the feline tumors were not only morphologically different but might have also contained amyloid deposits of different origin. However, because the amyloid of both tumors did not react with cytokeratin, AA, or AL antibodies, the origin of the amyloid in cat No. 1 remains unclear.
In sum, the feline APOTs displayed different morphological phenotypes, and their amyloid deposits seemed to be of different origin, resembling at least in one case the amyloid in human Pindborg tumors.
Footnotes
Acknowledgements
We wish to thank Professor Reinhold P. Linke from the Reference Center of Amyloid Diseases, amYmed GmbH, Martinsried, Germany, for performing and interpreting the immunohistochemical investigations and for his outstanding scientific support concerning the odontogenic ameloblast-associated protein (formerly, APin). We also thank Bettina Buck for her excellent technical assistance.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
