Abstract
Thirty-four peripheral nerve sheath tumors of four domesticated animal species were characterized and assayed for point mutation of the
Peripheral nervous system (PNS) tumors are relatively common in humans, but occur infrequently in domestic animals, with most reports in cattle and dogs. 8,10,19 In humans, schwannoma accounts for an estimated 8% of intracranial 3 and 29% of primary spinal tumors. 8,25 In humans, clinical and morphopathologic distinctions are recognized between schwannoma (World Health Organization [WHO] grade I), neurofibroma (WHO grade I), and malignant peripheral nerve sheath tumor (MPNST) WHO grade III and IV classification. Schwannoma in humans is considered a benign slowly growing neoplasm located anywhere in the PNS, but its most frequent intracranial site is the vestibular segment of the eighth cranial nerve. Neurofibroma is usually indolent, with a favorable prognosis. Multiple neurofibromas in humans are the hallmark of neurofibromatosis type 1 (von Recklinghausen's disease). MPNSTs are morphologically variable neoplasms with a distinct tendency toward divergent mesenchymal differentiation. More than one half of all humans affected have inherited mutations of the NF1 gene. 25
In contrast, in the veterinary literature, these differentiation patterns and subclassifications are less recognized, and they are all referred to as nerve sheath tumors or schwannomas. 4,9,19 Recent WHO classification of tumors of the nervous system and mesenchymal tumors of skin and soft tissues in domestic animals combine malignant and benign forms of neurofibroma and schwannoma under the title of peripheral nerve sheath tumor (PNST). 7,12
Macroscopically, benign animal schwannomas are usually encapsulated, globoid, lobulated masses of variable size and shape, depending on location of the neoplasm and animal species affected. 1,4,5,15–17 Malignant schwannomas are usually nonencapsulated and infiltrative tumors. Microscopically, the classical description of schwannoma is characterized by Antoni type A and B patterns and Verocay body formation. Antoni type A pattern is characterized by a parallel arrangement of Schwann cell fusiform nuclei, giving a palisaded pattern. Antoni type B areas are more loosely arraged with fewer cells, smaller, round dark nuclei, and space between cells. Verocay bodies are formed by palisaded arrays of tumor cell nuclei separated by dense, closely aligned, eosinophilic cell processes. 4,19–24 Malignant schwannomas are composed of less differentiated, pleomorphic, anaplastic populations of cells that infiltrate adjacent tissues or that metastasize to other organs. Schwannomas lacking these classical morphologic patterns are often difficult to differentiate from other spindle cell tumors such as fibroma, canine hemangiopericytoma, fibrous histiocytoma, melanosarcoma, and leiomyoma/sarcoma. In some cases, ultrastructural, immunohistochemical, and, when appropriate, genetic analyses are often required for an accurate diagnosis. MPNSTs are most commonly seen in dogs, and usually involve the brachial or lumbar plexus, often compressing and invading the spinal cord and brain. Histologically, cells are arranged in fascicles, whorls, or sheets, and range from spindle-shaped to plump fusiform cells with variable amount of fibrillar collagenous stroma to epithelioid arrangements. 12
This paper describes gross, histopathologic, and immunohistochemical features of PNSTs in various animal species. In addition, a specific genetic marker, a single point mutation of the
Materials and Methods
Histology and immunohistochemistry
Tumors of the PNS were derived from necropsy (16 animals) and surgical biopsy (18 animals) cases. They had been formalin-fixed and paraffin-embedded (Table 1) and were collected from the archival tissue repository of Texas A&M University, Department of Veterinary Pathobiology. During a period of 13 years (1986–1999), 50 cases diagnosed as neurofibromas, neurofibrosarcomas, undifferentiated sarcomas, PNSTs, or schwannomas were retrieved. New histologic sections were cut from each archival block and stained with hematoxylin and eosin (HE) and additional sections were prepared for immunohistochemistry (vimentin, S100 protein, neuron-specific enolase [NSE], and glial fibrillary acidic protein [GFAP]). Special stains such as Holmes' silver method for nerve cells, Alcian blue, and Masson-Fontana where used when required as ancillary methods. Examined neoplasms were reclassified as schwannomas based on their morphologic pattern and immunohistochemistry. For morphologic diagnosis of PNST we followed the recent
Animal species distribution and characterization of peripheral nerve sheath tumors. ∗
∗ V = Vimentin; GFAP = glial fibrillary acidic protein; NSE = neuron-specific enolase; MPNST = malignant peripheral nerve sheet tumor.
Immunohistochemical evaluation was performed as previously reported. 18 Briefly, serial histologic sections (5 µm thick) were deparaffinized in xylene, rehydrated through graded alcohols, and treated with 0.5% hydrogen peroxide-methanol for 30 minutes to quench endogenous peroxidase activity. To block nonspecific sites, tissue sections were incubated with 2–4% normal horse serum for 30 minutes. Primary antibodies against mouse-vimentin (1:50 dilution), rabbit S100 protein (1:150), and rabbit GFAP (1:300) were obtained from Dako (Carpinteria, CA). Biotinylated rabbit and mouse antisera (Dako) were used as secondary antibodies, and 3,3′-diaminobenzidine tetrahydrochloride-hydrogen peroxide was used as chromogen. Sections were slightly counterstained with Meyer's hematoxylin. For negative controls, the primary antibodies were replaced with appropriate normal serum. Tissue sections known to express the relevant antigens served as positive controls.
Analysis of neu oncogene
The DNA from paraffin-embedded tissues was isolated as previously described. 6 Briefly, HE-stained tissue sections were viewed with an Olympus microscope and the area of interest (e.g., areas of hypercelullarity, devoid of necrosis, hemorrhage, or artifacts) was marked with black ink on the back of the glass slide. A serial unstained section was examined under an Olympus inverted microscope and an identical area (identified on the HE section) was delineated using an Olympus ink marker objective. The marked area was microdisected with a thin, sterile, sharp spatula, placed in Eppendorf tubes, and digested according to previously published methods. 6
Genomic DNA extracted from an ENU-induced trigeminal nerve malignant schwannoma in a BD-IV rat, designated P41, served as a positive control for a codon 664 point-mutated c-
The polymerase chain reaction (PCR) was used to amplify DNA fragments spanning potential mutation sites from the c-
Single bands of 138-bp length were detected after amplification of DNA from both rat trigeminal and domestic animal schwannomas, using restriction fragment length polymorphism (RFLP) analysis. Point mutations that altered the DNA restriction sites may be detected as migration differences of restriction fragments in agarose gels.
Results
As a result of this investigation, 34 tumors were classified as PNSTs, of which 26 tumors were classified as benign schwannoma, 6 as malignant schwannoma, and 2 as MPNST.
Species distribution and anatomical location of schwannomas and MPNSTs in our study are given in Table 1. According to their necropsy or biopsy reports, the macroscopic appearance of schwannomas varied depending on animal species affected and location of the neoplasms. Their size ranged from few centimeters to 46 cm in diameter and weight ranged from 25 g to 26 kg. The largest were found in horses (horse No. 7: 31 × 46-cm, 26-kg intra-abdominal mass) and cattle (cow No. 12: 45-cm-diameter, 25-kg mediastinal mass). The largest tumor in a dog was an intrathoracic schwannoma attached to the heart (dog No. 23: 13 × 17 cm and weighing 2 kg). In general, the tumors were encapsulated, globoid or lobulated masses. On sections, most of the tumors were solid and lobulated; the largest one had a central core of hemorrhage and necrosis.
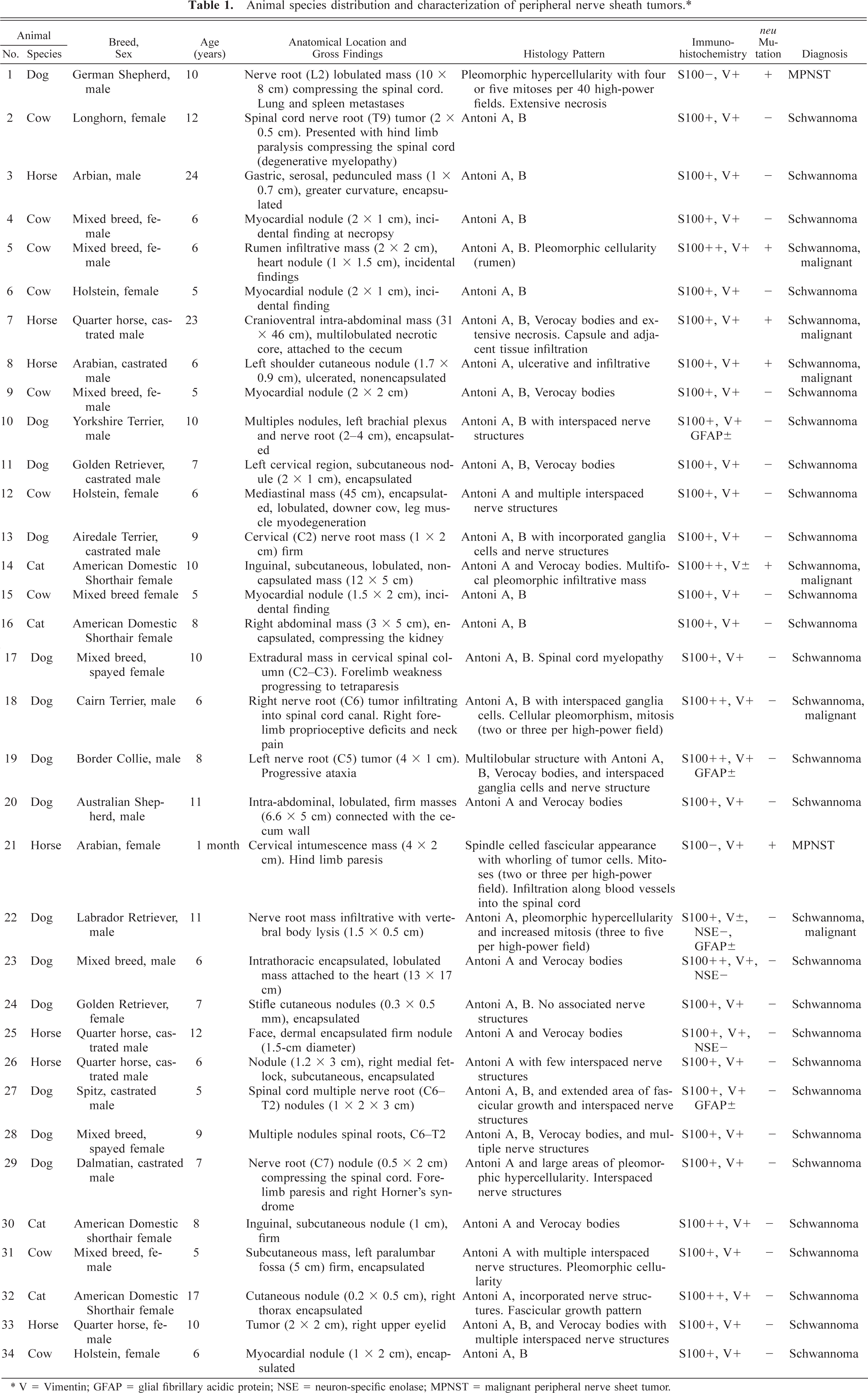
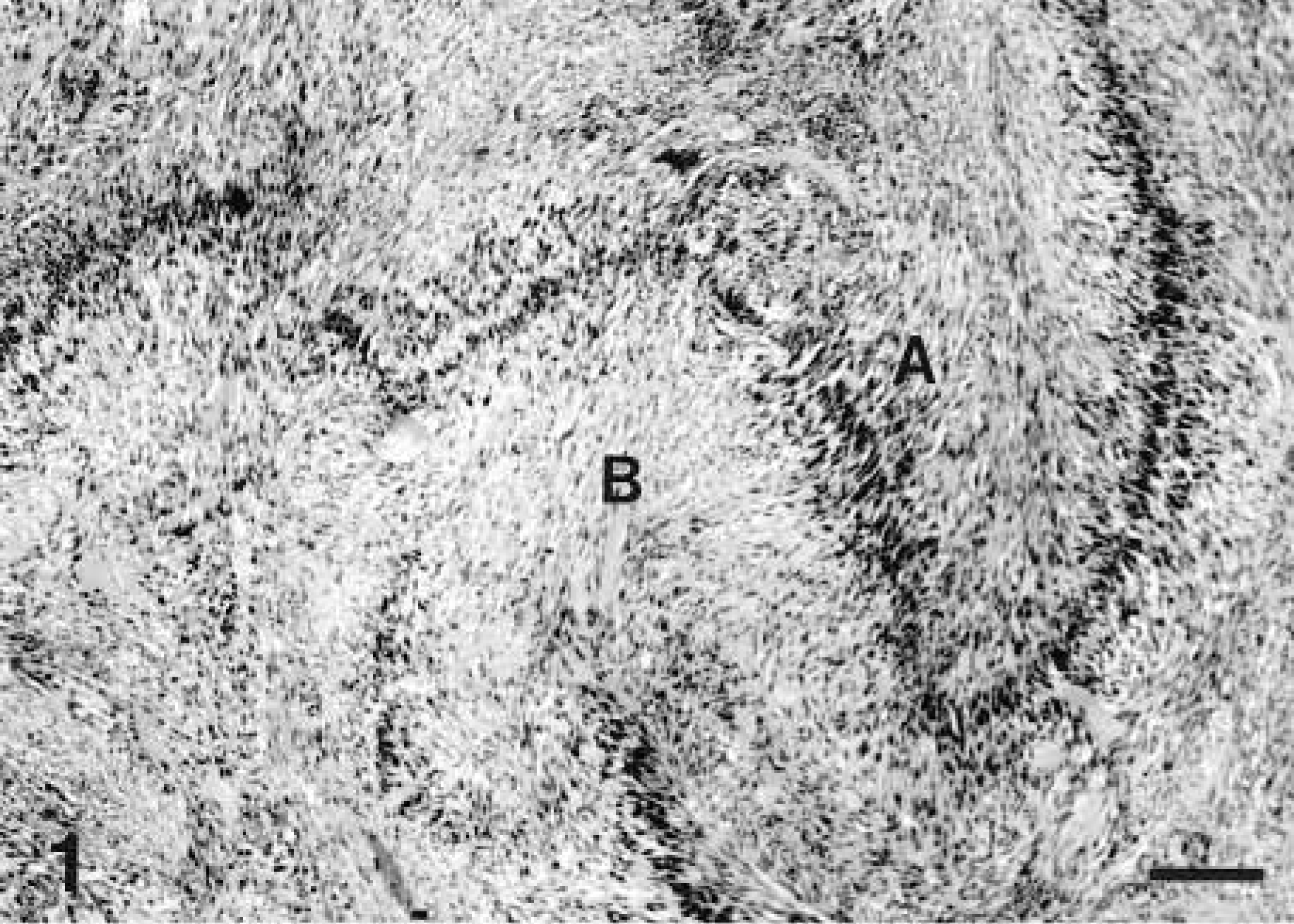
Microscopically, schwannomas were characterized by a pseudolobulated, pleomorphic pattern composed primarily of spindle-shaped cells either arranged in small dense palisades oriented in different directions (Antoni type A pattern) or loosely arranged tumor cells (Antoni type B pattern) (Figs. 1–3). Verocay bodies were also formed by parallel bodies of tumor cell nuclei separated by dense eosinophilic tissue (Fig. 2). This classical histologic pattern was less common in dogs where the predominant pattern was mostly of Antoni type A or a cellular fascicular growth pattern. In some tumors, the neoplastic cells were arranged in palisades or concentrically around small blood vessels, whereas polygonal or pleomorphic tumor cells without a specific arrangement predominated in other tumors or locations. The neoplastic cells had relatively abundant, faintly eosinophilic cytoplasm with indistinct cell margins and normochromatic spindle-shaped nuclei that were tapered instead of blunt-ended, in contrast to those of smooth muscle cells. Nuclear pleomorphism and mitotic figures varied from case to case; in some tumors, cellular atypia and increased nmbers of mitosis were prominent.
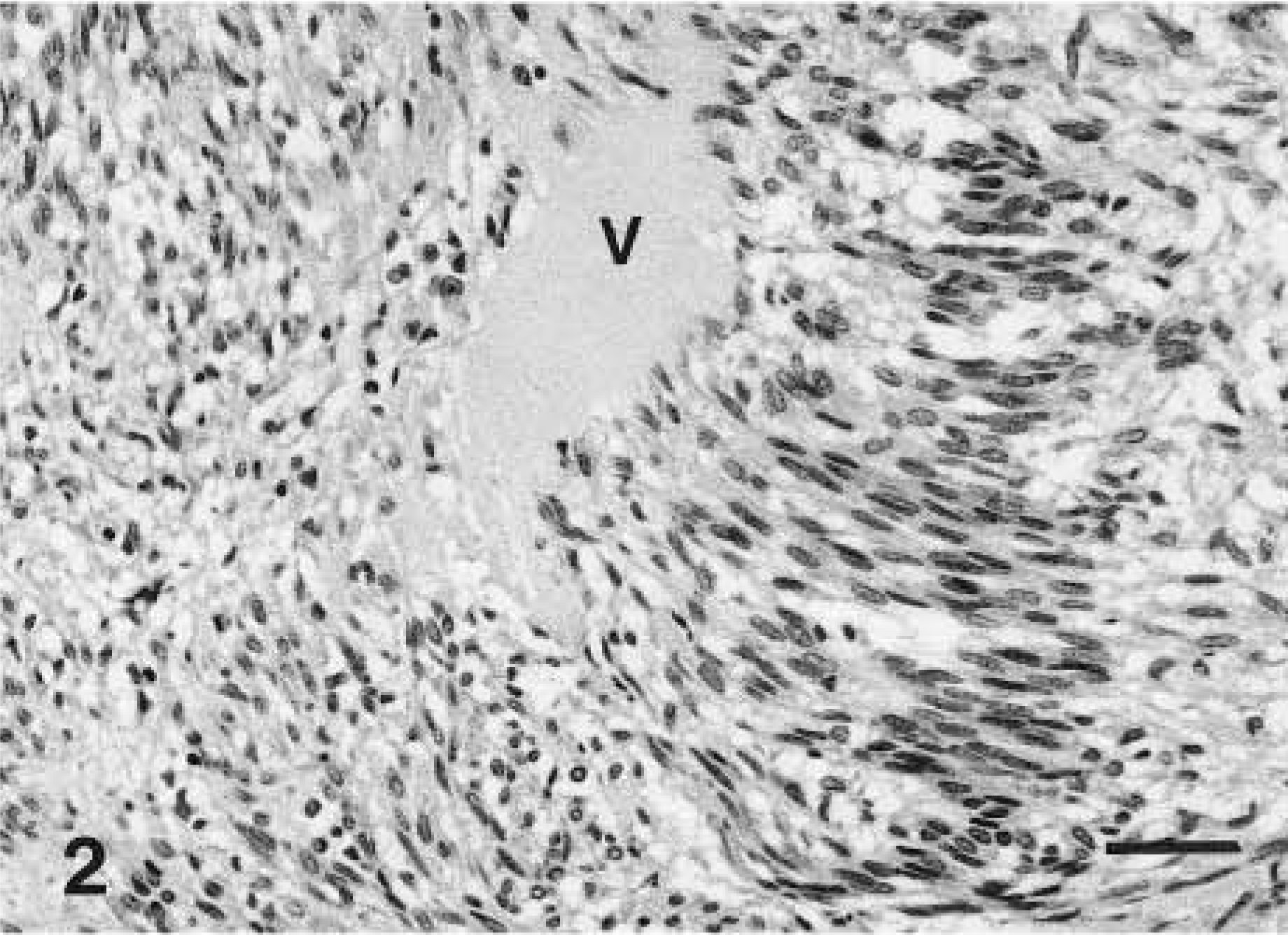
Schwannoma; horse No. 3. Typical histologic features of a benign schwannoma from a 24-year-old Arabian horse. This tumor was located on the greater curvature of the serosa of the stomach. Histology of this tumor illustrates a classical pattern of a schwannoma charaterized by cell nuclei forming a typical hypercellular palisading pattern (Antoni A) and hypocellular areas (Antoni B). HE. Bar = 200 µm.
Schwannoma; horse No. 3. Histologic features of a Verocay body (V) formed by palisaded arrays of tumor cell nuclei separated by dense, closely aligned, eosinophilic cells processes, taken from a schwannoma from a 24-year-old Arabian horse. HE. Bar = 200 µm.
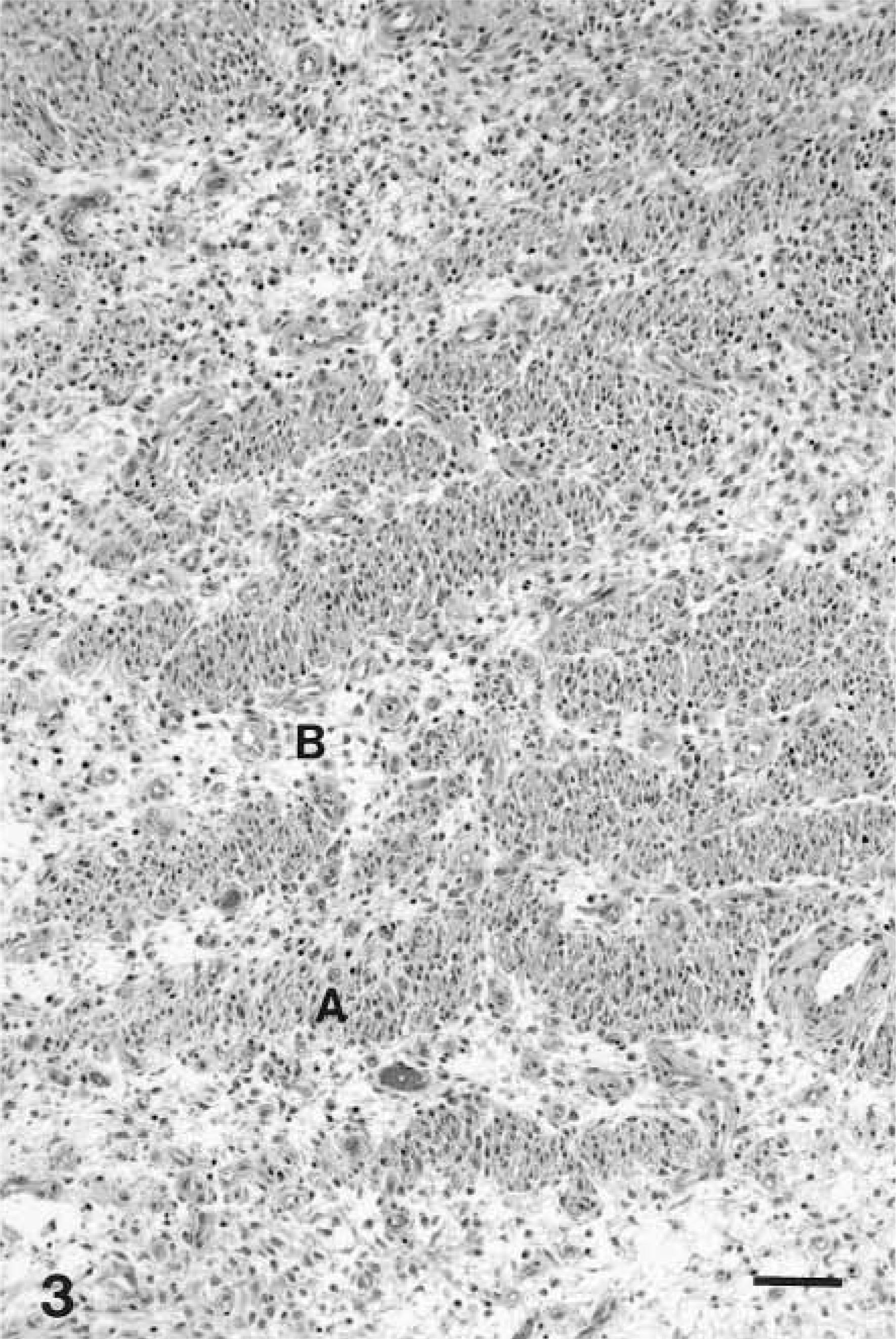
Schwannoma; dog No. 23. Histologic appearance of an intrathoracic schwannoma attached to the heart (surgical specimen) from a 5-year-old dog. Fig. 3A Biphasic pattern with hypercellular Antoni A areas. Fig. 3B Hypocellular Antoni B areas. Schwannoma cell nuclei are arranged in a typical palisading pattern. HE. Bar = 100 µm.
A dimorphic pattern composed of epithelioid and spindiloid cells was recognized in a benign canine schwannoma arising from the left fifth cervical spinal nerve root (dog No. 19). Besides the classical pattern (Antoni A and B, and Verocay body), this tumor exhibited areas composed of epithelioid cells arranged in densely packed nests showing pleomorphism, variably shaped and sized nuclei, and abundant eosinophilic cytoplasm. The central epithelioid cell population was surrounded by a field of spindiloid cells among which preexisting neurons (ganglia) were visualized. Epithelioid cells expressed vimentin and were negative for S100 protein. The spindiloid cells expressed vimentin and S100 protein and isolated cells expressed GFAP. This tumor was not invasive.
Six schwannomas were classified as malignant (tumor Nos. 5, 7, 8, 14, 18, 22) based on their cellular pleomorphism, anaplasia, increased mitoses, and local invasion. Malignant forms of schwannoma were poorly circumscribed and infiltrative. The species distribution and anatomical location of malignant schwannomas and MPNSTs,
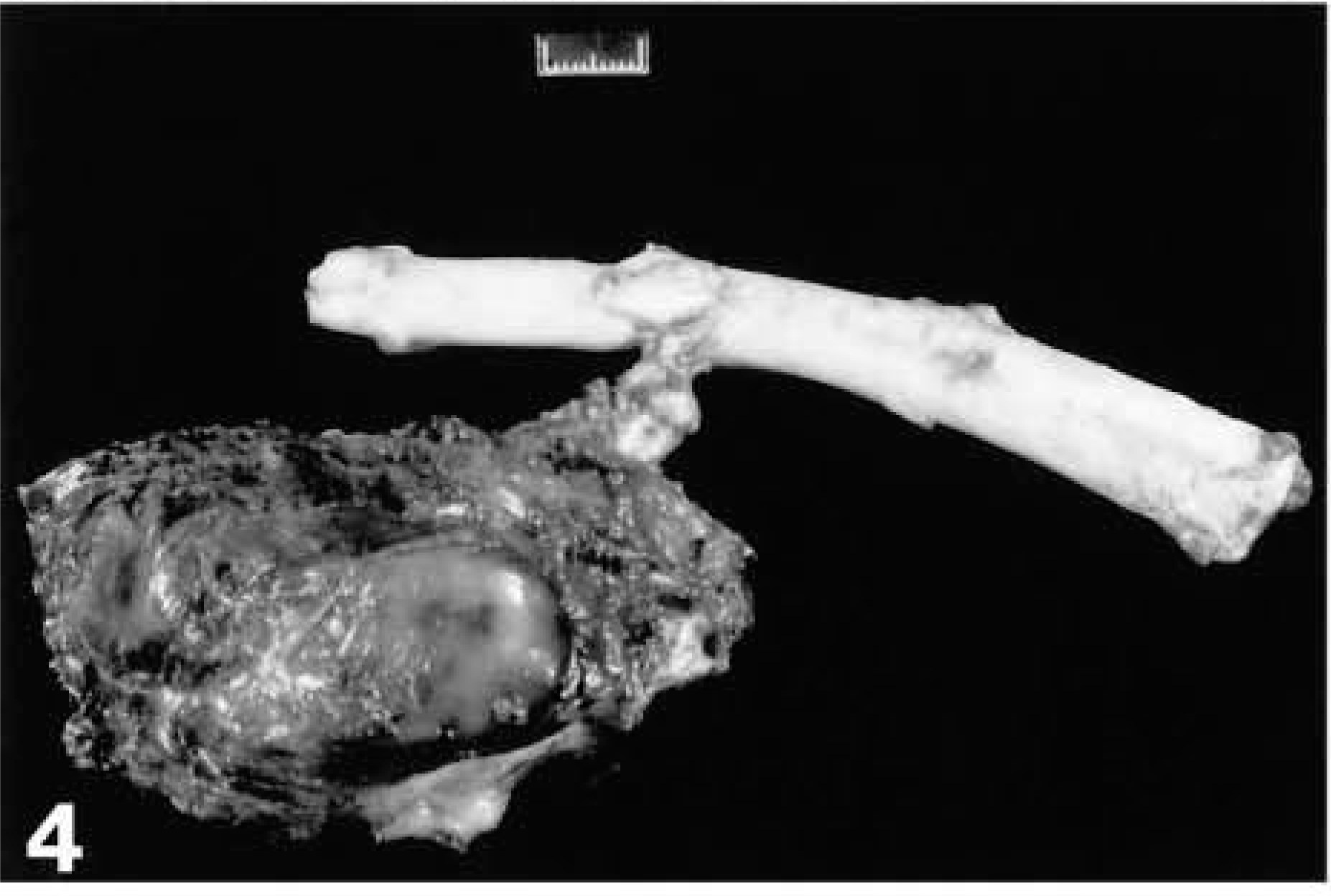
Malignant peripheral nerve sheath tumor (MPNST); dog No. 1. Gross appearance of a MPNST in a 10-year-old German Shepard Dog. This tumor developed from the second lumbar vertebral spinal cord root and metastasized to the lung and spleen. The tumor exhibited a
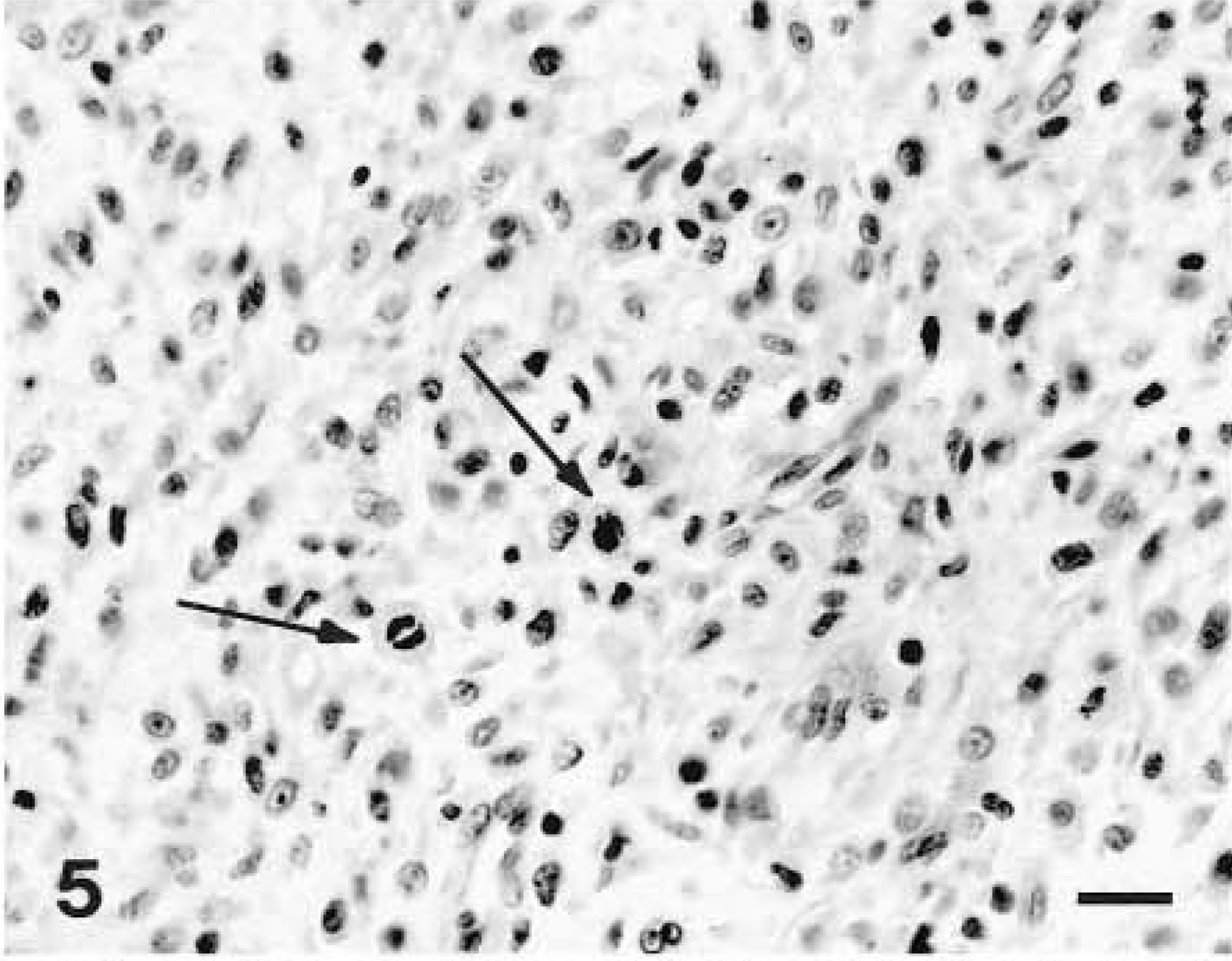
Malignant peripheral nerve sheath tumor; dog No. 1. Histologic section from the tumor illustrated in Fig. 4 showing densely packed epithelioid cells demonstrating pleomorphism, anaplasia, and mitotic figures (arrows). HE. Bar = 80 µm.
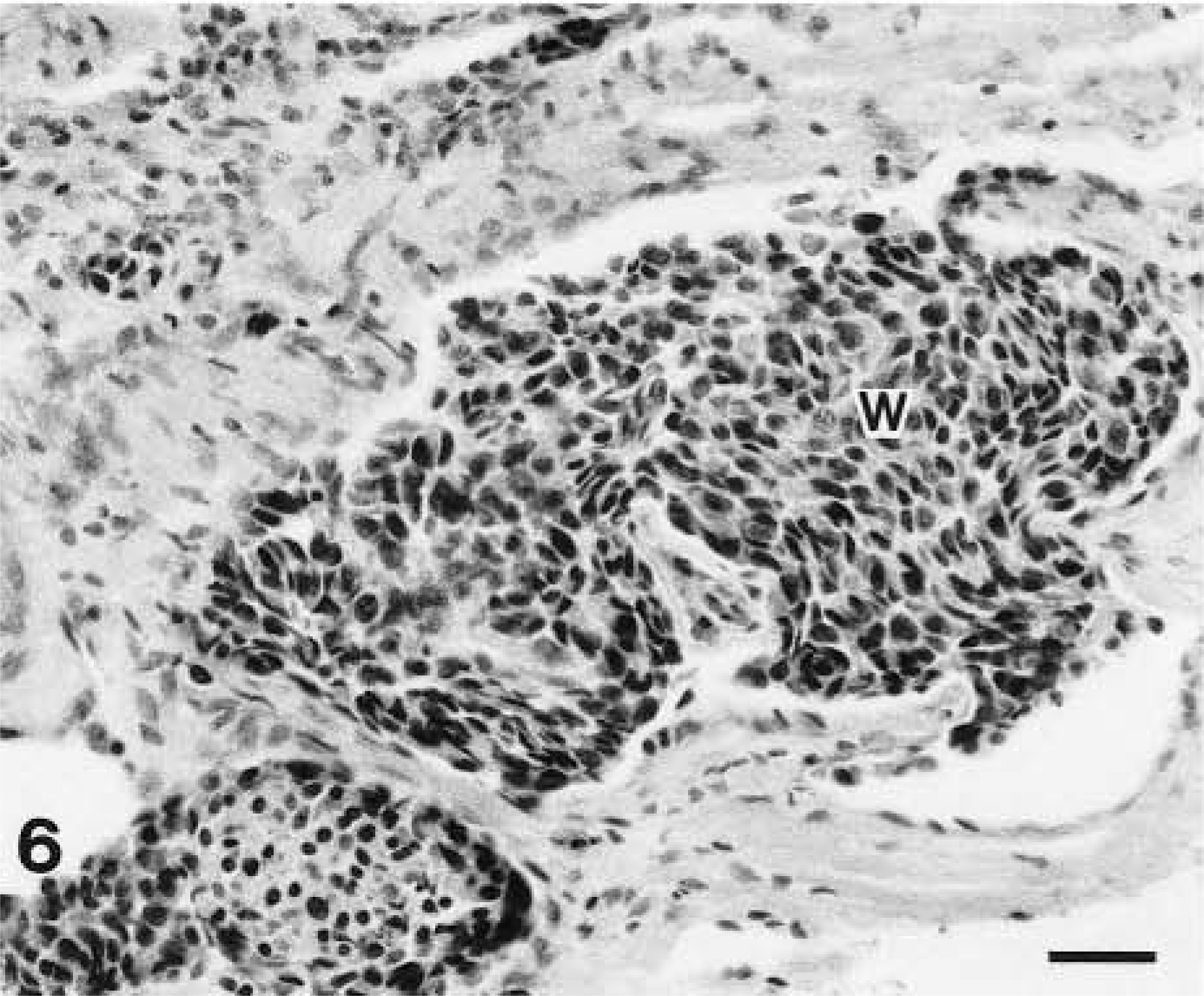
Malignant peripheral nerve sheath tumor; dog No. 1. Histologic appearance from the tumor mass illustrated in Fig. 4 demonstrating intravascular metastatic emboli (W). HE. Bar = 100 µm.
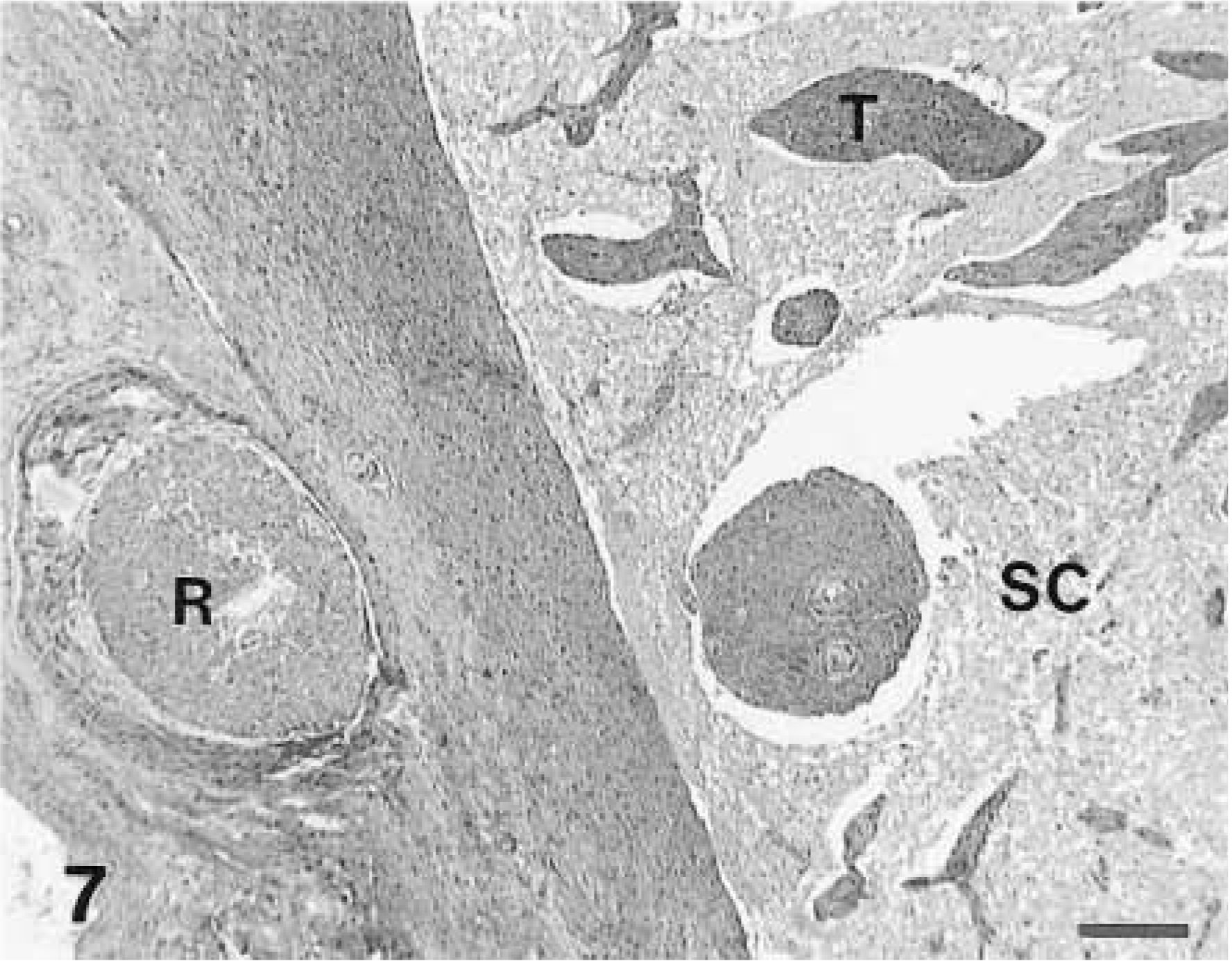
Malignant peripheral nerve sheath tumor; horse No. 21. Extramedullary tumor infiltrates along the Virchow-Robbin spaces (T) into the spinal cord white matter (SC). An extramedullary nerve root fascicle (R) is apparent. HE. Bar = 400 µm.
Immunohistochemistry revealed that all schwannomas included in this study expressed vimentin and S100 protein, but the staining intensity was variable (Figs. 8, 9). In addition, labeling for GFAP was weak and sparse in four of the canine schwannomas (dog Nos. 10, 17, 22, 27) originating from the spinal cord roots.
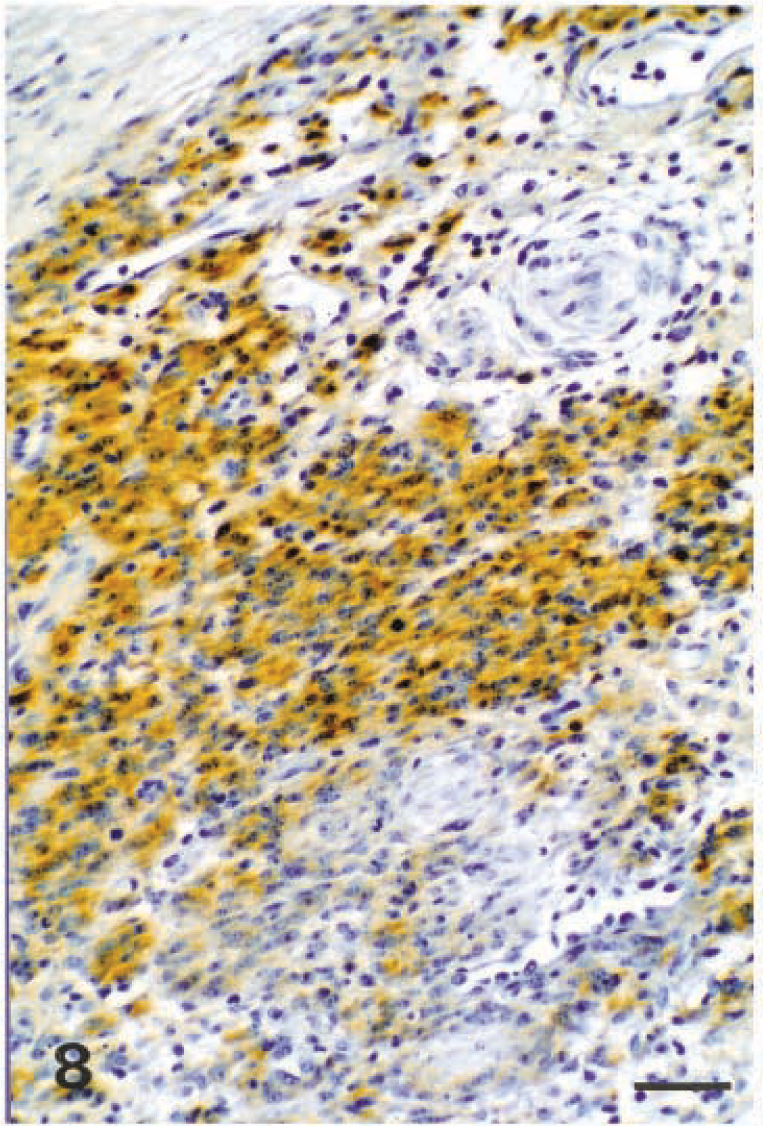
Schwannoma; dog No. 22. S100 protein immunolabeling of a canine malignant schwannoma. Avidin–biotin–peroxidase complex method and methyl blue counterstain. Bar = 125 µm.
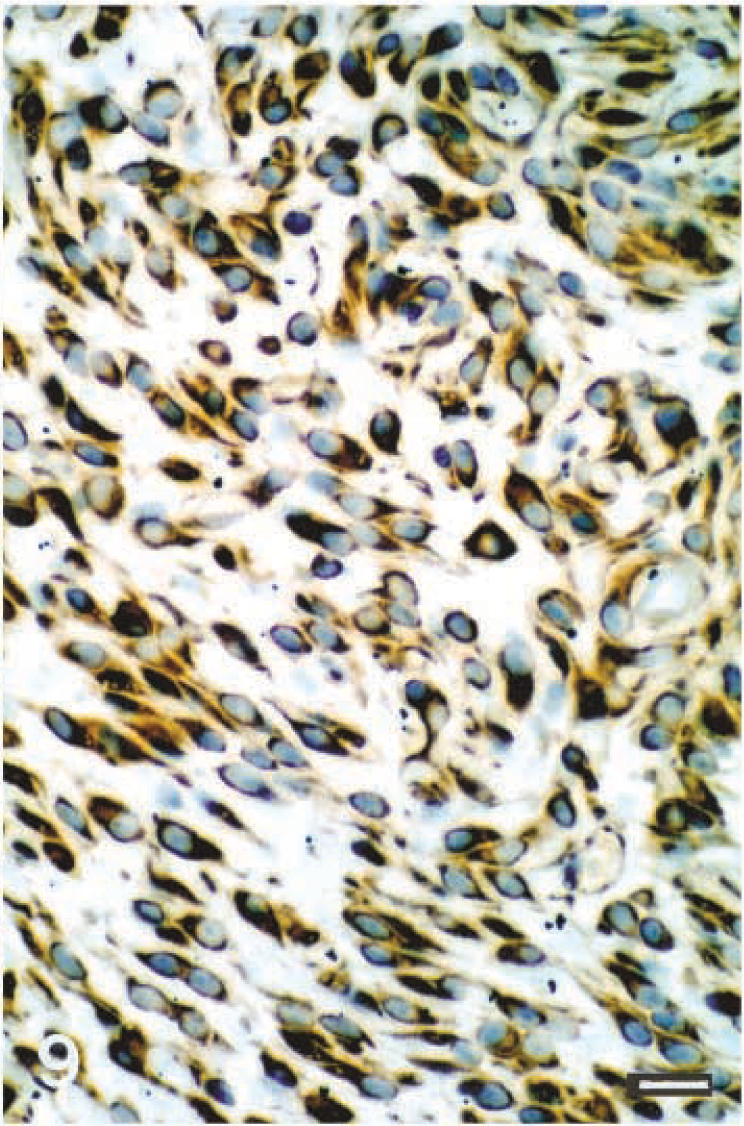
Schwannoma; dog No. 22. Vimentin immunolabeling of canine malignant schwannoma (case 22). Avidin–biotin–peroxidase complex method and methyl blue counterstain. Bar = 50 µm.
From a total of 34 PNSTs examined, 8 tumors were diagnosed as malignant (6 schwannomas and 2 MPNSTs) of which 6 (4 schwannomas and 2 MPNSTs) were found to have a specific point mutation of the
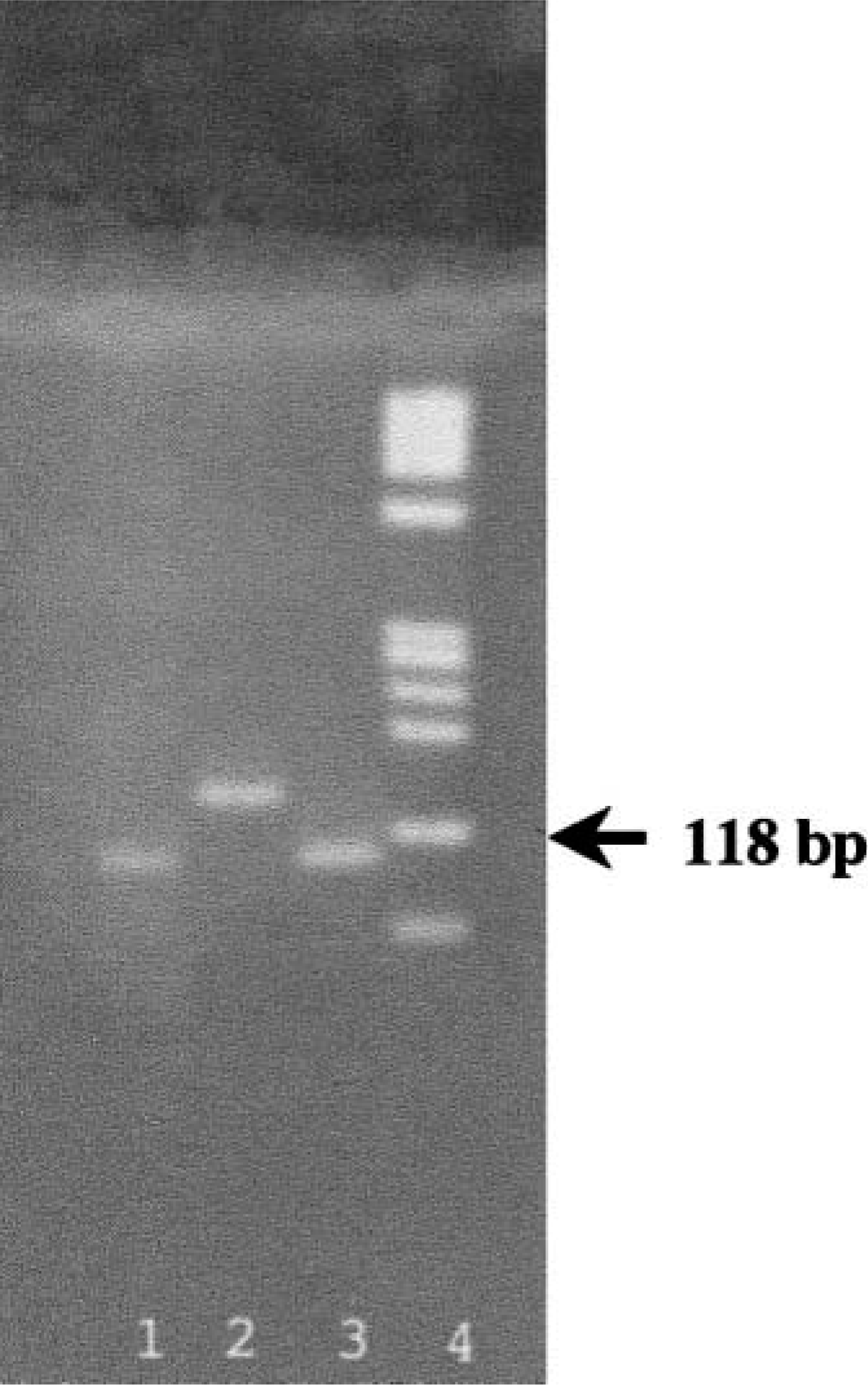
An example of the
Discussion
Initially, 50 PNSTs from archival formalin-fixed tissue were retrieved for morphoimmunohistochemical characterization and assayed for point mutation of the
The morphologic phenotype of PNSTs in domesticated animals is not always similar to the human counterpart. In the canine PNSTs, the presence of classical histologic patterns of Antoni type A and B and Verocay body are not the predominant patterns. Often, the histologic pattern is pleomorphic with cells arranged in fascicles, whorls, or sheets, and the cells range from spindle-shaped to epithelioid cells. This histologic pattern of canine schwannoma is more similar to the rat morphologic pattern than to the human pattern. Shwannomas of cattle and horses are more histologically similar to the human type than is the canine schwannoma.
Our study demonstrated that malignant schwannomas and MPNSTs in domesticated animals, in addition to having a characteristic morphologic pattern, carry a specific genetic marker, a point mutation of the
Previous reports in rats
2,10,13,14
have suggested the cell lineage-specific involvement of a T.A to A.T transversion mutation at nucleotide 2012 of the transmembrane region of the
This mutation is diagnostic for ENU-induced rat schwannomas and in the process of oncogenesis characterized a subset of Schwann cells that exhibit unrestrained proliferative activity in contrast to the differentiating wild-type counterpart cells. Schwann cells bearing the
In summary, our study confirmed the presence of
Footnotes
Acknowledgements
This work was supported by an interdisciplinary enhancement grant (246305) from Texas A&M University. We thank Mrs. Vollmar Rosemary and Ms. Ambrus Sandy for their technical assistance with histology and immunohistochemistry.
