Abstract
Mycobacterium tuberculosis is an old enemy of the human race, with evidence of infection observed as early as 5000 years ago. Although more host-restricted than Mycobacterium bovis, which can infect all warm-blooded vertebrates, M. tuberculosis can infect, and cause morbidity and mortality in, several veterinary species as well. As M. tuberculosis is one of the earliest described bacterial pathogens, the literature describing this organism is vast and overwhelming. This review strives to distill what is currently known about this bacterium and the disease it causes for the veterinary pathologist.
Naturally, we’re disposed to think about diseases just from our own point of view: what can we do to save ourselves and to kill the microbes? Let’s stamp out the scoundrels, and never mind what their motives are! In life in general, though, one has to understand the enemy in order to beat him, and that’s especially true in medicine.
—Jared Diamond in Guns, Germs, and Steel: The Fates of Human Societies 54
Tuberculosis (TB) is an ancient disease with a history interwoven with the evolution and migration of mankind, as well as with the origins of microbiology. The main etiologic agent of TB, Mycobacterium tuberculosis (Mtb), is thought to have evolved from an early progenitor in East Africa as early as 3 million years ago. 90 By the early 1800s, TB epidemics ravaged much of Europe and North America, resulting in 800 to 1000 deaths per 100 000 per year. 120 Today, approximately 9 million new cases of TB are identified per year, with almost 2 million deaths related to TB, making Mtb the single greatest cause of mortality due to a bacterial pathogen. 70
Mtb is a major component of the history of microbiology. In 1882, Robert Koch demonstrated the tubercle bacillus during a presentation of his famous postulates, which have since set the standard for the demonstration of infectious etiology. 119 Koch also developed tuberculin, a glycerine extract of pure Mtb cultures, which, although a failure in its proposed use as a therapy, could be used to detect latent infections by the measurement of skin reactions at the injection site. Today, tuberculin has been replaced by purified protein derivative (PPD), a precipitate of sterilized Mtb culture filtrate, for testing in humans.
A major breakthrough in the fight against TB occurred when Calmette and Guérin developed the attenuated vaccine strain, Bacille Calmette-Guérin (BCG), through multiple passages of Mycobacterium bovis on ox bile and glycerol-soaked potato slices between 1906 and 1919. 32 Although highly effective in preventing the childhood form of TB, the BCG vaccine has variable efficacy in adults. 67 As an attenuated strain, BCG has been enormously useful in TB research because, unlike Mtb, BCG does not require biosafety level 3 facilities. The other major breakthrough was the discovery of the antimycobacterial drugs streptomycin, isoniazid, the rifamycins, and pyrazinamide, in 1944, 1952, 1957, and 1980, respectively. 207 These discoveries led to a new era in TB prevention, research, and treatment.
Today, the absence of a completely protective TB vaccine, the slow development of new antimycobacterial drugs, the need for prolonged therapy regimens, and the emergence of multidrug-resistant (MDR) and extremely drug-resistant (XDR) strains of Mtb are issues that highlight the reemerging TB crisis. The MDR strains account for almost 5% of TB cases. 70 Because veterinary species are infected by Mtb, and because we can apply what is known about Mtb to the fight against M. bovis, which has reemerged in the United States and other countries due to persistence within wildlife reservoirs 218 and for which MDR strains have been reported, 105 it is critical that veterinary pathologists have a good basic understanding of Mtb. This review therefore strives to summarize the key components of the microbiology, cell biology, immunology, and pathology of Mtb that are pertinent for the veterinary pathologist.
The Bacterium
Mycobacteria are nonmotile, nonsporulating, weakly gram-positive, acid-fast bacilli that appear microscopically as straight or slightly curved rods, 1 to 4 μm in length and 0.3 to 0.6 μm wide. Mycobacteria are within the order Actinomycetales, which it shares with bacteria such as Corynebacterium, Nocardia, and Rhodococcus. These bacteria also express unique mycolic acids in the cell envelope that play a critical role in the structure and function of the cell wall. 14 The waxy cell wall confers many of the unique characteristics of this genus: acid-fastness, extreme hydrophobicity, resistance to drying, acidity/alkalinity, and many antibiotics, as well as distinctive immunostimulatory properties. 45
Mtb is a member of the slow-growing pathogenic mycobacterial species, characterized by a 12- to 24-hour division rate and prolonged culture period on agar of up to 21 days. Why Mtb grows so slowly is not well understood. Proposed mechanisms include limitation of nutrient uptake through the highly impermeable cell wall and slow rates of RNA synthesis. 96 During experimental infections, its metabolism can shift from an aerobic, carbohydrate-metabolizing mode to one that is microaerophilic and lipid metabolizing. 25 Mycobacteria are facultative intracellular bacteria that multiply within phagocytic cells, particularly macrophages and monocytes. Although many mycobacterial species are environmental, Mtb is strictly parasitic.
Mtb is a member of the M. tuberculosis complex, which is defined as the etiologic agents of TB in distinct hosts, and also includes M. bovis, M. africanum, M. canetti, and M. microti, with M. caprae and M. pinnipedii considered variants of M. bovis. It was previously assumed that Mtb evolved from M. bovis during the domestication of cattle, 190 a theory made widespread by the national bestseller, Guns, Germs, and Steel. 54 The genome sequencing projects for both species, however, revealed that M. bovis has several DNA deletions while maintaining 99.95% identity with Mtb and no new genetic material, 31 supporting the opposite scenario. Successive DNA deletions of regions of difference (RD) resulted in the branching off of members of the Mtb complex. 31 These genetic analyses indicate that members of the Mtb complex are the clonal progeny of an ancestral strain of M. canetti, also referred to as Mycobacterium prototuberculosis. 90
An important genetic difference between Mtb and BCG is the deletion of 16 loci in the BCG genome, designated RD1 through RD16. 19 RD1, specifically, was the primary deletion in the original attenuation of M. bovis to create BCG. 104 RD1 of Mtb is composed of 9 genes, which, together with genes lying outside the RD1 locus, constitute the ESAT-6 secretion system (ESX-1). 30 The major function of RD1 is thought to be the encoding and secretion of the major antigenic proteins, early secretory antigenic target–6 (ESAT-6) and culture filtrate protein–10 (CFP-10), 109 which are discussed in Table 1.
Virulence Factors of Mycobacterium tuberculosis

CDP-10, culture filtrate protein–10; DC, dendritic cell; DTH, delayed-type hypersensitivity; ESAT-6, early secretory antigenic target–6; IFN, interferon; IL, interleukin; NAD, nicotinamide adenine dinucleotide; RNI, reactive nitrogen intermediates; ROI, reactive oxygen intermediates; ROS, reactive oxygen species; TLR2, Toll-like receptor 2.
Cell Biology of Mtb
The fate of Mtb in the macrophage is one of the most intriguing aspects of Mtb pathogenesis and deserves explanation before a discussion of virulence factors. Mtb interacts with macrophages through multiple phagocytic receptors (Fig. 1; for an older but excellent review, see Ernst 60 ). Although macrophage responses to Mtb ligation of any or all of these receptors in vivo are unclear, some in vitro studies show that the ligation of particular receptors may determine the macrophage response. For instance, ligation of the Fc receptor (FcR) led to production of reactive oxygen intermediates (ROIs) and phagosomal-lysosomal fusion, whereas ligation of complement receptor 3 (CR3) or mannose receptor (MR) inhibited the respiratory burst and prevented phagosomal maturation. 7,111
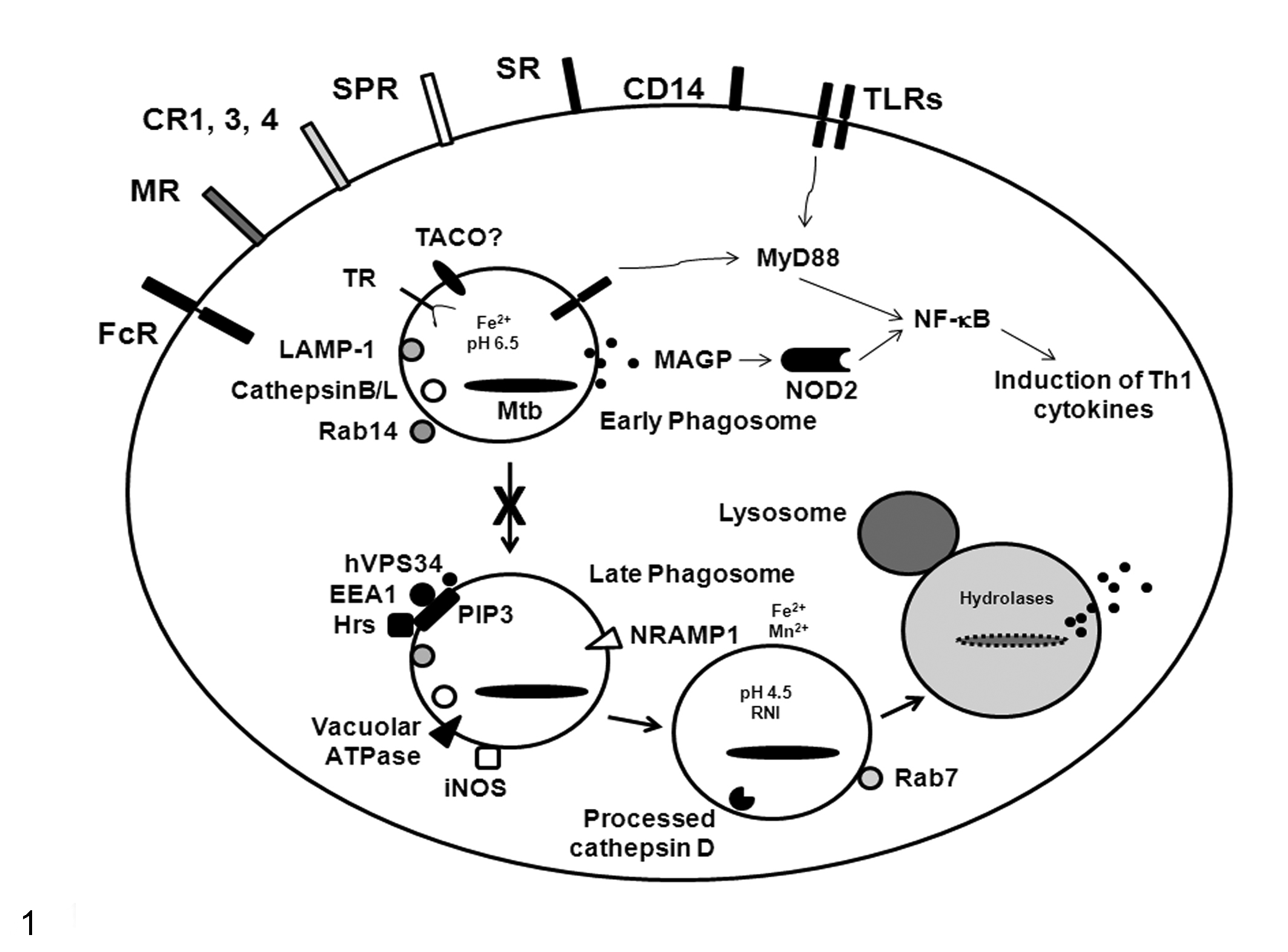
Cell biology of Mycobacterium tuberculosis (Mtb) infection. Mtb interacts with macrophages through a number of receptors that have been shown in vitro to differentially regulate the fate of the bacillus. Phagocytic receptors include the Fc receptor (FcR), mannose receptor (MR), complement receptors (CR1, CR3, CR4), surfactant protein receptors (SPR), and scavenger receptors (SR). CD14 is a tethering receptor that aids in Toll-like receptor (TLR) signaling. TLRs are expressed at both the plasma and phagosomal membranes and induce cytokine production via the MyD88 signaling pathway. Mycolylarabinogalactan-peptidoglycan (MAGP) from the Mtb cell wall can be released into the cytoplasm, where it is detected by the pattern recognition receptor, NOD2/CARD15. Both TLR and NOD2 signaling induce cytokine gene transcription via nuclear factor–κB (NF-κB). Once in the phagosome, live Mtb blocks phagosomal maturation (X). The mycobacterial phagosome actively retains the characteristics of an early endosome, such as tryptophan aspartate coat protein (TACO), although this is currently in debate. 65 Vacuolar proton ATPase, inducible nitric oxide synthase (iNOS), and natural resistance-associated membrane protein 1 (NRAMP1) are inhibited from associating with this phagosome, thereby avoiding pH reduction, reactive nitrogen intermediates (RNIs), and Fe2+/Mn2+ starvation, respectively.74,146,195 Phosphatidylinositol 3-phosphate (PI3P), a lipid involved in proper trafficking of lysosomal constituents to phagosomes, is also continuously eliminated from phagosomes containing live Mtb due to the hydrolytic activity of secreted lipid phosphatase, SapM. 211 Early endosomal autoantigen 1 (EEA1) and Hrs, both of which are trafficking molecules that associate with PI3P, are also prevented from associating with the Mtb phagosome.75,215 The Mtb cell wall component, lipoarabinomannan (LAM), potentially prevents phagosomal maturation through transient inhibition of cytoplasmic Ca2+ fluxes, which then impairs recruitment of the kinase (hVPS34) that produces PI3P. 210 TR, transferrin receptor; LAMP-1, lysosomal-associated membrane protein-1; Rab, small GTPases. Modified from Kaufmann. 115
Surface-expressed pattern recognition receptors (PRR), such as Toll-like receptor 2 (TLR2), also bind Mtb and initiate proinflammatory cascades in response to mycobacterial components, such as lipoarabinomannan (LAM), lipomannan, phosphatidylinositol mannosides (PIMs), 19-kDa lipoprotein, and trehalose dimycolate (TDM). 27,107,143,168 TLR4, TLR9, and the TLR adaptor molecule, MyD88, also appear to play important protective roles during Mtb infection in mice, as mice deficient in MyD88 or doubly deficient in TLR2 and TLR4 or TLR9 are more susceptible to infection and have uncontrolled bacterial growth. 11,77,181
The intracellular PRR, NOD2/CARD15, was shown to recognize the Mtb cell wall component mycolylarabinogalactan-peptidoglycan and to be at least partially required for IL-12p40 responses to this molecule in macrophages and dendritic cells (DCs). NOD2-deficient mice had increased bacterial burdens compared to wild-type mice at 6 months postinfection with Mtb. 55 NOD2 variants have also been demonstrated to be associated with susceptibility to TB in African American populations. 9
Both TLRs and NOD2 signal through the NF-κB signaling pathway, and mutations in the leucine zipper domain of NEMO (IκB kinase-γ), which is an activator of this pathway, have been associated with increased susceptibility to mycobacterial disease. 121 These mutations led to profound defects in interleukin (IL)–12 and secondary interferon (IFN)–γ production in monocytes from these patients. 66
Internalized Mtb resides in a phagosome, where, in resting macrophages, the bacteria block maturation, lysosomal fusion, and acidification. This inhibition is an active process because dead mycobacteria traffic to the lysosome. 7 Reduced acidification has been attributed to reduced or absent levels of vacuolar proton ATPase. 195 The mycobacterial phagosome retains the characteristics of early endosomes while excluding late endosomal markers. 40 Mycobacterial phagosomes are also prevented from association with inducible nitric oxide synthase (iNOS), thereby limiting exposure to damaging nitrogen radicals. 146 This protective environment also prevents effective antigen processing of Mtb. 201
Our understanding of the mechanisms controlling phagosomal maturation is incomplete and controversial. Prevention of mycobacterial phagosome maturation has been attributed to the active retention of tryptophan aspartate coat protein (TACO), as bacilli are readily trafficked to lysosomes and eliminated in TACO-deficient cells, but this is currently in debate. 65 Phosphatidylinositol 3-phosphate (PI3P), a lipid involved in proper trafficking of lysosomal constituents to phagosomes, is also continuously eliminated from phagosomes containing live Mtb. 211 In this study, it was shown that Mtb secretes a lipid phosphatase, SapM, which hydrolyzes PI3P, thereby inhibiting phagosome-late endosome fusion in vitro.
It is likely that Mtb cell wall lipids, such as LAM and TDM, both of which have been shown to prevent phagosomal maturation in vitro, 10,76 interact directly with the phagosomal membrane to mediate this effect. This likelihood is supported by a recent study that showed that M. avium required a closely apposed phagosomal membrane to prevent phagosomal maturation. 48 This study also showed that M. avium prevented from closely associating with the phagosomal membrane due to the presence of surface-bound beads, could be rescued from lysosomal conditions when the beads were shed.
Macrophages activated by cytokines such as IFN-γ and tumor necrosis factor (TNF)–α, however, can overcome this mycobacterial defense mechanism, potentially through the stimulation of reactive nitrogen intermediates (RNIs). Nitric oxide (NO), the product of iNOS, is highly toxic to intracellular mycobacteria. 36 The iNOS inhibitors aggravate the course of murine TB, and NOS2-deficient mice are highly susceptible to mycobacterial infection. 73,83 The role of iNOS in human TB is less clear, although iNOS expression is documented in human monocytes and alveolar macrophages. 149 The role of ROI is even murkier, although mice deficient in cytosolic p47, which is required for effective superoxide production, were partially impaired in controlling acute aerosol infections. 43 This ambiguity may be because certain mycobacterial cell wall components, such as LAM and phenolic glycolipid I, can scavenge oxygen radicals. 35,123 Amazingly, Mtb forced into phagolysosomes through antibody opsonization can still survive. 7
Natural resistance-associated membrane protein 1 (NRAMP1) is a membrane protein involved in innate immunity that is primarily expressed in macrophages and tissues of the reticuloendothelial system. This protein is expressed in the membrane of lysosomes in macrophages and is rapidly recruited to phagosomes. 87 The NRAMP1 gene (also called Slc11a1) has been linked with genetic resistance to BCG and other bacterial infections in the mouse by controlling intracellular microbial replication in macrophages. 128 NRAMP1 does this by acting as an efflux pump for the divalent cations Fe2+ and Mn2+, thereby preventing the use of the cations by the phagocytosed bacteria. 74 Mice strains susceptible to BCG infection carry a point mutation that affects NRAMP1 protein folding, leading to rapid degradation of this protein. 214
IFN-γ can also induce autophagy, the process whereby organelles are degraded and recycled, through the induction of p47 GTPases. 91 Induction of autophagy has been shown to lead to the destruction of intracellular Mtb, likely through forced interaction with antimicrobial autolysosomal contents, such as ubiquitin fragments. 4 Autophagy of Mtb can be induced by ligation of PRRs, such as TLRs. 50 Interestingly, Th2 cytokines such as IL-4 and IL-13 both inhibit autophagy and counteract IFN-γ-induced autophagy. 95
Virulence Factors
The virulence factors of Mtb can generally be divided into 2 groups: proteins and cell wall components. Because the literature on Mtb virulence factors is vast, the factors that have shown a phenotype in animal models when the gene is deleted or mutated, or for which the mechanisms are well described, are summarized in Table 1.
Pathogenesis and Lesion Development
Human Mtb infections usually begin by inhalation of aerosol droplets containing tubercle bacilli directly expectorated from an individual with “open” pulmonary disease. The infectious dose for a person is reported to be between 1 and 200 bacilli; however, as a single aerosol droplet can contain anywhere from 1 to 400 bacilli, it is unclear what is considered a biologically relevant dose. 12,173 The bacilli travel to the alveoli, where they are rapidly phagocytosed by alveolar macrophages. These macrophages are stimulated by ligation of TLRs and other PRRs to produce proinflammatory cytokines and chemokines, driving the recruitment of more leukocytes to the site of infection. Neutrophils and monocytes arrive first, phagocytose additional bacteria, secrete more cytokines and chemokines, and begin to organize the early granuloma. Dendritic cells also phagocytose Mtb and then migrate to regional lymph nodes to present mycobacterial antigens to lymphocytes.
Eventually, a well-organized granuloma develops, consisting of central, infected macrophages, surrounded by epithelioid macrophages, foam cells, and occasional multinucleated giant cells of the Langhans type, with peripheral recruited lymphocytes and a fibrous capsule (Fig. 2a). This structure is a fine balance between host containment of infection and protection of Mtb from IFN-γ-producing lymphocytes. Over time, the center of the granuloma undergoes necrosis, and the high lipid and protein content of the dead macrophages results in the caseous appearance grossly and histologically (Fig. 2a). The granuloma is maintained by a delayed-type hypersensitivity (DTH) response to the persistent presence of antigens and immunostimulatory lipids, as Mtb can survive within macrophages and extracellularly within the granuloma. 138 The caseum is predominantly composed of host-derived lipids, and a recent analysis of gene regulation in the immediately surrounding cells shows Mtb-driven dysregulation of host lipid metabolism. 117 The granuloma environment is considered hypoxic in guinea pigs, rabbits, nonhuman primates, and human beings, 5,213 which is an important consideration for mycobacterial metabolism and antibiotic therapy efficacy. It was previously believed that Mtb were sparse in the caseum, based on Ziehl-Neelsen acid-fast staining, with most of the detectable bacilli persisting at the periphery. 126 A recently developed acid-fast technique using Auramine O and Rhodamine B revealed that the majority of bacilli are actually in the necrotic core in guinea pig caseous granulomas, 102 but the viability of these bacilli remains unclear. Mtb adapt to this environment by preferentially using fatty acids in their metabolism, slowing down active replication, and increasing cell wall thickness, entering a so-called dormant state. 26
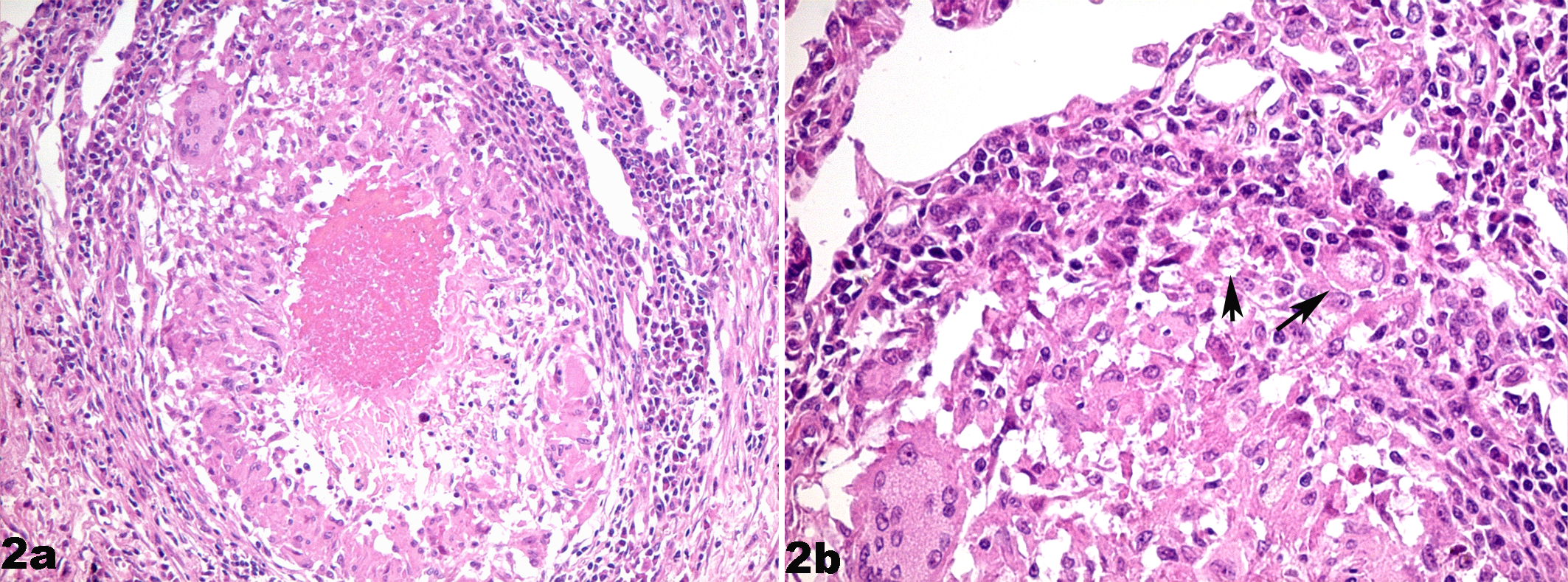
Lung; human. (a) The central caseum is surrounded by epithelioid macrophages, multinucleated giant cells, peripheral lymphocytes and plasma cells, and a fibrous capsule. Hematoxylin and eosin (HE). (b) This higher magnification of a portion of (a) contains foamy macrophages (arrows). HE. Courtesy of Dr. M. Kim.
The lesion at the primary site of implantation is termed the Ghon focus. Bacilli may spread before the formation of these organized granulomas via the lymphatics to regional lymph nodes, resulting in granulomatous lymphangitis and lymphadenitis, which, along with the Ghon focus, is called the primary Ranke’s complex. Hematogenous dissemination within the lung or to other organs can also occur during the early stage of this disease. Interestingly, the upper lung lobes of the human favor bacillary growth due to higher oxygen pressure and delayed immune responses. 160
Despite wide dissemination of Mtb during primary infection, the majority of infected, but otherwise healthy, individuals resolve these lesions without becoming symptomatic. Approximately 10% of infected individuals, however, develop severe, life-threatening disease or primary progressive disease at this stage. 41 This is believed to be caused by high bacterial load, increased bacterial virulence, immunosuppression, or genetic susceptibility. 41 In most cases, however, the initial infection subsides, and the collagen capsule contracts to form a scar, or the granuloma calcifies. Intriguingly, most TB patients have pulmonary lesions with heterogeneous morphology, 112 suggesting that each granuloma contains bacilli at different stages of latency and reactivation. 46,64 In chronic lesions, Mtb growth is predominantly in the foamy macrophages at the periphery of lesions or in chronic granulomas that have cavitated and accessed an airway. 112
In the absence of progressive disease, an infected individual can remain asymptomatic for years or decades, with bacilli existing in a poorly understood “latent” state, presumably held in check by the host immune system. This ability to establish a chronic asymptomatic infection, followed by reactivation and transmission years later to new uninfected hosts, lends to the tremendous success of Mtb as a pathogen. Any condition affecting the immune system, such as HIV or other infection, old age, malnutrition, malignant disease, immunosuppressive medication, or new infection, can lead to reactivation or secondary disease. 212
Five to 10% of latently infected individuals develop secondary disease at some point in their lives for reasons that are not always clear. 203 These individuals make up to 80% of clinical TB cases (at least in industrialized countries) and almost all cases of transmission. 81 Interestingly, extralesional or extrapulmonary tissues may be persistently infected with Mtb.98,148 In fact, nearly 15% of reactivating TB cases occur at extrapulmonary sites, such as the central nervous system, the skin, internal organs, and genitourinary tract, without accompanying lung lesions. 47 How reactivation occurs, particularly whether “latent” bacilli become proliferative or if reinfection or bacilli from other sites are responsible for reactivation, are questions that remain to be answered.
The most common form of secondary tuberculosis is usually limited to the lung, and lesions begin as an exudative bronchopneumonia and progress to classical caseous granuloma formation, followed by massive necrosis and cavity formation. 127 Eventually, there is breakdown of the fibrous capsule and communication with airways. These events allow for rapid growth of extracellular bacilli and spread into airways, leading to transmission or intrapulmonary spread. 33 Sputum analysis during this phase of TB reveals increases in macrophages and neutrophils. 155
Although symptoms of primary TB in most cases are generally subtle and easily overlooked, secondary cases are characterized by localized symptoms, such as coughing, hemoptysis, and pleuritic pain, as well as the generalized symptoms of fever, anorexia, night sweats, and cachexia. This propensity of TB to cause wasting away of patients garnered it the name consumption. In immunocompetent individuals, secondary TB can result in dissemination and death in 50% of cases and chronicity in 25% to 30% of cases. If the host is able to reestablish immune control over the disease, recovery is also possible, as is seen in 20% to 25% of cases. 15
Host Response
Cytokines
After phagocytosis of inhaled Mtb, alveolar macrophages are stimulated, primarily by ligation of TLR2 and TLR4, to secrete proinflammatory cytokines, such as TNF-α, IL-1β, IL-6, and IL-12. 28,143 TNF-α is a critical cytokine in the response to Mtb in that it can stimulate neutrophils and macrophages in an autocrine and paracrine fashion to stimulate apoptosis and ROI/RNI production, leading to the destruction of some phagocytosed bacilli. 79 TNF-α is also an essential cytokine in driving organized granuloma formation, and the recent development of anti-TNF-α drugs for the treatment of chronic inflammatory conditions has led to increased TB reactivation cases. 72 The role of TNF-α in granuloma formation is in part due to its own leukocyte recruitment activity, as well as TNF-α-dependent chemokine production. 3 TNF-α is also at least partially responsible for the fever and wasting that occur in progressive TB. 72
IL-1β has also been shown to play an important role in granuloma formation and has been correlated to disease activity and fever in infected patients. 63,202 IL-1 also acts in an autocrine and paracrine fashion to stimulate macrophages to produce TNF-α and IL-6 and upregulates T cell expression of IL-2 and its receptor, thus stimulating T cell proliferation. 200 IL-1α has shown some use as a biomarker for patients with pulmonary TB when used in combination with MIP-1β and epidermal growth factor. 38
IL-6 is produced in large amounts in the pleural fluid and bronchoalveolar lavage cells from TB patients, but this cytokine tends to antagonize the effects of TNF-α by preventing binding of TNF-α to macrophages and antagonizing its antimycobacterial activity. 21,202,220 IL-6 has also been shown to enhance mycobacterial growth both intra- and extracellularly 52 while inhibiting TNF-α and IL-1β production at the transcriptional level in vitro. 183 IL-6 also suppresses T cell proliferation and IL-2 production in response to antigenic stimulation. 208
IL-12 is an important cytokine driving T helper type 1 (Th1) differentiation and IFN-γ production, which is required for resistance to mycobacteria and other intracellular pathogens. 42 IL-12 also enhances cytotoxicity by stimulating the proliferation of antigen-specific cytolytic T cells and natural killer (NK) cells. 22 The importance of IL-12 is highlighted by cases of severe nontuberculous mycobacterial disease in patients with autosomal recessive mutations in the genes encoding either IL-12 or its receptor, IL-12Rβ1, most likely via decreased production of IFN-γ. 58
IFN-γ, a T cell cytokine that activates macrophages to produce reactive oxygen and nitrogen species, is probably the most important cytokine in the immune response to mycobacteria. This cytokine is mainly secreted by Th1 and CD8+ cytotoxic lymphocytes, NK cells, natural killer T (NKT) cells (cells with characteristics of both NK and T cells), professional antigen-presenting cells (APCs), and, to a lesser extent, B cells. 136 Mice defective in IFN-γ or its receptor are extremely susceptible to Mtb and BCG, with earlier death, high bacterial numbers, and progressive destructive granuloma formation. 71,110 Patients with genetic defects in IFN-γ receptor function or its signaling pathway (ie, STAT1) are particularly prone to chronic or uncontrolled nontuberculous mycobacterial infections. 58 Furthermore, treatment of these patients with exogenous IFN-γ can lead to clinical improvement. 103 Failure of AIDS patients to produce high levels of IFN-γ in the lung during pulmonary TB most likely contributes to their susceptibility to infection with Mtb. 124
A separate subset of T cells, termed Th17 cells, which produce IL-17A, IL-17F, IL-21, and IL-22, has been recently described. 159 IL-17, in particular, has been shown to play an important role in the immunopathology of autoimmune disorders and chronic infectious diseases, 56,57 mostly by the induction of chemokines and therefore excessive recruitment of leukocytes to the site of infection, with ensuing tissue damage. 154 This cytokine is produced primarily by γδ T cells in mice, in response to IL-6 and IL-23 during mycobacterial infection. 129 Studies in human DCs suggest that prior to IFN-γ production during Mtb infection, IL-23 is preferentially produced, suggesting early skewing toward a Th17 response. 85 In the absence of IL-17, neutrophils are found in lesser numbers, and granuloma formation in response to BCG is delayed, 205 suggesting a role for this cytokine in the initial formation of the granuloma. It is likely that a proper Th1-Th17 balance is required for control of Mtb.
Immunosuppressive cytokines such as IL-10 and transforming growth factor (TGF)–β, both of which suppress effective Th1 responses, play a role in active TB and tend to be transiently overexpressed in TB patients. 100 Higher numbers of regulatory T cells, which produce these immunosuppressive cytokines, were reported in the peripheral blood of TB patients and at the site of infection. 92 TGF-β is also associated with fibrosis during chronic Mtb infection. 97 IL-27, a member of the IL-12 cytokine family, has been shown to negatively regulate development of Th17-type cells during chronic inflammation. 194 Improved control of mycobacterial growth due to increased inflammatory cytokine responses in the lungs of mice lacking IL-27 receptor signaling has been reported. 162
IL-4 and other Th2 cytokines are elevated in patients with active TB, and high pulmonary IL-4 expression has correlated with cavitation. 206 IL-4 has a pathogenic role during late TB disease by downregulating protective Th1 responses. 24 Th2-skewed responses are also more commonly observed in TB patients living closer to the equator, possibly due to helminth coinfection, high exposure to saprophytic mycobacteria, or higher Mtb inoculum. 99 This effect may be responsible for the uneven efficacy of BCG vaccination in different parts of the world. 179
Cellular Response
TB and DTH lesions are thought to be examples of granulomas composed of classically activated macrophages, also referred to as M1 macrophages, in that they develop in response to concomitant stimulation with IFN-γ and microbial products. 2 Although Mtb contains several components known to ligate TLRs, which usually results in innately activated macrophages, infection elicits robust IL-12 production, supporting the M1 phenotype. These activated macrophages compose the majority of epithelioid macrophages within TB granulomas. The Langhans-type multinucleated giant cells, typical of Mtb granulomas, are the result of fusion of epithelioid macrophages and have recently been shown to be induced by the cell wall lipid lipomannan. 167 Foamy macrophages (Fig. 2b, arrows) predominate in advanced TB and are only induced by virulent mycobacteria in response to oxygenated mycolic acids. 165 These cells are thought to provide a nutrient-rich, protective reservoir for Mtb persistence, as these cells have reduced microbicidal and phagocytic activity. Foamy macrophages can also induce apoptosis of Th1 cells through the Fas/Fas ligand mechanism while suppressing their own apoptosis through high expression of the antiapoptotic Bcl2 molecule. 176
Neutrophils are one of the first responders to Mtb infection, and although their bactericidal activities, such as phagocytosis, ROI, RNI, antimicrobial peptides, and extracellular traps, are well characterized, their role in the immune response to Mtb is quite controversial. In mouse models, depletion of neutrophils was shown to lead to an increase in mycobacterial growth, whereas stimulation of neutrophilia led to decreases. 78 There are conflicting reports, however, regarding the ability of neutrophils to kill mycobacteria directly. 51,108 Nevertheless, neutrophils do play a role in cytokine and chemokine production, granuloma formation, and transference of microbicidal molecules to neighboring, infected macrophages. 174,185,197 Neutrophilic inflammation may also be an important part of TB immunopathology, in that there is greater neutrophil accumulation within lesions in TB-susceptible versus resistant animals. 61 This effect has recently been shown to be due to differential chemokine expression between these 2 models and may explain the earlier discrepancies in neutrophil studies. 116 Neutrophils have also been shown to be the predominant infected cell type in active TB patients, suggesting a permissive environment for bacillary replication. 62
Type 1 and 2 pneumocytes, as well as pulmonary fibroblasts and endothelial cells, have been reported to contain mycobacterial DNA in TB patients, 98 and both pneumocytes and alveolar endothelial cells can be infected in vitro, 144 but the role of these cells in disease is unclear. Mtb was shown in vitro to enter type 1 pneumocytes by macropinocytosis. 82 The M cells overlying the luminal aspect of bronchiolar-associated lymphoid tissue is also a proposed site of entry for Mtb. 199 As mentioned in Table 1, ESAT-6 is cytolytic for type 1 pneumocytes, 118 and necrosis of alveolar epithelium was shown to be a characteristic of virulent Mtb infection. 139 Both mouse and human lung and tracheal epithelial cells have been shown to express defensins after infection with Mtb, with data supporting a protective role by these cationic peptides. 177,178
Dendritic cells in the lung can phagocytose Mtb that has crossed the epithelial barrier. Mtb enters by binding the receptor DC-SIGN via the cell wall component ManLAM, with a lesser role played by CR3 and MR. 196 After phagocytosis, the DCs mature and migrate to regional lymph nodes, where they present mycobacterial antigens in the context of major histocompatibility complex (MHC) molecules, as well as CD1, to prime CD4+ and CD8+ T cells. 13,89 Dendritic cells infected with Mtb have an impaired ability to present lipid antigens, possibly due to delay of maturation mediated by cell wall components, and are retained at the lung for prolonged periods compared to DCs that have taken up model antigens. 163 Interestingly, it has been shown in vitro that more virulent strains of Mtb, the Beijing strains in particular, inhibit the maturation of DCs and the ability of DCs to present lipid antigens through decreased expression of CD1. 192
The activated T cells specific for mycobacterial antigens migrate to the lungs to participate in granuloma formation. The localized immune response to Mtb or the tuberculin reaction is the classical model for DTH. Mycobacterial antigens drive the differentiation of CD4+ T cells to Th1 cells. Th1 cells preferentially secrete IFN-γ, which is responsible for the development of DTH via macrophage activation. IL-12 is also critical for the induction of Th1 and IFN-γ responses and therefore DTH. Other important cytokines in this response are IL-2, which causes proliferation of the Th1 cells, and TNF-α and lymphotoxin, which mediate many of the vasodilatory effects and induce the secretion of more chemokines to recruit leukocytes to the reaction site. CD4+ T cells are important in the prevention of reactivation and both primary and secondary disease, as decreased CD4+ T cells accelerate TB in HIV patients. 219 CD4+ T cells are also relatively absent in cavitated TB lesions, HIV/TB coinfected patients are less likely to have cavitary disease, 29 and CD4-deficient mice have less necrosis in M. avium–induced lesions, 69 suggesting a role for CD4+ T cells in cavitation. CD4+ T cells in the bronchoalveolar lavage (BAL) fluid of patients with cavitary disease are also more Th2-skewed, secreting more IL-4 than IFN-γ. 206
αβ T cell receptor (TCR)–expressing CD4+ T cells are required for granuloma formation; transfer of these cells, but not CD8+ T cells, can protect immunodeficient mice from Mtb. 44 CD4+ T cells may also reduce bacterial numbers by the induction of apoptosis through the Fas (CD95) ligand system. 150 CD8+ T cells do play an important role, however, by destroying infected cells or killing intracellular bacteria via the secretion of granulysin and perforin, which lyse host cells and attack Mtb directly. 191 Recent studies in the nonhuman primate model, show an important role for CD8+ T cells in that both BCG-vaccinated or previously infected and antibiotic-cured rhesus macaques had markedly reduced protection against challenge with Mtb after CD8+ T cell depletion. 39
The CD1 family is composed of MHC class I–like molecules that bind distinct lipid antigens, particularly glycolipids, which are recognized by T cells. 17 CD1 molecules are therefore of great interest to TB researchers because Mtb abundantly releases lipid antigens during infection. 16 CD1 molecules have been implicated in the presentation of mycobacterial lipids and the immune response to Mtb. CD1d binds and presents the phosphatidylinositol mannoside, PIM4, to invariant NKT cells, which have recently been shown to be able to suppress intracellular Mtb replication both in vitro and in vivo through the innate production of IFN-γ in response to infection. 68,182 Mycobacterial diacylated sulfoglycolipids were shown to be presented by CD1b molecules and to stimulate specific T cells to recognize Mtb-infected cells and kill intracellular bacteria. 86 CD1b molecules also present free mycolic acids and LAM. 17,187 Treatment of mice with anti-CD1 antibody resulted in exacerbated early pathology and decreased reduction of type 1 cytokines after Mtb infection, although CD1d-deficient mice showed no greater susceptibility to infection. 18 In the context of human infection, CD1-restricted T cell responses to mycobacterial lipid preparations were observed in Mtb-infected individuals. 204
Natural killer cells are also mycobactericidal and can be activated in the absence of APCs. Stimulated NK cells secrete IFN-γ, IL-15, and IL-18, which play a crucial role in the regulation of CD8+ T cells during Mtb infection. 209 Natural killer cells also improve the cytolytic activity of γδ T cells and induce them to secrete IFN-γ. 222
Although cell-mediated immunity is far more critical for effective responses against Mtb, B cells and antibodies have been shown to play a role during TB. B cell–deficient mice were shown to have higher bacillary burden and exacerbated pulmonary lesions due to higher neutrophil recruitment after aerosol infection with Mtb. 132 Passive immunization of Mtb-infected SCID mice with sera from mice treated with Mtb extracts also reduced the number of bacilli in the lungs and the extent of granulomatous infiltration. 88 Treatment with antibodies specific for particular Mtb components, such as 19-kDa lipoprotein, 130 α-crystallin, 217 and LAM, 93 resulted in reduction of bacterial load and lesions in the lungs of infected mice. Antibody-opsonized mycobacteria are more efficiently internalized and killed by neutrophils and macrophages 49 and more effectively processed for antigen presentation by DCs. 198 Interestingly, although antibody production is typically associated with a shift to a Th2 response, BCG-induced antibodies were shown to enhance proliferation of IFN-γ production in mycobacterium-specific CD4+ and CD8+ T cells. 49 Unfortunately, diagnostic testing for antibody responses to Mtb components has poor sensitivity and specificity, rendering these tests relatively ineffective. 1
Animal Models of TB
The following is a description of animal models of TB that specifically use Mtb. There are several important TB models using Mycobacterium marinum (zebrafish) and M. bovis (rabbits, swine, calves), for which the reader is directed to other reviews.
Mice
The study of TB in laboratory animals has been challenging in that mice, for which the most reagents are available, are relatively resistant to Mtb and do not typically develop TB or a DTH response to Mtb in a manner similar to humans. Mice can be infected by aerosol, but their more complicated nasal scrolls, relative to humans, affect the final dose that is administered to the lungs. 172 Furthermore, mice can harbor high numbers of Mtb within lung tissue without showing clinical signs. 141 Mice also do not cough or form cavitary lesions, making them a poor model for transmission studies. 113 It is important to recognize, however, that the mouse model has been critical in the development of the standard anti-TB therapy that is used today (for an excellent review of this history, see Mitchison 147 ) and is still a required step in the development of new vaccines.
The histologic appearance of lesions in mice infected with Mtb by aerosol typically consists of multifocal aggregates of epithelioid and foamy, infected macrophages, with scattered multinucleated giant cells, lymphocytes, and plasma cells flooding alveoli, with significant lymphoplasmacytic perivascular cuffing. Often, there is the appearance of “inverted” granulomas, where the lymphocytes and plasma cells are centrally located, surrounding a venule or arteriole, with the infected macrophages located peripherally. This has potential significance for the pathogenesis of Mtb in mice, in that the centrally located, IFN-γ-producing T cells have greater access to the infected macrophages and may thus allow for better control of infection and reduced necrosis. 153 Fibrous capsules are also not observed histologically, which can affect the validity of antibiotic studies, as Mtb would be more easily accessed by drugs in the mouse lung. Nonetheless, chronic infection can be experimentally produced in mice, and caseous granulomas have been shown in sensitized mice in response to Mtb when infections were in proximity to lipids, such as adipose tissue or if preceded by lipid pneumonia, 106 or in certain mouse strains, such as I/St. 169 Necrotizing and cavitating granulomas with fibrosis can also be produced in mice by infection with M. avium, but in this model, infection is never controlled and bacillary burden increases until death. 20 In addition, because of their short life span, mice are poor models for the study of latent infection. Latency can be studied to a degree using the Cornell mouse model, in which mice are treated with antibiotics until a paucibacillary state of infection is reached, followed by immunosuppression by steroid administration. 137
Guinea Pigs
Guinea pigs develop robust DTH responses to mycobacterial antigens and, after infection with Mtb, reproduce many of the aspects of human infection, such as caseous and mineralized granulomas, primary and hematogenous pulmonary lesions, fibrous capsule formation, and dissemination. 142 Cavitation can occur with low-dose, chronic infections of guinea pigs but not as reliably as in the rabbit model. 221 They are also exceptionally vulnerable to infection by as little as a few inhaled mycobacteria, making them well suited for airborne Mtb transmission studies and as sentinel animals. 175
Guinea pigs, however, are not suitable for studies of latency because they are very susceptible hosts and often develop chronic progressive disease with extensive tissue destruction after very low-dose aerosol infection. 189 TB is also a primarily hematogenously disseminated disease in the guinea pig. 189 Pulmonary lesions in the guinea pig also contain a high proportion of granulocytes, particularly eosinophils, 152 which are not common features of the human disease. Furthermore, although inbred strains of guinea pigs are available, they are not well characterized with respect to Mtb infection, and immune responsiveness and reagents, although improving, are limited. It is also important to remember that guinea pigs have heterophils, rather than neutrophils, and that heterophils lack myeloperoxidase but instead contain antimicrobial cationic peptides that are not present in human neutrophils. 125 This difference is commonly overlooked by researchers and may be important when interpreting results in these species.
Rabbits
Rabbits infected with Mtb mount a moderate DTH response and form caseous granulomas and cavitary lesions. 133 Rabbits, including currently available inbred strains, are relatively resistant to Mtb, however, requiring the inhalation of 500 to 3000 bacilli to form one grossly visible tubercle at 5 weeks postinfection. 133 Most rabbits will also overcome disease completely, with few culturable bacilli. 131 This model is useful in the study of latent or paucibacillary TB states, however, without the use of antibiotics as in the Cornell model. Rabbits do need to be experimentally immunosuppressed, however, as they will not spontaneously reactivate disease. 134 There are minimal immune reagents, however, for this model, and the larger size of rabbits makes them more costly to use. There are inbred strains of rabbits, such as the Lurie and Thorbecke rabbits, which are more susceptible to Mtb infection. This susceptibility has been linked to suppressed macrophage antimycobacterial activity, decreased MHC class II expression, and impaired development of type IV hypersensitivity. 131,145 Rabbits, like guinea pigs, also have heterophils as opposed to neutrophils.
Nonhuman Primates
Nonhuman primates, such as the cynomolgus macaque, although expensive and challenging to work with, do provide an excellent model to study TB. Fifty percent of cynomolgus macaques infected via bronchoscope develop primary TB, whereas the other half become latently infected and can reactivate, and the full spectrum of granuloma types can be observed. 34 This model is also important for the study of coinfection with simian immunodeficiency virus and Mtb, as coinfection with HIV is particularly common in sub-Saharan Africa, and TB is the most common cause of death in HIV patients. 6 DTH responses in nonhuman primates are typically weak, with low rates of skin test positivity unless the more potent old tuberculin is used. 184 Although human reagents can often be used for analyses, because of the expense and difficulties of handling nonhuman primates, as well as the lack of inbred strains and potential for horizontal transmission within a colony, this animal model has not been widely used except in industry, as part of vaccine and treatment studies.
Conclusion
The tuberculous granuloma can be viewed as either a dance or a battleground between the pathogen and the host that has evolved over millennia. Changes in Mtb and in the granuloma environment can easily tip the scale in favor of one side versus the other. Although a tremendous amount of research has been performed in the study of TB, this disease continues to be a scourge upon humanity and veterinary species. Until the factors affecting each side of this battle are better understood, we are likely to continue to suffer this plague. The continued development of animal models and reagents, as well as the wide use of genetic and other screens to study virulence factors in Mtb, will help us in this endeavor. The veterinary pathologist can play an important role on the front lines, diagnostically, as well as in the interpretation of lesions produced during experimental infection.
Footnotes
Acknowledgements
I thank Dr. Elizabeth Rhoades for her help editing the parts of this review that composed the literature review of my dissertation and Drs. Kyle Rohde, Melinda Camus, and Andrew Moorhead for their help in editing this version. Thanks also to Dr. Mi-Jeong Kim for providing the human TB photomicrographs and to Dr. Helen Wainwright and Ms. Annalie Visser at the University of Cape Town, South Africa, for providing Dr. Kim with access to human pulmonary TB lesions. I also thank Dr. David Russell for his guidance and support during my graduate studies. I am grateful to the editors and reviewers of this manuscript, who provided excellent suggestions and constructive criticism. Finally, I would like to recognize and apologize to all of the researchers whose work I could not include in this review due to restrictions in the number of references I could include (my original reference list contained almost 500 citations). This review and my own research could not have happened without the tremendous effort of TB researchers past and present.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
