Abstract
Middle East respiratory syndrome coronavirus (MERS-CoV) is the cause of a severe respiratory disease with a high case fatality rate in humans. Since its emergence in mid-2012, 2578 laboratory-confirmed cases in 27 countries have been reported by the World Health Organization, leading to 888 known deaths due to the disease and related complications. Dromedary camels are considered the major reservoir host for this virus leading to zoonotic infection in humans. Dromedary camels, llamas, and alpacas are susceptible to MERS-CoV, developing a mild-to-moderate upper respiratory tract infection characterized by epithelial hyperplasia as well as infiltration of neutrophils, lymphocytes, and some macrophages within epithelium, lamina propria, in association with abundant viral antigen. The very mild lesions in the lower respiratory tract of these camelids correlate with absence of overt illness following MERS-CoV infection. Unfortunately, there is no approved antiviral treatment or vaccine for MERS-CoV infection in humans. Thus, there is an urgent need to develop intervention strategies in camelids, such as vaccination, to minimize virus spillover to humans. Therefore, the development of camelid models of MERS-CoV infection is key not only to assess vaccine prototypes but also to understand the biologic mechanisms by which the infection can be naturally controlled in these reservoir species. This review summarizes information on virus-induced pathological changes, pathogenesis, viral epidemiology, and control strategies in camelids, as the intermediate hosts and primary source of MERS-CoV infection in humans.
Keywords
Severe acute respiratory syndrome and Middle East respiratory syndrome (MERS) outbreaks have caused major public concerns in 2002–2003 and since 2012, respectively. Together with the ongoing COVID-19 pandemic, they indicate the need to study, prevent, and control emerging coronaviruses (CoVs) of zoonotic origin. Among these life-threatening virus infections, MERS-CoV infection has the highest case fatality rate (35%) among infected people. Severe pathogenic features of MERS-CoV infection in humans, including diffuse alveolar damage or even death, are mainly due to complex and dynamic processes characterized by massive infiltration of inflammatory cells into the lungs and induction of an inflammatory cytokine storm. 12 According to the World Health Organization, 2578 laboratory-confirmed cases and at least 888 fatalities have been reported as of December 2021. The Kingdom of Saudi Arabia, where MERS is endemic, is the country with the highest number of cases (specifically, 2167 laboratory-confirmed cases). Although most cases were diagnosed in countries from the Middle East, travel-associated cases have been documented in other regions of the world.14,26,40 So far, all cases reported in North America, Europe, and Asia had a history of travel to the Middle East. Furthermore, a major outbreak occurred in South Korea in 2015 with 186 cases and 38 associated deaths, highlighting the worldwide public health concern of MERS. 43 Thus, MERS cases identified outside the Middle East and without camel herds can also be at risk of endemic MERS-CoV.
Bats were initially suspected to be the hosts for MERS-CoV.47,61,62 Analysis of dipeptidyl peptidase-4 (DPP4, the MERS-CoV receptor) sequences, distribution of the DPP4 receptor in various bat species, in vitro infection studies with different bat cell lines, and in vivo data revealed that bats are potentially susceptible to MERS-CoV.16,28,52 Although bats are considered to be the primary host of the ancestor of MERS-CoV, 24 direct transmission from bats to human has not been proven. Rather, dromedary camels are considered the major source of MERS-CoV transmission to humans. In this review, we summarize the current state of knowledge on the pathological changes associated with MERS-CoV infection in camelids, and provide an overview of the viral epidemiology and immunopathogenesis of MERS-CoV infection in these reservoir species.
Epidemiological Patterns and Transmission of MERS-CoV Among Camelids
Serological surveys in dromedaries from the Arabian Peninsula (the Kingdom of Saudi Arabia, Qatar, Jordan, and Oman) and African countries (Tunisia, Egypt, Nigeria, Kenya, and Ethiopia) showed a high prevalence of neutralizing antibodies against MERS-CoV.22,25,32,35,53–56 The virus has been circulating widely in this species for a relatively long period, since MERS-CoV neutralizing antibodies have been found in retrospective studies in eastern Africa as early as 1983. 51 Further evidence for the importance of dromedaries as a susceptible host came after the detection of identical viral sequences in nasal swabs of dromedaries and infected humans.13,35,39 Currently, three MERS-CoV lineages (including those that have caused human infections) have been isolated from dromedary camels in the Arabian Peninsula and several African countries. 59 To date, no MERS-CoV antibodies or viral ribonucleic acid (RNA) have been detected in Bactrian camels.19,45,50
Camel-to-human transmission of MERS-CoV may occur in a variety of ways, but respiratory secretions are the most likely source of infection. Some studies have reported MERS-CoV RNA in milk and lymph nodes from infected animals, but no infectious virus was detected.9,32,55 However, fecal contamination of milk or meat during slaughter cannot be fully ruled out. Camel urine is also consumed in the Middle East for supposed health benefits, but no infectious virus has been detected in urine. 34 Thus, close contact between humans and dromedaries is the most probable route of virus transmission. Close contact with dromedaries can occur through camel festivals, races, sale barns, and parades. For instance, during the Hajj pilgrimage, dromedaries are animals of ritual significance. 46 Continuous and sporadic MERS outbreaks are considered a failure to control the zoonotic sources. Thus, gaining knowledge on the role of the reservoir host is essential for MERS prevention and global control.
Clinical and Pathological Manifestations of MERS-CoV in Camelids
Old World Camelids (Dromedaries and Bactrian Camels)
The clinical spectrum of MERS-CoV infection in humans ranges from mild respiratory symptoms to severe, life-threatening disease. By contrast, the viral infection merely causes mild upper respiratory tract disease in dromedaries, leading to a rapid clearance of the virus in about 1 or 2 weeks postinfection. 3 Clinical signs in MERS-CoV-infected Old World camelids have been described only in a few studies.2,3,36 The clinically apparent disease was generally mild, and signs were nonspecific and typical of a viral upper respiratory tract infection. Under natural conditions, MERS-CoV infection in dromedaries was most common in animals less than 4 years of age, with the highest prevalence observed in calves. The disease was frequently asymptomatic, but some animals showed nasal discharge, lacrimation, coughing, sneezing, fever, and transient anorexia.6,7,11,39,42
As in naturally occurring disease, experimental inoculation of dromedaries caused nasal discharge of serous to mucopurulent character in the first 2 weeks, and a mild, transient increase in body temperature during the first week after infection (Fig. 1).3,36 Minor nasal hemorrhages were also infrequently observed in dromedaries and were considered to be related to sample collection in most cases. 3

MERS-CoV experimental challenge, dromedary camel, 8 days postinfection (dpi). Mucoid discharge from the nostril.
Similar to dromedaries, experimental infection of Bactrian camels led to nasal discharge and coughing in the first week after infection. 2 Although some field studies reported MERS-CoV–specific antibodies in Bactrian camels of Mongolia, none of these samples turned out to be positive by virus neutralization test. 15 Consistently, natural infections in Bactrian camels have not been described to date.15,19
Pathologic findings occurring in naturally infected dromedaries have only been reported in two recent publications.10,11 One study investigated dromedaries under 2 years of age submitted for regular slaughtering procedures in the Kingdom of Saudi Arabia, some of which tested positive for MERS-CoV antigen and RNA in nasal swabs/turbinates. The animals showed variable lesions in the respiratory tract and some other organs. In the nasal turbinates, infiltration of mononuclear cells, exfoliation of epithelial cells, glandular degeneration, hemorrhages, and focal areas of mild epithelial hyperplasia were detected. In the trachea, the epithelium showed a loss of ciliated cells, vacuolation, erosion, and neutrophilic exocytosis. Additionally, interstitial pneumonia with thickening of alveolar septa by mononuclear cell infiltrates, type II pneumocyte hyperplasia, and increased numbers of alveolar macrophages occurred in all animals. One animal also had bronchopneumonia with segmented neutrophils in bronchioles and alveoli. Outside the respiratory tract, glomerular and tubular degeneration was observed in the kidneys of all animals, and splenic hyperplasia of the red pulp and depletion of the white pulp were detected in one animal. 11 Another study involved scanning electron microscopy (SEM) of nasal, tracheal, and lung samples from the same animals. There was massive ciliary loss, disordered arrangement of cilia, and goblet cell hyperplasia in the respiratory epithelium. 10 MERS-CoV antigen was detected in epithelial cells of the nasal turbinates, trachea, and bronchi but also apparently in pulmonary alveoli and in the kidneys. 11
In contrast to the described field cases, experimental infection of dromedaries with MERS-CoV typically produced lesions restricted to the upper respiratory tract, trachea, and bronchi.2–4,36,38 These included mild-to-moderate rhinitis, tracheitis, and bronchitis (Figs. 2, 3). The lesions were moderate at 4 and 5 dpi, mild at 14 and 28 dpi, and resolved after 42 dpi.2–4,36,38 The affected respiratory mucosa typically showed neutrophilic exocytosis, intraepithelial apoptosis or single-cell necrosis, epithelial erosions, glandular necrosis, and segmental epithelial hyperplasia or squamous metaplasia (Figs. 2, 3).3,36,38 Lesions were accompanied by mononuclear infiltrates and edema of the lamina propria and submucosa (Figs. 2, 3). Similar to natural infection, a massive ciliary loss in nasal turbinates, trachea, and bronchi was characteristic of the experimental disease in dromedaries (Figs. 4, 5).3,38 Importantly, the loss of cilia was not accompanied by extensive cell death or other profound alterations of ciliated cells, indicating that the virus might trigger ciliary loss or retraction in sublethally injured cells, as has been described for other respiratory viruses including CoVs.20,38,44 In contrast to field cases, interstitial pneumonia was not described in experimental infections. Accordingly, the viral antigen in experimentally infected dromedaries was predominantly located in epithelial cells of the upper respiratory tract with the highest antigen loads observed in the nasal turbinates (Figs. 6, 7).3,36,38 Additionally, viral antigen was found in single macrophages in the submucosa of nasal tissues (Fig. 6). 38 In contrast to natural infection, but in line with the lack of overt clinical disease, viral antigen was consistently absent from alveoli.3,36 Outside of the respiratory tract, histologic lesions were only detected in regional lymphoid tissues such as mandibular, retropharyngeal, and mediastinal lymph nodes or tonsils. These tissues had mild-to-moderate follicular hyperplasia with apoptosis of single lymphocytes.36,38 Viral antigen or RNA was documented in occasional cells within the tonsils, mediastinal, retropharyngeal, and cervical lymph nodes3,36 and in the gut-associated lymphoid tissue of the duodenum by immunohistochemistry (IHC) or in situ hybridization, respectively. 36 Therefore, the follicular hyperplasia observed in the lymphoid organs mostly resulted from local antigenic stimulation. The reported observation of single apoptotic lymphocytes was most likely a normal finding associated with lymphocyte homeostasis. However, it cannot be excluded that apoptosis of individual cells might also be triggered by virus infection. Viral antigen has not been found in other extrarespiratory tissues of experimentally infected dromedaries.3,36
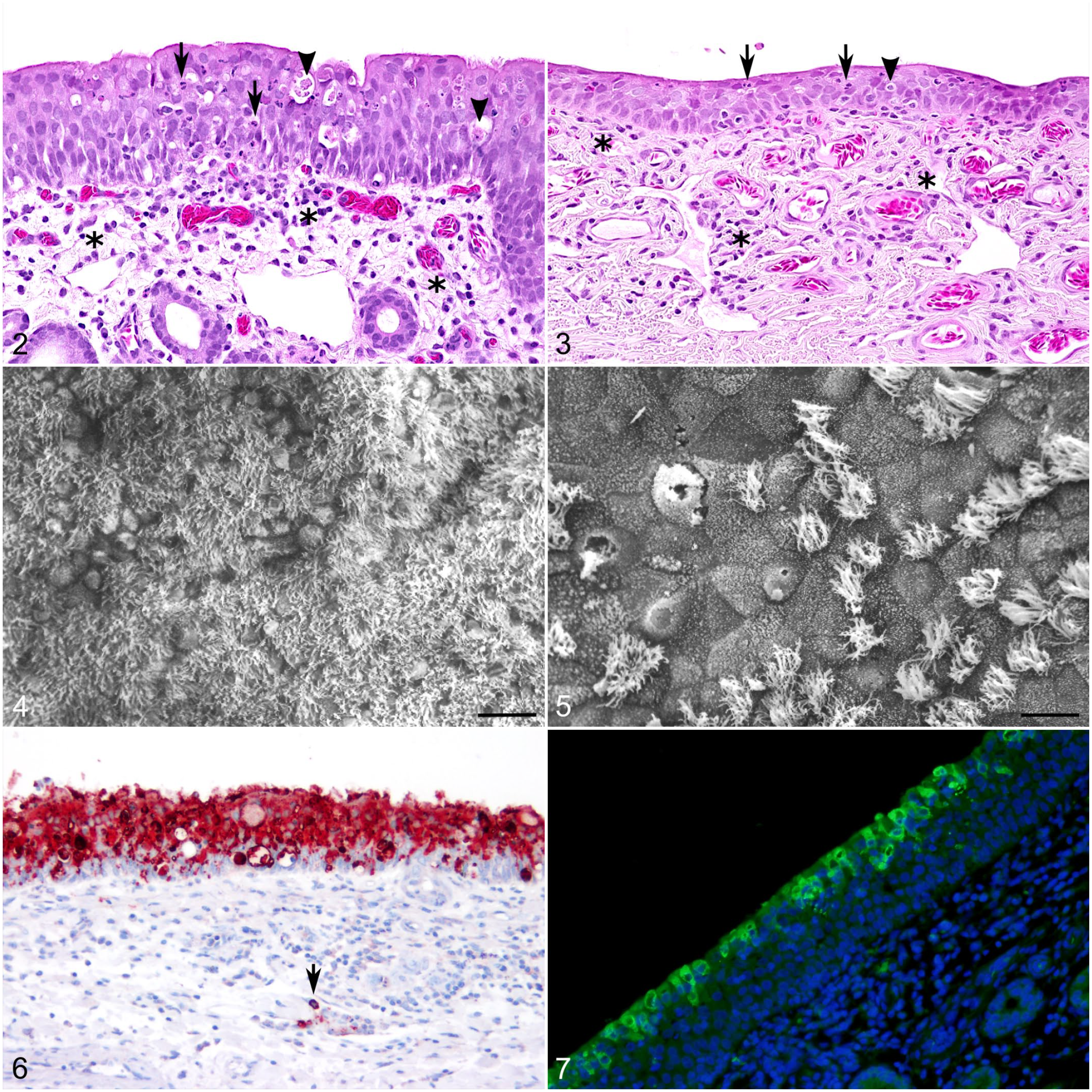
MERS-CoV experimental challenge, dromedary camel, 4 dpi.
In experimentally infected Bactrian camels, lesions are solely observed in the upper respiratory tract and trachea at 5 dpi and included mild-to-moderate lymphocytic sinusitis, rhinitis, and tracheitis accompanied by epithelial necrosis and squamous metaplasia of the respiratory epithelium. 2 Viral antigen was observed in the nasal turbinates, predominantly in the olfactory epithelium, and in the respiratory epithelium of the trachea, but not in any other location. 2
New World Camelids (Llamas and Alpacas)
Pathological features of MERS-CoV-infected New World camelids have been addressed by a limited number of studies. Overall, the outcome of MERS-CoV infection in New World camelids was comparable with that of infected dromedaries, indicating that the animals were mainly asymptomatic carriers of MERS-CoV.1,27,29,56,57,62
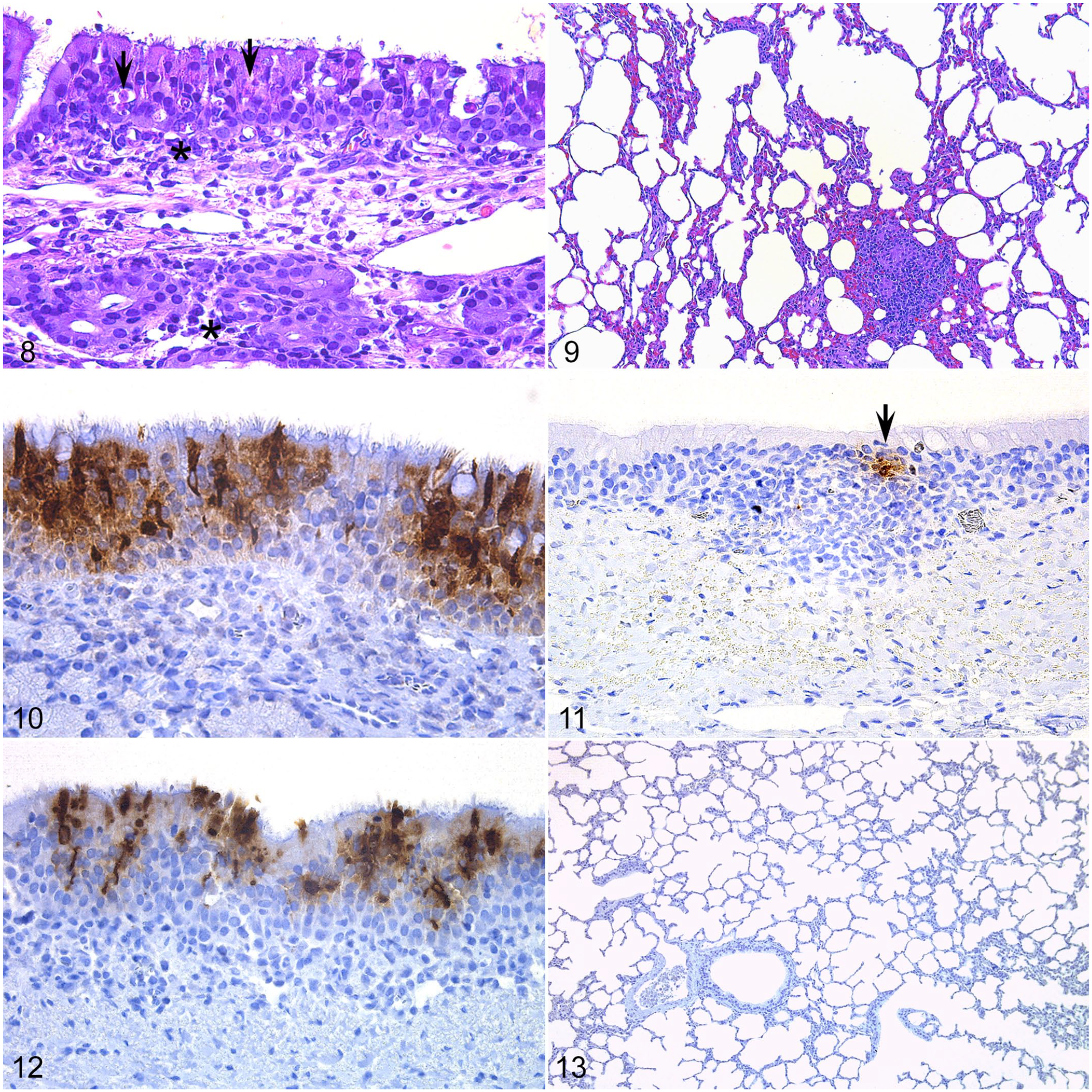
In the acute phase of disease induced by experimental MERS-CoV infection, histological lesions in New World camelids were mainly limited to the respiratory tract (Figs. 8, 9). Nasal turbinates showed mild-to-moderate lymphoplasmacytic rhinitis, characterized by segmental hyperplasia of the mucosa, epithelial vacuolation, slight intraepithelial infiltration of lymphocytes, and rare epithelial cell necrosis (Fig. 8).1,4 Consistently, the underlying lamina propria-submucosa was moderately infiltrated by macrophages, lymphocytes, and plasma cells (Fig. 8).1,4 These lesions tended to be multifocal, showing a similar degree as in dromedaries.
MERS-CoV experimental challenge, alpaca, 2 dpi.
No remarkable findings were observed in the trachea. Some bronchi showed mild epithelial hyperplasia, admixed with few lymphocytes that aggregated in the lamina propria (data not shown). The lungs had slight thickening of the alveolar septa with infiltration of few mononuclear inflammatory cells (Fig. 9). Such lesions were evident in the acute phase of the infection (2–3 days postinoculation) and gradually resolved.
Of note, New World camelids experimentally infected with MERS-CoV showed almost no significant ciliary loss across the respiratory mucosa, except for a single animal with moderate deciliation in the nasal mucosa on day 4 postinoculation. Extrarespiratory organs had no significant lesions.
Consistent with the histopathological findings, the nasal mucosa of the New World camelids displayed the highest amount of MERS-CoV antigen (Fig. 10).1,4 The trachea contained few viral antigen-positive cells mainly in areas displaying lesions (Fig. 11). 4 MERS-CoV antigen was also multifocally distributed in bronchial and bronchiolar epithelial cells but not in type I or II pneumocytes (Figs. 12, 13). Thus, New World camelids animal models develop similar subclinical disease and histopathological findings as seen in dromedaries in response to experimental MERS-CoV infection.
Pathogenesis of MERS-CoV in Camelids
The severe outcome of MERS-CoV infection is driven by dysregulated immune responses in humans, characterized by excessive production of various inflammatory cytokines and chemokines, leading to focal hemorrhagic and necrotizing pneumonia with extrapulmonary manifestations.8,12 In contrast, camelids show minimal or no clinical signs.1,3,68 Furthermore, experimentally inoculated alpacas and llamas could transmit infectious MERS-CoV to other noninoculated, naïve animals via close contact,1,58 indicating that these New World camelids species might be useful surrogates for dromedaries in experimental studies.
Different clinical outcomes of MERS-CoV infection in humans and camelids could be partially due to differences in the spatial and cellular distribution of DPP4, the main cellular receptor of MERS-CoV (Fig. 14). In humans, DPP4 protein was mostly found in the lower respiratory tract. The highest and most consistent expression was reported in type I and II pneumocytes and alveolar macrophages. 49 Additionally, DPP4 can be found in vascular endothelium and pleural mesothelium. 49 Interestingly, the expression of DPP4 was increased in the lungs of patients with chronic obstructive pulmonary disease and cystic fibrosis, which may explain the increased disease severity in patients with chronic pulmonary diseases. 49 DPP4 was absent or rare in the surface epithelium of the nasal cavity, trachea, and bronchi of humans, but it could be detected in subepithelial glands and mononuclear cells in these tissues. 49 In contrast to humans, the receptor protein in dromedaries was mainly expressed in ciliated epithelia of the nasal cavity, trachea, and bronchi, while alveolar epithelia and endothelia were rarely positive. 38 MERS-CoV antigen and mRNA generally colocalized with DPP4, and in vitro experiments showed that DPP4 was essential for virus entry, highlighting the determining role of the receptor in tissue and host tropism of the virus. 37 Nevertheless, other factors besides DPP4 contribute to efficient virus entry and replication and potentially also to disease severity. For instance, certain glycotopes of α2,3-sialic acids, which act as attachment factors for MERS-CoV, show a similar tissue distribution as DPP4 in humans and camelids. Interestingly, these glycotopes were absent from the surface epithelia of the respiratory tract of pigs and rabbits, and these animals did not shed virus efficiently despite robust DPP4 expression. Another attachment factor, CEACAM5, was also shown to facilitate MERS-CoV entry in conjunction with DPP4, and this protein was expressed on cell types susceptible to MERS-CoV infection in the human lung. 17 The distribution of CEACAM5 has not been investigated in camelids.
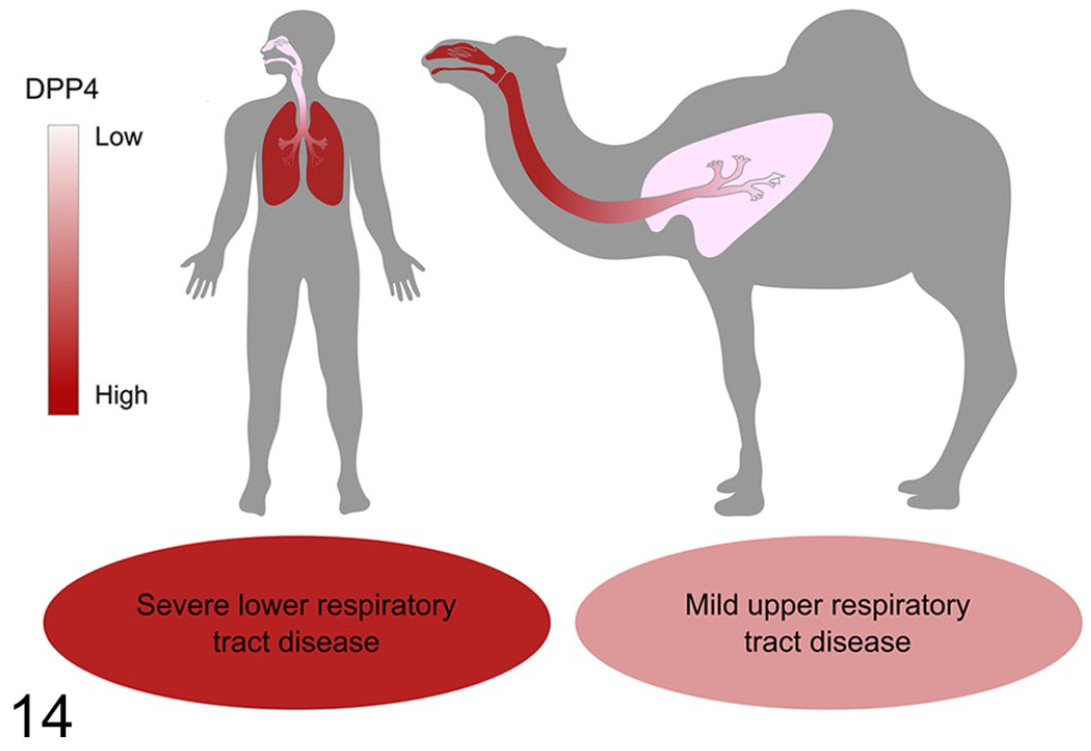
Differences in distribution of DPP4, the cellular receptor for MERS-CoV, in the respiratory tract of humans and dromedaries. In humans, DPP4 is predominantly expressed in the lower respiratory tract, mainly in type II pneumocytes, while it is largely absent in the nasal cavity and trachea. In contrast, the receptor is predominantly expressed in the nasal epithelium, trachea, and bronchi in dromedaries. The distribution of DPP4 corresponds to the distribution of virus following infection. Accordingly, human infection can result in fatal lower respiratory tract disease, while dromedaries usually only display mild and transient clinical signs of upper respiratory tract infection. Besides the differences in DPP4 distribution, other host factors potentially contribute to the different outcome of MERS-CoV infection in humans and camelids, including the distribution of attachment factors such as α2,3-sialic acids and CEACAM5, or differences in innate local immune responses (see section on Pathogenesis of MERS-CoV in camelids).
In addition to entry factors, local innate immune responses influence the outcome of MERS-CoV infection, as demonstrated in an alpaca model. These animals developed early and transient type I and III interferon responses concomitant with the peak of viral (MERS-CoV Qatar-15/2015 strain) infection. Cytokine profiling showed that MERS-CoV-infected nasal epithelial cells produced interferons but distant uninfected cells did not, while interferon-stimulated genes were simultaneously upregulated in both types of cells. Moreover, interferon-stimulated genes were moderately to highly induced in the lamina propria of nasal turbinates as well as in trachea and lungs, but without any detectable interferon mRNA in these tissues. These findings indicate that type I/III interferons produced by infected nasal epithelial cells seem to act in a paracrine/endocrine manner to induce interferon-stimulated gene expression along the whole respiratory tract, which may facilitate rapid virus clearance.
Concomitant to the mild and focal infiltration of some leukocytes in the nasal mucosa and submucosa, the IL10 expression was upregulated along with dampened transcription of proinflammatory cytokines and NLRP3 inflammasome components under NF-κB control, restricting a potential cytokine storm. Moreover, induction of chemokines (CCL2 and CCL3) in the lungs of MERS-CoV-infected animals correlated with a transient accumulation of leukocytes in the absence of IRF5 transcription, suggesting low abundance of M1 macrophages and, thus, controlled inflammation. Therefore, robust and well-timed type I/III interferon mucosal responses (prior or concomitant to maximal viral replication) and dampened inflammation in nasal epithelia may be key features for effectively controlling MERS-CoV infection in camelids that might limit it to a subclinical infection. Of note, low levels of infectious virus were observed in lungs of some alpacas at the peak of infection. In alpacas, DPP4 was abundantly expressed in lungs, strengthening the hypothesis that type I/III interferons produced in the nasal epithelia may act in an endocrine manner in lung cells to limit virus spread.
A comparative pathogenesis study of MERS-CoV in an alpaca model showed that clade B strains (Qatar-15/2015 and Jordan-1/2015) had higher viral replication in respiratory tissues and higher viral shedding than did the clade A EMC/2012 strain, confirming an enhanced replication fitness of MERS-CoV clade B strains in a camelid host. Such characteristics provide a rationale for the dominance of clade B strains in the Arabian Peninsula. Nevertheless, all three MERS-CoV strains led to very similar histopathological changes and innate immune gene profiles, highlighting that the same host mechanisms are responsible for counteracting the different strains of MERS-CoV that infect alpacas. 63
Control of MERS-CoV Infection in Camelids
Vaccination may be an effective strategy to disrupt camel-to-camel, camel-to-human, and subsequent human-to-human transmission of MERS-CoV. Multiple platforms have been used for MERS-CoV vaccine candidates, including inactivated whole virus, live attenuated virus, protein-based vaccines, vector-based vaccines, and DNA vaccines.48,60 However, only few vaccine candidates, encompassing two platforms (replication-deficient viral vector and recombinant protein), have been tested for efficacy in camelids infected with MERS-CoV (Table 1).4,7,36,58
Platforms and candidates of vaccines developed for camelids in response to MERS-CoV.

Abbreviations: MERS-CoV, Middle East respiratory syndrome coronavirus; MVA, Modified Vaccinia virus Ankara; S1, subunit 1; S, spike protein; DCs, dromedary camels; i.m., intramuscularly; i.n., intranasally.
A modified vaccinia virus Ankara vaccine expressing the MERS-CoV spike protein induced a high-level–specific neutralizing antibody response, and reduced excretion of infectious virus and viral RNA in vaccinated dromedaries after MERS-CoV challenge. 36 Here, animals were vaccinated via intramuscular and nasal routes. 36 Of note, protection from clinical signs and reduced lesions correlated with the presence of serum neutralizing antibodies to MERS-CoV. 36 In another study, MERS-CoV seronegative and seropositive dromedaries were vaccinated intramuscularly with a replication-deficient adenoviral-vectored vaccine (ChAdOx1) expressing the MERS-CoV spike protein. 7 After receiving a single intramuscular dose of ChAdOx1 MERS, seropositive dromedaries showed reduced MERS-CoV shedding as well as rhinorrhea and enhanced antibody response. 7 By contrast, at least two doses of vaccine were required to induce antibodies in young seronegative camels under 1 year of age. 7 Also, older animals that were previously seronegative responded more strongly to vaccination than younger animals. 7
Recombinant proteins are another potential vaccination strategy for camelids. In a recent study, llamas were either nonimmunized (naïve) or immunized with a recombinant S1 protein. Each group was later cohoused with nonvaccinated llamas that were experimentally infected with MERS-CoV. 58 While virus was transmitted to all in-contact naïve animals, none of the in-contact vaccinated llamas shed infectious MERS-CoV. Notably, the induction of strong virus neutralizing antibody responses was correlated with protection, demonstrating the high efficacy of the S1 subunit vaccine in blocking MERS-CoV infection in llamas. 58 Similarly, another study showed that a recombinant MERS-CoV S1 protein subunit vaccine completely protected alpacas against experimental MERS-CoV infection and reduced and delayed viral shedding in dromedaries. 4
Despite the promising results of MERS vaccine trails in experimental studies, the lack of licensed vaccines hinders the possibility of vaccinating dromedaries from the Arabian Peninsula, where MERS-CoV circulation is endemic. Thus, extensive implementation of precautionary measures is required to prevent camel-to-camel and camel-to-human transmission, including the use of personal protective equipment, improved infection control awareness, and education of farm and health care workers. 33 People who live in the Middle East should avoid contact with sick camels, drinking raw camel milk or camel urine, or eating raw camel meat, and follow general hygiene measures such as regular hand washing and disinfection of their clothes after contact with dromedaries. 33 Fortunately, as the abovementioned precautionary measures have been applied in the Arabian Peninsula where the virus is known to be endemic, MERS cases and deaths have been decreasing since 2016. 30 Nevertheless, increased surveillance of camel populations and workers in direct contact with infected herds are required to prevent MERS spillover from dromedaries to humans. 5
Conclusions and Future Directions
Camelids consistently develop minor clinical disease or asymptomatic infection upon MERS-CoV exposure. These species rapidly clear the virus from the upper respiratory tract and subsequently mount a robust adaptive immune response against the virus. Thus, camelids may not be a suitable animal model to recapitulate the severe disease that occurs in humans, but they may be useful as a model to develop effective vaccines for other camelids, to prevent future endemic in humans. Given that dromedaries are reservoir hosts of the virus, studies using MERS-CoV-infected camelids as challenge models have helped to fill critical knowledge gaps regarding zoonotic CoVs. Indeed, further detailed understanding on how camelids effectively control the virus without disease progression should aid the development of anti-MERS-CoV therapeutics and vaccines.
Due to the very high MERS-CoV prevalence in dromedaries as well as the continued zoonotic transmission to humans across the Arabian Peninsula, the MERS-CoV epidemic will most likely continue for years to come.31,48 Of note, MERS-CoV clade C strains, which form a separate group from those currently circulating in the Arabian Peninsula,18,21–23 are endemic in dromedaries across Africa. However, locally acquired zoonotic MERS-CoV infections have not been reported so far in Africa. 41 Thus, one could speculate that genetic or phenotypic differences in clade A/B versus clade C might be responsible for the observed differences in zoonotic potential. To shed light on potential evolutionary and molecular mechanisms for the restricted geographic circulation of different viral strains, it would be of interest to compare the pathogenesis and transmission capabilities of clade C African strains with Arabian strains in a camelid model. Such a study could address the question of how MERS-CoV is evolving and acquiring transmissibility and virulence.
Although some camel vaccine candidates were effective against MERS-CoV, there are still numerous knowledge gaps about the correlates of protection including the duration of neutralizing antibodies and the role of T cell-mediated immunity for viral clearance. Looking ahead, the development of effective human and camelid vaccines that elicit long-lasting humoral and cellular immune responses appears to be an ideal way to limit the continuous transmission of MERS-CoV to naïve animals and to humans. However, personal issues including the reticence of farmers to pay for a vaccine against MERS-CoV due to the absence of clinical signs in their camels account for the lack of interest in their vaccination. This may be tackled by education of camel owners. Moreover, compliance with infection control measures and continued surveillance of MERS-CoV among dromedaries with frequent close contact to humans is critical. Adoption of the “One Health” approach to improve preparedness and to mitigate the effects of the MERS epidemic is of paramount importance.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was in part supported by a BMBF (Federal Ministry of Education and Research) project entitled RAPID (Risk assessment in re-pandemic respiratory infectious diseases), 01KI1723G and by the Ministry of Science and Culture of Lower Saxony in Germany (14 - 76103-184 CORONA-15/20) and by the Zoonotic Anticipation and Preparedness Initiative (ZAPI project) (Innovative Medicines initiative [IMI] grant 115760 to NT), with contributions from EFPIA partners, supported by the European commission. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
