Abstract
Smallpox was one of the most devastating diseases known to humanity. Although smallpox was eradicated through a historically successful vaccination campaign, there is concern in the global community that either Variola virus (VARV), the causative agent of smallpox, or another species of Orthopoxvirus could be used as agents of bioterrorism. Therefore, development of countermeasures to Orthopoxvirus infection is a crucial focus in biodefense research, and these efforts rely on the use of various animal models. Smallpox typically presented as a generalized pustular rash with 30 to 40% mortality, and although smallpox-like syndromes can be induced in cynomolgus macaques with VARV, research with this virus is highly restricted; therefore, animal models with other orthopoxviruses have been investigated. Monkeypox virus causes a generalized vesiculopustular rash in rhesus and cynomolgus macaques and induces fatal systemic disease in several rodent species. Ectromelia virus has been extensively studied in mice as a model of orthopoxviral infection in its natural host. Intranasal inoculation of mice with some strains of vaccinia virus produces fatal bronchopneumonia, as does aerosol or intranasal inoculation of mice with cowpox virus. Rabbitpox virus causes pneumonia and fatal systemic infections in rabbits and can be naturally transmitted between rabbits by an aerosol route similar to that of VARV in humans. No single animal model recapitulates all known aspects of human Orthopoxvirus infections, and each model has its advantages and disadvantages. This article provides a brief review of the Orthopoxvirus diseases of humans and the key pathologic features of animal models of Orthopoxvirus infections.
Smallpox was a devastating human disease caused by Variola virus (VARV), a species of Orthopoxvirus, and it had a major influence in shaping global history. VARV is highly infectious by aerosol or microdroplet transmission in a susceptible population, and it is capable of rapidly causing massive epidemics originating from a single index case. 34,77 Because of its highly infectious nature, the disease created catastrophic epidemics in naïve populations but eventually existed endemically in many regions worldwide. In fact, it was maliciously used to decimate select human populations. The first recorded use of smallpox as a biological weapon was in 1763 during the French and Indian War, when British forces in North America distributed blankets used by smallpox patients to Native Americans. 34,46
The onset of Jenner’s refined vaccination procedure and, ultimately, the initiation of the World Health Organization (WHO) Global Smallpox Eradication Program led to the eradication of smallpox; the last natural case occurred in 1977 and the WHO officially declared it eradicated in 1980. 32,34,35 One key feature that facilitated its eradication was that it is strictly a human pathogen with no animal reservoir. Although smallpox no longer exists in nature, there are 2 WHO-sanctioned facilities where repositories of VARV still exist: the Centers for Disease Control and Prevention (Atlanta, GA) and the State Research Center of Virology and Biotechnology (Vector; Koltsovo, Novosibirsk Region, Russia). This virus is maintained at these 2 secure sites for research purposes because some believe that clandestine stocks of VARV may exist and that this virus or a potentially more virulent genetically engineered form could be used as a weapon of bioterrorism. 45,66 It is believed that the aerosol route of exposure would be used in a bioterrorism event because aerosolization allows for rapid viral dispersion over large areas and because the virus is relatively stable in the environment and highly infectious at low doses. 46 Some experts are also concerned that monkeypox virus (MPXV) or a conceivably engineered form of it might also be used as a bioterrorism agent. For these reasons and because smallpox was eradicated before the tools of modern molecular science were discovered, research using VARV and surrogate orthopoxviruses in animal models is ongoing to characterize features of the disease not possible before eradication.
Orthopoxviruses are large linear double-stranded DNA viruses that replicate in the cytoplasm of cells and have a high degree of antigenic similarity 13 —hence, the success of Jenner's vaccine that utilized cowpox virus (CPXV). The orthopoxviruses that can cause disease in humans are VARV, MPXV, CPXV, and vaccinia virus (VACV). Other noteworthy members of the Orthopoxvirus genus that have been the focus of animal model research include rabbitpox virus (RPXV) and ectromelia virus (ECTV), the causative agent of mousepox. The purposes of this article are to review the disease caused by smallpox in humans and the key pathologic features of animal models of Orthopoxvirus infection.
Smallpox in Humans
The most common clinicopathologic presentation of smallpox was a systemically virulent form of the disease known as variola major, which predominately manifested as a generalized pustular rash in a centrifugal pattern (ie, on the head and/or extremities before the trunk), with a case mortality rate of up to 30 to 40%. 34 After microdroplet aerosol exposure through close contact and seeding of the respiratory or oropharyngeal tissues, the virus replicated in the regional lymph nodes. A primary viremia developed whereby the reticuloendothelial system was infected, which corresponded with the incubation period (7–17 days). After replicating in the reticuloendothelial system, a secondary viremia ensued, and the infected individual experienced a sudden onset of prodrome, with fever, malaise, headache, and backache. After 3 to 4 days, enanthema and exanthema developed when the virus, either free or leukocyte cell-associated, localized in small blood vessels of the mucosae and dermis. It was during this time and up to day 10 of the rash that the disease was most contagious because of the high viral titers present in the oropharyngeal tissues. The exanthematous rash began with macules, which progressed to papules, vesicles, and then pustules that became umbilicated. Scabs subsequently developed and, if the person survived, eventually desquamated, often leaving pitted scars called pocks from which the word pox was derived. The scars were typically most severe on the face owing to the predilection of this virus for sebaceous glands, which are more numerous and larger in the face. Complications of the classic form of smallpox include keratitis, corneal ulcers, demyelinating encephalitis, and secondary bacterial infections, including pneumonia. 7,13,28,34
More severe but much less common manifestations of variola major, known as malignant or hemorrhagic smallpox, were associated with a near 100% case fatality rate; 34 the characteristic features of these forms of the disease were flat, smooth coalescing lesions or petechiae and frank hemorrhages, respectively. 46 Because pregnant women seemed particularly susceptible to developing hemorrhagic smallpox, compromised immunity was believed to be in part associated with this form of the disease. 46 A milder form of smallpox, in which fatalities rarely occurred, was known as variola minor or alastrim and caused a less severe clinical disease with fewer cutaneous lesions. 46
The most extensive pathologic descriptions of smallpox in humans date back to the early to mid-20th century. 10,11,22,68 Although natural VARV infection occurred primarily by respiratory exposure resulting in seeding of primary target tissues—such as the oral cavity, nasal passages, nasopharynx or oropharynx, and regional lymph nodes—close examination of these presumed primary sites of infection in natural cases did not reveal primary viral lesions in these tissues. 34 The most profound changes were secondary lesions associated with the characteristic rash in the skin and mucous membranes, and these lesions are well characterized: epithelial hyperplasia, ballooning degeneration and necrosis, vesicle and pustule formation, inflammation, and intracytoplasmic inclusions (Guarnieri bodies). Bronchopneumonia was reported to be the most frequent complication of the disease. 11,68 Some authors attributed the bronchopneumonia directly to VARV, 68 whereas others believed that bronchopneumonia was a complication of a secondary bacterial infection. 10,22 Although the cause of the bronchopneumonia is a topic of debate, it should be noted that mortality was not significantly altered in the postantibiotic era. 78 Inflammation in the interstitium surrounding bronchi and vessels was also reported, 22 as was degeneration and necrosis of alveolar epithelium. 10 Tubulointerstitial nephritis was the most commonly reported finding in the kidneys, and, in cases of hemorrhagic smallpox, there was striking pelvic hemorrhage. 10 Changes reported in the spleen and lymph nodes—namely, lymphoid hyperplasia—were nonspecific but considered associated with viral infection. 78 Hepatocellular fatty change (metamorphosis) and cloudy swelling (degeneration) were most consistently reported in the liver; 22,68 however, these lesions were most likely secondary to severe systemic disease rather than direct infection of the liver by VARV. Other lesions noted include myeloid hyperplasia and necrosis in bone marrow, necrotizing orchitis, and perivascular inflammation and demyelination in the brain. 78 In addition to hemorrhage in the renal pelvis, the lesions of hemorrhagic smallpox consisted of dermal petechiae; subpleural hemorrhages; hemorrhage in the testes, myocardium, and mucosa of the larynx, pharynx, and gastrointestinal tract; and a scarcity of megakaryocytes in the bone marrow, consistent with the clinical finding of thrombocytopenia. 22,34
Some authors consider either interstitial pneumonitis or tubulointerstitial nephritis as a contributing factor in the cause of death in smallpox patients; 78 however, the exact cause of death in patients with classic smallpox and the reason why it was so devastating in humans remain unknown. Because the disease was eradicated before modern molecular techniques were widely available, there remains much to learn about virus–host interactions, virulence factors, and the pathophysiology of this disease. 53,120 Seeding of other tissues, such as the liver, spleen, lymphoid tissues, and bone marrow, apparently occurred; however, VARV did not appear to replicate extensively enough to cause significant organ damage. 34 The only extensive viral cytopathic effects occurred in the skin and mucous membranes. In many cases, the skin lesions were so severe that the patients resembled those who had been severely burned; thus, the cause of death in many cases was attributed to shock secondary to volume depletion, hypotension, and renal failure. 34,78 There are many other theories regarding the cause of death, including circulation of antigen–antibody complexes and activation of the complement cascade 34 or deposition of immune complexes, viral pneumonia and subsequent airway compromise, bacterial sepsis, and viral cytopathic effects. 78 Historical references most commonly attribute death in smallpox patients (classic and hemorrhagic) to toxemia—now most likely believed to have been attributed to a cytokine storm and subsequent septic shock. Furthermore, VARV studies in nonhuman primates have demonstrated an upregulation of inflammatory cytokines, suggesting the crucial role of the immune system’s contribution in the pathogenesis of smallpox. 52,104 Although these theories remain a source of debate, ongoing animal studies will serve to elucidate the cause of death in animal models, compared to what is known of the human disease.
Classic smallpox cases had phasic high viremias, which were associated with the preeruptive and early eruptive stages. 34 In contrast to the classic form, hemorrhagic smallpox was associated with an overwhelmingly high, presumably cell-associated viremia, with continued release of large amounts of virus into the blood from secondary target tissues well into the course of the disease, which correlated with severity of infection. 25 Additionally, levels of antibody response and titers of neutralizing antibodies were strikingly low in hemorrhagic-type cases of smallpox, perhaps reflecting an inadequate immune response when compared to classic-type smallpox, rather than a difference in virulence. 25 The historically described toxemia associated with variola major—especially, the hemorrhagic syndrome—was most likely a result of a combination of sustained or phasic high viremia and an inappropriate immune response, including the production of various cytokines.
An inappropriate host immune response may have been the result of virulence factors. Two such factors associated with host immune evasion have been characterized for VARV: 26 smallpox inhibitor of complement enzymes (SPICE), which downregulates the human complement system, and high-affinity secreted chemokine-binding protein type II (CKBP-II), which binds chemokine receptors. Further investigation of these and other virulence factors will provide insight into pathogenesis and may provide potential strategies for therapeutic intervention. 69
Many aspects of smallpox remain a mystery, and further research on the disease and on countermeasures is needed. Because it is not possible to study naturally occurring smallpox and because experimental VARV infection of humans is ethically and morally prohibited, the US Food and Drug Administration established the animal efficacy rule, or “animal rule.” This rule facilitates the approval of therapeutic strategies and vaccines for microbial agents for which it would be unethical or impracticable to obtain efficacy data in humans, such as diseases caused by pathogens considered a bioterrorist threat. 129,130 Because no one model is expected to replicate all critical features of the human disease, information from a combination of animal models will be needed to fulfill the “animal rule.”
Animal Models of VARV Infection
Researchers in the 1950s and 1960s attempted to develop a nonhuman primate model of smallpox that mirrored ordinary-type or classic smallpox in humans, with little success. 42 -44,64,91,138 In the 1960s, wild-caught Asian macaques were inoculated with aerosolized VARV, and although infection occurred, the resulting disease (ie, pulmonary, systemic, or rash) was slight. In more recent studies, cynomolgus macaques were exposed to high-titer, small-particle aerosols of different strains of VARV; similar to the earlier efforts, viral infection was achieved in the macaques, but the resulting disease was relatively mild, transient, and not particularly faithful to smallpox in humans. 66 The virus–host interactions that result in an apparent resistance of the cynomolgus monkey to develop significant VARV-induced disease, pulmonary or otherwise, are uncertain. However, by adding intravenous (IV) inoculation to the aerosol exposure or by IV inoculation alone, 2 distinct diseases similar to variola major were induced: hemorrhagic disease with high-dose exposure and classic disease with a lower-dose exposure (as outlined below and summarized in Table 1 ). 52
Key Lesions in Animal Models of Orthopoxviruses a
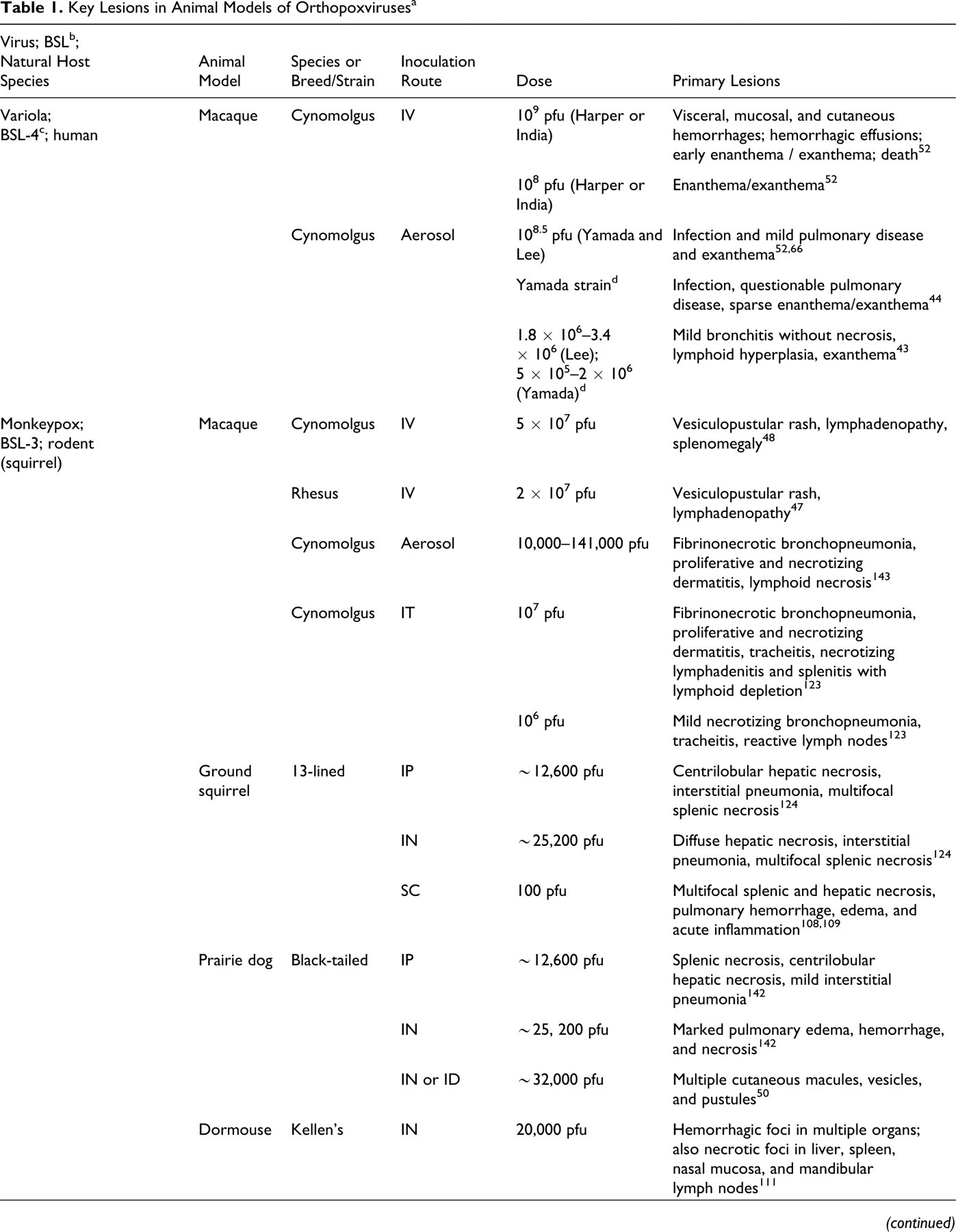
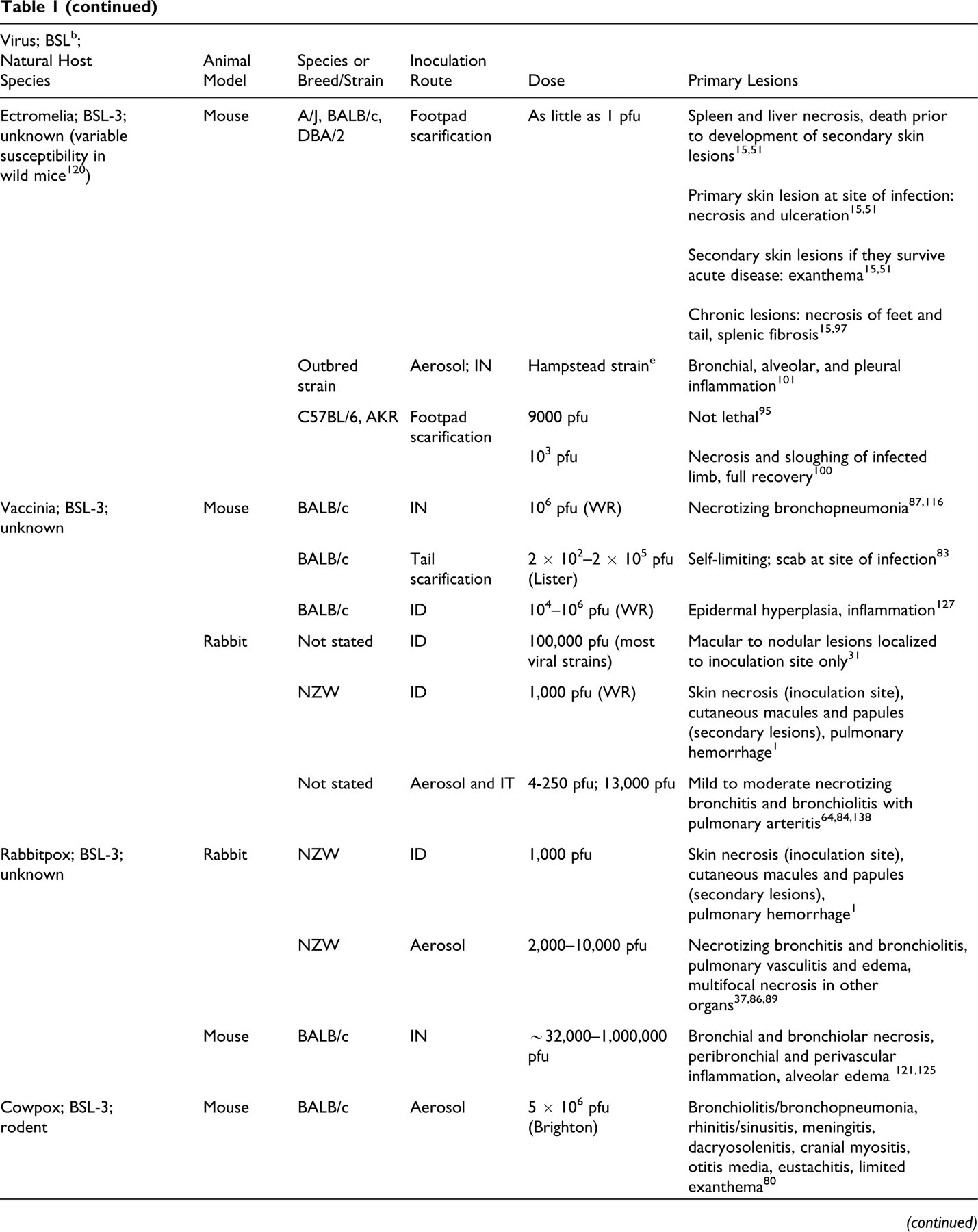
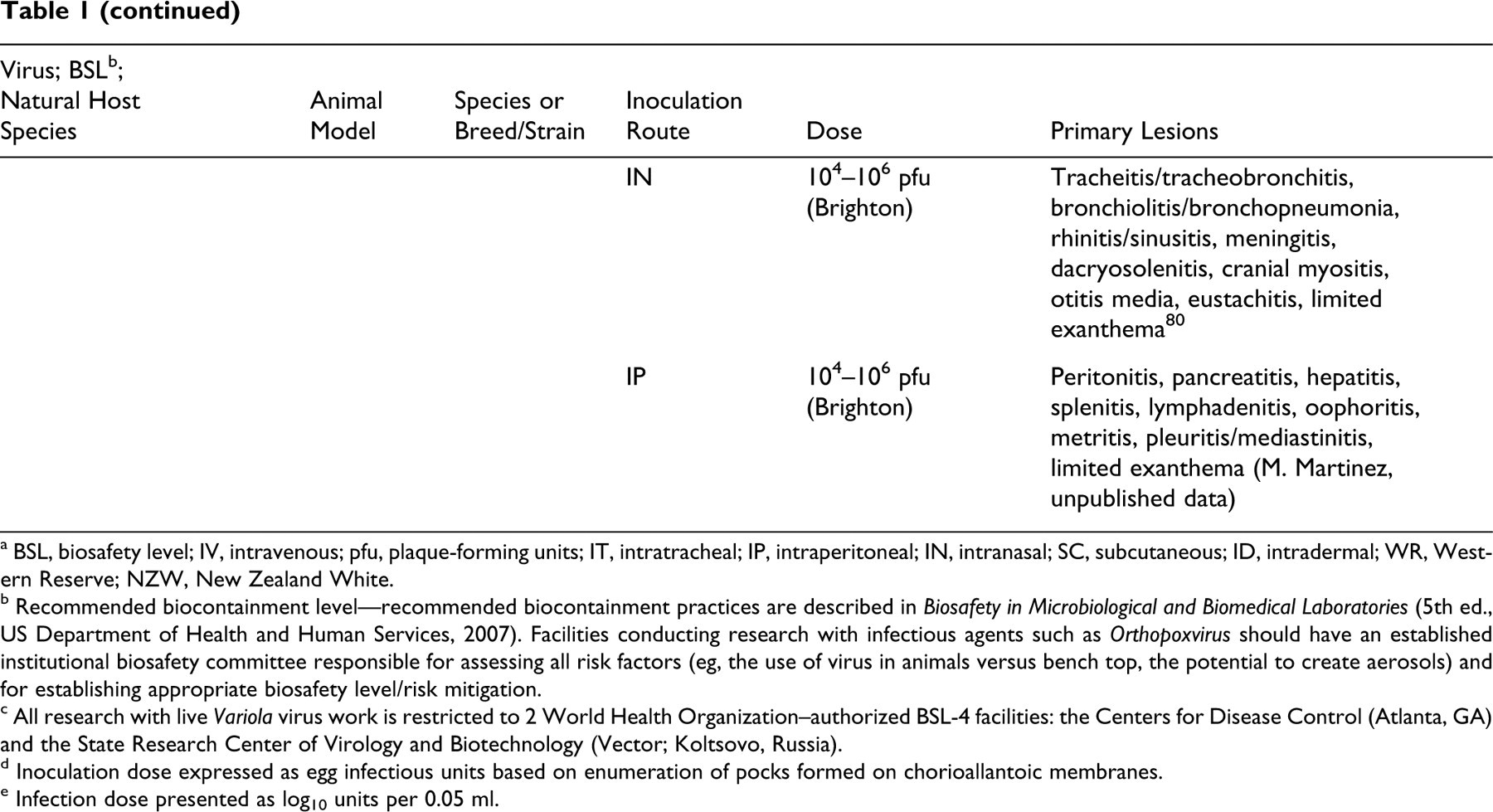
a BSL, biosafety level; IV, intravenous; pfu, plaque-forming units; IT, intratracheal; IP, intraperitoneal; IN, intranasal; SC, subcutaneous; ID, intradermal; WR, Western Reserve; NZW, New Zealand White.
b Recommended biocontainment level—recommended biocontainment practices are described in Biosafety in Microbiological and Biomedical Laboratories (5th ed., US Department of Health and Human Services, 2007). Facilities conducting research with infectious agents such as Orthopoxvirus should have an established institutional biosafety committee responsible for assessing all risk factors (eg, the use of virus in animals versus bench top, the potential to create aerosols) and for establishing appropriate biosafety level/risk mitigation.
c All research with live Variola virus work is restricted to 2 World Health Organization–authorized BSL-4 facilities: the Centers for Disease Control (Atlanta, GA) and the State Research Center of Virology and Biotechnology (Vector; Koltsovo, Russia).
d Inoculation dose expressed as egg infectious units based on enumeration of pocks formed on chorioallantoic membranes.
e Infection dose presented as log10 units per 0.05 ml.
Experimental IV inoculation seemingly bypassed the early stages in the pathogenesis of smallpox—namely, the initial transmission and infection of primary target tissues, as well as the incubation period, primary viremia, and associated prodrome. Experimental IV inoculation presumably created a secondary viremia that induced an acutely febrile disease that led to a hemorrhagic- or classic-type smallpox disease, depending on the dose used. 52
When a high viral inoculation dose (1 × 109 plaque-forming units [pfu]) was administered IV, alone or with aerosolized virus (1 × 108 pfu), a fulminant, uniformly fatal disease occurred that closely resembled the hemorrhagic form of smallpox in humans. 48,52 In addition to the early development of multifocal dermal hemorrhages (ie, petechial rash), cutaneous lesions in the later stages of the disease included vesicles and pustules (Fig. 1 ), with the typical centrifugal distribution, primarily on the extremities and face, similar to that reported in humans. Gross lesions seen at necropsy included petechial hemorrhages on serosal and mucosal surfaces of multiple organs and hemorrhagic visceral lymph nodes. Histologically, lymphoid depletion and lymphocytolysis were prominent in all lymphoid tissues, including the spleen, peripheral and visceral lymph nodes, gut-associated lymphoid tissue, and bone marrow. Renal tubular and hepatocellular degeneration were observed—lesions also reported in humans. Skin, oral, and mucocutaneous lesions included epidermal/epithelial degeneration and necrosis with the formation of vesicles and pustules (Figs. 2A, 2C), similar to lesions in humans. Immunohistochemistry demonstrated viral antigen in these lesions (Fig. 2B). In addition, viral antigen was widely demonstrated in mesenchymal cell types, such as fibroblastic cells and tissue histiocytes and macrophages, 52 suggesting widespread permissive infection and cell-to-cell spread through connective tissues of different organs. Early descriptions of infection of the reticuloendothelial (mononuclear phagocyte) system during the incubation period suggest a similar involvement in the human disease. 10,34
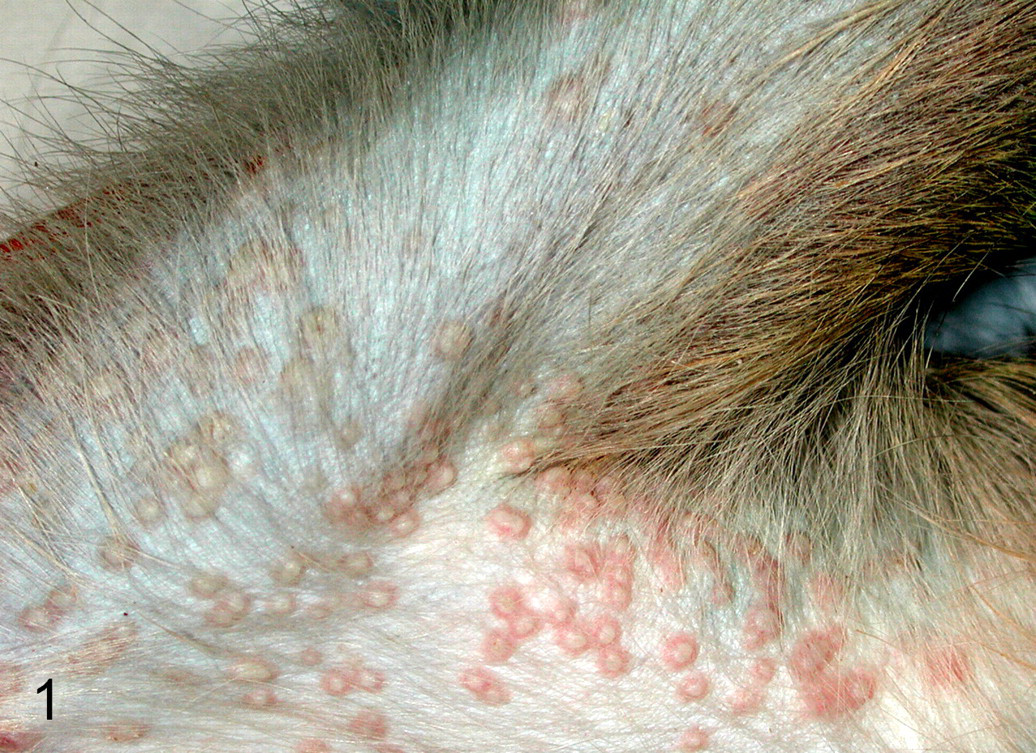
Skin; nonhuman primate. Medial surface of right arm and forearm of a cynomolgus macaque 10 days after aerosol and intravenous inoculation with Variola virus isolate (India 7124 strain). Numerous discrete pustules characterize the exanthema in this case. Note the circumscribing erythema associated with each pustule.
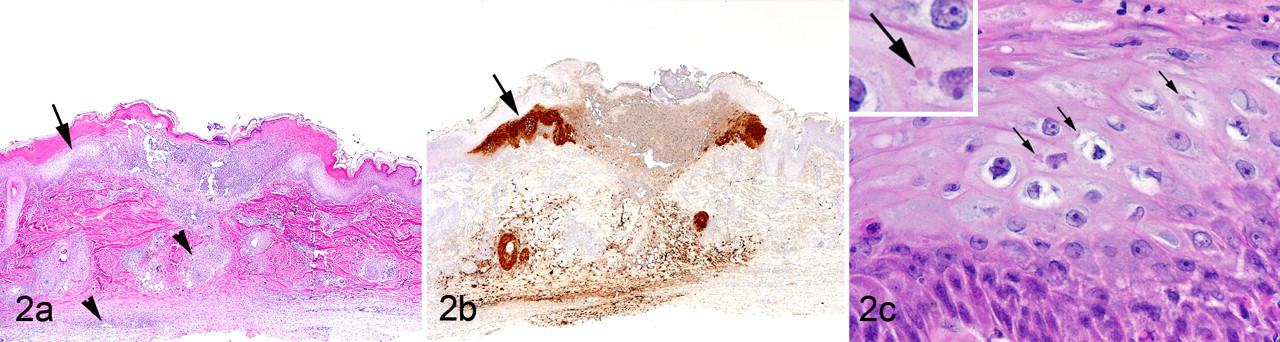
Skin; nonhuman primate. Umbilicated cutaneous pustule from a cynomolgus macaque 10 days after aerosol and intravenous inoculation with Variola virus isolate (India 7124 strain). A, note the sunken roof of the pustule formed by the stratum corneum. As in natural smallpox, umbilication centers on a hair follicle. Note also the pallor of the adjacent edematous yet intact epidermis (arrow), as well as the inflammation in the deep dermis and surrounding adnexal structures (arrowheads). HE. B, immunohistochemical staining for Orthopoxvirus antigen from a replicate section of skin in the first frame. Note immunolabeling of the intact adjacent epidermis (arrow), scattered cells (fibroblasts and macrophages) in subjacent dermis, and follicular epithelium. At this stage in lesion progression, localization of viral antigen has diminished within the pustule contents. Immunoperoxidase method using rabbit polyclonal antibody to vaccinia virus; hematoxylin counterstain. C, higher magnification of first frame. Note keratinocytes of the stratum spinosum. Intermediate zone reveals virus-induced intracellular edema, reflecting cellular (ballooning) degeneration. Small variably sized intracytoplasmic viral inclusions (Guarnieri-like bodies) are present (arrows; inset). HE.
A lower dose (1 × 108 pfu) of IV-inoculated VARV resulted in a more classic model of smallpox, and most monkeys survived. 52 Cutaneous vesicles and pustules developed in a centrifugal pattern, but dermal, mucosal, and serosal hemorrhages were not a predominant feature of this form of the disease (M. Martinez, unpublished data). In a subsequent study performed to assess the efficacy of an antiviral drug, VARV was administered to cynomolgus macaques (IV, 1 × 108 pfu), and the disease endpoints observed in control animals (lesions and death) were similar to those seen in humans. 48
Animal Models Utilizing Other Orthopoxviruses
Animal models utilizing other orthopoxviruses exist or are being developed and refined as models of human Orthopoxvirus infection. Owing to the narrow host range of VARV and the requirement that work with it be conducted under the most stringent biocontainment level—biosafety level 4—it is desirable to find models using closely related orthopoxviruses that cause disease in perhaps more facilitative animal species and that can be studied under less stringent biocontainment conditions. Table 1 and the following sections outline key aspects of the animal models of Orthopoxvirus infections currently being studied as surrogates for VARV, including their pathologic features.
Monkeypox in Africa
The first reported cases of spontaneous MPXV infection were in captive cynomolgus macaques with a widespread cutaneous pustular disease. 133 It was later determined that primates are not the natural host for this virus, which maintains an enzootic cycle in the rain forests of Central and West Africa, involving squirrels and possibly other rodents as natural reservoir hosts. 61,62
Human monkeypox was first reported in 1970 in the former Zaire (now known as the Democratic Republic of Congo), in a region where smallpox was thought to have been eradicated. 63,70,75 It is now believed that human monkeypox most likely existed previously but was mistaken for smallpox. 12 When smallpox vaccination programs were terminated after smallpox was declared eradicated, increasingly larger outbreaks of human monkeypox occurred in Central and West Africa during the 1980s and 1990s. In fact, the smallpox vaccination status, as determined by the presence of a smallpox vaccination scar, among a susceptible population in the outbreak areas influenced monkeypox case frequency as well as disease course and severity. In general, affected populations with higher numbers of vaccinated individuals experienced lower disease rates, and in those vaccinees that did develop disease, it was less severe. 57
Primary disease transmission, which is from animal to human, presumably involved exposure to infected rodent species (especially, squirrels). However, a WHO-sponsored epidemiological investigation of an upsurge of monkeypox cases during 1996 and 1998 in the Democratic Republic of Congo indicated that secondary transmission (person to person), rather than primary transmission (animal to human), accounted for the majority of those cases. 16,17,56,85,140 Monkeypox in humans is similar to classic variola major and is characterized by exanthema, fever, and respiratory signs. In contrast to smallpox, human monkeypox has a lower case mortality rate (approximately 10%), and there is striking peripheral lymphadenopathy, usually involving cervical and inguinal lymph nodes. 57 Affected individuals in West Africa usually have milder forms of the disease than do those in Central Africa, and there are genetic differences between Western and Central African strains of MPXV. 105,106 Although the disease is endemic in these areas of Africa, clinical studies of human monkeypox are difficult to conduct because of the sporadic nature of disease outbreaks, the poor accessibility to the remote areas in which outbreaks usually occur, and the lack of substantive medical and research facilities in these areas.
Monkeypox in the Western Hemisphere
In 2003, a shipment of many hundred African rodents destined for the exotic pet trade arrived in the United States (Texas) from Ghana. Some of these rodent species—rope squirrels (Funiscuirus sp), Gambian giant pouched rats (Cricetomys gambianus), and dormice (Graphiurus sp)—were later determined to be infected with MPXV and had transmitted the virus to captive prairie dogs (Cynomys sp) also being sold as pets. Some of the Gambian pouched rats and prairie dogs became ill, predominantly with conjunctivitis, skin lesions, and lymphadenopathy. 41,65,111
Consequently, the first-ever human monkeypox case in the Western hemisphere was discovered, and 71 cases of human monkeypox were eventually reported to the Centers for Disease Control and Prevention. 24 The majority of the 37 laboratory-confirmed cases involved contact with prairie dogs or other species of exotic pets. 24 Most of the infected individuals had relatively mild clinical signs; there were no fatalities, and no cases were determined to be solely a result of secondary transmission (person to person). 18,19 The MPXV involved in this outbreak, MPXV-USA, was a West African strain. 105,106
Nonhuman primates
Clinical disease caused by naturally acquired and experimental MPXV infection has been reported in several species of nonhuman primates. 2,82,107,133 Although rhesus and cynomolgus macaques are both susceptible, the latter are more sensitive to infection and usually display more severe clinical signs. 99,107,133
Researchers in the 1960s discovered that cynomolgus macaques inoculated intramuscularly with the virus developed a systemic viral infection and widespread vesicular–pustular rash, 137 and this was used as a model for studying disease pathogenesis and to test a potential antiviral therapeutic agent. 20,135,136 However, a large area of muscle and skin necrosis typically developed at the inoculation site, 136,137 and because of this undesirable sequela, the intramuscular route of inoculation is no longer used; nonhuman primate monkeypox models currently in development have primarily utilized IV, aerosol, or intratracheal (IT) inoculation routes.
Early studies involving IV inoculation of rhesus macaques with MPXV reported that infected animals had a widespread vesiculopustular rash with a primarily centrifugal distribution. 81,99 More recently, rhesus macaques given an IV dose of 2 × 107 pfu developed a severe disease that was uniformly fatal 7 to 14 days after inoculation. 47 Lesions present in these animals included a generalized vesiculopustular rash and marked lymphadenopathy. This rhesus model was used to test the efficacy of DNA-based smallpox vaccines. 47
After IV challenge with 5 × 107 pfu of MPXV, cynomolgus macaques develop a generalized vesiculopustular rash that begins on the head and extremities and then spreads to the rest of the body, much like human MPXV and VARV. 48 Clinically, there is fever with leukocytosis, and at necropsy there is prominent lymphadenopathy with splenomegaly and pulmonary edema. 48 This animal model has been used for efficacy studies of an antiviral therapeutic agent. 48,58
Aerosolized and IT routes of infection are the 2 principal respiratory models of MPXV infection that have been investigated. Aerosolization of pathogens is logistically and technically demanding and requires specialized facilities and equipment. This complicated method of delivery also presents challenges in assessing actual retained dose in real time, so more direct delivery methods may provide a more easily measured challenge dose. IT infection permits instillation of a precise dose of virus directly into the trachea without relying on specialized aerosol chambers. IT infection has traditionally been achieved by instilling a bolus of fluid in the trachea; however, a novel technique has recently been evaluated involving application of an aerosol via a microsprayer attached to a bronchoscope (A. Goff, personal communication).
An extensive pathological characterization of aerosolized MPXV in cynomolgus macaques has been described. 143 The pathogenesis of the disease, with systemic dissemination of the virus through a monocytic cell–associated viremia, is similar to that of VARV in humans. Death of the animals was attributed to fibrinonecrotic bronchopneumonia, and necrotizing lesions were present in target tissues, including lymphoid organs, skin and mucosal surfaces, and reproductive organs. Epithelial proliferation was a consistent feature in the skin and mucosal tissues. Intracytoplasmic inclusion bodies and/or multinucleated syncytial cells were occasionally present in the epithelium of the skin, oral cavity, tonsil, and gastrointestinal tract. Immunohistochemistry and electron microscopy identified antigen and viral particles, respectively, in tissues with morphologic alterations. With the exception of bronchopneumonia, the development of vesiculopustular rash (Fig. 3 ) and the distribution of lesions in these aerosol-challenged macaques were similar to those of macaques challenged with IV MPXV 48 and humans infected with MPXV and VARV. Intranasal (IN) challenge with MPXV has been described, and it produces lesions similar to those described in the aerosolized model. 105
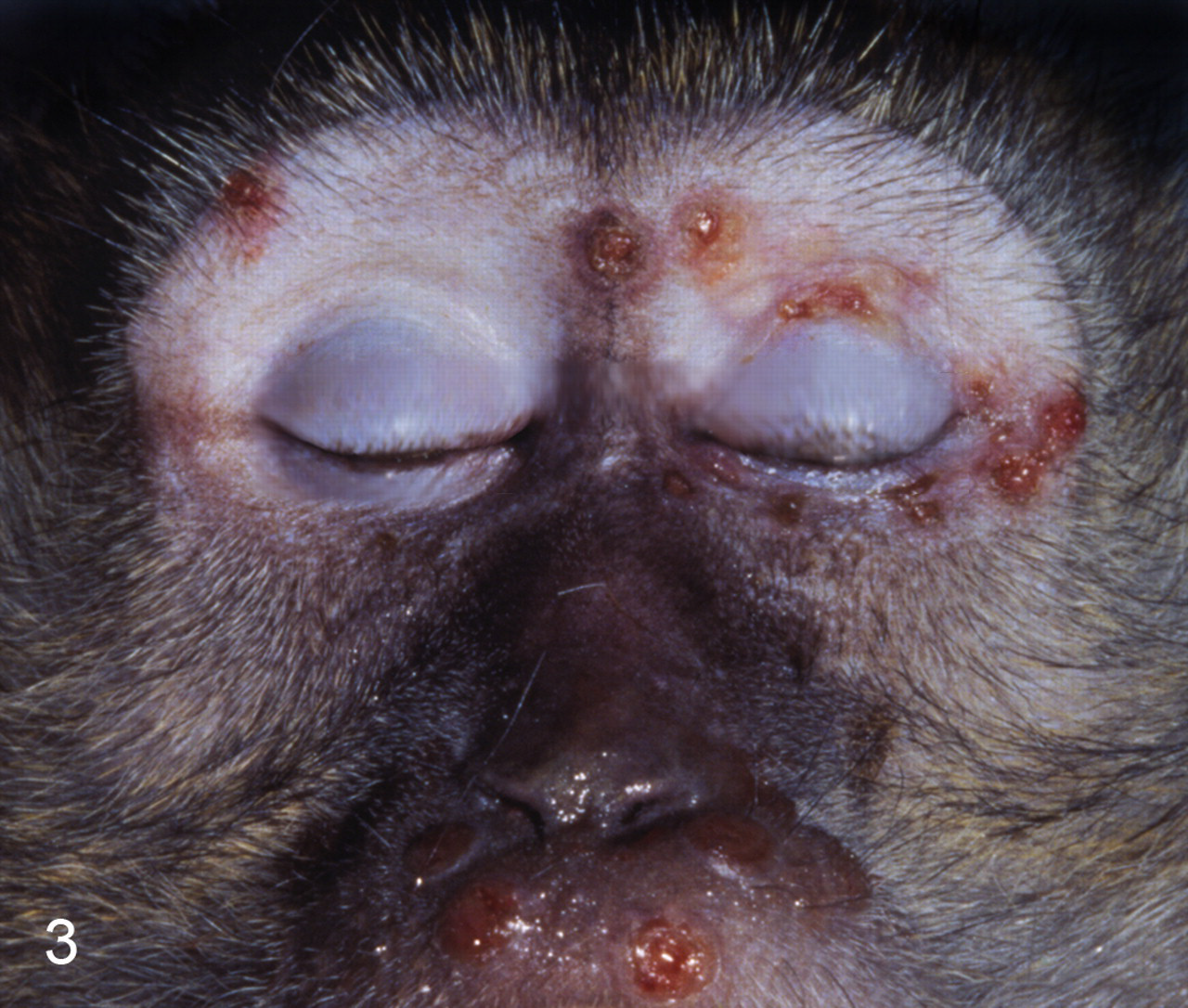
Skin; nonhuman primate. Cynomolgus macaque that died 17 days after experimental inoculation with aerosolized monkeypox virus (Zaire strain). Note the umbilicated and crusted lesions typical of this stage of the disease. Similar lesions are seen with intravenous inoculation with monkeypox virus.
The IT monkeypox infection model in cynomolgus macaques has been utilized to test the efficacy of vaccines and antiviral compounds. 122,123 Animals challenged with IT MPXV developed fibrinonecrotic bronchopneumonia and necrotizing lesions in the tongue, lymph nodes, and spleen. Histological evaluation revealed lymphoid depletion in multiple organs as well as proliferative and necrotizing lesions in the skin, with ballooning degeneration and intracytoplasmic inclusion bodies. Electron microscopy demonstrated poxvirus particles within keratinocytes and in the alveolar wall. 123
One of the advantages of using the aerosolized and IT models of MPXV virus infection is that they more closely mimic the natural route of transmission versus other models. However, the amount of virus delivered directly into the respiratory system in these models is much greater than what is naturally transmitted and thus may be more representative of an aerosolized bioterrorism event. 143
Rodents
Early attempts to experimentally infect animals with MPXV by various inoculation routes found that adult animals of some of the traditional laboratory species of rodents and rabbits usually developed only mild clinical signs, if any. 73,112 Generalized and lethal infections could be produced in infant mice, rats, and rabbits; 73,112 however, because of the small size of the animals and the immature status of their immune systems, infant rodents and/or rabbits are not currently used as animal models of MPXV infection. Cotton rats (Sigmodon hispidus) and Eurasian red squirrels (Sciurus vulgaris) were found to be susceptible to lethal MPXV infection when inoculated IV (cotton rats), IN, per os, or by skin scarification (red squirrels). 76,111 However, descriptions of the pathology findings in these animals are limited, and neither species has been further utilized as a model for studying the viral infection.
In recently published studies, several strains of mice with intact immune systems were found to be completely resistant to MPXV, or they developed only transient, nonlethal viral infections when inoculated intraperitoneally (IP), IN, or by foot pad injection. 49,92,119 However, mice with severe combined immune deficiency were found to be highly susceptible to MPXV when inoculated IP or IN. Consistent virus-associated histologic lesions in the mice challenged IP included ovarian necrosis and inflammation, intestinal serositis, and multifocal epidermal ballooning degeneration. 92 However, the severely compromised immune system of these mice limits their usefulness as a model of natural MPXV infection.
C57BL/6 mice lacking STAT1, which is a protein involved with interferon signaling, were reported to be highly sensitive to IN challenge with MPXV; significant histologic lesions in these animals included suppurative rhinitis and leptomeningitis. 119 This model was used in vaccine and antiviral efficacy trials. 119
Subsequent to the 2003 human monkeypox outbreak in the United States, there have been several attempts to develop rodent models of human smallpox and human monkeypox by experimentally infecting 13-lined ground squirrels (Spermophilus tridecemlineatus), black-tailed prairie dogs (Cynomys ludovicianus), and an African species of dormouse (Graphiurus kelleni) with different strains of MPXV using various viral doses and routes of inoculation. IP or IN inoculation of ground squirrels with high doses of MPXV-USA resulted in 100% mortality in 6 to 7 days for the IP route and 8 to 9 days for the IN route. 124 Significant histologic lesions for the IP route included centrilobular hepatocyte degeneration and necrosis with intracytoplasmic basophilic inclusion bodies. Squirrels inoculated IN had more diffuse hepatocellular necrosis with similar intracytoplasmic inclusions. For both inoculation routes, there were foci of necrosis in the spleen and interstitial pneumonia with foci of consolidation.
A subcutaneous (SC) route of inoculation was used to compare the pathogenicity of MPXV-USA and a Central African strain of MPXV in ground squirrels. 109 Both viral strains caused disseminated infections, with 100% mortality in 6 to 11 days postchallenge; however, the disease caused by the Central African MPXV strain was more rapid and severe. Although no gross lesions were described, the histologic lesions caused by both viral strains included pulmonary hemorrhage, edema, and acute inflammation, as well as foci of hepatic and splenic necrosis, with intracytoplasmic inclusions in hepatocytes. For all 3 routes of infection, the widespread severe disease that MPXV caused in ground squirrels is more similar to the hemorrhagic form of human smallpox than it is to classic smallpox.
IP inoculation of prairie dogs with a high dose of MPXV-USA resulted in 100% mortality within 11 days. 142 No gross lesions were described, but the most significant histologic lesions consisted of splenic necrosis, centrilobular hepatic necrosis, and interstitial pneumonia; intracytoplasmic inclusions were present in some hepatocytes.
When prairie dogs were inoculated IN with a high dose of MPXV-USA, 3 of the 5 (60%) challenged animals died 11 to 14 days postchallenge. 142 In contrast to the prairie dogs challenged IP, the animals that died after IN challenge had severe pulmonary edema, hemorrhage, and necrosis but no lesions in their spleens or livers. The 2 animals that survived the IN challenge developed transient mucopurulent nasal discharge and vesicles on lips and tongue, but these lesions resolved.
Both IN and intradermal (ID) routes of inoculation in prairie dogs have been used to compare the in vivo effects of MPXV-USA and a Central African strain of the virus. 50 Each viral strain by either inoculation route caused disseminated skin lesions that developed in a centrifugal pattern; these lesions were described as appearing first as macules, which then developed into vesicles, followed by pustules and, finally, scabs. No other descriptions of the gross or histologic lesions were provided. A more severe experimental disease was caused by the Central African strain of MPXV, and the only fatalities that occurred were in animals infected with this strain—specifically, 2 of the 4 inoculated ID died (50%) and 1 of the 4 inoculated IN died (25%).
The 13-lined ground squirrel and the prairie dog are not ideal species for use in poxvirus research. 111 The animals available for purchase are captured from wild populations, and there is currently no source of captive-bred and raised animals; thus, there is little control over pathogens to which these animals have been exposed and other factors affecting health. Additionally, the females produce only 1 litter of young per year, which greatly hinders the ability to establish a self-sustaining captive colony able to produce enough animals to meet research needs.
IN inoculation of dormice with various doses of a Central African strain of MPXV caused 100% mortality in 7 to 10 days with viral challenge doses of 200 and 2,000 pfu. 111 Similar results were reported when dormice were challenged with a West African strain of MPXV. In other experiments using the Central African MPXV strain and a challenge dose of 20,000 pfu, disseminated viral infections occurred, with viral titers detected in multiple tissues; gross lesions included hepatomegaly, hemorrhage in the upper gastrointestinal tract, and lymphadenopathy. The most prominent and consistent histologic lesions reported were foci of hemorrhage and necrosis in the nasal mucosa, liver, spleen, mandibular lymph nodes, as well as hemorrhagic foci in lungs, stomach, adrenal glands, and other organs. Viral inclusions were present in liver, spleen, and nasal mucosa. It was concluded that the severe disseminated disease produced in the dormice resembled the hemorrhagic form of smallpox more than it did other forms of smallpox or monkeypox in humans. 111 The primary disadvantage of this animal model is that limited numbers of these dormice are available in the United States for use in research; importation of wild-caught specimens has been banned, and only a small colony of laboratory-raised animals—specifically for use in Orthopoxvirus research—has been established at the National Institutes of Health (Bethesda, MD). 60
Ectromelia Virus
ECTV, the causative agent of mousepox, was first described in England in 1930 and caused a fatal disease in laboratory mice. 33 Susceptibility to ECTV is dependent on host factors—such as mouse strain, age, sex, and immune status—and virus factors. The virus is infectious at low doses and causes high mortality in susceptible mouse strains—including A/J, BALB/c, DBA/2, and C3H—whereas C57BL6 and AKR mice are resistant to severe disease. 14,30,51,110 Susceptible mouse strains succumb quickly and are not an efficient source of shedding and transmission, whereas strains that survive long enough to develop lesions are a source of infection, and resistant strains may be a source during asymptomatic infection. 51,97 ECTV infection in mice has been extensively studied, and the pathogenesis, with localized replication and systemic spread, is similar to the pathogenesis of VARV in humans. 33,59 Various routes of infection have been investigated, including ID, SC, IN, IT, IV, IP, intracerebral (IC), and aerosol. 33,110
The virus is naturally transmitted through abrasions in the skin, so footpad scarification is a commonly used method of infection in the mouse model. After localized replication in regional lymph nodes, primary viremia ensues, resulting in infection of liver and spleen, followed by secondary viremia and seeding of other tissues via a monocytic cell–associated viremia. 14,33 If mice survive the acute phase, they develop a generalized maculopapular rash similar to that seen in humans with smallpox. 30,33 Bilateral conjunctivitis may also be present, and large amounts of virus are shed in ocular discharge. 33 Those mice that develop the chronic form of the disease develop cutaneous ulceration and necrosis of the feet and tail (which can result in amputation) and a unique mosaic pattern of splenic fibrosis. 28,51,97
Histologically, there is necrosis in the liver, spleen, and lymphoid tissues, with variably discernable basophilic or eosinophilic intracytoplasmic inclusion bodies in infected cells, especially in hepatocytes at the periphery of necrotic foci. 97 Cutaneous lesions consist of epithelial hyperplasia, ballooning degeneration, and prominent intracytoplasmic inclusions, with development of erosion and inflammation. 97,102
Although it is not a natural mode of transmission, aerosolization of the virus has been used to mimic a poxvirus bioterrorism event. 96 With aerosol infection, the virus is taken up by alveolar macrophages and/or epithelium of the upper and lower respiratory tract and transported by macrophages to the regional lymph nodes and spleen. 33,101 Virus is also present in the olfactory epithelium and extends through the cribriform plate to the olfactory bulb. 101 IN infection of ECTV has been used as a route to more closely imitate the natural route of infection for smallpox. 33,96
The progression of ECTV-induced disease in mice can be measured by percentage body weight change and viral titers in the liver, spleen, and other tissues. 59 Body weight change is a simple variable to measure and is advantageous for evaluating antiviral drug efficacy. Another advantage of the ectromelia mouse model is the capability of evaluating an Orthopoxvirus in its host species. This is valuable not only because a low dose of virus causes lethal infections but also because so much is known about mouse genetics and immunology, providing a means for investigating immunomodulatory genes through targeted deletion. 33,120 A disadvantage of the mouse model of ectromelia infection is the hepatotropic nature of the virus in mice, a feature not observed in human infection with orthopoxviruses. 1 In fact, death in acute cases of mousepox is attributed to hepatic and splenic necrosis, presumably owing to the cytopathic effects of the virus. 120 Although hepatocellular degeneration was observed in human cases of smallpox, this did not appear to develop as a direct result of viral infection of the liver, and liver failure was not believed to be a cause of death, 53 as in mice infected with ECTV. An additional essential consideration in using this pathogen in research is the potential for spread of this virus through the research colony.
Vaccinia Virus
The origin of VACV is unknown. It has been suggested that VACV may have arisen as a hybrid of orthopoxviruses, from serial passages of Orthopoxvirus in the laboratory or in nature, or even from a now-extinct virus or natural host. 15,34 Whatever the origin, it is a distinct species of Orthopoxvirus that sometime during the 19th century, became widely used for smallpox vaccination; as such, a majority of clinical descriptions of VACV in humans are a result of vaccination. 34
The clinical features of VACV infection in immunocompetent individuals are localized to the site of vaccination. 13 The cutaneous lesion progresses through the classic stages of Orthopoxvirus disease—papule, vesicle, pustule, scab, and desquamated lesion. 7 Individuals occasionally display malaise and fever, indicating systemic spread. Adverse reactions include potential complications owing to generalized vaccinia, eczema vaccinatum, progressive vaccinia (vaccinia necrosum), myocarditis, and postvaccinal encephalitis. Individuals with compromised immune systems or other underlying diseases are much more likely to develop systemic infections, and because of this, the conventional VACV-derived smallpox vaccine is contraindicated for people with immune deficiencies, eczema and other forms of atopic dermatitis, and heart disease. 141
Although most clinical descriptions of VACV are associated with vaccination, over the past decade zoonotic outbreaks of VACV infection in Brazil have been reported in people who have direct contact with infected cattle. 126 The origin of this strain of VACV is unknown (ie, wild type versus survival of a vaccine strain); however, it persists in Brazil as a zoonotic disease. 126 Affected individuals develop lesions on the hands and forearms, as well as fever, malaise, and lymphadenopathy. 23,29,113 A similar zoonotic disease is transmitted by domestic Asian water buffalo (Bubalus bubalis) in India and is caused by buffalopox virus, a subspecies of VACV. 114 Additionally, individuals who drink unpasteurized milk from infected animals may develop oropharyngeal lesions. 28
Mice
The species of laboratory animal in which VACV has been most extensively studied is the mouse, and there is specific mouse and virus strain susceptibility. Mice have been infected via a variety of routes, including IV, IP, SC, ID, IN, and footpad scarification. Intracranial inoculation has been utilized to produce an animal model to study postvaccinal encephalitis, which is a serious but infrequent complication of smallpox vaccination in humans. 8 VACV infection of various immunodeficient strains of mice has been studied and used to test the efficacy of antiviral compounds. 88,118
The IN route has been extensively studied, and in susceptible mice such as BALB/c, the International Health Division (IHD) and Western Reserve (WR) strains of VACV cause lethal infection. 115,116,141 Pulmonary gross lesions are described in one report and are characterized by fluid accumulation, hemorrhage, and consolidation. 116 Pulmonary histopathology is described in a report of IN infection of BALB/c with a recombinant VACV characterized by peribronchial and perivascular inflammation and intra-alveolar edema. 128 Nelson reported a fatal necrotizing bronchopneumonia with IN instillation of VACV. 87 Williamson et al proposed a pathogenesis after infecting DBA/2 mice IN with the WR strain of VACV: respiratory infection, followed by viremia, followed by infection of the central nervous system (CNS). 139 However, the authors could not exclude direct CNS infection by extension through the cribriform plate.
The mouse model of VACV infection provides several advantages. The pathogenesis of IN VACV infection—with local replication, primary viremia, and systemic dissemination following secondary viremia—closely resembles human infection with smallpox. 59 The IN route of infection is an important model in mimicking disease that would be produced in a bioterrorism event via the aerosol route. Additionally, disease severity and systemic replication of the virus correlate with body weight loss, similar to the mouse model of ECTV. This variable is simple to measure and has proven useful in studies that assess antiviral compound efficacy by measuring its ability to inhibit virus-induced weight loss. 59,116
Other routes of VACV infection that have been extensively studied in mice include tail scarification, infection of the ear pinna, and footpad inoculation. Tail scarification usually produces a localized self-limiting infection, although virus recovery from the spleen, thymus, lung, and liver has been described. 14 An advantage of tail scarification is that it provides a means of evaluating smallpox vaccination; namely, the lesions progress from the pustular stage to the scab stage and, upon separation, to a scar that is similar to the “clinical take” in human smallpox vaccination. 83
According to Tscharke et al, vaccination of the ear pinna causes less distress for the mice than that of footpad inoculation, and sectioning of the tissue for histology is easier than it is with the tail. 127 The authors provided a pictorial representation of gross and histopathologic ear lesions over the course of the infection. The characteristic lesions consisted of dermal inflammation and epidermal proliferation, which was most severe at day 8 postinfection. Based on immunohistochemistry, antigen was demonstrated in the hyperplastic epithelium early in the course of infection, then in inflammatory cells present in the dermis before being cleared.
Rabbits
When most strains of VACV are inoculated into rabbits, they cause a relatively mild disease, with lesions usually localized to the primary site of infection: skin for ID inoculation and lungs for aerosol or IT inoculation. 31,64,84,138 However, strains of VACV classified as rabbitpox virus (discussed in the following section) do cause significant systemic infections in rabbits. 5,31 In a recent report, ID inoculation of rabbits with the WR strain of VACV unexpectedly produced a systemic disease, with high mortality in 9-week-old rabbits and lesser mortality (60%) in 6-month-old animals. 1 The disease course and gross lesions were similar to those described after ID inoculation of RPXV, except there were more numerous and prominent secondary skin lesions in the rabbits infected with the WR strain. No histopathologic findings were reported. Further studies are needed to determine whether infection of rabbits with the WR strain of VACV could serve as a useful animal model of systemic Orthopoxvirus infection.
Rabbitpox
In the 1930s, multiple outbreaks of a systemic and often fatal disease occurred in laboratory colonies of rabbits housed at the Rockefeller Institute for Medical Research in the United States. Because this rabbit disease was caused by a poxvirus and had many clinical and gross pathological features similar to those of smallpox in humans, it was named rabbitpox. 39,40,103 In the 1940s, outbreaks of a similar poxviral disease occurred in rabbits housed at the University of Utrecht (Netherlands). In contrast to the cases at the Rockefeller Institute, affected rabbits at the University of Utrecht typically had few skin lesions, if any; this disease became known as pockless rabbitpox. 54,55 During subsequent outbreaks of rabbitpox in Europe and the United States, the disease closely resembled the form originally seen in the Netherlands. 21,131 Rabbitpox has not been recognized outside of a laboratory setting, and there have not been any reports of a spontaneous outbreak of this disease in a rabbit colony since the mid-1960s.
Rabbitpox is caused by infection with RPXV, an orthopoxvirus considered to be a strain of VACV that is particularly virulent in rabbits. 67 Fenner demonstrated that there were distinct biological differences between the Utrecht and Rockefeller strains of RPXV. 31 Other experimental studies of rabbits using a variety of inoculation routes indicated that although the viral strains caused a systemic disease with similar lesions, the Utrecht strain was much more pathogenic and produced a more rapid and consistently fatal disease course. 5 However, when low doses of the Utrecht strain were administered, rabbits had a more prolonged disease and developed skin lesions similar to those seen with the Rockefeller strain. 138
All published rabbitpox animal studies in the last 40+ years have utilized the Utrecht strain of RPXV; the Rockefeller strain may no longer exist. Therefore, the following discussion of rabbitpox covers work done with only the Utrecht strain of RPXV.
Rabbits
When inoculated ID with RPXV, 9-week-old rabbits developed a systemic viral infection, with 100% mortality by day 9 postchallenge. 1 The same viral dose in 6-month-old animals caused a similar clinical disease but with 80% mortality. In both age groups, the initial gross lesions occurred at the sites of inoculation, which became red and swollen and then necrotic and black by days 3 to 5 postchallenge. These necrotic foci tended to increase in size until the animals succumbed. Secondary skin lesions developed on the ears, lips, and other sites away from the area of viral inoculation by days 5 to 8 postchallenge and appeared as round red-to-brown papules measuring up to 1 cm in diameter. Mucopurulent nasal and ocular discharges were seen around the same time as the secondary skin lesions; this was followed by the development of respiratory distress and then death. Postmortem examination of the rabbits revealed that the lungs were dark and marbled with foci of hemorrhage. However, there were no descriptions of the histologic appearance of any of the lesions in these rabbits. This rabbit model was subsequently used to study the efficacy of an antiviral compound. 1
Others working with ID RPXV inoculation of rabbits have presented limited histology findings, but these concerned only the skin lesions that developed at the sites of inoculation; early epidermal vacuolar degeneration accompanied by dermal edema and inflammation (primarily heterophilic) was followed by necrosis of the epidermis, adnexa, and dermis. 38,79,121
In the spontaneous outbreaks of rabbitpox that occurred, epidemiologic evidence pointed to inhalation of aerosolized viral particles as the most likely method by which most rabbits were infected. Experimental inoculation of rabbits with aerosolized RPXV was first reported in the 1960s. Although the infected rabbits in these experimental studies developed a fatal systemic viral disease and although the clinical signs and gross lesions were well described, the only histologic findings mentioned were brief descriptions of the pulmonary lesions and limited descriptions of the lesions in the testes and adrenals. 64,138
Researchers at the US Army Medical Research Institute of Infectious Diseases (USAMRIID) have more recently developed aerosolized RPXV inoculation of rabbits as an animal model for studying aerosolized Orthopoxvirus infection and have used this model in efficacy studies of poxvirus vaccines and an antipoxvirus therapeutic drug. 37,86,89 This model utilizes a small-particle (~ 1 μm) aerosol of RPXV and viral challenge doses that cause 100% mortality by 5 to 7 days postchallenge. The most consistent gross necropsy lesions are congestion and edema of the mandibular and mediastinal lymph nodes and edema of the lungs and mediastinum, often accompanied by foci of pulmonary consolidation and serous to serofibrinous thoracic exudates. Swelling of the head and neck owing to SC edema is also common. Rabbits that survive to day 6 or 7 postchallenge sometimes have 1- to 2-mm red-to-tan macules and papules on their lips, eyelids, and, occasionally, ears; larger skin lesions are occasionally present in areas that are shaved before viral challenge but are rare in unshaved areas (D. Nichols, unpublished data). Histologically, the most extensive and severe lesions are located in the respiratory tract and consist of necrotizing rhinitis, proliferative and necrotizing bronchitis and bronchiolitis (Figs. 4A, 4B), and multifocal pulmonary vasculitis, usually accompanied by moderate to marked pulmonary edema. Other common lesions include multifocal acute necrosis and inflammation in other organs—most frequently, the liver, gonads, and/or adrenal cortices. When present, skin lesions initially present with ballooning epidermal degeneration, accompanied by edema and heterophilic and histiocytic inflammation in the underlying dermis; later, epidermal vesicles and pustules develop. These findings agree with and expand on those reported in the 1960s. 64,138
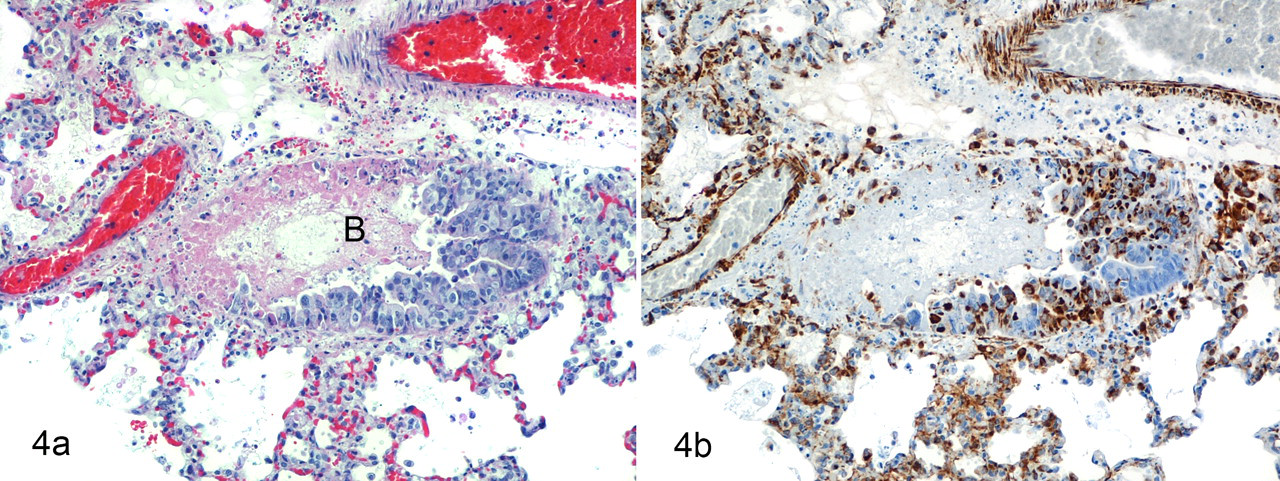
A, lung; rabbit. This animal died 6 days after experimental inoculation with a lethal dose of aerosolized rabbitpox virus. There is hypertrophy and hyperplasia of the epithelium lining a bronchiole (B), with focally-extensive epithelial necrosis and acute inflammation. Vasculitis, necrosis, hemorrhage, and fibrin are present in the adjacent interstitium. Similar lesions are seen in cynomolgus macaques exposed to aerosolized monkeypox virus and in mice exposed to aerosolized cowpox virus. HE. B, immunohistochemical staining for Orthopoxvirus antigens of a replicate section of rabbit lung from the first frame. Abundant intracellular and extracellular viral antigens are in areas of necrosis and inflammation, including smooth muscle cells in the walls of inflamed blood vessels. Immunoperoxidase method using mouse monoclonal antibody to vaccinia virus; hematoxylin counterstain.
Westwood et al reported that experimentally infected rabbits could transmit the disease to naïve sentinel animals housed in the same room. 138 Because there was no direct contact between the rabbits, it was assumed that the sentinels became infected by inhalation of aerosolized virus shed by the experimentally infected rabbits. Recent studies conducted at the USAMRIID have shown viral transmission from rabbits experimentally infected with aerosolized RPXV to noncontact sentinel animals housed in the same room; the affected sentinel rabbits usually had a more prolonged disease course with numerous skin lesions, which suggests that the viral infection dose was lower than that of the experimentally inoculated animals (A. Nalca and D. Nichols, unpublished data).
Likewise, rabbits inoculated ID with RPXV have transmitted the virus to sentinel rabbits. 1 The clinical signs, disease course, and gross lesions in sentinel animals that had direct contact with inoculated rabbits (ie, housed in the same cage) were similar to those present in the inoculated rabbits, except the sentinel animals did not develop primary skin lesions and they had pronounced swelling of the face and neck. No histopathology findings were reported. The onset of disease in these sentinel rabbits indicated that they had been infected at the time that the inoculated animals developed naso-ocular discharges; all together, this observation, the lack of any primary skin lesions consistent with direct-contact transmission of the virus to the sentinel rabbits, and the presence of the facial and cervical swelling in the sentinel animals suggest that the route of infection in the sentinel rabbits was inhalation of the virus into the upper respiratory tract. There was a delay of 1 to 2 days in the development of clinical signs in sentinel rabbits that did not have direct contact with ID-inoculated rabbits; one possible explanation is that the sentinels without direct contact were infected with a lower viral dose than were those with direct contact. That RPXV is readily and naturally transmitted between rabbits by an aerosol route, as is VARV among people, makes rabbitpox a useful animal model of smallpox.
Rabbits have also been infected with RPXV using IN, IV, SC, IP, and conjunctival routes of inoculation. 5,21 Each route produced an initial localized infection, followed by viremia and widespread viral dissemination with lesions in multiple organs. Because the publications pertaining to these experiments are all more than 40 years old and none of these inoculation routes are currently being used to study RPXV infections in rabbits, the diseases produced in these experiments are not discussed here.
Mice
Infection of mice with RPXV has been utilized to study the pathogenicity of the virus and several mutant strains. 9,27,79,121,125 Although routes of inoculation have included IV, IP, IN, and IC, most studies were based on clinical signs, mortality, and/or viral titers in various tissues. However, the only descriptions of lesions were the pulmonary changes present in animals infected IN; these mice had multifocal necrosis of bronchial and bronchiolar epithelium with alveolar edema and mixed inflammatory cell infiltrates around airways and blood vessels. 121,125
Cowpox Virus
As a zoonotic disease, cowpox in humans is rare but potentially severe, with the majority of recent cases traced to contact with infected domestic cats. CPXV is endemic in Europe, in some western states of the former Soviet Union, and in adjacent areas of northern and central Asia. 3,6,134 The name of the virus is based on the cutaneous lesions that it caused in domestic cattle; however, the natural reservoir hosts are wild rodents. Natural infection and disease occur in cattle, humans, domestic cats, pet rats, and various captive mammals in zoological collections. 29 In contrast to smallpox, CPXV infection in humans is due to direct implantation of CPXV into compromised skin and/or oral mucous membranes. Autoinoculation potentially contributes to the development of additional cutaneous and/or mucosal lesions. In immunocompetent individuals, infections are characterized by painful hemorrhagic pustules or eschars, usually on the hands or face, with edema, erythema, lymphadenopathy, and systemic involvement. Conjunctivitis and keratitis are also reported. Fatal disease rarely occurs and is not completely characterized, although isolated cases of heart failure and encephalitis have been reported and usually occur in immunocompromised individuals or those with eczematous disease. 4,74,90
Some of the earliest reports of poxvirus infection in rodents (presumably, CPXV infection) described dermal and pulmonary disease in white rats. 72 Subsequently, wild-caught Norwegian rats were experimentally infected with this ratpox virus by various routes and developed nasal, pulmonary, intestinal, and cutaneous lesions. 71 Presently, various strains of laboratory mice are known to be susceptible to CPXV infection by multiple routes of exposure, including ID, IT, IP, IN, and aerosol. The pathology resulting from IP, IN, or aerosol routes of exposure to CPXV is briefly discussed here.
Vinogradov et al reported that mice inoculated IP with CPXV developed viral infection in a range of cell types in the peritoneal cavity; however, the livers of these animals were not affected, in contrast to those of mice infected with ECTV. 132 Others have observed that IP inoculation of BALB/c mice with CPXV resulted in a rapidly lethal disease with a wide dissemination of virus throughout the peritoneal cavity (M. Martinez, unpublished data). Virus-induced inflammation, necrosis, and hemorrhage involved the liver, pancreas, uterus, ovarian bursa, serosa/peritoneum, abdominal adipose tissue, and uncommonly, the spleen and ovary. Virus-induced inflammation, necrosis, and hemorrhage of the diaphragm, mediastinum, thoracic adipose tissue, pleurae, and lymph nodes were present and were attributed to migration of infected peritoneal macrophages into the thoracic cavity.
Efforts to develop a mouse model of respiratory CPXV infection were supported by the acceptance that smallpox was transmitted by respiratory exposure. Specifically, development of such a model focused on small-particle aerosol or IN microdroplet inoculation of various strains of mice. 94,110,117,125 Thompson et al showed that IN inoculation of wild-type CPXV resulted in pulmonary disease in BALB/c mice; however, the upper respiratory tract was not examined. 125 Martinez et al showed that the virus-induced lesions in the upper and lower respiratory tracts of BALB/c mice that were inoculated with a lethal dose of aerosolized or IN CPXV were similar between the 2 routes of inoculation, although the distribution and severity differed slightly. 80 Time-dependent progression of virus-induced inflammation of the lungs, nasal passages, and sinuses was present in both groups in terms of extent and severity; however, IN inoculation was associated with more extensive and severe lesions in nasal passages and hilar pulmonary regions, whereas aerosol exposure produced less severe upper respiratory disease and more peripherally distributed pulmonary lesions. Both groups also had virus-induced inflammation in the bronchiolar vessels, pleura, mediastinum, trachea, perineurium of olfactory nerves, meninges of the olfactory bulbs, and skin. Similar to others' observations, hemorrhage was a component of CPXV-induced lesions. 80,98,125
Analysis of the various CPXV mouse models has provided a more comprehensive understanding of Orthopoxvirus infection. These models can provide data pertinent to virus immunomodulation, virus–host interaction, and host immune response, and they can identify and assess the safety and efficacy of various compounds or vaccines with activity against orthopoxviruses. 36,93
Summary
The global eradication of smallpox is one of the greatest public health successes in history. However, because the threat exists for intentional or accidental release of VARV and other orthopoxviruses infectious for humans, development of medical countermeasures to these viruses is essential. Fundamental to investigating these countermeasures is the development of animal models of Orthopoxvirus infection, including a thorough characterization of the lesions and pathogenesis in host and adapted species. Although such characterization has been adequately described for a few of the animal models, most of them currently have only limited pathology descriptions. Thus, further work in this area is clearly needed. Additionally, because smallpox was eradicated before modern molecular techniques were discovered, much effort is being made to study the interaction between the host immune reaction and orthopoxviruses. 104 This aspect of the disease is critical to explore because, in addition to the viral cytopathic effects, a cytokine storm is one of the proposed mechanisms of the pathogenesis and cause of death in human smallpox. 59,120
Inhalation of aerosolized viral particles is believed to be the route by which VARV and MPXV are naturally transmitted, and virus aerosolization is the means most likely to be used in a bioterrorism event. Thus, research utilizing aerosolized orthopoxviruses has been an important focus of animal model development, but there is value in exploring animal models using other routes of infection. For example, skin scarification in the ectromelia mouse model provides insight into the pathogenesis of a systemic Orthopoxvirus disease via a natural route of infection; furthermore, IC inoculation in the VACV mouse model provides a model to study postvaccinal encephalitis, a serious complication of smallpox vaccination. Although an IV route of infection is an unnatural route of entry and the primary viremia is bypassed, the utility of animal models employing IV inoculation of orthopoxviruses is based on the following premise: If protection by medical countermeasures is successful in an animal infected by this rigorous challenge route, then certainly that same compound would be effective against another route of infection. 53
Because they have anatomical and immunological features most similar to those of humans, nonhuman primates are generally regarded as the fundamental species for animal models of smallpox (and many other human diseases). However, nonhuman primates are relatively expensive to purchase and maintain, and they require much larger housing space than that of rodents or rabbits. Nonhuman primates also potentially harbor zoonotic diseases (eg, herpes B virus) and can be physically difficult and dangerous to handle. The housing and handling issues are particularly significant under biocontainment conditions, where space is limited and personnel must wear special protective equipment that makes the simplest tasks more difficult to perform.
Rodent and rabbit Orthopoxvirus models offer the advantage of utilizing species that are much less expensive than nonhuman primates, require less housing space, and are easier and less dangerous to handle. Therefore, experimental studies with rodents or rabbits can more readily accommodate large numbers of animals, thereby increasing the statistical power of the experimental results. In addition, most of the commercially available strains of rodents (and, to a lesser extent, rabbits) offer the opportunity to conduct studies with relatively uniform and well-characterized populations of animals. The primary disadvantages of rodent and rabbit animal models are their anatomic and immunologic differences with humans. In addition, the small body size of these species, compared to that of nonhuman primates, causes more limitations on the volume of blood and other biologic samples collected from individual animals.
Animal models of MPXV infection serve a dual function: They not only provide strong surrogates for studying smallpox but can act as models of human monkeypox. However, there is utility in studying host-adapted orthopoxviruses, such as ECTV in mice and RPXV in rabbits, because these viruses have naturally evolved to cause a systemic infection in their host, thus mimicking VARV infection in humans. 59
There is no one animal model that can recapitulate all aspects of human Orthopoxvirus infections, and each model has its advantages and disadvantages. Therefore, certain features of the various animal models will need to be linked to provide a more complete picture of the human disease, thus fulfilling the requirements of the US Food and Drug Administration’s “animal rule.” Adequately defining the pathology of animal models of orthopoxviruses will serve as a key factor in this process and in the evolution of investigating medical countermeasures to these viruses.
Footnotes
Acknowledgements
We gratefully acknowledge Mr Neil Davis, Mr Jeff Brubaker, and Ms Gale Krietz for excellent technical assistance; Ms Chris Mech for immunohistochemical preparations; Dr Bernard Moss for providing the antibody used for Variola virus immunohistochemistry studies; and Mr Larry Ostby, Mr Bill Discher, and Ms Sissy Riley for photographic assistance. The views, opinions, and/or findings contained herein are those of the authors and should not be construed as an official Department of Army position, policy, or decision unless so designated by other documentation. All US Army Medical Research Institute of Infectious Diseases studies mentioned in this review involved protocols approved by the Institutional Animal Care and Use Committee, and research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals at the times the studies were conducted. Experiments involving animals adhered to principles stated in the Guide for the Care and Use of Laboratory Animals of the National Research Council. The US Army Medical Research Institute of Infectious Diseases is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
