Abstract
Recently, several cases of human cowpox virus (CPXV) infections were reported in France and Germany, which had been acquired through close contact with infected pet rats. The animals exhibited respiratory signs or skin lesions and died shortly after purchase. After natural infection of white rats with CPXV in the USSR in 1978, a peracute pulmonary form, a milder dermal form, and a mixed form exhibiting features of both have been described. To the best of the authors’ knowledge, 3 experimental cowpox virus infection studies using rats have been performed to date; however, neither results of histomorphological examinations nor immunohistochemical analyses have yet been reported in rats after experimental infections. To investigate the impact of the infection route on the clinical course, the development of lesions, and tropism, rats were infected intradermally, intranasally, or by a combination of both routes. The authors found a correlation between clinical manifestation, pathology, and infection routes. Intradermal and contact exposure yielded a mild dermal form, characterized by the development of vesiculopustular dermatitis. In contrast, intranasally infected animals died peracutely, showing severe dyspnea. Occasionally, a combination of the dermal and the respiratory form occurred after intranasal infection. Immunohistochemically, CPXV antigen was detected in the epithelial and mesenchymal cells of the upper respiratory tract and affected skin lesions and rarely in mesenchymal cells of lymph nodes. This is the first histomorphological and immunohistochemical analysis of CPXV in rats after experimental infection.
The genus Orthopoxvirus (OPV), family Poxviridae, includes 4 species that are pathogenic for humans: variola (causing smallpox), monkeypox, cowpox (CPXV), and vaccinia virus. In Europe and parts of northern and central Asia, CPXV is endemic. 22 Wild rodents are considered reservoir hosts, and natural infections and disease have been described in cattle, humans, domestic cats, pet rats, and captive mammals such as elephants in zoological gardens. 9 The termination of smallpox vaccination and hence a growing naive population could be relevant for increasing frequency of CPXV infections in humans. 6
Most human CPXV cases are caused by contact with infected cats; however, several cases of CPXV infections in humans, which had been acquired through close contact to infected pet rats, have been reported recently in France and Germany. The animals exhibited respiratory signs or skin lesions and died shortly after purchase. Affected owners frequently developed fever when skin lesions were in the pustular stage, as well as associated regional lymphangitis and lymphadenopathy. 1,6,18
In natural infection of white rats with CPXV, 3 different clinical patterns were described in the USSR in 1978: a peracute pulmonary form, a milder dermal form, and a mixed form. 14 The pulmonary form was characterized by dyspnea, loss of appetite, and flaccidity and ended lethally within 4 days after onset of clinical signs. Macroscopic examination revealed diffuse or focal pneumonia with serous or hemorrhagic pleural exudate. The intestines and stomachs were distended, but skin and mucosal rash were not observed. 14
The milder dermal form came along with papular rash, rhinitis and conjunctivitis with hemorrhagic discharge, occasional anorexia, flaccidity, and necrosis of the tail and hind feet with rare deaths. At necropsy, there were dermal eruptions, focal pneumonia, and mucosal exanthema. 14 Histomorphological data were not presented. To study CPXV infections, various mouse models have been developed in the past using intradermal, intratracheal, intraperitoneal, and intranasal infection routes. 7 These CPXV mouse models have provided a deeper understanding of Orthopoxvirus infection, including virus-host interactions and pathogenesis, especially regarding viral immunomodulation.
To the best of our knowledge, 3 experimental cowpox virus infection studies using rats have been performed to date; however, neither results of a histomorphological examination nor results of an immunohistochemical analysis of CPXV have been reported in rats after experimental infection.
In the first experiment, intranasal infection as well as infection by scarification of wild rats (Rattus norvegicus) with CPXV led to 100% mortality with respiratory signs, proliferative skin lesions, hemorrhages, and distension of the stomach. 13 Both infection routes induced a similar course of disease but was less severe and prolonged after scarification. 13 In the second experiment, infection of white rats with various isolates by intranasal, oral, and contact infection induced clinical manifestations. Skin lesions were not detectable after intranasal infection, and the highest virus concentrations were found in the lungs. A detailed description of the clinical course, gross lesions, and histopathology was not given. 15
The aim of the third experiment was to investigate the role of rats either as potential reservoir hosts or as accidental hosts of CPXV. 12 Pet rats and laboratory rats were infected (intranasally or intranasally combined with scarification) with a pet rat–derived cowpox virus strain. The results indicate that at least with regard to this CPXV strain, rats might be a reservoir or accidental host for CPXV. After an incubation period of 6 days, the animals developed a severe, often fatal disease with large amounts of virus detected in oropharyngeal secretions. Virus shedding could be demonstrated before the onset of clinical signs, which enhances the potential for humans to be infected by rats during the incubation period. Furthermore, it was found that tissues from lesions in the respiratory tract and skin were most likely to give positive results in titration and real-time polymerase chain reaction (PCR). In numerous other tissues (including brain, liver, and salivary gland), low loads of CPXV DNA were detected. 12 Whether these findings were due to viremia or tissue-specific variable CPXV replication is unclear.
The present study is complementary to the recent, third experiment. The aim of this study was to elucidate potential replication sites and pathogenesis of CPXV infections in rats. We analyzed whether the infection route has an influence on the outcome of the disease. To this end, clinical signs, pathologic changes, and viral antigen tropism of CPXV in the rats after experimental intradermal, intranasal, or combined infection were compared.
Material and Methods
Cowpox virus strain ratpox 09 was used, which originated from a diseased rat that had bitten 2 girls who subsequently developed cutaneous lesions at the site of the bite (Meyer, unpublished data). The virus was propagated on Vero cells (CCLV-RIE 109) and stored at –70°C until use. Wistar rats (6 weeks old, female and male; Charles River, Germany) were infected with 106 TCID50 (100 μl) per animal: 5 animals were infected intranasally (group IN), and 5 animals were infected intradermally by cautiously scarifying the dorsal base of the tail and applying the virus (group ID). In a third group, 5 rats were infected via both routes (group ND). To each group, 2 contact animals were added 1 day postinfection (dpi). Two additional rats served as noninfected negative controls. The animals were monitored daily for clinical signs. Moribund rats (IN and ND groups) were anesthetized with isofluorane and euthanized by exsanguination. For histopathological investigation, 2 rats in the ID group were sacrificed at 11 dpi to compare histomorphological data with the IN and ND animals. Finally, all surviving infected rats were euthanized at 23 dpi, when the general condition was good but dermal lesions were still visible. The contact animals were killed 36 dpi after complete recovery. All animal experiments were reviewed and approved by the responsible state ethics animal welfare/Institutional Animal Care and Use Committee (IACUC; LALLF M-V/TSD/7221.3–2.1.-005/09).
Tissues of skin, nasal cavity, trachea, lung, esophagus, larynx, mandibular lymph node, salivary gland, heart, brain and liver were sampled from all rats in 4% phosphate-buffered neutral formaldehyde and processed for paraffin embedding. Paraffin-wax sections (3–4 μm) were dewaxed and stained with hematoxylin and eosin (HE). To determine the distribution of CPXV, immunohistochemistry was performed using the avidin-biotin-complex (ABC) method as previously described. 3 Slides were incubated with a rabbit anti-CPXV serum (kindly provided by Bundeswehr Institute of Microbiology, München, Germany; dilution: 1:2000 in Tris-buffered-saline, TBS). A biotinylated goat anti-rabbit IgG1 (Vector, Burlingame, CA, diluted 1:200 in TBS) was applied for the ABC method. Positive control sections of monkeypox virus–infected tissues (kindly provided by K. Mätz-Rensing, German Primate Center, Göttingen, Germany) were included, and a rabbit serum against bovine papillomavirus (BPV, 1:2000) was used as nonrelated control antibody.
Results
Clinical Observations
The clinical signs and gross lesions are summarized in Table 1. The survival rate is shown in Figure 1.
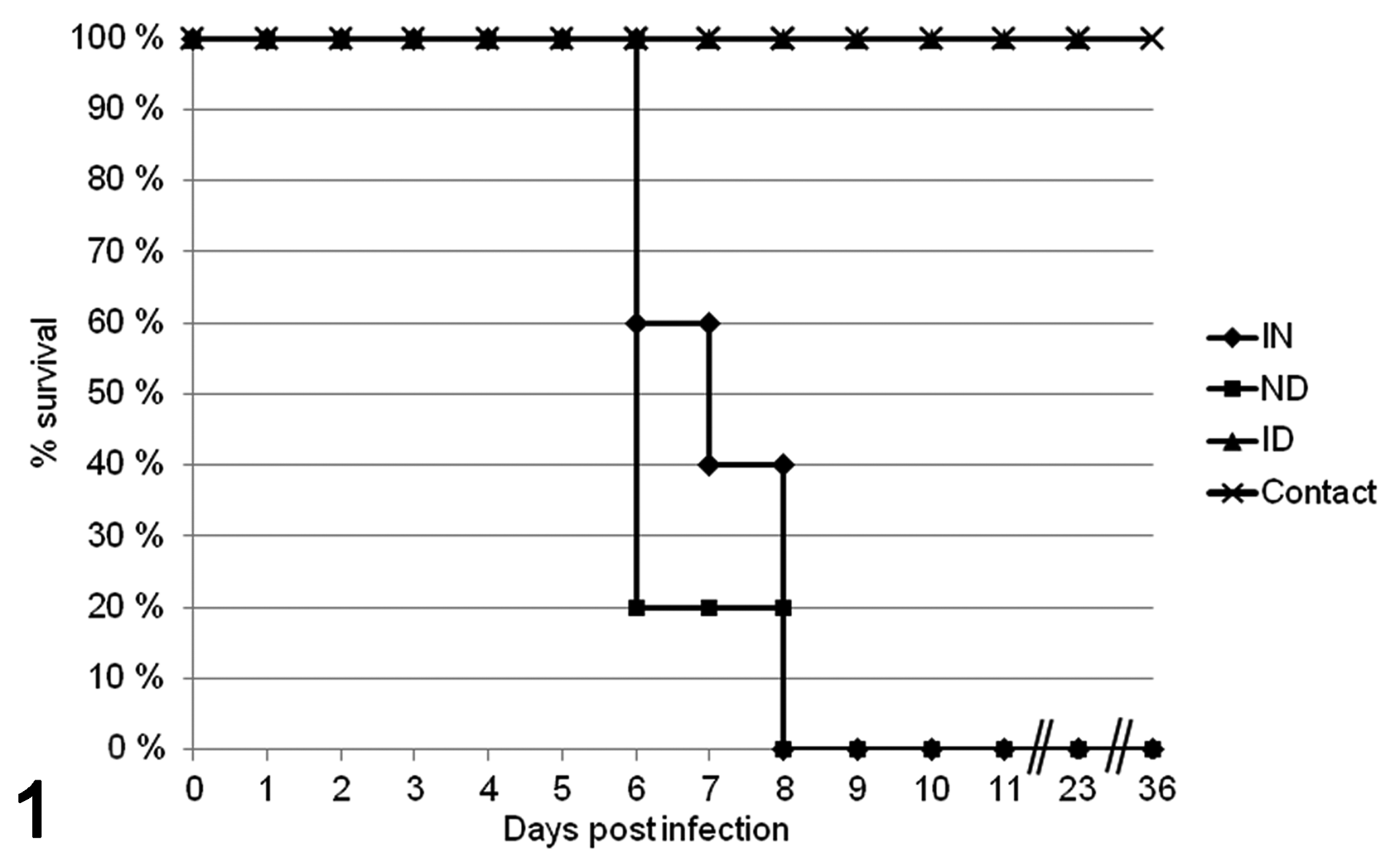
Survival rate of rats after intranasal (IN), intradermal (ID), combined intranasal and intradermal (ND), and contact infection with cowpox virus.
Clinical Observations and Gross Lesions in Rats After Intranasal (IN, 5 Rats), Intradermal (ID, 5 Rats), Combined Intranasal and Intradermal (ND, 5 Rats), and Contact Infection (6 Rats) With Cowpox Virus
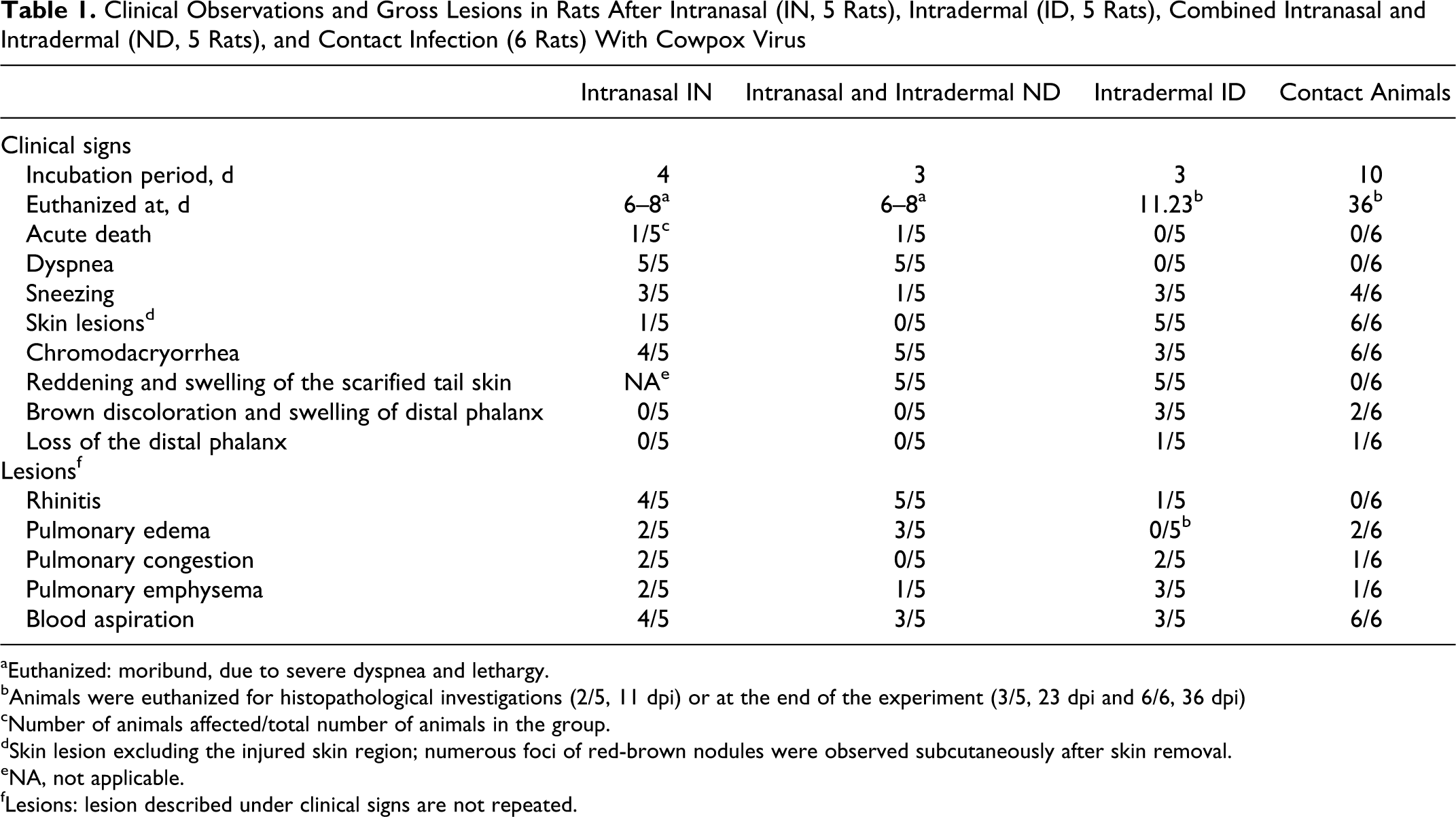
aEuthanized: moribund, due to severe dyspnea and lethargy.
bAnimals were euthanized for histopathological investigations (2/5, 11 dpi) or at the end of the experiment (3/5, 23 dpi and 6/6, 36 dpi)
cNumber of animals affected/total number of animals in the group.
dSkin lesion excluding the injured skin region; numerous foci of red-brown nodules were observed subcutaneously after skin removal.
eNA, not applicable.
fLesions: lesion described under clinical signs are not repeated.
Intranasal infection route (IN)
After an incubation period of 4 days, the infected animals showed slightly dried red-brown discharge (chromodacryorrhea) around the eyes and nose (4/5 rats) or sneezing (3/5). The animals died (1/5) or were euthanized (4/5) after showing severe dyspnea (5/5) and lethargy (4/5) on days 6 to 8 postinfection. At 6 dpi, 1 animal showed a sharply demarcated cutaneous erosion, 2 mm in diameter, at the dorsal aspect of the right hind foot (Fig. 2).
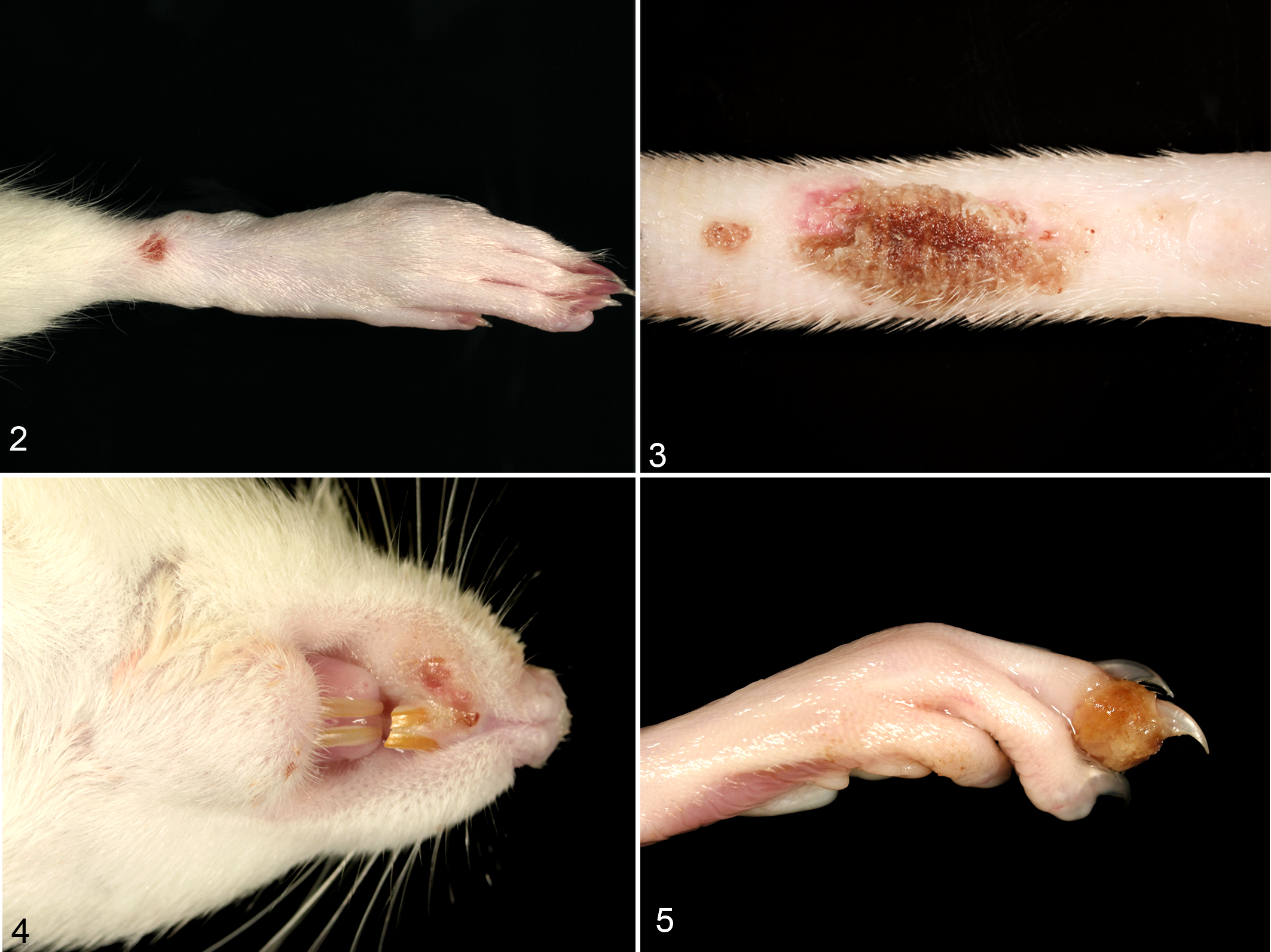
Skin. Rat, 6 days after intranasal cowpox virus (CPXV) infection. Singular pox lesion at the dorsal aspect of the hind limb.
Intradermal infection route (ID)
All rats consistently exhibited reddening and swelling of the skin of the scarified tail on day 3 postinfection (Fig. 3). Multifocal cutaneous proliferative and necrotizing areas, up to 4 mm in diameter, were observed at 10 dpi especially on the limbs, nose, lips, tongue, and inguinal region (Fig. 4, 5/5 rats). For histopathological investigation only, 2 rats were sacrificed at 11 dpi. From 16 dpi onwards, chromodacryorrhea (3/3), sneezing (3/3), and brown discoloration of the swollen distal part of the digits (Fig. 5, 3/3) were found. One animal lost the affected distal phalanx by automutilation. The remaining 3 rats were euthanized at 23 dpi.
Intranasal and intradermal infection route (ND)
In this group, all rats showed reddening and swelling of the injured skin region (3 dpi) and chromodacryorrhea. One animal exhibited sneezing (4 dpi). The rats had to be killed due to severe dyspnea and lethargy 6 dpi (3/5 rats) and 8 dpi (1/5). One animal died 6 dpi after being handled and exhibiting dyspnea.
Contact animals
The 6 sentinel animals consistently developed cutaneous erosion (6/6) after an incubation period of 10 days, and 2 rats had swollen distal phalanges (11 dpi). One animal lost a distal phalanx. Chromodacryorrhea (6/6, starting 13 dpi) and sneezing (4/6, starting 17 dpi) were observed. The skin lesions were completely healed by 36 dpi.
Histopathology
Intranasal infection route (IN)
Within the upper respiratory tract, a focally extensive, proliferative, and necrotizing rhinitis (4/5 rats) was characterized by degenerated and necrotic mucosal epithelium with large intracytoplasmic eosinophilic inclusion Guarnieri bodies. Affected regions were infiltrated by numerous neutrophils, lymphocytes, and macrophages. The nasal cavity was filled with cellular debris and fibrin, and frequently colonies of coccoid or rod-shaped bacteria were present. The pharynx and larynx exhibited a focally extensive, partially proliferative, and partially erosive to ulcerative-necrotizing pharyngitis (laryngitis) (3/5) with the same characteristics as described for the rhinitis (Figs. 6, 7).
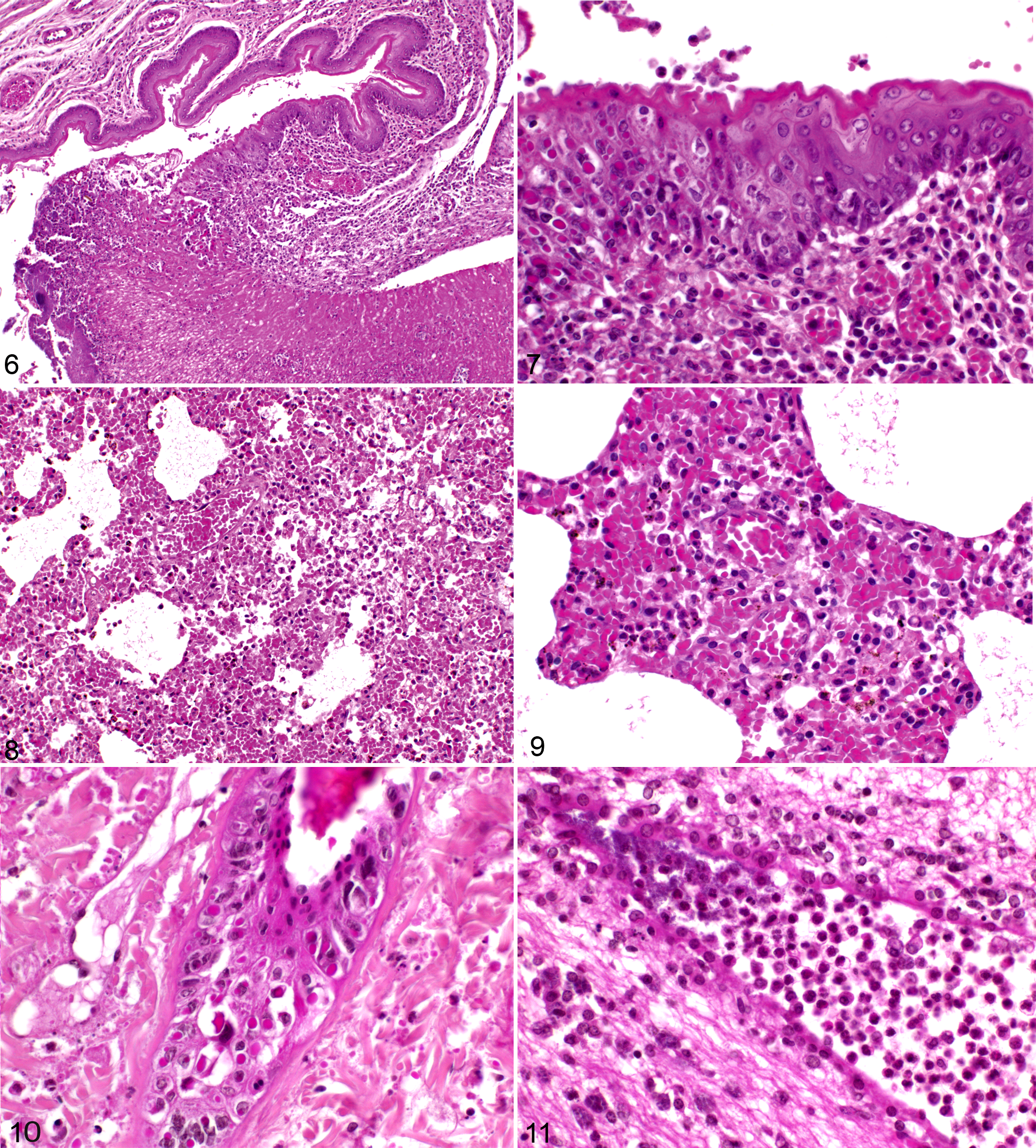
Histopathology of rats, aged 6 weeks, infected with cowpox virus (CPXV) intradermally, intranasally, and by a combination of both routes.
In 1 animal, there was acute bronchointerstitial pneumonia primarily with infiltrates of lymphocytes and fewer neutrophils and histiocytes (Figs. 8, 9). Furthermore, congestion (2/5), alveolar edema (2/5), and emphysema (2/5) were present. Multifocally, alveolar and bronchial lumina (4/5) were filled with red blood cells, which was interpreted as aspiration of blood subsequent to exsanguination.
Within the mandibular lymph nodes, necrosis of lymphocytes admixed with infiltration of viable and degenerate neutrophils was observed in the hilus regions (2/5).
The affected skin showed proliferative and necrotizing dermatitis and folliculitis with epithelial ballooning degeneration and necrosis and parakeratotic hyperkeratosis (1/5). Large numbers of intracytoplasmic, eosinophilic inclusion bodies were found within the surface epithelium and partially in the epithelium of the hair follicles and sebaceous glands (Fig. 10).
Occasionally, moderate to severe suppurative meningoencephalitis (2/5) with multifocal malacia (1/5) and intralesional rod-shaped bacteria and (perivascular) hemorrhage was detected (Fig. 11).
Intradermal infection route (ID)
At 11 days postinfection, a focally extensive, proliferative, or erosive to ulcerative-necrotizing rhinitis (1/5), as already described, was frequently found and the larynx of infected animals was equally affected (1/5). Occasionally, the necrosis of the nasal mucosa was not accompanied by inflammation (1/5).
Animals euthanized at 11 dpi and 23 dpi showed acute bronchointerstitial pneumonia with mixed cellular (lymphocytes and fewer neutrophils and histiocytes) infiltration (3/5). Congestion (2/5), emphysema (3/5), and blood aspiration (3/5) were found.
The most affected cutaneous region was the scarified skin of the tail with proliferative to necrotizing and ulcerative, mixed cellular dermatitis and folliculitis with epithelial ballooning degeneration and necrosis and parakeratotic hyperkeratosis (2/5, euthanized 11 dpi) with rare subepithelial edema (1/5). The skin lesions observed on the limbs, nose, lips, tongue, and inguinal region showed proliferative necrotizing and erosive dermatitis and folliculitis (restricted to haired skin). Intracytoplasmic, eosinophilic inclusion bodies were found within the surface epithelium and partially in the hair follicles and sebaceous glands (2/5). The scarified skin of rats euthanized at 23 dpi showed focal parakeratotic hyperkeratosis (3/5), serocellular crusts, and epithelial ballooning degeneration with intracytoplasmic, eosinophilic inclusion bodies in epithelial cells (2/5).
Intranasal and intradermal infection route (ND)
Rhinitis (5/5) and pharyngitis (4/5) were seen frequently as already described. There was acute bronchointerstitial pneumonia with infiltrates of lymphocytes and fewer neutrophils and histiocytes (5/5). Several animals showed pulmonary edema (3/5), emphysema (1/5), and blood aspiration (3/5).
Within the dermis, restricted to the skin of the tail, a necrotizing, mixed cellular dermatitis and folliculitis was observed as described to the ID infection route (5/5). Suppurative meningoencephalitis (3/5), multiple foci of malacia (1/5), and intralesional rod-shaped bacteria and perivascular hemorrhage (2/5) were present.
Contact animals
In the sentinels, rhinitis, laryngitis, or pneumonia did not occur. Within the lung, pulmonary edema (2/6), congestion (1/6), emphysema (1/6), and blood aspiration (6/6) were found.
Immunohistochemistry
Intranasal infection route (IN)
CPXV antigen was detected in the upper respiratory tract, the skin, and mandibular lymph nodes. The respiratory and olfactory epithelium within the nasal cavity or the squamous epithelium in the apical nasal cavity as well as subjacent mesenchymal cells (fibrocytes, histiocytes, or macrophages) showed marked immunopositivity (4/5 rats). The respiratory and squamous epithelium and mesenchymal cells of the pharynx were also affected (3/5 rats, Figs. 12, 13). Antigen was detected in all layers of the squamous epithelium of the nasal skin (2/5 rats) and was found in mesenchymal cells of the mandibular lymph node (2/5, Fig. 14). CPXV antigen was not detectable in any other tissues investigated.
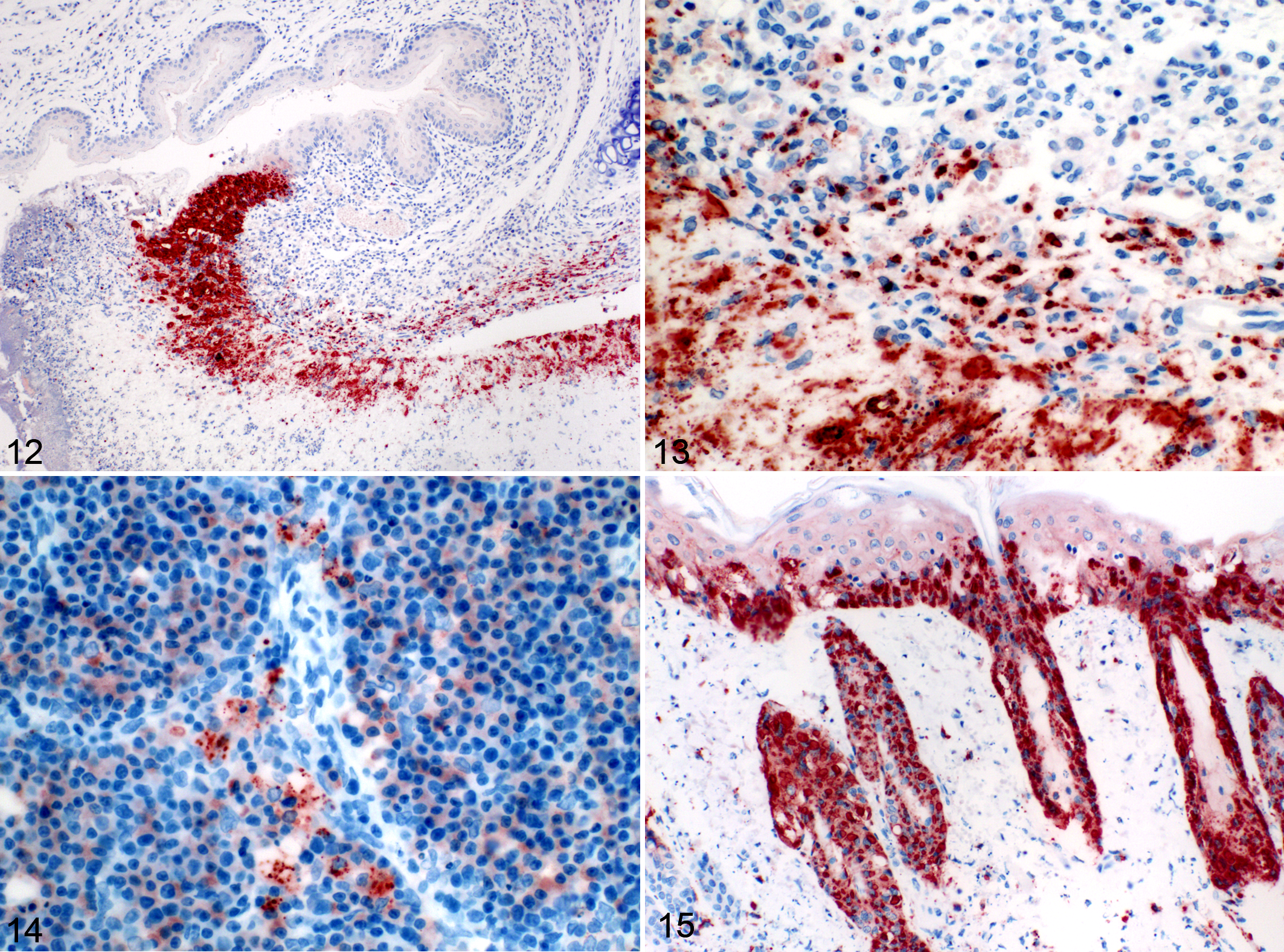
Immunohistochemistry of rats, aged 6 weeks, infected with cowpox virus intradermally, intranasally, or by a combination of both routes. Avidin-biotin-complex (ABC) method, AEC staining (red), hematoxylin counterstain (blue).
Intradermal infection route (ID)
Cowpox virus antigen in rats euthanized 11 dpi was restricted to the upper respiratory tract and skin. Immunolabeling in respiratory and squamous epithelial as well as mesenchymal cells in the nasal cavity (2/5 rats) and the pharynx (1/5 rats) was detected. Within the skin, the squamous epithelium, the dermal sebaceous glandular and hair follicle epithelium, and subjacent mesenchymal cells showed marked immunopositivity (2/5 rats, Fig. 15). In 1 animal euthanized at 23 dpi, CPXV antigen was detected in the hair follicle epithelium. Cowpox virus antigen was not observed in any other tissues.
Intranasal and intradermal infection route (ND)
As described for the ID infection route, the presence of antigen was restricted to the upper respiratory tract and the skin. Immunolabeling was detected in respiratory and squamous epithelium as well as mesenchymal cells in the nasal cavity (4/5 rats) and the pharynx (4/5 rats). The squamous epithelium of the skin, the dermal sebaceous glandular and hair follicle epithelium, and subjacent mesenchymal cells showed marked antigen labeling (3/5 rats). Viral antigen was not detected in any other tissues.
Contact animals
No CPXV antigen was detected at 36 dpi.
Discussion
Our results demonstrate that the course of CPX disease is strongly related to the infection route. Intradermal infection resulted in a dermal form with pustules and crusts predominantly affecting the hairless body areas. Some animals exhibited sneezing, rhinitis, and pneumonia, but dyspnea and death did not occur. Intranasal and combined intranasal/intradermal infection resulted in lethal infection associated with severe dyspnea and rhinitis. Pneumonia was found frequently after combined and rarely after intranasal infection. Cutaneous pox was detectable only sporadically.
The dermal form was observed in contact animals, emphasizing that experimental intradermal infection might be a useful model to mimic natural infection in rats for pathogenetic studies. Remarkably, after natural infection in pet rats, a variety of outcomes, including subclinical infection, skin lesions, respiratory signs, and fatal disease, were observed. 1,6,14,18 This indicates that, for the outcome of the disease, several factors could be relevant. These include the virus strain and dose, the infection route, the rat species, a preexisting illness, or immunodeficiency. Different survival rates of laboratory and pet rats were observed, depending on the dose of CPXV, the application route, and breed, respectively. 12 To date, there are limited data regarding the differences in the pathogenesis of CPXV infection in laboratory and pet rats (fancy rats). Kalthoff et al 12 found that 9 of 9 Wistar rats inoculated with a high dose of CPXV succumbed to the disease and 1 of 10 fancy rats survived.
Different survival rates and excretion patterns between Wistar and fancy rats are probably related to the small number of animals used or may be due to the differences in the age of the animals (4 weeks vs 6 weeks). 12 Even the volume of the virus inoculum has an influence on the pathogenesis, as shown in BALB/c mice with a syncytium-forming variant of cowpox virus: a high-volume infection produced a more rapid lethal disease associated with severe pneumonia, whereas a low-volume infection remained primarily confined to the upper respiratory tract. 20 Factors such as immunosuppression can influence the course of CPXV infection. Immunosuppressive diseases—for example, in cats—caused by concurrent infection with feline immunodeficiency virus (FIV) or feline leukemia virus (FeLV) have also led to fatal complications during cowpox virus infection. 1,2 Remarkably, concurrent infection with feline parvovirus did not lead to a severe course of CPXV infection. 19
The development of cutaneous pox might be initiated by at least 2 routes: (1) infection of the epidermis after external mechanical stress or (2) infection of the epidermis as a sequela of viremia followed by virus replication in mesenchymal dermal cells and spread to basal keratinocytes of the adjacent epidermis. The latter route of infection is supported by experimental data showing that various organs and serum samples (unpublished data) contain CPXV DNA indicating viremia. 12 In addition, the histopathological and immunohistological indications for initial epidermal spread starting from the basal layer further underline this view (Fig. 15). Potentially, the pathogenesis of the dermal form has common features with ectromelia infection of mice (BALB/c). The ectromelia virus replicates in mice, its natural host, in the epidermal and dermal cells. With lymphocytes or macrophages or as free virus, ectromelia virus reaches the regional lymph nodes. Following the primary viremia, the virus is found in internal organs, including spleen and liver. In lymph nodes, spleen, and liver, necrosis can occur. After the secondary viremia, virus is again detected in the skin. 5 Based on our morphological data, CPXV antigen was detected in the epidermal cells and cutaneous mesenchymal cells (fibrocytes, histiocytes, or macrophages) and sporadically within the regional lymph nodes (mandibular lymph node) associated with lymphocyte necrosis. Further internal organs were not affected. Hence, the spread of CPXV might be triggered by macrophages, or CPXV may spread independently as a free virus. Lymph nodes could be important replication sites for CPXV. It is possible that relevant CPXV replication in a broader spectrum of tissues is not present. Additional kinetic studies might clarify if replication in a broader spectrum of organs occurs with an earlier time point.
Intranasal and combined intranasal and dermal inoculation was associated with severe dyspnea, rhinitis, pharyngitis, and pneumonia. Very likely, 2 animals died of suffocation. Possible causes for dyspnea could be pneumonia, alveolar edema, obstructive rhinitis, or pharyngitis. In the current study, the cause for dyspnea remains unclear. Pneumonia, rhinitis, and pharyngitis were confirmed in animals with and without significant dyspnea.
Because intradermally infected animals and sentinel animals did not develop dyspnea, we hypothesize that the outcome of dyspnea might be related to the infection dose or the volume of inoculum. Infected animals excrete virus, and due to personal grooming and close contact to cage mates, a viral uptake via the respiratory tract occurs, but the amount of virus might be insufficient to trigger the respiratory form.
Pneumonia is described as a primary lesion in several orthopoxvirus infection models. In rabbits (rabbitpox virus), macaques (monkeypox virus), and mice (vaccinia virus), necrotizing or fibrinonecrotic bronchopneumonia occurs. 7 In humans, infection with smallpox leads to bronchopneumonia, which is described as a frequent complication of the disease. However, it remains unclear if bronchopneumonia is directly related to the virus infection or due to secondary bacterial infections. 7 Aerosolized and intranasal CPXV infection induces necrotizing fibrinous and hemorrhagic bronchopneumonia in mice (BALB/c). 16 Remarkably, pneumonia has not been described after intracerebral, intravenous, intradermal, cutaneous (scarification), subcutaneous, intrafollicular, or intraperitoneal inoculation in mice. The same is true for guinea pigs, chicks, or rabbits. In the present study, acute bronchointerstitial pneumonia with mixed cellular infiltration was found infrequently and could not be associated with a specific inoculation route (IN, 1/5; ID, 3/5; ND, 5/5). It remains unclear whether these lesions were virus induced or due to secondary bacterial or viral infection. Further studies are needed to investigate the influence of antibiotics and antiviral drugs on the development of lung lesions.
In the present study, multifocal to coalescing red foci in the lungs were present in rats that were euthanized by exsanguination (16/21 animals). Hemorrhage in the lungs has been described after experimental infection. 13 However, the histopathology of the current animal experiments confirmed aspiration of blood subsequent to exsanguination.
Hemorrhagic exudates were described after intranasal and natural infection of rats with CPXV. 13,18 In the current study, chromodacryorrhea (not hemorrhage) was observed. In rodents, the lacrimal fluid contains porphyrins. 10 Overproduction of porphyrins due to infectious diseases or reduced personal grooming results in dark red crust, a condition called chromodacryorrhea. 10,21
The sporadic occurrence of meningoencephalitis in 2 rats of the ID group and 3 rats of the ND group was considered a secondary effect of CPXV infection based on the detection of intralesional rod-shaped bacteria. Further microbiological investigation in 3 of these animals revealed Proteus sp. and Escherichia coli in lung, skin, and nasal mucosa specimens.
By comparison, concurrent bacterial septicemia was found in a cynomolgus monkey after aerosol infection with monkeypox virus. The bacterial infection was associated with increased dissemination of the virus to the liver, spleen, and bone marrow and resulted in a more rapid fatal clinical course. 23 In the present study, the occurrence of meningoencephalitis was not associated with an increased dissemination of the viral antigen or a more rapid clinical course or death. Numerous poxvirus genes are associated with immunomodulatory effects, promoting secondary viral and bacterial infections. 11,17
Viral antigen detection and the occurrence of large intracytoplasmic eosinophilic inclusion bodies were restricted to the upper respiratory tract (nasal cavity, larynx), the affected skin, and sporadically in macrophages of the mandibular lymph node. This appears to be contrary to recent data. After intranasal infection using virus isolation techniques, Maiboroda 13 detected high to moderate amounts of virus in lungs, rectum, liver, kidneys, spleen, and testes. Also, Kalthoff et al 12 showed that animals that had succumbed after intranasal CPXV infection or intranasal infection combined with scarification displayed a high viral load in a broad set of tissues, tested by virus isolation and PCR. Whether their results reflect virus replication in these tissues, viremia, or a combination of both remains unclear without in-depth pathomorphological and immunohistochemical investigation, including electron microscopy. The immunohistochemical detection of CPXV antigen does not prove replication in the affected tissue. The detection of intranuclear inclusion bodies, however, indicates replication because incomplete virions formed in the cytoplasm were coated in the process of migration through the inclusion body membrane in the late phase of replication, as shown by ultrastructural investigation. 8
However, our data indicate that a widespread viral detection using virus isolation or PCR methods might be due to viremia.
The present study demonstrates that the course of CPXV disease after experimental infection is strongly related to the infection route, leading to a mild dermal, a lethal respiratory, or a lethal mixed form. Because natural infections in pet rats are associated with a variety of outcomes, several factors of potential relevance for the clinical course were discussed.
The respiratory form was associated with severe dyspnea. Pneumonia, rhinitis, or pharyngitis could not be determined as a cause for the dyspnea. Whether pneumonia is the consequence of CPXV infection or is due to secondary bacterial infection remains unclear.
The detection of viral antigen and intracytoplasmic eosinophilic inclusion bodies was restricted to the epithelium and subjacent mesenchymal cells of the upper respiratory tract, the affected skin, and sporadically in macrophages of the mandibular lymph node. Macrophages might play a role in the spread of CPXV, and it is likely that CPXV replication in a broader spectrum of tissues did not occur.
Footnotes
Acknowledgements
The authors would like to thank Gabriele Czerwinski, Gerda Busch, and Mareen Grawe for excellent technical assistance and Thomas C. Mettenleiter for critical reading and comments.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
