Abstract
Despite numerous advances in the diagnosis and control of infectious diseases of nonhuman primates in the laboratory setting, a number of infectious agents continue to plague colonies. Some, such as measles virus and Mycobacterium tuberculosis, cause sporadic outbreaks despite well-established biosecurity protocols, whereas others, such as retroperitoneal fibromatosis-associated herpesvirus, have only recently been discovered, often as a result of immunosuppressive experimental manipulation. Owing to the unique social housing requirements of nonhuman primates, importation of foreign-bred animals, and lack of antemortem diagnostic assays for many new diseases, elimination of these agents is often difficult or impractical. Recognition of these diseases is therefore essential because of their confounding effects on experimental data, impact on colony health, and potential for zoonotic transmission. This review summarizes the relevant pathology and pathogenesis of emerging and reemerging infectious diseases of laboratory nonhuman primates.
Emerging and reemerging infectious agents remain a threat to laboratory-reared nonhuman primates (NHPs). Such animals are critical to a number of biomedical research programs and are used extensively in drug discovery and development. In contrast to those of many other laboratory animal species, infectious diseases have proven difficult to eliminate from primate colonies, and various pathogens may affect animal and colony health. Moreover, despite significant advances in the diagnosis of infectious diseases, unrecognized or adventitious agents are common in NHPs and have the potential to confound experimental work. 84 This review focuses on some of the better-characterized emerging and reemerging infectious agents of concern to facilities that utilize NHPs in biomedical research programs. Here we describe their clinical presentations, gross and histologic lesions, and means of diagnosis.
In recent decades, much progress has been made in excluding select pathogens from NHP colonies. All colony management schemes, for example, include protocols for elimination of mycobacteriosis. At least 4 levels of specific pathogen–free status have been defined, depending on which viral agents have been successfully eliminated. Viruses targeted for exclusion include simian retrovirus, simian T-lymphotropic virus, simian immunodeficiency virus (SIV), and Macacine herpesvirus 1 (B virus). 99 Agents such as the former two (simian retrovirus and simian T-lymphotropic virus) are undesirable owing to their immunomodulatory effects, whereas Mycobacterium tuberculosis and B virus also pose a zoonotic threat to humans. In addition, research protocols may call for populations of animals naïve to particular pathogens. Colonies of macaques free from cytomegalovirus and Helicobacter pylori, for example, have been successfully developed. 10,125
Despite these successes, elimination of many other infectious diseases from NHP colonies has proven difficult for several reasons. The standard approach—based on cesarean derivation and isolation used to produce specific pathogen–free populations of many species of laboratory animals—is difficult to reproduce in primates, given that parental input is critical in the normal social development of infants and juveniles. This approach may lead to future poor reproductive performance and behavioral issues. 56,128 Common primate housing and breeding strategies that are used to enhance socialization and welfare may promote transmission of many agents. The continued use of feral and free-ranging source colonies introduces variability in exposure that is difficult to control. In addition, the long life span and high value of many NHP species can restrict options available in the event of infection of animals. Finally, transmission of infectious agents such as M. tuberculosis and measles virus from human handlers poses a constant and continued threat to captive primate populations.
A variety of emerging and reemerging infectious diseases may affect NHP health and research programs (Table 1 ). Concerns arise through resurgence of previously identified agents, identification of truly novel pathogens, and recognition of the potential effect of previously described infectious agents on experimental work. Factors that promote the emergence and reemergence of infectious diseases include the following: importation of primates from sources with poor microbial quality control; mixing of primates from different sources; poor separation of primates from other primate and nonprimate species; experimental induction of immune dysfunction or dysregulation; and inadvertent transmission of adventitious agents during experimental manipulation. 7,84,134 Of these factors, importation of animals from nondomestic sources may introduce and pose the most significant risk.
Emerging and Reemerging Infectious Agents of Nonhuman Primates a
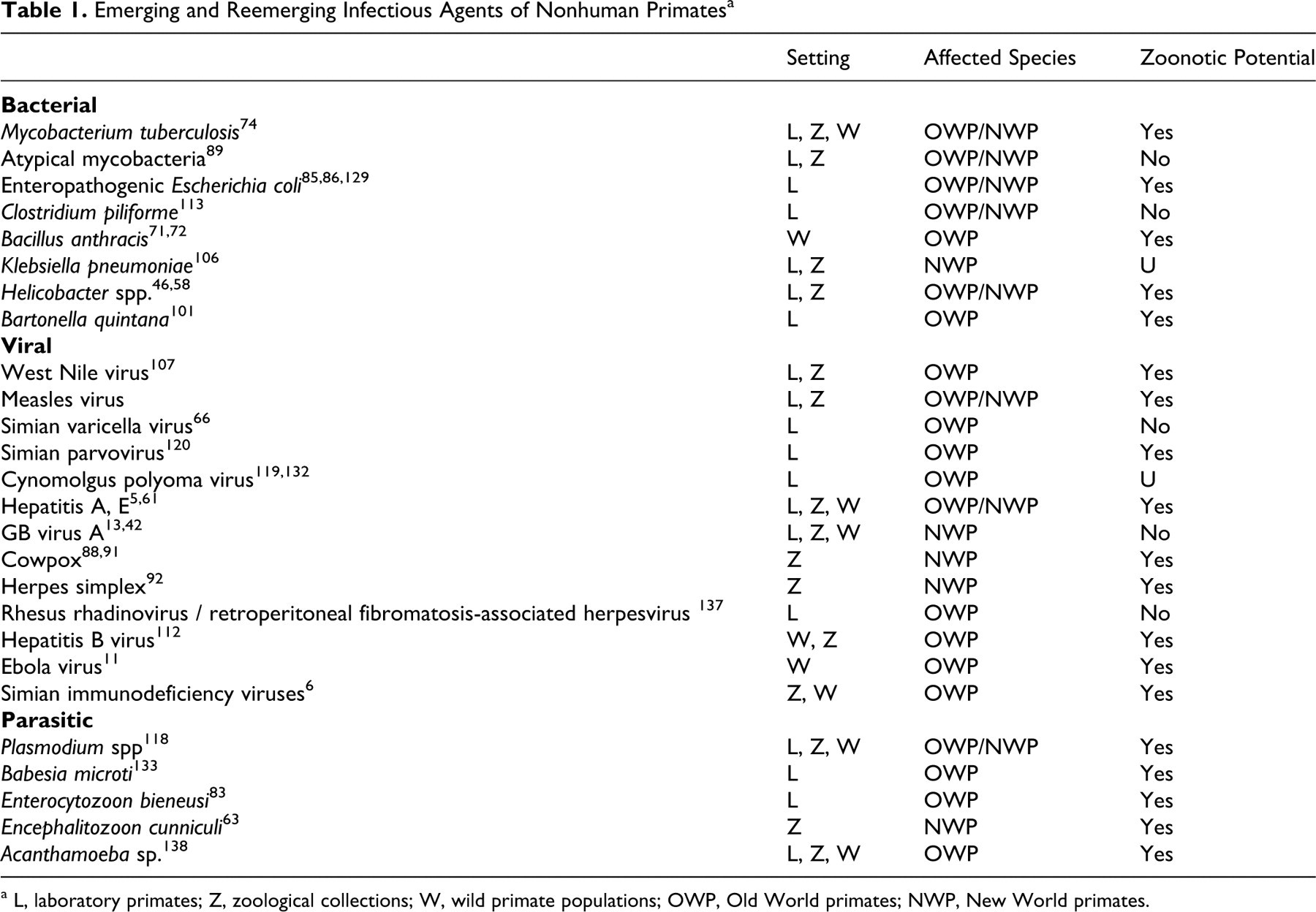
a L, laboratory primates; Z, zoological collections; W, wild primate populations; OWP, Old World primates; NWP, New World primates.
The impact of these infectious agents on individuals may differ per the population under evaluation. The relevant diseases in captive laboratory primates vary on the basis of source (imported versus domestic), housing configuration (single-caged versus corral versus free-ranging), and experimental use. The spectrum of diseases in populations of captive NHPs in zoological collections may be different owing to exposure to agents and other species not typically found in a research setting. Finally, wild NHPs experience different stressors and exposures than do captive animals. The burden from environmental stressors and human contact may be more profound than that observed in captive populations, and evidence suggests that epizootics resulting from cross-species transmission of agents may have a devastating effect on wild populations. Recent descriptions of Ebola virus and anthrax in great apes suggest a significant adverse impact on conservation efforts. 11,71,72
The result of infection with agents may have different outcomes or consequences based on host and environmental factors. Agents may be uniformly pathogenic in the normal host-producing disease and pathology that may be detrimental to the animal. This is often the case with cross-species transmission of pathogens to inadvertent hosts, such as the transmission of herpesvirus simplex from humans to Cebidae. Agents may produce variable pathogenic outcome in the normal host depending on genetic and environmental factors. Thus, simian retrovirus may be controlled by the host’s immune response or lead to progressive immunodeficiency. Often, agents may result in subclinical infection with few, if any, clinical consequences to the infected animal. Finally, pathogenic outcomes may be observed in the face of immunosuppression or immunodeficiency with such opportunistic infections as lymphocryptovirus and cytomegalovirus. 20,137 This last category can be problematic in experimental studies involving immune modulation and so result in the reactivation of previously unrecognized latent infection and overt pathology. 134
Morphologic alterations often provide essential information in the differential diagnosis of infectious diseases in NHP species. This review summarizes current information on relevant emerging and reemerging viral, bacterial, and parasitic agents of NHPs used in biomedical research.
Viruses
Measles Virus
Etiology
Measles virus is an enveloped, negative-sense, single-stranded RNA virus of genus Morbillivirus, family Paramyxoviridae. As a morbillivirus, measles virus is closely related to the etiologic agents of canine distemper, rinderpest, and peste des petits ruminants. Though often considered a historical disease of captive primates and one largely controlled in domestic colonies, in recent years measles virus has repeatedly been imported from Southeast Asia, and it poses a significant threat to naïve colonies and human handlers.
A variety of strains are used in experimental models of infection. These strains are classified as attenuated or nonattenuated based on laboratory culture conditions that influence their receptor tropism and in vivo virulence. In general, strains isolated and passaged in primary human peripheral blood mononuclear cells or lymphoblastoid cell lines, such as B95-8 or B95-a, remain virulent, whereas those passaged in human or monkey kidney cell lines, HK or Vero, do not.
Epizoology
Remarkable progress has been made toward eradication of measles from human populations worldwide through comprehensive vaccination programs. Globally, the number of reported human fatalities has fallen more than 60% in the past decade. Unfortunately, Indochina and the Indopacific region, from whence most macaques are imported, maintain relatively high incidence of the disease. 22 Even nations with well-established vaccination programs maintain a persistent low incidence of measles owing to interference of maternal antibodies with effective vaccination of children. Thus, importation of infected animals remains a concern and may pose a significant threat to domestic colonies.
Although most, if not all, NHP species are susceptible to infection, humans are the only natural host of measles. Seropositive animals of many species, including macaques, chimpanzees, spider monkeys, and owl monkeys, have been identified during import quarantine. 79,100 One survey of large numbers of macaques imported from India revealed an absence of exposure at the site of export, but seroprevalence reached 100% by only 4 weeks after import into the United States. 98 Seroconversion to measles has been identified in wild populations of macaques, but it appears to depend on intimate contact with local, presumably unvaccinated, human populations. 62
The virus itself is highly contagious and, like other Paramyxoviridae, measles is spread through contact, fomites, and aerosols. In experimentally inoculated animals, infected cell numbers from bronchioalveolar lavage fluid peak at 6 days postinfection, before or around the time of the eruption of the characteristic skin rash, suggesting that infected animals are capable of aerosol transmission before the onset of suggestive clinical signs. 100 In a colony setting, the disease often presents as an epizootic. Reported mortality rates among captive NHPs are typically around 25% but vary widely, depending on species and concurrent disease processes. 26,75,79
Clinical disease
The presentation of measles in many Old World NHPs is similar to that described in humans. Signs develop at about 1 week postinfection and consist of conjunctivitis, a maculopapular rash (particularly evident on the face and sparsely haired skin of the ventrum), and minute pathognomonic white eruptions in the oral cavity (known as Koplik’s spots). As disease progresses, infection of cells in the lung and gastrointestinal tract results in bronchopneumonia and diarrhea. Like other morbillivirus infections, measles virus is immunosuppressive, and the clinical picture may be compounded by opportunistic agents such as Shigella, Campylobacter, Candida, cytomegalovirus, and adenovirus. 26
Many cases in NHPs, however, run an atypical or even entirely subclinical course. 57 Signs are variable and heavily influenced by host species and virus strain. Rash and conjunctivitis are more consistent and pronounced, for instance, in rhesus and Japanese macaques than in cynomolgus macaques. 26,40 In an experimental infection of rhesus macaques with 6 different wild-type and culture-attenuated viral strains, only one wild-type strain (Bilthoven) produced clinical disease. 8 In another study, telemetric measurements of body temperature revealed only a slight elevation (about 0.5°C) eclipsed by the animals' normal circadian variance of about 2°C, making fever an unreliable indicator of infection. 40
New World primates may lack many of the hallmark signs common to humans and other Old World primates. In most cases, they fail to develop conjunctivitis or cutaneous lesions, and death occurs as a result of immune suppression and enterocolitis. Encephalitis is observed with intracranial inoculation but not with peripheral inoculation. 2 Viral strain differences also play a role. Infection of moustached tamarins with the attenuated Edmonston strain of measles virus, which fails to produce clinical disease in rhesus macaques, has resulted in a 25% mortality in the tamarins. Another Vero cell–attenuated strain, JM, has proven to be 100% fatal. 3 Observations of a colony of owl monkeys demonstrated seroconversion of a few cohoused animals but no signs of clinical disease. 100
Pathogenesis
Measles virus is believed to gain access to the body through tonsils or the respiratory tree. Two of its receptors have been well characterized: CD46, an inhibitory complement receptor, and CD150, the signaling lymphocyte activation molecule. The latter is leukocyte restricted whereas the former is more broadly expressed. Receptor tropism varies among virus strains and is heavily influenced by the culture condition in which they are maintained. Isolates from clinical cases raised in primary human peripheral blood mononuclear cells retain affinity for both receptors, whereas those raised in lymphoblastoid cell lines lose tropism for CD46. 81 Other receptors exist that permit low-efficiency infection of CD46 and CD150−cells, including a yet-uncloned epithelial receptor. Measles was traditionally thought to first infect respiratory epithelial cells through the apical cytoplasm and then, after local replication, leukocytes and so gain access to the circulation. Infection of rhesus macaques with an epithelial receptor–binding incompetent strain of measles virus, however, has demonstrated an abililty to recapitulate systemic disease but not result in infection of respiratory epithelium, suggesting that the port of entry may be CD150+ dendritic cells or other leukocytes. 37,73
Circulating virus is primarily cell associated, with plasma virus burden one to two orders of magnitude less than cell-associated burden. 143 In blood and lymphoid tissue, CD150+ B lymphocytes and both CD4+ and CD8+ T lymphocytes—but not monocytes, natural killer cells, or neutrophils—harbor infection. Infected cells in peripheral tissues are morphologically consistent with lymphocytes and dendritic cells. Epithelial cells of the lung and trachea are infected, whereas those of the gastrointestinal tract are not. 37
Both humoral and cell-mediated immunity play roles in controlling infection. In naïve animals, immunoglobulin M responses occur by day 7, peak by 2 weeks, and subsequently decline. Immunoglobulin G and neutralizing antibody titers appear at 2 weeks, peak at 3, and remain elevated for months or longer. Cytotoxic T lymphocyte responses arise as late as 4 weeks postinfection. 143 Although vaccines designed to elicit neutralizing antibody titers are effective at preventing infection, experimental depletion of CD8+ and CD20+ lymphocytes has revealed that cell-mediated immunity is critical for control of disease. Viral kinetics and disease progression of CD20-depleted monkeys are generally indistinguishable from those of controls. Monkeys depleted of CD8+ or both CD8+ and CD20+ cells, however, have higher viral loads, longer courses of disease, and more extensive and severe cutaneous lesions. 105
The mechanisms of immune suppression are complex and still under active investigation. Depletion of lymphocytes, thymic dysinvolution, aberrant cytokine responses, and suppression of peripheral lymphoproliferation all play roles. 114 Concurrent measles virus infection may impair immune responses to other agents and interfere with routine diagnostic assays. Of principal concern is interference with intradermal skin testing for M. tuberculosis.
Pathology
Gross and histologic lesions in Old World primates resemble those of humans. The rash begins as erythematous and maculopapular and develops crusts as the lesion matures. Histologically, infiltrates of mononuclear cells concentrate around hair follicles. Immunohistochemical methods reveal virus within keratinocytes and cells morphologically consistent with dendritic cells. 37 The epidermis shows focal parakeratotic hyperkeratosis and may contain syncytial epithelial cells with as many as 20 nuclei (Fig. 1 ). 57
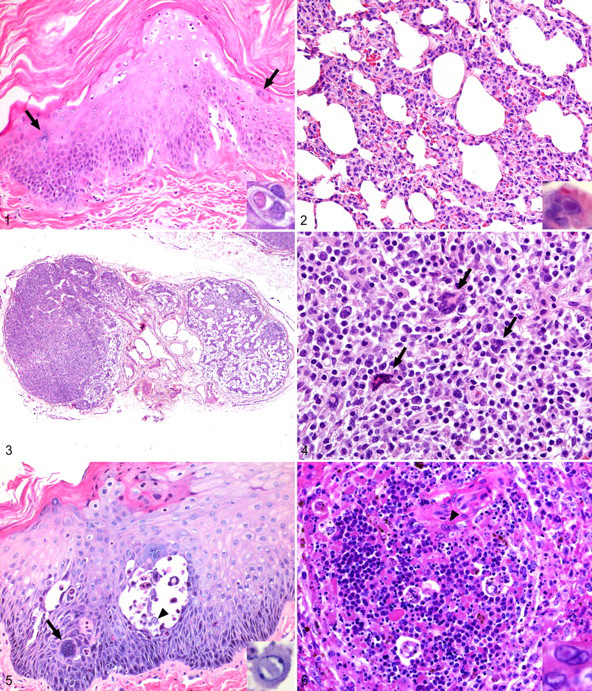
Pulmonary lesions consist of bronchopneumonia with mononuclear interstitial infiltrates and type II pneumocyte hyperplasia. Alveoli contain mononuclear cells, neutrophils, and occasional multinucleated giant cells (Fig. 2). Syncytial epithelial cells bearing eosinophilic intracytoplasmic or less frequently intranuclear inclusion bodies are the hallmark of the disease but may not be present in all cases. The lesion may be complicated by secondary bacterial infection resulting from epithelial damage and immune suppression. 26,57
Epithelial syncytia occur in numerous other organs, including the salivary gland, pancreatic duct, thyroid, and liver. 57 Lymphoid necrosis and depletion may be seen in any of the secondary lymphoid organs and may be marked (Fig. 3). Large antigen-positive multinucleated syncytial cells termed Warthin-Finkeldey cells may be observed (Fig. 4). Lymphoid changes may persist for prolonged periods following resolution of cutaneous and pulmonary pathology interfering with interpretation of experimental results.
Occasionally, cuffs of mononuclear inflammatory cells form around cerebral vessels. Viral antigen may be demonstrated in nearby neurons by immunohistochemical staining. Experimental intracerebral inoculation may cause encephalitis that shares some features with the subacute, sclerosing panencephalitis lesions seen in humans. 1
Lesions in New World animals are subtler. Gross lesions are restricted to splenomegaly and lymphadenomegaly. 3,75 Rash and conjunctivitis do not occur. Histologically, the major lesions consist of gastritis and colitis with occasional inclusion-bearing epithelial syncytial cells and rare foci of necrosis in the kidney, liver, pancreas, and salivary gland. Lymphoid organs are characterized by lymphoid depletion as in macaques. The characteristic pneumonitis, with or without epithelial syncytial cell formation, was reported as a common finding in a colony of naturally infected common marmosets, as well as cotton-top and saddle-back tamarins. Syncytia may also be found in bronchiolar epithelium, the liver, and pancreatic ducts. 3
A high clinical index of suspicion should be maintained for measles virus infection in animals recently imported from Southeast Asia. Such animals may show typical cutaneous and respiratory pathology as described above, but these lesions may have resolved with only subclinical lymphoid changes remaining. Morphologic features in tissue are characteristic, and diagnosis may be made by demonstrating viral antigen in tissue sections by immunohistochemistry. Seroconversion may assist in the diagnosis of acute cases. Presence of antibodies indicates previous exposure to natural infection or vaccine strains.
Simian Varicella Virus
Etiology
Simian varicella virus (Cercopithecine herpesvirus 9) is a double-stranded DNA virus of genus Varicellovirus in the Alphaherpesvirinae subfamily of the Herpesviridae. Several strains, named for institutions in which outbreaks occurred, are recognized, including Delta herpesvirus, Liverpool vervet virus, and Medical Lake virus. 13,27,94 Most recent publications of experimental disease that refer to simian varicella virus employ a passaged strain of Delta herpesvirus. The genome of Delta herpesvirus has been sequenced and shows very high homology to varicella zoster virus, the causative agent of human varicella (chickenpox) and zoster (shingles). 53 Like measles, simian varicella is largely well controlled in modern colony settings, but outbreaks occur on occasion. The virus is lymphotropic, and infection serves as a confounder to immunological studies. Though often considered a historical disease, simian varicella virus has been recognized with increasing frequency during the course of experimental immunomodulation and should remain high on the differential diagnosis of febrile animals with exanthema.
Epizoology
Natural and experimental infections have demonstrated the susceptibility of African and Asian Old World primates, including patas monkeys, vervets, and pigtail, Japanese, cynomolgus, and rhesus macaques. 43,109,115 Epidemiologic studies of simian varicella viruses in the wild are lacking, and the natural host for simian varicella viruses is unknown. The virus is shed in respiratory secretions, and animals are contagious before the onset of clinical signs. 51 In colony settings, the disease often manifests as an epidemic, with mortality rates reaching 50 to 60%. 124 Epidemics are often linked to recent imports but in some cases result from reactivation of latent infection. Recovery typically results in long-lasting immunity, but immunosuppression may trigger recrudescence and clinical disease. 52
Clinical disease
The course of disease may range from inapparent infection without detectable viremia to systemic disease and death. Clinical signs appear 1 to 2 weeks after exposure and may be rapidly progressive, with death ensuing within 24 hours. Nonspecific signs of anorexia and lethargy are common. 52 The earliest sign is that of exanthematous dermatitis, which is particularly evident on glabrous skin but typically spares the palms and soles. 14 This distribution differs from the viral exanthema caused by orthopoxviruses in which lesions first appear on the extremities and progress centrally. Lesions begin as papules and progress to vesicles that may become hemorrhagic, rupture, and encrust over the course of 3 to 4 days. 109,115 The vesicles are of uniform size, typically < 5 mm in diameter, and may coalesce over the ventrum. Vesication of oral and esophageal mucosae is less common but has been reported. 14,109
Simian varicella virus is hepatotropic, and elevation of serum alanine aminotransferase and aspartate aminotransferase may be detected as early as day 3 postinfection. 52 Other reported clinicopathologic abnormalities include a neutrophilic leukocytosis, thrombocytopenia, and azotemia. 39 Clinical signs of bronchiolitis and pneumonia may occur. 136 Death may occur within 2 days following the appearance of a rash.
Pathogenesis
Following an initial round of replication in respiratory epithelium, simian varicella virus disseminates via infected B and T lymphocytes. 140 In acute infection, the virus exhibits broad cellular tropism. Viral antigen is abundant in the liver, lung, lymph nodes, adrenal cortex, bone marrow, and trigeminal and dorsal root ganglia, but infection spares the brain and spinal cord. 39 The onset of exanthema roughly coincides with the appearance of a specific humoral immune response. 52 Viral genome remains detectable in circulating lymphocytes up to a year postinfection and possibly later, although coculture of these lymphocytes with susceptible Vero cells fails to recover infectious virus. 140 The virus remains latent in trigeminal and dorsal root ganglia and may establish latency even in the absence of detectable viremia. 80
Pathology
Histologically, cutaneous lesions begin as foci of degeneration within the epidermis. These lesions vesicate and fill with acantholytic keratinocytes and interstitial fluid. Vesicles form at the stratum basale or low within the spinous layer. 14 Vesicles may become hemorrhagic before rupturing owing to infection and necrosis within the underlying dermis. Syncytia form among the keratinocytes, and many keratinocytes contain intranuclear inclusions (Fig. 5). 136 The underlying dermis is edematous and contains areas of necrosis and infiltrates of mononuclear inflammatory cells, some of which contain intranuclear inclusions. 14
Grossly, the lung contains multifocal to coalescent foci of hemorrhage and edema. 109 Histologically, lesions consist of bronchiolitis and necrotizing alveolitis with hemorrhage and exudation of fibrin. Intranuclear inclusions may be seen in respiratory epithelium.
The liver ranges from normal to friable with coalescent yellow-gray foci of necrosis. Infection results in multifocal, random necrosis with intranuclear inclusion bodies in hepatocytes. Inflammation is minimal and primarily neutrophilic. 14,27 Spleen and lymph nodes are characterized by multifocal necrosis, and mononuclear cells in affected areas bear characteristic intranuclear inclusions (Fig. 6). Acutely infected ganglia are infiltrated by lymphocytes, but neuronal cell bodies are not necrotic, and inclusions are seen only in nonneuronal cells. 39
The morphologic features of simian varicella virus infection in tissue, necrosis (vesiculation), syncytia formation, and intranuclear inclusions are characteristic but not specific. The major differential diagnoses are disseminated infections caused by members of the Simplexvirus genus of the Alphaherpesvirinae, such as herpes simplex virus and B virus. Definitive diagnosis should be based on immunologic or molecular evidence. Immunohistochemistry techniques have been developed for demonstrating B virus and herpes simplex virus in fixed tissue specimens. 121 Diagnosis of any of these viruses can be accomplished by way of viral isolation from lesions or detection of viral nucleic acid. Diagnosis of recent infection can be made by demonstrating seroconversion. Animals with preexisting antibodies are infected for life and may pose a risk for reactivation and transmission to contacts.
Rhesus Rhadinovirus
Etiology
Rhesus rhadinovirus (RRV; Macacine herpesvirus 5) is a recently recognized virus belonging to the genus Rhadinovirus of subfamily Gammaherpesvirinae of the Herpesviridae family. RRV is part of the RV2 subgroup of the Rhadinovirus genus. Infection is usually clinically silent, but disease manifests under conditions of virally induced immune suppression. Related viruses have been recognized in Macaca nemestrina and M. fascicularis.
Epizoology
Several rhadinoviruses of New World species have been described, but RRV and the related retroperitoneal fibromatosis-associated herpesvirus (RFHV; discussed below) have been identified in macaques. RRV DNA is readily detectable in saliva; the virus is readily transmitted 139 ; and seroprevalence surveys of infected colonies reveal a greater-than-90% rate of seroconversion. 36 A study of 90 macaques based on real-time polymerase chain reaction (PCR) and conducted at the California National Primate Research Center identified RRV DNA in blood or saliva of 81% of animals tested. 139
Clinical disease
Experimental infection with RRV alone results in fever beginning 2 days postinoculation and persisting as long as 2 weeks. Lymphadenopathy develops 2 weeks postinfection and resolves within 3 months. Circulating B cell counts may be increased up to two times normal. Coinfection with SIV exacerbates these signs, resulting in persistent massive lymphadenopathy and splenomegaly. 87,141
Pathogenesis
Genomic analysis has revealed that RRV encodes analogs of several immunomodulatory genes, including IL-6, macrophage inflammatory protein, and interferon regulatory factors, which may explain its propensity to drive lymphoproliferation. 117 In experimental coinfection of RRV and SIV, RRV has been shown to attenuate SIV antibody responses, but its potential in modulating the immune response to other pathogens remains unknown. 87
Pathology
RRV-induced lymphadenopathy begins as follicular and parafollicular hyperplasia. Follicular hyperplasia is severe, and lymphocytes may often be arranged in characteristic concentric circles around follicles. The parafollicular region is expanded by a mixture of immunoblastic, mitotically active lymphocytes and plasma cells. Numerous small blood vessels lined by plump hypertrophic epithelial cells infiltrate the paracortex. As lymphadenopathy resolves, the walls of these vessels become hyalinized. 87,141
Diagnosis
A number of options are available for the diagnosis of RRV infection, including serology and viral isolation. Because of high prevalence rates, detection of the RRV in individual animals or tissues cannot be equated to causality, and additional work is required to define disease associations.
Retroperitoneal Fibromatosis-Associated Herpesvirus
Etiology
RFHV is a rhadinovirus closely related to RRV but classified under the RV1 subgroup. Table 2 summarizes differences between RRV and RFHV. RFHV is the simian analogue Kaposi sarcoma herpes virus (Human herpes virus 8), the cause of Kaposi sarcoma in human AIDS patients. Like RRV infection, patent RFHV infection usually becomes apparent following immune suppression.
Comparison of Retroperitoneal Fibromatosis-Associated Herpesvirus and Rhesus Rhadinovirus
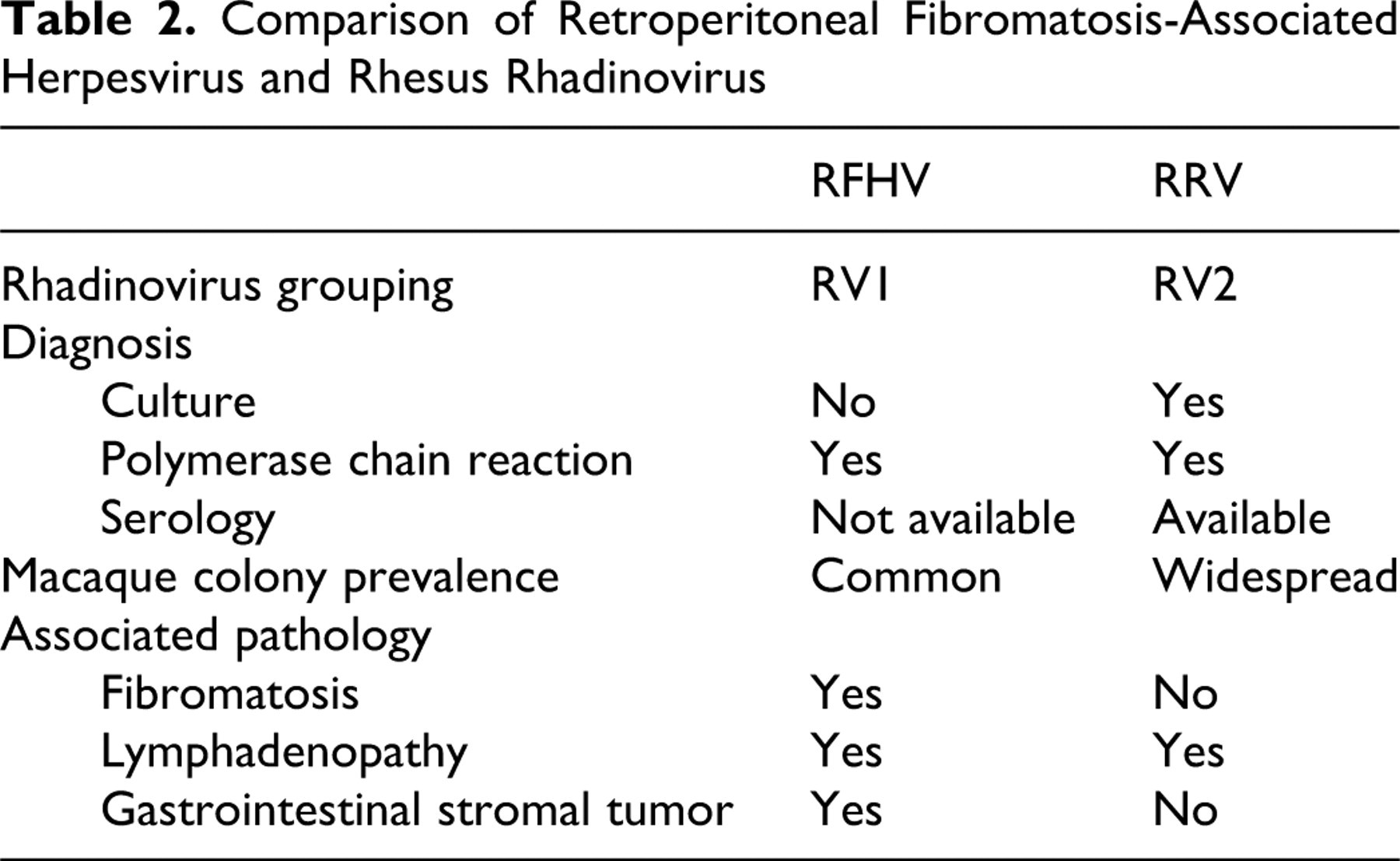
Epizoology
A convenient serologic assay for detection of RFHV is lacking. The prevalence of this agent probably varies from colony to colony, but at least one study screening animals for viremia using PCR found 5 out of 6 animals positive. 15 Like RRV, RFHV can be isolated from saliva, albeit at lower levels. The California National Primate Research Center study revealed only a 44% prevalence for RFHV as compared with 81% for RRV. Interestingly, a significant rate of coinfection with both RRV and RFHV was observed in this cohort, and all RFHV-infected animals were infected with RRV. 139 The incidence of RFHV-associated lesions has sharply declined in recent years, but this may be because of the elimination of simian retrovirus-2 (an immunosuppressive comorbidity often associated with manifestation of RFHV pathology) from colonies of macaques. 137
Clinical disease
RFHV pathology manifests under conditions of immune suppression. Lesions are most commonly reported in animals infected with simian retrovirus-2 but may manifest in monkeys infected with SIV. The classical lesion of RFHV is retroperitoneal fibromatosis for which the virus is named. Retroperitoneal fibromatosis is a locally invasive mesenchymal proliferative disorder that typically arises from the mesentery, often in the vicinity of the ileocecal junction. Clinical signs are nonspecific gastrointestinal disturbances that may be masked by other opportunistic infections in the face of immunosuppression. RFHV infection also results in the related lesion of subcutaneous fibromatosis and has been observed in association with gastrointestinal stromal cell tumors in SIV-infected macaques. 12
Pathogenesis
The mechanisms of RFHV pathogenesis are largely unknown, but like RRV, the viral genome also contains an analog of IL-6. 110
Pathology
Grossly, localized retroperitoneal fibromatosis is characterized by one or more nodules of firm fibrous tissue, typically arising from the retroperitoneum above the ileocecal mesentery. In the progressive form, the neoplasm may extend throughout the body cavity and mesentery to surround the intestines. Invasion of the diaphragm may allow the mass access to the pleural cavity
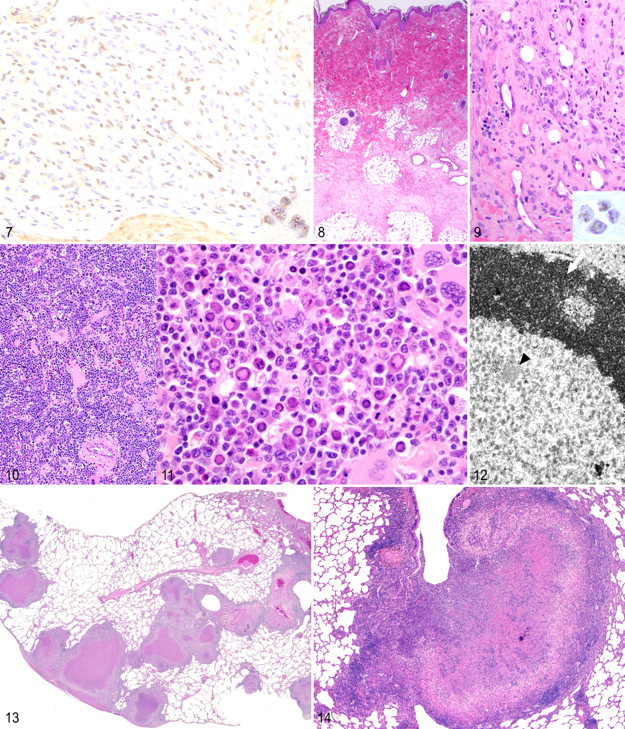
Histologically, retroperitoneal and subcutaneous fibromatosis are somewhat similar to Kaposi sarcoma (Fig. 9). The tumors consist of spindle cells arranged in either “proliferative” or “sclerotic” patterns. Proliferative areas comprise plump spindle cells set within a haphazard extracellular matrix. These areas are richly vascular and often infiltrated by lymphocytes and plasma cells. In contrast, the sclerotic form consists of more mature spindle cells separated by greater amounts of matrix material with less vascularization and inflammation (Fig. 8). The neoplasm does not invade epithelial basement membranes. 49 Immunohistochemically, cells are positive for vimentin and c-kit, a phenotype shared with gastrointestinal stromal cell tumors. 12 Labeling of retroperitoneal or subcutaneous fibromatosis lesions with antibodies directed against the latency-associated nuclear antigen (LANA) of Human herpes virus 8 reveals characteristic punctate nuclear staining (Fig. 9). 18
RFHV-associated gastrointestinal stromal cell tumors are unencapsulated but well-demarcated masses arising from the intestinal wall. They are composed of densely cellular bundles of plump spindle cells sometimes separated by mature collagen bundles. Unlike typical GISTs, some tumors are infiltrated by eosinophils, lymphocytes, macrophages, and lesser numbers of neutrophils and plasma cells. Nearby submucosal vessels may have thick, proliferative intimae. Immunohistochemical staining localizes RFHV LANA to the nuclei of the spindle cells (Fig. 9 ). 12
Diagnosis
Diagnosis of RFHV is difficult because the virus has not been cultured and serologic assays have not been developed. PCR may be used to detect viral nucleic acid in tissues and peripheral blood mononuclear cells, but its sensitivity is unknown for detection of low-level or latent infection. A monoclonal anti–Human herpes virus 8 LANA antibody cross-reacts with RFHV LANA but not RRV and has utility in viral localization by immunohistochemistry. 12
Simian Parvoviruses
Etiology
Simian parvoviruses are recently recognized nonenveloped single-stranded DNA viruses of the Erythrovirus genus of the Parvoviridae family (note that an individual virion may contain either a plus or minus sense strand). The human parvovirus, B19, is their closest relative. At least 3 species have been identified, including the original simian parvovirus from cynomolgus macaques, pigtail parvovirus from pigtail macaques, and rhesus parvovirus from rhesus macaques. 45,55,102 A survey of human handlers of NHPs revealed a significant seroconversion rate to simian parvovirus, indicating potential for zoonosis. 17 Like RRV and RFHV infections, simian parvovirus infections typically go undetected unless the host is immunosuppressed. Infections have taken on increasing importance in animals undergoing solid organ transplantation drug regimes.
Epizoology
Transmission is thought to occur naturally via respiratory secretions and fomites, but it may also occur through transplantation of infected tissues. Seroprevalence estimates of captive animals range from 35 to 50%. 16
Clinical disease
Clinical signs may be subtle or absent in healthy animals. 103 In immunosuppressed animals, disease is more severe. The most consistent clinical finding is a normocytic, normochromic, nonregenerative anemia, but the clinical picture may be compounded by other effects of immunosuppression. 102
Pathogenesis
Viremia is detectable as early as 2 days postinoculation and typically resolves by 2 weeks. Viremia coincides with a period of reticulocytopenia, followed by a rebound reticulocytosis as the virus infects erythroid progenitors and is subsequently cleared. 103
Pathology
Bone marrow lesions consist of erythroid and myeloid dysmaturation with megakaryocytic hyperplasia. Infection results in bizarre erythroid precursor cells characterized by nuclear lobulation and blebbing. Many cells contain large, poorly defined, and eosinophilic intranuclear inclusion bodies (Figs. 10 , 11). 102 Ultrastructural analysis of the inclusions reveals scattered 24-nm particles occasionally arranged into small arrays (Fig. 12 ). 102
Diagnosis
In fulminant cases, the morphologic features in bone marrow are diagnostic and can be confirmed by ultrastructural examination or in situ hybridization. Antemortem diagnosis and detection of subclinical infection is more problematic and must rely on detection of antibodies in serum and viral nucleic acid in whole blood.
Bacteria
M. tuberculosis
Etiology
Mycobacteria are a group of aerobic, nonmotile, rod-shaped, acid-fast bacilli. Although structurally “gram positive,” mycobactetria contain lipids within their cell walls that interfere with uptake of Gram stain reagents. Although opportunistic infection with “atypical” species such as Mycobacterium avium and Mycobacterium intracellulare are frequently diagnosed in immunocompromised animals, the species of greatest concern are M. tuberculosis and Mycobacterium bovis. These agents present a perennial problem in the laboratory setting owing to their communicability, zoonotic potential, and ability to establish latent infection. Though largely controlled in domestically bred animals, M. tuberculosis remains a significant threat in animals imported from regions of the world with high rates of human infection.
Epizoology
An extensive survey of nearly 23,000 imported NHPs, including African green monkeys, rhesus macaques, and cynomolgus macaques, revealed an overall prevalence of 0.4%. 23 Nearly all infected shipments contained cynomolgus macaques that were often asymptomatic and sometimes negative on tuberculin skin tests. 23 Such asymptomatic and test-negative animals have the potential to precipitate epizootics once integrated into a research colony. The agent readily spreads through aerosol, and a single animal can easily infect an entire room, even if animals are individually housed. In one report, a rhesus macaque that had completed a 6-month quarantine, including 17 negative tuberculin skin tests and 2 unremarkable thoracic radiographs, was later euthanized owing to clinical signs of pneumonia. Necropsy revealed pulmonary, hepatic, and splenic granulomas, whereas culture yielded M. bovis. Over the next 2 months, 43 of 45 cohoused animals converted from tuberculin negative to positive and were euthanized. 144 Many similar scenarios have been reported and so underscore the difficulties inherent in screening for disease and containing the outbreak once an infected animal is identified. 47,64,93
Both New World and Old World species are susceptible. Most reports of mycobacteriosis in Old World animals involve rhesus and cynomolgus macaques, but African monkeys and great apes are susceptible as well. 64 Sporadic cases of pulmonary and disseminated mycobacteriosis have been reported in owl monkeys, squirrel monkeys, and capuchins, but these species are believed to be relatively resistant. 60,68,123
Clinical disease
Although cynomolgus and rhesus macaques appear equally susceptible to infection, cynomolgus monkeys are much less likely to develop clinical disease. 47 At low doses of inoculum, experimentally infected cynomolgus monkeys develop chronic, latent infection in approximately 40% of cases. 19 Clinical signs of active disease consist of weight loss, cough, and dyspnea. Monkeys often hide signs of illness in the presence of their caretakers, however, so respiratory signs may not be detected until late in the course of disease. In the laboratory setting, animals are likely to be euthanized before infection proves lethal.
Pathogenesis
Following infection, pulmonary macrophages are drawn to mycobacterial lesions by components of the mycobacterial cell wall. Mycobacteria survive phagocytosis by phagosome escape and inhibition of phagosome–lysosome fusion. 44 Containment of infection relies on the host’s ability to mount an appropriate Th1 response and sequester bacteria within granulomas. Tumor necrosis factor–alpha expression by macrophages in response to mycobacterial antigens is necessary for recruitment of lymphocytes, induction of an appropriate cytokine response, and formation of granulomas. 108 If the host mounts an effective Th1 cytokine response, macrophages are potentiated through interferon gamma and interleukin-1 beta. Interferon gamma causes destruction of mycobacteria via autophagy of pathogen-laden phagosomes, whereas interleukin-1 beta reestablishes phagosome–lysosome fusion through unknown mechanisms. 44 An inappropriate host response permits infected macrophages to migrate from the site of the primary lesion and seed granulomas in distant organs.
Even in the face of an effective immune response, mycobacteria may persist either in small populations of slowly dividing cells or in a state of metabolic dormancy. The necrotic center of a granuloma is an hypoxic environment that may trigger bacteria to enter dormancy. 4,142 Immunocompromise of the host may lead to reactivation of infection.
Pathology
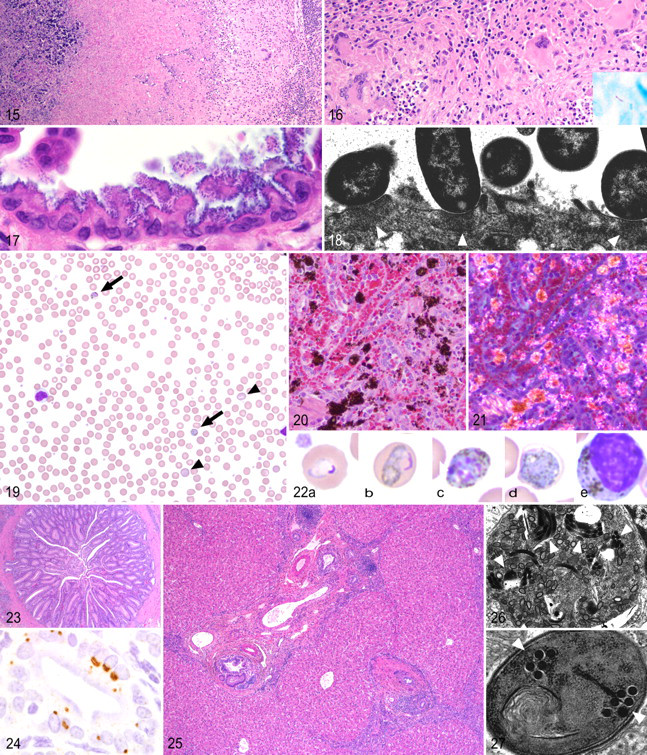
In patent infections, caseous granulomas (“tubercles”) are readily apparent on gross examination of the lung (Fig. 13 ). Granulomas may become confluent, liquefy, or cavitate (Fig. 14 ). The disease spreads through the pulmonary lymphatics, seeding tracheobronchial lymph nodes or distant organs and producing granulomas in the liver, spleen, and other viscera. 67 Histologically, lesions range from prototypical granulomas—consisting of caseous, necrotic cores encircled by epithelioid macrophages, Langhans giant cells, and lymphocytes—to poorly organized pyogranulomas. Occasionally, airways fill with neutrophils and necrotic debris. 76 Granulomas from monkeys with chronic or latent disease are encapsulated and often mineralized with fewer epithelioid macrophages and giant cells (Fig. 15 ). Acid-fast bacilli may be found within the caseous core or within the surrounding macrophages, but often multiple sections and granulomas must be examined to identify the pathogen (Fig. 16 ). 19
Diagnosis
The lack of a convenient, sensitive, and specific antemortem diagnostic test remains a persistent difficulty in maintaining a tuberculosis-free colony. The intradermal skin test with mammalian old tuberculin and subjective visual scoring of the delayed-type hypersensitivity response remains the standard means to screen animals and colonies. The skin test is insensitive in latently infected animals and must thus be repeated periodically to identify newly acquired or recrudescent infections. 74,77 False positives based on presumptive exposure to environmental atypical mycobacteria are common. Recently, 2 commercial diagnostic kits have become available for use in NHPs: The PRIMAGAM (Prionics AG, Zurich, Switzerland) test measures IFN-γ production by whole blood leukocytes in response to mycobacterial antigens, whereas the PrimaTB STAT-PAK (Chembio Diagnostic Systems, Medford, NY) detects antibodies to several mycobacterial proteins in serum samples. 48,78 Neither is offered as a replacement for the skin test, but when used in combination with the other, they both may help to classify otherwise equivocal results.
All 3 tests rely on a functional immune system. Immunosuppressive experimental manipulation or comorbidities such as measles virus infection may cause false-negative results. In our experience, animals may repeatedly test negative or equivocal on all 3 tests and still be confirmed positive at necropsy. Culture and isolation remains the gold standard for a positive diagnosis of M. tuberculosis but may take several months to complete. More rapid assays, such as use of PCR to detect mycobacterial nucleic acid, are being developed, but the sensitivity and specificity have not been rigorously addressed in NHPs.
Enteropathogenic Escherichia coli
Etiology
Seven distinct diarrheagenic strains of Escherichia coli are recognized on the basis of the lesions they produce: enteropathogenic E. coli (EPEC), enterohemorrhagic E. coli (EHEC), enteroaggregative E. coli, enterotoxigenic E. coli, enteroinvasive E. coli, Shiga-toxin-producing E. coli, and diffusely adhering E. coli. Among the diarrheagenic strains, both EPEC and EHEC produce a characteristic attaching and effacing (A/E) lesion on histopathology. 130 EPEC is differentiated from EHEC in that it lacks Shiga-like toxins that induce hemorrhage. EPEC associated with the A/E lesion was identified as a major cause of colitis in marmosets, SIV-infected rhesus macaques, and cotton-top tamarins. 85,86,129 The role of pathogenic E. coli in producing gastrointestinal disease in NHPs has only recently been recognized.
Epizoology
An outbreak in a marmoset colony resulted in hemorrhagic diarrhea in animals from 68 of 72 cages. 129 In a study of macaques dying of simian AIDS, the A/E lesion was identified histologically in 28% of cases. In 7% of cases, E. coli was the only enteric pathogen recognized. 87 Following an outbreak of acute colitis in a colony of cotton-top tamarins, 42% of the other animals were found by PCR to harbor EPEC. The presence of EPEC correlated with the degree of inflammatory disease, as determined by colon biopsy. 86
Clinical disease
Animals may carry EPEC asymptomatically. In marmosets, diarrhea is acute and variably hemorrhagic. 129 Patent infections in macaques and tamarins result in colitis, which manifests as acute or chronic nonhemorrhagic diarrhea. In SIV-infected animals, it is often identified in concert with other opportunistic agents, including Cryptosporidium, Enterocytozoon bieneusi, atypical mycobacteria, amoebae, and Balantidium. These animals typically present with chronic diarrhea, wasting, and dehydration. 85 In cotton-top tamarins, the organism is associated with acute hemorrhagic colitis or identified in association with chronic ulcerative colitis. 86
Pathogenesis
Pathogenicity is conferred by a number of genes encoded by pathogenicity islands in chromosomal, episomal, and phage DNA. The capability for A/E is encoded by a chromosomal pathogenicity island called the locus of enterocyte effacement, 95 which contains several genes that encode a type III secretion system, the bacterial attachment protein intimin, and the translocated intimin receptor (Tir)—a transmembrane protein that the bacterium injects into a eukaryotic host cell by means of the type III secretion system. 65,96 E. coli bind to host cell membranes through several molecular interactions, but the close attachment of EPEC to the intestinal epithelium is mediated by intimin–Tir binding. 65 Tir also promotes restructuring of the host cell cytoskeleton to form flat actin “pedestals” beneath the adherent bacteria. Microvillus architecture is distorted and lost, leading to malabsorption. 111
Pathology
Case reports do not describe gross lesions with the exception of diarrhea. Histologically, EPEC produce a characteristic A/E lesion in the colon. In severe infections, the ileum and distal jejunum may rarely be colonized as well. Colonic glandular epithelium is often markedly hyperplastic with loss of nuclear polarity, increased mitotic rate, and loss of goblet cells, adopting a “cobblestone” or “tombstone” appearance. Enterocytes are hyperbasophilic and vacuolated. Necrosis and sloughing of epithelium lead to flattening of the remaining epithelial cells. Microvilli are lost. The inflammatory response is primarily neutrophilic and is often mild in comparison to the severity of clinical signs. 85,86 The lesions in marmosets include marked neutrophilic and lymphoplasmacytic typhlocolitis with neutrophilic vasculitis of the submucosal vessels and erosion of the epithelium. 129
The bacteria themselves are easily visualized on routine hematoxylin and eosin staining. They appear as minute, darkly basophilic dots that line up along the apical surface of mucosal epithelial cells (Fig. 17 ). Ultrastructurally, the bacilli are closely apposed to the host cell membrane. The cytoskeletal “pedestals” are visible as slightly electron-dense, roughly semicircular areas in the host cell cytoplasm beneath each bacterium (Fig. 18 ). 85,86
Diagnosis
Enteropathogenic E. coli are easily overlooked with routine culture if the lab in question does not speciate lactose-fermenting organisms from fecal samples. An in vitro adhesion assay is available to screen strains for aggregative behavior. 32 PCR for specific virulence factors is useful for identifying strains. Recently, a multiplex real-time PCR pooled-colony screening assay was developed capable of detecting and distinguishing among 6 pathogenic strains. 9
Protozoa
Plasmodium spp.
Etiology
Plasmodium spp., the etiologic agents of malaria, compose a genus of mosquito-borne apicomplexan hemoparasites of several vertebrate taxa. Nearly all NHPs are susceptible to infection, but natural infection is rare in rhesus macaques, marmosets, and owl monkeys. In the laboratory setting, splenectomized owl monkeys are used as experimental models of disease. 59 Cynomolgus macaques control infection well and are frequently subclinically infected when imported. 30,90 If immunocompromised by stress or as part of an experimental procedure, monkeys may develop fulminant recrudescent disease. 127 With the marked increase in importation of cynomolgus macaques in the past 10 years, subclinical infection with Plasmodium spp has increasingly been recognized as a potential confounder of experimental work.
Parasites have a complex life cycle that begins when an infected anopheline mosquito inoculates a vertebrate host with sporozoites, which invade hepatocytes to form exoerythrocytic schizonts that release merozoites into the blood stream. Merozoites enter erythrocytes and morph into trophozoites that feed on hemoglobin growing into schizonts or gametocytes. Schizonts rupture their erythrocytes to release more merozoites into circulation. Male and female gametocytes fuse when taken up by a mosquito during a blood meal, and they undergo a further round of schizogeny to form sporozoites, thus completing the life cycle.
The life cycle has slight differences among species. Malaria parasites are unique in that erythrocytic schizogeny is synchronized. Depending on species and strain, merozoites are released at 24-hour (quotidian), 48-hour (tertian), or 72-hour (quartan) intervals. In addition, the exoerythrocytic schizonts of some species enter a dormant state capable of reactivation and reinfection of an aparasitemic host. The dormant parasites are known as hypnozoites.
Many Plasmodium spp. that naturally infect NHPs have been proven to infect humans as well. 28 Plasmodium knowlesi, a parasite of cynomolgus and pig-tailed macaques, is an emerging disease in human populations of Southeast Asia. P. knowlesi infections present a diagnostic challenge because the parasite is difficult to distinguish from the human parasite, Plasmodium malariae, but is much more virulent. 31,69 Table 3 presents a summary of the more common species.
Epizoology
Published statistics on the prevalence of infection of imported animals are mostly lacking. Studies of wild primates have revealed great variability in rates of infection depending on geography and species. One study of more than 3,000 NHPs from the Amazon valley revealed an overall prevalence of 10% for Plasmodium brasilianum. 33 Older reports of prevalence in imported cynomolgus macaques range from 2 to 25%. 127
Clinical disease
Compared with uninfected animals, chronically infected animals are usually asymptomatic but generally show significant decreases in hematologic parameters, such as hematocrit, hemoglobin concentration, and mean corpuscular volume. 38 Acutely infected animals show nonspecific signs of pyrexia, inappetence, and lethargy. 104 Other signs result from hemolytic anemia and circulatory dysfunction, including pale icteric mucous membranes, prolonged capillary refill time, tachycardia, ejection murmurs, dehydration, and tachypnea. 116,127 Complete blood counts reveal severe regenerative anemia with intraerythrocytic trophozoites, schizonts, and gametocytes visible on blood smears (Fig. 19 ).
Pathogenesis
Like those of many diseases, lesions produced by malaria parasites result from activities of both the parasite and the host. The parasite itself is injurious to the host in several ways. Parasitized erythrocytes are removed by the reticuloendothelial system resulting in extravascular hemolysis and anemia. Erythrocytic schizogeny results in intravascular hemolysis compounding anemia and causing hemoglobinuric nephrosis at high parasite burdens. Additionally, some species evade the reticuloendothelial system by causing parasitized cells to lodge in the microvasculature where they cause local ischemic damage and inflammation. 34
An excessive host inflammatory response can be detrimental. The mechanisms by which Plasmodium spp induce inflammation are diverse and include signaling by Toll-like receptors, Nod-like receptors, and the release of proinflammatory mediators such as uric acid, histamine, and complement factor C5a. 41 TLR2 and TLR9 are of particular interest because they include malarial glycophosphatidylinositol and malarial pigment-associated DNA among their repertoire of recognized patterns. Parasites excrete a hemoglobin metabolite, hemozoin, which provokes antigen-presenting cells to produce a variety of inflammatory cytokines and may alter dendritic cell maturation and function. 29
Pathology
The severity of lesions correlates with the severity of disease. In fulminant disease, gross lesions are those of hemolytic crisis, including icterus, hydropericardium, hydroperitoneum, and hydrothorax. The spleen and liver are enlarged and congested. Kidneys are discolored red-brown with hemoglobin. Infarction of the extremities may occur. With cerebral involvement, the brain is edematous, and cerebral vessels are congested. 104 In some cases hemozoin pigment imparts a gray-green or gray-brown color to the lung, brain, abdominal fat, and serosa. 126
Microscopically, Kupffer cells and splenic macrophages contain deposits of granular brown to black intracytoplasmic hemozoin pigment (Fig. 20 ). Hemozoin may be distinguished from hemosiderin by its white birefringence when viewed under polarized light but is difficult to differentiate from artifactual acid hematin (Fig. 21 ). Spleens are enlarged owing to follicular hyperplasia. Deposits of fibrin and necrotic lymphocytes may be found within germinal centers. Hepatic central lobular zones undergo hydropic and fatty degeneration, consistent with hypoxic change. Acute kidney injury is common. Tubules are variably necrotic and contain cellular and hemoglobin casts. Hematopoietic cells accumulate in the congested vasa recta. 126 Species that cause infected erythrocytes to adhere to vessel walls result in plugs of parasitized erythrocytes lodged in small capillaries. 104
Diagnosis
Diagnosis of severe parasitemia is easily accomplished by routine examination of blood smears (Fig. 19 ). Chronic infections in cynomolgus macaques are more difficult because of lower parasite burdens and the cyclical rhythm of schizogeny. Several daily consecutive smears may be necessary to confirm infection. Speciation by morphology requires an experienced microscopist and a sufficient parasite burden to provide examples of different life stages (Fig. 22). 28 Nested PCR assays have been described for diagnosis and speciation of human Plasmodium spp. capable of reliably detecting as few as 10 parasites per microliter of blood. 122 Sensitive real-time PCR and nucleic acid sequence–based amplification assays have also been developed. 97 There is currently no antemortem test for dormant exoerythrocytic organisms.
Fungi
E. bieneusi
Etiology
E. bieneusi is a microsporidian parasite recognized to infect a variety of vertebrate hosts. Microsporidia are a group of eukaryotic obligate intracellular parasites traditionally classified as protozoa. Recent biochemical, phylogenetic, and genome structural analyses, however, classify them as descendents of zygomycete fungi. 50,70 Their life cycle is simple, with only 2 stages: an extracellular infective spore and an intracellular meront, or Plasmodium. The defining morphologic feature of Microsporidia is the presence of a coiled polar tube.
E. bieneusi is an emerging pathogen in the human population as a result of the AIDS epidemic. First recognized in 1985 in a Haitian AIDS patient, it has since been reported in a number of nonhuman animals species, including healthy and immunosuppressed macaques. 35,54,82,83 With the increasing use of rhesus macaques as an AIDS model, E. bieneusi infections have become common in experimental animals.
Epizoology
A retrospective study of necropsy specimens from SIV-infected macaques of our colony revealed an overall prevalence of E. bieneusi of 10%, or 35% considering only animals with histopathologic evidence of hepatobiliary disease. 83 PCR analysis of fecal material revealed an even higher prevalence: 34% in SIV-infected animals and 17% in healthy controls. 82
Clinical disease
E. bieneusi is a common cause of chronic diarrhea and biliary tract disease in human AIDS patients, and it has been reported as a cause of transient, self-limiting diarrhea in immunocompetent individuals. 135 In macaques, the biliary tree is the most common site of infection. Specific clinical signs have not been reported in experimentally inoculated SIV-infected monkeys, but they may be masked by other opportunistic infections. 54,131 Infected animals do have significant elevations in serum alkaline phosphatase. 82
Pathogenesis
Owing to the relative novelty of the organism and the lack of in vitro and rodent models, little is known about the pathogenesis of E. bieneusi infection. Both immunologically normal and immunocompromised macaques are readily inoculated orally, and they begin shedding spores with about a week following infection. The infection rapidly disseminates to the hepatobiliary tree and can be detected in the bile within 2 to 3 weeks of inoculation. In immunologically normal animals E. bieneusi can be detected in bile for prolonged periods following experimental inoculation suggesting that this may be the site of persistence for the pathogen. 54,131
Pathology
Histopathology of immunocompetent animals is subtle—typically, a mild lymphoplasmacytic cholangitis. In immunocompromised animals, fulminant cholangiohepatitis, cholecystitis, and choledochitis may occur. Grossly, the bile duct and gall bladder mucosa are thickened, and the bile is translucent and watery. Histologically, bile ductules are markedly proliferative and accompanied by bridging portal fibrosis. In rare cases, concentric layers of fibrous tissue surround bile ductules (Fig. 23). Portal areas are accompanied by infiltrates of lymphocytes and plasma cells sometimes arranged in nodular aggregates. The gall bladder and bile duct mucosa are proliferative and inflamed (Fig. 25). Infected mucosal epithelial cells are often partially or wholly extruded into the lumen. 83
E. bieneusi less commonly causes a nonsuppurative polyserositis and pleuritis. Affected serosa is edematous and the mesothelium is reactive and proliferative. The organism can be localized to mesothelial cells and macrophages. 24
Organisms are difficult to visualize with routine hematoxylin and eosin staining. They may appear as 1.0- to 1.5-μm-diameter pale ringlike structures in extruded cells. A clue to their presence is provided by the indentation of the host cell nucleus, caused by the enlarging multinucleated plasmodial stage. The organism is easily demonstrated by special stains, such as Weber’s modified trichrome or Calcofluor White M2R (Sigma, St. Louis, MO). 54,83 In situ hybridization probes and monoclonal antibodies have also been developed to localize the organisms in tissue (Fig. 24). 21 Ultrastructurally, members of the genus Enterocytozoon may be distinguished from other genera by a number of characteristic features—most notably, cytoplasmic electron-lucent inclusions that persist throughout most of the life cycle, development of polar tube complexes before plasmodial division, and the presence of 5 to 7 polar tube doublets visible within the sporoblast and spore cytoplasm (Figs. 26 , 27). 25
Diagnosis
Infected animals shed spores in their feces, which can be detected by trichrome or Calcofluor staining of fixed fecal smears, but PCR amplification of fecal DNA is more sensitive. 21,54
Microbial Quality Control in NHPs
To facilitate the detection and control of potential pathogens, all facilities that house NHPs should implement comprehensive microbial quality programs. Clearly for some agents, complete pathogen eradication and measures to prevent reintroduction to protect animals, personnel, and research programs are warranted. 84 However, elimination of all infectious agents from NHP colonies may not be a realistic goal and, in fact, may not be desirable. If animals are to be used to model human conditions, the presence of infectious agents that closely approximate those observed in human populations may allow for the better assessment of the effect of drugs, stressors, and experimental manipulations. Because it has not been possible to eliminate all pathogens from NHP colonies, it is important to define the spectrum of microbial agents present within a population of animals and understand how these agents influence experimental work and colony health. 84
Microbial quality control programs are multifaceted and, at a minimum, include (1) prescreening and quarantine measures; (2) routine preventative health examinations with tuberculosis testing; (3) serological and molecular testing for common viral agents; (4) vigorous diagnostic approach to animals with undefined illnesses; (5) complete medical records that document clinical, experimental, and housing histories; and (6) complete necropsy and histologic evaluation of all animals that die or are euthanized. The veterinary pathologist plays a key role in integrating and interpreting these data and must be familiar with the full spectrum of infectious diseases present within NHP populations.
Emerging and reemerging infectious diseases remain a threat to NHP colonies and research programs. These threats relate to the importation of animals from nondomestic sources, the influence of immunomodulatory effects of experimental manipulation, and the recognition of previously unknown agents. Their impact varies from subtle but significant confounders of experimental work to diseases with overt morbidity and mortality. All facilities with NHPs should implement comprehensive microbial quality programs, and we should seek methods to integrate and bridge disease surveillance across facilities.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health / National Center for Research Resources Grant Nos. P51 RR000168 and T32 RR000700.

