Abstract
Most papillomaviruses (PVs) are oncogenic. There are at least 100 different human PVs and 65 nonhuman vertebrate hosts, including wild rodents, which have species–specific PV infections. Florid papillomatosis arose in a colony of NMRI-Foxn1nu/Foxn1nu (nude) mice at the Advanced Centre for Treatment Research and Education in Cancer in India. Lesions appeared at the mucocutaneous junctions of the nose and mouth. Histologically, lesions were classical papillomas with epidermal hyperplasia on thin fibrovascular stalks in a verrucous pattern. Koilocytotic cells were observed in the stratum granulosum of the papillomatous lesions. Immunohistochemically, these abnormal cells were positive for PV group-specific antigens. With transmission electron microscopy, virus particles were observed in crystalline intranuclear inclusions within keratinocytes. The presence of a mouse PV, designated MusPV, was confirmed by amplification of PV DNA with degenerative primers specific for PVs. This report is the first of a PV and its related disease in laboratory mice.
Papillomaviruses (PVs) are small nonenveloped DNA viruses that are species and anatomic site-specific. They form the Papillomaviridae family. PVs infect a variety of mammals, birds, and reptiles, inducing a variety of benign and malignant neoplasms. 1,24 More than 100 different human PVs have been identified, and virtually all human cervical cancers are caused by mucosotrophic oncogenic PVs. 10,11 Most PV infections causing cervical cancer can be prevented by commercially available recombinant PV vaccines. 9,18 Although the conception of the efficacious human papillomavirus (HPV) vaccine was established by the beginning of the 1990s, it was not widely accepted until its efficaciousness was proven with the canine oral PV (COPV) model. In those studies, recombinant viruslike particles of COPV were 100% efficacious in preventing initial infection of COPV but not the other PVs. 4 No therapeutic treatment for precancerous or malignant HPV-related cancers is currently available. 1 Since the early development of transgenic technology, genetically engineered mice were created that expressed PV genes, usually from human or bovine PVs. Although interesting information was obtained, little data could be translated to human diseases caused by HPVs. Although nonhuman PV model systems have been critical for advancing our knowledge of this group of oncogenic viruses, a naturally occurring PV infection in laboratory mice would provide a major new tool for investigating the molecular pathogenesis and new treatment approaches for this group of viruses.
The canine PV (primarily COPV), rabbit PV (primarily cottontail rabbit cutaneous PV, which induces nonproductive squamous cell carcinomas in the domestic rabbit), and bovine PV (types 1, 2, and 4) were historically used as the primary animal models for understanding the pathogenesis of the viral-induced cancers and vaccine development. 24 All are large animals in the context of laboratory animal species. PVs are prevalent in many species and are known to infect wild species of rats and mice, 12,13,17,23,26,27 as well as other wild rodent species. 24 As such, laboratory mice should be naturally infected as well, but owing to the high levels of husbandry practiced by many institutions today, infectious diseases in mouse colonies are considered all but eliminated. Laboratory mice provide numerous advantages as a biomedical tool over the current PV models in that they are relatively safe, easy to handle, and numerous immunological and biological reagents are readily available for detailed molecular studies. We report here identification of a cutaneous laboratory mouse PV (MusPV) and the lesions associated with this infection.
Materials and Methods
Mice
All mice were housed at the Advanced Centre for Treatment Research and Education in Cancer (ACTREC), Kharghar, Navi Mumbai, India. NMRI-Foxn1nu/Foxn1nu (nude) mice were inbred and procured from the National Institute of Virology, Pune, India. Swiss mice procured from the National Institute of Virology, Pune, India, were bred in-house. Mutant bare (S/RV/Cri-ba/ba) mice were found in a colony of these Swiss inbred mice, which were subsequently bred and maintained in the ACTREC. Mice were maintained in vivarium that controlled humidity (55 ± 5%), temperature (22 ± 2°C), and light (cycle, 12:12 hours) under specific pathogen-free conditions for Mycoplasma pulmonis, hantavirus, lymphocytic choriomeningitis virus, Clostridium piliformae, CAR bacillus, Sendai virus, ectromelia virus, pneumonia virus of mice, Kilham rat virus, and Reo3 virus tested by commercially available ELISA-based serology techniques. Myocoptes spp. were observed in modules of normal mice but not in those of the nude mice. No ectoparasites were found by histopathology in any cutaneous sections. Nude and bare mice were housed in an individually ventilated caging system with polycarbonate cages (360-cm2 floor area) at a maximum capacity of 5 mice per cage. Mice were allowed free access to autoclaved feed pellets (made in-house by ACTREC) and ultraviolet-treated water. The ACTREC is registered with the Committee for the Purpose of Control and Supervision of Experiments on Animals, Ministry of Environment and Forests, Government of India, for breeding and conducting experiments on small laboratory animals. All rules and regulations were followed for breeding and maintenance of the small laboratory animals.
Tissue Samples
To prevent the spread of infections, mice that were observed with clinically evident papillomas were euthanized by carbon dioxide asphyxiation. Naturally occurring papillomas from NMRI nude mice were collected at the time of necropsy and either snap-frozen in liquid nitrogen and stored at –80°C for molecular studies or fixed in neutral buffered 10% formalin, embedded in paraffin, processed routinely, sectioned at 6 μm, and stained with hematoxylin and eosin. Serial sections were tested for the presence of PV group–specific antigens as previously described. 22 Conventional bacterial cultures were done on papillomas. Coagulase-positive Staphylococcus spp was isolated from some but not all papillomas. In all cases, the isolated Staphylococcus was not associated with ulcers or erosions and, as such, was interpreted to be a secondary isolate.
Electron Microscopy
Representative samples from papillomas were minced with a razor blade, fixed in 2% buffered gluaraldehyde, embedded in LX-1112 plastic embedding medium (Ladd Research Industries, Inc, Burlington, VT), mounted on 200 mesh copper grids, stained with uranyl acetate and lead citrate, and viewed under a Philips CM-12 transmission electron microscope. Nuclei in the stratum granulosum of the papillomas were scanned for intranuclear viral particles as previously described. 8
PV DNA Detection by Polymerase Chain Reaction Amplification and Sequencing Analysis
Viral DNA was extracted with routine methods and amplified by polymerase chain reaction with the MY9/11 and GP5+/6+ primer sets as previously described with modifications. 15 Briefly, the annealing temperature used for MY primers was 55°C and for GP primers, 45°C. Forty amplification cycles were carried out with high-fidelity Taq polymerase (Invitrogen, Carlsbad, CA). To obtain DNA sequencing data, amplified DNAs were extracted from the gel with a kit (QIAquick Gel Extraction Kit, Qiagen, Germantown, MD) and sent to the DNA core facility at University of Louisville for sequencing. DNA data was put through the BLAST program at the National Center for Biotechnology Information (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Transmission Studies
Papillomas were collected aseptically at the time of necropsy, homogenized in phosphate buffered saline, filtered through 0.22-μm Millipore filters (Millipore Corporation, Bedford, MA), and injected at 2 locations on the dorsal lumbar midline of 5 female S/RV/Cri-ba/ba mutant 8-week-old mice. These bare mutant mice have diffuse alopecia throughout life, 16 thereby facilitating viewing of lesions that are grossly visible. The bare mutant mice are immunocompetent and survive when maintained under conventional environmental conditions. Mice were observed weekly for 5 months.
Results
Florid papillomatosis (Fig. 1) appeared in a closed colony of NMRI-Foxn1nu/Foxn1nu mice at the ACTREC. All affected mice were euthanized to avoid spread of the infection to other animals in the colony. The lesions appeared as clusters of light brown to white papillary projections around the mouth and nose at the mucocutaneous junction. The papillomas were observed in other haired skin sites but in low multiplicity. The papillomas were limited to the cutaneous surface and did not extend onto the mucosa. Individual lesions ranged from 3 mm in diameter at the base, projecting at least 1 to 2 mm above the skin surface and upward of 10 to 15 mm (Fig. 1). Histologically, the edges of the lesions were acanthotic, adjacent to papillary projections of hyperplastic epidermis on thin fibrovascular stalks (Figs. 2, 3). Cells within the stratum granulosum had clear, unstained cytoplasm similar to the pale, clear, or hollow (koilocytotic) cells described for most productive PV infections (Fig. 3). 24
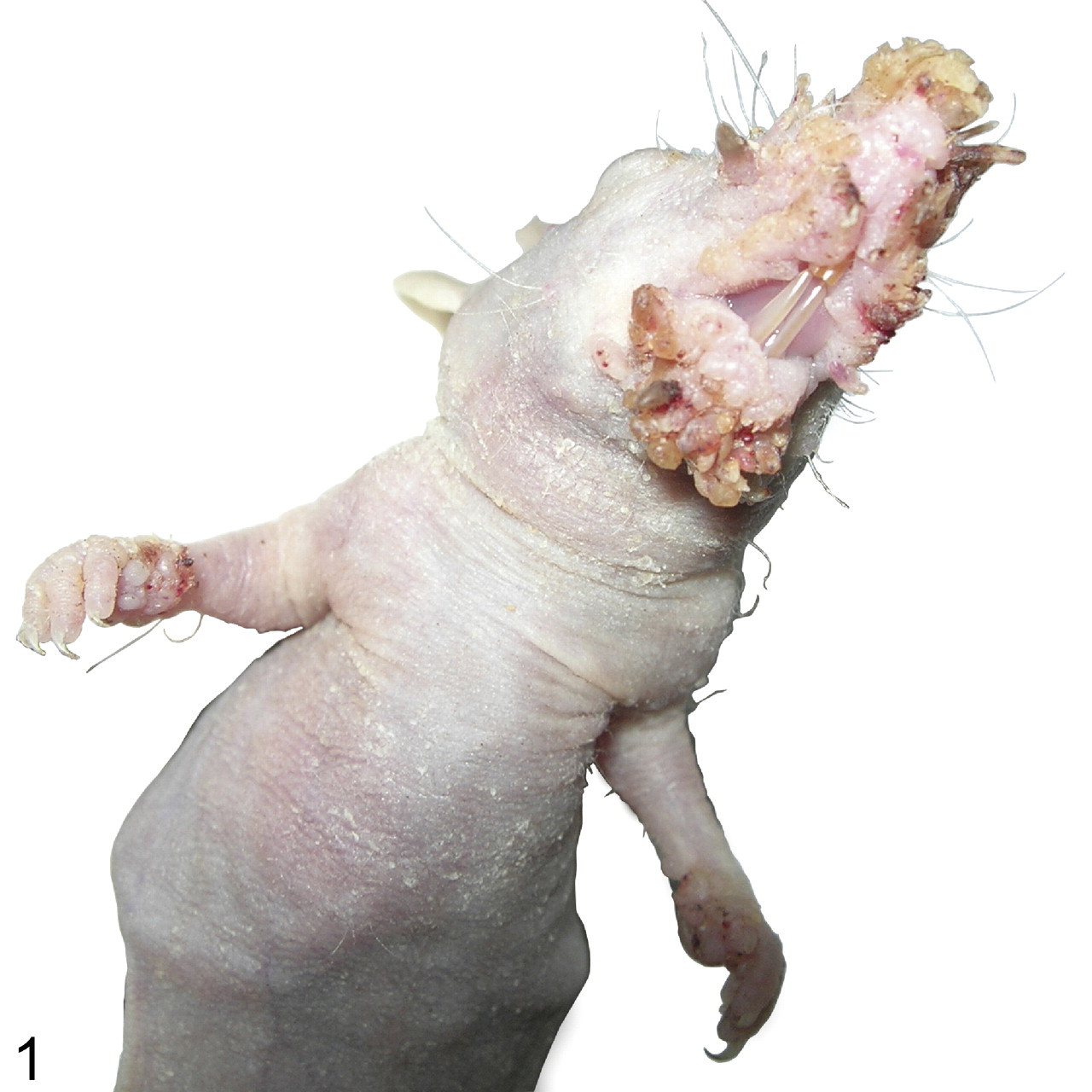
Florid papillomatosis around the nose and mouth of an NMRI-Foxn1nu/Foxn1nu (nude) mouse.
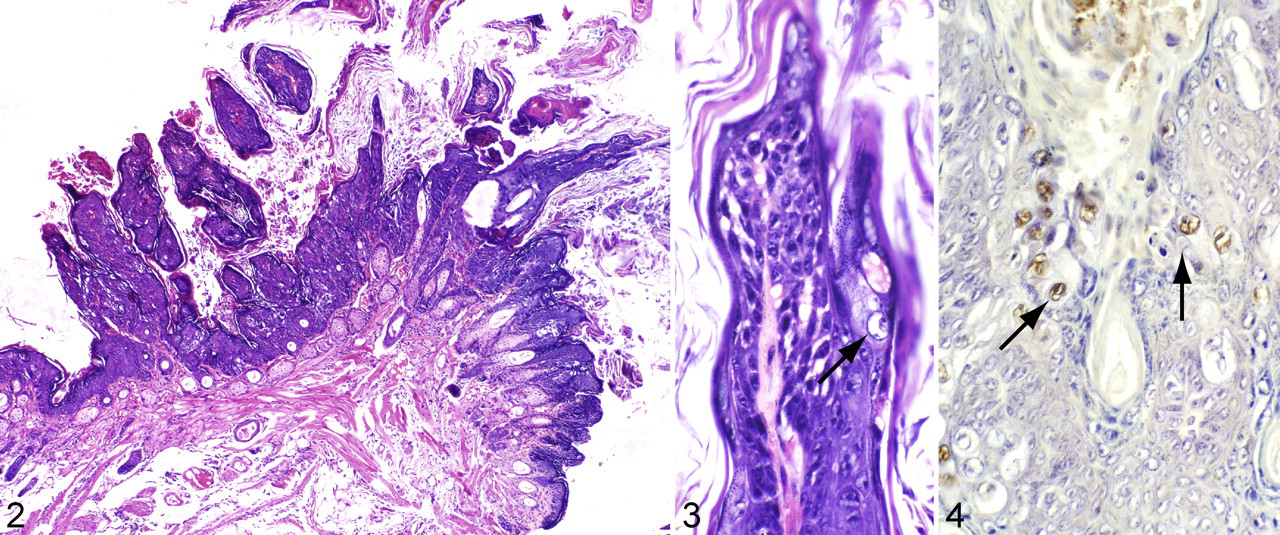
Immunohistochemistry demonstrated the presence of PV group–specific antigens within koilocytotic cells (Fig. 4). PV cross-reactive antigens exist mainly on L1 major capsid proteins, which would be overexpressed during PV productive infection. Virus particles were present within nuclei of koilocytes (Fig. 5). Although not all the cells observed under the electron microscope contained virus particles, some cells were filled with viruses that formed crystalline structures.
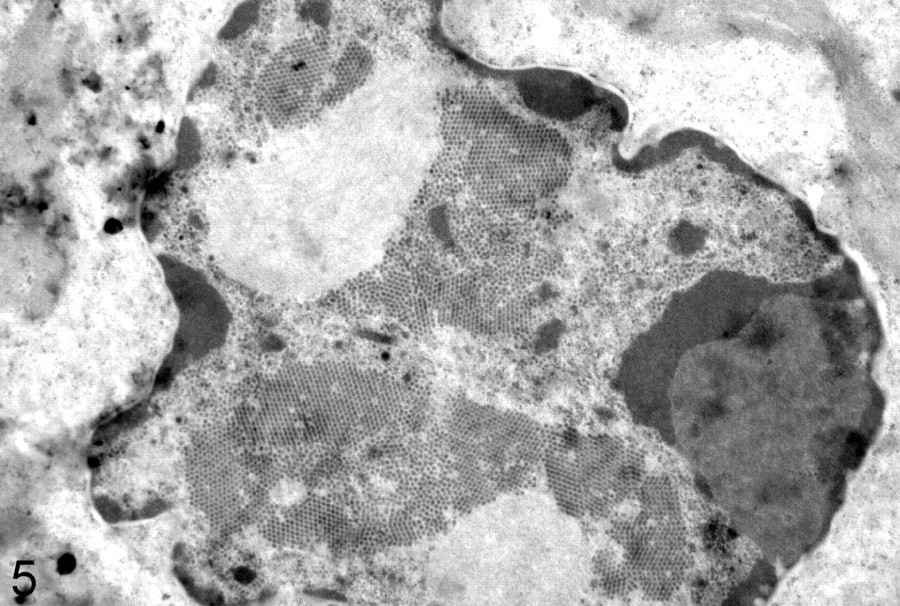
Nucleus of a koilocyte with numerous uniformly sized virus particles, many in crystalline arrays.
To determine if the agent associated with the cutaneous papillomas in nude mice was a PV, DNA was extracted from 4 papillomas harvested from 4 NMRI-Foxn1nu/Foxn1nu nude mice. Degenerative primers commonly used for the detection of HPVs, including MY9/11 and GP5+/6+, were used to detect PVs in these samples. Only MY9/11 primers amplified the PV in these samples (Fig. 6). The sequencing analysis of the MY9/11 amplicon, with BLASTn (n for nucleotide), revealed that this PV was most likely new (Fig. 7). Protein sequence analysis of the translated 347 DNA, by BLASTx (x for translated nucleotide), showed that the closest mammalian PV was the Mastomys coucha PV2, with 83% similarity.
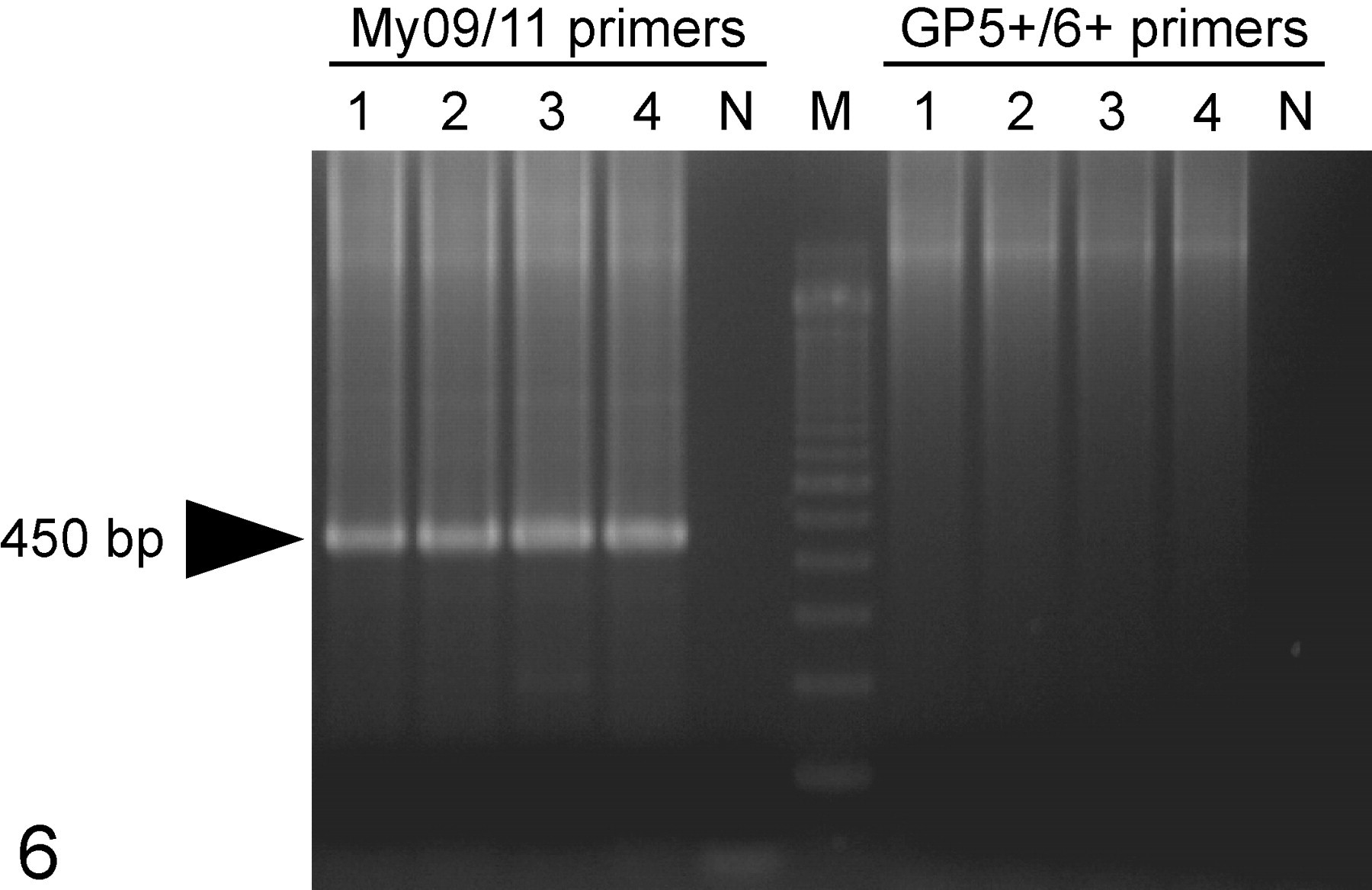
Degenerative primers commonly used for the detection of human papillomavirus DNA in tissues (MY9/11 and GP5+/6+) were used in the low–molecular weight DNA samples isolated from the nude mouse papillomas positive for papillomavirus antigens. Only MY9/11 primers amplified the papillomavirus DNA in these samples.

DNA sequence analysis of MY9/11 amplicon of the novel mouse papillomavirus (proposed name, MusPV) and its translated amino acid sequence.
The original papillomas on the nude mice were spontaneous, but these could be transferred to other immunocompetent laboratory mice. Five S/RV/Cri-ba/ba mice were inoculated with this PV virus at 2 sites with cell-free suspensions from papillomas harvested at the time of necropsy from affected NMRI-Foxn1nu/Foxn1nu . As a result, 3 of the 5 mice developed the papillomas 2 to 3 mm in diameter at both injection sites (Fig. 8); the other 2 mice developed papillomas only at one site. Papillomas were first evident 3 weeks after inoculation, and they completely regressed by 8 weeks postinoculation; as such, no skin or lesions were tested to confirm the presence of PV antigens or DNA.
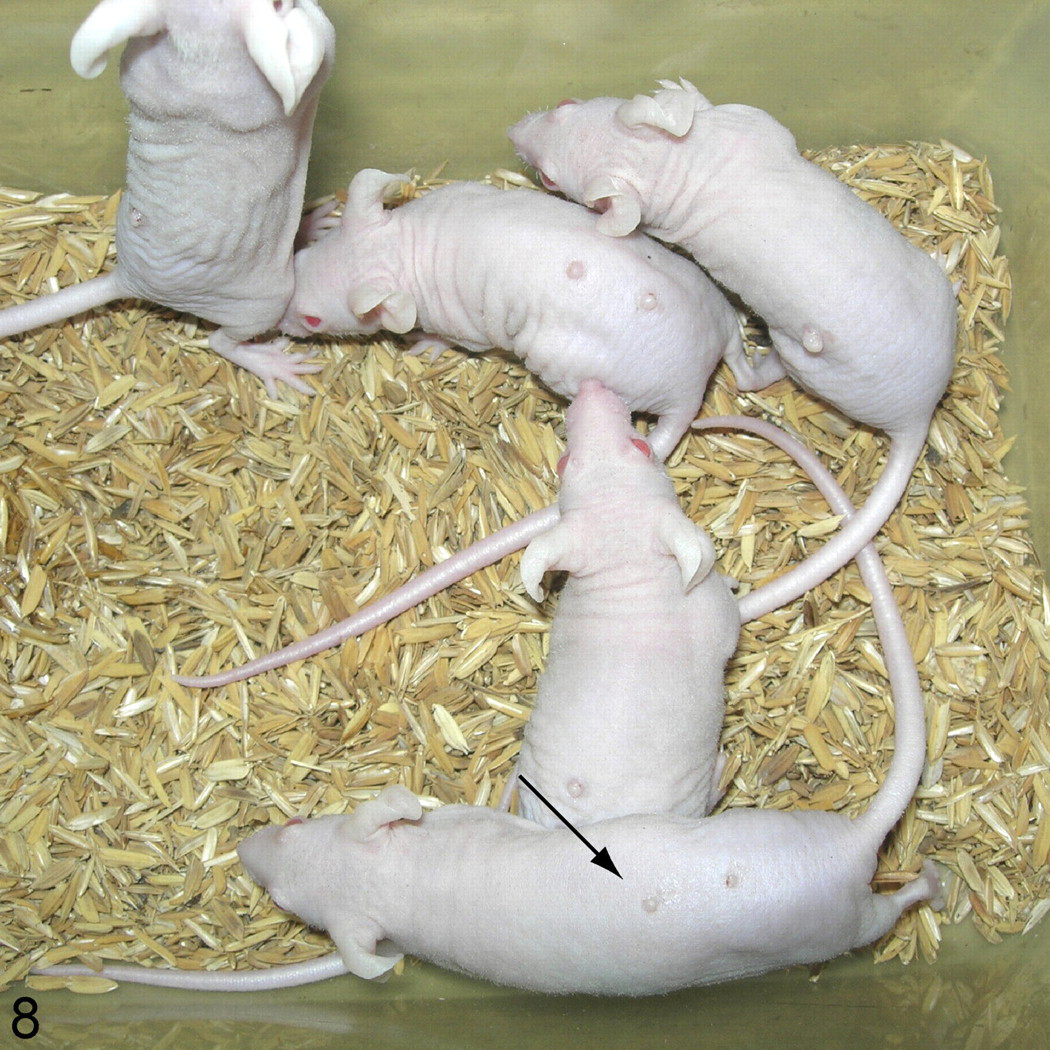
S/RV/Cri-ba/ba (bare) mutant mice injected with cell-free material from homogenized papillomas developed papules of 2 to 3 mm in diameter at the injection sites.
Discussion
High levels of husbandry, barrier maintenance, and pathogen screening in today’s laboratory mouse colonies worldwide limit the infectious diseases to which mice are exposed, enabling high-quality biomedical research to be conducted. Laboratory mice do spontaneously develop papillomas and squamous cell carcinomas that are not associated with PV infections, even in some of the highest barrier level facilities 2,20 and in response to chemical carcinogenesis protocols. 3,21,25,28 Routine screening of papillomas and squamous cell carcinomas in laboratory mice from colonies worldwide for more than 3 decades failed to identify a PV association, which was rather surprising given that these viruses infect most species examined (J.P.S., A.B.J., S.G., unpublished data).
We report here the first transmissible laboratory mouse PV, with polymerase chain reaction confirmation, with identification of PV group–specific antigens in the stratum granulosum of the papillomas and virus particles within the nuclei of koilocytes, and with successful transmission to immunocompetent mice. We have shown that the antibody used for immunohistochemistry is broadly cross-reactive between the PVs of birds and mammals 22 but does not react with other relatively close-related viral genera. 5,7 In addition, the epitopes that correspond to specific PV sequences strongly support its specificity for PVs. 6 These combined data indicate that this PV naturally infects immunodeficient mice (nude mice that lack T cells) 19 but is transmissible to immunocompetent mice.
Nomenclature of nonhuman PVs is built on the first 2 letters of the host genus and species, followed by PV and a number (ie, when more than one virus is characterized as affecting that species; the number represents the type of PV, based on the order in which it was first identified and published). 24 Because laboratory mice are generally derived from various subspecies of Mus musculus, 14 MmPV would be the recommended designation. However, MmPV was used for the harvest mouse PV (ie, Micromys minutus). 13,23 Given that this is a spontaneous PV infection identified in laboratory mice of the genus Mus, we thus propose MusPV for this novel virus. PVs are highly species specific and, often, anatomic site-specific. Although it is possible that the nude mice were infected from wild rodents, transmission studies have demonstrated that lesions can be induced in immunocompetent mice; therefore, this should be considered a mouse PV.
Although adding another PV and host to the ever-growing list of species infected is a minor point, the fact that this laboratory mouse PV is transmissible makes it an important new observation. It is now possible to do simple transmission studies with the vast numbers of spontaneous and genetically engineered mouse mutants to investigate the pathogenesis of this infection and possibly determine the key genetic/protein components of resistance, susceptibility, and even progression to squamous cell carcinoma.
Footnotes
Acknowledgements
We acknowledge the technical support of C. Caple for her work on the tissue prepared for electron microscopy.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was supported by the University of Louisville Clinical and Translational Science Pilot Grant Program.
