Abstract
A case is reported of fatal cardiomyopathy in an 8-year-old female German Shepherd after standard chemotherapy with doxorubicin for splenic hemangiosarcoma. The main gross lesion was a moderate bilateral cardiac ventricular dilation with diffusely pale myocardium. Histological analysis revealed severe multifocal vacuolar degeneration of cardiomyocytes, myocytolysis, myofibril loss, myocardial fibrosis, and edema. Myocardial fiber vacuolization and myocytolysis were highly suggestive of doxorubicin cardiotoxicity.
After undergoing a splenectomy for hemangiosarcoma, an 8-year-old female German Shepherd was treated with a combination protocol of doxorubicin and cyclophosphamide administered every 3 weeks for a total of 6 treatments. The cumulative dose of doxorubicin was less than 240 mg/m2 according to published protocols.3,5,15 All of the parameters (clinical chemistry and blood values) were within normal limits for the duration of the chemotherapy. After the sixth treatment, the dog was referred for sudden onset of general weakness. Heart failure with atrial fibrillation was diagnosed by electrocardiography, and soon afterward the animal died.
Gross and Microscopic Findings
At necropsy, the main gross lesion was a severe dilation of both ventricles. Moreover, mild pulmonary edema and diffuse vascular stasis were observed. Myocardium was pale with thin white longitudinal streaks visible beneath the epicardium. The entire heart was collected and fixed in formalin. Sections from the atrial and ventricular free walls, interventricular septum, and left papillary muscle were prepared by standard methodology and stained with hematoxylin and eosin and Mallory trichrome stain. Histologically, all the samples had similar pathological changes: Groups of severely damaged myocytes were admixed with normal-appearing myocytes (Fig. 1). There was no recognizable specific topographic pattern of lesions. Single scattered cells or groups of myocytes contained single or (more rarely) multiple and coalescing irregularly sized smooth-contoured empty vacuoles ranging from 4 to 36 μm in diameter (vacuolar degeneration/adria cells) (Fig. 2). When visible, the nucleus was displaced to the periphery. Scattered myocytes occasionally had perinuclear areas of homogeneous pale eosinophilic cytoplasm, with myofibrils recognizable only at the periphery of the cells (myofibril loss). Scattered hypereosinophilic angulated myocytes, fragmented myofibers, and microhemorrhages were also present. Myofibers were separated by mild interstitial edema. A moderate to large increase in collagen fibers was multifocally visible in the interstitium between myocytes (myocardial fibrosis) (Fig. 3). A small number of lymphocytes and plasma cells infiltrated these areas.
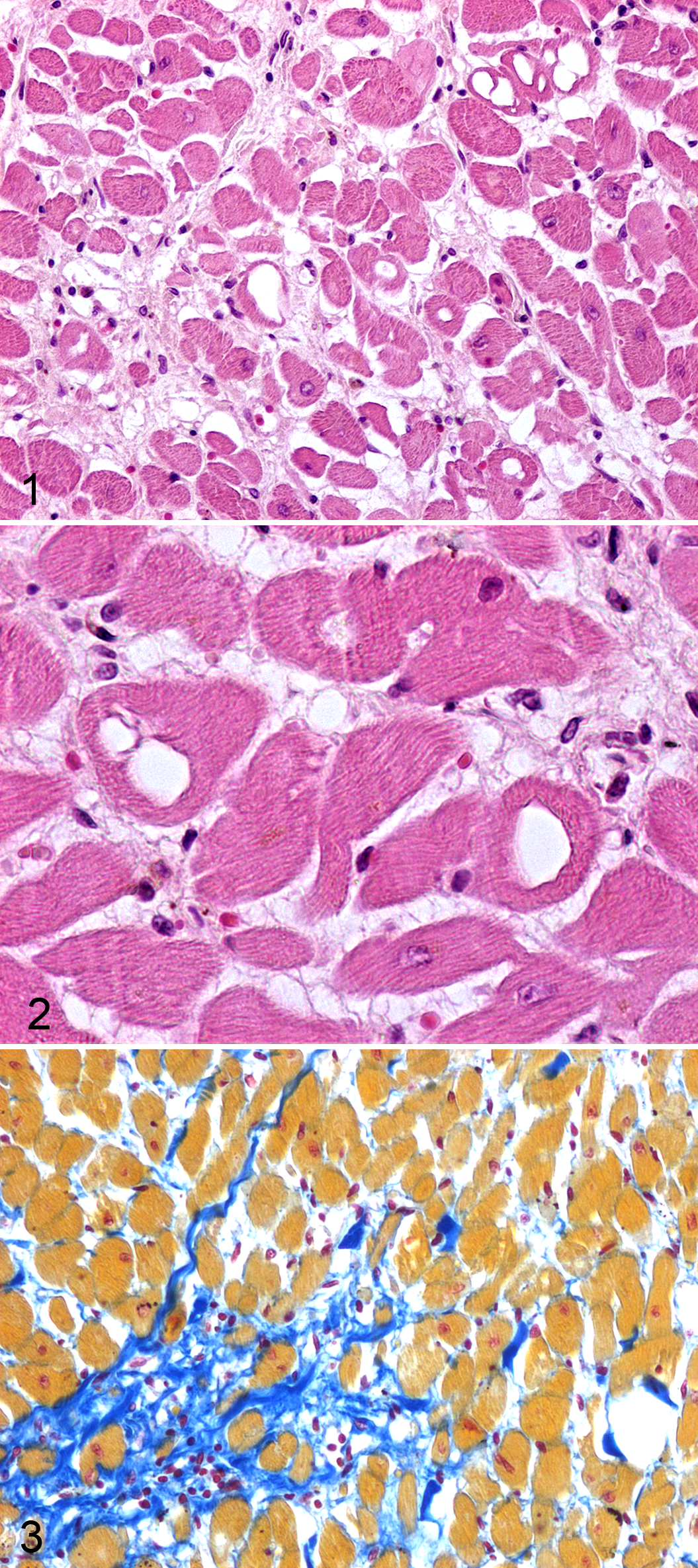
Diagnosis
A diagnosis was made of severe diffuse subacute to chronic cardiomyopathy. Vacuolar degeneration was considered a typical finding associated with doxorubicin treatment.
Discussion
Although doxorubicin is known to be a dose-limiting cardiotoxic drug that can cause irreversible dilated cardiomyopathy in dogs and people,10–12,14,15,18 it is currently one of the most widely used antineoplastic drugs. In humans, the myocardial damage caused by doxorubicin after chemotherapy can remain asymptomatic for months to years before manifesting with insidious effects followed by abrupt, rapidly progressive biventricular failure and death.12,14,15 Noninvasive techniques are poor indicators of early lesions and subclinical myocardial injury,9,10 and the gold standard for early diagnosis of ensuing doxorubicin-associated cardiotoxicity is serial right ventricular endomyocardial biopsies.1,4,14,15 In human cardiac biopsies, myocardial vacuolar degeneration (adria cells), myocytolysis, and interstitial edema are considered highly suggestive of doxorubicin cardiomyopathy.2,12,18 The mechanism of doxorubicin-induced myocardiopathy is not entirely known. The majority of the evidence indicates that free radicals are involved;15,18 the chemical structure of doxorubicin is prone to free radical generation, and increased oxidative stress may lead to subcellular changes, culminating in the death of myocytes.15,18 In dogs as in human beings, long-term administration of doxorubicin causes diffuse myocardial damage; however, there are currently no sensitive predictor tests available for monitoring canine patients.6,7,11,17 In dogs and human beings, the most sensitive test for monitoring early doxorubicin-associated cardiotoxicity is myocardial biopsy; 16 however, the invasiveness of this procedure precludes this technique from being routinely employed. Dogs are generally considered to be more sensitive than human beings to the cardiotoxic effects of doxorubicin. In dogs, cardiomyopathy and congestive heart failure is reported to occur at a total dose ranging from 122 to 400 mg/m2 body surface area,6–8,10,16,17 whereas in human beings, it occurs at a total dose greater than 550 mg/m2 body surface area. 7 Moreover, significant individual variation in susceptibility to anthracycline-induced cardiotoxicosis has been reported in human beings and dogs.10,12,13,15 In the present case, according to standard veterinary chemotherapy protocols, a cumulative dose of doxorubicin less than 240 mg/m2 was used; this dosage is recommended5,15 to minimize the cardiotoxic effects. Although the dog’s health was unremarkable during the course of the treatment, it experienced sudden-onset fatal cardiac failure. Dogs have been used as an experimental model of doxorubicin-induced cardiomyopathy, and cardiac lesions in dogs are similar to those seen in human beings.2,5,10,16
As interest in veterinary chemotherapeutic treatment increases, pathologists will begin to see more doxorubicin-induced myocardial lesions during routine diagnostic activities. It is therefore important that they be familiar with typical doxorubicin-induced myocardial histological changes.
Footnotes
Acknowledgement
We wish to thank Mr M. Colombo for photographic support.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
