Abstract
This study aims to investigate the expression of metalloproteinases (MMPs) and their tissue inhibitors (TIMPs) in chronic doxorubicin cardiomyopathy in a rabbit model and to evaluate the effects of bone marrow-derived mesenchymal stem cell (MSC) transplantation in this disease. Thirty-nine 3-month-old New Zealand rabbits were divided into 4 groups: group 1 (n = 9) was the untreated control. Groups 2–4 were treated with 6 weeks of doxorubicin (3 mg/kg). Group 2 (n = 6) received no further treatment. In group 3 (n = 9), animals were treated with culture medium (CM) alone. In group 4 (n = 15), autologous MSCs (1.5–2.0 X 106/ml) were injected in the left ventricular (LV) wall. Hearts were stained with HE and picrosirius red. MMP-1, −2, −3 and −9 and TIMP-2 and −3 were detected immunohistochemically. The mRNA levels were determined by real-time polymerase chain reaction. The results confirmed that doxorubicin treatment resulted in minimal myocardial fibrosis and showed that expression of MMPs increased and TIMP-3 decreased. The injection procedure resulted in increased myocardial fibrosis in groups 3 and 4. After MSC injection, MMP-1, MMP-2, and TIMP-3 expression was higher than that in group 2. CM injection led to more fibrosis, elevated TIMP-3, but diminished MMP-1 and MMP-2 expression compared with MSC injection. The mRNA levels of MMPs and TIMPs were not significantly different among all groups. In conclusion, chronic doxorubicin cardiomyopathy was characterized by increased MMP and decreased TIMP-3 expression. MSCs injection into the LV resulted in marked differences of collagen content and MMP/TIMP expression in the whole heart, although significant numbers of living MSCs were not detected after 4 weeks.
Keywords
Stem cell therapy is gaining importance as a potential new therapy for patients with advanced heart failure. 4, 14, 20 Induction of differentiation of bone marrow–derived mesenchymal stem cells (MSCs) in vitro into cells with beating myotubules by treatment with 5-azacytidine was first reported in 1995 by Wakitani et al. 35 Even without pretreatment, MSCs have recently been shown to home to the site of myocardial infarction, 3, 4 differentiate into cardiomyocyte-like cells, 31, 32 and integrate functionally and morphologically in the myocardium. 4, 39 Since then, the body of evidence has been growing that transdifferentiation plays a minor role, if any, in the therapeutic effects of stem cell transplantation. Using echocardiography, Scorsin et al. 28 observed improvement of fractional shortening 1 month after transplantation of fetal cardiomyocytes into doxorubicin-treated hearts in mice. Interestingly, this improvement of functional parameters was observed, although no evidence of cell survival over this course was found. It seems that as yet unidentified paracrine mechanisms may be involved in this therapeutic process. 27
In dilative cardiomyopathy, not only disturbances in the contractile apparatus of the cardiomyocytes but also alterations in interstitial architecture of the myocardium play a role in the pathogenesis of heart failure. 6, 10, 11, 12, 15 Extracellular matrix (ECM) integrity is maintained by a balance between the activity of matrix metalloproteinases (MMPs), a family of enzymes that degrade ECM proteins, 18, 19 and their tissue inhibitors (TIMPs). 6, 12, 15 Cardiomyocytes, fibrocytes, and endothelial cells express 1 or more types of MMP/TIMP. 12, 33 The activity of MMPs and TIMPs varies with the type, etiology, and stage of the cardiac disease; 10, 24, 30, 31 but in human idiopathic dilative cardiomyopathy in general, MMP levels are high and TIMP levels are low. 38 Doxorubicin is commonly used in anticancer therapy in humans and animals, but dilative cardiomyopathy is an undesired side effect. To our knowledge, MMPs and TIMPs have not been studied in doxorubicin cardiomyopathy in any species.
We hypothesized that alterations of ECM metabolism play a role in doxorubicin-induced cardiomyopathy and may be influenced by stem cell injection. The validity of the doxorubicin cardiomyopathy model has been established by previous studies in rabbits, 2 mice, 28 rats, 26 and dogs. 25 Thus, the aim of this study was first to investigate the expression of MMPs and TIMPs in chronic doxorubicin cardiomyopathy in the rabbit model and, second, to evaluate the effects of MSC transplantation in this disease.
Material and Methods
All experiments were conducted in accordance with guidelines published in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (USA) and were under protocols approved by the Institutional Animal Care and Use Committee at Leipzig University and Regierungspraesidium (Germany).
Animals
Three-month-old male healthy New Zealand white rabbits (Charles River, Germany), housed individually under conventional caging conditions, were used in this study. After a 2-week acclimatization period, animals were randomly assigned either to the healthy untreated control (group 1, n = 9) or to the doxorubicin treatment group (n = 30). Doxorubicin hydrochloride (Ribodoxo-L 50) (3 mg/kg of body weight) was administered once a week for 6 weeks intravenously followed by an interval of 14 days without treatment. These treated animals were then randomly subdivided into 3 groups: 1 with no further therapy (group 2, n = 6), a group with intramyocardial injection of culture medium (CM) (group 3, n = 9), and a group receiving autologous MSCs (group 4, n = 15). The time course of the experiment is summarized in Table 1.
Experimental design for examination of the effects of mesenchymal bone marrow–derived stem cells in doxorubicin-induced cardiomyopathy.

∗Echocardiograms were obtained on days 0, 42, 57, and 84.
— = no treatment or application
Isolation, Expansion, and Labeling of MSCs
Harvest and expansion of mesenchymal bone marrow stem cells were achieved in accordance with previously described methods. 22 To avoid immune rejection, we gave the animals of group 4 autologous femoral bone marrow cells (BMCs).
On day 50 of the experiment, a femoral bone marrow aspirate was performed aseptically in the animals of group 4. Marrow cells were flushed out with phosphate buffered saline, pH 7.4, and ethylenediaminetetraacetic acid (2 mM) (PBS-EDTA) to obtain a maximum amount of cells. The BMCs were fractionated over 1.073-g/ml Ficoll solution (Sigma, Steinheim, Germany), and mononuclear cells were obtained according to Tomita et al. 32 After 2 washings, BMCs were resuspended in 12-ml Dulbecco's Modified Eagle Medium (DMEM, Sigma) with 10% fetal bovine serum, 100-U/ml penicillin G, and 100-μg/ml streptomycin (Sigma) added. To induce differentiation, we cultured cells with 10-μM 5-azacytidine (Sigma) for 24 hours; we then washed them, seeded them on gelatin-coated Petri dishes, and cultured them at 37°C in a humidified atmosphere with 5% CO2. The MSCs were isolated on the basis of their ability to adhere to the culture plate. Hematopoietic and other nonadherent cells were removed twice by a medium change on days 52 and 54. Prior to cell transplantation, adherent cells were detached by trypsin-EDTA, passaged twice, and labeled with 30-μl/ml Vybrant cell-labeling solution (1,1′-dioctadecyl-3,3,3′,3′,-tetramethylindocarbocyanine perchlorate [DIL]; Invitrogen, Paisley, UK) for 30 minutes under light protection at 37°C, followed by 2 washes with PBS. DIL is a red lipophilic fluorochromatic dye which accumulates in the membrane of rough endoplasmic reticulum. 8 Cells were further passaged twice, harvested, and used for cell transplantation.
CM Injection and Stem Cell Transplantation
On day 57, thoracotomy was performed in animals of groups 3 and 4, under general anesthesia through the left intercostal space to expose the heart. The free lateral wall of the left ventricle (LV) was identified, and either 1-ml pure CM (group 3) or 1-ml CM containing 1.5–2.0 × 106 stem cells/ml was injected transepicardially into the myocardium in a circular manner at 4 locations (diameter of the injection area, ∼1.5 cm) within 2 minutes. A thoracic drain was inserted before closing the chest, and the rabbits were treated with antibiotics and analgesics.
Clinical Observations
Clinical observations together with cardiac auscultation were performed regularly.
Echocardiographic Studies
Echocardiograms were obtained on days 0, 42, 57, and 84. Results were evaluated by statistical methods.
Gross Examination
On day 84, animals in all groups were euthanized by heart explantation under general anesthesia. The hearts were inspected grossly, and transverse sections were made through the injection area. A representative section was fixed in 10% buffered formol saline. One section was snap-frozen in liquid nitrogen and stored at −80°C for mRNA analyses.
Histologic Analysis
Formalin-fixed specimens were embedded routinely in paraffin wax, sectioned at 3 μm, and stained with HE. Picrosirius red–stained slides 9 were used for fibrillar collagen measurements. For representative measurements, heart cross-sections were divided into equal areas (0.12 cm2) (LV wall, n = 5; right ventricular [RV] wall, n = 5; interventricular septum [IVS], n = 5) and digitized. The mean value of collagen deposition was generated. The area (percentage) of extracellular red-stained collagen fibers was computed from the fields, providing a composite measure of interstitial, perivascular, and replacement fibrosis. Additionally, investigations of picrosirius red–stained slides were viewed with polarized light to demonstrate the collagen types I (yellow-red) and III (green). 37 Deparaffined unstained slides were examined by fluorescence microscopy to identify the DIL-labeled stem cells.
Immunohistochemistry
Using the peroxidase-antiperoxidase (PAP) technique, serial sections were immunolabeled with the following primary mouse monoclonal antibodies: MMP-1 (Chemicon, MAB 3307, Hofheim, Germany), MMP-2 (Chemicon, MAB 3308, Hofheim, Germany), MMP-3 (Chemicon, MAB 3312, Hofheim, Germany), MMP-9 (Chemicon, MAB 3309, Hofheim, Germany), TIMP-2 (Lab Vision, MS1485, Fremont, California), and TIMP-3 (Chemicon, MAB 3318, Hofheim, Germany). The secondary antibody was rat anti-mouse immunoglobulin G (Dianova, Hamberg, Germany), and mouse PAP complex (Dako, Hamberg, Germany) was used. Diaminobenzidinehydrochloride (Sigma-Aldrich Chemie, München, Germany) was used as chromogen, and slides were counterstained with Papanicolaou's stain (Merck, Darmstedt, Germany). Negative controls were obtained by substituting irrelevant mouse antibody as the primary antibody.
All slides were read blind. The staining intensity of the cardiomyocytes was evaluated semiquantitatively and graded as follows: (±) minimal, (+) mild, (++) moderate, and (+++) marked. Fibrocytes with a positive or negative signal were counted digitally (Imaging System 3.2, Olympus DP-Soft, Olympus, Hamburg, Germany). For a representative measurement, 5 high-power fields (each 0.1 mm2) excluding the injection zone were randomly chosen from the LV, RV, and IVS. The mean number of cells counted in these 5 fields was generated.
Assay of mRNA Profiles
To assess the mRNA expression of MMP-1, -2, -3, -9 and TIMP-2, -3, we performed real-time polymerase chain reaction (PCR). Frozen tissue samples of the RV, LV, and IVS from 6 animals in each group were prepared with an Ultra-Turrax (IKA Werke, Detmold, Germany) using Trizol Reagent (Invitrogen, Karlsruhe, Germany). An aliquot of total RNA (500 ng) was reversely transcribed into cDNA using a Sensiscript Kit (Qiagen, Hilden, Germany) and random primers (Invitrogen) according to the instructions of the manufacturers. To quantify mRNA expression levels, we used 1-μl aliquot of the cDNA in real-time PCR containing the gene-specific primers (Table 2) and LightCycler FastStart DNA Master SYBR-Green I reaction mix (Roche Diagnostics, Mannheim, Germany). The specific expression levels were analyzed using LightCycler software and expressed in relation to the individual 18S rRNA intensity in relative units. Each PCR was performed in triplicate with a variability <10%. PCR conditions for each of the MMP and TIMP mRNA were as follows: annealing temperature 60°C and elongation time 30–40 seconds at 21–40 PCR cycles.
Sequences of primers for MMPs and TIMPs in real time PCR.∗
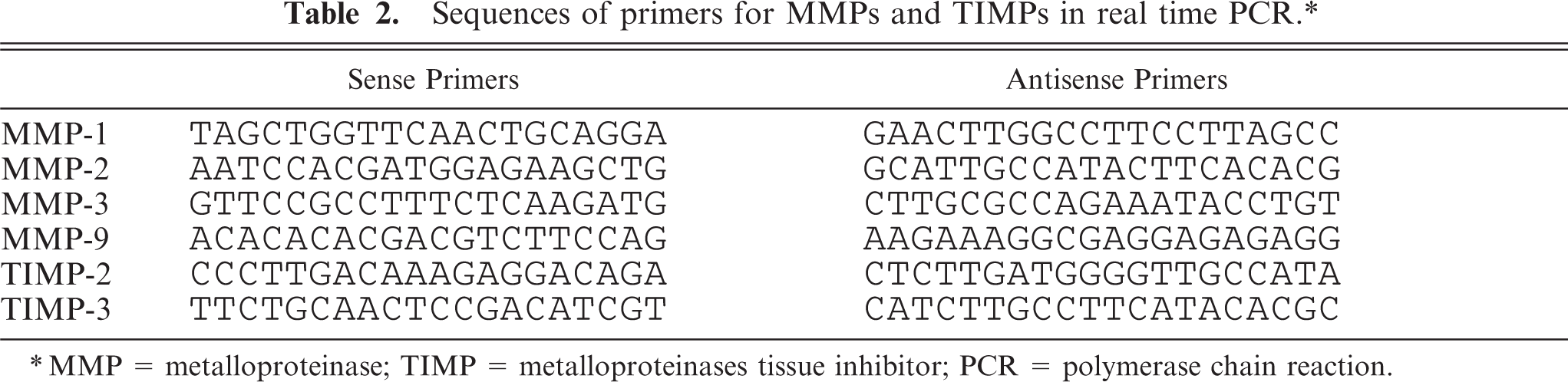
∗MMP = metalloproteinase; TIMP = metalloproteinases tissue inhibitor; PCR = polymerase chain reaction.
Statistical Analysis
Statistical analyses included the Shapiro-Wilk test and 1-way analysis of variance. For post hoc analyses, the Levene and Bonferroni tests were applied. A paired t-test was performed as appropriate. Results were considered statistically significant if p ≤ .05. All data analyses were performed by using SPSS software, Version 11.5 (SSPS Inc. Headquarters, Chicago, IL, USA).
Results
Clinical Observations
Doxorubicin administration caused clinical signs of heart failure (cyanosis, dyspnea) from treatment day 20 on, which was confirmed by echocardiography on day 42. Serial echocardiography detected a decline in the ejection fraction and fractional shortening in doxorubicin-failing hearts (groups 2–4) compared with those in the untreated controls (group 1). MSC injection (group 4), but not CM injection (group 3), resulted in a significantly better heart function as reported previously. 7
Gross Findings
Macroscopically, moderate LV dilation was seen in all doxorubicin-treated animals. The injection sites of CM or MSCs were characterized by focal mild-to-moderate epicarditis (Fig. 1).
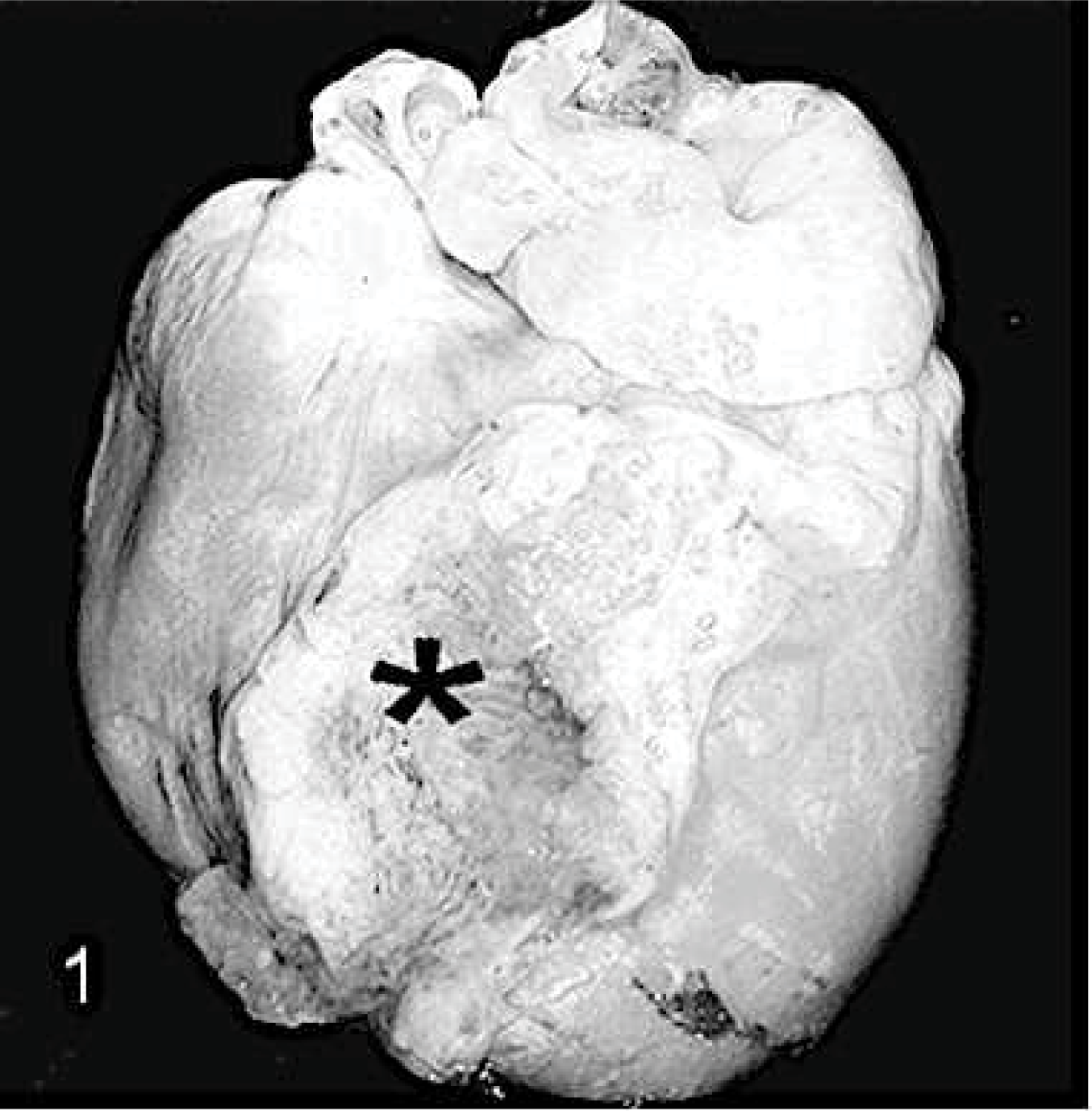
Heart; rabbit No. 28, group 4. Macroscopically, focal epicarditis (asterisk) on the left ventricle is present in the area of transepicardial stem cell injection.
Histopathologic Findings
The hearts of the untreated controls (group 1) were normal: The epicardium was composed of only a few type I collagen fibers and several adipocytes around coronary vessels. Cardiomyocytes were arranged in the typical pattern with few intermingled type I collagen fibers and single type III collagen fibers. Fibrocytes were characterized by slender hyperchromatic nuclei and small amounts of cytoplasm. Interstitial and perivascular collagen fibers (Fig. 2) constituted ∼7% of the myocardial area (Table 3), and fibrocytes numbered 65 ± 10 per high-power field. The endocardium consisted of endothelial cells with few underlying type I collagen fibers.
Area of myocardial type I and III collagen fibers(mean ± SD) in the different groups and heart locations.
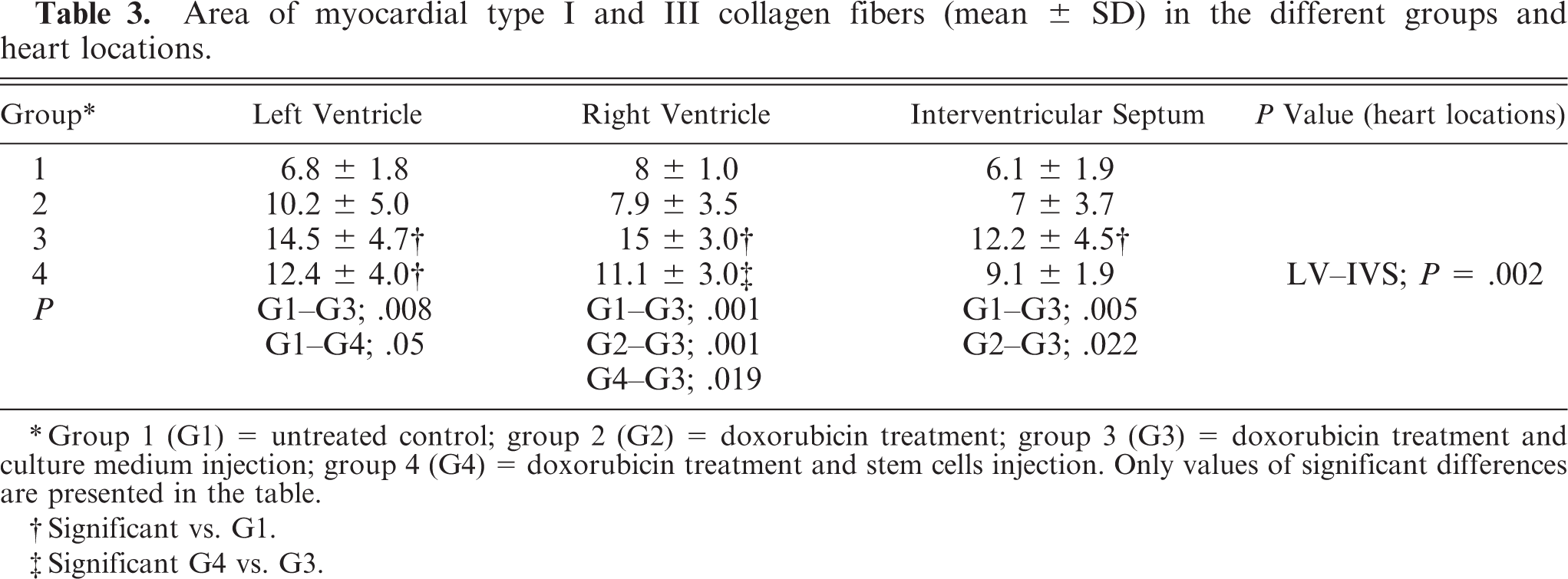
∗Group 1 (G1) = untreated control; group 2 (G2) = doxorubicin treatment; group 3 (G3) = doxorubicin treatment and culture medium injection; group 4 (G4) = doxorubicin treatment and stem cells injection. Only values of significant differences are presented in the table.
†Significant vs. G1.
‡Significant G4 vs. G3.
Intensity of expression of MMPs and TIMPs in the cardiomyocytes(exclusive to the injection site) in the different groups detected by immunohistochemistry.∗

∗Staining intensity in cardiomyocytes: – negative; (±) = minimal; + = mild; ++ = moderate; +++ = marked.
†Group 1 = untreated control; group 2 = doxorubicin treatment; group 3 = doxorubicin treatment and culture medium injection; group 4 = doxorubicin treatment and stem cells injection.
‡MMP = metalloproteinase; TIMP = metalloproteinase tissue inhibitor.
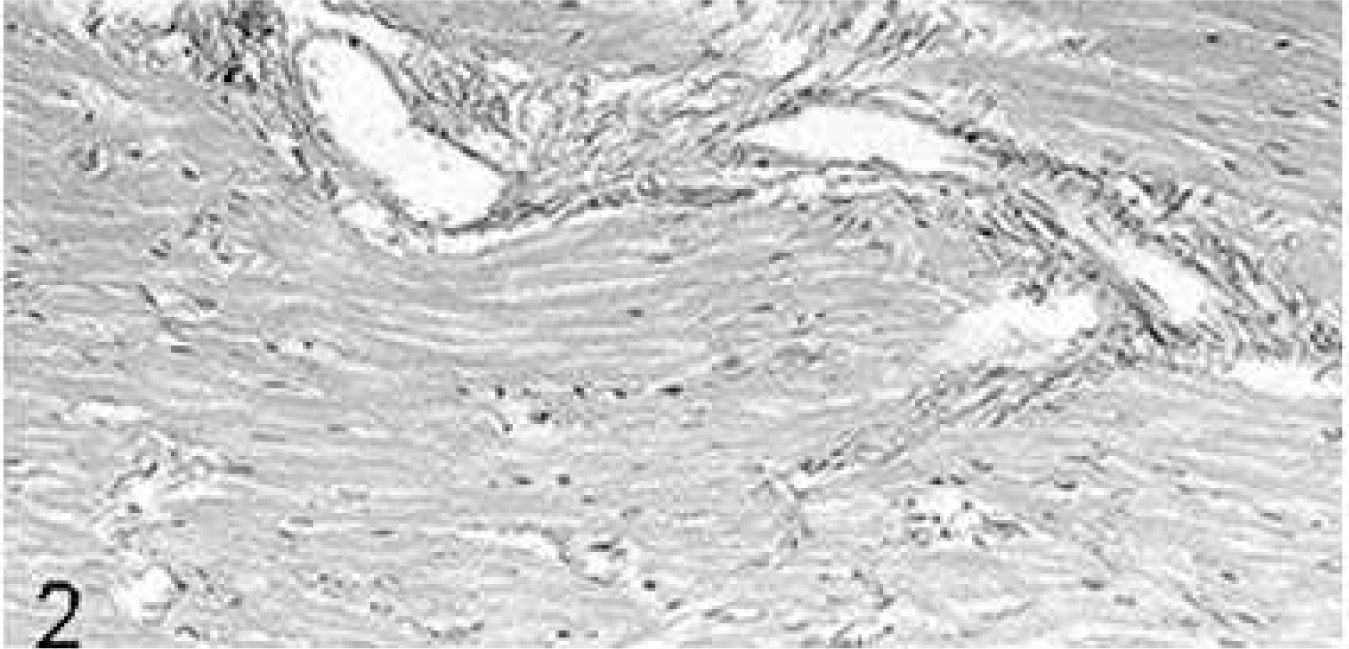
Heart; rabbit No. 1, group 1. In the untreated controls, intramural coronary vessels are surrounded by normal small amounts of collagen fibers. Picrosirius red stain.
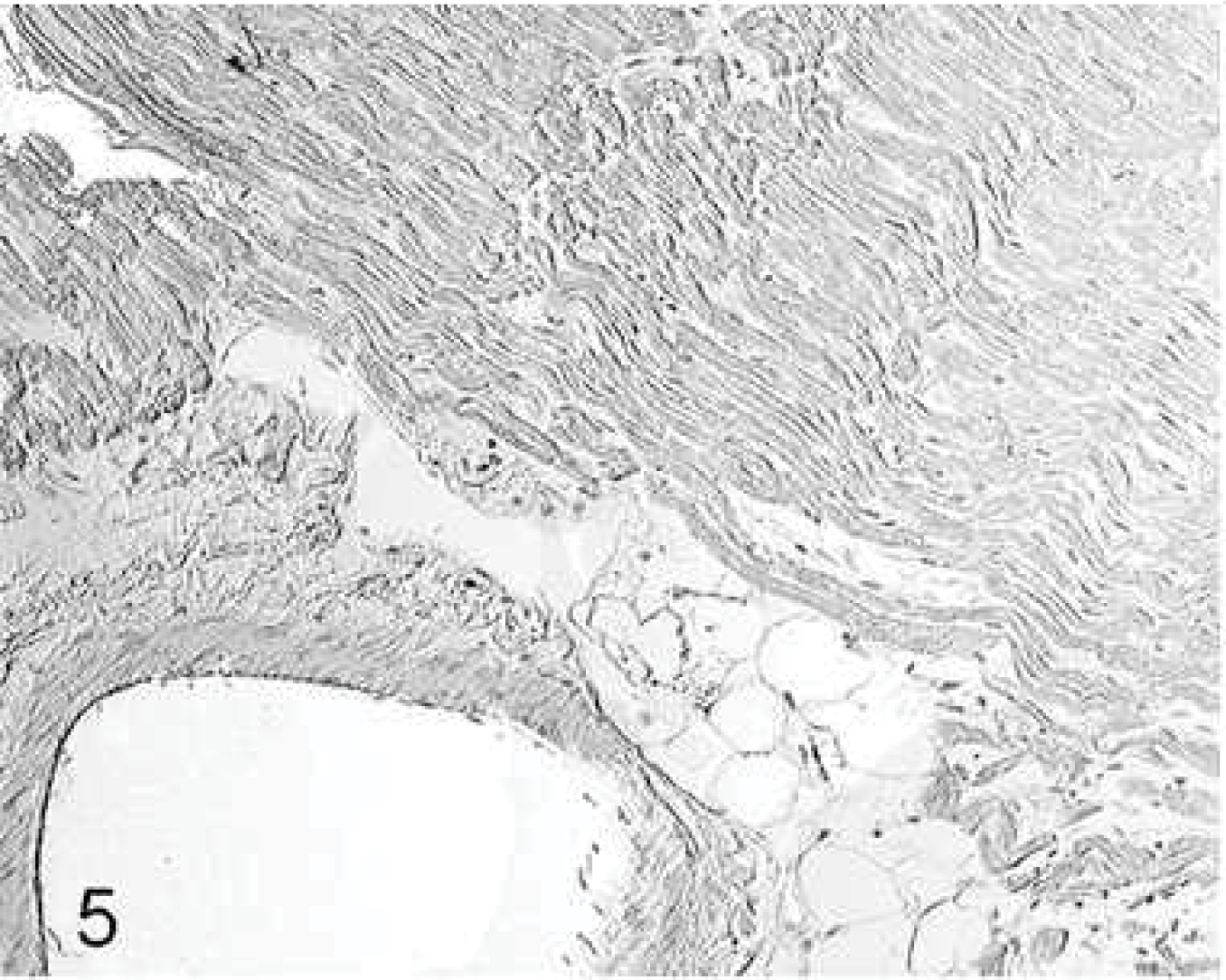
In the hearts of all doxorubicin-treated animals (groups 2–4), single lymphocytes and macrophages were distributed in the interstitium. Mild angiosclerosis was apparent in some coronary vessels. In group 2, type I and III collagen fibers were not significantly (P = .852) increased compared with those in the untreated controls (group 1). In group 3, collagen fibers were mainly perivascular (Fig. 3), significantly increased compared with those in group 1. In group 4, the collagen content was significantly increased in the LV only. In the RV, the fibrotic area in group 3 was significantly greater than that in group 4 (Table 3). The number of cardiac fibrocytes (70 ± 5) was not significantly different among the groups.
Heart; rabbit No. 17, group 3. Injection of culture medium induces marked perivascular fibrosis spreading into the surrounding myocardium. Picrosirius red stain.
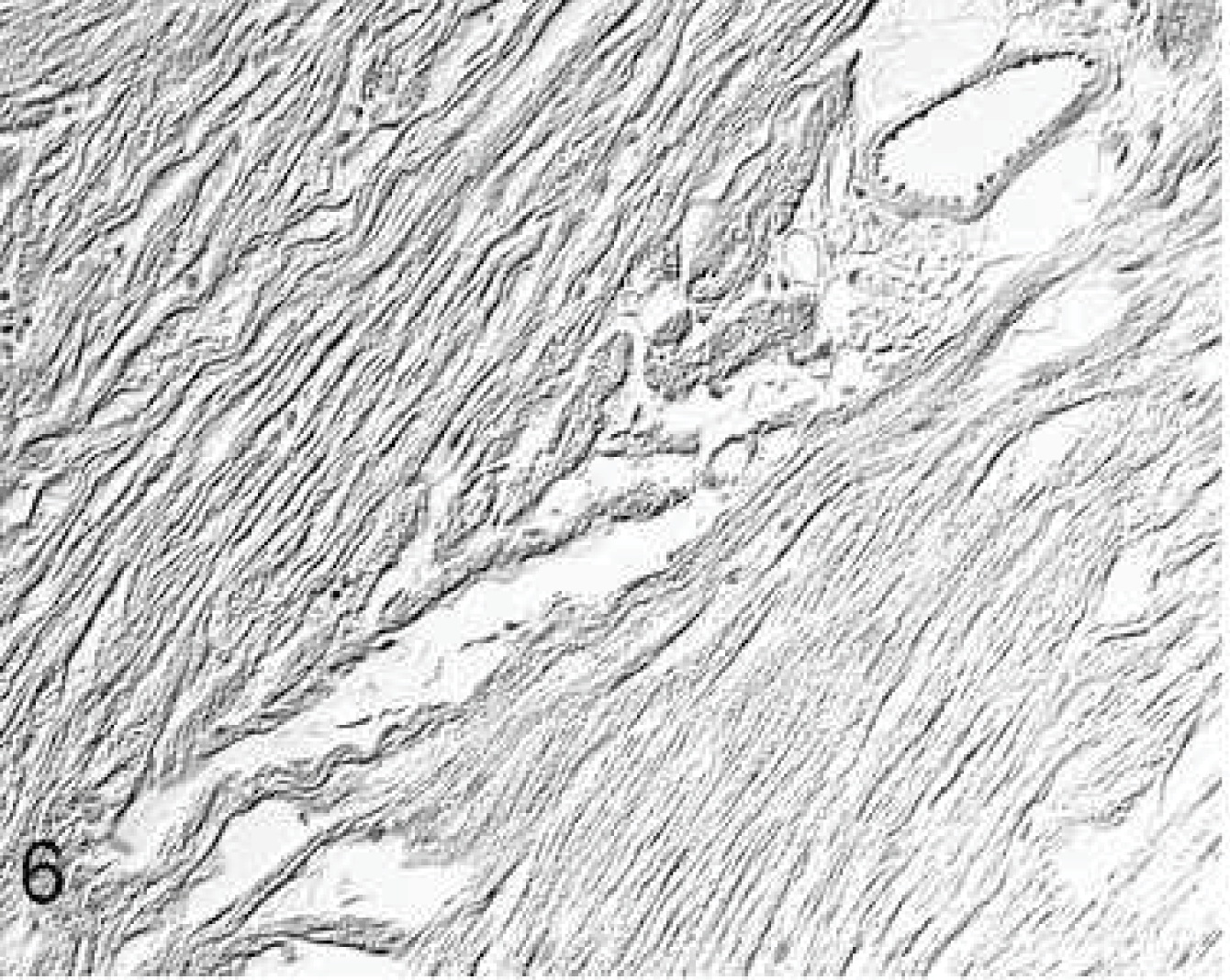
At injection sites, focal mild-to-moderate chronic fibroblastic inflammation was noted in the epicardium (group 3: mild, n = 3; moderate, n = 6; group 4: mild, n = 9; moderate, n = 6). Well-demarcated fibrotic foci extending from the epicardium into the myocardium were interpreted as injection canals (Fig. 4). These were mainly composed of type III collagen fibers, with ∼10% type I collagen fibers, large spindle-shaped fibroblasts, a few macrophages and lymphocytes, and single neutrophilic granulocytes.
Heart; rabbit No. 21, group 3. The injection site is characterized by focal fibroblastic epicarditis and a focal myocardial scar extending from the epicardium into the myocardium. Picrosirius red stain.
Stem cells could not be identified in HE-stained slides. Using fluorescence microscopy, we saw single large cells (1–2 cells/high-power field), with oval nuclei showing a specific intracytoplasmic signal deriving from DIL loading in the sites of MSC injection (group 4); however, from their morphology, it was unclear whether they were original MSCs or whether they were macrophages with phagocytosed remnants of MSCs. In the sites of CM injection (group 3), no cells with a specific intracytoplasmic signal were identified.
Immunohistochemical Findings
The group-specific findings of MMP and TIMP expression in cardiomyocytes and MMP-2 and TIMP-3 expression in fibrocytes are presented in Tables 4– 7. In all groups, the staining intensity of MMPs and TIMPs in cardiomyocytes was slightly higher in the RV than in the LV or IVS. Only TIMP-3 expression in fibrocytes was significantly different among the RV, LV, and IVS in most groups (Table 6). MMP-1 and MMP-9 expression was present in cardiomyocytes, but not in fibrocytes, endothelia, and vascular smooth muscle cells. MMP-2, TIMP-2, and TIMP-3 were expressed in cardiomyocytes as well as in fibrocytes, endothelia, and vascular smooth muscle cells. Macrophages and neutrophilic granulocytes were positive for MMP-9 and TIMP-3, and lymphocytes expressed TIMP-3.
Metalloproteinase-2 expressing myocardial fibrocytes (%) exclusive to the injection sites in the different groups and heart locations (mean ± SD).
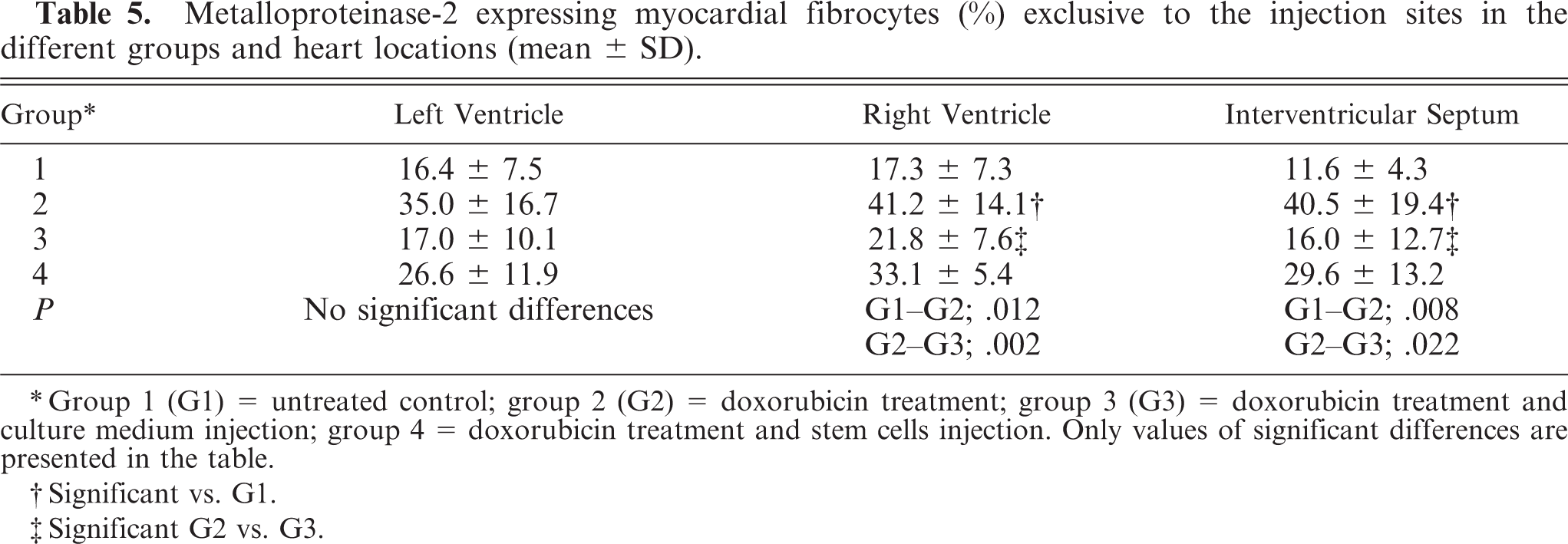
∗Group 1 (G1) = untreated control; group 2 (G2) = doxorubicin treatment; group 3 (G3) = doxorubicin treatment and culture medium injection; group 4 = doxorubicin treatment and stem cells injection. Only values of significant differences are presented in the table.
†Significant vs. G1.
‡Significant G2 vs. G3.
Metalloproteinase tissue inhibitor-3 expressing myocardial fibrocytes (%) exclusive to the injection site in the different groups and heart locations (mean ± SD).
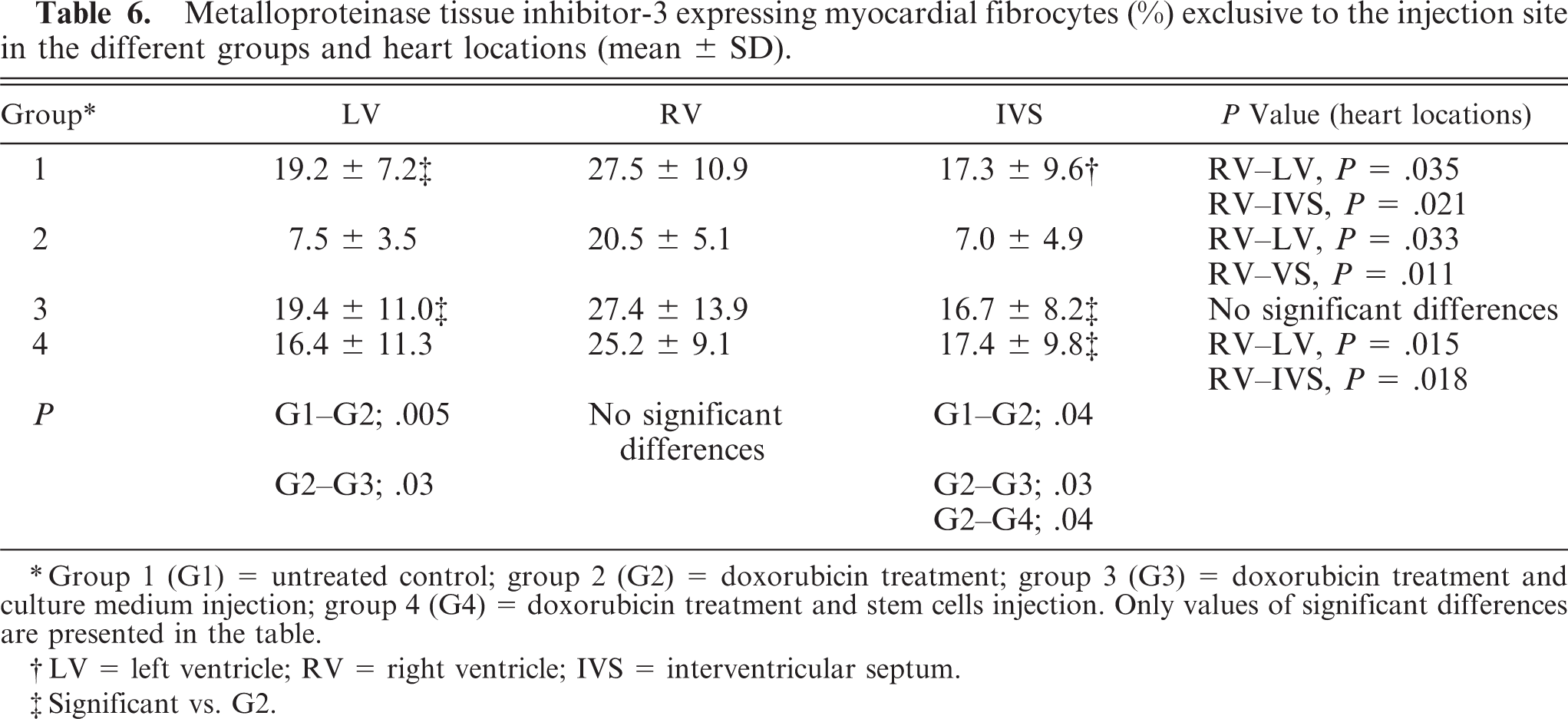
∗Group 1 (G1) = untreated control; group 2 (G2) = doxorubicin treatment; group 3 (G3) = doxorubicin treatment and culture medium injection; group 4 (G4) = doxorubicin treatment and stem cells injection. Only values of significant differences are presented in the table.
†LV = left ventricle; RV = right ventricle; IVS = interventricular septum.
‡Significant vs. G2.
Immunohistochemical detection of MMP and TIMP expression in the epicardial and myocardial fibrocytes at the injection sites of culture medium(group 3) and stem cells (group 4).

∗MMP = metalloproteinases; TIMP = metalloproteinases tissue inhibitors. (+) = <10% of fibrocytes are positive; + = <20% of fibrocytes are positive; ++ = <50% of fibrocytes are positive; +++ = >50% of fibrocytes are positive; − = all fibrocytes are negative.
Group 1: In the untreated controls, MMP-1 (Fig. 5) and MMP-9 (Fig. 6) expression in cardiomyocytes was mild and diffuse (Table 3). MMP-2 was minimally expressed in perivascular cardiomyocytes and was also mildly expressed in ∼15% of the fibrocytes (Table 5). TIMP-2 and TIMP-3 (Fig. 7) were mildly and moderately, respectively, expressed in all cardiomyocytes and moderately expressed in ∼20% of fibrocytes (Table 6).
Heart; rabbit No. 4, group 1. In untreated controls, expression intensity of metalloproteinase-1 (MMP-1) in cardiomyocytes is minimal and absent in fibrocytes and vessels. Immunohistochemistry, MMP-1, Papanicolaou's counterstain.
Heart; rabbit No. 4, group 1. In untreated controls, metalloproteinase-9 (MMP-9) expression in cardiomyocytes is mild and absent in fibrocytes and vessels. Immunohistochemistry, MMP-9, Papanicolaou's counterstain.
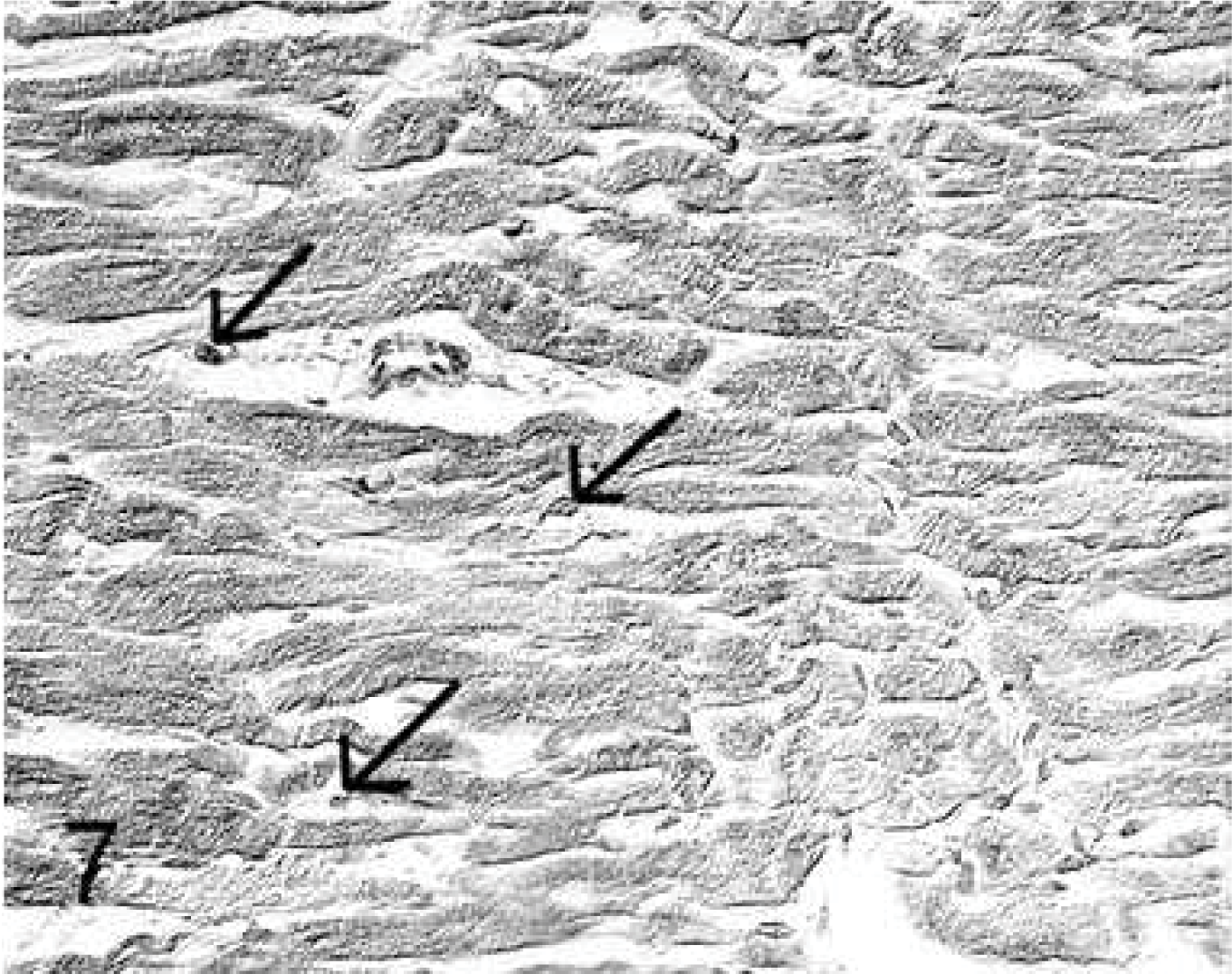
Heart; rabbit No. 6, group 1. In untreated controls, expression of metalloproteinase tissue inhibitors-3 (TIMP-3) in cardiomyocytes is mild but intense in numerous fibrocytes (arrows) and in endothelia. Immunohistochemistry, TIMP-3, Papanicolaou's counterstain.
Group 2: Doxorubicin treatment resulted in mildly increased expression of MMP-1 and MMP-2 in perivascular cardiomyocytes. In contrast to the untreated controls (group 1), marked MMP-9 expression was seen diffusely in cardiomyocytes. MMP-3 expression was limited to a few small foci of the myocardium. When compared with all groups, MMP-2-positive fibrocytes (∼40%) were increased, and TIMP-3-expressing fibrocytes (∼10%) were decreased.
Group 3: Injection of CM induced moderate MMP-1 and MMP-2 expression intensity in perivascular cardiomyocytes, while MMP-9 was marked in all cardiomyocytes. Compared with group 2, TIMP-3 staining in cardiomyocytes was increased and ∼20% of fibrocytes expressed TIMP-3 and MMP-2.
Group 4: MSC injection resulted in marked MMP-1 (Fig. 8) and MMP-2 expression in perivascular cardiomyocytes, and ∼30% of fibrocytes expressed MMP-2. MMP-9 expression was also marked and was noted in all cardiomyocytes (Fig. 9). Moderate expression of TIMP-2 and TIMP-3 was also seen in all cardiomyocytes and in ∼20% of fibrocytes.
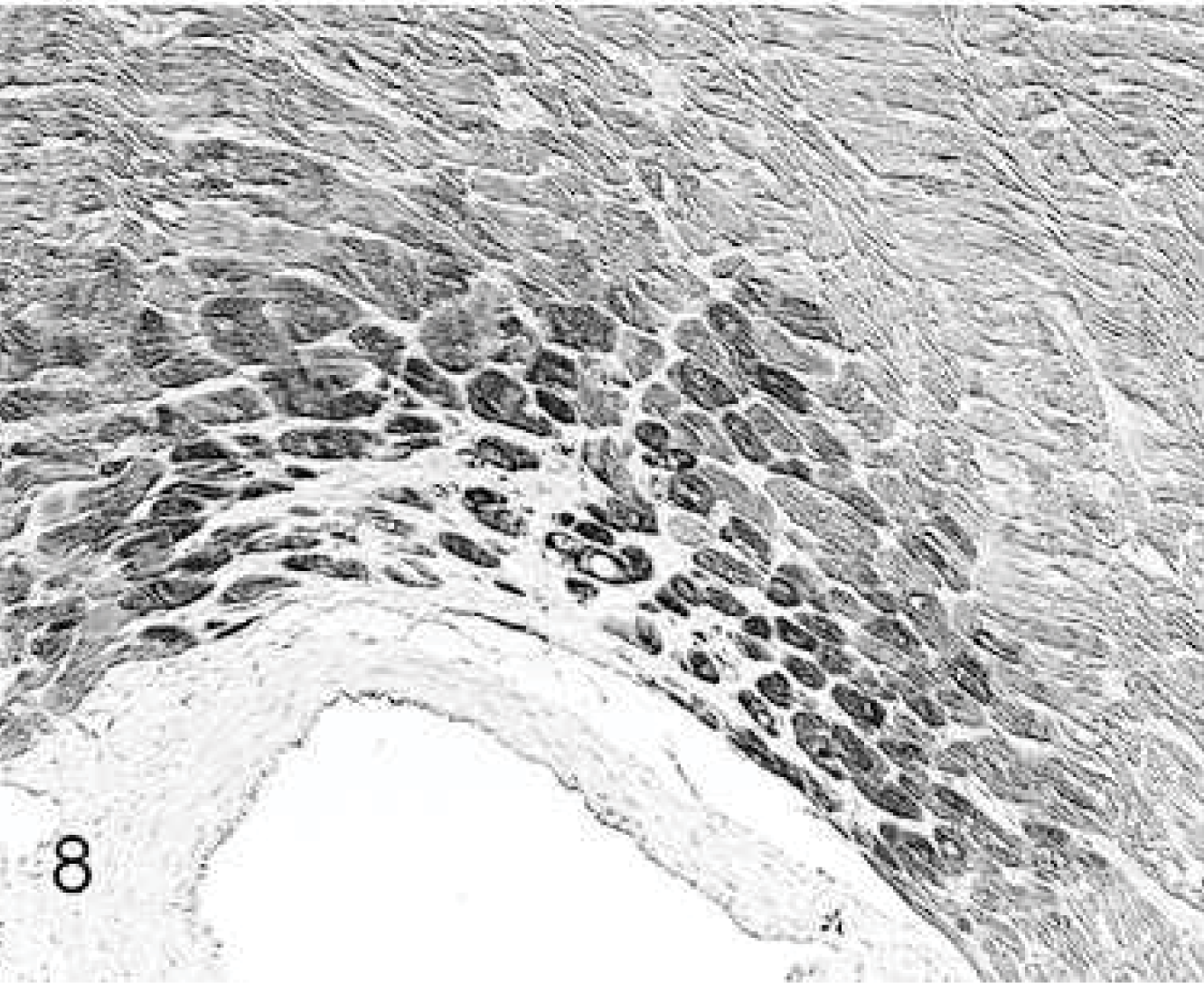
Heart; rabbit No. 29, group 4. After stem cell injection, the expression intensity of metalloproteinase-1 (MMP-1) is markedly increased in perivascular cardiomyocytes. Immunohistochemistry, MMP-1, Papanicolaou's counterstain.
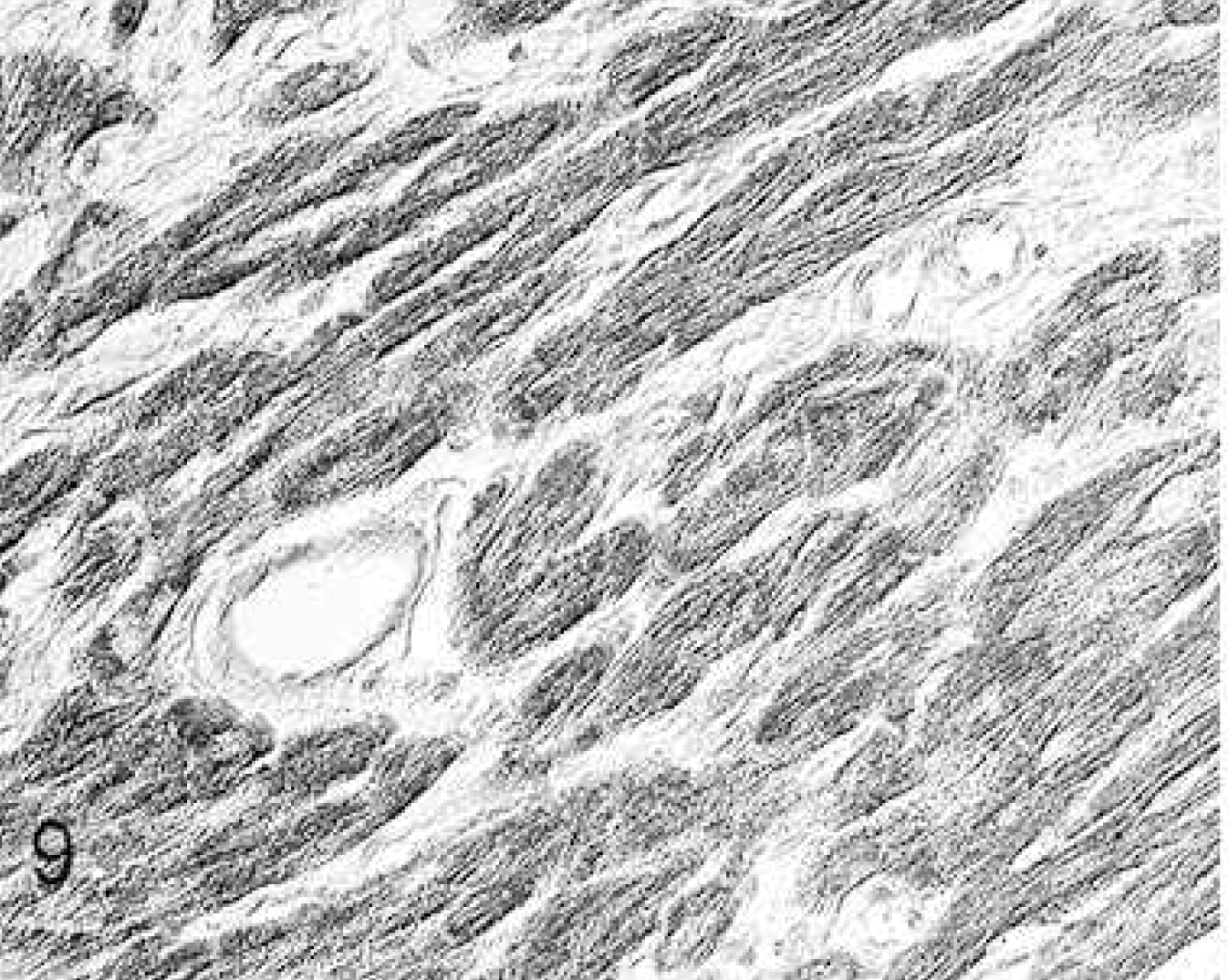
Heart; rabbit No. 29, group 4. After stem cell injection, the expression of metalloproteinase-9 (MMP-9) in all cardiomyocytes is moderately increased, but fibrocytes and vessels are negative. Immunohistochemistry, MMP-9, Papanicolaou's counterstain.
Injection sites (groups 3 and 4): In general, in cardiomyocytes bordering the injection site, there was an increased expression of MMP-1 and MMP-2 compared with the remaining myocardium. A greater number of MMP-1- and MMP-2-positive fibrocytes was seen in CM injection sites than in MSC injection sites (Table 7). Furthermore, all fibrocytes within the injection sites expressed TIMP-2 and TIMP-3 (Fig. 10) intensively.
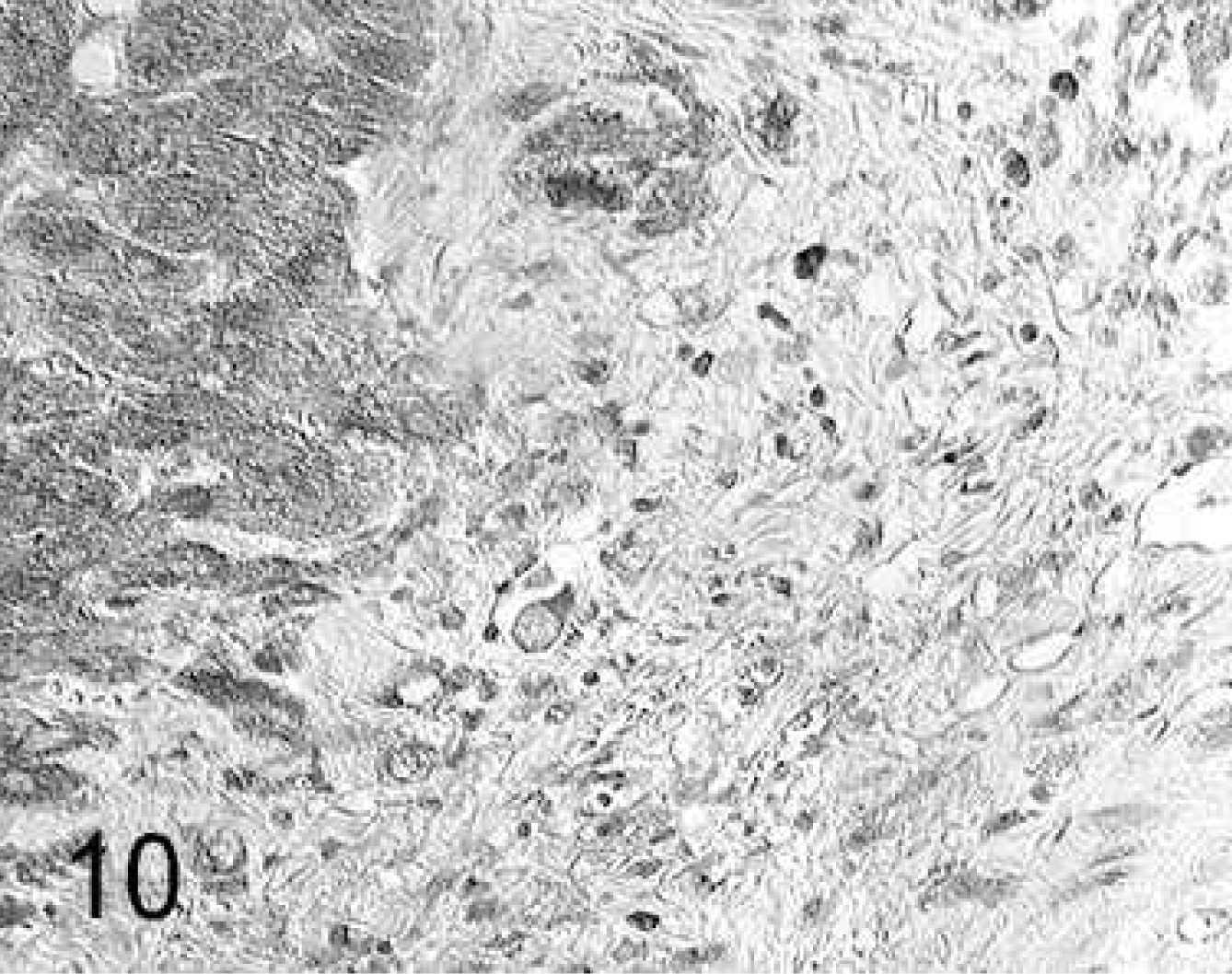
Heart; rabbit No. 18, group 3. In the culture medium injection site, the fibroblasts express metalloproteinase tissue inhibitor-3 (TIMP-3) intensively, and in the periphery of the scar, the expression intensity in cardiomyocytes is increased. Immunohistochemistry, TIMP-3, Papanicolaou's counterstain.
On the basis of the semiquantitative and statistical results, it can be summarized that doxorubicin treatment results in minimally increased myocardial fibrosis but marked increase of MMP-1, -2 and -9 and decreased TMP-3 expression.
Injection of either CM or MSCs increased myocardial fibrosis. CM injection resulted in increased TIMP-3 expression and a higher degree of fibrosis. MMP expression was similar compared with that in group 2, whereas MSC injection led to a higher MMP-1 and MMP-2 and normal TIMP-3 expression.
mRNA Assays
The mRNA values for MMP-3 were very low compared with the values of other MMPs and TIMPs. All mRNA levels of MMPs (Figs. 11, 12) and TIMPs (data not shown) were not significantly different among the 4 groups or among the 3 locations of the heart (RV, LV, and IVS) (data not shown). No correlation between the mRNA levels and the immunohistochemical findings, which showed marked group-specific differences, could be detected.
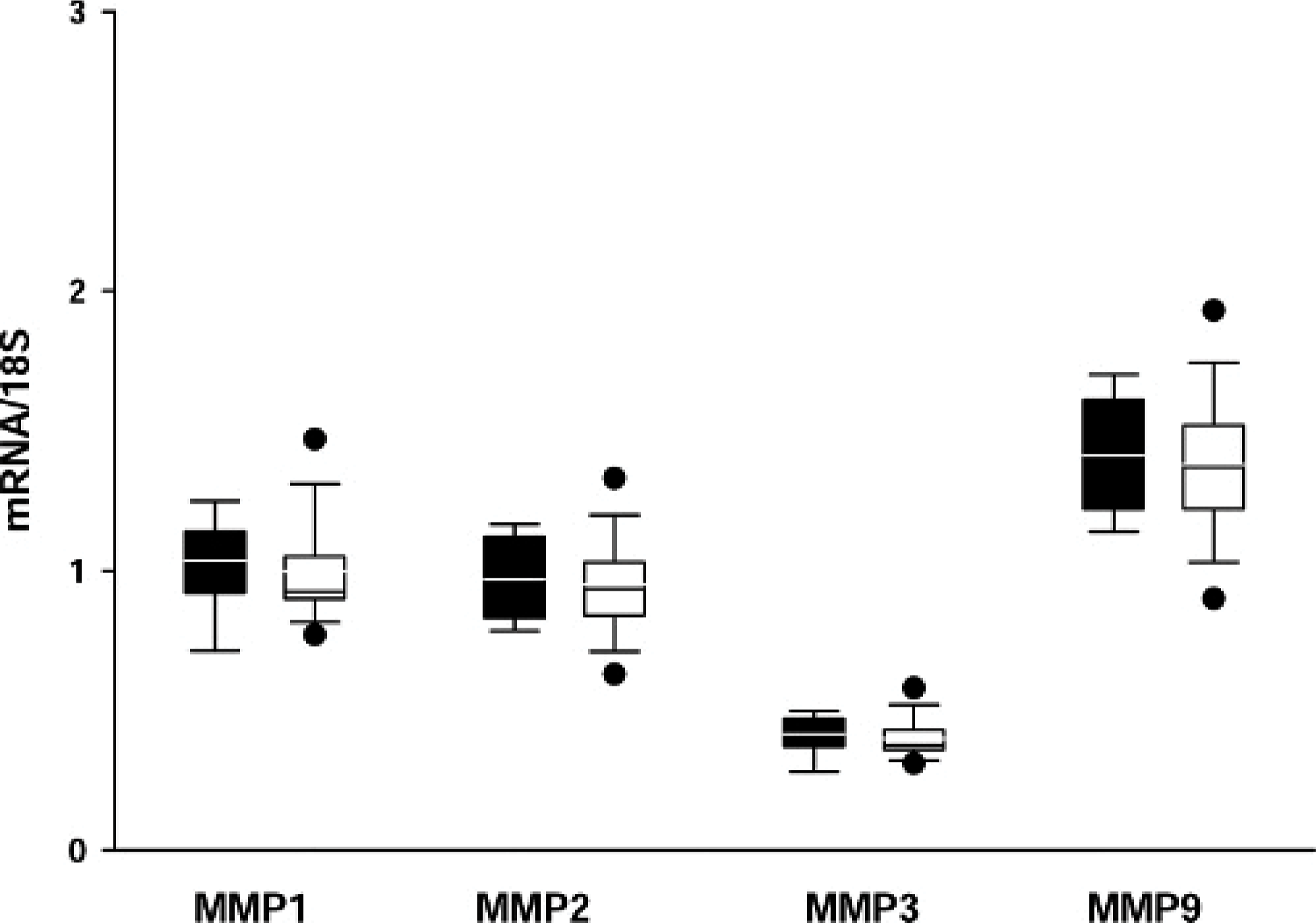
Transcription levels of metalloproteinases (MMPs) in the left ventricle (LV), group 1 (white) compared with group 2 (black). MMP-3 levels are markedly lower than other MMP levels; no significant differences are seen in the transcription levels of all MMPs between the untreated controls (group 1) and the doxorubicin-treated animals (group 2).
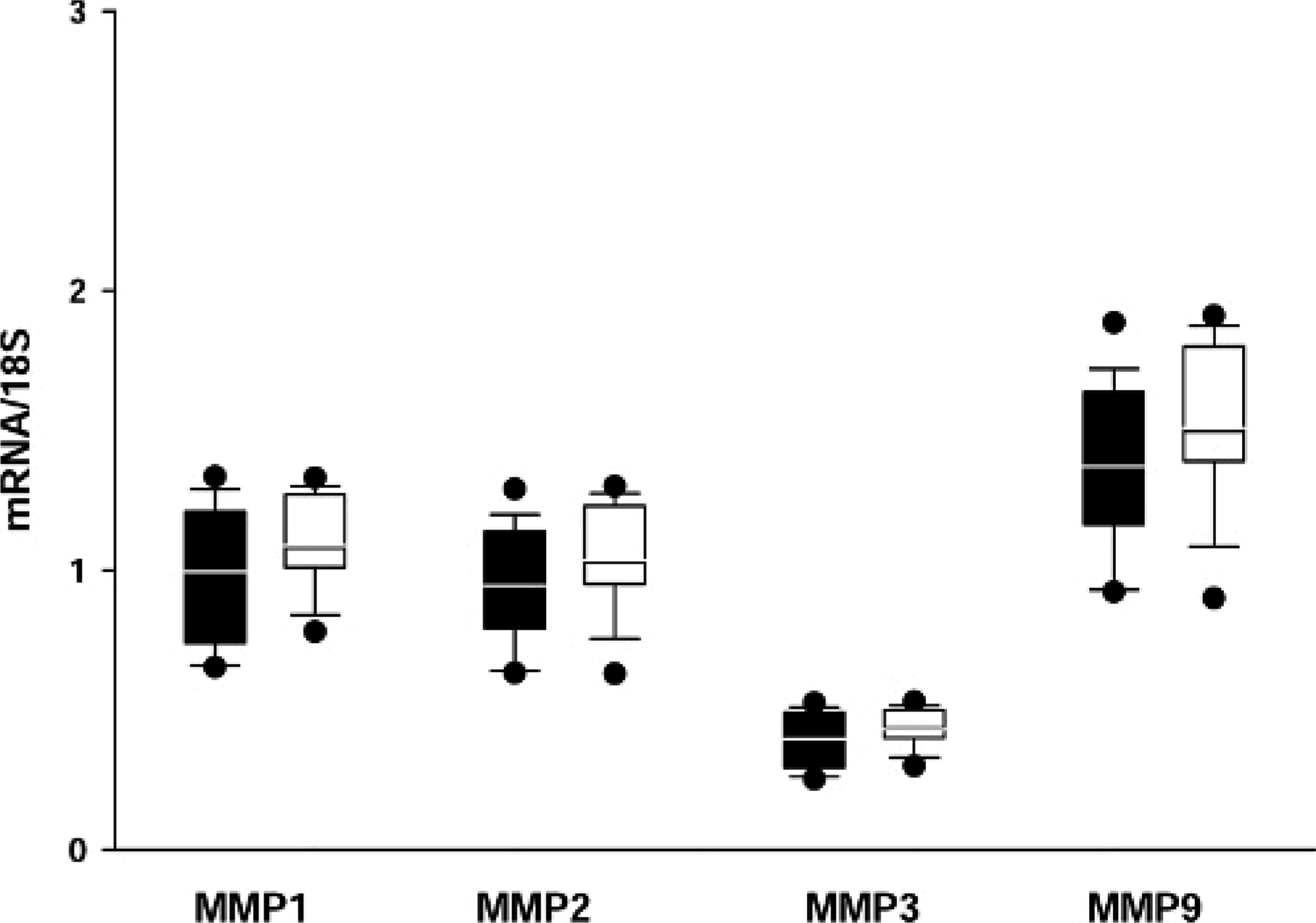
Transcription levels of metalloproteinases (MMPs) in the left ventricle (LV), group 3 (white) compared with group 4 (black). MMP-3 levels are markedly lower than other MMP levels; no significant differences are seen in all MMPs among the rabbits with culture medium injection (group 3) and stem cell injection (group 4).
Discussion
In this study, we investigated first the expression of MMPs and TIMPs in chronic doxorubicin cardiomyopathy in a rabbit model and, second, the effects of stem cell injection in this disease.
In accordance with the literature, 33, 36 in normal hearts (group 1), the expression intensity of MMPs and TIMPs was very mild. Cardiomyocytes were positive for MMP-1, -2, -9 and TIMP-2 and -3, whereas in fibrocytes, MMP-2 and TIMP-2 and -3 expression was detected. Endothelia expressed MMP-2 and TIMP-2 and -3.
Successful induction of heart failure in the doxorubicin-treated rabbits was confirmed by echocardiographic data 7 and ß-receptor analysis. 5 Corresponding to the literature, 13, 34 collagen fibers were minimally increased mainly in perivascular areas in doxorubicin cardiomyopathy (group 2). In vitro studies in rat heart–derived embryonic myocytes (H9c2) showed that doxorubicin-induced a rapid increase of MMP-2 and MMP-9 expression and activation. 29 This was confirmed in vivo by our rabbit model. MMP-1, MMP-2, and MMP-9 were increased in cardiomyocytes, and the number of TIMP-3-positive fibrocytes decreased.
Myocardial fibrosis was increased after CM injection (group 3) and MSC injection (group 4). Interestingly, the fibrosis of the RV was significantly higher in group 3 than in group 4, although injection was into the LV. The relationship of this diffuse fibrosis to the surgical intervention and local injection lesion is not readily explained. MSC injection, however, resulted in less myocardial fibrosis than injection of CM alone and was accompanied by higher perivascular MMP-1 and MMP-2 and lower fibrocyte TIMP-3 expression.
Significant differences among the groups were detected only by immunohistochemistry and not by PCR, possibly due to the fact that the investigations were carried out in the chronic phase of the disease. Post-transcriptional mechanisms controlling the protein translation rate, the half-life of specific proteins or mRNAs, and the intracellular location and molecular association of the protein products of expressed genes may be insufficiently sensitive to predict protein expression levels from quantitative mRNA data. Similar difficulties, reported by Peterson et al., 21 were that mRNA levels and total amounts of protein were not comparable in all cases. In our study, the clinical findings correlated with the morphologic and immunohistochemical differences among the groups but not with the PCR results.
We conclude that there are marked differences of extracellular matrix metabolism between medium and stem cell injection groups at the local injection site as well as in the whole heart. In MSC injection sites, local MMP-1 and -2 expression of fibrocytes was less intensive than in CM injection sites, while TIMP-2 and -3 expression in fibrocytes was intensive in the injection sites in both groups; however, the repair process in CM injection sites seemed more active than that in stem cell injection sites. To our knowledge, no comparable studies have been reported to date. Further investigations using in situ zymography to identify the cells that are involved in the remodeling processes are indicated.
Only a few cells were identified as integrated original stem cells or more likely phagocytosed debris of MSC so that it is unlikely that MSCs had a direct effect on the heart overall. While Agbulut et al. 1 identified single stem cells from 2 days up to 2 weeks after injection of unpurified BMCs in acute doxorubicin cardiomyopathy in transgenic mice, Scorsin et al. 28 found no evidence of transplanted cell survival over the follow-up period, although they observed an improvement of fractional shortening by echocardiography 1 month after transplantation of fetal cardiomyocytes into doxorubicin-treated hearts. As discussed in other studies, paracrine mechanisms appear to be more likely than direct effects of stem cells. 20, 23 Dhein et al. 5 demonstrated that down-regulation of ß-adrenoceptors was attenuated, and noradrenaline and adrenaline plasma levels were normal in the stem cell–treated rabbits of our study. To our knowledge, the effects of stem cell injection on the extracellular matrix metabolism have not been investigated in detail in any other study. MMPs are regulated at multiple levels, including transcription, translation, secretion, and activation. 17 Plasmin, TGFß1, TNFa, and IL-1ß are some examples for the mediators that are involved in the MMP regulation; 16 however, it remains unclear which are secreted by the stem cells and how the beneficial effects are induced.
In conclusion, doxorubicin cardiomyopathy is characterized by minimally increased myocardial fibrosis but a marked increase of MMP-1, -2, and -9 and decreased TMP-3 expression. MSC injection improved clinical heart function 7 and ß-receptor expression 5 but induced myocardial fibrosis and increased MMP expression. In contrast, injection of CM resulted not in better heart function 7 but in a higher degree of myocardial fibrosis and TIMP-3 expression than MSC injection did. A relevant number of stem cells could not be identified after 4 weeks; thus, paracrine mechanisms of stem cells in doxorubicin cardiomyopathy have to be considered.
Footnotes
Acknowledgements
This work was supported by the German Heart Foundation “Deutsche Stiftung für Herzforschung” grant F/08/03 held by Stefan Dhein. We thank K. Schneider and C. Ullmann (Experimental Laboratory Department of Cardiac Surgery, Heart Center, Leipzig, Germany), A. Richter (statistics), and J. Kacza (Morphometry, Veterinary Anatomy).
