Abstract
Dilated cardiomyopathy (DCM), the most common form of cardiomyopathy in the dog, most often occurs in certain breeds. The objective of this study was to describe a rapidly progressive form of DCM that has been recently recognized in juvenile Toy Manchester Terrier dogs (TMTs). The clinical history and gross findings were reviewed in a group of 14 TMTs, and histologic sections of heart were examined in 12 of those 14 TMTs with DCM. Histochemical and histomorphometric analyses were employed to compare the heart in TMTs affected by DCM with that of control dogs. TMTs ranged in age from 10 to 58.3 weeks, with males and females being equally affected. Affected TMT hearts contained foci of degeneration and loss of myofibers with fibrosis and mild lymphoplasmacytic infiltrates. Less prominent features included foci of acute myofiber degeneration and necrosis with or without intralesional mineralization and mild to moderate suppurative and lymphoplasmacytic infiltrates. Morphometric quantification demonstrated that the right ventricle was more severely affected (P ≤ .05) than the left ventricle with variable involvement of the interventricular septum. Immunohistochemistry for canine parvovirus was negative in all heart samples. However, the absence of parvoviral antigen does not rule out a possible viral or autoimmune cause. The presence of these myocardial lesions among closely related dogs suggests a genetic contribution to this disease process in the TMT.
Dilated cardiomyopathy (DCM) is the most prevalent form of cardiomyopathy in the dog and occurs mostly in specific breeds. 29 It is characterized by dilation and impaired systolic function of the left ventricle or both ventricles and, to a lesser degree, diastolic dysfunction. 29 Although dogs may develop DCM at any age, it is typically an adult-onset disease affecting young to middle-aged animals. Exceptions to this general rule have been noted in the juvenile Portuguese Water Dog (PWD) 14 and in a litter of Doberman Pinscher puppies. 49
Dysfunction of sarcomeres contributes to the functional and structural alterations of DCM. 20 In the dog, these structural alterations may be reflected microscopically as myofiber attenuation (atrophy), imparting a long wavy appearance to the fibers, or as myofiber degeneration with fibrosis and adipose tissue infiltration. 44 This variation in microscopic lesions and the tendency for one histologic pattern to predominate over the other have led some authors to argue that there are 2 histologically distinct forms of idiopathic canine DCM. 44
The attenuated wavy fiber form of DCM has been described in medium- to giant-breed dogs, 40,42,45 and specifically in Newfoundland dogs, 43 PWDs, 14 and 3 Doberman Pinscher littermates. 49 The fatty infiltration-degenerative form predominates in Doberman Pinschers with DCM 19 and Boxers with arrhythmogenic right ventricular cardiomyopathy (ARVC). 3
Irrespective of the histologic type, the underlying cause in most cases of canine DCM remains undetermined. 44 It has been postulated that canine DCM is the final result of a variety of myocardial insults arising from familial/genetic diseases, nutritional deficiencies, infectious diseases, metabolic disorders, immunologic derangements, and toxin/drug- and tachycardia-induced myocardial hypokinesis. 44 Genetic/heritable diseases are the most recognized causes of DCM in the mature dog, 29 whereas canine parvovirus is a recognized cause in puppies and juvenile dogs. 11,23 The overrepresentation of DCM within certain breeds and specific families of dogs suggests a genetic basis. 29 Through pedigree analysis, specific modes of inheritance have been reported in the Boxer, 34 Irish Wolfhound, 13 Doberman Pinscher, 30 and PWD. 14
A new and rapidly progressive form of DCM has recently been recognized in juvenile Toy Manchester Terriers (TMTs). Most affected dogs are younger than 1 year and typically die suddenly without signs of heart disease. These findings are similar to DCM in the juvenile PWD. 14 The purpose of this study was to characterize the clinical, gross, and microscopic features of DCM in juvenile TMTs.
Materials and Methods
Heart Tissue
A written request was sent to veterinary laboratories across Canada and the United States for cases of TMTs that had a history of sudden death and/or had a postmortem diagnosis of DCM, as well as animals that died as a result of noncardiac disease; 47 laboratories were contacted, and 11 (including our laboratory) participated in the study. Case selection criteria included availability of clinical history, postmortem reports, and histologic slides or paraffin-embedded blocks from affected hearts. The archives of the Atlantic Veterinary College, University of Prince Edward Island laboratory were searched following the same selection criteria as for external cases.
The following information was extracted from all postmortem reports: sex, age, heart weight, body weight, and gross and microscopic appearance of the heart. When available, the gross thicknesses of the left ventricle (LV), right ventricle (RV), and interventricular septum (IVS) were used to calculate RV wall thickness to LV wall thickness (RV/LV) ratios. The heart weight to body weight ratio (HW/BW) was calculated when the data were in the postmortem reports. TMTs with microscopic evidence of myocardial lesions were placed in the study group (TMTCM, for TMT cardiomyopathy). Heart tissue sections from 1 TMT with no cardiac lesions were included in the control group.
The control group consisted of sections of histologically normal heart tissue from 10 dogs that had died of noncardiac disease, including bronchopneumonia with bronchitis or bronchiolitis (n = 4), fibrinonecrotizing bronchopneumonia and necrotizing enteritis (n = 1, polymerase chain reaction [PCR] negative for canine parvovirus), hemorrhagic enteritis (n = 1, fluorescent antibody test negative for canine parvovirus), asphyxiation (n = 1), organophosphate toxicosis (n = 1), severe hepatic lipidosis (n = 1), and death of unknown cause (n = 1, only heart submitted). Selection of control hearts was further based on breed and age (age range of 6 weeks to 10 months, to approximate the age range of TMTCM). Preference was given to breeds that are most phylogenetically similar to TMTs. 38 The final control group included a Chihuahua, a Pug, a Standard Manchester Terrier, a TMT, 3 mixed breed dogs, a Miniature Pinscher, a Miniature Schnauzer, and a Yorkshire Terrier.
Light Microscopic Examination and Morphometry
The specimens were processed by routine histologic methods, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (HE), Masson’s trichrome (MT), Alcian blue (AB; pH 2.5), phosphotungstic acid–hematoxylin (PTAH), von Kossa (vK), and periodic acid–Schiff (PAS) with light green counterstain. The histologic sections of TMTCM hearts (n = 12) were reviewed and grouped according to myofiber morphology and lesion duration. The first group contained myofiber loss, attenuation, and fibrous tissue replacement; the second included acute myocardial degeneration and necrosis. Microscopic changes were also localized to LV, RV, and IVS. All sections were reviewed for evidence of inflammation, viral inclusions, bacteria, or protozoa.
For morphometry of the myocardial fibrous lesion area, the Masson’s trichrome–stained heart sections were mounted on an inverted microscope (IM-35; Zeiss, Munich, Germany) with an E-XY stage encoder (Microcode II; Boeckeler Instruments, Tucson, AZ), and a live image was acquired using a high-resolution video camera (Dage 68; Michigan City, IN) connected to a computer image analysis system (Optronics DEI-750 video camera controller; Meyer Instruments, Houston, TX). Each section of heart tissue was outlined and the total area quantified using an image analysis program (Bioquant; Bioquant Image Analysis Corporation, Nashville, TN) and stereology toolkit. Outlining consisted of manually tracing along the epicardial, endocardial, and cut surfaces at ×2.5 magnification using the stereology toolkit trace function. The sections of heart tissue were examined for lesions at ×6.3 magnification; lesions were outlined and the total lesion area for each tissue section was determined. The percent myocardial area affected (cPMAA) was calculated for each tissue section as (total lesion area/total tissue section area) × 100.
Immunohistochemistry
Paraffin-embedded tissue from each dog in the TMTCM group was submitted to an external veterinary diagnostic laboratory (Prairie Diagnostic Services, Saskatoon, SK, Canada) for parvovirus antigen detection. Methods included protease pretreatment, followed by application of primary monoclonal antibody (CPV1-2A1, 1:800; Custom Monoclonals, West Sacramento, CA). Antibody binding was detected using a streptavidin-biotin complex technique adapted for an automated slide stainer (BMK iVIEW DAB Paraffin detection kits on Benchmark staining platform; Ventana Medical Systems, Tucson, AZ).
Statistical Analysis
All summary data are presented as mean and standard deviation (SD) or median. The data entry and statistical analyses were carried out using a statistical software package (Minitab 15; Minitab, Inc, State College, PA). Paired t-tests were used to compare the cPMAA between the RV, LV, and IVS, and the differences between the paired observations were verified for normal distribution and to identify potential outliers using the Shapiro-Wilk test. Regression analyses and scatterplots were used to examine the relationship between age in weeks and cPMAA; RV, LV, and IVS were examined. Model verification included assessing normality of residuals (Ryan-Joiner test) to identify potential outliers or influential observations. In all cases, differences were considered significant if P ≤ .05.
Pedigree Analysis
The genetic relationships among affected dogs were determined using 10-generation pedigrees. The pedigrees were merged into 1 database containing 772 individuals. A subset pedigree containing several more closely related dogs was drawn using the Cyrillic software package. 12 Pedigree statistics were computed using PEDSTATS. 50 Inbreeding coefficients for each dog in the pedigree were calculated using the MTDFREML software package. 4
Results
Signalment and Clinical History
Postmortem reports from internal and external cases were available from 15 TMTs that died between 1988 and 2010. One of those 15 TMTs was placed in the control group due to absence of microscopic lesions and a confirmed cause of death (organophosphate toxicosis) that was not cardiac related. Postmortem reports were available for 14 TMTs that were confirmed to be affected based on microscopic lesions identified by microscopic examination of submitted slides or paraffin-embedded tissues (TMTCM Nos. 1–12) or based on microscopic cardiac lesions described in the submitted postmortem reports (TMTCM Nos. 13 and 14).
The clinical histories of the TMTCM dogs included sudden death without premonitory signs (4/14), death following brief listlessness and/or respiratory distress (5/14), and dogs found dead (2/14). In 5 of 14 TMTCM dogs, terminal events occurred 1 day or less following events such as general anesthesia/surgery or exercise (Table 1). The age of TMT dogs ranged from 10 weeks to approximately 58.3 weeks. No sex predilection was apparent (6 females and 8 males). Six of the 8 males were cryptorchid (4 bilateral, 1 unilateral, and 1 unspecified; Table 1). No information was available regarding the presence or absence of cryptorchidism in the remaining 2 males.
Age, sex, clinical history, heart lesion size measured as a percentage of myocardium area affected, and inbreeding coefficients in 14 Toy Manchester Terriers (TMT) with dilated cardiomyopathy (CM).
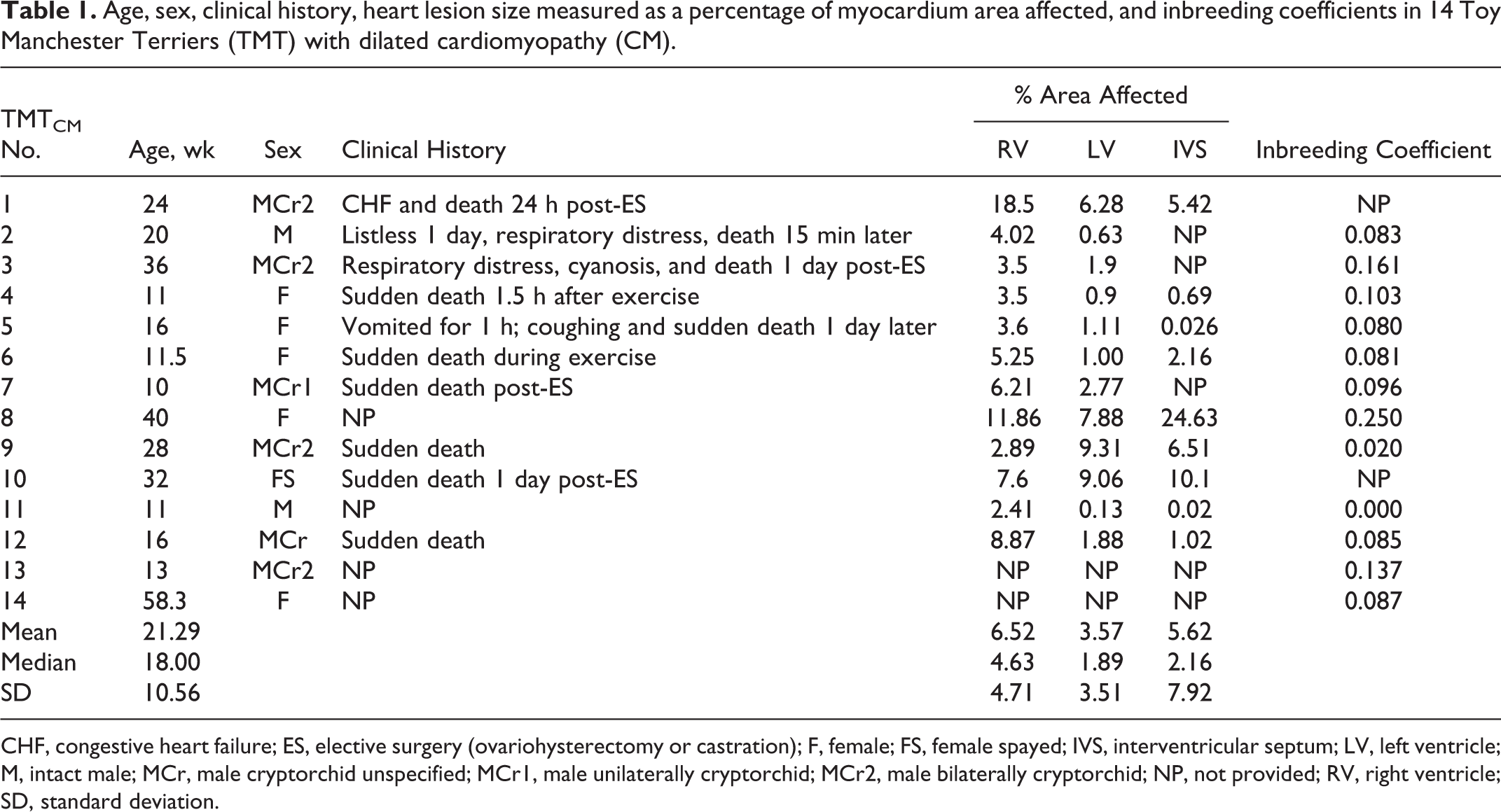
CHF, congestive heart failure; ES, elective surgery (ovariohysterectomy or castration); F, female; FS, female spayed; IVS, interventricular septum; LV, left ventricle; M, intact male; MCr, male cryptorchid unspecified; MCr1, male unilaterally cryptorchid; MCr2, male bilaterally cryptorchid; NP, not provided; RV, right ventricle; SD, standard deviation.
Gross Findings
In 7 of 14 cases, gross cardiac changes were described and included mild left atrial appendage enlargement, LV dilation, LV wall thickening, and/or cardiomegaly (TMTCM Nos. 1, 5, 7, 8, 9, 12, and 14). Six (TMTCM Nos. 1, 5, 8, 9, 12, and 14) of those reports had the necessary data to calculate the HW/BW and RV/LV ratios. Five dogs (TMTCM Nos. 1, 5, 8, 9, and 14 with ratios of 1.07, 1.44, 1.07, 1.08, and 1.00, respectively) had increased HW/BW ratios (normal range is 0.58%–0.94% of total body weight), 48 and the remaining dog had a decreased ratio (TMTCM No. 12 with a ratio of 0.56). Five of the reports describing gross cardiac changes also contained sufficient data to calculate the RV/LV ratio (TMTCM Nos. 1, 5, 10, 12, and 14); for TMTCM No. 7, only the RV/LV ratio was recorded. Four of 6 RV/LV ratios were within the normal range of 1:1.47 to 1:2.79, 48 whereas 2 cases had a moderately to markedly decreased RV/LV ratio (TMTCM Nos. 7 and 12; 1:4 and 1:6, respectively). For both cases with decreased RV/LV ratio, the hearts were described as having left ventricular wall thickening in the absence of dilation. The only consistent extracardiac change was moderate to marked pulmonary congestion and pulmonary edema in 11 of 14 TMTCM dogs.
Light Microscopic and Immunohistochemical Findings—Cardiac Tissue
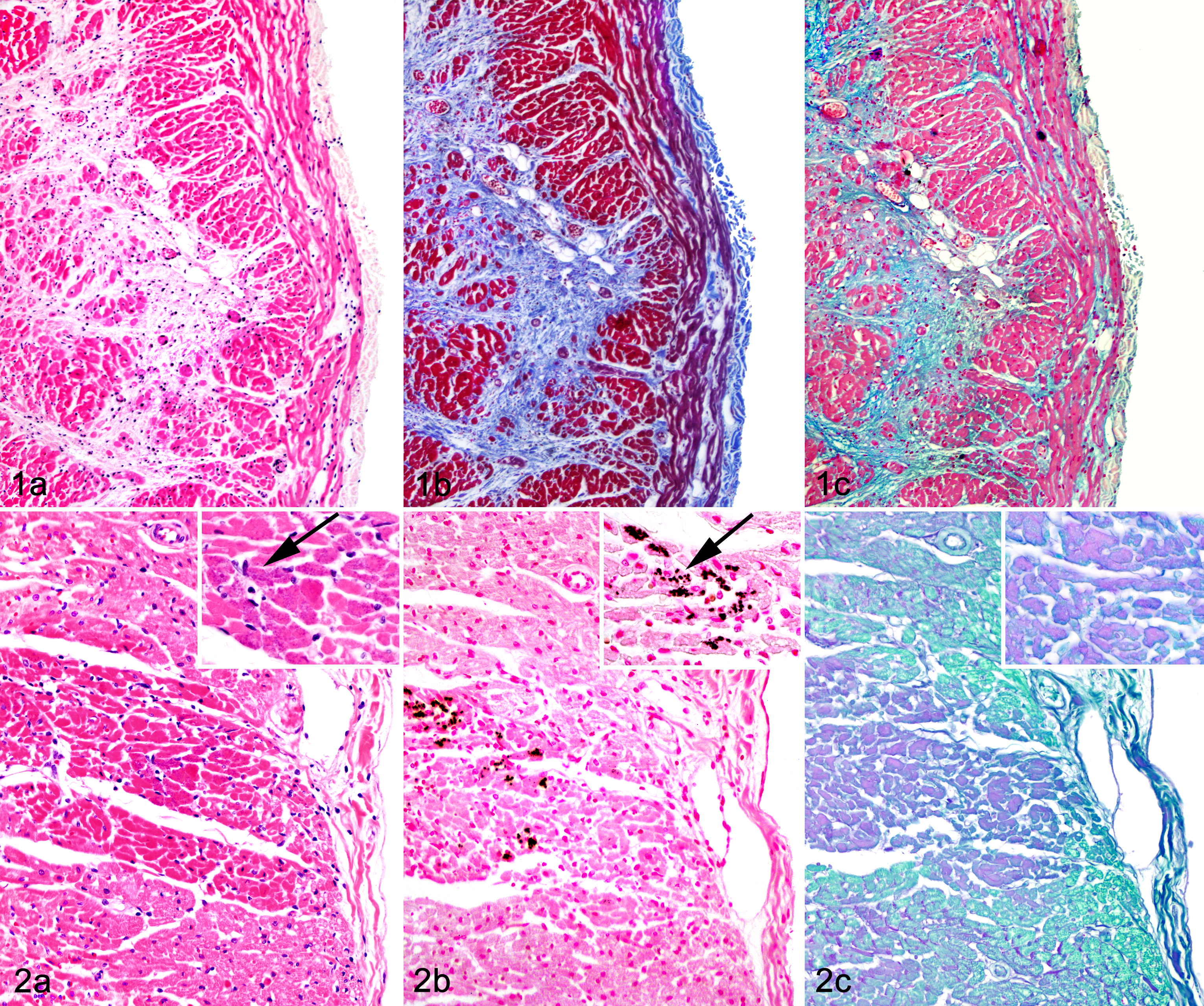
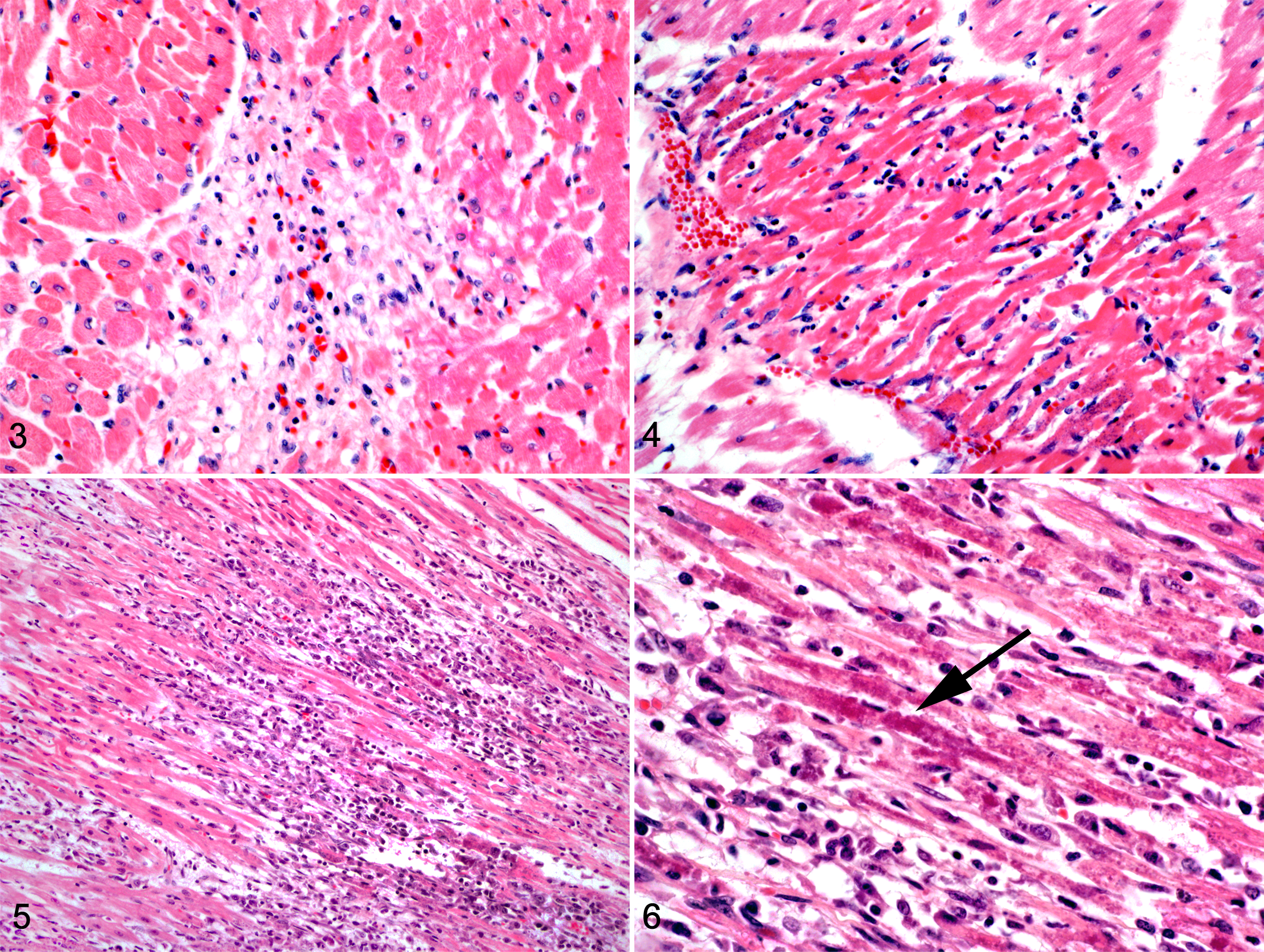
Heart tissue sections were available from the RV and LV for 12 of 14 TMTCM cases (TMTCM Nos. 1–12), and 9 of 12 cases (Table 1) included sections of IVS. Microscopic changes in hematoxylin and eosin (HE)–stained sections were extensive and largely consistent among the TMTCM animals. Most myocardial lesions were subacute to chronic and consisted of multiple, irregular foci of cardiomyocyte loss with stromal collapse and replacement by variably edematous fibrocollagenous tissue (Fig. 1a, Table 2). The fibrocollagenous tissue commonly dissected or surrounded myofibers and less commonly bridged affected foci. Areas of fibrosis were interspersed with scattered adipocytes and few lymphocytes, plasma cells, and macrophages (TMTCM Nos. 1, 3, 5–12). With trichrome stain, mild to moderate amounts of fibrillar collagen were in the foci of fibrosis (Fig. 1b). With AB staining, these areas stained faint blue, indicating the presence of acidic sulfated mucosubstances such as mucopolysaccharides and glycosaminoglycans (Fig. 1c). Cardiomyocytes entrapped within the stroma were commonly shrunken and angular or had a loss of striation and hypereosinophilia with or without flocculent changes (degenerating and necrotic cardiomyocytes). Other cardiomyocytes contained central to perinuclear zones of clearing or finely granular eosinophilic material within the cytoplasm that was PAS and AB negative. Many sections (most obvious in TMTCM Nos. 2 and 4 but also noted in Nos. 6, 8, 9, 11, and rarely in 12) also contained scattered foci of acute myocardial degeneration and necrosis (Fig. 2a). Unlike the mild lymphoplasmacytic inflammation interspersed among the fibrous lesions in most sections (Fig. 3), TMTCM No. 2 had predominantly acute lesions that consisted of mild interstitial edema and mild to moderate lymphoplasmacytic and neutrophilic infiltrates (Fig. 4). Although inflammation was not prominent in most cases, in TMTCM No. 7, moderate to marked inflammatory infiltrates expanded and effaced the RV and LV myocardium (Fig. 5). The infiltrates consisted of numerous macrophages, lymphocytes, plasma cells, and less numerous degenerated neutrophils, commonly centered around and admixed with necrotic and degenerated cardiomyocytes (Fig. 6).
Histologic myocardial lesions in 14 Toy Manchester Terriers (TMT) with dilated cardiomyopathy (CM).
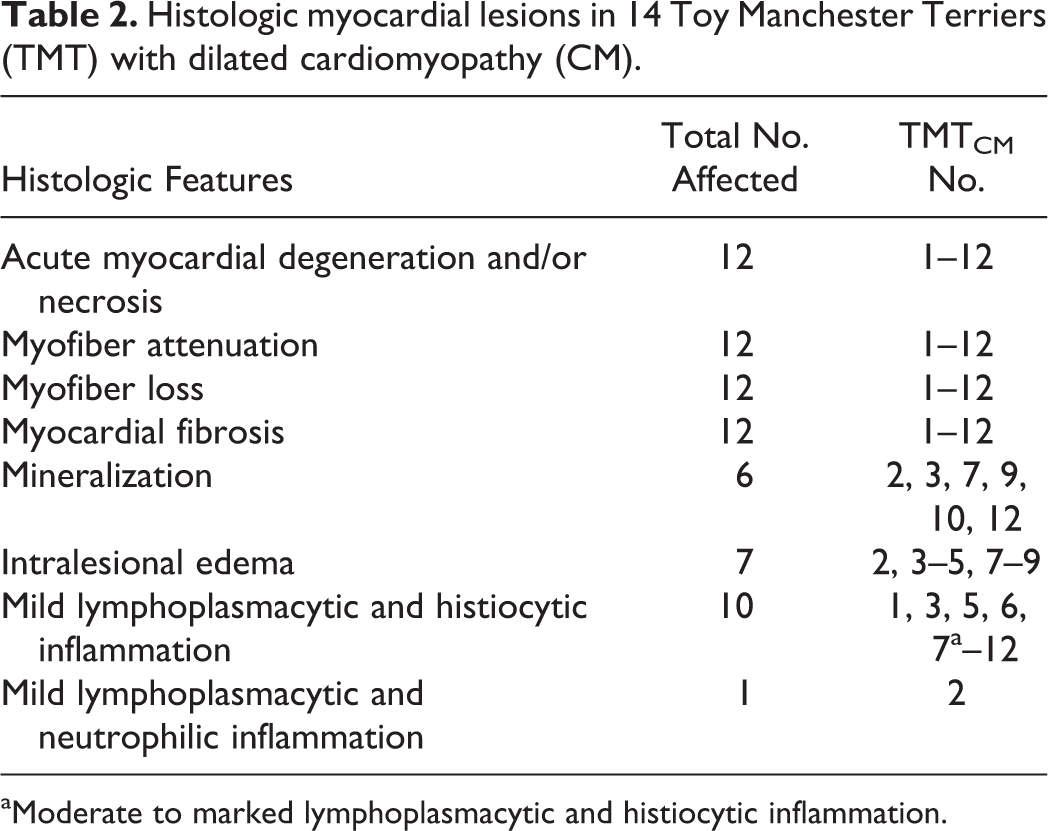
aModerate to marked lymphoplasmacytic and histiocytic inflammation.
Within the more acutely affected areas, there were deposits of granular to glassy basophilic/amphophilic material within the sarcoplasm of degenerating myofibers (TMTCM Nos. 7, 9, 10, and 12; Figs. 2a, 5, and 6). With von Kossa histochemistry, this material was black-brown, consistent with calcium phosphate (Fig. 2b). In 2 dogs (TMTCM Nos. 2 and 3), cardiomyocytes without basophilic deposits in HE-stained sections had von Kossa–positive deposits. No evidence of fibrosis, myocardial degeneration/necrosis, or mineralization was detected in any of the control sections.
With PAS, the TMTCM heart sections had scattered individual to variably sized clusters of cardiomyocytes with granular to homogeneous glassy, pale pink to purple cytoplasm (Fig. 2c) that corresponded to foci of acute myocardial degeneration and necrosis within and around the areas of fibrosis. In most control sections (Nos. 1–9, 11), rare individual or small clusters of faintly PAS-positive cardiomyocytes were seen.
In the areas of acute myocardial degeneration and necrosis (most obvious in TMTCM No. 2), the PTAH-stained cardiomyocytes had loss of striation with mild clumping or granularity of the sarcoplasm.
No viral inclusions, protozoa, or bacteria were found in any section. Immunohistochemistry did not reveal the presence of canine parvovirus antigens within any sections of heart tissue.
Morphometric Analysis
Morphometric analysis of the MT-stained sections from RV, LV, and IVS from the TMTCM group (n = 12) revealed variability in the cPMAA within each heart region and a significant difference (P = .041) in the percentage area affected between the RV and LV (Table 1). No significant differences in cPMAA were noted between RV and IVS or LV and IVS. Examination of the cPMAA for the IVS revealed a single value that was disproportionately larger than any other value (TMTCM No. 8; Table 1) for this region and could represent an outlier. Removal of that value from the data set reduced the mean (SD) of the cPMAA from 5.62 (7.92) to 3.24 (3.70), bringing these values closer to that of the LV.
Microscopic Findings—Extracardiac Tissue
Extracardiac tissues were provided for 10 TMTCM animals. Lung tissue (n = 10) showed changes consistent with acute congestion and edema. Similarly, congestion was identified in all sections of liver (n = 9).
Skeletal muscle was submitted for 3 TMTCM animals (TMTCM Nos. 4, 6, and 12). Two sections of skeletal muscle (TMTCM No. 4) contained individual or occasional small clusters of swollen myocytes, with hyalinized or flocculent sarcoplasm (degenerate and necrotic myocytes). Occasional degenerating fibers contained basophilic, granular material (consistent with mineral), and small numbers of macrophages and fewer neutrophils infiltrated rare necrotic myocytes. The sections of skeletal muscle from TMTCM Nos. 6 and 12 were histologically unremarkable.
Pedigree Analysis
Pedigrees were available for 12 of 14 TMTCM dogs (n = 12; TMTCM Nos. 2–9, 11–14). Analysis of the 10-generation pedigrees revealed 3 affected half-siblings (TMTCM Nos. 5, 6, and 8) with the same sire and 2 (TMTCM Nos. 3 and 7) with the same dam. Other relationships include a full-avuncular pair (TMTCM No. 3 was full uncle of No. 6) and several half-avuncular pairs (TMTCM No. 13 was half-uncle of Nos. 5, 6, and 8; No. 7 was half-uncle of Nos. 5 and 6). Any remaining relationships between affected individuals were more distant than grandparents. High inbreeding coefficients were calculated in 10 of 12 affected dogs (Table 1) in comparison to the entire population (inbreeding coefficient population mean [SD] = 0.047 [0.076]; Table 1).
Discussion
In this study of heart disease in TMTs, the clinical histories were similar. All but 1 dog was 10 months of age or less. Prior to sudden death, all the TMTs were overtly healthy. Similar numbers of males and females were represented. Most dogs were either found dead or died rapidly following a short period of listlessness, collapse, or respiratory distress. Cardiac disease in these dogs could have gone clinically undetected, similar to the occult myocardial disease in the Doberman Pinscher 10 or Boxer. 28
The sudden death of the TMTs in this study and the absence of premonitory clinical signs suggest that death probably was a result of a sudden fatal dysrhythmia. The stress associated with surgery, general anesthesia, and/or physical exertion could have triggered arrhythmias and, potentially, sudden death in susceptible dogs. Five of the 14 TMTs had a history of physical exertion or elective surgery prior to acute onset of clinical signs or sudden death. Clinically significant arrhythmias have been documented in the perioperative period in young, healthy dogs undergoing routine surgery such as ovariohysterectomy or orchiectomy. 17 Similar to the TMTs in this study, German Shepherds with inherited ventricular arrhythmias died suddenly during or following exercise. 35 The overrepresentation of cryptorchid dogs in our study may be attributed to the longer duration of general anesthesia typically required for cryptorchid surgical sterilization (when compared with routine castration), which potentially could cause prolonged myocardial depression. However, 2 of the cryptorchid TMTs died without undergoing castration, so there could be another unidentified association, possibly genetic, between cryptorchidism and cardiomyopathy to account for this overrepresentation. 15 The young age, short and abrupt clinical course, and absence of a sex predilection for cardiomyopathy in TMTs bear similarity to the DCM in juvenile PWDs; however, the subtle nature of the gross cardiac lesions in TMTs is strikingly different from those of the juvenile PWD. 14
Gross cardiac enlargement was noted in half the TMTs and, with the cardiac weight data, suggests mild cardiomegaly. One dog had a mildly decreased heart weight that could merely reflect normal variation rather than decreased myocardial mass. A moderately to markedly decreased RV/LV ratio was identified in 2 of 6 TMTs; this could indicate thinning of the RV free wall or thickening of the LV. However, ventricular wall thickness is modified by factors such as rigor mortis and dilation, such that simultaneous ventricular hypertrophy and dilation could result in an apparently “normal” wall thickness 5 and mask hypertrophy. 48 The variation in gross morphology in these dogs could reflect diversity in cardiac alterations or the subjectivity of visual interpretation by different veterinary pathologists. Because the gross cardiac changes were subtle in many of the TMTs, it can be assumed that sudden death occurred before substantial dilative change could take place.
Microscopic alterations are found in most cases of canine DCM, but none are pathognomonic or indicative of a specific cause. 14 These changes include 1 or more of the following features: myofiber degeneration, atrophy and necrosis, interstitial fibrosis in areas of myofiber loss, infiltration of adipocytes, and variable mononuclear (lymphoplasmacytic and histiocytic) inflammation. 40 Microscopic changes have been assigned to 2 distinct histologic forms of canine DCM: the attenuated wavy fiber type and the fatty infiltration–degenerative form. 44 Components of both histologic forms have been reported in the dilated hearts of affected Estrela Mountain Dogs 24 and Newfoundland dogs. 43
The histologic changes noted in the TMTs of this study were uniform, with minor variations among individual dogs. Notably, adipose tissue replacement of lost cardiomyocytes was not a prominent feature. Of the 2 described histologic forms, 44 the microscopic lesions in these juvenile TMTs (cardiomyocyte degeneration, loss with replacement by interstitial fibrocollagenous stroma) bore most resemblance to the fatty infiltration–degenerative form. This is in contrast to the attenuated wavy fiber form described in juvenile PWDs with DCM. Loss of cardiomyocytes and replacement by fibrous stroma, such as seen in the TMT hearts, are subacute to chronic lesions as it takes days to weeks for fibrous stroma replacement to occur. Acute myocardial necrosis and mineralization were also present in the affected TMT hearts, but these changes were much less conspicuous in most dogs. Application of PAS histochemistry highlighted an atypical cytoplasmic change in some acutely injured myofibers. This change could be associated with altered membrane permeability and diffusion of plasma proteins into the sarcoplasm of damaged muscle cells. 21 Overall, there appeared to be a mixture of acute and chronic lesions in the TMTCM group consistent with repeated waves of myocardial injury and repair.
Despite the chronicity of the majority of the heart lesions, none of the study dogs had a clinical history of cardiac disease prior to sudden death. This combination of myocardial lesions, short clinical course, and sudden death is unique among juvenile dogs with DCM. However, there are some similarities between the clinical disease course, progression, and myocardial lesions of these TMTs and the occult or asymptomatic DCM described in adult Doberman Pinschers and Boxers. 10,28 Like affected Boxers, Doberman Pinschers with the occult form of DCM frequently have gross and histologic changes consistent with the fatty infiltration–degenerative form. 10 The Doberman Pinschers that died as a result of congestive heart failure had cardiac lesions comparable to those that died suddenly with no clinical signs of heart disease. 10 Similar to the Doberman Pinschers and the TMTs in this study, Boxer dogs that died suddenly of occult DCM had grossly normal hearts with similar microscopic cardiac lesions that predominated in the RV. 28 Microscopic examination of heart tissue was necessary for the diagnosis of cardiomyopathy in some Boxer dogs 28 and in the TMTCM group.
Sudden death in Doberman Pinschers and Boxers, with similar mild gross cardiac lesions and fibrofatty histologic lesions, is thought to occur as the result of fatal arrhythmias. 28,39 The same may be postulated for the TMTCM dogs. Both acute inflammatory and chronic fibrotic myocardial lesions have been implicated in the creation of pro-arrhythmic substrates in human patients with cardiac disease. 1,2 In the acute inflammatory processes, inflammation of cardiomyocytes and the supporting interstitium provokes fluctuations in membrane potential and can result in arrhythmogenesis. 2 Arrhythmic events and sudden death have also been linked to or associated with myocarditis in dogs. 6 Similarly, myocardial scar tissue consisting of surviving cardiomyocytes, fibroblasts, myofibroblasts, and extracellular matrices can also mechanically and electrically interact to form arrhythmogenic substrates. 1,36 Although myofiber degeneration and fibrosis involved less than 20% of the myocardium in the majority of examined TMT heart sections, the lesion distribution and the combination of the chronic and acute injury could have created similar arrhythmogenic substrates.
The distribution of lesions in the TMTCM group involved sections of RV, LV, and IVS, in a similar distribution to that reported in Boxers, 3 Estrela Mountain Dogs, 24 Doberman Pinschers, 10 Newfoundland dogs, 43 and English Cocker Spaniels. 41 Morphometric variability was detected in the degree to which the different sections of myocardium were affected by fibrosis. Overall, the RV cPMAA was significantly larger than that of the LV, indicating a greater degree of fibrosis in the RV in most dogs. In 2 dogs, the LV had a larger cPMAA than the RV, indicating some variability with regard to the most affected region of the heart. Following removal of a single outlier (TMTCM No. 8), the mean cPMAA for the IVS was closer to that of the LV, suggesting the lesion severity within these regions was similar. Although a larger sample size was sought, the number of dogs in the study group was substantial given the rarity of this breed and the difficulty in clinical diagnosis. Due to the retrospective nature of this study, cardiac sampling was not standardized. Despite these limitations, the differences in the degree to which each of these heart regions was affected could reflect changes in these respective regions as a whole. If the RV is more extensively affected than the LV in TMTs, then the distribution and severity of these histological changes may be similar to those observed in Boxer dogs. 3 Therefore, sections of RV may be most valuable in the diagnosis of DCM in the TMT. The lesion distribution could also provide insights into the possible mechanism/cause of the disease. 31,33
In most cases of canine DCM, the underlying cause is not determined, 44 and other myocardial diseases can produce identical clinical manifestations. It is clear that gross dilative change seen at necropsy may be the result of a variety of myocardial insults or underlying mechanisms. However, the histologic changes could be more specific for a particular disease process than previously thought. 44 For example, fatty infiltration–degeneration may be a consequence of cardiomyocyte loss due to inflammatory insults such as myocarditis 9 or resulting from other injurious stimuli, including antineoplastic agents. 26 The attenuated wavy fiber form could be associated with acute myocardial ischemia 18 or represent an early change due to cytoskeletal defects. 46
Examination of the hearts and additional tissues from the TMT group did not reveal evidence of valvular heart disease, vascular disorders, or extracardiac disease that could account for the gross and microscopic changes described in this study. Underlying genetic, immune-mediated, and/or infectious conditions were also considered as potential causes for these heart lesions. Because many of the heart sections from the TMTCM group had mild to rarely moderate inflammation, the tissues were examined carefully for intranuclear viral inclusions, bacteria, and/or protozoa, but no infectious agents were identified by light microscopy. All heart sections from the TMTCM group were also immunohistochemically negative for canine parvoviral antigen. Given these results and the mildness of the inflammation in most heart sections, it is tempting to dismiss canine parvovirus as a possible cause. However, viral myocarditis is thought to exist as a triphasic disease process. 22,25 Initially, injury is induced either by direct myocardial injury due to viral replication or through the host’s immune response to the viral agent (viral stage). 22,25 The viral phase may transition into the autoimmune phase, in which the immune response continues despite elimination of the virus. 22,25 In the chronic phase of this continuum, the autoimmune response can persist, despite the absence of viral antigen, potentially leading to cardiac remodeling and DCM. 22,25 With this understanding of viral and postviral myocarditis, the mild to moderate myocardial inflammation and minimal cardiac dilation in the TMTCM group bears some resemblance to the autoimmune or chronic phase of the viral myocarditis continuum.
The absence of detectable canine parvoviral antigen within the TMTCM group could indicate that viral antigen was no longer present in sufficient quantities for immunohistochemical detection or that canine parvovirus was never present as an inciting agent. 27 Myocarditis typically occurs as a result of common viral infections in humans; however, a broad range of infectious, immune, and toxic causes have also been implicated. 22 Autoimmune cardiac disease has been associated with other infectious agents (eg, Trypanosoma cruzi) in dogs. 8 Noninfectious causes of inflammation can also lead to autoimmune cardiac disease and dilation. 22 Cellular components that escape autophagy and the ubiquitin-proteasome pathways (eg, mitochondrial DNA, abnormal proteins, damaged organelles) have been linked to the development of autoimmune myocarditis and dilated cardiomyopathy following a variety of stressors and injuries (including external hemodynamic stress, ischemia, defects in genes coding for sarcomeric proteins, and viral infections). 37,47 In addition to canine parvovirus, 11 other potential causes of cardiac injury and autoimmune disease must be considered as inciting factors for sudden arrhythmias, cardiac dilation, and death of the TMTs in this study.
Circulating autoantibodies against myosin heavy chain and α-cardiac actin were significantly higher in dogs with spontaneous DCM in comparison to normal counterparts. 7 Similarly, antimitochondrial antibodies and serum antinuclear antibodies (ANA) have been identified in some related English Cocker Spaniels with DCM. 16 The relatedness of the English Cocker Spaniels, as well as the strong association between DCM and specific complement C4 phenotype, suggests an inherited susceptibility to the development of autoimmune disease. 16
Heritability of DCM and modes of transmission have been established in many affected breeds. Autosomal dominant modes of transmission have been reported in the Boxer, Irish Wolfhound, 13 and Doberman Pinscher, 30 whereas an autosomal recessive mode of inheritance has been documented in the PWD. 14 A splice site mutation in a gene encoding for a mitochondrial protein has been associated with the development of DCM in the Doberman Pinscher. 32 Similarly, ARVC appears to be associated with the presence of a striatin gene mutation in Boxers. 31
The presence of a phenotypically distinct form of cardiomyopathy in this relatively rare breed suggests that genetic factors contributed to the cardiac disease in this cohort of TMTs. The process of selection for a toy variety of Manchester Terriers narrowed the gene pool and resulted in sharing of more alleles with each other than with other dogs. The presence of myocardial lesions among close relatives, such as half-siblings, is further evidence for a genetic contribution. The prevalence in various regions of both Canada and the United States suggests that this condition is unlikely to be caused solely by an environmental factor. The presence of close relatives among the TMTCM group and the high inbreeding coefficients are consistent with a recessive or multigenic mode of inheritance for the TMT cardiomyopathy phenotype, when the allele(s) for the disease has a higher frequency in a subset of the population. Dominant or X-linked modes of inheritance are unlikely due to the lack of transmission from parent to offspring and the prevalence of the trait in females, respectively. Based solely on the current data, it is not possible to determine whether this trait was a simple recessive or a quantitative trait caused by combined genetic and environmental factors.
Further work is necessary to identify a specific genetic cause for this form of cardiomyopathy. The collection of tissue and DNA samples from additional affected dogs is crucial to attain the sample size necessary for a genetic association study using modern high-density single-nucleotide polymorphism arrays. Identification of smaller subpedigrees with multiple affected individuals would facilitate statistical evaluation of the likelihood of recessive and multigenic modes of inheritance. If cardiomyopathy is a quantitative trait, a larger collection of samples could provide information on interactions between genetic and environmental factors affecting cardiomyopathy in this breed.
In conclusion, we describe a previously unrecognized form of juvenile-onset DCM in TMT dogs. Most of the affected dogs appeared healthy prior to sudden death, suggesting an occult form of disease in this breed. The affected hearts had minimal to mild gross evidence of cardiac hypertrophy or dilation, but histologically the myocardium had prominent loss of cardiomyocytes with interstitial fibrosis and less prominent areas of acute degeneration and necrosis. The fibrous lesions were most severe in the RV compared with the LV or IVS. The absence or mild degree of gross cardiac changes indicates that death occurred in many dogs before clinically significant cardiac dilation and hypertrophy developed. Further investigations should include electrocardiographic and/or echocardiographic evaluation of juvenile TMTs to detect conduction abnormalities or cardiac dysfunction. Further pedigree analysis and genetic testing is necessary to confirm that this disease is heritable in the TMT.
Footnotes
Acknowledgments
The following institutions contributed tissues and postmortem reports used in this study: Texas A&M University, New Brunswick Department of Agriculture and Fisheries, Nova Scotia Department of Agriculture, Purdue University, Oregon State University, Michigan State University, Louisiana State University, University of Guelph, and University of Tennessee. We thank those who searched laboratory databases for potential cases and the dog owners and referring veterinarians whose pets or patients were included in this study. A very special thanks to Amanda Kelly (Toy Manchester Terrier breeder and liaison for the Canadian and American Manchester Terrier Clubs) and the Canadian and American Manchester Terrier Clubs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is partially funded by the Atlantic Veterinary College Companion Animal Trust Fund.
