Abstract
The amyloid of canine amyloid-producing odontogenic tumor (APOT) was evaluated biochemically and immunohistochemically. The N-terminal amino-acid sequence of purified amyloid protein from a canine APOT was strikingly similar to the sequence in both rat ameloblastin and porcine sheathlin. Immunohistochemically, the amyloid in APOT from 9 dogs was strongly reactive with anti-rat ameloblastin, anti-porcine sheathlin, and anti-canine APOT amyloid and weakly reactive with anti-porcine amelogenin but negative for antibodies to cytokeratins, vimentin, desmin, α-smooth muscle actin, amyloid A, glial fibrillary acidic protein, or S100 protein. The neoplastic epithelial cells of APOT were focally reactive with antibodies to ameloblastin, sheathlin, amelogenin, and canine APOT amyloid. The similarity in amino-acid sequence of the amyloid protein of canine APOT to that of enamel proteins, such as ameloblastin, sheathlin, and amelogenin, and the expression of these antigens in both APOT amyloid and in the neoplastic cells suggest that the amyloid of canine APOT is derived from enamel proteins secreted by ameloblasts.
Amyloidosis is a heterogeneous group of disorders characterized by the deposition of amyloid proteins derived from any of at least 25 different precursor molecules. Amyloid is considered a pathologic substance that appears histologically as an extracellular, amorphous, congophilic protein with apple-green birefringence under polarized light. Amyloidosis can be categorized as systemic or localized. Common human types of localized amyloidosis include amyloidosis of endocrine organs associated (or not) with neoplastic conditions and cerebral amyloidosis of Alzheimer’s disease. 21,51 Localized amyloid deposits with or without association with neoplasia also have been described in the human mammary gland, 41,52 respiratory tract, 26,34 gastrointestinal tract, 5,38 genitourinary tract, 13,17 skin, 24,25 and bone. 23
In animals, localized amyloidosis has been associated with neoplastic conditions such as calcifying epithelial odontogenic tumor (CEOT), 2,29 pancreatic endocrine tumor, 28 medullary thyroid carcinoma, 15 ameloblastoma, 10 mammary carcinoma, 43 intestinal carcinoid, 40 extramedullary plasmacytoma, 31,36 and myeloma. 12 CEOT is a rare neoplasm of the tooth-forming apparatus that produces a mineralized substance and amyloid. 30 The origin of the amyloid in human and animal CEOT is often debated but is still unknown. 2,19,27,29,37,42,46 Recent studies indicate that the amyloid in human CEOT is secreted by the neoplastic epithelial cells, but its precise nature remains unclear. 27,39,42
CEOT in veterinary medicine is classified as amyloid-producing odontogenic tumor (APOT). 9,14 In the present study, we partially sequenced the amino acids of the major amyloid protein of canine APOT and compared the amino-terminal sequence with that of the enamel proteins rat ameloblastin and porcine sheathlin, which are transcribed in cells of the epithelial root sheath in normal incisor tooth germ. 16,47,48,50 We examined the immunohistochemical profile of the amyloid using antibodies to ameloblastin, sheathlin, amelogenin, and amyloid protein isolated from a canine APOT.
Methods
Amyloid Extraction, Purification, and Sequencing
Amyloid protein was extracted from the APOT of a 12-year-old male Shih Tzu dog (Table 1, dog No. 9) by a modification of the procedure of Pras et al. 33 The proteins prepared by high-pressure liquid chromatography (HPLC) were separated by sodium dodecyl sulfate–polyacrylamide slab-gel electrophoresis (SDS-PAGE) according to Laemmli. 22 The polypeptide bands thus separated were transferred to a polyvinylidene fluoride membrane by the method of Towbin et al 45 and stained with Coomassie brilliant blue R-250. The band of amyloid protein was excised from the membrane, and its NH2-terminal amino-acid sequence was determined by an Applied Biosystems (Applied Biosystems, Inc. Foster City, CA) 490 protein sequencer.
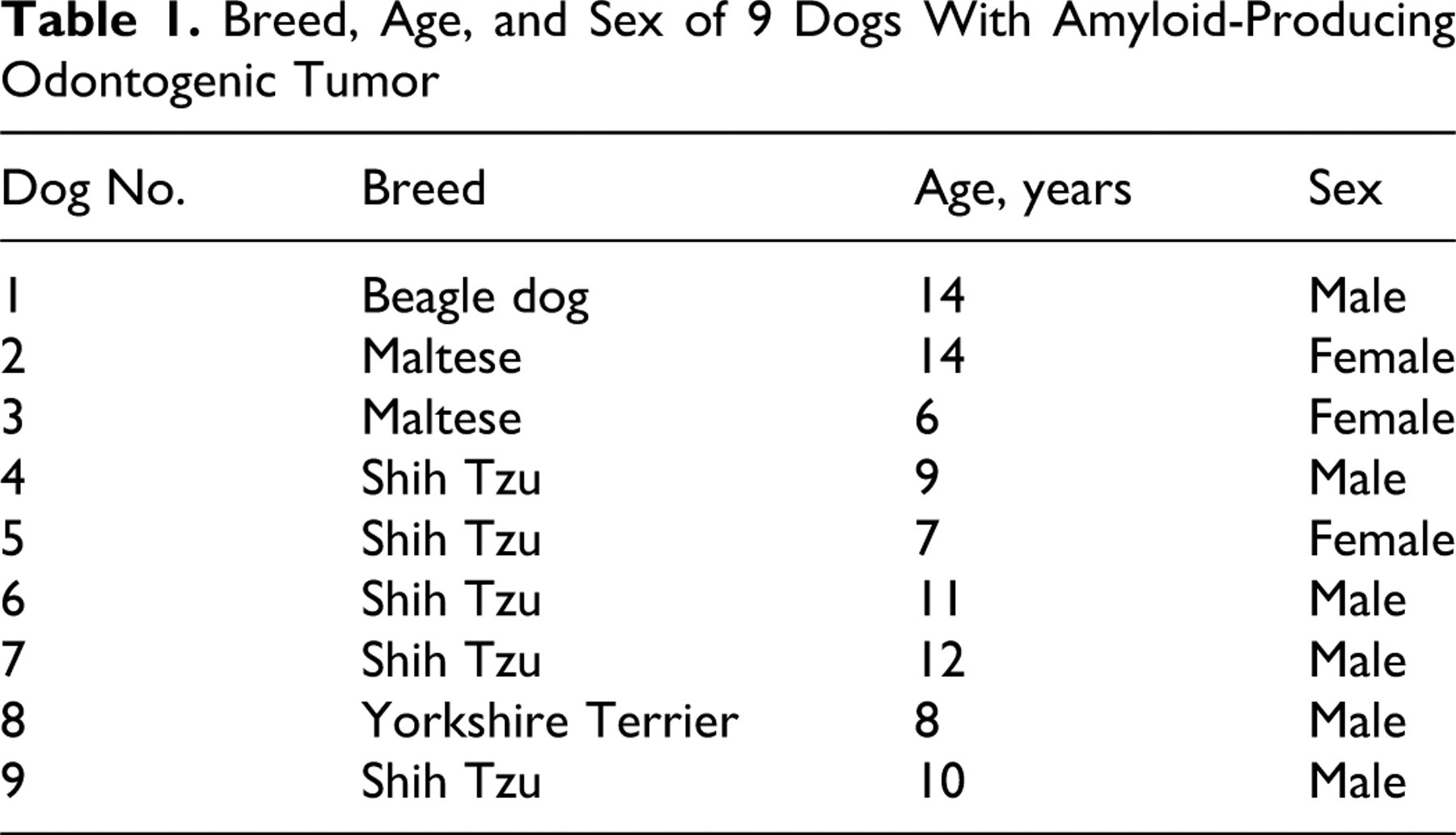
Breed, Age, and Sex of 9 Dogs With Amyloid-Producing Odontogenic Tumor
Histopathology
Neoplastic tissues from 9 dogs with APOT (Table 1) and normal tooth germ from 3 canine fetuses were fixed in 10% neutral buffered formalin, decalcified in 5% formic acid solution for 24 hours, and neutralized in 5% sodium sulfate for another 24 hours. The decalcified tissues were processed routinely, embedded in paraffin, sectioned (4 μm), and stained with hematoxylin and eosin. Additional sections were stained by Congo red with and without prior 5% potassium permanganate treatment. Congo red–stained sections were examined under polarized light. Undecalcified tumor tissues were also processed as described above, and sections were stained by the von Kossa method.
Antibodies
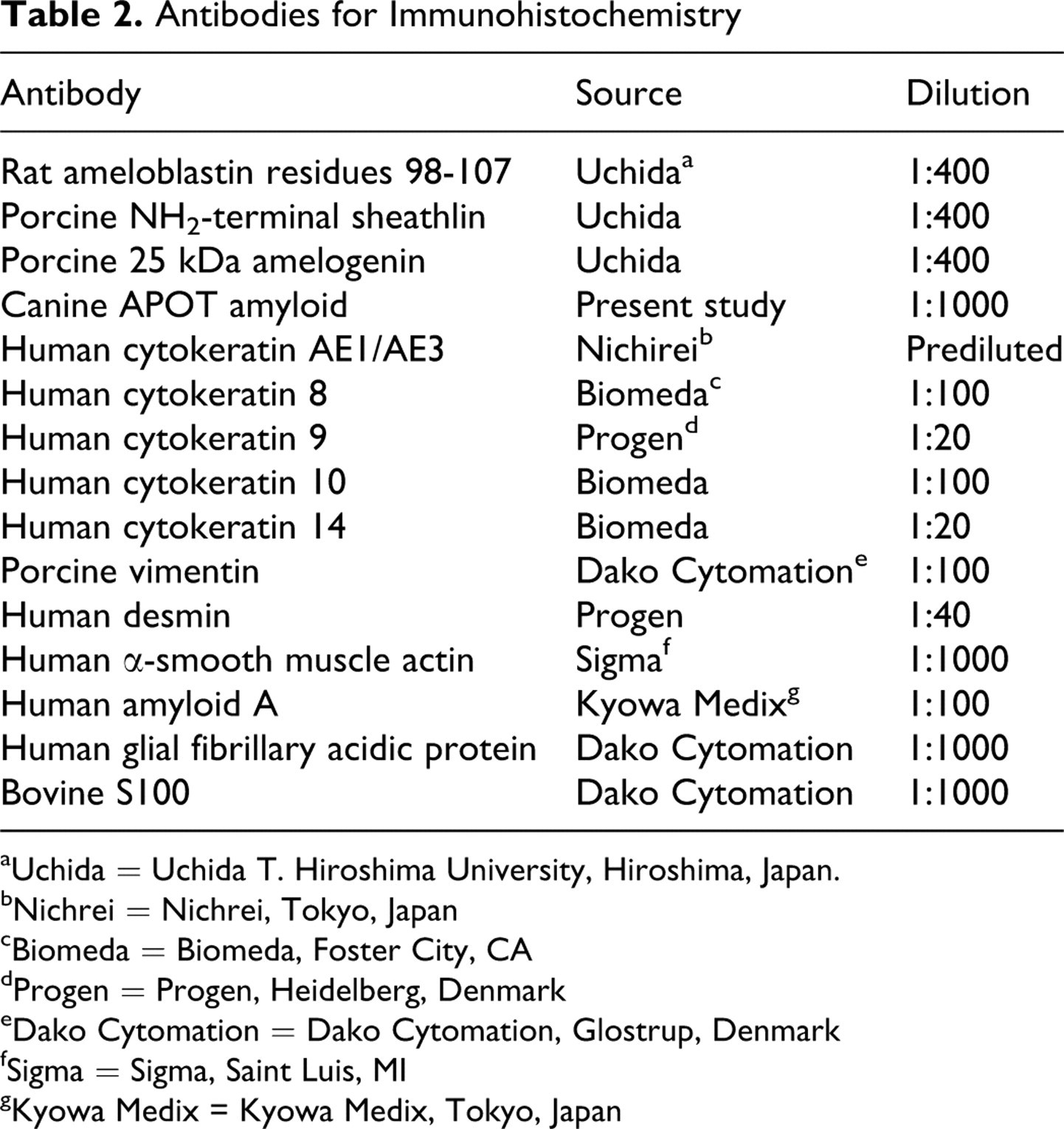
Rabbits (4-week-old females) were injected intramuscularly with the purified APOT amyloid protein plus complete Freund’s adjuvant for 2 months. The polyclonal amyloid antibody fraction (anti-canine APOT amyloid polyclonal antibody) was prepared from the resulting rabbit hyperimmune sera by affinity chromatography using Sepharose 4B coupled with purified amyloid protein. Anti-rat ameloblastin (residues 98-107), anti-porcine sheathlin (NH2-terminal), and anti-porcine amelogenin polyclonal antibodies were prepared. 42,50 Anti-porcine vimentin, anti-human desmin, anti-human α-smooth muscle actin (α-SMA), monoclonal anti-human amyloid A, anti-human glial fibrillary acidic protein (GFAP), polyclonal anti-bovine S100 protein, and anti-human cytokeratins (CK) AE1/AE3, CK 8, CK 9, CK 10, and CK 14 were also used for immunohistochemical evaluations of in situ amyloid protein and neoplastic epithelial cells of this tumor. Details of the antibodies are summarized in Table 2.
Antibodies for Immunohistochemistry
aUchida = Uchida T. Hiroshima University, Hiroshima, Japan.
bNichrei = Nichrei, Tokyo, Japan
cBiomeda = Biomeda, Foster City, CA
dProgen = Progen, Heidelberg, Denmark
eDako Cytomation = Dako Cytomation, Glostrup, Denmark
fSigma = Sigma, Saint Luis, MI
gKyowa Medix = Kyowa Medix, Tokyo, Japan
Immunohistochemistry
Histologic sections of APOTs from 9 dogs and normal tooth germ tissue from 3 canine fetuses were examined immunohistochemically using the avidin–biotin–peroxidase complex (ABC) procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA). Endogenous peroxidase was blocked in deparaffinized sections by incubation in 3% H2O2 for 10 minutes. All sections were incubated with primary antibody at 4°C for 16 hours, with biotinylated secondary antibody for 30 minutes at room temperature, and with ABC for an additional 30 minutes. The immunologic reaction was detected with 0.05% 3,3′-diaminobenzidine as chromogen. Tooth germ from a normal canine fetus was used for immunohistochemical controls. Control procedures included the use of excess antigen in relation to immune sera and nonimmune sera instead of the primary antibody. Omission of the primary antibody abrogated the immunohistochemical reaction.
Results
Amino-Acid Sequence Analysis of Amyloid Fibril Protein
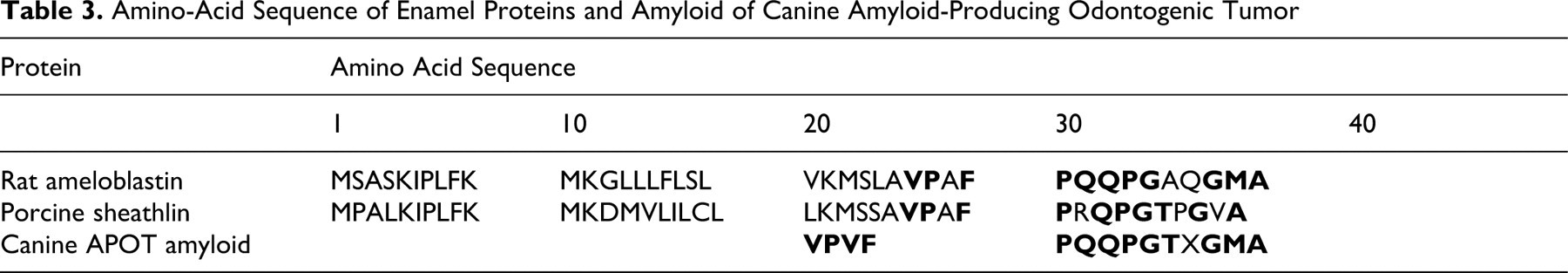
SDS-PAGE of amyloid protein produced a band that was slightly smaller than 14.4 kDa (Fig. 1). The NH2-terminal amino acid sequence of the amyloid protein was determined (Table 3). The first 14 amino acid residues of the protein were Val-Pro-Val-Phe-Pro-Gln-Gln-Pro-Gly-Thr-X-Gly-Met-Ala. A homology search using FASTA (Sequence Similarity Search Served by Genome Net: http://FASTA.genome.ad.jp) revealed that the amyloid protein had striking similarity in NH2-terminal amino-acid sequence to that of rat ameloblastin (11/14 identical amino acids) and porcine sheathlin (10/14 identical amino acids) (Table 3).
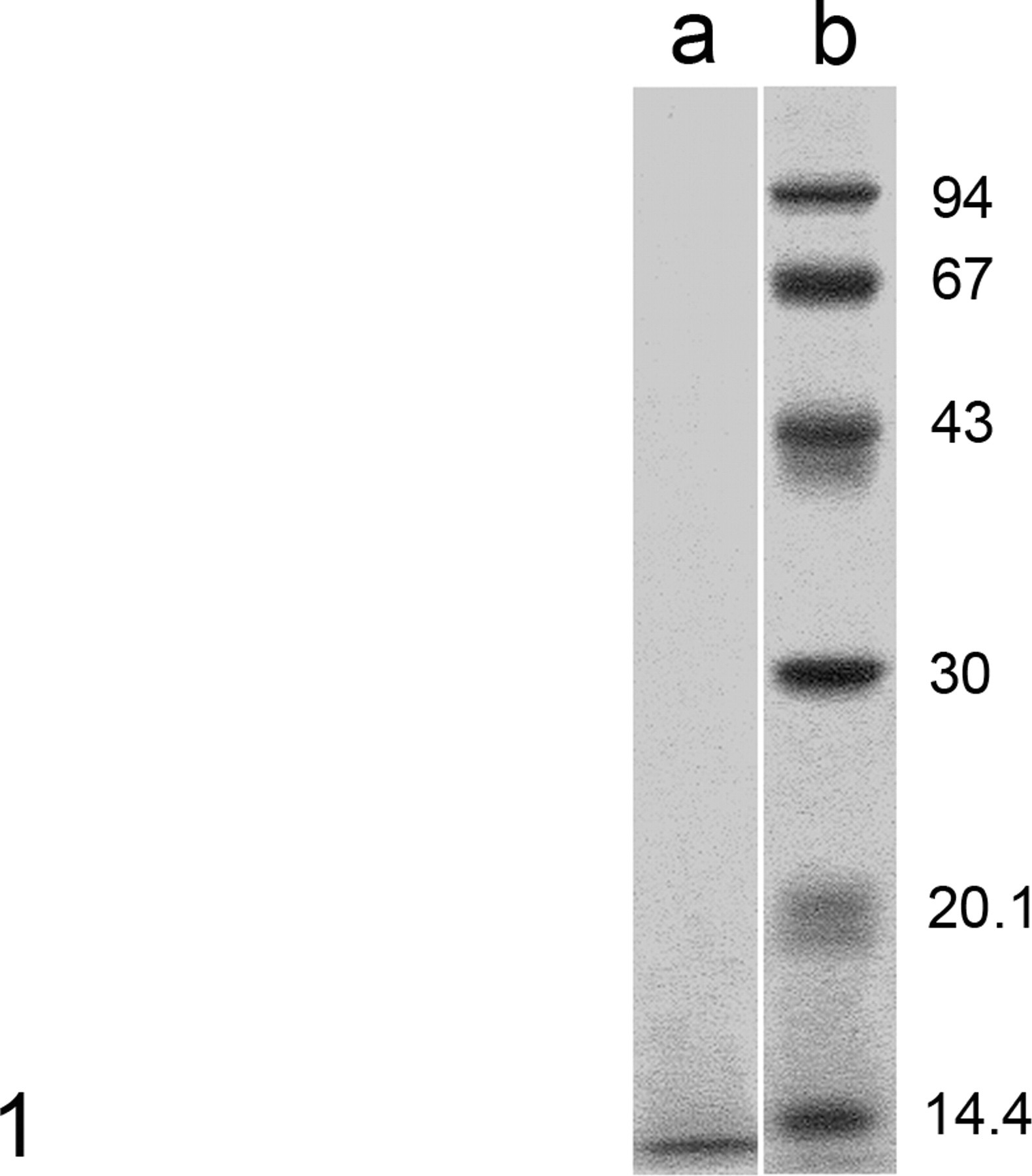
Amyloid-producing odontogenic tumor (APOT); dog No. 9. Sodium dodecyl sulfate–polyacrylamide slab-gel electrophoresis (SDS-PAGE) of canine APOT amyloid produced a polypeptide band slightly less than 14.4 kDa (column a). Molecular weight markers are shown in column b. APOT, amyloid-producing odontogenic tumor.
Amino-Acid Sequence of Enamel Proteins and Amyloid of Canine Amyloid-Producing Odontogenic Tumor
Histopathology
In all dogs, the APOT consisted of islands and sheets of neoplastic odontogenic epithelium with interstitial deposits of homogeneous eosinophilic material. The deposits were congophilic (Fig. 2) both with and without 5% potassium permanganate pretreatment and had apple-green birefringence in polarized light (Fig. 3). Mineralized foci, labeled by von Kossa’s method, were scattered throughout the tumors including within the amyloid deposits.
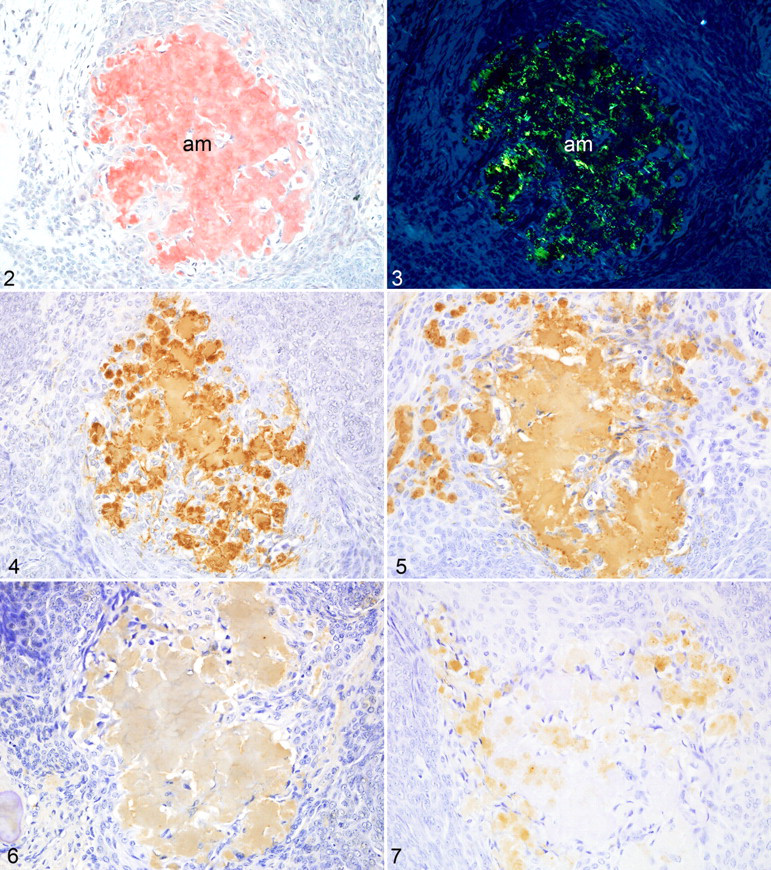
Immunohistochemistry
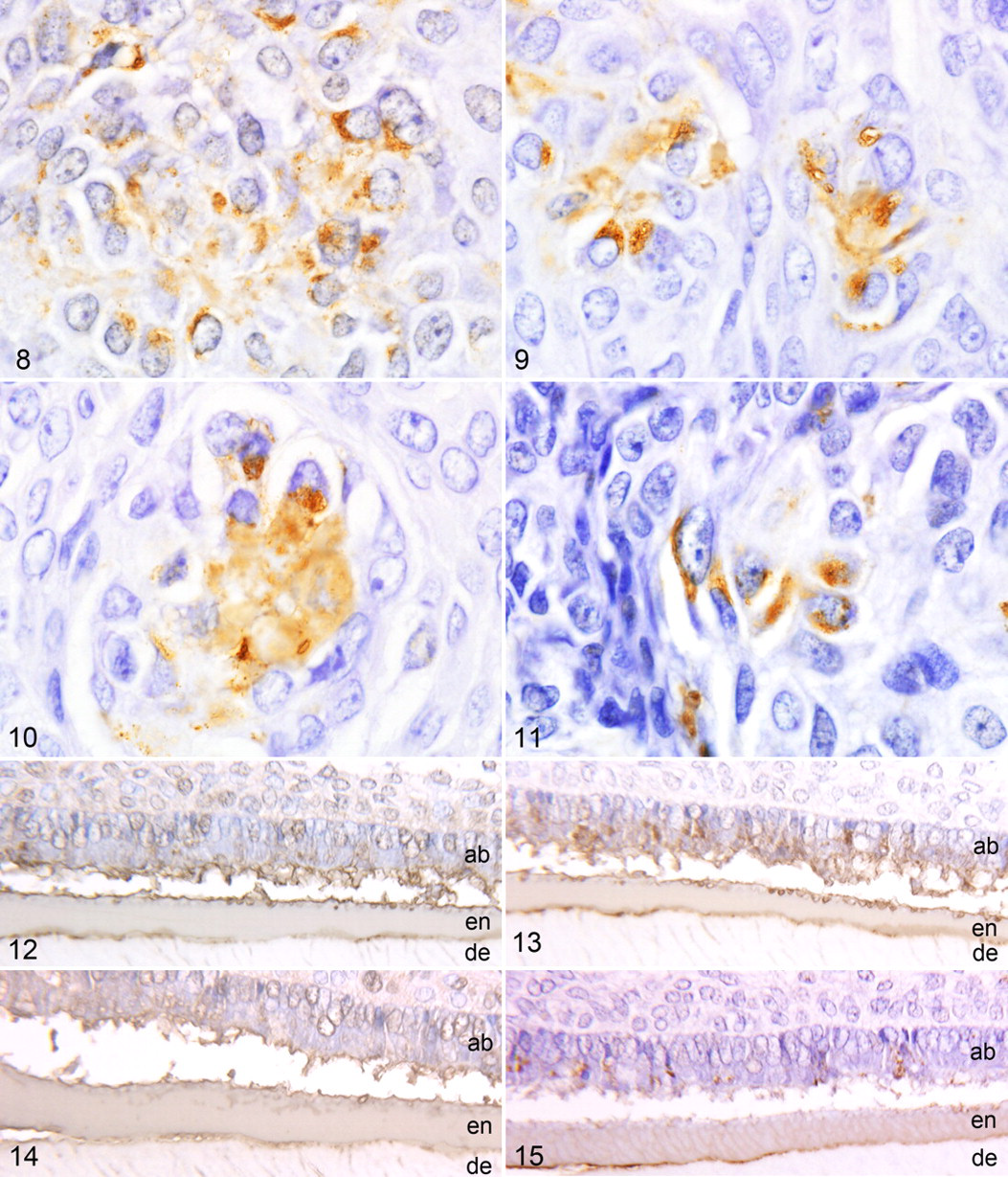
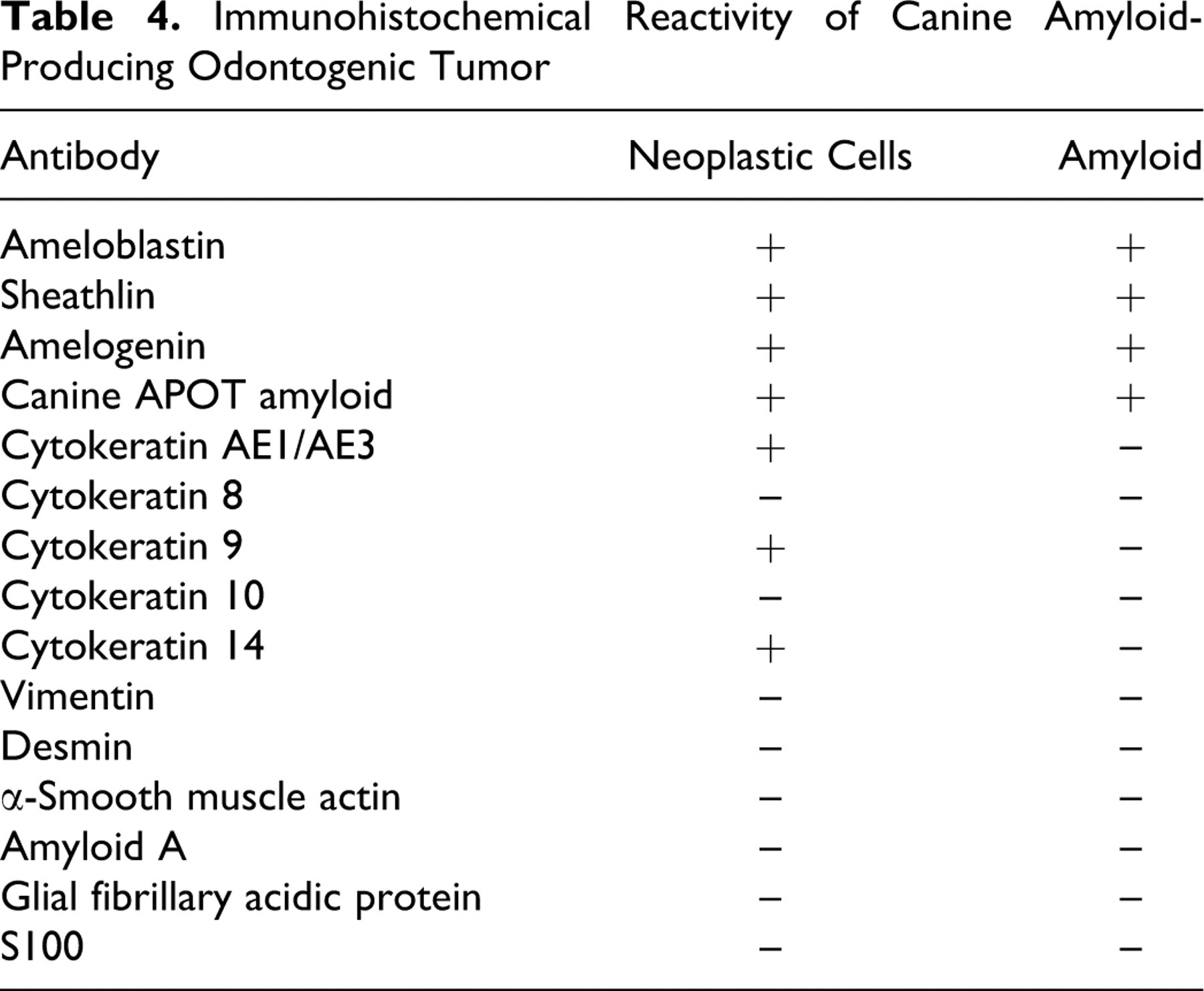
The amyloid in all tumors reacted strongly with antibodies to ameloblastin (Fig. 4 ), sheathlin (Fig. 5 ), and canine APOT amyloid (Fig. 6 ). Focal weak reactivity was noted with antibody to amelogenin (Fig. 7). Some neoplastic epithelial cells also expressed the above antigens (Figs. 8–11). The amyloid did not express CK AE1/AE3, CK 8, CK 9, CK 10, CK 14, vimentin, desmin, α-SMA, amyloid A, GFAP, or S100 protein. However, the neoplastic epithelial cells in all tumors expressed CK AE1/AE3, CK 9, and CK 14. In the bell stage of developing tooth germs from 3 canine fetuses, immature enamel matrix and the cytoplasm of ameloblasts strongly expressed ameloblastin (Fig. 12), sheathlin (Fig. 13), amelogenin (Fig. 14 ), and canine APOT amyloid (Fig. 15 ). Immature dentine matrix, odontoblasts, dental papilla, and dental follicles in tooth germs did not express these antigens. Immunohistochemical results are summarized in Table 4.
Immunohistochemical Reactivity of Canine Amyloid-Producing Odontogenic Tumor
Discussion
In this study, the presence of enamel proteins, specifically, ameloblastin, sheathlin, and amelogenin, was demonstrated in the amyloid and the neoplastic epithelial cells of canine APOT. The list of enamel proteins in the developing tooth germ includes ameloblastin, amelogenin, sheathlin, enamelin, tuftelin, and others, with new proteins found as studies continue. 35,42 These proteins, produced by ameloblasts at different stages of development, are considered to play various roles in the modulation of mineral deposition and crystal growth during tooth morphogenesis. 35
Ameloblastin was cloned from an enamel protein in rat incisor tooth germs. 4,7,18 Sheathlin, considered to be part of the same protein as ameloblastin, was recently cloned from porcine tooth germs. 16 An immunohistochemical study using antibodies that recognize different regions of that protein showed that sheathlin is synthesized by ameloblasts and secreted into the immature enamel matrix with posttranslational and postsecretory modifications and that cleaved NH2-terminal polypeptides concentrate in the prism sheath. 49 It is assumed that sheathlin, its cleavage products, or both play important roles in amelogenesis, such as mediation of the contribution of ameloblasts to the mineralized matrix and the modulation of crystal growth. 49 Immunohistochemical results suggest that the protein is homogeneously distributed in the surface layer of the tissue (ie, in newly secreted enamel) where it appears as the nascent 62-kDa molecule. 47,50
Amelogenins form the bulk of the enamel matrix (90%) during the secretory stage of enamel development 35 and are initially secreted as a 25-kDa nascent protein in porcine tissues. 50 In other species, amelogenin varies slightly in molecular mass. 8,35,44 Nascent amelogenin is spatially restricted to the newly secreted enamel and is processed relatively rapidly so that the subsurface, bulk, and inner enamel is dominated by a 20-kDa amelogenin molecule in the enamel matrix. 3,6
In the present study, the amino-acid sequence of canine APOT was closely homologous to that of both rat ameloblastin and porcine sheathlin. Immunohistochemically, the amyloid proteins of canine APOT reacted strongly with polyclonal antibodies to rat ameloblastin, porcine sheathlin, porcine amelogenin, and canine APOT amyloid. Immature enamel matrix and ameloblasts in the developing tooth germs of canine fetuses also reacted with these antibodies. The antibodies to ameloblastin and sheathlin used in the present study recognized the NH2-terminal residues of the enamel proteins in canine developing tooth germs. Thus, the proteins of the amyloid in canine APOT share similar amino-acid sequence with ameloblastin, sheathlin, and amelogenin, which indicates that these enamel proteins may be precursors to the amyloid of APOT. The immunohistochemical characteristics of neoplastic cells of canine APOT and ameloblasts in developing canine tooth germs suggest that this odontogenic neoplasm is derived from the ameloblast.
Canine APOT is a neoplasm characterized by the presence of odontogenic epithelium as seen in ameloblastoma with extracellular deposition of amyloid. 14 The tumor has been referred to as calcifying epithelial odontogenic tumor (CEOT) in the literature, because of the suggested similarity to human CEOT. 14,29,46 However, canine APOT has morphologic features that differ in several respects from those of human CEOT. 11 Thus, amyloid-producing odontogenic tumor (APOT) was proposed as a more appropriate name for the canine neoplasm. 9,11,14
The origin of amyloid in human CEOT and animal APOT has been studied. 19,20,29,32,37,42,43 The amyloid of canine and feline APOT did not react immunohistochemically with antibodies to human cytokeratins, human vimentin, chicken desmin, laminin, human collagen (types I, III, IV), or amyloid A; the neoplastic epithelial cells reacted only with anti-human cytokeratins. 29 Breuer et al 2 evaluated animal APOTs immunohistochemically with a panel of antibodies to amyloid A (AA protein), immunoglobulin light chain protein in amyloidosis (AL protein), amyloid protein in familial amyloidotic polyneuropathy (AF protein), amyloid fibril proteins in senile cardiac amyloid (ASc1 protein), amyloid β protein in senile plaque amyloid (AB protein), cytokeratins, vimentin, desmin, and laminin and suggested that the amyloid in CEOT was epithelial in origin based on the exclusive immunoreaction to anticytokeratin and antilaminin 2 Another investigator reported that the neoplastic epithelial cells expressed cytokeratins AE1/AE3, neuron-specific enolase, and S-100 protein but that the amyloid deposits did not express cytokeratins AE1/AE3. 46
Basement membrane components 37 and cytokeratins 1,19,42 have been proposed as the origin for the amyloid; however, in another study, the amyloid lacked immunoreactivity for basement membrane components (laminin 1 and 5, collagen type IV, fibronectin) or pan-cytokeratins. 32 Immunohistochemical detection of amelogenin in the human CEOT showed only feeble reactivity to intercellular amyloid-like material. 20,27 Takata et al 42 examined the immunohistochemical localization of sheathlin in 5 cases of the human CEOT; neither the amyloid nor the neoplastic cells reacted with antisheathlin antibody. 42
In the present study, the immunohistochemical characteristics of amyloid in canine APOT are clearly different from those in human CEOT. Immunohistochemistry with antibodies to enamel proteins has not been applied to odontogenic tumors of animals. Ameloblastoma is thought to be derived from ameloblasts, based on the histologic features of the odontogenic epithelium, such as peripheral palisading with apical nuclei and basilar cytoplasmic clearing, and prominent intercellular bridges in the centers of nests of neoplastic cells. 14 Ameloblasts are odontogenic epithelial cells that produce enamel proteins; these proteins can be demonstrated immunohistochemically in the cytoplasm of ameloblasts and in the enamel of developing tooth germs. 42 Therefore, immunohistochemistry using antibodies to enamel proteins is recommended to study the pathogenesis of odontogenic tumors.
We conclude that the precursor protein of amyloid fibrils in canine APOT may be derived from enamel proteins produced by ameloblasts. Thus, we propose that the canine amyloid-producing odontogenic tumor (APOT) would more properly be named canine amyloid-producing ameloblastoma (APA).
Footnotes
Acknowledgements
We thank Dr. Takashi Takata, the Department of Oral Pathology, and Dr. Takashi Uchida, the Department of Oral Anatomy, Hiroshima University, for providing the ameloblastin, sheathlin, and amelogenin antibodies; Dr. Tomotaka Shinoda for isolation of the amyloid material; and Dr. Tohru Takahashi, Takahashi Animal Hospital, Hokkaido, and Dr. Mitsuru Kuwamura, Osaka Prefecture University, Osaka, for the offer of the materials.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
