Abstract
A 10-year-old, male, Shih-Tzu dog presented with swelling of the right lower jaw caused by a mass arising from the right mandibular gingiva. Radiographic examination revealed bone lysis of the right wing of the mandible. Histopathologically, the growth was characterized by indistinctly lobulated nests, islands, and strands of proliferating odontogenic and squamous epithelial cells, intermingled in close association with large numbers of irregular extracellular deposits of amyloid and amorphous calcified substance. Immunohistochemically, both epithelial components stained strongly positive for cytokeratin (AE1/AE3); the squamous epithelial cells also reacted strongly with neuron-specific enolase (NSE) and S-100 protein, whereas the odontogenic epithelial cells displayed weak immunoreactivity to NSE and partial reactivity to S-100 protein. The amyloid deposits were AE1/AE3-negative. The growth was diagnosed as an amyloid-producing odontogenic tumor.
A 10-year-old, male, Shih-Tzu dog had developed a sublingual gingival mass over the right mandible about half a year ago. It grew fast recently and caused significant swelling of the right lower jaw. The dog had normal spirit and appetite, but nausea was noted by the owner about once a day, while the mass gradually enlarged recently. Oral examination revealed the loss of the first premolar tooth with the second and third ones partially dislocated and embedded in the gingival growth. The mass was approximately 3.0 × 2.7 × 2.2 cm and almost occupied the entire sublingual space (Fig. 1, inset ). Radiographic examination revealed 2 indistinct irregular radiopaque foci in the lower jaw with bone lysis in the right wing of the mandible. The mass was partially excised and submitted for pathologic examination. Histopathologically, the mucosal epithelium covering the gingival growth was markedly thickened with formation of prominent irregular down-growing and anastomosing cords. The lamina propria of mucosa contained some irregularly dilated lymphatics and was somewhat compressed by the underlying partially demarcated and nonencapsulated tumor growth (Fig. 1). The growth was composed of indistinctly lobulated irregular nests, islands, and strands of proliferating epithelial cells supported by varying amounts of fibrous stroma. Some of the epithelial cells were polyhedral with abundant deeply eosinophilic cytoplasm and evident intercellular bridges, resembling squamous epithelial cells (Fig. 2); while others were arranged in a palisading pattern and had relatively little but more basophilic cytoplasm resembling the odontogenic epithelial cells (Fig. 3). In some areas, the fibrous stroma was in a relatively loose arrangement and sometimes cradled by the palisading odontogenic epithelial cells in a cap-shaped pattern (Fig. 3). Small cysts lined by flattened odontogenic epithelial cells were also occasionally observed. In addition, there were many variably sized globular or nodular deposits of a homogenous eosinophilic substance. These deposits stained strongly positive to Congo red and displayed an apple green birefringence under polarized light (Fig. 4). They were distributed in the stroma in close association with the proliferating epithelial components, especially the odontogenic epithelial cells (Fig. 3). These deposits were thus considered as amyloid. Spherical to amorphous calcified corpuscles and discrete to confluent calcification, confirmed by von Kossa's staining, were frequently seen in the amyloid deposits and fibrous stroma. Based on the histopathologic findings, the gingival growth was diagnosed as an amyloid-producing odontogenic tumor.
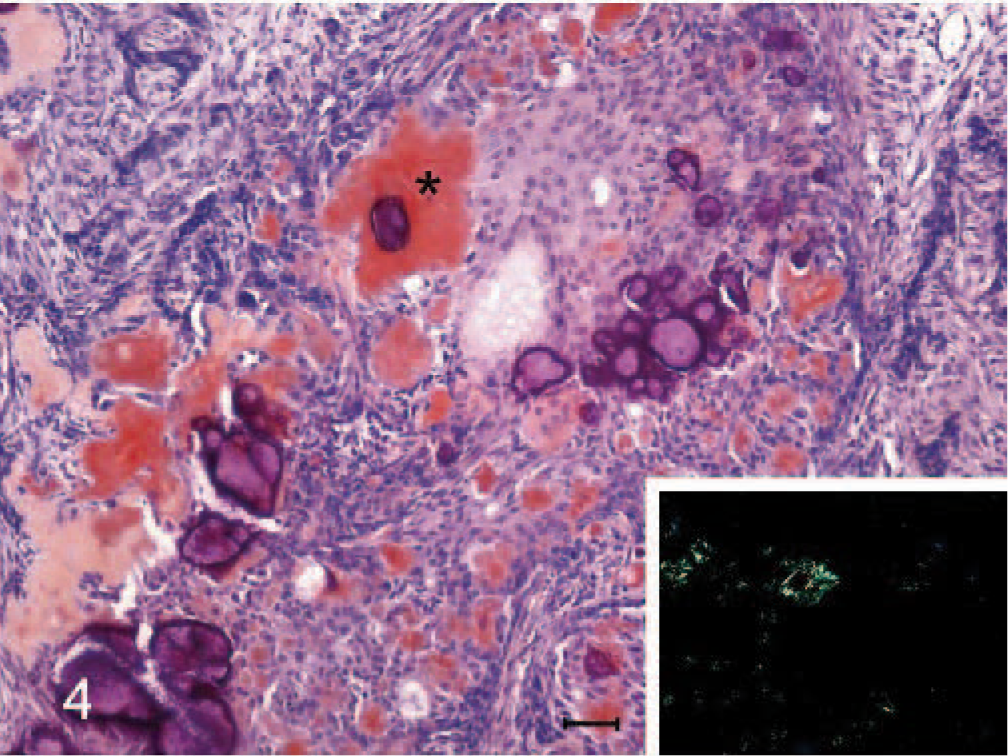
Amyloid-producing odontogenic tumor, right mandibular gingiva; dog. The deposits of the homogenous eosinophilic amyloid-like substance (∗) stain positively to Congo red. Congo red stain. Bar = 30 μm. Inset: The typical apple green birefringence of the Congo red–positive deposit under polarized microscope. Congo red stain.
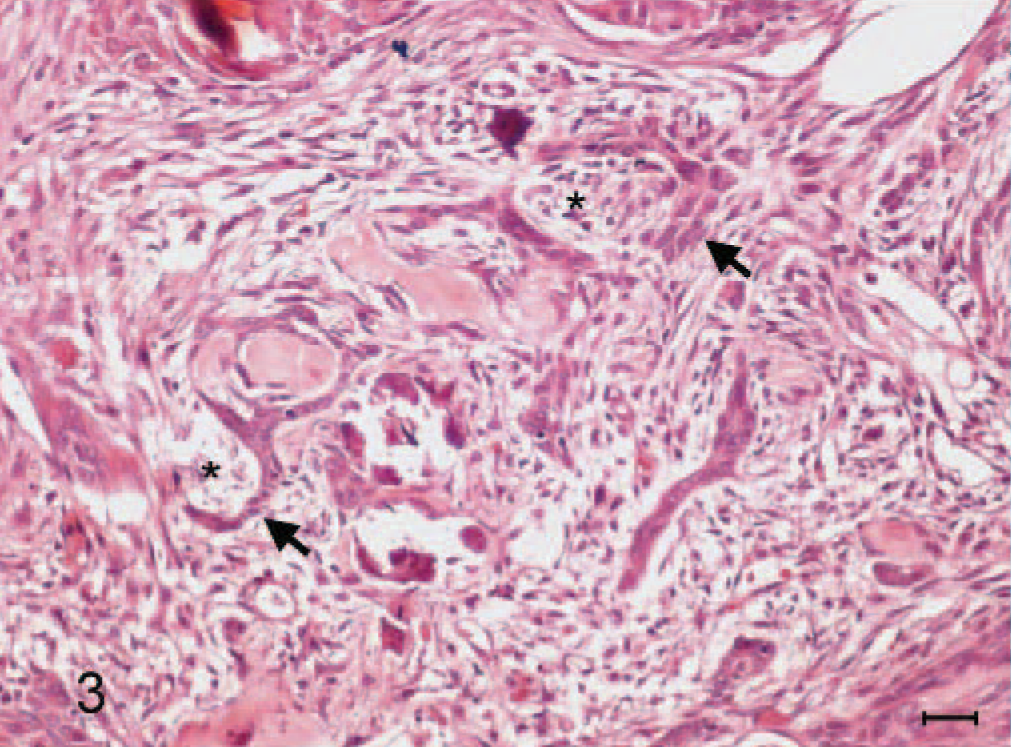
Amyloid-producing odontogenic tumor, right mandibular gingiva; dog. Loosely arranged fibrous stroma (∗) is sometimes cradled by the palisading hyperchromatic odontogenic epithelial cells (arrows). HE stain. Bar = 20 μm.
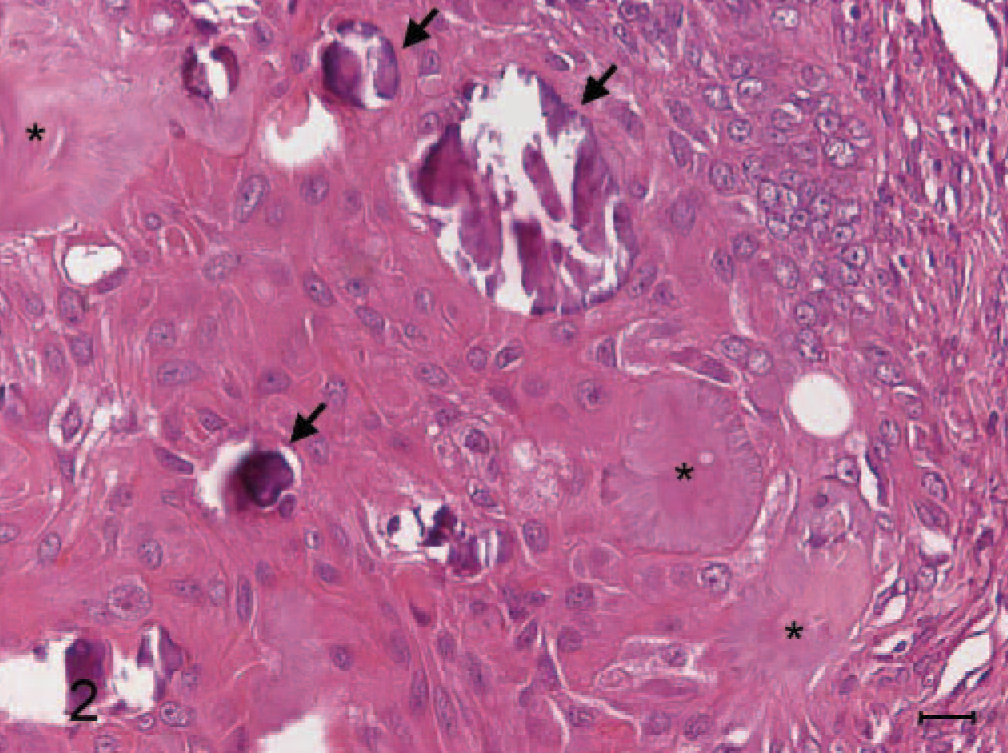
Amyloid-producing odontogenic tumor, right mandibular gingiva; dog. An island of proliferating epithelial cells that are polyhedral with abundant deeply eosinophilic cytoplasm and evident intercellular bridges, resembling squamous epithelium. Areas of calcification (arrows) are seen in close association with the epithelial component. Several islands of amyloid (∗) can also clearly be seen. HE stain. Bar = 15 μm.
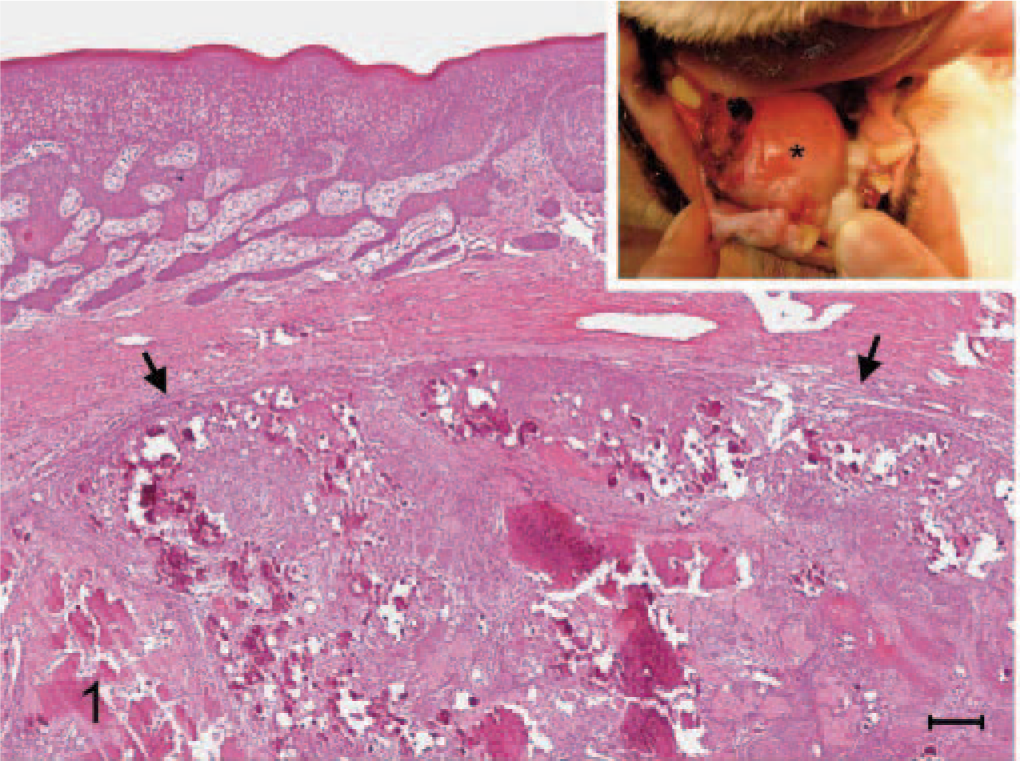
Amyloid-producing odontogenic tumor, right mandibular gingiva; dog. The growth (arrows) is partially demarcated from the overlying mucosa and contains indistinctly lobulated nests, islands, and strands of proliferating epithelial cells with numerous deposits of amorphous calcified substance. The surface mucosal epithelium is markedly thickened. HE stain. Bar = 100 μm. Inset: The gingival mass (∗), 3.0 × 2.7 × 2.2 cm, occupies almost the entire sublingual space.
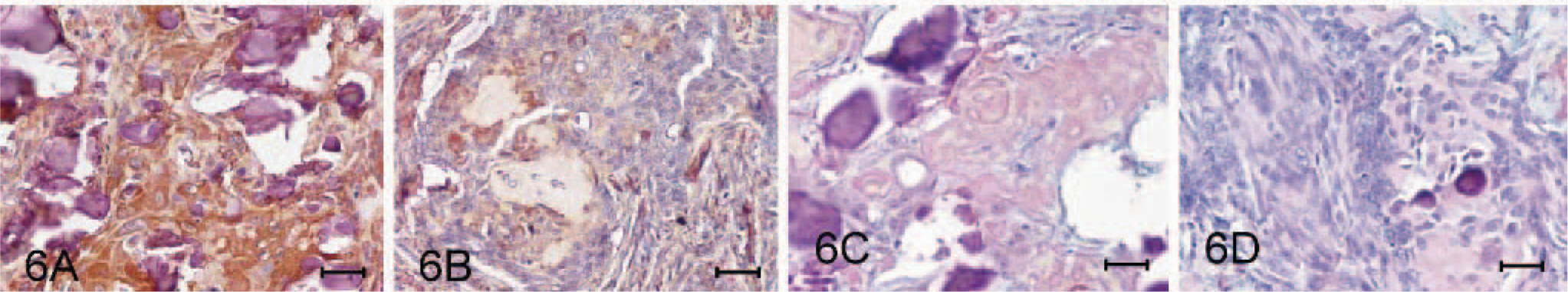
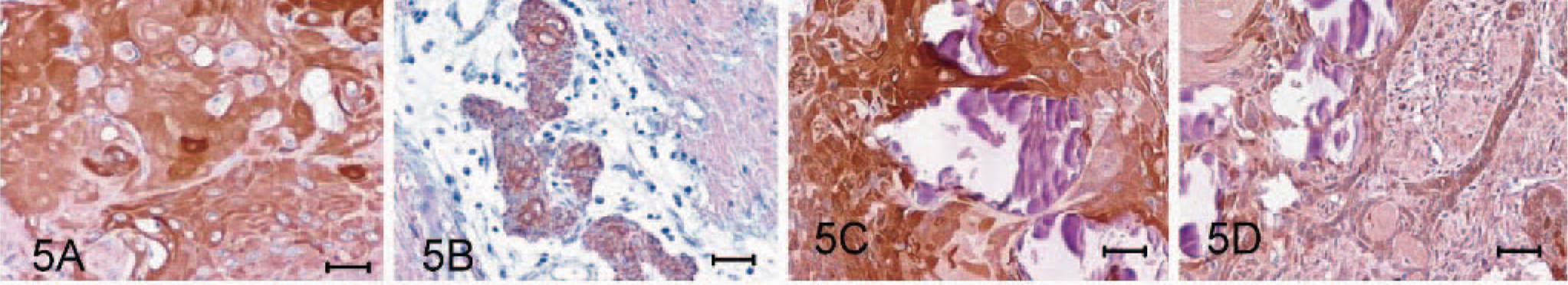
Immunohistochemical staining using a 1:50 dilution of mouse anti-human cytokeratin (AE1/AE3) (Dakocytomation, Glostrup, Denmark), 1:150 dilution of rabbit anti-human S-100 protein (Biomeda, Foster city, CA), 1:100 dilution of rabbit anti-bovine neuron specific enolase (NSE) (Biomeda), or 1:100 dilution of mouse anti-human glial fibrillary acidic protein (GFAP) (Dakocytomation) was performed on sections of formalin-fixed paraffin-embedded tissues with the avidin-biotin complex method (Vectastain Kit, Vector Labs, Burlingame, CA). Diaminobenzidine (DAB) was used as the chromagen (Sigma Chemical Co., St. Louis, MO). Tissue samples of normal gingiva and brain from a dog dying from other disease were taken as the positive and negative controls for immunostaining. Both squamous and odontogenic epithelial components of the growth stained strongly positive for cytokeratin (AE1/AE3) (Fig. 5A, B), while the cells cradled by the odontogenic epithelial cells were not. No reactivity was seen in the amyloid deposits. The squamous epithelial cells also stained intensively for NSE (Fig. 5C) and S-100 protein (Fig. 6A); however, those odontogenic epithelial cells displayed weak and weak-to-no reactivity to NSE (Fig. 5D) and S-100 protein (Fig. 6B), respectively. Both epithelial components were completely negative for GFAP (Fig. 6C, D).
Amyloid-producing odontogenic tumor, right mandibular gingiva; dog.
Amyloid-producing odontogenic tumor, right mandibular gingiva; dog.
Amyloid-producing odontogenic tumor (APOT), an epithelial odontogenic tumor, is a rare and poorly understood oral neoplasm in animals.6,12,15 This tumor may be presented as an epulis clinically.5 It is histologically benign but usually locally invasive and can cause destruction of bone and displacement of teeth. Thus, it is usually considered as low grade of malignancy and rarely metastasizes. Similar clinical behaviors were also seen in the present case. After the surgery, the rostral one fourth of the right mandible was loosened resulting in fracture at the mandibular symphysis and destabilization of the mandible. The dog is currently alive with a reasonable life quality.
On the basis of the inductive interactions between dental epithelium and mesenchyme, epithelial odontogenic tumors can be classified into 2 groups, the one with no induction of mesenchymal tissue and the one with induction of mesenchymal tissue. Ameloblastoma, adenomatoid ameloblastoma, and calcifying epithelial odontogenic tumor (CEOT) are considered to be noninductive, while the mesenchymal induction is evident in ameloblastic fibroma, dentinoma, ameloblastic odontoma, complex odontoma, and compound odontoma.15 Amyloid-producing odontogenic tumor, which has only been reported in animals,6,9 was originally referred to as the counterpart of human CEOT.1,15 However, as the number of the reported APOT cases increased, the histopathologic differences between these 2 types of tumors became distinct, and some of the canine tumors reported as CEOT previously have been rediagnosed as APOT by others.6,9 The dominant microscopic differences between these 2 distinct tumors are in the proliferating epithelium and mesenchyme. In CEOT, the epithelial component is composed of sheets of eosinophilic epithelial cells that display significant nuclear pleomorphism with no inductive change in connective tissue;6,10 while palisading arrangement of more basophilic cells accompanied with proliferation of closely associated pulp-like mesenchyme are seen in APOT.1,4,6 In the present dog, no nuclear pleomorphism seen in the proliferating squamous epithelial cells in conjunction with the characteristic cap-arrangement of the palisading hyperchromatic odontogenic epithelial cells and formation of mesenchyme among the epithelial components led to the final diagnosis of APOT.
As described first by Pindborg in 1958,11 CEOT was initially considered to be odontogenic origin and to possibly develop from the immature enamel organ of the unerupted tooth based on the observation that the tumor cells displayed characteristics of squamous epithelium.4 Later on, other possibilities, including arising from rests of the dental lamina or from the basal cells of oral epithelium, were also suggested for those cases of CEOT without unerupted teeth.7 More recently, dental lamina complex or its remnants have been suggested as a more suitable general source for CEOT.10 As for APOT in animals, in the literature, there is a lack of complete dissection and enough descriptions regarding whether there is any reminiscent tooth. In the present dog, no tooth component was found in the partially removed tumor growth, and it is not known whether there was any in the remaining unremoved portion of the growth.
During the cap stage development of the tooth, the enamel organ differentiated from the dental lamina becomes cap-shaped, and the cells lining the concave face of the enamel organ in contact with the dental papilla begin to differentiate into the future ameloblasts. As the enamel organ develops further, the cells forming the main bulk of the enamel organ become enlarged and encircled by the ameloblasts. The cell arrangement seen in the present case also somewhat resembles the arrangement of the cap stage of tooth development. Whether this resemblance implies that APOT may also develop from the dental lamina complex or its remnants as suggested for CEOT requires further investigation.
It is known that tooth is derived from the neural crest, which is a group of neural precursor cells in the embryologic stage.14 However, to our knowledge there has been no report providing information about the immunohistochemical staining characteristics of the neural tissue markers such as NSE, GFAP, and S-100 protein in animal odontogenic tumors. In the present dog, both squamous and odontogenic epithelial cells reacted to the anti-NSE and anti-S-100 protein antibodies but not to the anti-GFAP antibody; the more differentiated squamous epithelial cells displayed stronger reactivities to the antibodies of both neural tissue markers, whereas the reactivity of odontogenic epithelial cells for S-100 protein was only limited in some areas. Normal tooth development results from interactions between primitive oral epithelium and neural crest cell–derived mesenchyme. All tissues of the tooth, except enamel, and its supporting apparatus are derived from the neural crest; evidence has shown that certain neural tissue proteins produced by neural crest–derived cells may participate in epithelial-mesenchymal interactions in tooth morphogenic events.13 Expression of neural tissue markers, including NSE, GFAP, and S-100 protein, has been demonstrated in human odontogenic tumors and normal tooth differentiation.13 Thus, it is speculated that the different immunoreactivities seen in the present dog may be due to different stages of differentiation of the proliferating epithelial cells.
The present study demonstrated, for the first time, the expression of neural tissue markers in animal odontogenic tumor. Although the immunohistochemical staining result cannot prove directly the involvement of neural crest–derived cells in the histogenesis of APOT, it at least suggests that under certain pathologic conditions, the expression of neural proteins does occur in the process of odontogenic tumor development in animals.
The amyloid component seen in animal odontogenic tumors has not been thoroughly examined. Whether it is a secreted substance or a degenerative product remains controversial. However, it has been demonstrated that the amyloid deposits in human CEOT are reactive to anticytokeratin antibodies, including cytokeratins 1, 3, 6, 8, 13, 16, and cytokeratins AE1 and AE3;2 the amyloid in animal CEOT does not react with amyloid-specific antibodies but produces a low to medium–grade reaction with anticytokeratin and antilaminin antibodies;3 and it has been suggested that the amyloid is of epithelial origin2,3 and may derive from degenerative keratin filaments originating from the tumor squamous epithelium.2 The amyloid component in a more recently reported APOT from a Bengal tiger was also cytokeratin-positive,8 while in our dog the amyloid deposits showed no reactivity for cytokeratins AE1 and AE3. Thus, the association between amyloid and epithelial cells or keratin requires further elucidation in animal APOT.
The present dog provides another classic, but rare, APOT in animal and in conjunction with other reports may provide more information for veterinary pathologists to diagnose and understand the possible origin of this rather unique tumor.
Footnotes
Acknowledgement
We are grateful to Miss H. C. Wang in Animal Technology Institute Taiwan for assistance in immunohistochemistry.
