Abstract
The present study describes the pathologic changes and cellular apoptosis in the central nervous system (CNS) of fetal and neonatal small ruminants infected with border disease virus (BDV), as demonstrated by immunohistochemistry and in situ hybridization. Abortions of ewes and goats were observed, as were births of lambs and kids with poor survival rates and nervous signs. Lesions included cerebellar hypoplasia, porencephaly, hydranencephaly, and nonsuppurative meningoencephalomyelitis with hypomyelinogenesis. Viral antigens and RNA were present in neuropil, glial, and neuronal cells, especially in periventricular areas, cerebellum, and brainstem. TUNEL positivity and labeling of anti-bax and anti-caspases 3, 8, and 9 were detected in BDV-infected CNSs, especially in glial and neuronal cells. The double immunostaining and TUNEL assay revealed that in BDV-infected animals, not only were BDV-infected glial and neuronal cells undergoing apoptosis, but so were uninfected cells in close vicinity of BDV-infected cells. The expression of activated caspases 3, 8, 9; bax; and TUNEL in glial and neuronal cells of the infected fetal and neonatal kids were significantly (P < .05) higher than those of the infected fetal and neonatal lambs. Yet, the expression of bcl-2 in the CNSs of the infected fetal and neonatal lambs was higher (P < .05) in neuronal and glial cells than in those of the infected fetal and neonatal kids. The results suggest that cell death in the BDV-infected CNS is induced by intrinsic and extrinsic cascades of apoptotic pathways.
Border disease, first reported in 1959 from the border region of England and Wales, is a congenital infectious viral disease affecting sheep and occasionally goats. 6,8,10,14–16 Border disease virus (BDV), closely related to bovine viral diarrhea virus and classical swine fever virus, is classified in the genus Pestivirus of the family Flaviviridae. 13 BDV infections have been reported worldwide and so lead to significant losses in the small ruminant population. 8,10,24,25,28
In October 2004, we encountered newborn twin lambs showing nervous clinical signs and abnormal fleece, with long and straight birth coats, in a sheep flock in Aydin-Kuyucak, Aegean region of Turkey. Pathologic and immunohistochemical examinations indicated pestivirus infection in the lambs. Pestivirus isolated from the lambs was characterized as BDV by sequencing of reverse transcriptase polymerase chain reaction (RT-PCR) products. Phylogenetic analysis using fragments of the 5′-NTR and Npro of the isolates revealed that the viruses formed a distinct cluster as a BDV-7, 19 which is characteristic for certain geographic regions, much like what has been found for classical swine fever virus. The aim of the present study was to understand the pathogenesis of BDV in infected brain tissues of the fetal and neonatal small ruminants by analyzing apoptosis and cellular tropism of the virus.
Materials and Methods
History and Animals
Animal materials were collected from 5 flocks at different localities in the Aydin and Mugla provinces of the Aegean region of Turkey, which were generally vaccinated against brucellosis, enterotoxemia, and foot-and-mouth disease. Infertility, abortion, stillbirth, and neonatal mortality with nervous signs such as ataxia, paresis, paralysis, and involuntary muscular tremors were the most conspicuous findings. Additionally, abnormal fleece, with long and straight birth coats (hairy shaker), was observed in many sick lambs. A total of 21 small ruminants were obtained from these flocks (Table 1 )—namely, from 1 farm with sheep and goats (flock A), 3 sheep farms (flocks B, C, D), 1 goat farm (flock E). Of the lambs, 8 were neonates and 5 were fetuses. Of the kids, 5 were neonates and 3 were fetuses. For control purposes, 10 animals (3 neonatal and 2 fetal lambs; 3 neonatal and 2 fetal kids) that had died at 3 to 5 days of age or were aborted from other causes and did not have brain lesions were obtained from flocks that were considered uninfected by pestivirus, based on negative serology. These animals had no evidence of pestivirus or bluetongue virus in brain, lymphoid organs, and lungs by RT-PCR and immunohistochemistry (IHC).
Comparison of Immunohistochemistry and In Situ Hybridization for Severity and Distribution of Border Disease Virus Antigen and Nucleic Acid in the Brain Tissues a
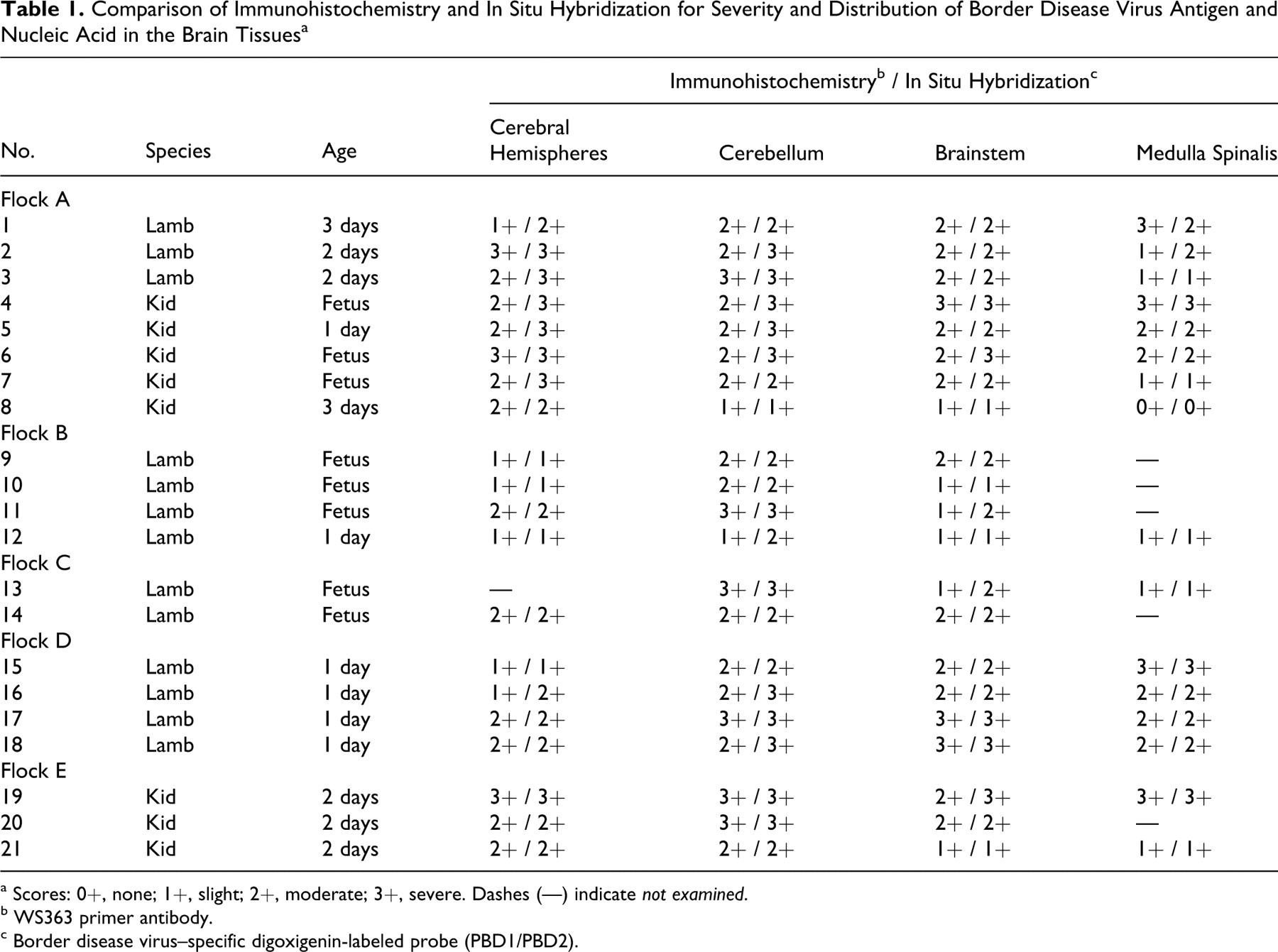
a Scores: 0+, none; 1+, slight; 2+, moderate; 3+, severe. Dashes (—) indicate not examined.
b WS363 primer antibody.
c Border disease virus–specific digoxigenin-labeled probe (PBD1/PBD2).
The animals chosen for detailed study were subjected to necropsy. Following the gross examination, tissue samples were fixed in 10% neutral buffered formalin, dehydrated in ethanol, embedded in paraffin wax, sectioned at 5 μm, and stained by routine methods with hematoxylin and eosin (HE). Replicated sections were used for immunoperoxidase labeling, and selected brain sections were used for in situ hybridization (ISH) and detection of apoptosis.
In Situ Hybridization
BDV reference Moredun cytopathic was used for preparation of the digoxigenin-labeled (DIG-labeled) probe. First, the 5′-NTR of the pestivirus genome from BDV Moredun was amplified with panpesti-generic primers (324/326), 26 and a 225–base pair BDV-specific amplicon was obtained by RT-PCR with a pair of primers, PBD1 and PBD2. 27 The RT-PCR products were purified using a cleanup kit (GenElute PCR Clean-Up Kit NA1020, Sigma, St Louis, MO). The template DNAs were labeled with the PCR DIG Probe Synthesis Kit (No. 11636090910, Roche, Mannheim, Germany). After labeling, the products were analyzed in 1% agarose–ethidium bromide gels in Tris–acetate buffer and visualized on a ultraviolet transilluminator.
ISH was performed on each brain section with the DIG-labeled probes (panpesti-generic primers 324/326; BDV-specific amplicon PBD1/PBD2) and the ISH detection system core kit (DD130-60K, Biogenex Technology, San Ramon, CA). The optimal conditions, including probe dilutions and hybridization temperatures, were determined by titration experiments. Paraffin sections (5 μm) on poly-L-lysine-coated glass slides were dewaxed in xylene and rehydrated through a graded series of alcohol; the slides were then washed for 5 minutes in sterile deionized water containing 0.2% RNase block. Tissues were then digested at 37°C for 20 minutes in proteinase K (Roche), 200 mg/ml in phosphate-buffered saline (PBS; pH 7.3). One of the serial sections was treated with RNase A at 100 mg/ml in 10mM Tris-HCl (pH 7.4) for 30 minutes at 37°C, to remove target RNA as a specificity control. After digestion, tissues were fixed in 4% paraformaldehyde in PBS, RNase-free, for 10 minutes. After rinsing with deionized water with RNase block, the slides were washed again in RNase-free deionized water for 5 minutes. Hybridization was carried out overnight in a humidity chamber at 42°C in a mixture containing heat-denatured DIG-labeled probes (approximately 60 ng/μl) in hybridization solution. Posthybridization washes comprised washing twice in 2× PBS with 0.1% Tween-20 and once with 1× PBS with 0.1% Tween-20, before blocking with 1% blocking solution and IHC using biotinylated anti-DIG antibody (room temperature for 30 minutes). After washing with 1× PBS with 0.1% Tween-20, the sections were covered with streptavidin–peroxidase and incubated for 30 minutes. Finally, the immunoreactivity was revealed with 0.08% (w/v) 3,3-diaminobenzidine tetrahydrochloride (DAB, Sigma) and 0.045% (v/v) H2O2 in PBS. The slides were lightly counterstained with Mayer’s hematoxylin.
Immunohistochemistry
IHC was performed on 5-μm paraffin sections using the standard avidin–biotin–peroxidase complex (ABC, DakoCytomation, Glostrup, Denmark) method. We used monoclonal antibodies against mouse anti-pestiviruses group-specific antigen (WB103/105 nonstructural protein 2-3, CVL, PA0801) and mouse anti-BDV-specific antigen (WS363 envelope rns, CVL, PA0825). For detection of apoptosis signals, sections of BDV-infected and uninfected brains were labeled with mouse anti-bax, mouse anti-bcl-2, and mouse anti–caspases 3, 8, and 9 antibodies (Santa Cruz Biotechnology Inc, Santa Cruz, CA). Mouse anti–glial fibrillary acidic protein antibody (Sigma) was also applied to detect gliosis or loss of astrocytes. Negative control tissue sections were incubated with normal mouse serum.
Terminal Deoxynucleotidyl TUNEL Assay
Labeling of apoptotic cells in sections of BDV-infected and uninfected brains were performed with a TUNEL (transferase-mediated dUTP nick end labeling) assay (in situ cell death detection kit; POD, Roche). Briefly, the paraffin sections were dewaxed and hydrated to water before digestion with Proteinase K (20 μg/ml; Roche) for 15 minutes at room temperature. Slides were then treated with 3% (v/v) H2O2 for 5 minutes to quench the endogenous peroxidase activity before incubation with 50 ml of the TUNEL reaction mixture containing terminal deoxynucleotidyl transferase and fluorescein–dUTP (29-deoxyuridine 59-triphosphate) in a humidified chamber at 37°C for 1 hour. The sections were checked by fluorescence microscopy. The reaction was stopped by washing slides in PBS. After that, slides were incubated with anti-fluorescein–POD conjugate diluted 1:3 in 100mM Tris-HCl, 150mM NaCl (pH 7.5), and 1% blocking reagent for 40 minutes at room temperature. After washing 3 times for 15 minutes in PBS, the sections were stained by incubation with the chromogenic substrate DAB for 5 to 15 minutes at room temperature. Negative controls included the substitution of the TUNEL reaction mixture for label solution without terminal transferase in sections both infected and uninfected with BDV.
Double Staining: IHC and TUNEL
A double-staining method that combined IHC (to detect BDV antigen) and TUNEL assay was performed on BDV-infected brain paraffin sections to demonstrate colocalization of viral antigen and TUNEL positivity. For this purpose, the TUNEL assay was applied first with horseradish peroxidase as conjugated enzyme and with DAB as chromogen, followed by detection of BDV antigen based on ABC method as conjugated enzyme and 3-amino-9-ethylcarbazole as the chromogen. ABC and TUNEL assay protocols were as described above.
Data Analysis
The scores of IHC and ISH were based on the proportion of positively stained cells observed in a total of 10 high-power fields measured at 400×. The scores were as follows: 0+, absent; 1+, weak immunopositivity (1–3% positive cells); 2+, moderate immunopositivity (4–9% positive cells); 3+, strong immunopositivity (> 10% positive cells).
Semiquantitative analysis of apoptosis signals in the BDV-infected and uninfected cells of the CNSs of 21 animals (13 lambs and 8 kids) were assessed with a grading system based on the total number of positive cells. For quantification of immunostaining for anti-bcl-2; anti-bax; anti–caspases 3, 8, and 9; and TUNEL assay, the BDV-infected and uninfected areas with the highest density of positive staining were chosen (cerebellum, cerebral hemispheres, brainstem, and spinal cord). All positive cells for TUNEL assay and apoptosis markers from a total of 10 high-power fields measuring 0.020 mm2 were counted. To calculate the total number of positively stained cells, an ocular grid of 100 (10 × 10) squares was used at 400× magnification.
Independent-sample t test for statistical analyses was performed with SPSS 10.0 to examine apoptosis markers among BVD-infected and uninfected animals. Statistical significance was set at P < .05.
Results
Gross Lesions and Histopathologic Findings
At necropsy, fetal lesions in the brain included porencephalic cysts in the cortex of the cerebral hemispheres (case Nos. 4, 5, 6, and 19), cerebellar hypoplasia (case Nos. 4, 5, 13, 15, 16, 19, and 20), hydranencephaly (case No. 13), and hydrocephalus (case No. 15).
The most consistent and severe microscopic changes were in the periventricular areas, cerebellum, brainstem, and cerebral hemispheres and occasionally in medulla spinalis, as characterized by nonsuppurative and/or necrotizing meningoencephalomyelitis and often accompanied by hypomyelinogenesis. Periventricular leukomalacia with gliosis and infiltration of mononuclear cells in ependyma throughout the cerebrospinal axis were conspicuous. IHC with anti–glial fibrillary acidic protein antibody exhibited no significant changes in astrocyte distribution and frequency in sections of CNSs, except for periventricular areas. However, a slight alteration of the blood-brain barrier was detected in vessels containing a strong perivascular infiltrate, with a loss of contact between astrocyte cytoplasmic processes and vessel walls.
The frontal lobes of the cerebral hemispheres, thalamus, hypothalamus, periventricular areas, and brainstem had focal or multifocal malacia (some of which included dystrophic mineralization) within the gray and white matter. The parenchyma adjacent to the malacic lesions showed neuronal loss, neuronal eosinophilia or neuronal necrosis with dystrophic mineralization, mononuclear inflammatory reactions, focal or diffuse gliosis, and neuronophagia. Hypoplastic cerebella had the most severe necrotic and dysplastic changes, with Purkinje cell loss and focal-to-diffuse depletion of granule cells. Hypoplastic cerebella had areas of disorganization of the cellular layers of cerebellar cortex, reduction or rarefaction of the internal granular layer associated with degeneration and necrosis of granule cells, and widening of the normally thin external granular layer. In some cases, stratification in the folia was not recognizable because of partial or complete destruction of the cerebellar cortex, loose arrangement of Purkinje cells (necrotic or with dystrophic mineralization), and ectopic granule cells.
Distribution of Viral Nucleic Acid and Antigens in the Brain Sections
Table 1 presents the results. ISH and IHC revealed that tissue localization of viral nucleic acid and antigen was complementary to the histopathologic findings. Results of ISH with both DIG-labeled probes and IHC with pestivirus group–specific antigen (WB103/105) and BDV-specific antigen (WS363) showed a correlation between the intensity and distribution of viral nucleic acid and viral antigen; however, ISH showed more positive cells in some areas.
The labeling of viral RNAs and antigens was mainly detected in neuronal cytoplasm and cell processes of the cerebral hemispheres, brainstem, and spinal cord (Fig. 1 ). ISH and IHC labeling was also found in glial and ependymal cells, especially in periventricular areas (Fig. 2 ). In the cerebellum, labeling of viral RNAs and antigens was observed in the cytoplasm and cell processes of Purkinje cells, in neurons of the molecular and granular layers, and in glial cells of the substantia alba (Fig. 3 ). Linear labeling of viral RNA and antigen extended from meningeal and/or parenchymal vessels to neuropil, particularly in the cerebral hemispheres, cerebellum, and brainstem. There was no IHC or ISH labeling in the control slides.
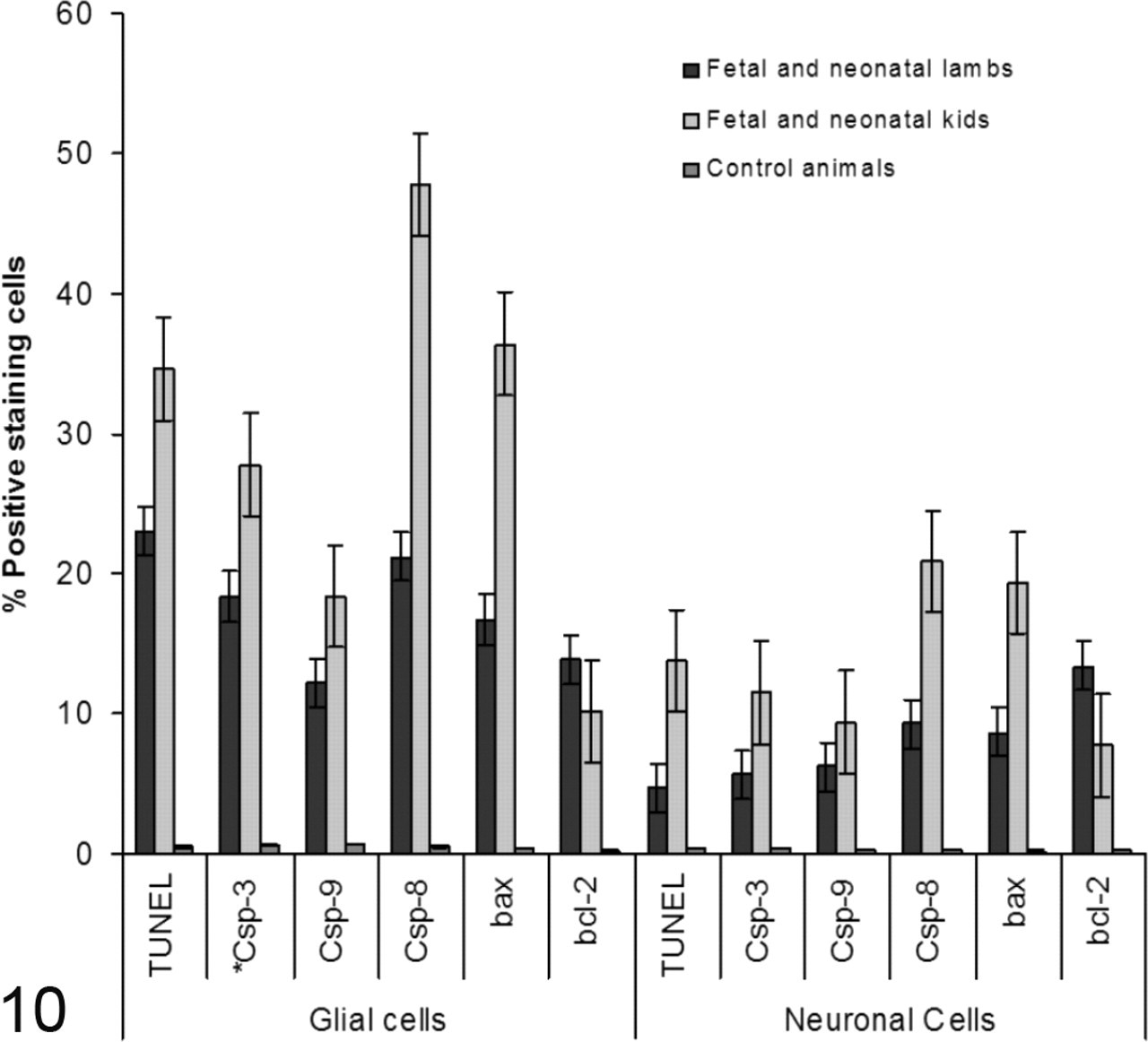
Semiquantitative analysis of TUNEL; bax; bcl-2; caspases 3, 8, and 9 levels (mean ± SEM) in border disease virus–infected and uninfected cells of the brain sections of infected animals (n = 21; 13 lambs and 8 kids) and uninfected control animals (n = 10; 5 lambs and 5 kids). *Csp, caspase.
Demonstration of Apoptosis
The TUNEL staining revealed a distinct population of the cells that labeled for DNA fragmentation in the CNSs of BDV-infected animals. TUNEL positivity was predominantly in the nucleus of the glial cells, mainly in the brainstem, cerebellum, and cerebral hemispheres and, to a lesser extent, medulla spinalis (Fig. 4 ). In some cases, TUNEL staining was diffuse and/or globular (resembling apoptotic bodies formed by fragmented nuclear material) in the cytoplasm of glial cells, especially in the cerebellum. Based on the morphologic appearance of the glial cells, the TUNEL reactivity was mainly in astrocytes and oligodendroglia and, to a lesser extent, microglia.
TUNEL positivity was weaker in neuronal cells than in glial cells. The TUNEL assay exhibited strong positivity in the granule cells of the external and internal granular layers, in the ectopic granule cells in the molecular layer, and occasionally in the Purkinje cells in the cerebellum of both species. TUNEL positivity was especially found in neurons of the brainstem. Additionally, TUNEL positivity was detected in meningeal cells and infiltrated mononuclear cells of the meninges, as well as in endothelial cells in inflamed areas. No TUNEL reactivity was observed in cells of perivascular infiltrates. In contrast, only a few positive cells could be detected in the brain tissues of uninfected animals. Uninfected animals had significantly (P < .001) fewer apoptotic cells in all regions examined than did infected animals. In sections of BDV-infected and uninfected brains, the substitution of the TUNEL reaction mixture with labeling solution without terminal transferase gave negative results.
The double immunostaining and TUNEL assay in BDV-infected kids and lambs revealed that not only were BDV-infected glial and neuronal cells undergoing apoptosis, but so were uninfected cells in the vicinity of BDV-infected cells (Fig. 5 ). Yet, absence of TUNEL positivity in BDV-infected glial and neuronal cells in some areas in lambs was conspicuous, whereas its absence in the infected cells observed in kids was restricted with only a few cells. As in the TUNEL assay, labeling of anti-bax and anti-caspases 3, 8, and 9 was found, especially in the glial cells and neuronal cells of BDV-infected regions of the CNS (Figs. 6–9). Figure 10 presents the results, including the mean scores of apoptotic signals in cerebral hemispheres, cerebellum, brainstem, and medulla spinalis of the BDV-infected animals and uninfected control animals. The expression of activated caspase 8 was higher than that of activated caspases 3 and 9 in the glial and neuronal cells of both species infected with BDV. Furthermore, the expression of activated caspases 3, 8, and 9; bax; and TUNEL in the glial and neuronal cells of the infected fetal and neonatal kids were significantly (P < .05) higher than those of the infected fetal and neonatal lambs. In general, the expression of bcl-2 in the CNSs of the infected fetal and neonatal lambs was higher (P < .05) in neuronal and glial cells compared with those of the infected fetal and neonatal kids. The expressions of TUNEL; caspases 3, 8, and 9; bax; and bcl-2 in infected animals were higher than those of uninfected animals. The differences between infected and uninfected animals were statistically significant (P < .001). IHC with normal mouse serum resulted in no labeling of structures in CNS of the BDV-infected animals. Also, IHC with pestivirus antibody did not label CNS tissues of uninfected animals.
Discussion
Apoptosis, as characterized by specific morphological and biochemical changes, is important in neural development and in a variety of neurological diseases, including viral infection. 18,20 Apoptosis is caused by a family of intracellular cysteine proteases termed caspases. Conventionally, apoptosis may be initiated by the extrinsic pathway involving death receptors or by the intrinsic pathway induced by intracellular stimuli that transmit a signal to the mitochondria. 3,9 The extrinsic pathway is mediated by interaction of a ligand with death receptors such as Fas to activate caspase 8. The intrinsic pathway involves an alteration of the mitochondrial membrane potential leading to mitochondrial membrane permeabilization and so results in release of cytochrome c and in activation of caspase 9. In both pathways, the activation of the executioner caspase 3 is the key inducer of downstream effectors of apoptosis. 12
Mechanisms of virus-induced apoptosis are an important aspect of the pathogenesis of viral infections. Although a common defense mechanism of virus-infected cells is the induction of apoptosis to prevent virus replication and production of progeny, little information exists regarding the mechanism by which BDV induces or prevents cell death. For noncytopathic bovine viral diarrhea virus, viral infection does not cause any visible alteration or synthesis of interferon in the infected cells, but it does inhibit apoptotic cell death in vivo as well as in vitro. 5,7,23,29 This situation suggests that evasion of this innate defense mechanism may be crucial for the establishment of persistent infection. 1,22 In contrast, cytopathic bovine viral diarrhea virus results in massive destruction of the cellular structure and induces apoptotic cell death following viral infection. 2,4,11,21,29
The present study clearly demonstrated that apoptosis signals by TUNEL assay were induced in the glial and neuronal cells of fetal and neonatal small ruminants naturally infected with BDV. What is striking is that the many TUNEL-positive glial and neuronal cells containing BDV viral antigens, as demonstrated with the double stain, indicates a potential association between direct viral infection and apoptosis in these cells. The observation of TUNEL-positive cells without viral antigen might indicate a rapid cell death upon infection before detectable viral protein synthesis. Alternatively, as suggested by Bielefeldt-Ohmann et al, 4 in brain tissue that is inflamed as a result of bovine viral diarrhea virus infection, inflammatory mediators elaborated by the infiltrating macrophages and glial cells—including nitric oxide, type I interferon, and other excitotoxic signals—may trigger the caspase-dependent apoptotic cascade, ultimately resulting in neuronal loss, demyelination, and malacia.
For the validity of the TUNEL assay, we tested the specificity of the method by additional immunostaining for enzymes and cleaved proteins that are considered specific for apoptosis, including caspases 3, 8, and 9 and bax. The results display similar localization of TUNEL-positive and activated caspase 3 signals of various cellular elements, suggesting the presence of classical apoptosis in a caspase-dependent manner, affecting glial cells, endothelial cells, and neurons in BDV-infected animals. Yet, activated caspase 3 in occasional neuronal cells may play a role in neuroprotection by mediating a state of preconditioning following sublethal challenges for apoptotic disintegration of the nuclear matrix. 4,17 The demonstration of activated caspase 9 with a distribution similar to that of activated caspase 3 therefore suggests that the intrinsic pathway of apoptosis was most important in the present animals.
This study evaluated the level of caspase 8 and 9 expressions in glial and neuronal cells in kids and lambs, and the results suggest that apoptosis in the CNS involves intrinsic and extrinsic pathways. However, the expression of caspase 8 in glial and neuronal cells in the fetal and neonatal kids was greater than that of the fetal and neonatal lambs. That cytopathic BDV is generally responsible for border disease of kids suggests, as a defensive mechanism against viral infection, the elimination of possible cytopathic BDV-infected neurons by the extrinsic pathway of apoptosis in kids. 4 Yet, the appearance of weak glial and neuronal apoptosis and the strong expression of neuronal bcl-2 in the infected areas in the fetal and neonatal lambs suggests that bcl-2, as an anti-apoptotic protein, plays an important role in protecting neuronal and glial cells infected with possible noncytopathic BDV—in support of the findings of in vitro studies. 1,7 More detailed comparative in vivo and in vitro experimental studies are needed to better understand mechanisms underlying the interaction of cytopathic and noncytopathic BDVs and their hosts.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
