Abstract
The pathogenesis of 4 isolates of turkey-origin reovirus (NC/SEP-R44/03, NC/98, TX/98, and NC/85) and 1 chicken-origin reovirus (1733) was examined by infecting specific pathogen free (SPF) poults. These turkey-origin reovirus (TRV) isolates were collected from turkey flocks experiencing poult enteritis and are genetically distinct from previously reported avian reoviruses. Microscopic examination of the tissues collected from the TRV-infected poults revealed different degrees of bursal atrophy characterized by lymphoid depletion and increased fibroplasia between the bursal follicles. To understand the relationship between virus spread and replication, and the induction of lesions, immunohistochemical staining (IHC) for viral antigen, in situ hybridization (ISH) for the detection of viral RNA, and terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay for the detection of apoptosis in affected tissues was performed. Both IHC and ISH revealed viral antigen and RNA in the surface epithelial cells of the bursa, in macrophages in the interstitium of the bursa and, to lesser degree, in splenic red pulp macrophages and intestinal epithelial cells. Increased apoptosis of bursal lymphocytes and macrophages was observed at 2 and 5 days postinoculation. No lesions were found in tissues from poults inoculated with the virulent chicken-origin strain, however viral antigen was detected in the bursa and the intestine. Although all TRVs studied displayed similar tissue tropism, there were substantial differences in the severity of the lesions produced. Poults inoculated with NC/SEP-R44/03 or NC/98 had moderate to severe bursal atrophy, whereas poults inoculated with TX/98 or NC/85 presented a mild to moderate bursal lymphoid depletion. The lymphoid depletion observed in the bursa appears to be the effect of an indirectly induced apoptosis and would most likely result in immune dysfunction in poults infected with TRV.
Avian reoviruses belong to the Orthoreovirus genus in the Reoviridae family and have been associated with a wide range of disease presentations affecting avian species.28 The vast majority of reovirus characterization has been performed in chickens and/or with chicken origin reoviruses. Diseases associated with avian origin reoviruses include viral arthritis/infectious tenosynovitis, malabsorption syndrome, stunting–runting syndrome, and various disorders characterized by pericarditis/myocarditis, chronic respiratory syndrome, nephritis, and multifocal necrotic hepatitis.1,3,4,8–11,21,25,31,38,47,50 Avian reoviruses have also been associated with immunosuppression24,40 and atrophy of the bursa of Fabricius with characteristic lymphoid depletion and proliferation of connective tissue.23,24,27,29,36,38,48 Studies in turkeys involving experimental reovirus infection have produced viral arthritis2,19,30 and poult enteritis,7,9,20,26,35,42 although descriptions of microscopic lesions caused by turkey-origin reoviruses (TRVs) are limited.
Poult enteritis mortality syndrome (PEMS) is a highly infectious disease of young turkeys characterized by diarrhea, stunting, increased feed consumption, and increased time-to market. In the more severe forms, runting, immune dysfunction, and mortality occur.5 Reovirus has been isolated from turkeys with PEMS9,42 and its milder form, poult enteritis complex.15,44 Recently, several TRVs collected from commercial turkey flocks affected with poult enteritis were shown to be genetically distinct from previously reported chicken reoviruses.15,39 These TRVs caused significant body weight decreases and moderate to severe bursal atrophy in specific pathogen free (SPF) poults and commercial poults.44 In this study, we complement these findings by comparing the microscopic lesions induced by these TRVs in SPF poults and determining the viruses spread and replication and its relationship with the lesions observed. We accomplish this by studying the distribution of viral antigen using immunohistochemistry (IHC), viral RNA using in situ hybridization (ISH), and viral-induced apoptosis using the terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) method.
Materials and Methods
Samples
Tissue samples were collected from SPF Small White Beltsville turkeys inoculated at 2 days of age with 1 of 4 different strains of TRV as described by Spackman et al.44 Briefly, 4 TRV isolates were evaluated: NC/85, TX/98,39 NC/98,15 and NC/SEP-R44/03. A virulent chicken-origin strain, 1733,37 was used as a reference isolate. Birds were divided into groups of 20 and housed in Horsfal isolators with ad libitum access to feed and water. Each group was inoculated with 0.2 ml of the appropriate virus at 2 days of age (approximately 104.0 tissue culture infecting dose [TCID50]/bird) by the intratracheal and oral routes (0.1 ml each route). Sham inoculated control birds were inoculated with sterile 50% DMEM/50% F12 cell culture medium. At 2, 5, 7, and 9 days postinoculation (DPI) 2 birds from each group were euthanized with a CO2 overdose, and the bursa, thymus, spleen, liver, heart, proventriculus, duodenum, pancreas, jejunum, ileum, and ceca were collected and fixed in 10% neutral buffered formalin. Infection status was confirmed by testing all intestinal tissues for virus by real-time reverse-transcriptase polymer chain reaction (RT-PCR).44
Histopathology
Paraffin-embedded tissues were sectioned, mounted, stained with hematoxylin and eosin (HE), and examined by light microscopy. The examiner was blind to the treatment groups for all samples that were evaluated. Lesions were scored as follows: −, no lesions; +/−, minimal; +, mild; ++, moderate; +++, severe.
Immunohistochemistry
A peroxidase IHC technique using convalescent or hyperimmune sera from reovirus-inoculated turkeys or chickens was used to identify reovirus-infected tissues. All procedures were done at room temperature except when noted. Tissue sections were cut (4-μm thick) from paraffin-embedded tissue samples and mounted on charged glass slides (Superfrost/Plus, Fisher Scientific, Pittsburgh, PA). Paraffin was melted from slides (15 minutes at 60°C) and removed by immersion in Citrisolv (Fisher Scientific) twice for 3 minutes each time. Slides were then washed twice in 100% ethanol for 3 minutes and rehydrated by sequentially immersing the slides through graded ethanol washes for 3 minutes each. Endogenous peroxidase activity was blocked by immersing the slides in 0.3% hydrogen peroxide for 15 minutes. Antigen retrieval was performed by heating the slides in citrate buffer (Citra, Biogenex, San Ramon, CA), maintaining the slides at 100°C for 10 minutes in a microwave oven. Immunohistochemistry staining was performed with an automated stainer (Autostainer Plus, DakoCytomation, Carpinteria, CA). Two blocking reactions were performed: one using Power Block for 10 minutes (Biogenex, San Ramon, CA), another using Protein Block (goat sera) for 2 hours (Biogenex).
Antibodies from 3 sources were evaluated for use as primary antibody: turkey convalescent sera, turkey hyperimmune sera, and chicken hyperimmune sera. A single antibody was not developed for this purpose because the cross-reactivity among these isolates, which are genetically variant in the gene that encodes the major virus antigen, has not been determined. TRV isolate-specific primary antibodies used were: (1) turkey convalescent sera collected at 21 DPI; (2) turkey hyperimmune sera produced by infection of turkeys at 2 days of age and boosting at 21 days by the intraperitoneal route and collection at 35 DPI, and (3) chicken hyperimmune sera produced the same way but in chickens. Antibodies were diluted 1:100 in streptavidin–peroxidase antibody diluent (Biogenex) before use. Slides were incubated with the primary antibody for 1 hour.
The secondary antibody was Goat anti-Turkey IgG HRP labeled or Goat anti-Chicken IgG HRP labeled (Southern Biotechnology Associates, Inc, Birmingham AL) and was diluted 1:100 in antibody diluent (Biogenex). The reaction was visualized with the 3,3′ diaminobenzidinc (DAB) substrate kit for peroxidase (Zymed Laboratories Inc, San Francisco, CA). After IHC staining, sections were counterstained with hematoxylin, air dried, coverslipped, and examined by light microscopy.
Positive controls consisted of tissue sections from reovirus-infected poults in which the infection had been confirmed by RT-PCR amplification.44 Negative controls consisted of tissues collected from sham-inoculated poults, for which infection status was confirmed to be negative by RT-PCR. The intensity of staining in each section was scored as follows: −, no antigen staining; +/−, rare; +, infrequent; ++, common; +++, widespread staining.
In situ hybridization
A 402-base pair (bp) probe, which targeted the Sigma-C gene of the NC/SEP-R44/03 TRV isolate, was produced by RT-PCR. The probe corresponds to nucleotides 500 to 902 of the NC/SEP-R44/03 S1 genome segment, which spans the putative overlapping p17/sigma C gene junction (GenBank accession number 800797). The probe was labeled with digoxigenin by nick translation (DIG-Nick translation mix, Roche, Indianapolis, IN). Two micrograms of the RT-PCR product in a total reaction volume of 20 μl was incubated at 14°C for 2 hours. Labeling completion was verified by testing an aliquot of the reaction mix on 2.5% agarose gel electrophoresis staining with ethidium bromide. Smearing and disappearance of the 402 bp band were used to judge the adequacy of labeling.
A commercial detection kit for ISH (InnoGenex Universal ISH Kit, InnoGenex, San Ramon, CA) was used according to the manufacturer's instructions. Briefly, tissue sections were cut (4-μm thick) from the same paraffin-embedded tissue samples as used for histopathology and IHC and mounted on charged glass slides (Superfrost/Plus, Fisher Scientific). Slides were deparaffinized and rehydrated as previously described, and then treated with proteinase K for 10 minutes. Postfixation was carried out in 1% formaldehyde for 10 minutes after washing in phosphate-buffered saline (PBS). Hybridization was performed at high stringency with 2 ng/μl of denatured probe at 37°C for 16 hours in 50 μl of hybridization mix under a plastic coverslip in a humidified chamber. Excess probe was removed by washing in 2× PBS for 10 minutes at room temperature and was followed by 2 washes in 1× PBS for 3 minutes each. After incubation with Power Block reagent for 5 minutes, the sections were incubated with antidigoxigenin antibody for 2 hours. After 2 separate washes with 1× PBS, the sections were treated with streptavidin–alkaline–phosphatase conjugate for 20 minutes at room temperature. After 2 separate washes with 1× PBS, the signal was developed using 5-bromo-4-chloro-3-indolyl phosphate (BCIP) and nitroblue tetrazolium salt (NBT), which produces an insoluble blue precipitate indicating hybridized molecules. Sections were counterstained with nuclear fast red. Positive and negative controls were from the same sources as those used for the IHC procedure. Staining was observed by examination under a light microscope.
TUNEL assay
Apoptotic cells were detected with a commercial in situ cell death detection kit (DeadEnd Colorimetric TUNEL System, Promega Corp, Madison, WI) according to the manufacturer's instructions. Briefly, tissue sections were cut (4-μm thick) from the same paraffin-embedded tissue samples used for histopathology and IHC and mounted on charged glass slides (Superfrost/Plus, Fisher Scientific). Slides were deparafinized and rehydrated as previously described. Slides were washed in 0.85% NaCl and PBS for 5 minutes each and fixed by immersion in 10% neutral buffered formalin for 15 minutes. After 2 washes in PBS for 5 minutes each, the slides were treated with 20 μg/ml proteinase K for 10 minutes. Tissue sections were refixed by immersing the slides in 10% neutral buffered formalin for 5 minutes each. Slides were washed again twice in PBS, and excess liquid was removed. The tissues were covered with 100 μl of equilibration buffer for 10 minutes. The equilibration buffer was then blotted off, and the tissues were covered with 100 μl of terminal deoxynucleotidyl transferase (TdT) reaction mix, containing a biotinylated nucleotide mix and TdT enzyme. Tissue sections were covered with plastic coverslips to ensure even reaction mix distribution and incubated at 37 C for 60 min inside a humidified chamber to allow the end-labeling reaction to occur. The reaction was terminated by immersing the slides in 2× standard sodium citrate for 15 minutes. Slides were washed twice in PBS for 5 minutes to remove unincorporated biotinylated nucleotides. Endogenous peroxidases were blocked by immersing the slides in 0.3% hydrogen peroxide for 15 minutes. Slides were washed again in PBS, and the streptavidin–horseradish–peroxidase solution diluted at 1:500 in PBS was added to each slide; the slides were incubated for 30 minutes at room temperature. After the slides were washed twice in PBS, 100 μl of a solution containing the DAB substrate, the DAB chromogen, and hydrogen peroxide was applied to each slide and allowed to develop until there was a light-brown background (approximately 10 minutes). After the slides were rinsed several times in deionized water, they were mounted and coverslipped. Positive and negative controls were from the same source as those used for the IHC. Staining was observed under a light microscope.
Results
Histopathology
The distribution and severity of histologic lesions and reovirus antigen staining by tissue and virus isolate are summarized in Table 1. The thymus, heart, pancreas, and proventriculus, which presented no lesions, very mild lesions, or were IHC-negative, were excluded from the table.
Distribution and severity of histologic lesions and viral antigen in poults inoculated with turkey-origin reoviruses: NC/SEP-R44/03 (SEP/44), NC/98, TX/98, NC/85 or a chicken-origin reovirus (1733).∗
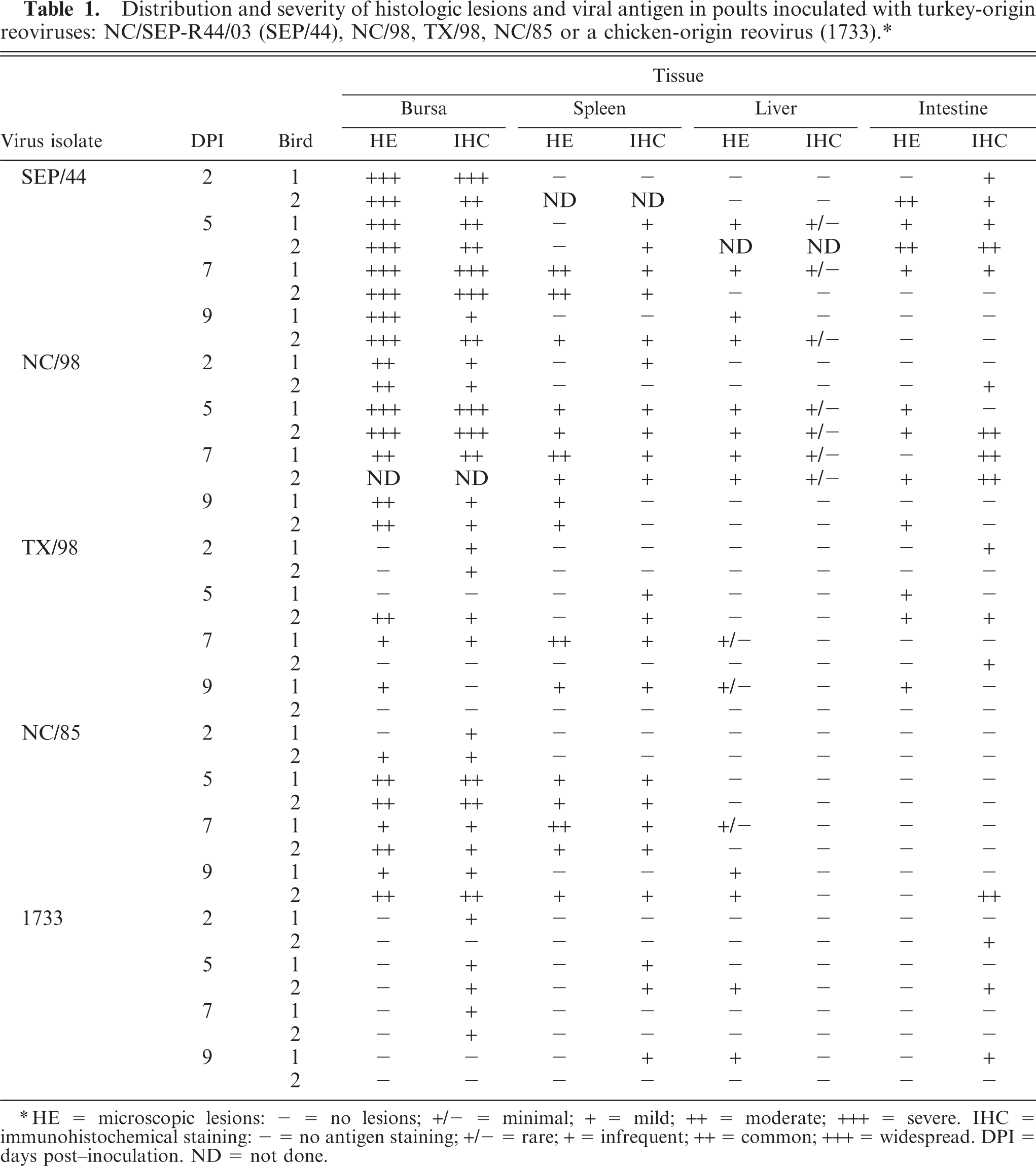
HE = microscopic lesions: − = no lesions; +/− = minimal; + = mild; ++ = moderate; +++ = severe. IHC = immunohistochemical staining: − = no antigen staining; +/− = rare; + = infrequent; ++ = common; +++ = widespread. DPI = days post–inoculation. ND = not done.
The major histologic lesions associated with TRV infection in poults was bursal atrophy characterized by lymphoid depletion and fibroplasia. Mild to moderate lymphoid depletion and histiocytic and ellipsoidal hyperplasia were present in the spleen. Mild crypt hyperplasia and heterophil and lymphocytic infiltration in the submucosa were observed in the jejunum and duodenum. Mild to moderate lymphocytic infiltration was present in the liver, pancreas, heart, and proventriculus, but there were no lesions in the thymus. Lymphocytes were found around the portal veins in the liver, between myocardial fibers in the heart, and between the acinar epithelium of the pancreas. In the proventriculus, lymphocytic infiltrates were present in the proventricular glands and in the lamina propria of the submucosa. No lesions were observed in tissues collected from sham inoculated poults at any time. Poults inoculated with the chicken-origin reovirus isolate 1733 had lymphocytic infiltrates in the liver at 5 and 9 DPI, but no other microscopic lesions.
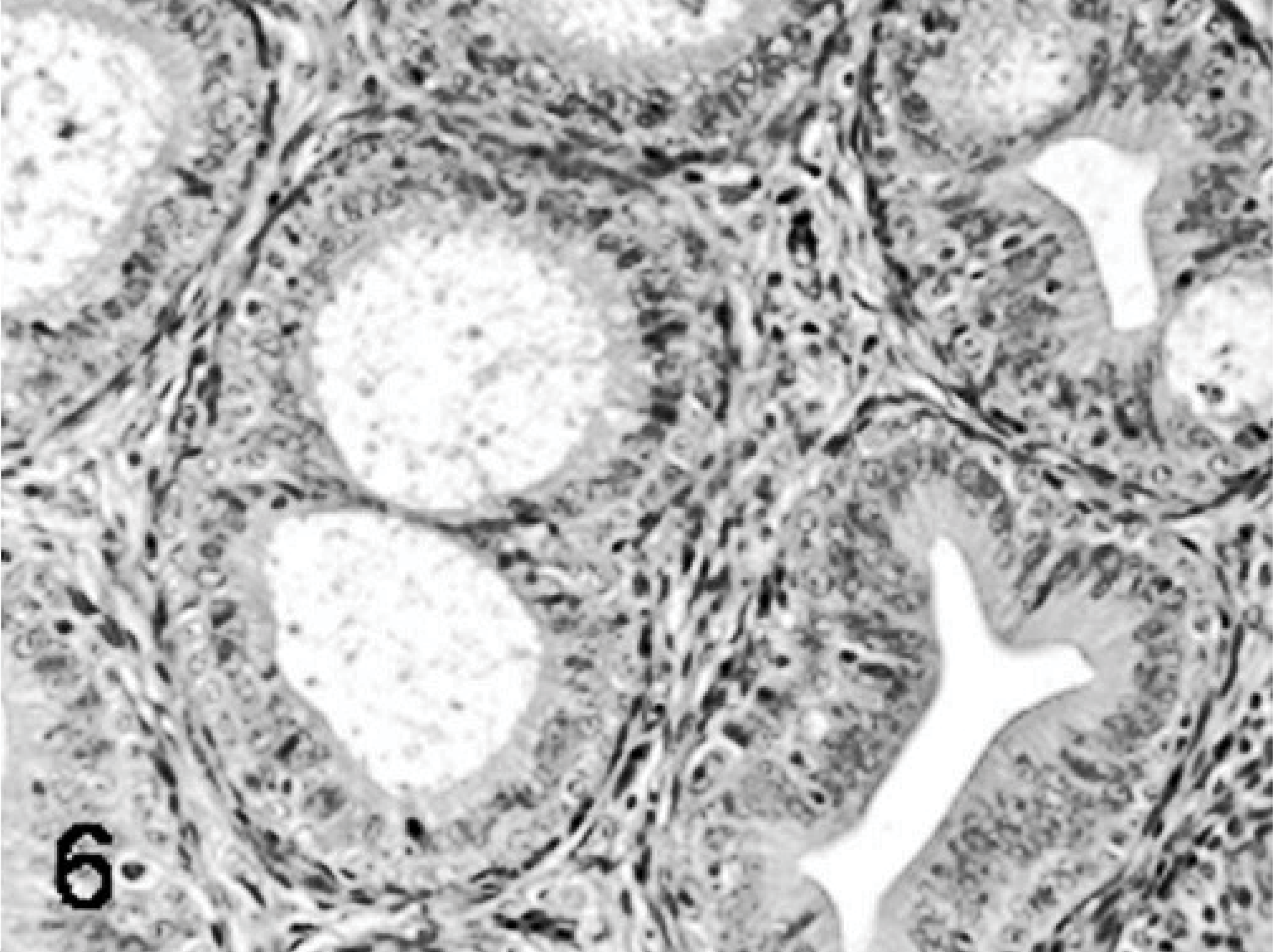
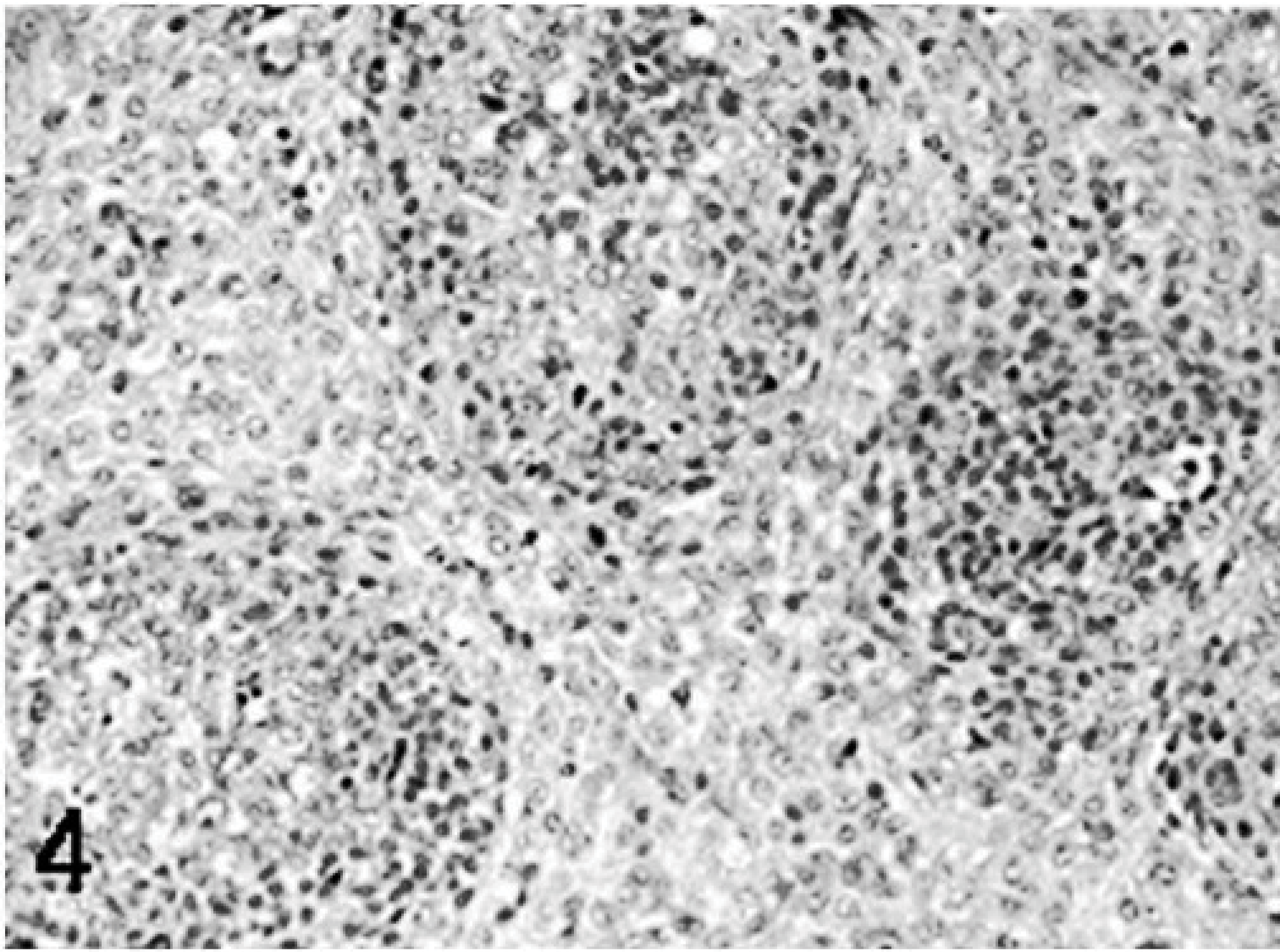
Poults inoculated with reovirus isolate NC/SEP-R44/03 had severe bursal atrophy at 2, 5, 7, and 9 DPI. At 2 DPI active lymphocellular depletion indicated by pyknotic and karryorhectic nuclei was observed. Macrophage proliferation and mild heterophil infiltration was also present. At 5, 7, and 9 DPI there was severe lymphoid depletion in bursal follicles. Both cortical and medullary regions of the bursal follicles were affected (Figs. 1– 6). Fibroplasia was present surrounding the bursal follicles. Follicles containing large macrophages with foamy cytoplasma and multiple large intrafollicular cysts lined with a pseudostratified epithelium were observed at 7 and 9 DPI (Figs. 3 –6). Mild lymphoid depletion in the spleen periellipsoidal and periarteriolar sheaths, coupled with histiocytic and ellipsoidal hyperplasia, was also observed at 7 and 9 DPI. Mild crypt hyperplasia with heterophil and lymphocytic infiltration was observed in the submucosa of the duodenum and jejunum at 2, 5, and 7 DPI. Two poults also presented loss of the intestinal villi tips in the jejunum and individual enterocyte cell death in both jejunum and duodenum. Lymphocytic infiltrates were observed in the liver of several poults at 5, 7, and 9 DPI and in the heart and proventriculus of 1 poult at 9 DPI. One of the poults at 7 DPI had pancreatic degeneration characterized by necrosis of the pancreatic acinar epithelium and increased numbers of macrophages and fibrosis.
Bursa; higher magnification; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 9 DPI. Marked epithelialization of the bursal follicles. The reticular epithelium undergoes metaplasia giving rise to acinar structures lined by a psuedostratified epithelium. HE.
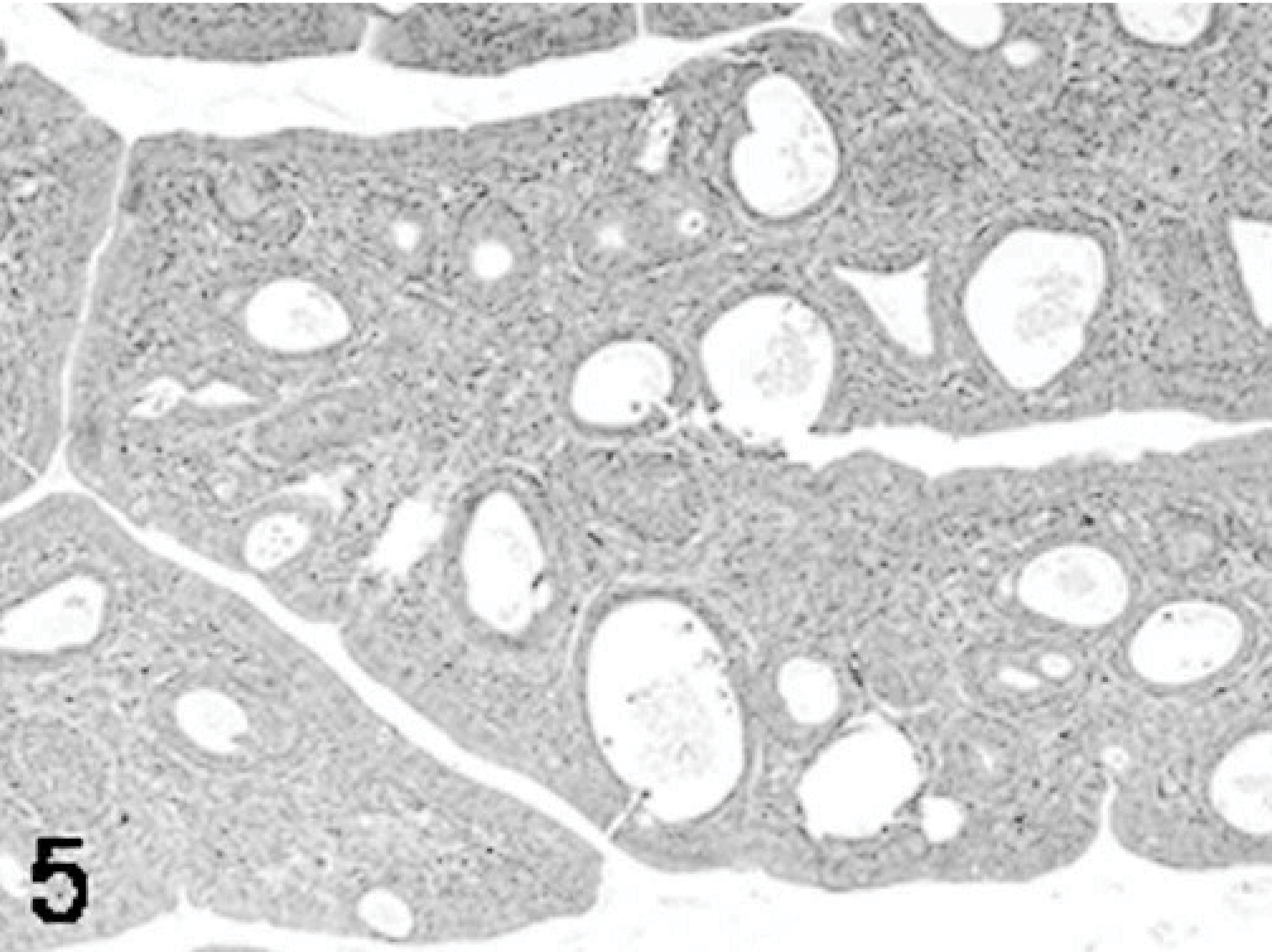
Bursa; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 9 DPI. Bursal atrophy and stromal fibroplasia. Intrafollicular cysts lined with pseudostratified epithelium. HE.
Bursa; higher magnification; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 5 DPI. Note both cortical and medullary follicle lymphoid depletion and stromal fibroplasia. HE.
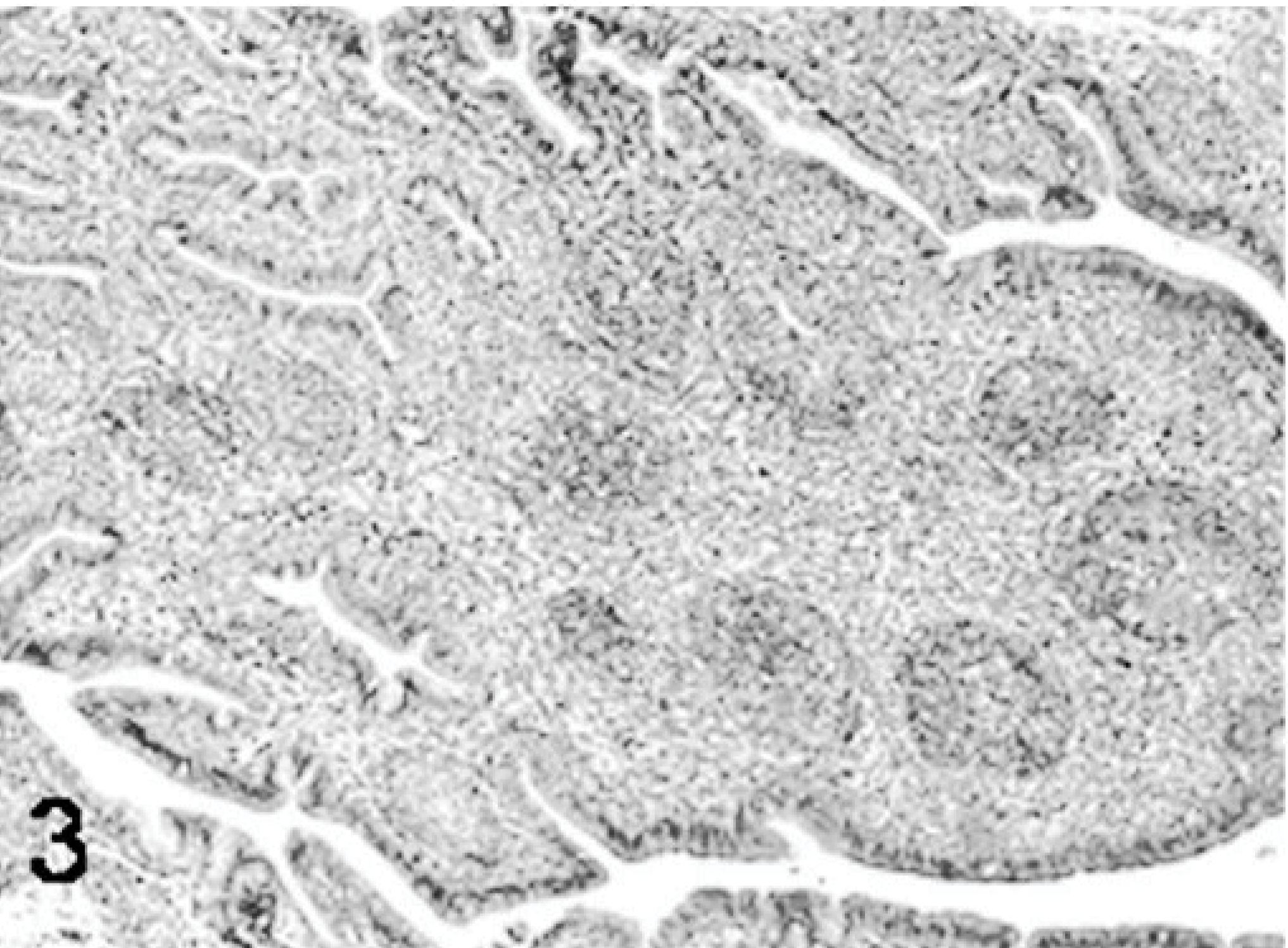
Bursa; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 5 DPI. Bursal atrophy with follicle lymphoid depletion and stromal fibroplasia. HE.
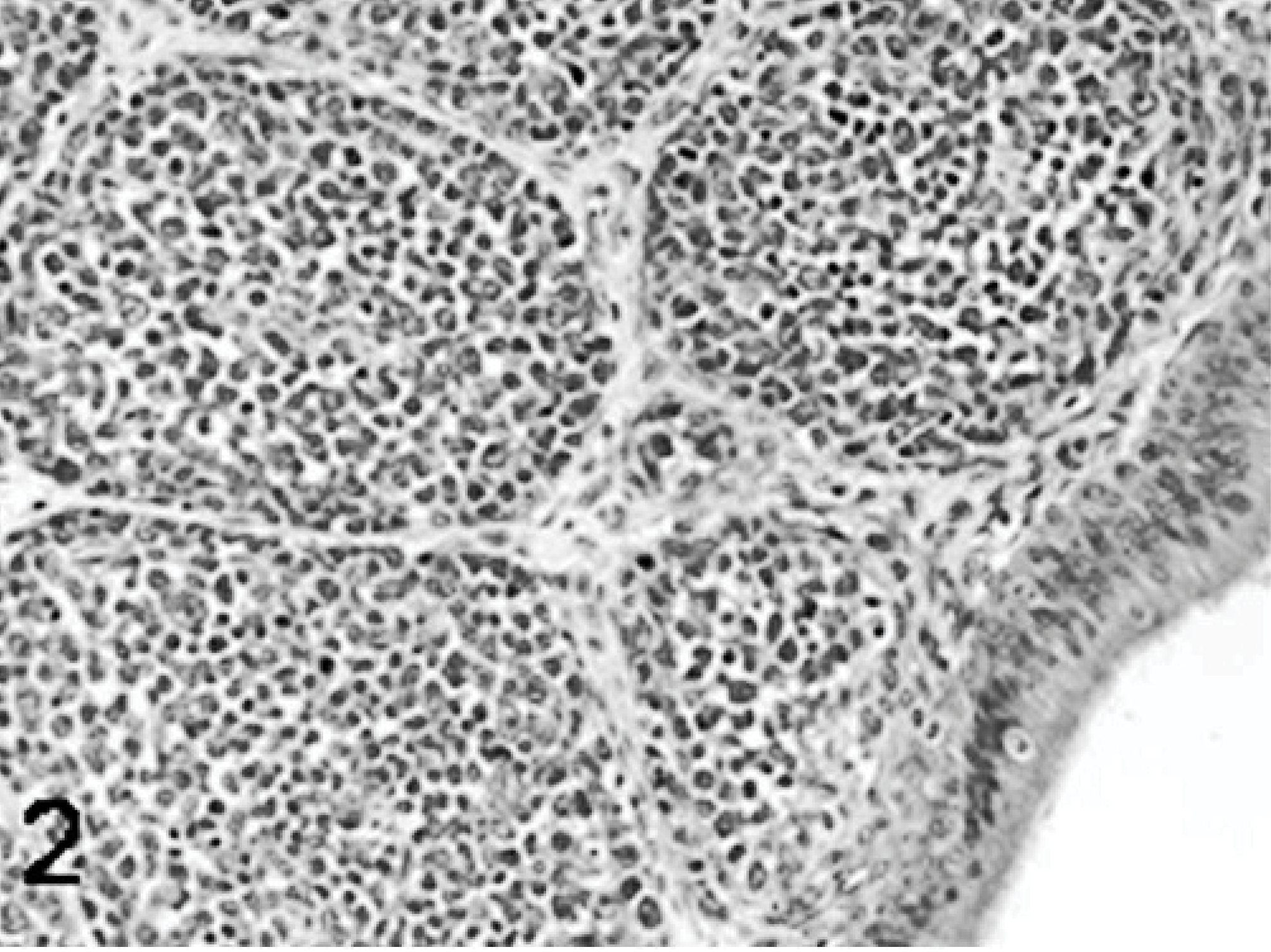
Bursa; higher magnification; poult sham inoculated; 5 DPI. HE.
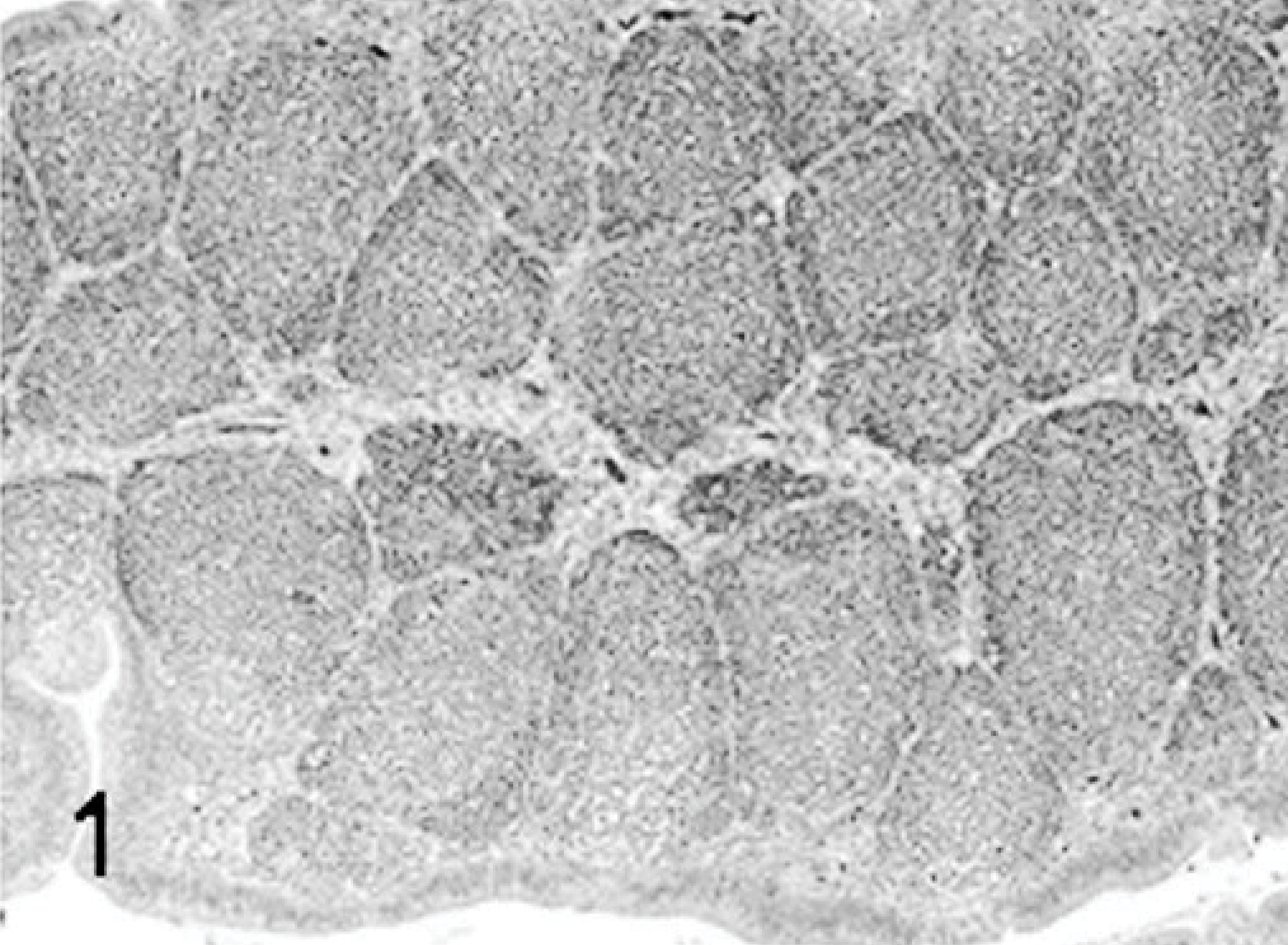
Bursa; poult sham inoculated; 5 DPI; HE.
Poults inoculated with the NC/98 TRV isolate had moderate to severe bursal lymphoid depletion and fibroplasia at 2, 5, 7, and 9 DPI. Lymphoid depletion was also present in the spleen at 5, 7, and 9 DPI. Lymphocytic infiltrates were observed in the liver at 5 and 7 DPI and in the pancreas at 9 DPI. Mild crypt hyperplasia and death of the epithelial cells on the tips of the villi were observed in the duodenum and jejunum at 5, 7, and 9 DPI. Proventriculitis was observed in one of the poults at 2 DPI.
Poults inoculated with the TX/98 and NC/85 TRV isolates had mild to moderate bursal lymphoid depletion at 5, 7, and 9 DPI. Mild lymphoid depletion in the spleen and lymphocytic infiltrates in the liver were observed at 7 and 9 DPI. Mild lesions or no lesions were present in the intestine. Two poults inoculated with NC/85 had lymphocytic infiltrates in the pancreas at 9 DPI. Lymphocytic infiltrates were present in the proventriculus at 5, 7, and 9 DPI.
Viral antigen and nucleic acid detection in tissue
Tissues from TRV-infected poults, but not those from the control poults, contained detectable TRV antigen and RNA. Positive staining by IHC was characterized by the presence of intracytoplasmic dark-brownish granules. No specific peroxidase-positive staining was seen in the tissues of the control poults.
Of the 3 primary antibodies used, the turkey convalescent sera gave the best results. The use of turkey hyperimmune sera increased the background staining, whereas the staining obtained with the chicken convalescent sera was weak. In spite of the difference in the staining quality, the 3 antibodies gave the same staining pattern. The nonspecific background, characterized by a diffuse light-brown staining, was reduced by the use of protein blocks.
In the bursa, peroxidase-positive staining was seen in the cytoplasm of the surface epithelial cells, stromal cells, and in macrophage at days 2, 5, 7, and 9 DPI in all TRV-inoculated poults. Viral antigen was first detected in the bursa at 2 DPI primarily in the surface epithelium and adjacent stomal cells (Fig. 7). This staining was more frequently seen in the follicular-associated epithelium (Fig. 7). At 5, 7, and 9 DPI, macrophages present in the interfollicular area also stained for viral antigen (Fig. 8).
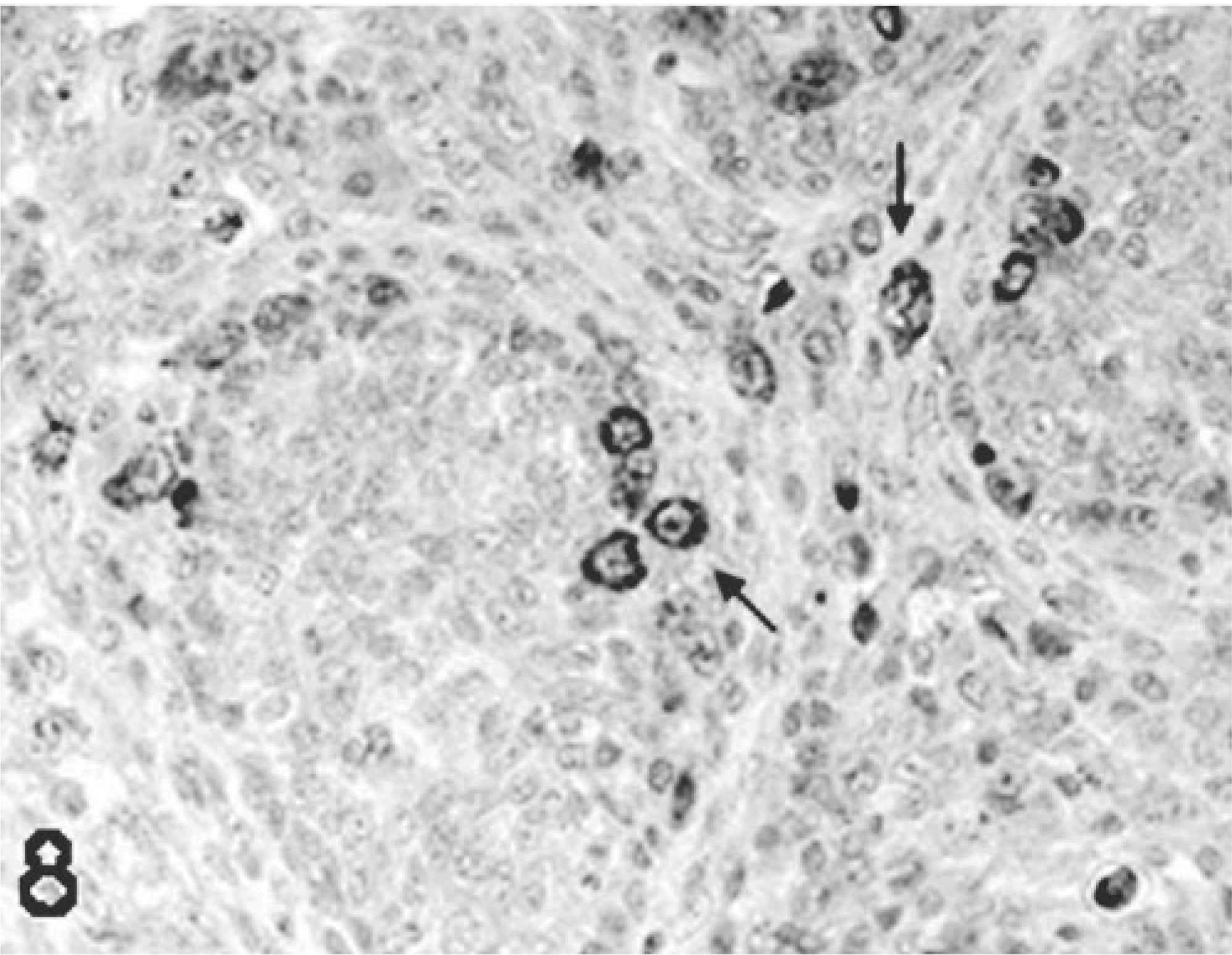
Bursa; poult inoculated at 2 days of age with TRV isolate NC/98; 7 DPI. Viral staining in macrophages present in the interfollicular area, and in the follicles (arrows). Immunoperoxidase labeling, hematoxylin counterstain.
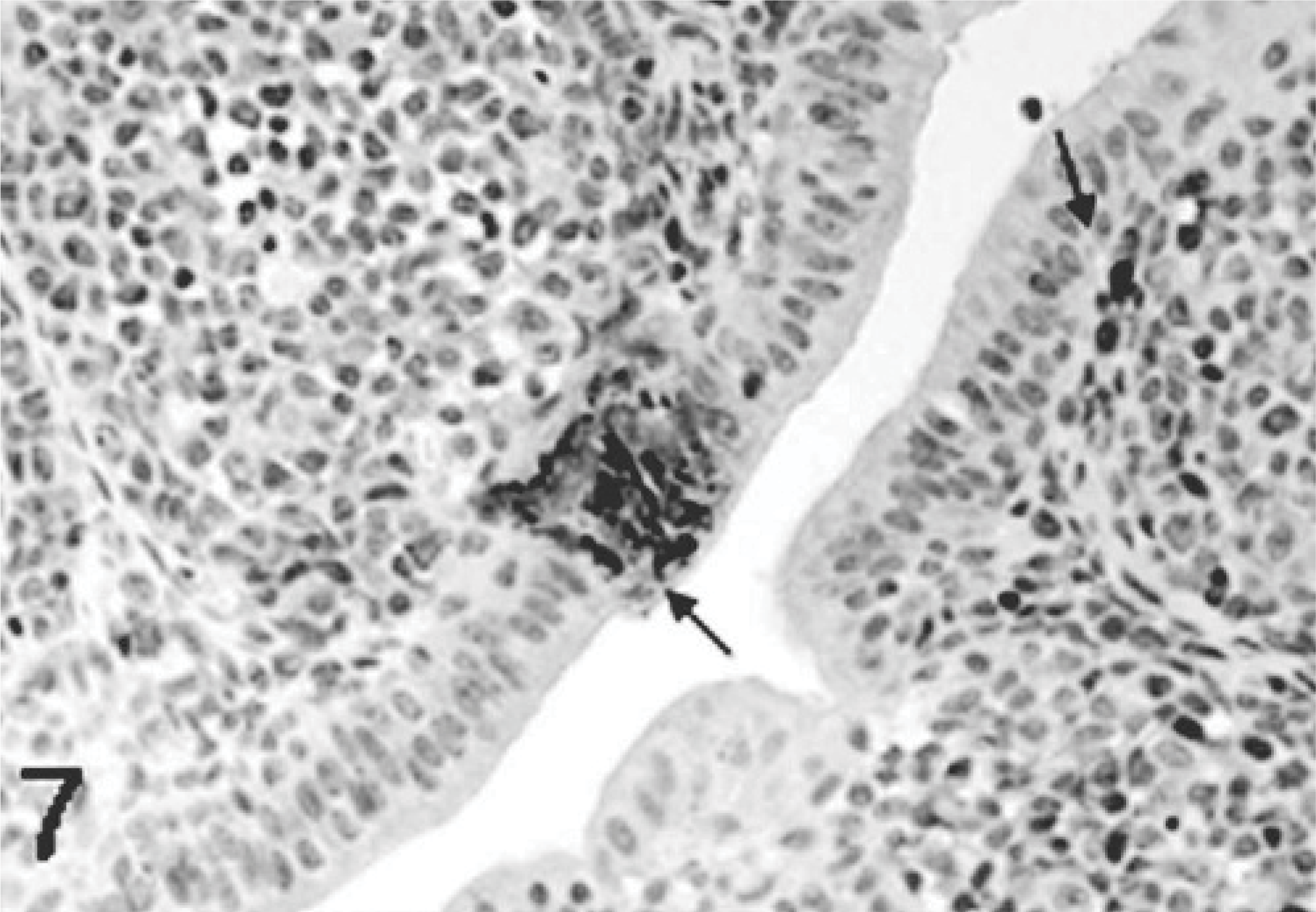
Bursa; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 2 DPI. Viral antigen present in the bursal surface epithelium, follicular associated epithelium, and adjacent stromal cells (arrows). Immunoperoxidase labeling, hematoxylin counterstain.
Positive staining was also observed in the cytoplasm of the enterocytes at the tip and middle section of the affected villi (Fig. 9) and in large mononuclear cells (most likely macrophages) in the splenic red pulp. In the liver there was no staining or very faint staining, mostly in Kupffer cells. The lesions observed in the liver, although mild, did not correlate with the antigen staining; this was most likely because the viral load was very low and was mostly associated with uptake of the virus-infected cell debri by Kupffer cells. The same was observed in the pancreas where no staining was observed; the exception was the poult that had pancreatic degeneration, where viral antigen was present in the intra-lesional macrophages.
Jejunum; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 5 DPI. Viral staining is present in the cytoplasm of the enterocytes at the tip and middle section of the affected villi (arrows). Immunoperoxidase labeling, hematoxylin counterstain
The viral staining present in the bursa of poults inoculated with reovirus isolates NC/SEP-R44/03 (SEP/44) and NC/98 was strong and widespread in contrast to the staining present in the bursas from poults inoculated with isolates TX/98, NC/85, and 1733, where it was less common.
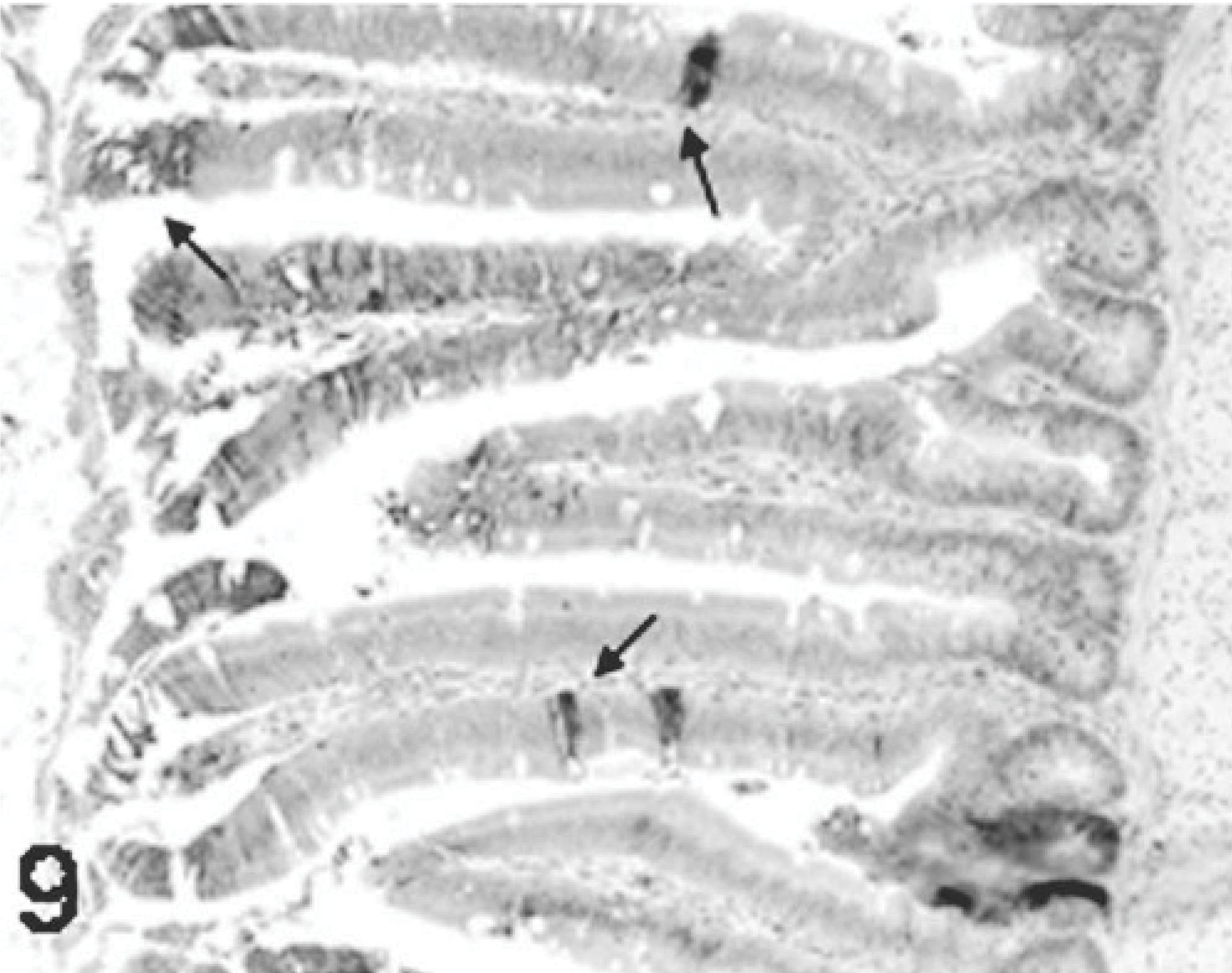
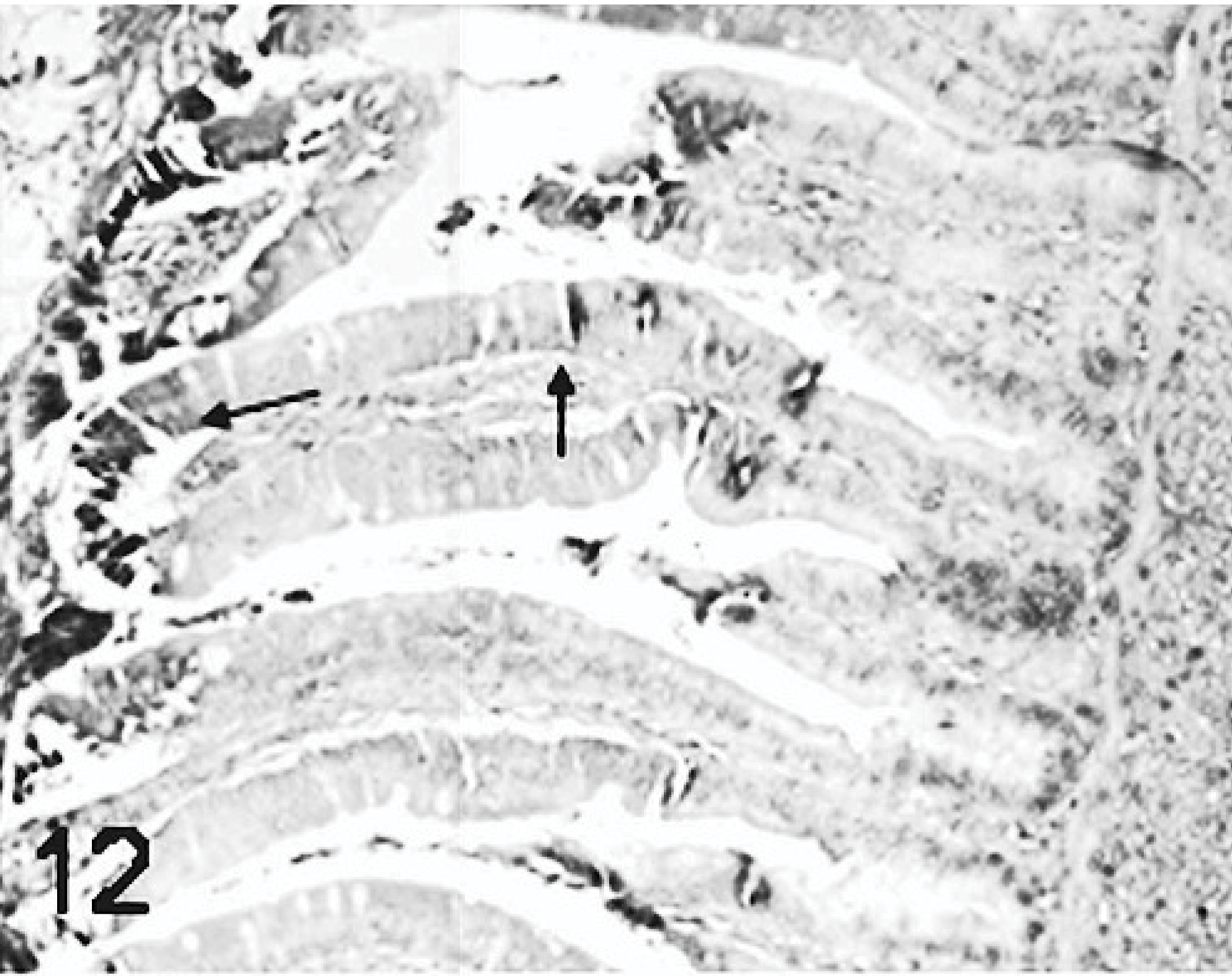
Results of ISH were similar to IHC. In all TRV-inoculated poults, positive cytoplasmic staining was found in the bursal epithelial cells lining the lumen of the organ (Fig. 10) and within macrophages in connective tissue surrounding the follicles (Fig. 11). In the small intestine, positive cytoplasmic staining of TRV RNA was observed in the surface enterocytes but not in the other cells (Fig. 12). Mild staining for viral RNA was also present in splenic macrophages and to a lesser degree in the liver Kupffer cells. No ISH staining was found in the rest of the tissues examined. No virus-specific staining was detected in the controls.
Jejunum; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 5 DPI. ISH. Viral RNA staining is present in the cytoplasm of the enterocytes at the tip and middle section of the affected villi (arrows). Digoxigenin labeling, BCIP-NBT staining fast red counterstain.
Bursa; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 5 DPI. ISH. Viral RNA staining in macrophages present in the interfollicular area (arrows). Digoxigenin labeling, BCIP-NBT staining, fast red counterstain.
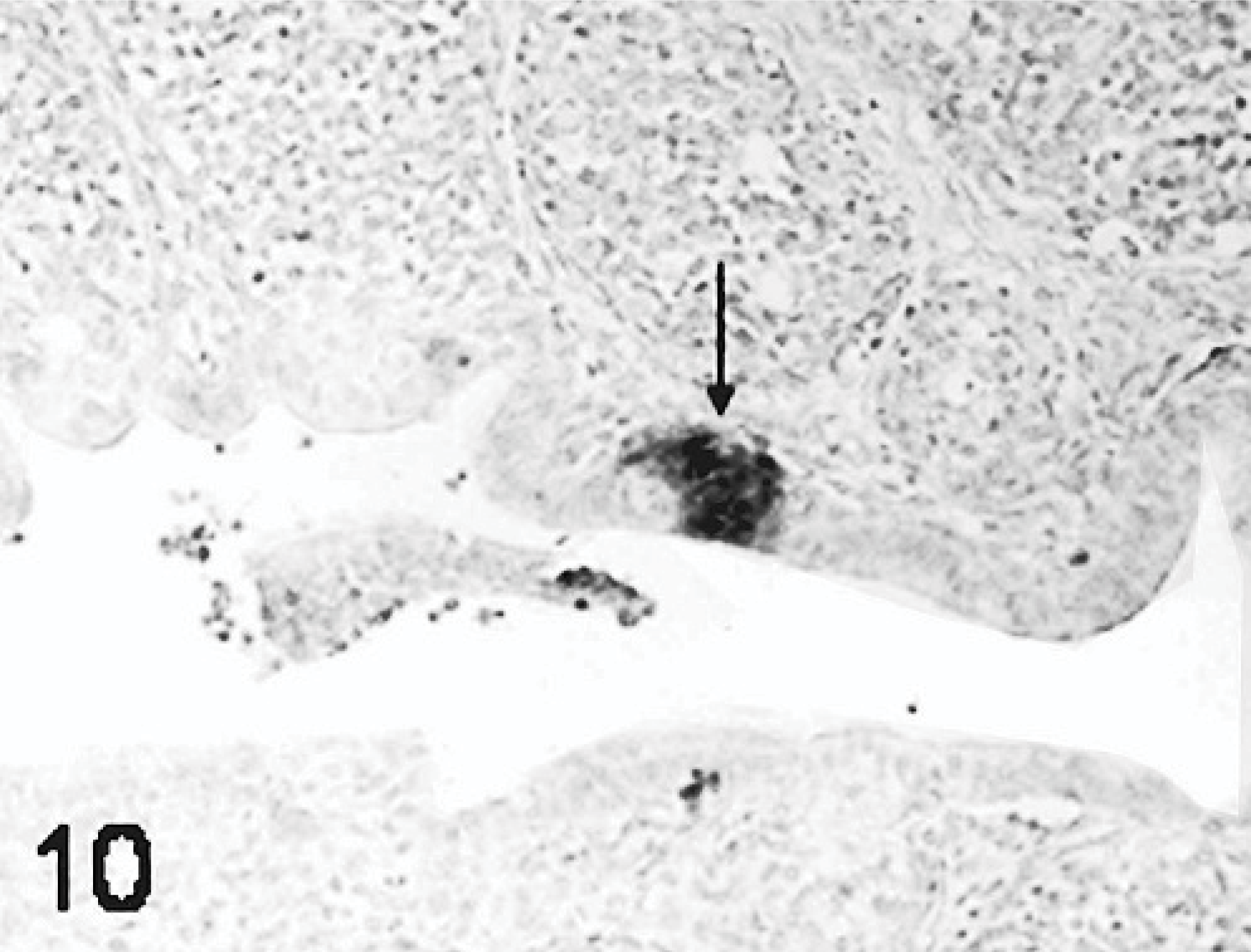
Bursa; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 2 DPI. ISH for viral RNA. Viral RNA present in the bursal surface epithelium and adjacent stromal cells (arrows). Digoxigenin labeling, BCIP-NBT staining, fast red counterstain.
Apoptosis detection
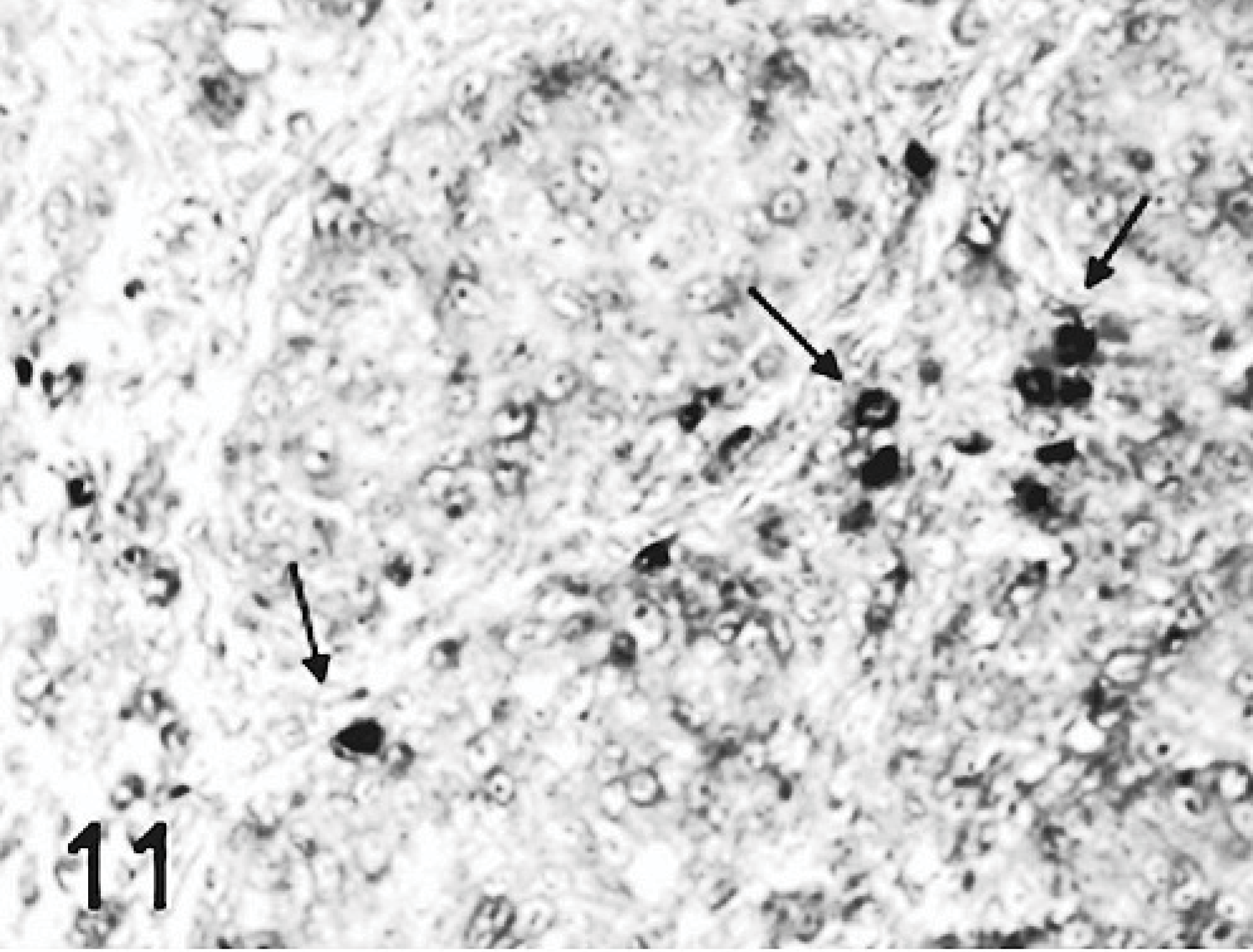
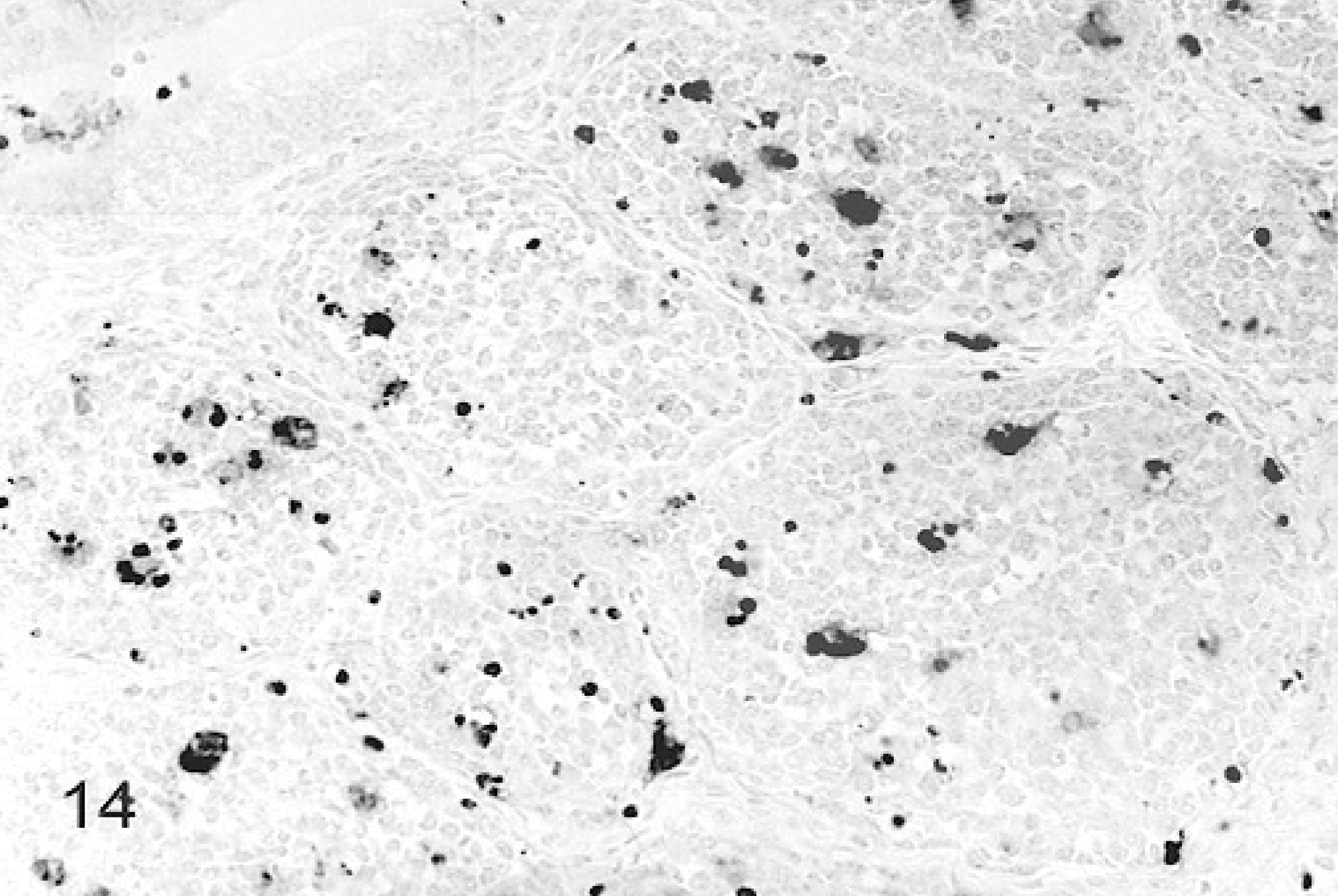
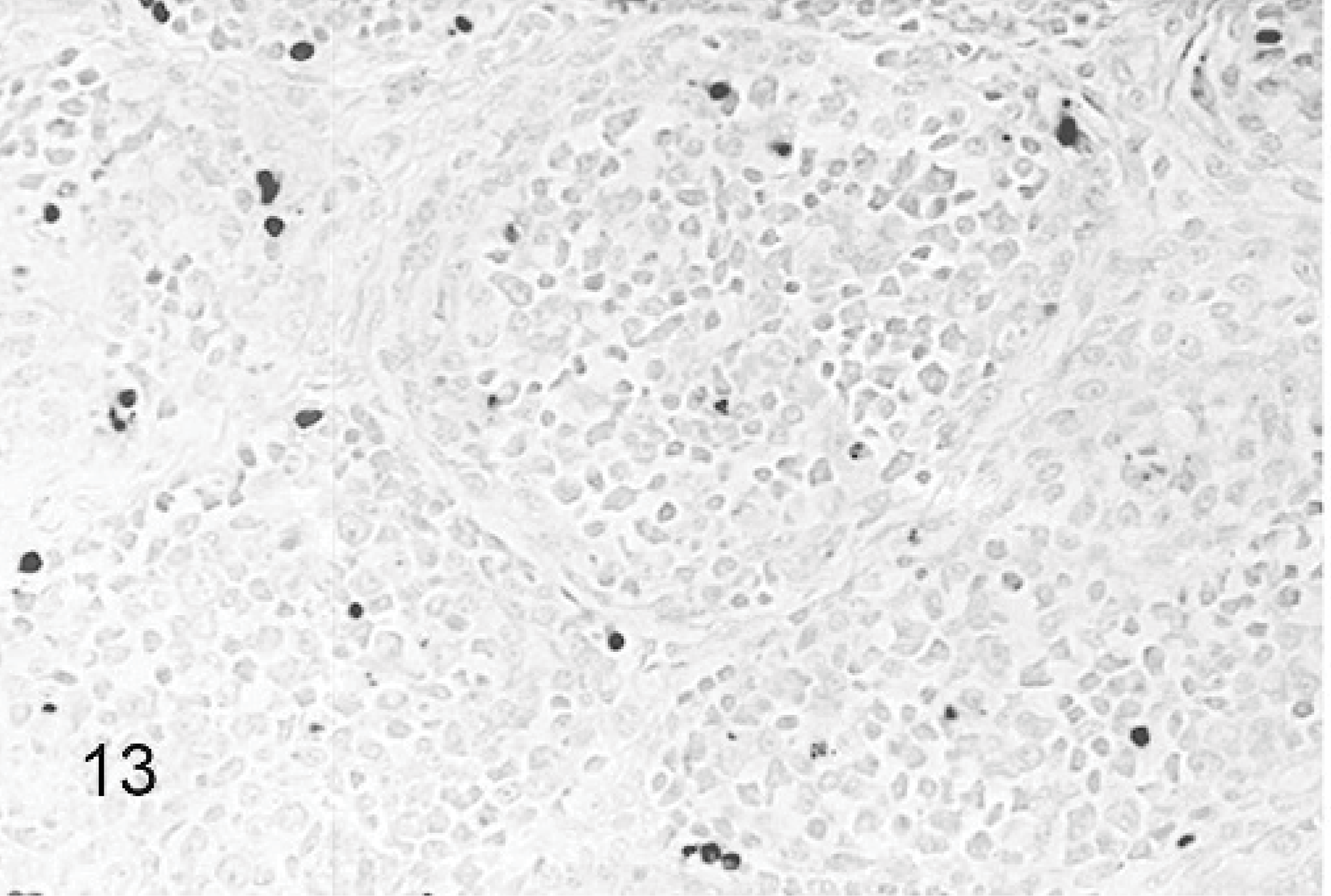
An increase in apoptosis staining was observed in bursas from all TRV-inoculated poults between 2 and 5 DPI as compared to the sham-inoculated controls (Figs. 13, 14). The minimal apoptotic staining in the bursa of control birds represents normal cellular turnover during maturation of B lymphocytes.22 Apoptosis staining was strongest in bursae from poults inoculated with TRV strains NC/SEP-R44/03 or NC/98. Birds from these groups also had increased apoptosis staining in the spleen. No apoptosis staining was observed in the other tissues examined.
Bursa; poult inoculated at 2 days of age with TRV isolate NC/SEP-R44/03 (SEP/44); 2 DPI. TUNEL staining.
Bursa; sham-inoculated poult; 2 DPI. TUNEL staining.
Viral antigen RNA staining and staining for apoptosis did not always correlate. The cells undergoing apoptosis in both the bursa and spleen were mostly macrophages, but lymphocytes, which did not stain for viral antigen or RNA, also stained for apoptosis.
Discussion
This study describes the distribution of TRVs in the tissues of experimentally infected SPF turkey poults and the microscopic lesions associated with TRV infection. Immunohistochemistry and ISH were used for detecting the virus and, in terms of staining intensity and tissue distribution of viral signals detected, revealed similar results. Among all of the tissues examined, only the bursa, intestines and spleens were consistently positive for the presence of virus. The specific cell types infected were limited to bursa surface epithelial and stroma cells, macrophages, and enterocytes. The localization of peroxidase-positive, brownish granules in the cytoplasm of the affected cells by IHC and bluish granules by ISH is consistent with the intracellular distribution of reovirus.28,46 The early presence of staining in the bursa supports previous speculation that the bursa may serve as a primary site of replication for reovirus during an infection.46–48
The pattern of virus spread and replication observed among the reovirus isolates in turkeys was similar, but different from that reported for chicken origin reovirus.13,16 For both chickens and turkeys, intestine and bursa are primary entry and replication sites, and the spleen is among the first tissues infected after initial replication in both species. This may be due to viremia. However, the liver does not appear to be a site of replication for turkey reovirus as it is for chicken reovirus. No evidence of multifocal hepatic necrosis similar to that described in chickens21,48 was observed in the turkeys, nor was there clear evidence of viral replication in the liver, although this organ has been considered a primary target of avian reoviruses.3,16,21,27,28,47,53 In chickens, reovirus does not replicate in the thymus nor does it cause detectable alteration in peripheral T cell subpopulations.40 Similarly, no lesions or viral replication were present in the thymus of TRV-infected poults. The effect of TRV infection on the gastrocnemius tendon was not evaluated in this study because lesions in the tendons are usually not present until the second week after inoculation.36,37 Studies of the persistence of TRV and its effect on tendons and other organs need to be performed.
Avian reovirus grows in macrophage cultures, indicating that macrophages may be a target cell for this virus.45,52 Virus staining was present in macrophages in the bursa and spleen, indicating that macrophages are also a target cell in turkeys. However, in the early stages of the infection the viruses were primarily found in epithelial cells of the intestine and the bursa. Similar staining patterns have been observed in reovirus-infected psittacines.38
In a study with poults inoculated with a PEMS-associated reovirus, increased crypt depth was observed in intestinal sections.9 Intestinal lesions have also been reported for chicken-origin avian reoviruses37 but their effect on body weight gain is not clear.17,43,49 Reoviruses isolated from cases of malabsorption syndrome in chickens induced lesions such as vacuolar degeneration and sloughing of enterocytes in the small intestine of chickens.43 In the present study, mild lesions and mild viral antigen and RNA staining, with a few exceptions, were present in the intestine, indicating that although the viruses can infect and replicate in the enterocytes, it does not cause the tissue damage associated with the enteritis characteristic of poult enteritis complex; however, a concomitant infection with another agent could possibly cause it. The poults in this experiment presented mild clinical disease although some TRV isolates did cause significant body weight decrease as indicated by Spackman et al.44
An effect reported for reovirus infection is the induction of immunosuppression, predisposing the host to infection with other pathogens, which would account for the diversity of syndromes associated with reovirus.23,25,33,40 The bursal lymphoid depletion caused by the TRV isolates could induce a transient and possibly permanent immunosuppression in young infected turkeys. A difference in pathogenicity was observed between the TRVs studied. Two of the isolates, NC/SEP-R44/03 and NC/98, induced severe lesions in the bursa, whereas the other 2, TX/98 and NC/85, induced mild to moderate lesions. Therefore, the effect on the immune response of the poults to other pathogens may be affected to a different degree depending on the reovirus strain involved. Further molecular and antigenic characterization needs to be completed for these TRVs.
Reovirus infection induces apoptosis in cells in vitro and in vivo.6 In mammals, there is an excellent correlation between the location of virus infection, the presence of tissue injury, and apoptosis, indicating that apoptosis is a critical mechanism by which disease is triggered in the host.6 Mammalian6 and avian18,41 reovirus-induced apoptosis has been well documented, and it has been suggested that bystander apoptosis could result from the release of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) or other death ligands from reovirus-infected cells.6 In this study, apoptosis was present not only in virus antigen-positive cells of the bursa and spleen, but also in cells that were negative for viral staining, like lymphocytes. This would explain the lymphocyte depletion observed in both organs, although lymphocytes are not a target for reovirus replication. This virus-independent or “bystander” lymphoid depletion was also mentioned in a previous report of reovirus infection in psittacines 38 and was identified in several viral infections of other avian species.12,14,32 The lack of inflammation in the bursa observed with TRV infection most likely is due to these viruses killing lymphocytes by apoptosis instead of necrosis, as reported with variant strains of infectious bursal disease virus (IBDV).51 The severe bursal atrophy observed with the NC/SEP-R44/03 and NC/98 isolates of TRV is similar to that observed with IBDV infection in chickens. The death of bursal B-cells before 2 weeks of age, as observed with turkeys infected with NC/SEP-R44/03 and NC/98, can result in permanent humoral immunosuppression.34 This immunosuppressive effect of TRVs is being evaluated.
Footnotes
Acknowledgements
We acknowledge Diane Smith and Scott Lee for technical assistance with this paper. This work was supported by US Department of Agriculture Current Research Information System (CRIS) project #6612-32000-036. Mention of trade names or commercial products in this manuscript is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
