Abstract
Concurrent infection with peste des petits ruminants virus (PPRV) and pestivirus was diagnosed in stillborn twin lambs. With the flock history, the findings of epidermal syncytial cells and necrotizing bronchitis/bronchiolitis prompted testing for PPRV infection, and PPRV antigen was detected by immunohistochemistry (IHC) in the skin, lungs, kidneys, rumen, and thymus. Macroscopic anomalies that were typical of border disease included scoliosis, brachygnathism, prognathism, arthrogryposis, hydranencephaly, cerebellar hypoplasia, and hairy fleece; pestiviral antigen was detected by IHC in the brain, liver, lungs, and kidneys. Tissues from both lambs were positive by reverse transcriptase-polymerase chain reaction (RT-PCR) for PPRV and pestivirus. To the authors' knowledge, PPR has not been reported previously as a congenital infection or in combination with pestiviral infection.
Keywords
Peste des petits ruminants (PPR) is a contagious viral disease of small ruminants that is characterized by pseudomembranous oral lesions, bronchointerstitial pneumonia, and enteritis. 4, 14, 16, 29 The mortality rate is higher in goats than in sheep in Turkey because the PPR virus (PPRV) strain Tu00 is highly virulent for goats. 7, 10, 22 The Tu00 strain belongs phylogenetically to Lineage 4. 7, 10, 22, 29 Infection with PPRV is generally thought to be transmitted via oral and/or respiratory routes, and the virus is present in lacrimal and oral secretions in high concentration. 10, 16, 17 Vertical transmission of PPRV has not been reported.
Border disease (BD) is characterized by various congenital anomalies including hydranencephaly, cerebellar hypoplasia, arthrogryposis, and abnormal hair coat. 2, 3, 17, 23, 25, 28 A pestivirus in the family Flaviviridae is the etiologic agent of BD. 11, 25 When twin fetuses are infected, BD may differ in severity and type of lesions in each fetus such that one fetus has classical lesions, such as hydranencephaly and hypomyelination, whereas the other fetus has milder white matter cavitation and porencephaly. 12, 18, 23, 25, 26
In this study, we report the pathologic, immunohistochemical, and molecular diagnostic features of concurrent PPRV and BDV infection in stillborn twin lambs. A 2.5-year-old ewe in dystocia in the fifth month of pregnancy was presented to the Gynecology and Theriogenology Clinics of the Faculty of Veterinary Medicine, Kirikkale University. Dead twin lambs were extracted from the birth canal and subjected to complete necropsy examination. Tissue samples were fixed in 10% neutral buffered formalin and embedded in paraffin. Serial sections (4-μm thickness) were cut for histopathologic and immunohistochemical examinations. Unfixed samples of skin, spleen, lymph node, lung, and brain were submitted for virologic analyses.
There were 33 ewes in the flock of origin. According to the attending veterinarian, clinical findings consistent with PPR, such as oral mucosal erosions, coughing, nasal discharge, and diarrhea, were observed in this flock. PPR was enzootic during 2004–2005 in Kirikkale Province, 14 and this flock had not been vaccinated against PPRV infection. Twelve ewes aborted during the lambing season; surviving lambs were weak, and some had arthrogryposis. In addition, pestivirus was detected by RT-PCR and IHC in a blind, incoordinated lamb that died at 2 months of age.
IHC was performed according to the LSAB-2 System HRP kit (Dako, Glostrup, Denmark) instructions. For PPRV immunostaining, tissue sections were digested with 0.1% proteinase K (VMRD, Inc., Pullman, WA, USA) for 10 minutes then incubated with rabbit anti-rinderpest (RP) antibody (Institute for Animal Health, Pirbright, Surrey, England, UK) at a dilution of 1 : 500 for 50 minutes. To demonstrate pestiviral antigens, tissue sections were digested with 0.1% protease-XIV (Sigma, Providence, RI, USA) for 5 minutes and then incubated with 1 : 1024 diluted antimouse bovine viral diarrhea virus (BVDV) 1&2 antibody (VMRD, Inc.) for 50 minutes. Aminoethyl carbasole (AEC) and 3,3′-diaminobenzidine (DAB) were used as chromogens. Tissue sections from animals with RT-PCR–confirmed PPRV or pestivirus infection were used as positive controls. Tissue sections from 2 animals with no clinical or histologic evidence of either PPRV or pestivirus infection were used as negative controls.
The RT-PCR methods for detection of PPRV 22 and pestiviral RNA 13 have been published. The PPRV primers were selected to detect the F coding gene of the virus. For pestiviral RT-PCR, the selection of primers directed to the p125 protein coding region of the BVDV genome was based on the close relationship between ruminant pestiviruses. The selected primers not only detect all type 1 viruses within the family but also distinguish between cytopathogenic and noncytopathogenic strains by the size of the resultant DNA product. Initial denaturation was performed at 94°C for 6 minutes. The reaction mixtures were then cycled 35 times at 94°C for 45 seconds, 55°C for 1 minute (60°C for 1 minute for PPRV) and 72°C for 1 minute. The final extension step in 72°C was performed for 10 minute. The PCR products were analyzed by electrophoresis after ethidium bromide staining in agarose gels.
Lamb 1
The skin was coated with keratinaceous debris (Fig. 1). The cerebral hemispheres were reduced to a sac-like layer of cortical tissue around a fluid-filled cavitation (Fig. 2). Cerebellar hypoplasia was severe. Only the basal brain structures, including pons, medulla oblongata, optic chiasm, and portions of the olfactory bulbs, were distinguishable. The kidneys were speckled with irregular cortical surfaces. Scoliosis with up to 90° deviation of the second through seventh cervical vertebrae was present. Congenital anomalies included inferior brachygnathia, gingival hypertrophy, and arthrogryposis that involved carpal, tarsal, stifle, and elbow joints.
Histologically, the thinned cerebral cortices were apposed to the pia mater and composed of few glial cells, necrotic neurons, and vessels. The normal laminar arrangement of neurons was not evident. In the skin, parakeratotic hyperkeratosis and epidermal necrosis were present. Numerous syncytial cells were in the epidermis and atrophic follicular epithelium. The lungs were affected by necrotizing and hyperplastic bronchitis/bronchiolitis and interstitial mononuclear cell infiltration. Other lesions included nonsuppurative interstitial nephritis and myocarditis. In the heart, interstitial myocarditis was accompanied by vasculitis, hemorrhage, and multifocal necroses.
Lamb 2
The fleece lacked crimp and was hairy instead of woolly (Fig. 3). A 3-cm diameter defect of porencephaly effaced the right parietal-occipital lobe of the cerebrum (Fig. 4). Subcortical cavitations throughout both cerebral hemispheres and cerebellar hypoplasia were also noted. Arthrogryposis, scoliosis, and gingival hypertrophy were observed as in lamb 1 (Fig. 5, 6).
Histopathologic findings in the brain included hyperemia, multifocal gliosis, central chromatolysis, necrosis, and neuronal calcification. Spongiosis was present in the white matter of the parietal lobes, cerebellum, medulla oblongata, and spinal cord.
According to immunoperoxidase findings; in lamb 1, PPRV antigens were detected in the epidermis. Immunoreactivity was conspicuous in epithelial and syncytial cells of the stratum spinosum as well as in the superficial keratinaceous material (Fig. 7). In the kidneys, immunoreactivity was observed in proximal tubular (Fig. 8) and renal pelvic epithelial cells, endothelial cells of cortical vessels, and interstitial macrophages. Bronchial and bronchiolar epithelial cells (Fig. 9), alveolar macrophages, and thymic reticular cells were also immunopositive.
Immunoreactivity for pestiviral antigens was present in bronchiolar epithelial cells and alveolar macrophages (Fig. 10). In the liver, granular cytoplasmic immunoreactivity was observed in hepatocytes and macrophages of the portal area (Fig. 11).
In lamb 2, PPRV immunoreactivity was observed only in bronchiolar epithelial cells and ruminal mucosa. Pestivirus immunoreactivity was strong in cerebrocortical neurons and motor neurons of the spinal cord (Fig. 12) and moderate in renal proximal tubular epithelial cells and thymic reticular cells.
Results of RT-PCR correlated well with IHC results for detection of PPRV and pestivirus in tissue samples from both lambs (Table 1 Table 2) and indicated that each lamb was infected in utero with both viruses. The spleen and lymph node samples were positive by RT-PCR for both viruses in both lambs. However, lung tissue was tested only for PPRV RNA and was negative. The presence of pestiviral RNA in the brain of both lambs indicates the possibility of viral persistence.
The primer sets used for amplification of peste des petits ruminants virus and pestivirus RNA using reverse transcriptase polymerase chain reaction.

Immunoperoxidase and reverse transcriptase polymerase chain reaction (RT-PCR) results based on tissue samples of affected twin fetuses.

Test was not performed.
PPR has been reported periodically from different regions of Turkey since 1996. 14, 19, 22, 29 The first PPR outbreak in Kirikkale Province occurred in October 2004; pathologic and virologic findings of infected animals have been reported. 14 Epidemiologic studies in the Eastern and Southeastern regions 5 and in the coastal and inland areas of Anatolia indicated that goats and sheep had high pestivirus seropositivity and increased incidence of abortion. 20
In the current case, the flock history plus histologic findings of syncytial cells in the epidermis and necrotizing bronchitis/bronchiolitis prompted us to conduct IHC and RT-PCR for PPRV infection. The results of IHC corroborated RT-PCR results; PPRV viral antigen was widespread with detection in rumen, skin, and kidneys in addition to its usual localization in pulmonary alveolar macrophages and reticular cells of the thymus. 14, 29 The unusual and widespread tissue distribution of PPRV in stillborn lambs may indicate transplacental passage of the PPRV to the amniotic fluid that subsequently contaminated fetal skin by direct contact and the rumen and lungs by swallowing and aspiration, respectively. The presence of viral antigen in the kidneys suggests that the virus also circulated in fetal blood.
Congenital malformations such as hydranencephaly, porencephaly, cerebellar hypoplasia, and arthrogryposis are characteristic of aborted fetuses from pestivirus-infected ewes. 9, 17, 23, 25, 27, 28 In the stillborn lambs of this report, pathologic, immunohistochemical, and RT-PCR findings corroborated the assumption that these malformations resulted from pestivirus infection.
PPR is quite contagious; transmission is generally thought to be horizontal. 8, 22 To the authors' knowledge, vertical transmission of PPRV or its detection in fetal tissues has not been documented. However, although PPRV is not considered an abortifacient virus, abortion has been reported in PPR outbreaks in goats, 1, 24 including a 1996 report of 105 abortions in 9 different flocks. 15 In the present study, the detection of PPRV infection in stillborn twin lambs suggests that vertical transmission of PPRV is possible. In contrast, pestiviruses readily invade and cross the placenta and can cause severe lesions in placentomes. 21 Furthermore, pestiviruses can inhibit synthesis of interferon, so pestivirus-infected cells may be more susceptible to infection by a heterologous virus. 6, 8 In this study, the presence of hydranencephaly and cerebellar hypoplasia indicate that pestivirus infection occurred before 80 to 90 days of gestation. 8, 21 We hypothesize that placental and fetal pestiviral infection early in gestation may have facilitated PPRV transmission through an injured placental barrier to the fetuses.
In conclusion, we report in utero infection with PPRV, suggesting transplacental transmission, which has not been reported previously. Further investigations are needed to determine the prevalence of congenital infection in flocks with enzootic PPR and whether concurrent pestiviral infection predisposes ovine fetuses to infection with PPRV.
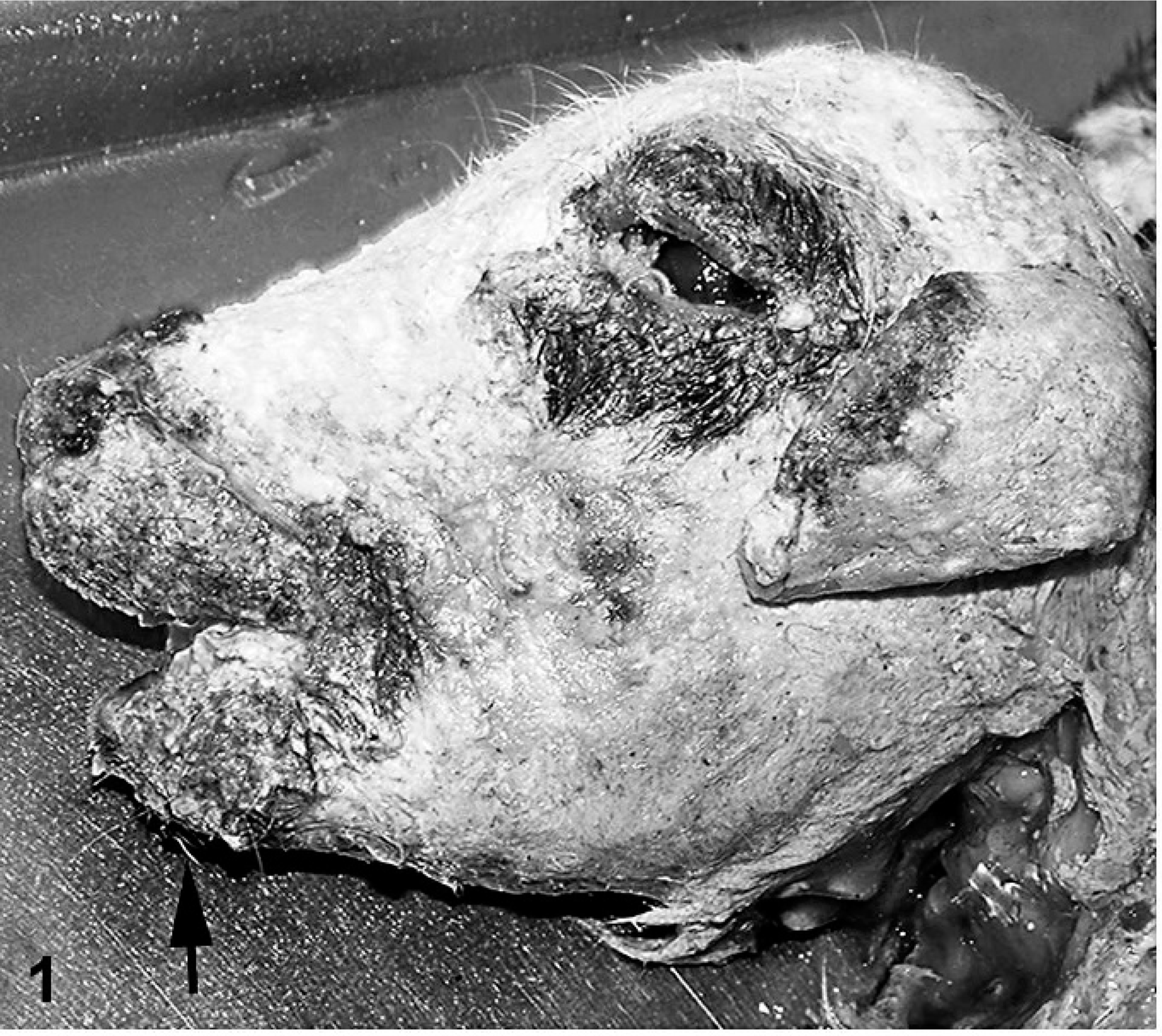
Head; stillborn lamb 1. Keratinaceous material coats the skin. Note brachygnathia inferior (arrow).
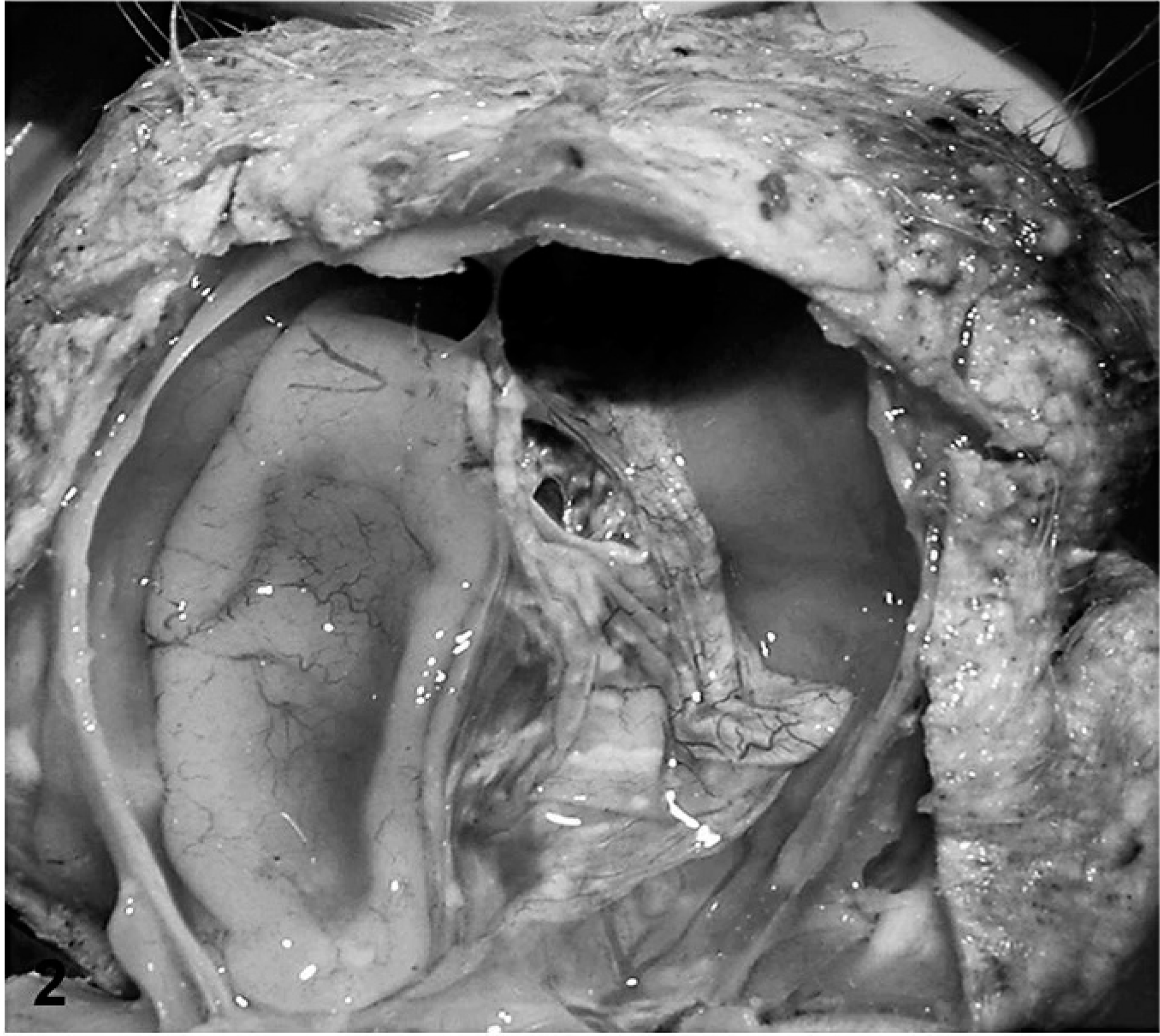
Head; stillborn lamb 1. Hydranencephaly is severe with only a thin shell of cerebral cortex adhered to the meninges.

Head; stillborn lamb 2. Long straight hairs compose the fleece. Note inferior prognathism (arrow).
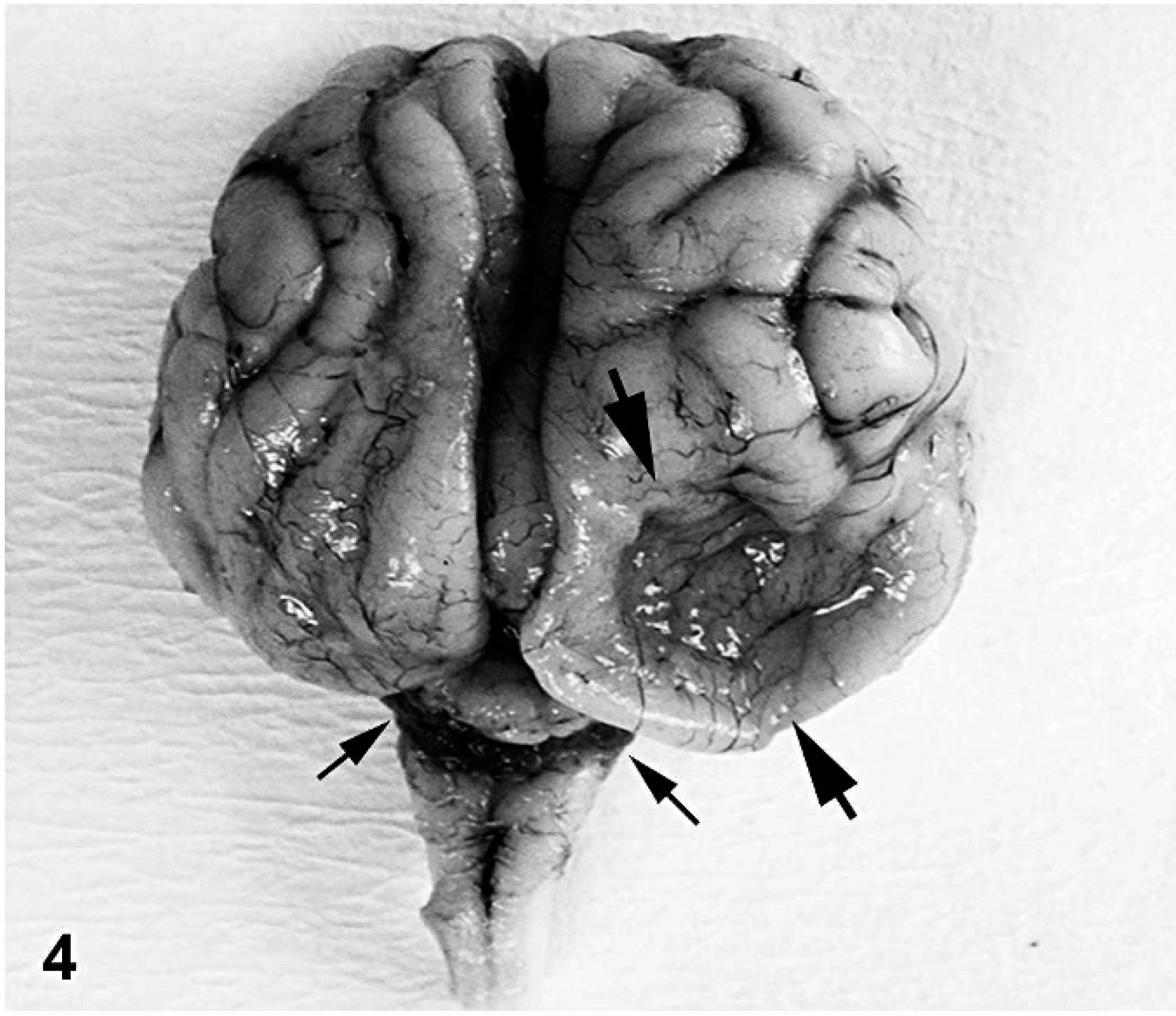
Head; stillborn lamb 2. Porencephaly in the right parietal-occipital lobe of the cerebrum (thick arrows) and severe cerebellar hypoplasia (thin arrows).
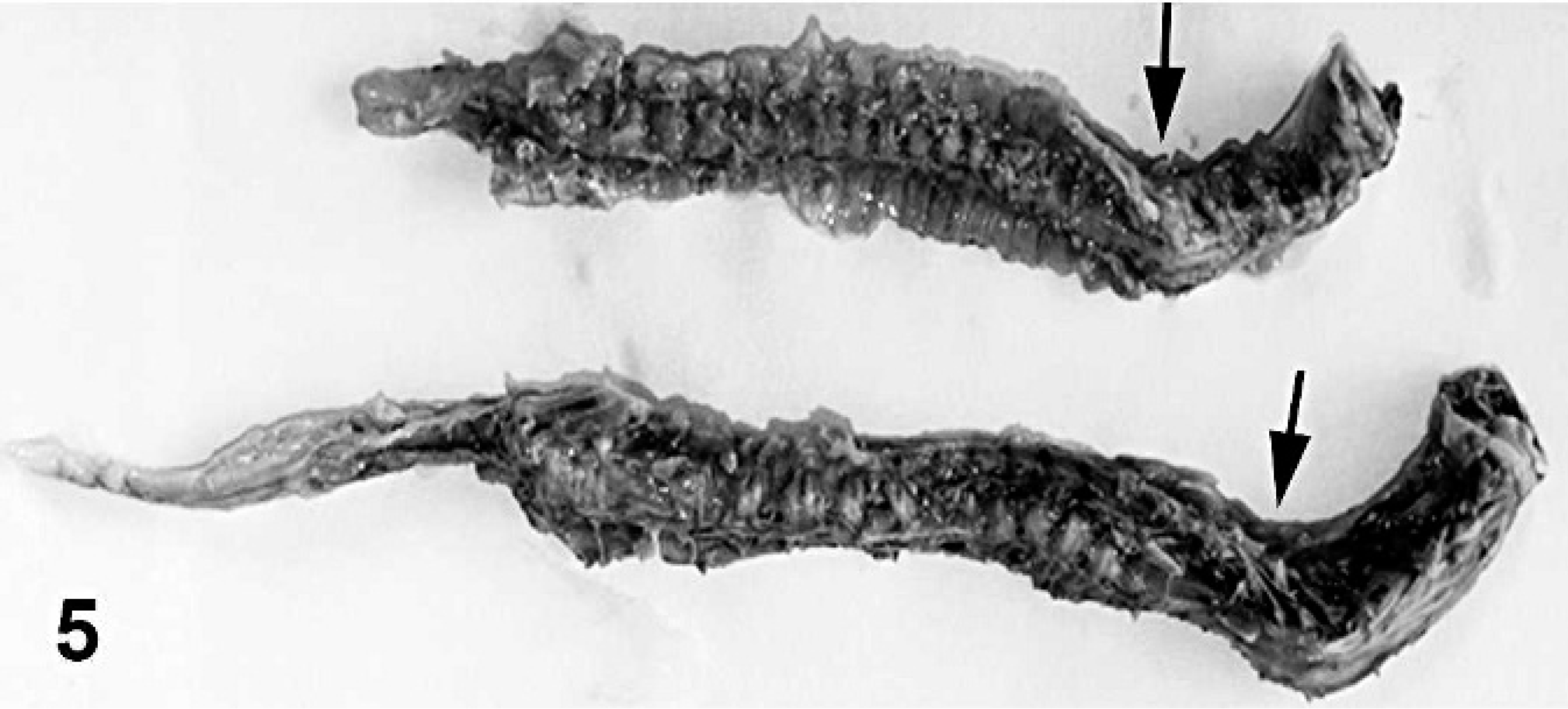
Vertebral column; stillborn lambs. Both lambs had scoliosis, with up to 90° rotation of the second to seventh cervical vertebrae (arrows).

Fore and hind limbs; stillborn lamb 1. Arthrogryposis is present to a varying degree.
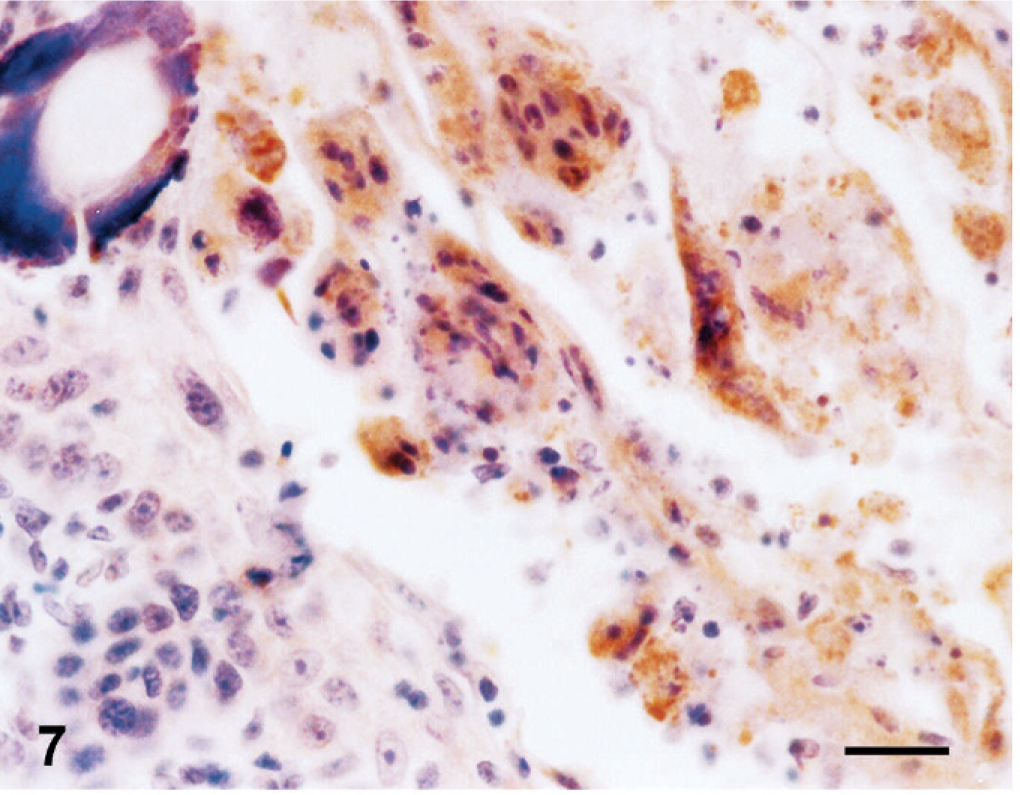
Skin; stillborn lamb 1. Peste des petits ruminants viral antigen–positive reaction in surface keratinaceous debris in epithelial cells and syncytial cells. Anti-rinderpest virus antibody, streptavidin peroxidase method, Mayer's hematoxylin counterstain. Bar = 30 μm.
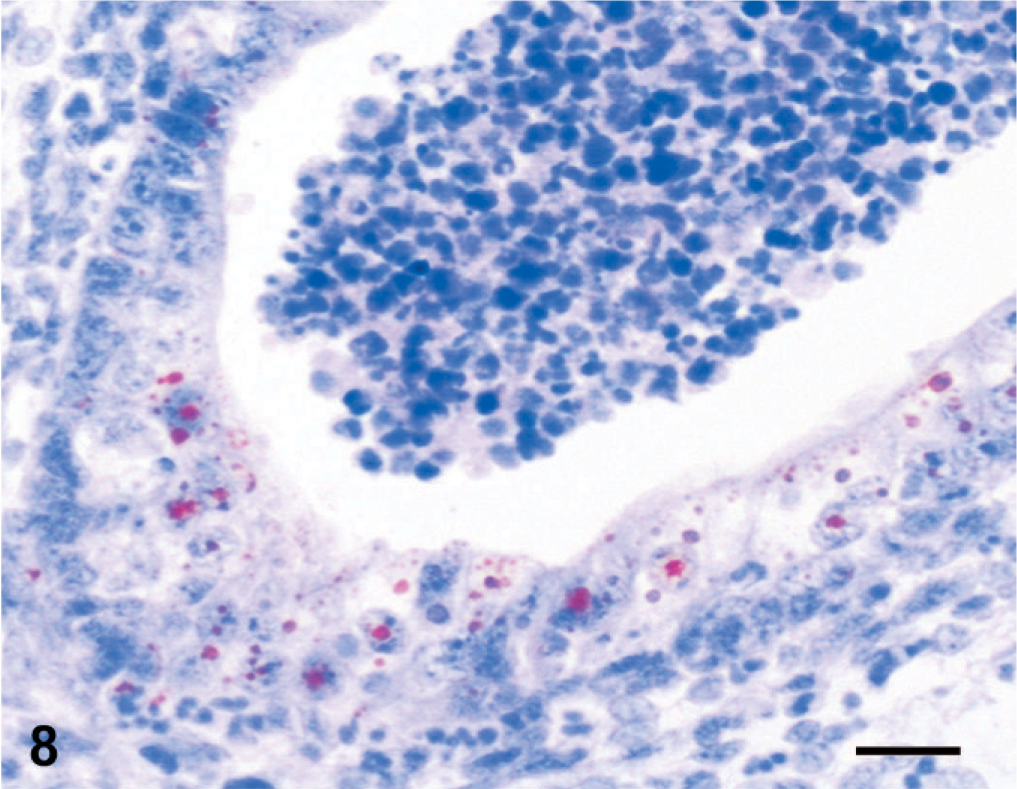
Kidney; stillborn lamb 1. Immunoreactivity for peste des petits ruminants virus in proximal tubular epithelium and necrotic luminal material. Anti-rinderpest antibody, streptavidin peroxidase method, Mayer's hematoxylin counterstain. Bar = 30 μm.
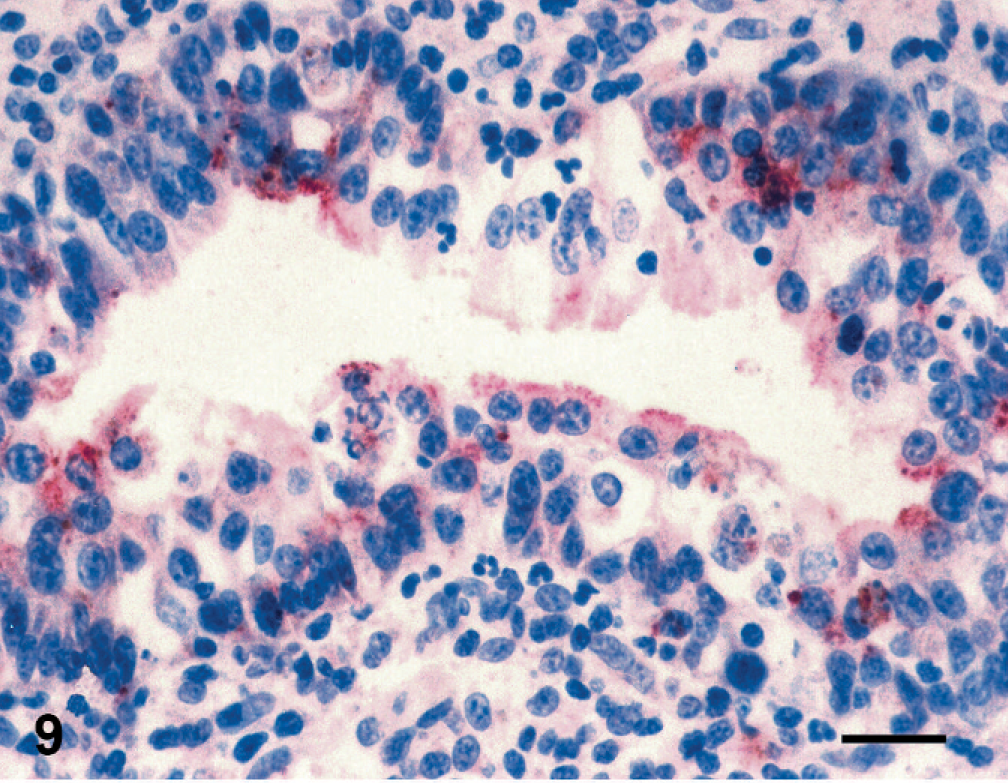
Lung; stillborn lamb 1. Cytoplasmic immunoreactivity for peste des petits ruminants virus in bronchiolar epithelial cells. Anti-rinderpest antibody, streptavidin peroxidase method, Mayer's hematoxylin counterstain. Bar = 30 μm.
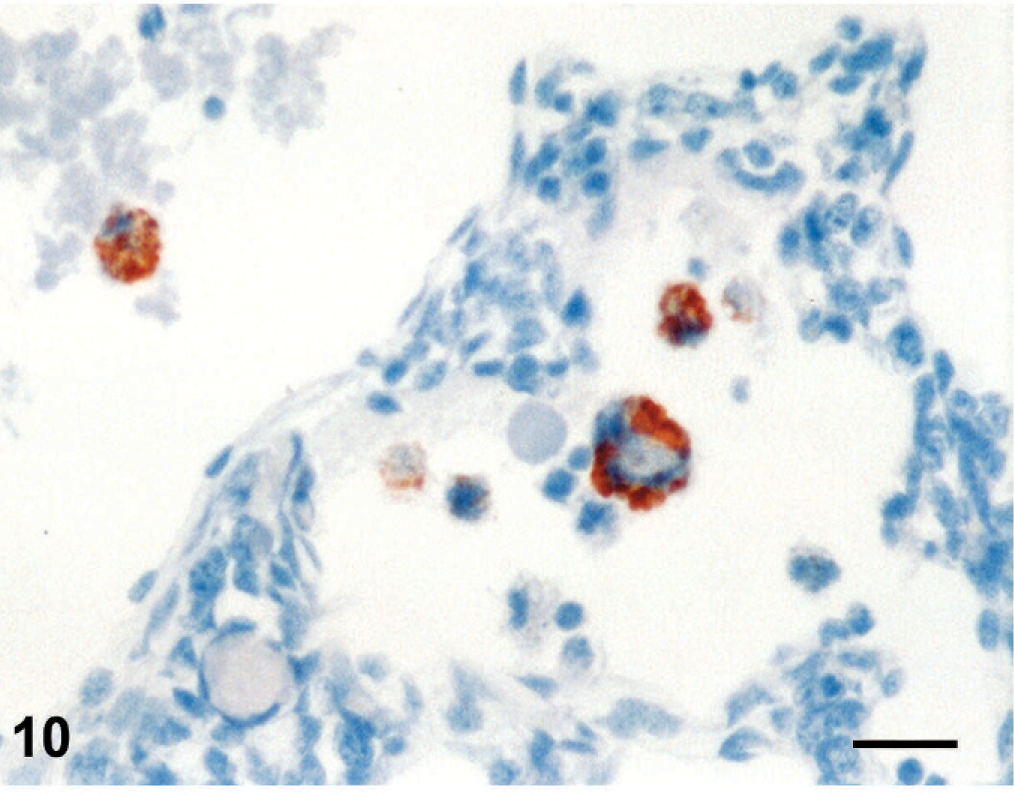
Lung; stillborn lamb 1. Pestivirus-positive immunostaining in alveolar macrophages. Anti-bovine viral diarrhea virus 1&2 antibody, streptavidin peroxidase method, Mayer's hematoxylin counterstain. Bar = 25 μm.
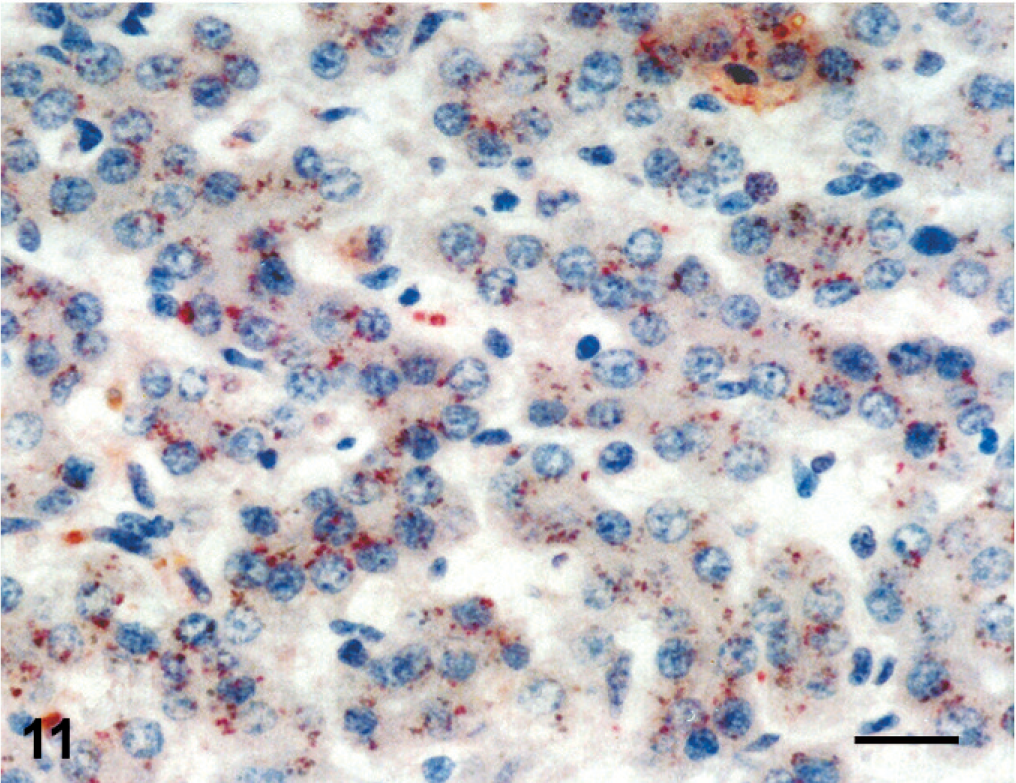
Liver; stillborn lamb 1. Granular cytoplasmic immunoreactivity for pestivirus in hepatocytes. Anti-bovine viral diarrhea virus 1&2 antibody, streptavidin peroxidase method, Mayer's hematoxylin counterstain. Bar = 25 μm.
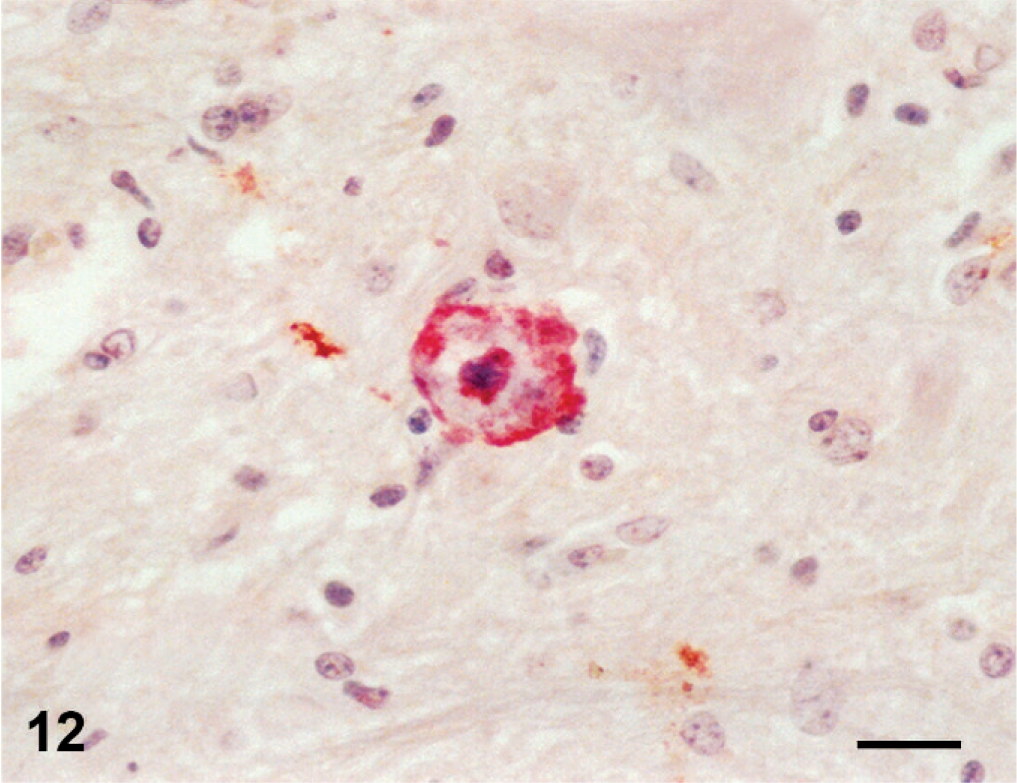
Spinal cord; stillborn lamb 2. Cytoplasmic immunoreactivity for pestivirus in a motor neuron. Anti-bovine viral diarrhea virus 1&2 antibody, streptavidin peroxidase method, Mayer's hematoxylin counterstain. Bar = 25 μm.
Footnotes
Acknowledgement
This work was supported by grants from the Kirikkale University Scientific Research Council (Project Nos. 2005/15 and 2006/19).
