Abstract
The pathobiology of H5N1 high-pathogenicity avian influenza (HPAI) virus infection in wild waterfowl is poorly understood. This study examined the pathobiology of A/chicken/Korea/IS/06 (H5N1) HPAI in 5 migratory waterfowl species—mute swans (Cygnus olor), greylag geese (Anser anser), ruddy shelducks (Tadorna ferruginea), mandarin ducks (Aix galericulata), and mallard ducks (Anas platyrhynchos)—following intranasal inoculation or contact exposure, from which all birds became infected. In mute swans, this virus had strong vascular endothelial cell tropism, producing acute severe disease and 100% mortality; the virus was detected in various parenchymal cells; and necrotic and inflammatory changes were noted in a range of organs, including pancreas, brain, spleen, heart, oral cavity, adrenal gland, lung, and liver. The ruddy shelducks had 100% mortality, but time to death was delayed, and the lesions were primarily restricted to the brain, heart, pancreas, and spleen. The mandarin ducks had only a single mortality, with lesions similar to those in ruddy shelducks. The greylag geese became infected, developed neurological signs, and had residual meningoencephalitis when examined at termination but lacked mortality. The mallards had asymptomatic infection. These results indicate variation in the pathobiology of H5N1 virus infections in different species of wild waterfowl, ranging from severe, acute systemic disease with 100% mortality to asymptomatic infection of respiratory and gastrointestinal systems.
Avian influenza (AI) is caused by infection with a type A influenz virus of the family Orthomyxoviridae. 41 Low-pathogenicity AI viruses have been isolated from numerous wild and domestic avian species, 37,41 and wild waterfowl are regarded as the primordial reservoir hosts of these viruses. 46 However, high-pathogenicity (HPAI) viruses arise from mutation of low-pathogenicity AI viruses as they circulate in poultry. 34,43 These HPAI viruses produce a severe systemic disease with near 100% mortality in chickens, turkeys, and other gallinaceous birds 31,41 but usually do not cause infection, clinical disease, or death in domestic waterfowl or wild birds, especially aquatic birds of the order Anseriformes (ducks, geese, swans). 1,6,25
Since the isolation of H5N1 HPAI virus in 1996 from a domestic goose in Guangdong Province, China, descendants of this virus have developed variable abilities to infect and cause disease in domestic ducks and wild aquatic birds under natural and experimental settings. 3 –5,7,11,14,38 In experimental infections, H5N1 HPAI viruses that were isolated between 1997 and 2001 in Hong Kong produced acute lesions and high mortality in chickens and other gallinaceous birds but only asymptomatic infections in domestic ducks or captive-raised aquatic birds. 27,31 However, the diversity of avian species for which H5N1 HPAI viruses are potentially infective, pathogenic, and lethal changed dramatically in late 2002, as shown by virus isolation from a variety of dead captive waterfowl in Penfold Park and Kowloon Park in Hong Kong, as well as from various waterfowl (ducks, geese, and swans), American flamingos (Phoenicopterus ruber), and more recently, tree sparrows (Passer montanus) in China. 9,11,20 In 2005, H5N1 HPAI virus was isolated from migratory birds found dead in Qinghai Lake of western China, a major breeding site for migratory birds, including bar-headed geese (Anser indicus). 10,23 In quick succession, mortality was found among domestic poultry and aquatic birds in western Siberia, Mongolia, Europe, Africa, and the Middle East and, in late 2006, reappeared in domestic ducks and chickens in South Korea and Japan. 35 The infection of a variety of wild birds under natural conditions raises concerns about the ecology, epidemiology, and pathobiology of these H5N1 HPAI viruses. 8,12
This study was initiated (1) to determine the susceptibility of mute swans (Cygnus olor), greylag geese (Anser anser), ruddy shelducks (Tadorna ferruginea), mandarin ducks (Aix galericulata), and mallard ducks (Anas platyrhynchos) to intranasal inoculation with or contact exposure to the A/chicken/Korea/IS/06 (H5N1) HPAI virus and (2) to describe the lesions and the distribution of virus in each species. These birds were chosen because they are naturally occurring species in South Korea and migrate to and from mainland Asia where H5N1 HPAI virus has become endemic.
Materials and Methods
Virus
A/chicken/South Korea/IS/06 (Ck/Kr/06) (H5N1) AI virus was isolated from tissues collected from affected chickens during an outbreak in November 2006 (Avian Disease Division, National Veterinary Research and Quarantine Service, South Korea). Genetically, the virus is closely related to the clade 2.2 A/Bar-headed Gs/Qinghai/5/2005 and A/whooper swan/Monogolia/244/2005 (H5N1) HPAI viruses. 3,22 Virus-infected chorioallantoic fluid of second passage in 10-day-old embryonating chicken eggs was diluted 1:300 in brain–heart infusion medium (BHI) and used as the inoculum.
Animals
Mute swans, greylag geese, ruddy shelducks, mandarin ducks, and mallards were obtained from commercial captive-breeding sources and were sham or virus inoculated or contact exposed at 7, 7, 12, 8, and 12 weeks of age, respectively. Each species was housed in self-contained isolation units (Mark 4; Controlled Isolation Systems, San Diego, CA), ventilated under negative pressure with HEPA-filtered (ie, high-efficiency particulate air filtered) inlet and exhaust air. Feed and water were provided ad libitum. All birds used in this study were cared for in accordance with the guidelines of the Institutional Animal Care and Use Committee. All experiments were performed in a biosafety level 3–enhanced facility certified by the US Department of Agriculture.
Experimental Design
For each species, birds were separated into sham and virus-exposed groups. The sham groups contained 1 to 2 birds that were intranasally inoculated with 0.1 ml of noninfected chorioallantoic fluid diluted 1:300 in BHI medium. The sham birds were euthanized on the same day when the last virus-inoculated or contact-exposed bird died or were euthanized.
The virus-inoculated group contained 2 birds that were each inoculated intranasally with 106.0 EID50 of the Ck/Kr/06 (H5N1) AI virus in a 0.1-ml dose. Twenty-four hours after intranasal inoculation, a naive bird from each species was placed in direct contact with the virus-inoculated birds (ie, contact exposed). The birds were monitored daily for illness or death.
Oropharyngeal and cloacal swabs were collected in BHI medium containing antimicrobial compounds (100 μg/ml gentamicin, 100 units/ml penicillin, and 5 μg/ml amphotericin B) from the virus-inoculated birds at 1, 2, 4, 7, and 14 days postinfection (DPI) and from the contact-exposed birds at 1, 3, 6, and 13 days postexposure (DPE). At 14 DPI (or 13 DPE), serum was collected from all surviving birds for serologic testing, and the birds were euthanized with intravenous sodium pentobarbital (100 mg/kg body weight). Necropsies were performed on all birds that died or were euthanized, and routine tissues were collected for histopathological and immunohistochemical evaluation.
Histopathology and Immunohistochemistry
Collected tissues were fixed by submersion in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Sections were made at 5 μm and were routinely processed and stained with hematoxylin and eosin (HE). A duplicate 5-μm section was immunohistochemistry stained with a mouse-derived monoclonal antibody (P13C11) specific for type A influenza virus nucleoprotein antigen (SEPRL, Athens, GA) as the primary antibody. Procedures for immunohistochemistry followed those previously described. 29 Fast red was used as substrate chromogen, and slides were counterstained with hematoxylin. Demonstration of viral antigen was based on chromogen deposition in the nucleus, which was often accompanied by chromogen deposition within the cytoplasm.
Virus Detection by Quantitative Real-Time Reverse Transcriptase Polymerase Chain Reaction
RNA was extracted from oropharyngeal and cloacal swab material as follows: 250 μl of swab material was added to 750 μl of TRIzol LS (Invitrogen Inc, Carlsbad, CA). The samples were mixed by vortexing and incubated at room temperature for 10 minutes; then, 200 μl of chloroform was added. The samples were vortexed again, incubated at room temperature for 10 minutes, and then centrifuged for 15 minutes at approximately 12,000 × g. The aqueous phase was collected, and RNA isolation was completed by extracting the RNA from the aqueous phase with the MagMAX AI/ND viral RNA isolation kit (Ambion, Inc, Austin TX) in accordance with the kit instructions using the KingFisher magnetic particle processing system (Thermo Scientific, Waltham, MA).
Quantitative real-time reverse transcriptase polymerase chain reaction (qRRT-PCR) for the influenza matrix gene was performed as previously described. 36 Samples with cycle threshold (CT) values over 38 were considered suspect and were confirmed as positive or negative by conventional reverse transcriptase polymerase chain reaction for the NS1 gene followed by gel electrophoresis. 39 Because the NS1 test is not quantitative, CT values for the NS1-positive samples were calculated based on the matrix gene qRRT-PCR test.
Serologic Assays
Agar gel precipitin and hemagglutination inhibition (HI) tests were performed on the serum (preinoculation and postinoculation) by using standard procedures. 42 The HI tests were performed by using a 0.5% suspension of chicken erythrocytes in phosphate-buffered saline.
Results
Sham-Inoculated Controls
There was no morbidity or mortality observed in sham birds of the 5 species. These birds lacked evidence of infection, including absence of antibodies against AI virus at the beginning of the study and at 14 DPI; furthermore, no AI viral genome was detected by qRRT-PCR from oropharyngeal or cloacal swabs collected during the study. Histologically, random lymphoid nodules were occasionally observed in the liver and kidney. Nonspecific immunohistochemical staining, which was restricted to cytoplasmic granules of a few individual cells, was infrequently observed in the lymphoid tissues, including the spleen, tonsils, thymus, cloacal bursa, and liver. Immunohistochemical staining of this nature has been interpreted as nonspecific staining of mast cell granules. 29 In ruddy shelducks, the spleen had random foci of mild to moderate hemosiderosis.
Infectivity and Clinical Features
Morbidity and mortality following virus exposure to the Ck/Kr/06 H5N1 HPAI virus varied among the 5 species (Table 1 ). Virus inoculation and contact exposure in all mute swans and ruddy shelducks produced clinical disease—typically, listlessness, ruffled feathers, and neurological signs such as incoordination and head tremors, which terminated in death (Table 1). The contact-exposed mandarin duck died after exhibiting listlessness for a few days, but the virus-inoculated mandarin ducks lacked clinical signs. Virus inoculation or contact exposure produced mild neurological signs of incoordination in greylag geese with recovery by 10 DPI or 11 DPE. By contrast, virus inoculation and contact exposure in mallard ducks did not produce clinical signs or death.
Clinical Features in Waterfowl Following Intranasal Inoculation or Contact Exposure With H5N1 High-Pathogenicity Avian Influenza Virus a
a Intranasally sham-inoculated control birds for each species lacked clinical and pathological evidence of virus infection.
b Ill/dead birds of total birds.
c Numbers in parentheses indicate “(days postinfection or days postexposure)” or the range therein.
The Ck/Kr/06 virus replicated in all virus-inoculated birds of all 5 species, and the virus spread to all contact-exposed birds (Table 2 ). Virus detection was more frequent from the oropharynx than cloaca, and the peak quantity was highest in oropharyngeal swab samples (ie, lowest CT values) for all species except for mallards, where the highest quantity was from a cloacal swab sample. Peak oropharyngeal swab quantity ranged from a low genome detection with 44.6 CT value in mallard ducks to a high genome detection with 25.5 CT value in mute swans. The duration of oropharyngeal virus shedding was relatively short for mallard ducks (4 DPI or 3 DPE), but virus was detected up to 7 DPI or 6 DPE in mandarin ducks and greylag geese and up to 10 DPI in virus-inoculated ruddy shelducks. Antinucleoprotein and anti-H5 hemagglutination inhibition antibodies were detected in all surviving greylag geese, mandarin ducks, and mallard ducks (Table 2), indicating that all birds became infected.
Immunohistochemical Demonstration of Viral Antigen, Real-time Reverse Transcriptase Polymerase Chain Reaction for Avian Influenza Viral Genome, and Serological Data in Waterfowl Following Intranasal Inoculation or Contact Exposure With H5N1 Virus a
a IHC, immunohistochemistry; qRRT-PCR, quantitative real-time reverse transcriptase polymerase chain reaction; AGP, agar gel precipitation; HI, hemagglutination inhibition; CT, cycle threshold; NT, not tested because of lack of survivors.
b For qRRT-PCR, the quantity of viral genome is inversely proportional to CT value (ie, the lower the value, the greater the quantity of genome detected).
c Positive birds of total tested birds.
d Numbers in parentheses indicate “(days postinfection or days postexposure)” for IHC or “(days detected, days postinfection or days postexposure)” for qRRT-PCR.
e Virus titer range from positive swab samples based on qRRT-PCR.
Gross Lesions
All mute swans had multiple white necrotic foci of 0.1 to 10.0 mm in diameter, with moderately coalescent hemorrhage in the pancreas and petechial hemorrhages in the epicardium and coronary fat of the heart. The lungs were moderately congested with edema in a single bird that died at 3 DPI. All 3 mute swans had hepatomegaly with friable parenchyma and congestion, a few petechial hemorrhages in the mucosa of the proventriculus, and moderately enlarged and congested in the spleen and kidney.
All 3 ruddy shelducks were severely dehydrated with mild to moderate dilation of the small intestine (especially the duodenum); they also had empty gastrointestinal tracts and green discolored feathers around the cloaca. The nasal sinuses and pharynx had excessive mucus accumulation. Two of 3 birds had multiple, pale, white 0.1- to 0.5-mm-diameter necrotic foci in the pancreas. The 2 virus-inoculated ruddy shelducks had congested and enlarged livers, spleens, and kidneys.
The contact-exposed mandarin duck that died 5 DPE had severe dehydration and green discolored feathers around the cloaca associated with diarrhea. In the intestine, especially the duodenum and jejunum, there was moderate congestion, severe dilation, and lack of luminal contents. The lungs were congested with ecchymotic hemorrhages. The spleen and kidney were moderately congested and increased in size. The liver was severely friable.
The 2 virus-inoculated mandarin ducks lacked lesions when examined at 14 DPI. The contact-exposed greylag goose euthanized 13 DPE had a 1.5-cm necrotic area extending from meninges into one cerebral hemisphere but not in the cerebellum. The 2 virus-inoculated geese lacked lesions when examined at 14 DPI. The virus-inoculated and contact-exposed mallards lacked lesions when examined at 14 DPI and 13 DPE, respectively.
Histology and Immunohistochemistry
Histological lesions and the corresponding viral antigen were distributed among multiple tissues in the mute swans, ruddy shelducks, greylag geese, and mandarin ducks but were lacking in all mallard ducks. Tables 3 and 4 summarize the tissue distribution and severity of histological lesions and AI viral staining.
Average Severity of Histological Lesions in Waterfowl Exposed to H5N1 Virus by Contact With Infected Birds or Intranasal Inoculation a

a —, no lesion; ±, minimal; +, mild; ++, moderate; +++, severe. Number in parentheses indicates number of birds with histopathological changes. Mallard ducks lacked histological lesions and the corresponding viral antigen.
b Histological lesions of 1 bird that died at 5 days after contact exposure.
Average Distribution of Avian Influenza Viral Antigen in the Tissues From Waterfowl Exposed to H5N1 Virus by Contact With Infected Birds or Intranasal Inoculation a

a —, no staining; ±, minimal; +, mild; ++, moderate; +++, severe. Parentheses indicate number of birds with avian influenza viral antigen in the tissue. Mallard ducks lacked histological lesions and the corresponding viral antigen.
b Antigen distribution of 1 bird that died at 5 days after contact infection.
Mute swans
Of the 5 species investigated, the most widespread distribution of histological lesions and viral antigen was observed in the mute swans. In this species, the most significant lesions were found in the oral cavity, pancreas, brain, lung, liver, spleen, heart, and adrenal gland. The oral mucosa had severe vacuolar degeneration and/or coagulative necrosis of squamous epithelium with associated intraepithelial viral antigen (Figs. 1A, 1B). The pancreata had moderate to severe, multifocal to confluent acinar necrosis with associated viral antigen in necrotic cells. The brain had focally extensive neuronal degeneration and necrosis, including vacuolation of the neuropil in the cerebrum and medulla. Mild to moderate lymphocytic perivascular cuffs, mild perivascular edema, and randomly scattered gliosis were also observed in the cerebrum and cerebellum. Viral antigen was detected in neurons, glial cells, neuropil, ependymal cells, epithelium of choroid plexi, and cerebellar Purkinje cells and neurons of granular layer. Moderate to severe multifocal coagulative necrosis of hepatocytes with minimal heterophilic to lymphocytic infiltration was identified, along with mild proliferation of Kupffer cells and necrosis of lymphocytes around portal areas.
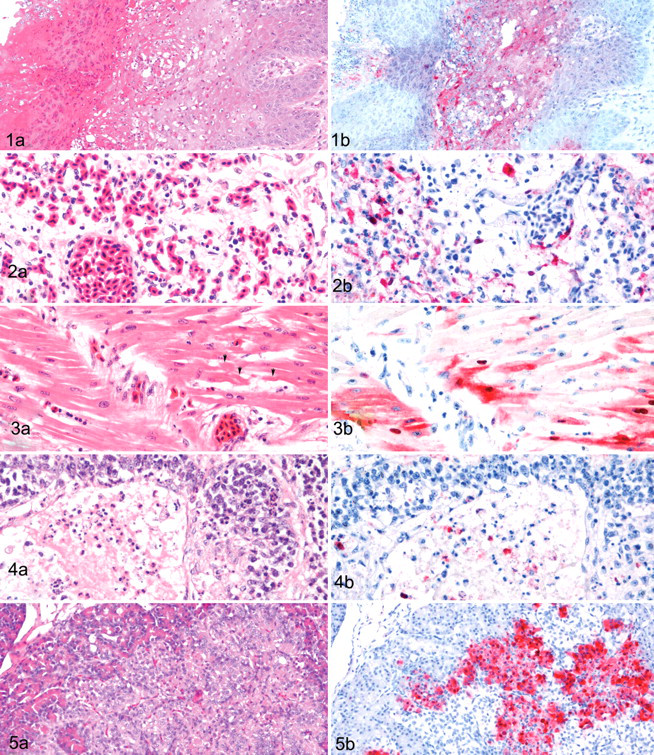
Mild to severe histiocytic interstitial pneumonia was found with vascular-oriented pulmonary damage, including edema, congestion, hemorrhage, and, rarely, microthrombosis of capillaries (Fig 2A). The interstitial pneumonia was most severe, and the greatest quantity of viral antigen was detected in histiocytes and endothelium of blood vessels (Fig. 2B) of the mute swan that died at 3 DPE. In the nasal cavity, 2 virus-inoculated swans had moderate degeneration to necrosis of mucosal epithelium with congestion and/or hemorrhage. Viral antigen was found in the epithelium, submucosal histiocytes, and adherent cellular debris.
The heart had random, multifocal myocardial degeneration to necrosis with mild hemorrhage and moderate congestion (Fig. 3A). The spleen had severe lymphoid depletion from apoptosis and necrosis with edema and severe congestion. The corticotrophic cells and, less frequently, the chromaffin cells of the adrenal gland had moderate to severe multifocal to confluent areas of cytoplasmic vacuolar degeneration to coagulative necrosis. In addition, the cloacal bursa had severe lymphocyte depletion from lymphocytic necrosis and apoptosis not related to physiological regression (Fig. 4A). Viral antigen was associated with the histological lesions, commonly in cardiac myofibers (Fig. 3B), hepatocytes and Kupffer cells, splenic histiocytes, corticotrophic and chromaffin cells in the adrenals, and histiocytes and necrotic cellular debris in the cloacal bursa (Fig. 4B).
In the absence of histological lesions, viral antigen was infrequently identified in the parasympathetic ganglia within the submucosal and myenteric plexus of the small and large intestines; tubular epithelium of the kidney; vascular endothelium in intestine, heart, nasal cavity, and brain; skeletal muscle; epidermis; and pulp of feather follicles.
Ruddy shelducks
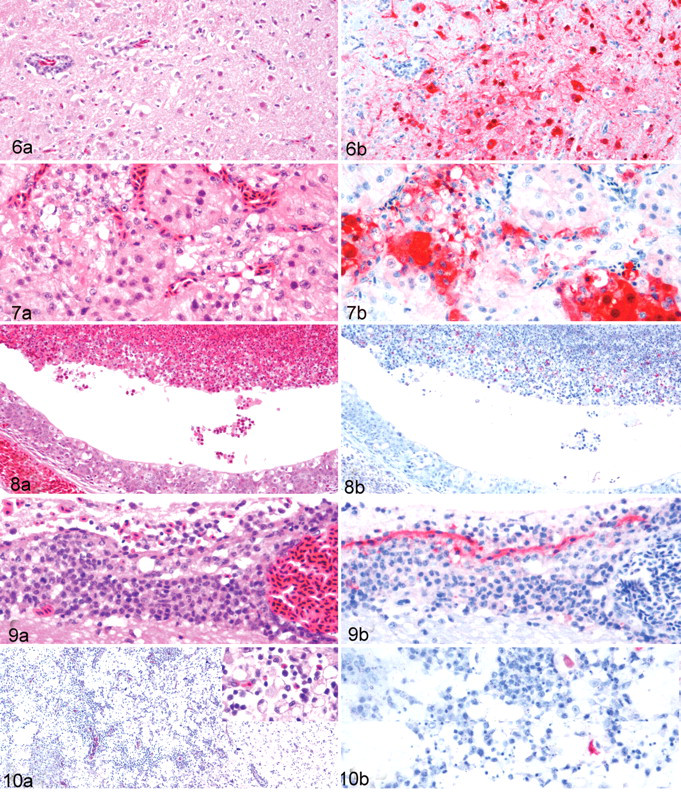
The distribution of lesions and antigenic staining paralleled those observed in the swans. However, the histological lesions, especially in the lungs, were generally less prominent and antigenic staining, less frequent. The pancreas, brain, heart, adrenal gland, and oral cavity were the tissues most affected in this species. The pancreata commonly had multifocal to confluent necrosis of exocrine parenchyma (Fig. 5A) and mild vacuolar degeneration of islets, with viral antigen being most consistent in acinar epithelium (Fig. 5B) and less frequently in islets cells. The cerebrum of the brain had moderate neuronal degeneration and necrosis, mild to moderate lymphocytic perivascular cuffs, mild perivascular edema and lymphocytic meningitis, and randomly scattered gliosis (Fig. 6A). The cerebellum had moderate necrosis of Purkinje cells, focal necrotic foci in the molecular layer, and some lymphocytic perivascular cuffs. Viral antigen was demonstrated in neuropil, neurons, glial cells (Fig. 6B), Purkinje cells and dendritic cells, granule cells, and ependymal cells. The heart had randomly scattered foci of myocyte degeneration with associated viral antigen. The adrenals commonly had degeneration and necrosis of corticotrophic cells and, less frequently, chromaffin cells, with associated viral antigen in the lesions (Figs. 7A, 7B). Mucosal epithelium of the oral cavity had focal degeneration and/or necrosis with abundant viral antigen. The spleen exhibited slight lymphocyte depletion, moderate sinusoidal congestion, and mild to moderate hemosiderosis. The liver had lysis of individual hepatocytes and mild proliferation of Kupffer cells. However, no viral antigen was detected in the liver or spleen. Viral antigen was demonstrated rarely to infrequently in scattered myofibers within the muscular layers of ventriculus (gizzard) and small and large intestines, ganglion cells in the myenteric plexi of the digestive tract, and histiocytes in the lungs. Lesions and viral antigen were absent in the trachea and skeletal muscle. An incidental finding was mild intestinal coccidiasis.
Mandarin ducks
One contact-exposed bird died at 5 DPE and had histological lesions similar to those in ruddy shelducks, but the distribution and intensity of viral antigen were more restricted in the mandarin duck, mainly affecting the nasal cavity, brain, and pancreas. Moderate to severe necrotizing rhinitis and sinusitis were present, and viral antigen was demonstrated in the necrotic cellular debris (Figs. 8A, 8B). The brain had multifocal moderate lymphocytic meningitis (Fig. 9A), diffuse moderate mononuclear perivascular cuffs, mild perivascular edema, mild focal gliosis, mild focal neuronal degeneration and necrosis, and minimal lymphocytic inflammation in the choroid plexus. Viral antigen was randomly distributed in the brain within ependymal cells, neurons, glial cells, and the epithelium of the choroid plexus (Fig. 9B). Moderate to marked multifocal necrotizing pancreatitis with mild lymphocytic inflammation was found, and viral staining was consistently demonstrated in pancreatic acinar epithelium. Additional histological changes observed in other organs included mild multifocal necrosis of cardiac myofibers, mild hepatocyte necrosis with minimal Kupffer cell hyperplasia, moderate lymphocytic depletion owing to apoptosis and necrosis in the spleen, and mild perivascular cuffs in the muscular layers of small intestine. Viral antigen was not detected in any other tissues.
The 2 intranasally infected mandarin ducks that were euthanized at 14 DPI lacked lesions and viral antigen, except for mild lymphocytic cuffs around blood vessels and peripheral nerves in the tunica muscularis of the small intestine (duodenum and jejunum).
Greylag geese
All greylag geese were euthanized at 14 DPI or 13 DPE and had lesions restricted to the brain, primarily in the cerebrum and midbrain. The brain lesions consisted of diffuse, severe malacia characterized by severe destruction of the neuropil, variable degrees of neuronal loss, moderate dystrophic calcification of necrotic neurons and neuropil, mild to moderate gitter cell and mononuclear cell infiltrates, severe lymphocytic perivascular cuffs, focal hemorrhage, and moderate spongiform degeneration (Fig. 10A). Surrounding the malacia was moderate neuronal satellitosis, marked mononuclear perivascular cuffs, and astrogliosis. The meninges had severe mononuclear cell infiltration with moderate edema. The cerebellum of the contact-exposed bird had focal neuronal necrosis. Viral antigen was infrequently detected in necrotic neurons and neuropil and rarely in glial cells and gitter cells within the malacia areas (Fig. 10B). Minor lesions that were identified included occasional urate deposition within kidney tubules and mononuclear cell infiltration around blood vessels and peripheral nerves. Viral antigen was lacking in all tissues other than the brain.
Mallard ducks
Specific lesions were lacking in all tissues, and viral antigen was not demonstrated.
Discussion
The clade 2.2 H5N1 HPAI viruses, represented by the prototype virus A/Bar-headed goose/Qinghai/1A/05, have spread from Asia to Europe and Africa, and they have been associated with mortality in swans, geese, and some wild ducks. However, mortality in the most common duck species, the mallard, has been infrequent. 8 In this study, we examined the infectivity and pathogenicity of a clade 2.2 virus, Ck/Kr/06, for 5 waterfowl species that migrate between mainland Asia, Korean Peninsula, and Japan. All 5 waterfowl species became infected following virus inoculation or contact exposure and shed virus, mainly from the oropharynx and to lower frequency and quantity than from the digestive tract. However, there were species-related variability in the infections, as evident by differences in virus genome quantity detected in oropharynx and cloaca, severity of disease, distribution of viral antigen and lesions in tissues, and mortality patterns. In field cases associated with clade 2.2 H5N1 HPAI viruses in the European Union during the winter of 2006, most cases of mortality were identified in swans, and fewer cases were seen in ducks and geese, respectively. 24,33 However, within wild ducks, the mortality varied from a few to many, depending on the individual species. 19 Experimentally or in natural cases, the most severe infections in wild ducks were reported in wood ducks (Aix sponsa), tufted ducks (Aythya fuligula), and Eurasian pochards (Aythya ferina), whereas mallards were asymptomatically infected or had low mortality rates. 3,17,19
Intranasal inoculation or contact exposure to the Ck/Kr/06 virus produced fatal infections in all mute swans, all ruddy shelducks, and 1 mandarin duck within 10 DPI or DPE. The vascular endothelial tropism (mute swans) and broad distribution of necrotic and inflammatory lesions in multiple visceral organs (mute swans, ruddy shelducks, and mandarin duck) with associated AI viral antigen staining suggest similar pathogenetic mechanisms for disseminated infections, as reported in gallinaceous birds and some other waterfowl infected with various HPAI viruses. 18,21,29,30,32 The current data are also consistent with field observations and other experimental studies that have shown (1) that swans and ruddy shelducks are highly susceptible to infection and the pathological effects of H5N1 HPAI viruses and (2) that such infections can be lethal, resulting from multiorgan virus replication and associated lesions. 3,4,8,16,17,19,24,26,44,45
In chickens and other gallinaceous species, vascular damage (eg, severe pulmonary edema, congestion, microthrombosis) and viral antigen in the vascular endothelium have been commonly reported and may be responsible for fatal outcome. 40 However, such widespread vascular damage and viral antigen in endothelium have been less commonly reported in waterfowl, most recently in black swans and wood ducks but rarely in domestic Pekin ducks or mallards infected with HPAI viruses. 2 –4,30 However, of 5 species examined in this study, only the mute swans had widespread vascular damage with antigen detected in the endothelium of capillaries, small arteries, and veins in various visceral organs, including lungs, intestine, heart, nasal cavity, and brain. These lesions could explain the early mortality possibly attributed to ischemia from vascular thrombosis and multiple organ failure or widespread dissemination of virus with multiorgan replication and damage. 40 Previous experimental studies in swans have identified endothelial tropism, but such tropism was less prominent in natural infections, suggesting multiple pathological mechanisms involved in disease and death. 4,16,26,44 Such endothelial tropism was most prominent in black swans that died at 2 and 3 DPI and less prominent in trumpeter (Cygnus buccinator), whooper (Cygnus cygnus), and mute swans that died at 4 to 9 DPI. 4
In contrast to endothelial tropism, mute swans, ruddy shelducks, and mandarin ducks had multiple virus-associated necrotic lesions in many critical organs, suggesting that failure of one or more organs (eg, heart, adrenals, pancreas, brain, liver) was responsible for clinical signs and death. Moderate to severe necrotizing pancreatitis, myocarditis, and neuronal necrosis in brain or meningoencephalitis with associated viral antigen have been most commonly identified, and such tissue tropisms for H5N1 HPAI viruses have been demonstrated in multiple avian species, including migratory waterfowl. 3,4,16,17,26,28,44 However, some waterfowl species have less severe disease signs and outcomes, typically affecting only one or two systems (eg, nervous system). In our study, the greylag geese developed nonfatal clinical neurological disease, and at termination of the study, neurotropism was confirmed by severe cerebral malacia and meningoencephalitis and demonstration of viral antigen in areas of inflammation. Clinically and histopathologically, these neuropathological findings resemble those seen in domestic geese experimentally infected with clade 0 H5N1 HPAI virus isolated during 1997 30 and in Canada geese infected with clade 1 H5N1 HPAI virus from Vietnam. 28 This indicates that these recent clade 2.2 viruses have retained their tissue tropism for the central nervous system of geese but that they can have pathophysiological effects for other visceral organs. Despite no mortality in our experimental study, we assume that infected geese in their natural habitat succumb to starvation, dehydration, or predation because of the persistent neurological dysfunction. 13
Despite considerable variation in quantity of viral genome detected and length of time for virus detection, mallards have had limited infections and no mortality with H5N1 HPAI viruses in experimental studies, including A/duck meat/Anyang/01 (clade 0), A/turkey/Turkey/1/2005 (clade 2.2), Ck/Kr/06 (clade 2.2), and A/Whooper swan/Mongolia/244/2005 (clade 2.2). 3,17 Similar asymptomatic infections have been produced in other duck species with H5N1 HPAI viruses, including northern pintail (Anas acuta), blue-winged teal (Anas discors), redhead duck (Aythya americana), common teal (Anas crecca), Eurasian wigeon (Anas penelope), and gadwall (Anas strepera). 3,17 By contrast, experimental infections in the domestic duck, which is the same genus and species as the mallard and thus has the mallard as the ancestor, has produced a range of outcomes, from asymptomatic infections to neurological disease to severe systemic infection with death. 14,15,27,40 Factors that determine severity of disease and fatal outcomes include virus strain and plaque morphology, dose of virus, age of experimental ducks, duck strain, and route of inoculation. Compared to the other 4 waterfowl species in our study, the mallards had the lowest quantity of viral genome detected in oropharyngeal and cloacal swabs and the shortest period of detection (4 DPI or 3 DPE) without clinical signs and pathological changes. Similarly, previous studies reported that when infected with other clade 2.2 H5N1 HPAI (ie, Qinghai-like) viruses, 3,17 domestic ducks lacked clinical or pathological evidence but could become infected and excrete different levels of virus, depending on the virus strain—that is, maximum titer of 103.5 TCID50/ml of A/turkey/Turkey/1/2005 (H5N1) and shed for 4 days or less, maximum titer of 102.1 EID50/ml of A/duck meat/Anyang/01 (H5N1) and shed for 2 days or less, and no virus shed with A/Whooper Swan/Mongolia/244/05 (H5N1). Interestingly, the authors of these studies drew opposite conclusions regarding the role that mallards play as potential long-distance vectors of H5N1 HPAI virus.
These results indicate a significant variation in the pathobiology of A/Chicken/Korea/IS/06 virus infections in different species of wild waterfowl. The mute swans had strong vascular tropism, 100% mortality, and the most diverse, acute, severe organ involvement. Ruddy shelducks and mandarin ducks had limited multiple-organ involvement with low to high mortality; greylag geese had pathobiology limited to the nervous system without mortality; and mallards had asymptomatic infection without clinical signs, thereby demonstrating the diverse clinical and biological outcomes following infection of waterfowl with H5N1 HPAI viruses.
Footnotes
Acknowledgements
We thank Joan Beck for the organization of experiments and technical support. We thank Kira Moresco, James Doster, and Roger Brock for assistance in the technical aspects of this project. We appreciate Jaehong Kim, Junhun Kwon, and Younjeong Lee for providing the virus isolated from chickens in South Korea.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
This study was supported by the US Department of Agriculture, Current Research Information System project No. 6612-32000-048-00D.
