Abstract
The ongoing H5N1 Asian epidemic is currently affecting a number of avian species including ducks. These birds are an important part of the poultry industry in the affected countries, and it is likely that they are acting as a reservoir of infection. Ten Pekin ducks were challenged with 100 μl containing 107 50% egg infective dose of the highly pathogenic avian influenza virus (HPAIV) A/Duck/Vietnam/12/05 (H5N1), administered by an intra-nasal and oral route. Clinical symptoms were recorded twice a day up to 14 days postinfection (dpi). Clinical signs were first noted at 2 dpi, with conjunctivitis and slight depression, and progressed over a period of 1–3 days to severe neurologic signs consisting of torticollis, incoordination, tremors, and seizures. Survival times varied from 3 to 7 dpi. On postmortem examination, hemorrhages were observed in the duodenum, ceca, proventriculus, ventriculus, trachea, pancreas, and brain. Histologic lesions, as well as immunohistochemistry positivity, were recorded in the pancreas and brain. In situ hybridization revealed viral antigen associated with acinar pancreatic cells, bronchial epithelial cells, and with cells of the central nervous system as well as neurons of the submucosal plexus of the duodenum. Our experimental findings agree with those previously observed in ducks naturally infected with HPAIV H5N1 viruses, confirming the acquired viral neurotropism and pancreatotropism, as previously noted in other avian species, as well as in humans.
Highly pathogenic avian influenza (HPAI) is an acute, generalized, fatal disease etiologically restricted only to those avian influenza virus strains of the subtypes H5 and H7, which code for a furin-sensitive cleavage site in their hemagglutinin protein and, therefore, are capable of inducing systemic infections. 24 Wild aquatic birds, including ducks, are the natural reservoir of influenza type A viruses and play an important role in the ecology and propagation of the viruses. Viruses representative of all 16 hemagglutinin and all 9 neuraminidase subtypes have been isolated from waterfowl. From this reservoir, influenza A viruses can occasionally be transmitted to other avian and mammalian hosts, including humans, and can cause outbreaks of severe disease. Influenza viruses in wild aquatic birds have long been in a state of evolutionary equilibrium, and infected hosts usually show no signs of disease. 38 However, during the 1999–2000 H7 avian influenza outbreak in Italy, mortality was observed in 2 domestic geese and 2 Muscovy ducks. 6 Since late 2002, H5N1 outbreaks in Asia have caused mortality among waterfowl in recreational parks, domestic flocks, and wild migratory birds. 8, 30 Between late 2003 and early 2005, H5N1 avian influenza spread in an unprecedented fashion across 10 Asian countries, resulting in 53 human fatalities in Thailand, Vietnam, and Cambodia and the death and slaughter of more than 150 million birds, as reported by the World Health Organization (http://who.int/csr/disease/avian_influenza/en/). During the Asian H5N1 outbreak of 2003–2005, domestic ducks were among the species affected by the epidemic, although they were not as dramatically affected as the more susceptible host, the chicken.
These recent developments, together with the lack of in-depth knowledge on the pathogenesis of highly pathogenic avian influenza virus (HPAIV) infection in ducks, have stimulated the following investigations focusing on the clinical and pathologic findings and viral spread in Pekin ducks experimentally infected with the HPAIV A/duck/Vietnam/12/05 (H5N1) using immunohistochemistry (IHC) and in situ hybridization (ISH).
Materials and Methods
Viruses
The challenge was carried out with strain A/Duck/Vietnam/12/05 (H5N1) HPAIV. The challenge strain was selected from among 20 H5N1 isolates originating from 20 different Vietnamese outbreaks in 2005. Four out of the 20 virus isolates responsible for disease and high mortality rates in Vietnamese duck flocks were typed and titrated according to Council Directive 92/40/EEC (now replaced by Council Directive 2005/94/EC, http://eur-lex.europa.eu).
The strain A/Duck/Vietnam/12/05 (H5N1) was isolated in Hanoi province during an Asian influenza (AI) outbreak in domestic ducks, confirmed on 19 July 2005. The number of virus passages in embryonated eggs has been minimized in order to avoid any possible adaptation of the isolate to egg embryos. The A/Duck/Vietnam/12/05 (H5N1) was inoculated in specific-pathogen free embryonated eggs, and the 50% egg infective dose (EID50) was calculated according to the Reed and Muench method. 21 The titer of the challenge virus was 10 7, 25 EID50/100 μl. The challenge virus was diluted in sterile phosphate buffered saline in order to obtain a viral solution of 107 EID50/100 μl.
Animals and experimental design
Fifteen 1-day-old commercial male and female Pekin ducks, clinically healthy and serologically negative to AI, were housed in biosafety level 3 facilities. Birds were fed ad libitum on a commercial feed appropriate for the growing period. Well water was provided ad libitum.
On day 52 of age, 100 μl containing 107 EID50 of the challenge virus was administered to 10 ducks by intranasal and oral route. The viral dose was equally divided between intranasal and oral routes. Five animals received a sham-inoculated control and were kept separately as negative controls. In order to evaluate the pathogenicity of the A/Duck/Vietnam/12/05 (H5N1) HPAIV, as well as the mortality and virus replication in duck tissues, birds were to be observed for 14 days postinfection (dpi), clinical signs were to be recorded twice a day, and birds that survived until the end of the observation period were to be euthanized.
Sampling
All ducks that died were immediately necropsied for determination of gross and histopathologic lesions. Skeletal muscles (leg, deep pectoral, and superficial pectoral), liver, the thoracic portion of trachea, lung, duodenum, cecal tonsils, pancreas, and brain were collected from each animal and immediately fixed in 10% neutral buffered formalin for histologic examination.
Histopathology and immunohistochemistry
After 24 hours, fixation samples were processed, paraffin-embedded, and stained with HE. Three-micrometer sections were cut, mounted on positively charged SuperFrost Plus microscope slides (Menzel, Braunschweig, Germany), dewaxed, and rehydrated. The antigen retrieval was performed by pressure cooking for 25 minutes in citrate buffer pH 6. The sections were incubated with the primary monoclonal antibody against type A influenza virus nucleoprotein (NP) (kindly supplied by Dr. D. E. Swayne, Southeast Poultry Research Laboratory, US Department of Agriculture, Athens, GA), applied at 1 : 2,000 dilution. The EnVision AP (DAKO K1396, Carpinteria, CA) detection system and nuclear fast red (DAKO K1396) were used as chromogens. Sections were counterstained with Mayer's hematoxylin. One positive and 1 negative control section were included in each IHC run.
ISH
Paraffin-embedded 3.5-μm sections were dewaxed and rehydrated successively in 100%, 90%, 70%, and 50% ethanol and finally in MilliQ sterile water (Millipore, Billerica, MA). After treatment with proteinase K (10 μg/ml; Roche, Mannheim, Germany) for 30 minutes at 37°C, the sections were fixed with 4% paraformaldehyde (Sigma-Aldrich, Milano, Italy) for 2 minutes at room temperature (RT) and then incubated in triethanolamine buffer containing 0.5% acetic anhydride for 10 minutes at RT. Dehydration was performed in 50%, 70%, 90%, and absolute ethanol, and sections were air-dried for about 15 minutes.
Sections were prehybridized in hybridization buffer without probe for 1 hour at 37°C. The probe (kindly supplied by Dr. R. Wagner, Institut fuer Virologie, Philipps-Universitaet, Marburg, Germany) is a single-stranded ribonucleic acid (RNA) (riboprobe), resulting from the transcription of a deoxyribonucleic acid (DNA) insert subcloned into a commercially available plasmid (pBluescript KS+, Stratagene, La Jolla, CA), labeled with digoxigenin (DIG). It is a 365-base pair (bp)-long fragment of the NP of influenza A virus A/FPV/Rostock/34 (H7N1) (Genbank M22576). The homology between the H7N1 and the H5N1 NP was 94%.
The DIG-RNA probe against type A influenza virus nucleoprotein was incubated in hybridization buffer (50% vol/vol formamide, 1% Denhardt's solution, 100 μg/ml transfer RNA, 100 μg/ml salmon testes DNA, 10% dextran sulfate, 1X sodium chloride-sodium citrate buffer (SSC), 0.5-M sodium chloride [NaCl]; Sigma-Aldrich) at 80°C for 5 minutes to allow possible probe secondary structure to relax. Finally, 250 ng of DIG-RNA probe were added to tissue sections, and the slides were incubated in a sealed humid chamber overnight at 42°C. Sections were washed in 50% formamide buffer/2X SSC 3 × 15 minutes at 42°C and successively in 2X SSC 3 × 15 minutes at 42°C. Sections were incubated with RNase A (20 μg/ml; Roche, Mannheim, Germany) and RNase T1 (1 U/ml; Roche) for 30 minutes at 37°C and then were washed 2 × 20 minutes in 50% formamide buffer/2XSSC at 42°C and 2 × 20 minutes in 0.2XSSC at 42°C. All steps of immunodetection were performed in a sealed humid chamber. Sections were incubated in 1% normal sheep serum (Sigma-Aldrich) in tris(hydroxymethyl)aminomethane (Tris)-hydrogen chloride/NaCl buffer for 30 minutes at RT and successively in mouse antidigoxigenin (1 : 100 in Tris 1M; Roche) overnight at 4°C. After 3 washes of 5 minutes each in Tris 1M, sections were incubated in polyclonal rabbit anti-mouse immunoglobulins (1 : 25 in Tris 1M; DAKO) for 30 minutes at RT and successively, after 3 washes of 5 minutes each in Tris 1M, with alkaline phosphatase-anti-alkaline phosphatase mouse monoclonal (1 : 25 in Tris 1M; DAKO) for 30 minutes at RT. Sections were washed 3 × 5 minutes in Tris 1M and finally were stained with chromogenic substrate system for alkaline phosphatase (Liquid Permanent Red, DAKO) for 30 minutes, counterstained with methyl green, and mounted in Ultramount (DAKO).
Sections of chorion allantoic membrane (CAM) of HPAIV experimentally infected chicken embryos were used as positive controls. In addition, CAM of uninfected chicken embryos was introduced in each ISH reaction as a negative control.
Results
Mortality and clinical signs
Intranasal and oral administration of HPAIV A/Duck/Vietnam/12/05 (H5N1) resulted in the death of all 10 ducks. Clinical signs started at 2 dpi, with conjunctivitis and slight depression, and progressed over a period of 1–3 days to severe neurologic signs consisting of torticollis, incoordination, tremors, and seizures (Table 1). Survival times varied from 3 to 7 dpi. Uninfected ducks appeared healthy throughout the period of clinical monitoring and were euthanized at 14 dpi.
Clinical signs, histopathology, and results of IHC and ISH of ducks intranasally and orally inoculated with HPAIV A/Duck/Vietnam/12/05 (H5N1).
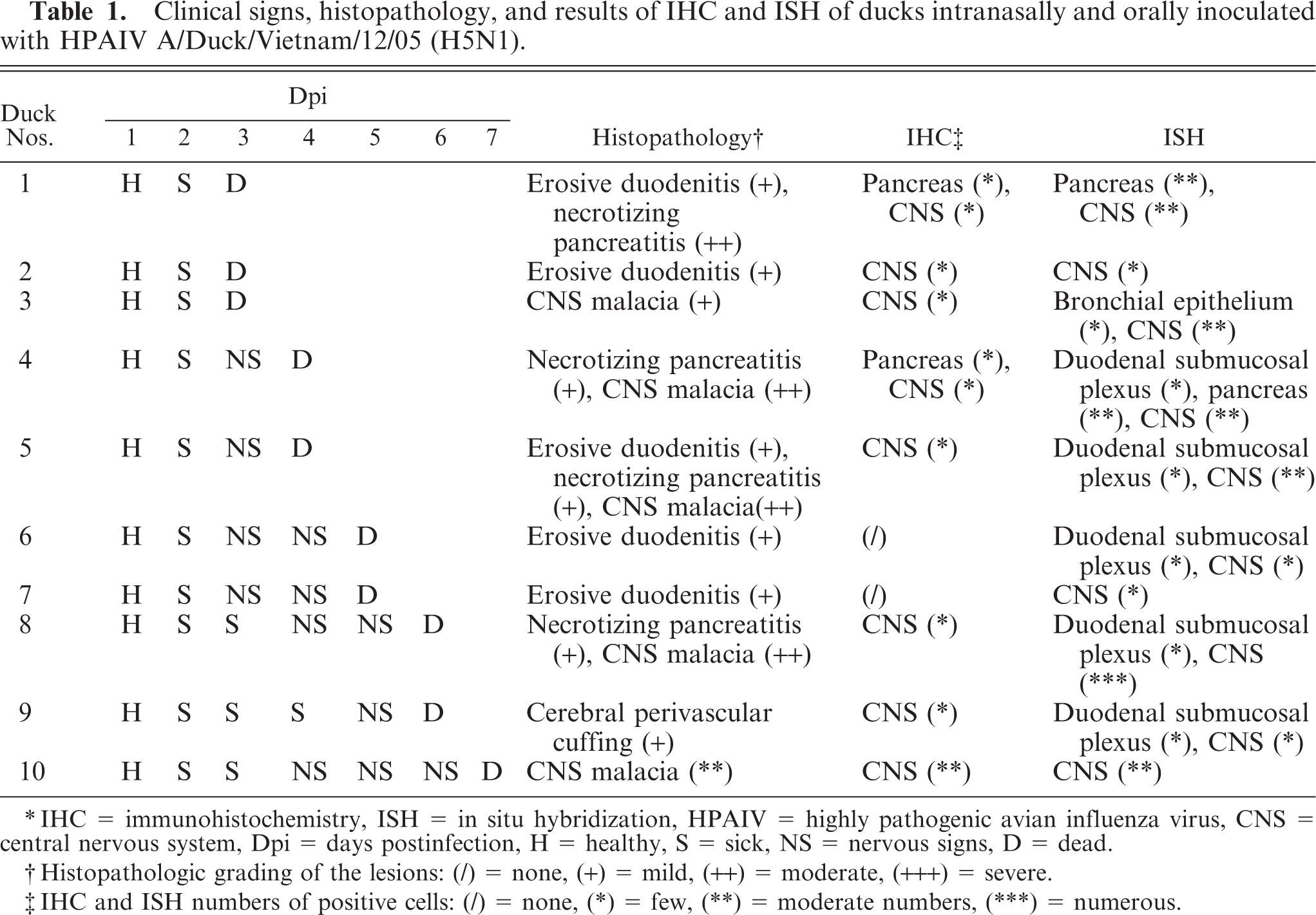
∗ IHC = immunohistochemistry, ISH = in situ hybridization, HPAIV = highly pathogenic avian influenza virus, CNS = central nervous system, Dpi = days postinfection, H = healthy, S = sick, NS = nervous signs, D = dead.
† Histopathologic grading of the lesions: (/) = none, (+) = mild, (++) = moderate, (+++) = severe.
‡ IHC and ISH numbers of positive cells: (/) = none, (∗) = few, (∗∗) = moderate numbers, (∗∗∗) = numerous.
Gross pathology
At necropsy, mild lesions consisting of petechial hemorrhages in the duodenum, ceca, proventriculus, and ventriculus were observed in infected ducks. Congestion of the duodenal mucosa with hemorrhagic content was observed in 5 birds (50%). Hemorrhages of the pancreas (3/10, 30%) and trachea (2/10, 20%) were also present in some birds. No gross lesions were observed in negative controls.
Histopathology
Histologic lesions were mainly observed in the brain (6/10, 60%) and pancreas (4/10, 40%) of the ducks that developed clinical signs. Multifocal, moderate-to-severe necrosis of the neurons and glial cells (Fig. 1) and occasional neuronophagia were observed in cerebral gray matter. Rarely, vessels showed multifocal perivascular cuffing, with lymphocytes and fewer histiocytes. No histologic lesions were observed in either the cerebellum or brain stem. In the pancreas, multifocal necrosis of the acinar cells was present (Fig. 2), while no lesions of the islet cells were observed. In 5 ducks, histologic examination revealed sloughing of the epithelium of the upper part of the villi and rare foci of hemorrhages in the mucosa of the duodenum (5/5, 50%). No further lesions were detected among ceca, skeletal muscles, liver, the thoracic portion of trachea, and lung. No lesions were observed in the uninfected ducks.
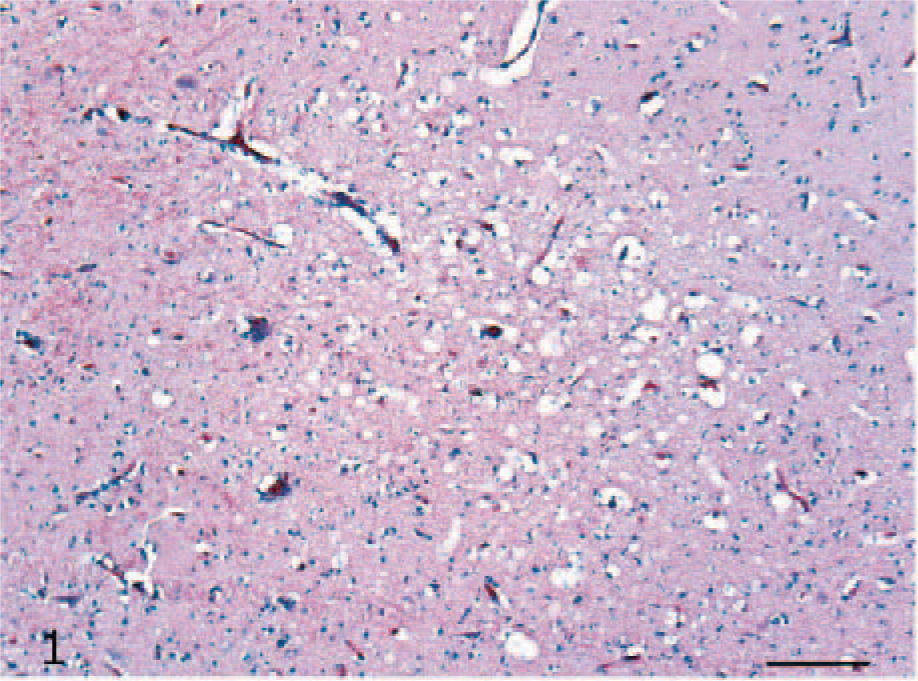
Cerebrum; H5N1 experimentally infected duck. Discrete focus of necrosis and congestion. HE. Bar = 50 μm.
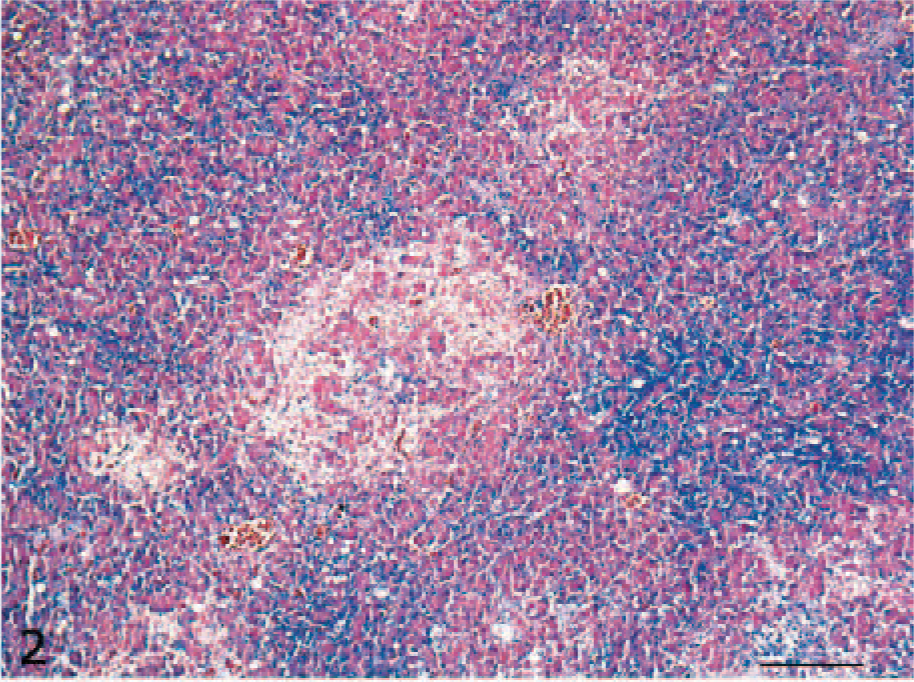
Pancreas; H5N1 experimentally infected duck. Focal necrosis of the acinar pancreatic cells. HE. Bar = 100 μm.
IHC
Viral antigen was detected in the cerebrum, brain stem, and pancreas, mainly in association with histologic lesions. Intranuclear and intracytoplasmic staining was seen in neurons and glial cells (Fig. 3) of the cerebral gray matter and brain stem in 8 of the infected ducks (8/10, 80%). In the pancreas, immunolabeling was detected in the nucleus and cytoplasm of necrotic acinar cells of 2 ducks (2/10, 20%) (Fig. 4). No nucleoprotein antigen was detected in negative controls.
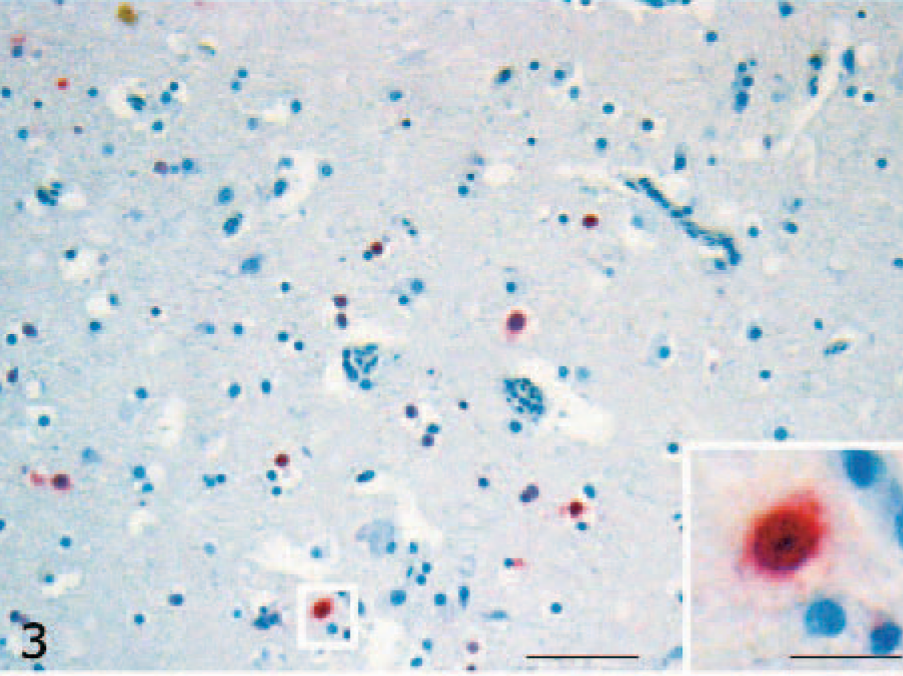
Cerebrum; H5N1 experimentally infected duck. Intranuclear and intracytoplasmic immunohistochemical staining for avian influenza virus (nucleoprotein in a limited number of cerebral neurons) (
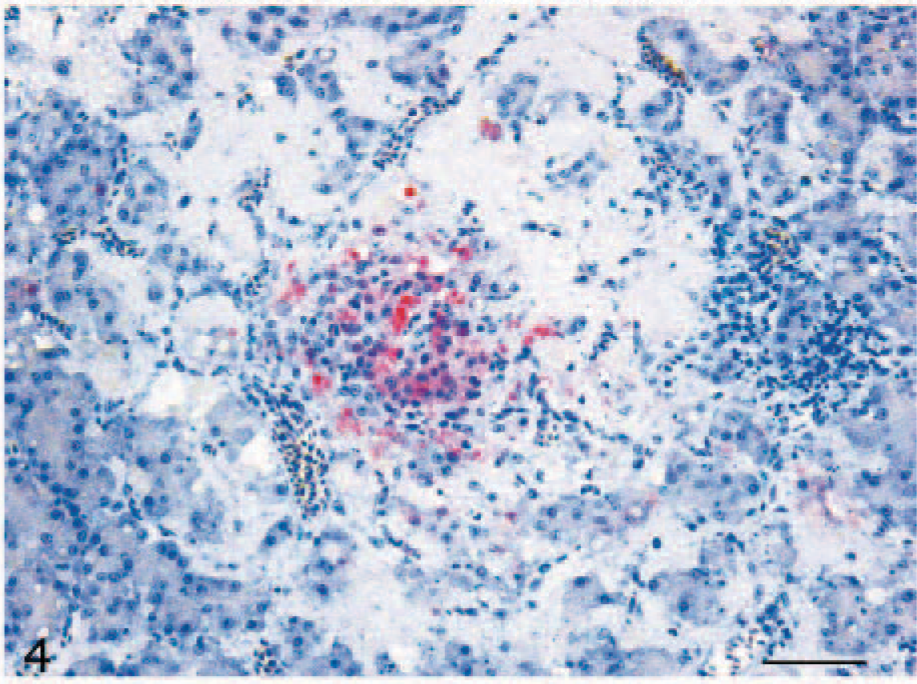
Pancreas; H5N1 experimentally infected duck. Intranuclear and intracytoplasmic staining for avian influenza virus nucleoprotein in necrotic acinar cells. EnVision AP (DAKO) with Mayer's hematoxylin counterstain. Bar = 100 μm.
ISH
Viral RNA was detected in neurons and glial cells of the cerebral gray matter (Fig. 5) and brain stem of all symptomatic ducks. Furthermore, positive staining was observed in cerebellar Purkinje neurons. The ISH-positive reaction was of stronger intensity and wider distribution than IHC immunolabeling in all the organs evaluated. Viral RNA was also observed in nervous cells of the submucosal plexus of the duodenum in 5 infected ducks (5/10, 50%) (Fig. 6). In addition, ISH revealed viral RNA associated with necrotic cells of the pancreas of 2 ducks (2/10, 20%) and in epithelial cells of the bronchi of 1 duck (1/10, 10%). No immunolabeling was observed in negative controls. The results of histologic examination, IHC, and ISH are reported inTable 1.
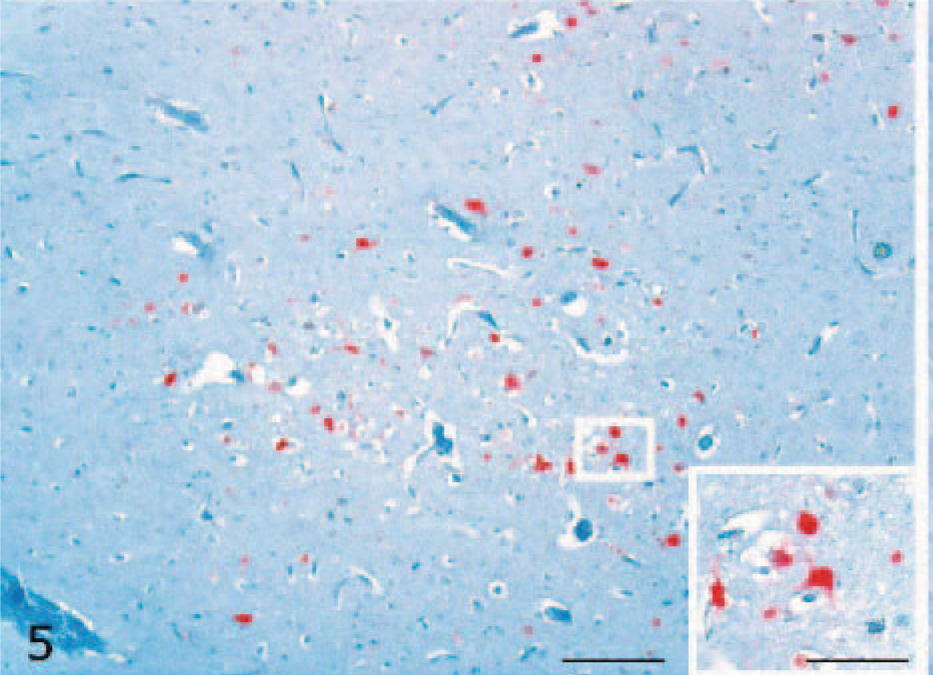
Cerebrum; H5N1 experimentally infected duck. Intranuclear and intracytoplasmic detection of viral nucleoprotein ribonucleic acid in cerebral neurons (
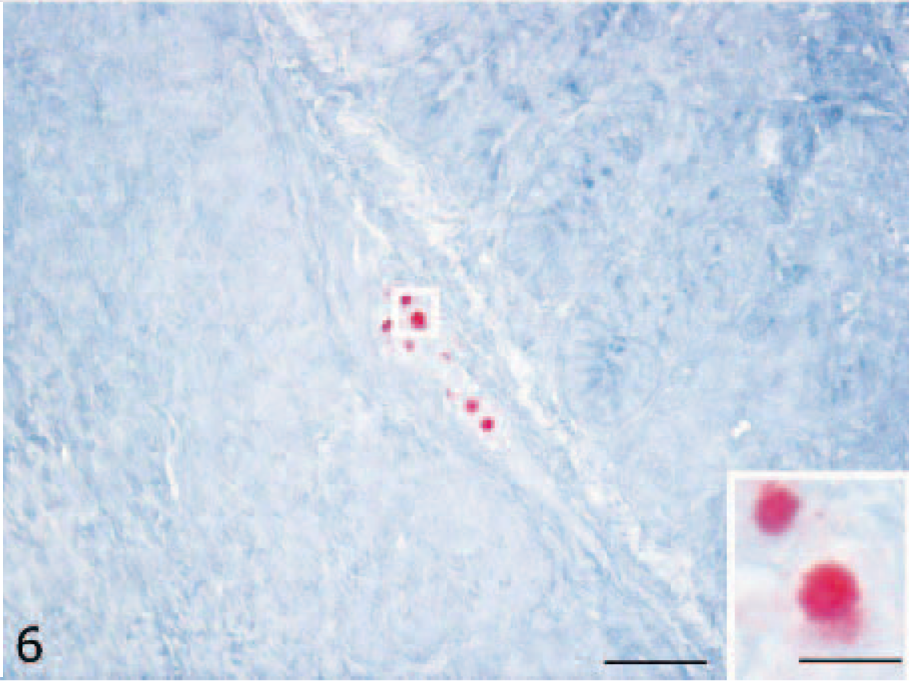
Submucosal duodenal plexus; H5N1 experimentally infected duck. Viral ribonucleic acid in the nucleus of nervous cells (
Discussion
Waterfowl, the natural reservoir of all influenza A viruses, usually carry the infection without signs of disease. 1, 2, 12, 33, 38 Most studies emphasize the ducks' resistance to disease development after experimental infection with H5N1 influenza virus. 7, 25 In the past 5 years, there have been more frequent reports of ducks developing severe disease, such as neurologic signs and mortality after infection with HPAI viruses during outbreaks. 6, 8 The H5N1 avian influenza viruses isolated in late 2002 in Hong Kong, which caused the first cases of lethal influenza virus infection reported in wild aquatic birds since 1961, were antigenically distinct from previously described H5N1 viruses and were neurotropic and pathogenic in ducks. 30 For this reason, the relatively new phenomenon of viruses being pathogenic in their natural reservoir is intriguing, and there is considerable scientific interest in better understanding the pathogenesis of H5N1 viruses in ducks.
In the present study, intranasal and oral administration of HPAIV A/Duck/Vietnam/12/05 (H5N1) resulted in high morbidity and mortality in Pekin ducks, within 7 dpi. Clinically, the affected ducks showed severe neurologic signs. Histopathology, IHC, and ISH demonstrated a marked neurotropism of the virus. Furthermore, pancreatic damage associated with virus replication was observed. Previous studies demonstrated that in those avian species susceptible either naturally or experimentally to selected HPAIVs, the virus tends to consistently localize to both the brain and the pancreas, with variable or minor lesions in other organs. 19, 20 Interestingly, although no clinical signs of disease were observed in DK/Anyang/AVL-1/01 high-pathogenicity H5N1 virus–inoculated ducks, infectious virus could be detected in lung tissue, cloacal, and oropharyngeal swabs. This virus was unique among the H5N1 isolates in that infectious virus and viral antigen could also be detected in brain and muscle tissue of ducks. 34, 35 The susceptibility of emus, domestic geese, domestic ducks, and pigeons to the experimental infection with the H5N1 HPAIV that emerged in Hong Kong in 1997 has been also investigated. Both emus and geese developed pancreatitis, meningoencephalitis, and mild myocarditis, and viral antigen was detected in affected organs by IHC. In contrast, viral infection in ducks was pneumotropic. Pigeons were resistant to viral infection. 19 Among several other avian species, the brain was consistently affected, but microscopic lesions and viral antigen were also observed widely distributed in other organs. 18, 20, 36
Our findings agree with those recorded in ducks and geese during the outbreak of H7N1 avian influenza virus, which occurred in Italy during 1999 and 2000. Mortality and nervous signs were recorded, and histologic examination revealed a limited number of necrotic foci of the acinar cells of the pancreas and mild lymphocytic encephalitis with perivascular cuffing. IHC positivity was observed in the pancreas and central nervous system (CNS) in both species. 6 In a recent study, the susceptibility of experimentally infected pigeons to the HPAIV H5N1, associated with the neurotropism of the virus, was demonstrated. 13 These studies indicate that there is a significant variation in the pathogenicity of H5N1 HPAIVs in avian species, even though the neurotropism is a recurrent feature among birds that eventually succumb to infection. Infections of the CNS with HPAIV of the subtype H5 have been observed also in a wide variety of mammalian species, particularly in mice 22 and rats, 4 but also in humans 11 and feline species, including domestic cats 23 and tigers, 3 in ferrets 39 as well as in dogs. 28 However, it is important to note that these infections with H5 HPAIVs in mammalian species have largely been only successfully induced experimentally through selected routes of inoculation, 22, 23, 39 as opposed to those in avian species that are both spontaneously and experimentally induced through a variety of routes. 7– 9, 13, 14, 36
There is evidence for at least 2 routes of virus entry into the nervous system. It has been demonstrated that HPAIV replicates in endothelial cells during the early stage of infection in chickens, and virus spread in the body occurs following viremia. 5, 14, 32 After penetrating the blood-brain barrier, infection is propagated to neurons and glial cells. 27 A second route of CNS invasion has been demonstrated in experimentally infected mice, by ascending trans-synaptic transmission of H5N1 via vagal, olfactory, and trigeminal nerve fibers. 16, 17, 29 In cats, a third route of entry from the intestinal lumen has been postulated. The submucosal and myenteric plexuses of the small intestine may have been infected directly from the intestinal lumen via nerve fibers projecting through the mucosa and interacting directly with surface epithelial cells. 23 In our investigation, no viral genome was detected in endothelial cells. In contrast, viral RNA detection in nervous cells of the submucosal plexus was evident. However, since all the examined birds spontaneously succumbed to viral infection, no pathologic data about the early stages of the infection are available and no conclusion about the route of entry of the virus in the CNS can be drawn.
Influenza A viruses have generally been found to replicate preferentially in the intestinal epithelial cells of ducks. 10, 37 However, the newer H5N1 viruses are shed primarily from the upper respiratory tract. 8, 30 In our study, neither viral antigen nor viral RNA was detected in the epithelium of the intestinal tract. In addition, only 1 duck had a few bronchial epithelial cells positive for viral RNA by ISH, while all ducks that died with clinical symptoms were positive for virus in the CNS. These results could indicate that the immune-privileged nervous system is the preferred site of replication of the HPAIV A/Duck/Vietnam/12/05 (H5N1) in ducks according to the time course of the infection and the sampling times of the study. Furthermore, neurotropism is likely to be the predominant factor in the production of morbidity and mortality relative to infection with this HPAIV virus in ducks. A recent study showed that the recent Asia-wide outbreak of H5N1 influenza was caused by a dominant H5N1 Z genotype. 15, 31 This homogeneity in virus genotype contrasts with the pathogenic potentials of the virus isolates, which ranged from nonpathogenic to lethal. Therefore, H5N1 pathogenicity in ducks does not correlate with genotypes and may be due to genetic traits more subtle than genotypes, such as an individual allogene or even just a few specific amino acids. 31 A reverse genetic approach identified a single amino acid substitution, Glu-to-Lys at position 627 in the PB2 protein, which affects replicative efficiency but not cell tropism, of Hong Kong H5N1 influenza A viruses in mice. 26 The molecular basis of the neurotropism of certain AI virus strains remains to be further elucidated.
Through the application of IHC, the localization of viral antigen was found to closely correlate to clinical manifestations of disease and the histologic lesions detected. In some samples, viral demonstration in necrotic pancreatic foci was not possible, presumably due to the extensive necrosis of affected cells. In contrast, both IHC and ISH were able to reveal viral infection in individual cells before the development of histologic lesions. ISH was more sensitive than IHC, revealing a small amount of viral RNA in some samples where viral nucleoprotein has not been detected by IHC. ISH has been previously applied in studying the AI pathogenesis and the cell tropism of the virus. 9, 16 This method, applied on paraffin-embedded sections, appears to be very useful in the study of the pathogenesis of the AI infection, particularly in the case of low viral-replication rates.
In conclusion, the pathologic findings of HPAIV A/Duck/Vietnam/12/05 (H5N1) have been investigated in experimentally infected Pekin ducks. The virus showed high pathogenicity, associated with marked CNS and pancreatic damage. IHC and ISH detected virus spread even in cells and tissues where histologic lesions were not present, showing the strong viral neurotropism in ducks.
Footnotes
Acknowledgements
We acknowledge the skillful technical assistance of the personnel of the Virology Laboratory and of the Animal Care staff of the Istituto Zooprofilattico Sperimentale delle Venezie. We thank Alberto Masiero for assistance with photographs. Dr. David E. Swayne, Southeast Poultry Research Laboratory, US Department of Agriculture, Agricultural Research Service, Athens, GA, is gratefully acknowledged for providing the influenza A nucleoprotein monoclonal antibody. Dr. Ralph Wagner, Institut fuer Virologie, Philipps-Universitaet, Marburg, Germany, is gratefully acknowledged for providing the ISH probe, and Dr. Bjorn Keiner, for the information provided. The work has been financially supported by European Project AVIFLU QLRT-CT2001-01454.
