Abstract
The weakly beta-hemolytic porcine spirochete Brachyspira murdochii is considered a normal intestinal commensal. In the present study, however, a field case of B murdochii–associated catarrhal colitis was identified in a pig, as characterized by extensive spirochetal colonization of the surface epithelium. Experimentally, 8 weaned pigs were challenged with the B murdochii isolate, reproducing catarrhal colitis in 2 animals. By applying fluorescent in situ hybridization using a species-specific oligonucleotide probe targeting 23S rRNA, B murdochii organisms were found in high numbers and were closely associated with the surface epithelium in the pigs with catarrhal colitis. The results indicate that, when present in high numbers, B murdochii is low pathogenic for pigs.
Spirochetes—long, helically coiled bacteria—are often found in the large intestines of animals affected by diarrhea. The most common porcine intestinal spirochetes comprise several species of the genus Brachyspira: B hyodysenteriae, B pilosicoli, B intermedia, B innocens, and B murdochii.2 The first 2 species are well-known porcine pathogens causing swine dysentery, severe mucohemorrhagic colitis, and the less severe disorder spirochetal colitis. 3,5,6 The importance of the other spirochetes as intestinal pathogens has not yet been investigated thoroughly. In addition to affecting pigs, B murdochii has been isolated from hens 13 and rats (wild and laboratory) 14 without history of intestinal disease. Hitherto, B murdochii has not been considered to be pathogenic, 2,4,11 but the recent detection of B murdochii by polymerase chain reaction (PCR) in cases of porcine colitis negative for B hyodysenteriae and B pilosicoli warrants speculation on the potential pathogenecity of the bacterium. 15
The aim of this study was to report a case of naturally acquired colitis and experimental colonic infection caused by B murdochii in pigs. For demonstration of the spirochete in formalin-fixed tissue, fluorescence in situ hybridization (FISH) was developed and applied using an oligonucleotide probe specific for B murdochii.
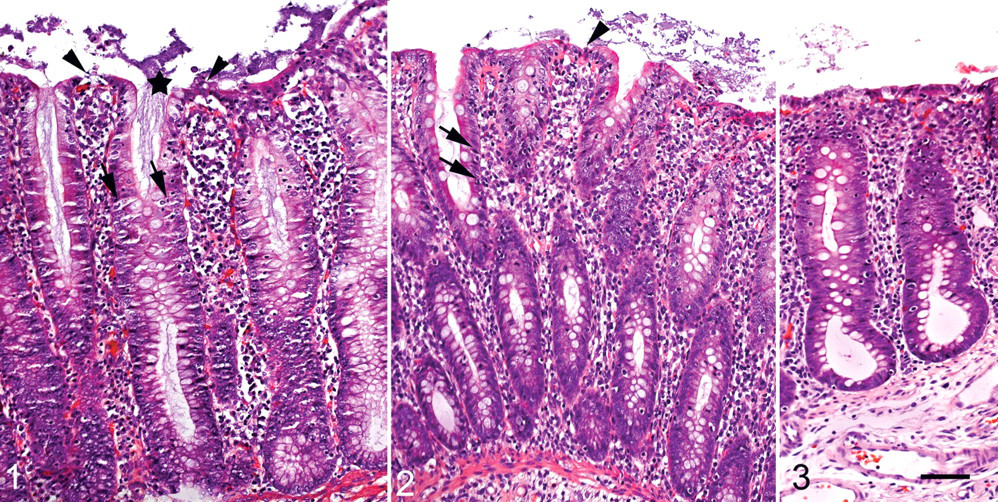
From a farm with suspicion of spirochete-associated colitis, a 9-week-old pig with typical symptoms was euthanized. Intestines, including formalin-fixed ileum and colon samples, were then submitted for routine laboratory examination. The formalin-fixed samples were processed for normal histopathology, embedded in paraffin, and sectioned at 3 µm. Macroscopically, the colonic mucosa was thickened without signs of blood or excess mucus. The colonic contents were liquid. Microscopically, there was catarrhal colitis with large numbers of spirochetes colonizing the surface epithelium and the upper part of the crypts. Sloughing of surface epithelial cells, hyperplasia of crypt enterocytes, necrosis of single crypt enterocytes, and infiltrate of mononuclear cells in lamina propria characterized the colitis, as shown in Fig. 1. Based on in situ hybridization according to Boye et al, 1 with a hybridization and washing temperature of 45°C, the spirochetes were identified as Brachyspira spp. Hybridization testing for B hyodysenteriae and B pilosicoli was negative. The extensive spirochetal colonization, including frequent infiltration of surface epithelium and the associated colitis, strongly indicated a pathogenic importance of the spirochete, which was identified as B murdochii (see below).
Feces culture 12 yielded only a weakly beta-hemolytic spirochete. 12 The isolate (02-11287) was biochemically identified as B murdochii 2,11 and further characterized by sequencing of the gene for 16S ribosomal RNA, revealing 100% homology with the type strain for B murdochii ATCC 51284. The 23S rDNA comprises longer regions of hypervariability than does the 16S rDNA and therefore provides a better target molecule for molecular detection methods. 10
A specific oligonucleotide probe (Table 1 ) was designed on the basis of 23S rRNA gene sequences from the Probe Design function in the software ARB (Technische Universität München, Munich, Germany; see http://www.arb-home.de/). Testing the specificity of the novel probe was done in formalin-fixed tissue samples, each containing 1 of 6 reference strains—B hyodysenteriae (B 78), B pilosicoli (P 43), B intermedia (ATCC 51140), B innocens (ATCC 29796), B murdochii (ATCC 51284), and B aalborgi (ATCC 43994)—revealing only hybridization signal in sections with B murdochii.
Sequences, Target Sites (Escherichia coli Numbering), and Specificities of rRNA-Targeted Oligonucleotide Probes Used for In Situ Hybridization
A challenge study was designed in an attempt to fulfill Koch postulates in respect to colitis. From a high-health herd known to be free from B hyodysenteriae and B pilosicoli, 14 six-week-old pigs were obtained and allocated into 2 groups of 8 and 6 but otherwise fed and housed in the same manner. The group of 6 pigs was kept as noninoculated control animals, whereas the 8 pigs were challenged with B murdochii (02-11287) similar to that described by Lindecrona et al. 9 Approximately 1 × 106 colony-forming units were given to each pig by stomach tube daily for 3 consecutive days. Although the doses of inoculums were smaller compared with those of previous challenge studies with B hyodysenteriae and B pilosicoli, 5,6,8,9 5 pigs started shedding B murdochii 6 days postinfection (p.i.). A similar early shedding, indicating a relative immediate colonization of the pig, is common for B hyodysenteriae, whereas the first shedding of B pilosicoli is normally seen a few days later. 5,6,8,9 The shedding was intermittent with 60% of the feces samples being positive between day 6 and 20 p.i. at the end of experimentation (see Table 2 ). The control pigs were culture negative throughout the experimentation. During the study, diarrhea was infrequently observed; a change in fecal consistency from normal to loose and watery was seen in 3 challenged pigs and no control pigs. In sum, the 3 pigs had diarrhea for 6 days (as listed in Table 2). The pigs were euthanized between day 16 and 20 p.i. Compared with diarrhea based on infection with the highly pathogenic B hyodysenteriae, the observed diarrhea was insignificant but similar to some of those reported on infections with the pathogenic B pilosicoli. 6 In the present study, the pigs were given a standard commercial feed without supplement of soy proteins, which are known to facilitate clinical swine dysentery. 6 At the end of study, 6 challenged pigs were shedding B murdochii. No other Brachyspira spp, enteropathogenic Escherichia coli, or Salmonella enterica were isolated from any of the pigs.
Experimental and Naturally Acquired Brachyspira murdochii Infection in Pigs: Culture, Histopathology, and Fluorescence In Situ Hybridization (FISH) of B murdochii
a Days indicate diarrhea postinfection; negative sign (–) indicates no diarrhea.
b B murdochii culture in feces: No. of reisolations (total No. of samples). Samples were taken on days 6, 9, 13, 16, 18, and 20 postinfection.
c Histopathology of small and large intestines.
d B murdochii in colon by fluorescence in situ hybridization: +, few to moderate number; ++, high number.
e Lawsonia intracellularis detected focally in ileum.
f Catarrhal colitis with sloughing of surface epithelial cells, elongation of crypts, loss of single crypt enterocytes, goblet cell hyperplasia, and infiltrate of mononuclear cells in lamina propria.
At necropsy, the small intestines of the challenged pigs were normal, whereas the spiral colon was slightly distended by gas and liquid contents. The colonic mucosa appeared normal or with mild reddening and increased thickness of the wall, especially in pig Nos. 7 and 8. The small and large intestines of the control pigs were normal. Tissue samples from ileum, cecum, and colon were fixed in formalin and processed as previously described, including immune staining for Lawsonia intracellularis 7 and porcine circovirus type 2 (PCV2) 7 and FISH 1 with the oligonucleotide probes listed in Table 1.
Microscopically, catarrhal colitis was evident in the colon of 2 challenged pigs (Nos. 7 and 8) and was characterized by moderate elongation of crypts with hyperplasia of enterocytes and mucous metaplasia and infiltration of the lamina propria by lymphocytes, small mononuclear cells, and macrophages (Fig. 2). Moreover, focal erosions with sloughing of epithelial cells were observed with single necrotic enterocytes in the crypts and in the extrusion zones between the crypts. The small intestines were normal. With the exception of pig No. 3, which had focal proliferative ileitis caused by L intracellularis, the other challenged pigs, as well as the control pigs, revealed no or only minimal histologic intestinal lesions (Fig. 3). Furthermore, the lengths of 10 randomly selected crypts from each colon were measured, revealing a mean mucosal thickness of 630 ±32 µm and 526 ±27 µm for pigs Nos. 7 and 8, respectively, compared with 425 ±38 µm for the other pigs. The mean mucosal thickness of the field case was 594 ±25 µm. In addition, all pigs were negative for PCV2.
In situ hybridization with the Brachyspira-specific probe and the B murdochii–specific probe revealed identical results: low to extensive colonization by B murdochii of the spiral colon in 5 challenged pigs (negative results for the control pigs, as well as pig Nos. 1, 2, and 6). Pigs Nos. 1 and 2 were additionally culture negative for B murdochii at necropsy.
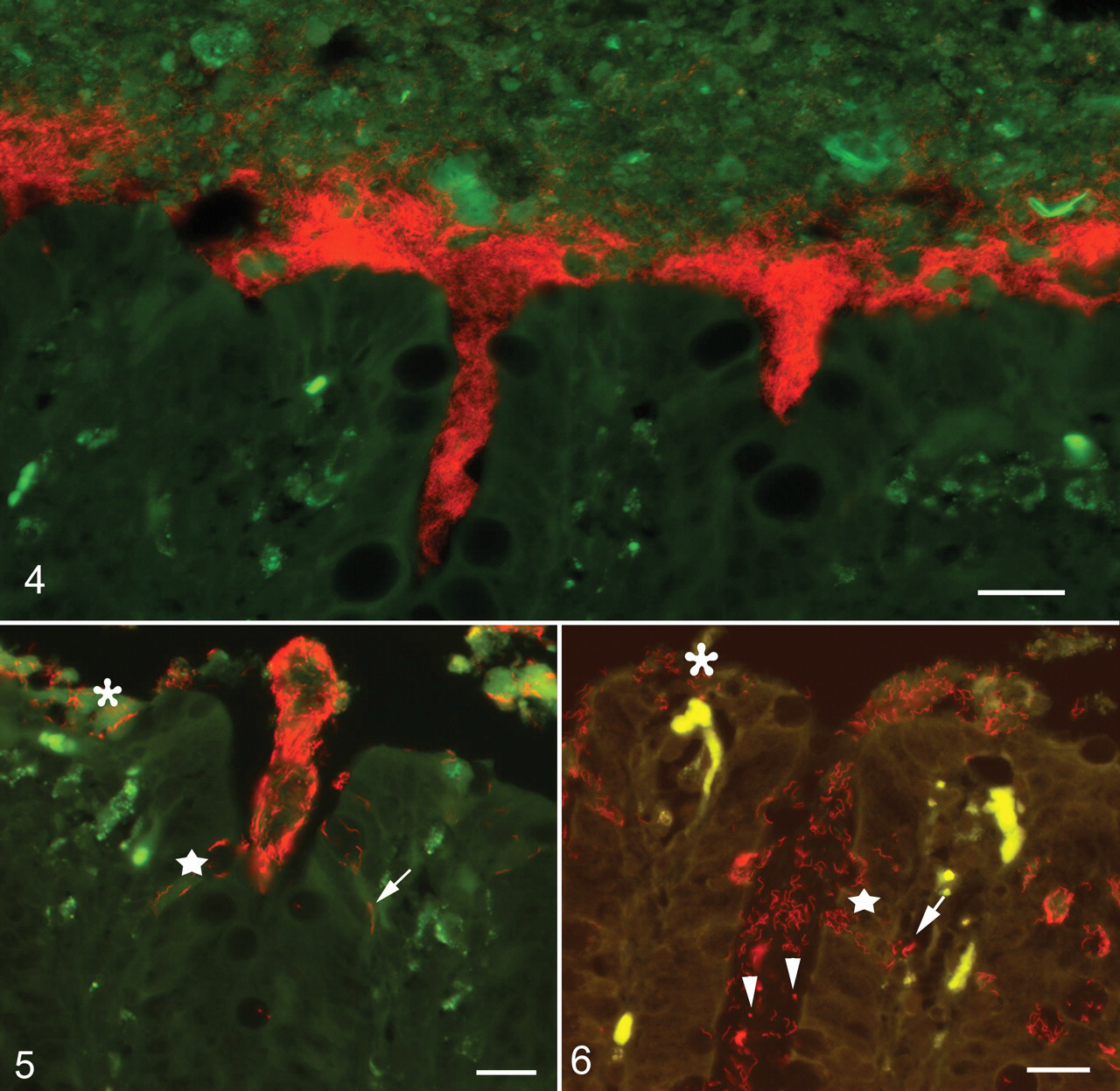
B murdochii were demonstrated in high numbers and closely associated with the surface epithelium, only in the 2 pigs with catarrhal colitis (Fig. 4 ), as well as invading the epithelium in the extrusion zone between crypts, and in some foci invading the surface and crypt epithelium (Fig. 5). This is in accordance with the observations in the naturally acquired B murdochii infection, which was confirmed by FISH with the species-specific probe (Fig. 6). Compared with swine dysentery and intestinal spirochetosis, the histopathological changes associated with B murdochii infection were much less severe, especially in respect to surface and crypt lesions. 5,6 On the contrary, the increased thickness of the mucosa (crypt elongation) and the goblet cell hyperplasia were comparable to that seen in intestinal spirochetosis. 6
The fact that 3 pigs, low to moderately colonized by B murdochii, only showed minimal lesions compared with more obvious lesions of colitis in the severely colonized pigs suggests that propagation of the organism to high numbers (including epithelial invasion) is essential for inducing mucosal lesions. Similarly, high numbers of spirochetes were found in silver-stained sections in 5 field cases of colitis in which B murdochii was detected by PCR in an Austrian study. 15 Concomitant infection with B pilosicoli, PCV2, or B innocens was, however, detected in 3 of the 5 cases, whereas the last 2 cases also revealed polyserositis/peritonitis. 15 Under Danish conditions—in which B murdochii–associated colitis was revealed in 2 to 4% of the routine FISH examinations for Brachyspira—the bacterium was in some cases found as a concomitant infection with L intracellularis (unpublished result). The concomitant infection with L intracellularis in this study (pig No. 3) did not facilitate the propagation of B murdochii. The importance of B murdochii as a cause of colitis on a herd level warrants further studies.
In conclusion, by applying FISH with a species-specific oligonucleotide probe targeting 23S rRNA, this study demonstrated naturally acquired B murdochii–associated colitis in a pig without concomitant bacterial infections. In an attempt to fulfill Koch postulates, we found that B murdochii is able to colonize the large intestines of healthy pigs under experimental conditions. Colitis was found in only the 2 pigs with high numbers of spirochetes closely associated with the mucosa, suggesting that B murdochii is of low pathogenicity under these circumstances. The diagnostic importance of culturing the bacterium from feces only is uncertain, however, because the method is not quantitative. Thus, development of quantitative methods such as real-time PCR will be useful in future studies.
Footnotes
Acknowledgment
We wish to thank the staff at the National Veterinary Institute for practical and technical assistance.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
