Abstract
Mammary intraepithelial lesions (IELs) are noninvasive epithelial proliferations that include ductal hyperplasia (DH), atypical DH (ADH), and ductal carcinoma in situ (DCIS). In women, IELs are associated with increased risk of invasive breast cancer and form a basis for therapeutic decisions. Similarly, in female dogs, IELs are common in tumor-bearing glands and in non-tumor-bearing glands. To determine the prevalence and types of spontaneous IELs, mammary glands from 108 female dogs without clinical mammary disease were evaluated histologically and immunohistochemically. Within this population, 56 dogs (52%) had at least one type of spontaneous IEL, including DH (49 dogs), ADH (14 dogs), low-grade DCIS (19 dogs), intermediate-grade DCIS (12 dogs), and high-grade DCIS (1 dog). Twenty-one dogs had two or more different IEL types. In 23 of 24 dogs with atypical IELs (ADH or DCIS), immunohistochemical expression was determined for estrogen receptor α (ER-α), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER-2/neu), and Ki-67. For all markers examined, low-grade DCIS had significantly lower scores than did adjacent nonlesional gland; PR expression was significantly decreased in low-grade DCIS compared to other atypical lesions. Sixty-one lesions were ER-α negative (12 ADH, 36 low-grade DCIS, 13 intermediate-grade DCIS), and no lesions overexpressed HER-2/neu. Based on the dog’s prevalence of spontaneous mammary IELs that precede clinical mammary disease, the remarkable histologic similarity between canine and human IELs, and the loss of ER expression in certain IELs in both species, the dog shows promise as a model for human breast preneoplasia.
Cancer is the second-leading cause of death in people in the United States, and breast cancer is the most common malignancy in women, estimated to account for 27% of expected new cancer cases in 2009. 17 Mammary intraepithelial lesions (IELs) are noninvasive proliferative lesions of the breast, such as ductal hyperplasia (DH), atypical DH (ADH), and ductal carcinoma in situ (DCIS). Whereas DH is associated with only a slight increase in relative risk for developing invasive carcinoma, 1,7,33 IELs with atypia, such as ADH or DCIS, are treated as high risk for subsequent cancer development. 14,19 Mammary IELs are detected with increasing frequency through routine screening and mammography. Because these lesions may precede invasive carcinoma, IELs provide the opportunity to intercept the progression of breast cancer through early detection and targeted therapy.
In human breast cancer, prognostic markers, such as hormone receptors (estrogen receptor [ER] and progesterone receptor [PR]) or human epidermal growth factor receptor 2 proto-oncogene (HER-2/neu), are used to predict the therapeutic response to hormone (tamoxifen) or monoclonal antibody (trastuzumab) treatments, respectively. 25,31 These drugs are cornerstones of breast cancer therapy in women but are often ineffectual in tumors negative for these markers.
An animal model of spontaneous mammary IELs would facilitate comparative research on preinvasive breast lesions. Established mouse models fail to meet some of the criteria for modeling human premalignant breast disease. 22 In addition, lesions in the mouse mammary gland are not spontaneous as in human disease but rather are induced by a variety of methods, including mammary tumor viruses, chemical carcinogens, radiation, hormonal stimulation, and genetic manipulation. 22 Furthermore, an animal model of ER-negative lesions is lacking, even though ER is expressed in only 25% of high-grade DCIS in women and is absent in about 30% of all breast cancers. 8,27
As in women, mammary neoplasia is the most common spontaneous malignancy in female dogs. 24 In a retrospective study, our laboratory examined canine mastectomy specimens and found a high prevalence of spontaneous IELs adjacent to tumors, as well as remarkable morphologic similarities between canine and human mammary IELs. 3 We also demonstrated a decrease in ER expression in IELs with atypia (ADH and DCIS). 3 Based on these findings, further assessment of the dog as an animal model of IELs was pursued.
The objectives of the current study were (1) to determine prevalence of IELs in adult female dogs without a clinical history of mammary disease and (2) to compare expression of ER, PR, HER-2/neu, and the proliferation marker Ki-67 among different grades of IELs and between IELs and nonlesional mammary gland.
Materials and Methods
Animals
This cross-sectional postmortem study included 108 adult female dogs with no clinical history of mammary disease; in particular, the dogs had no palpable mammary masses. The animals were either retired hounds purchased through a breeding facility or were female dogs used in terminal research projects unrelated to mammary gland disease at Purdue University. Breeds included 88 hounds (mostly coonhounds and foxhounds), 2 beagles, 2 mixed-breed dogs, and 1 Labrador retriever; 15 dogs had no breed designation. Seventy-eight dogs were confirmed to be sexually intact by histological examination of ovaries; the remaining dogs had been ovariohysterectomized, or the sexual functional status was not documented. The dogs ranged from 1.5 to 13.0 years old (median, 8 years), with more than 70% of dogs at least 6 years old. Acquisition, handling, and euthanasia of the dogs were in accordance with the Purdue University Animal Care and Use Committee guidelines.
Morphology
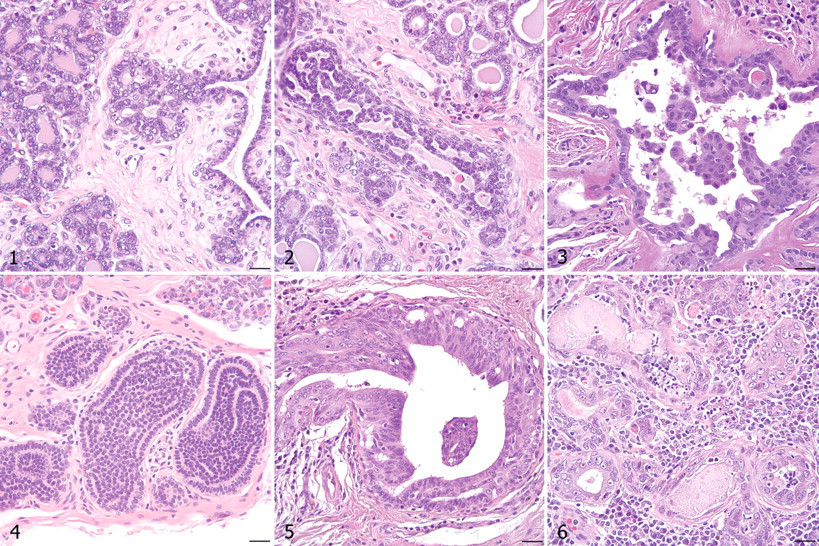
All mammary glands from each dog were collected immediately following euthanasia and were immersion fixed in neutral-buffered formalin. Gross specimens were serially sectioned from the lateral to the medial aspect of each gland; labeled according to dog, gland, and section number to map potential lesions to specific glands; and subsequently paraffin embedded. Each dog had an average of 38 mammary tissue blocks (range, 7–140 blocks), and a total of 979 mammary glands were examined histologically (average, 9 glands per dog). Formalin-fixed, paraffin-embedded 5- to 6-µm sections were routinely processed, stained with hematoxylin and eosin, and evaluated for the presence of IELs. Lesions were categorized as described by Antuofermo et al 3 in consultation with an MD pathologist (S.S.B) according to criteria for IELs of the human breast 33,36 with a focus on DH, ADH, and low-, intermediate-, or high-grade DCIS. Features applicable to DH included dual proliferation of epithelial and myoepithelial cells, indistinct cell borders, nuclear streaming, absence of atypia, lack of polarity, and irregular fenestrations (when present). Lesions categorized as ADH had increased cellular atypia characteristic of DCIS but with dual proliferation of epithelial and myoepithelial cells, lack of polarity, and irregular fenestrations. Also classified as ADH were lesions with cytologic and architectural features consistent with intermediate-grade DCIS but confined to only one duct cross-section. Lesions with mild architectural disarray, cytologic atypia, and/or proliferation were not included in the IEL category if they were associated with significant inflammation. Noninvasive lesions consisting solely of proliferating epithelial cells with cytologic features of malignancy were classified as DCIS. DCIS typically arose in interlobular or intralobular ducts but often spread throughout an entire mammary lobule. Lesions of DCIS were further categorized as low, intermediate, or high grade to reflect an increase in cellular atypia and a decrease in polarity with increasing grade. Table 1 lists the cytologic and architectural characteristics of low- versus high-grade DCIS. 36 Intermediate-grade DCIS incorporated those lesions with a nuclear grade between low- and high-grade DCIS. Figures 1 through 6 depict canine mammary glands without IELs and with DH, ADH, and grades of DCIS. Any benign mammary tumors identified histologically were classified according to the Armed Forces Institute of Pathology and World Health Organization criteria for canine mammary neoplasms. 24
Features of Low- and High-Grade Ductal Carcinoma In Situ a

a Summarized from the Consensus Conference on the Classification of Ductal Carcinoma In Situ. 36
Immunohistochemistry
Formalin-fixed, paraffin-embedded mammary tissue sections (collected and processed as described above) from 23 dogs with atypical IELs were serially sectioned at 5 µm and mounted on charged slides for immunohistochemistry (IHC). Sections were dried overnight in an oven at 50°C. After deparaffinizing and rehydrating tissues, heat-induced epitope retrieval was performed in citrate buffer, pH 6.0 (S2369; Dako Corp, Carpinteria, CA), using a food steamer for 20 minutes at 90–95°C. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide followed by application of a nonspecific protein binding block (X0909; Dako Corp). Sections were then incubated at room temperature with one of four primary antibodies as designated in Table 2 . Simultaneously, serial sections used as negative control tissues were incubated with either universal negative control mouse immunoglobulin (N1698; Dako Corp) or rabbit immunoglobulin (N1699; Dako Corp). The ER-α antibody used in our previous study 3 had been discontinued, requiring the validation and use of a different ER-α clone in the present study. In addition, a rabbit monoclonal PR antibody was selected over the previously used mouse monoclonal antibody in an attempt to achieve the highest specific immunolabeling. 41 Positive control tissues were canine uterus (ER and PR), a canine mammary carcinoma known to express HER-2/neu, and canine lymphoma (Ki-67). Immunoreactivity was detected using the labeled streptavidin–biotin–peroxidase method (K0675; Dako Corp) followed by visualization with 3,3′-diaminobenzidine (K3466; Dako Corp). Tissues were counterstained with hematoxylin (MHS32; Sigma-Aldrich, St. Louis, MO). For ER, PR, and Ki-67, the estimated percentage of cells labeled and the labeling intensity of positive cells were recorded. Intensity was graded on the following scale: 0 = no reactivity, 1 = weak specific labeling, 2 = moderate, and 3 = strong. Semiquantitative IHC scores were calculated by multiplying the estimated percentage of cells labeled by the intensity of labeling. For HER-2/neu analysis, the staining was titrated such that normal mammary lobules showed moderate, complete membranous labeling. In this system, strong complete membranous labeling would be interpreted as overexpression according to American Society of Clinical Oncology / College of American Pathologists guideline recommendations for HER-2 testing in breast cancer. 43 Lesions were scored by two authors (P.M., M.A.M.). ER expression was assessed in 60 mammary glands and included 19 ADH, 38 low-grade DCIS, and 16 intermediate-grade DCIS lesions. Expression of PR was evaluated in 57 glands and included 15 ADH, 34 low-grade DCIS, and 17 intermediate-grade DCIS lesions. Immunoreactivity of HER-2/neu was reviewed in 53 glands and included 19 ADH, 34 low-grade DCIS, and 12 intermediate-grade DCIS lesions. Expression of Ki-67 was assessed in 53 glands and included 12 ADH, 34 low-grade DCIS, and 11 intermediate-grade DCIS lesions. DH, other benign lesions, and tumors were not evaluated immunohistochemically for expression of ER, PR, HER-2/neu, and Ki-67.
Antibodies Used in Immunohistochemistry a
a ER-α, estrogen receptor α; PR, progesterone receptor; HER-2/neu, human epidermal growth factor receptor 2.
Statistical Analysis
Morphologic lesions were expressed with descriptive statistics. Immunohistochemical data were analyzed with nonparametric methods on semiquantitative IHC scores using SPSS. The Kruskal–Wallis test was used to compare groups (ADH, low-grade DCIS, and intermediate-grade DCIS) for each antibody. The Mann–Whitney test was applied for post hoc comparison in the case of significant differences. Pairwise comparison between IHC scores in atypical IELs and adjacent nonlesional mammary gland was performed using the Wilcoxon signed–rank test. With all statistics, results were considered significant when P ≤ .05 except in post hoc analysis, in which the α level was adjusted downward (Bonferroni correction).
Results
Morphology
Spontaneous mammary IELs identified within the sample population included DH, ADH, and low-, intermediate-, and high-grade DCIS (Figs. 2–6). Table 3 lists the number of dogs with each type of mammary IEL, as well as the dogs in which benign tumors were discovered histologically, for a total of 58 dogs with proliferative mammary lesions. Fifty-six dogs (51.9%) had at least one type of IEL; 21 dogs (19.4%) had two or more different lesion types simultaneously. IELs with atypia (ADH or DCIS) were identified in 24 dogs (22.2%). Of the 11 dogs with nonpalpable benign neoplasms detected histologically, 9 (81.8%) also had IELs. Of the 56 dogs with IELs, 49 (87.5%) were confirmed to be sexually intact by histological examination of ovaries. Of the 24 dogs with atypical IELs, 18 were at least 8 years old; age was not specified in the remaining 6 dogs with atypical lesions. Only 2 of 13 dogs younger than 8 years old had IELs, and both were categorized as DH. DH and ADH frequently formed micropapillary projections (Figs. 2, 3), whereas low- and intermediate-grade DCIS were solid or formed cribriform fenestrations (Figs. 4, 5). The high-grade DCIS lesion was predominantly solid with areas of necrosis and lymphoplasmacytic inflammation (Fig. 6). Mammary IELs ranged from focal, affecting as few as one duct cross-section, to multifocal, incorporating multiple ducts in a single tissue section, in multiple sections, or in multiple glands. Although IELs were identified in all glands of the mammary chain, the prevalence of lesions in the cranial thoracic glands (gland 1) was 25 to 50% of the prevalence in any other gland; higher-grade lesions, such as intermediate- or high-grade DCIS, were not identified in cranial thoracic glands. Table 4 outlines the distribution of 209 individual IELs among the mammary glands.
Number of 108 Female Dogs With Proliferative Mammary Lesions a
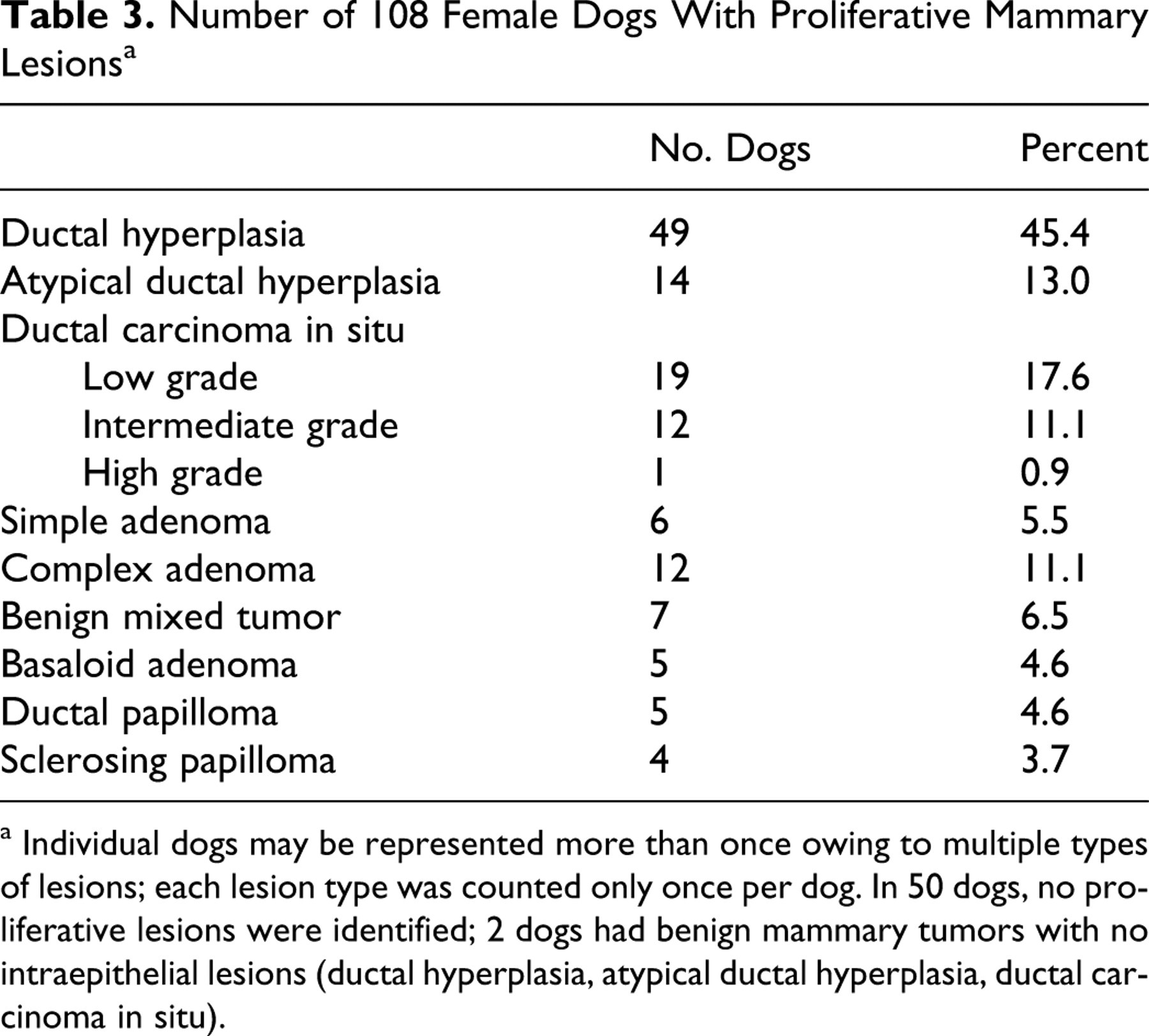
a Individual dogs may be represented more than once owing to multiple types of lesions; each lesion type was counted only once per dog. In 50 dogs, no proliferative lesions were identified; 2 dogs had benign mammary tumors with no intraepithelial lesions (ductal hyperplasia, atypical ductal hyperplasia, ductal carcinoma in situ).
Distribution of Canine Mammary Intraepithelial Lesions by Gland a
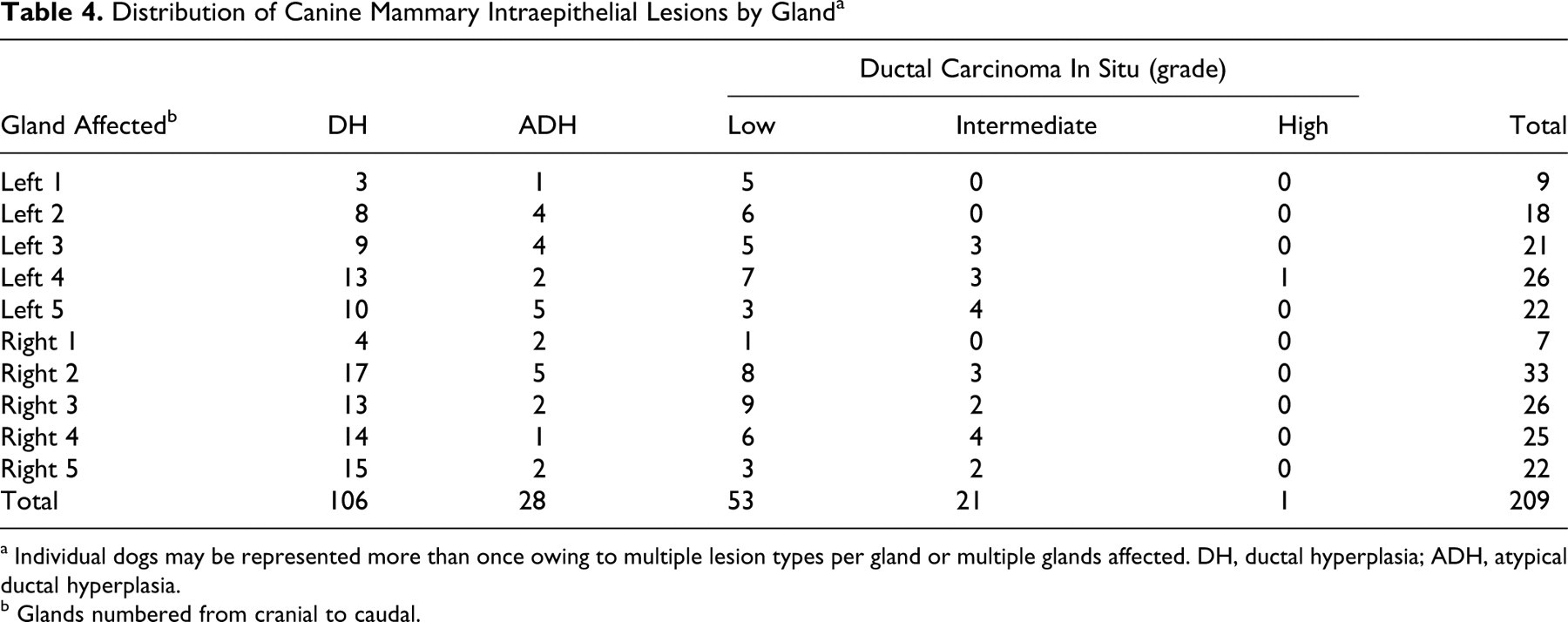
a Individual dogs may be represented more than once owing to multiple lesion types per gland or multiple glands affected. DH, ductal hyperplasia; ADH, atypical ductal hyperplasia.
b Glands numbered from cranial to caudal.
Immunohistochemistry
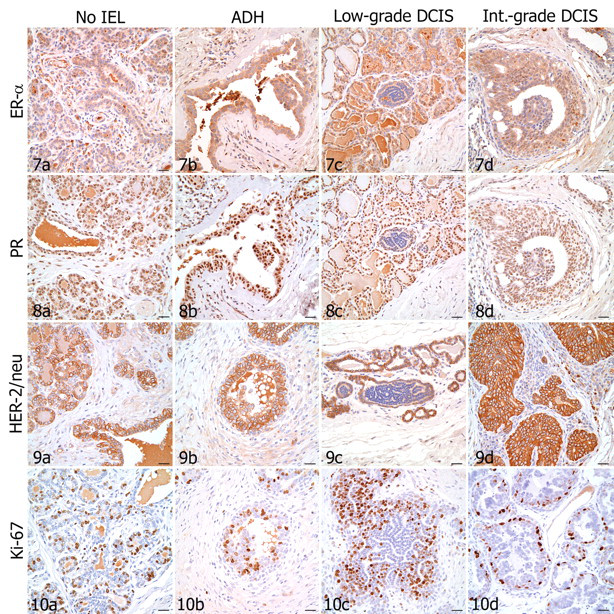
Table 5 lists the median and range of percentage of cells positively immunolabeled and the intensity of immunoreactivity for ER, PR, and Ki-67 in IELs and in adjacent nonlesional mammary gland. None of the IELs showed HER-2/neu overexpression. Low-grade DCIS had a notable paucity of immunoreactivity for all antibodies tested (Figs. 7C , 8C, 9C, 10C). The single case of high-grade DCIS was quantitatively insufficient for IHC analysis. Also, insufficient lesional tissue or inadequate fixation of some other sections precluded IHC scoring.
Immunohistochemistry of Atypical Mammary Intraepithelial Lesions in Dogs a
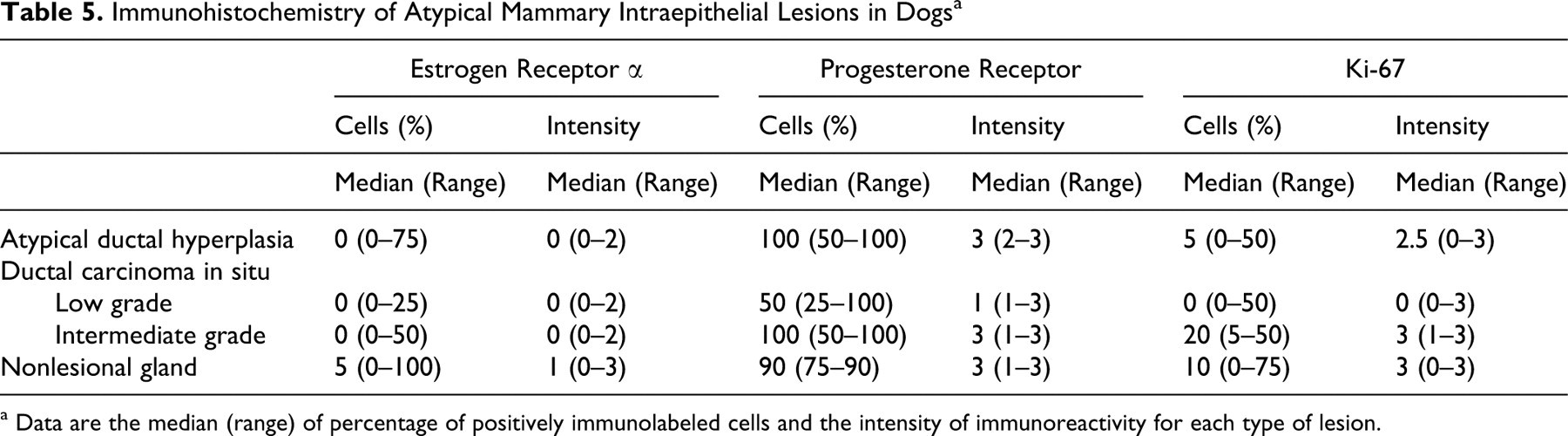
a Data are the median (range) of percentage of positively immunolabeled cells and the intensity of immunoreactivity for each type of lesion.
ER
In nonlesional mammary gland adjacent to IELs, 0 to 75% of cells (rarely up to 100% of cells) expressed ER-α, with intensity ranging from no reactivity to moderate (Fig. 7). Atypical DH had significantly lower ER-α expression compared with that of adjacent nonlesional gland (P = .023). Low-grade DCIS had significantly lower ER-α expression than that of adjacent nonlesional gland (P < .001) and ADH (P = .001). Low-grade DCIS also had lower ER-α expression than did intermediate-grade DCIS, but these results were not statistically significant (P = .036, not significant after Bonferroni correction). ER expression in intermediate-grade DCIS did not significantly differ from that in adjacent nonlesional gland. Of 73 atypical IELs, 61 (83.6%)—including 12 of 19 ADH, 36 of 38 low-grade DCIS, and 13 of 16 intermediate-grade DCIS—were ER-α negative (0% of cells immunolabeled). Seven of 19 ADH, 2 of 38 low-grade DCIS, and 3 of 16 intermediate-grade DCIS had ER-α immunoreactivity; these 12 ER-α positive IELs were from 4 of the 23 dogs tested.
PR
Nonlesional mammary gland typically had moderate to strong nuclear expression of PR in 75 to 90% of cells (Fig. 8). In low-grade DCIS, 25 to 100% of cells expressed PR; however, the intensity of immunoreactivity was typically weak to moderate. Low-grade DCIS had significantly lower PR expression when compared with that of adjacent nonlesional gland (P < .001), ADH (P < .001), and intermediate-grade DCIS (P < .001). Atypical DH and intermediate-grade DCIS had moderate to strong PR expression in 50 to 100% of cells. PR expression in ADH and intermediate-grade DCIS did not significantly differ between the two lesions or from that in adjacent nonlesional gland.
Interrelationship between ER and PR
Sixty-one atypical IELs had sufficient tissue for ER-α and PR IHC in serial sections. Of the 61 lesions, 14 had strong, diffuse PR expression (100% of cells immunolabeled, intensity = 3). Of these 14 IELs, 8 were ER-α negative (ER–/PR+) and 6 were ER-α positive (ER+/PR+); only 1 IEL with strong, diffuse PR expression was low-grade DCIS. In contrast, 20 of the 61 atypical IELs had weak PR expression (intensity = 1) in 25 to 100% of cells. Of these 20 IELs with weak PR expression, all were ER-α negative, and 19 of 20 were low-grade DCIS.
HER-2/neu
As expected, nonlesional mammary gland showed moderate, diffuse membranous expression of HER-2/neu (Fig. 9). None of the IELs showed HER-2/neu overexpression. In general, the intensity of HER-2/neu reactivity in all types of IELs tested was significantly lower than that in adjacent nonlesional gland, including ADH (P = .001), low-grade DCIS (P < .001), and intermediate-grade DCIS (P = .010). Of 65 atypical IELs, 6 (9.2%) were completely negative for HER-2/neu, including 2 of 19 ADH and 4 of 34 low-grade DCIS. Interestingly, 5 of these 6 IELs were also ER-α negative.
Ki-67
The intensity of nuclear expression of Ki-67 (Fig. 10) was typically moderate to strong in nonlesional mammary gland, although the percentage of scattered immunoreactive cells varied greatly among sections (from 0 to 75%). In low-grade DCIS, Ki-67 immunoreactivity typically occurred in 5% of cells or less. Low-grade DCIS had significantly lower Ki-67 expression compared with that of adjacent nonlesional gland (P < .001) and intermediate-grade DCIS (P < .001) and, although not statistically significant, lower expression compared with that of ADH (P = .043, not significant after Bonferroni correction). In addition, intermediate-grade DCIS had increased Ki-67 expression compared with that of ADH, but these results were also not statistically significant (P = .084). Expression of Ki-67 in ADH and intermediate-grade DCIS did not differ significantly from that in adjacent nonlesional gland.
Discussion
In this study, spontaneous IELs were detected in over half the female dogs without clinical mammary disease. Thus, approximately 1 in every 2 dogs examined had at least one IEL, and approximately 1 in every 5 dogs had more than one type of IEL. Histologically, the lesions were similar to human IELs and included DH, ADH, and grades of DCIS, which provided evidence that adult female dogs develop spontaneous mammary IELs with high prevalence, even in the absence of tumors. Because higher-grade IELs have been detected adjacent to mammary carcinomas in dogs, 3 the present report—wherein the sample population was selected to be clinically free of mammary tumors—provides additional support that IELs arise first and may act as a precursor to the development of invasive carcinoma. Unfortunately, because this was a postmortem study, follow-up was not possible to determine the risk of subsequent mammary cancer development. Although this study focused on detection of IELs, several benign tumors were identified histologically, including simple adenoma, complex adenoma, benign mixed tumor, basaloid adenoma, ductal papilloma, and sclerosing papilloma. Adenosis, sclerosing adenosis, and lobular hyperplasia were reported in our previous study 3 but were not evaluated in the present study, because the examined mammary glands were in varying stages of physiologic hyperplasia or atrophy (ie, pseudolactation, lactation, or involution).
The current findings are supported by previous studies in which spontaneous preinvasive epithelial lesions were described in the canine mammary gland and, in some reports, compared to similar lesions in humans. In a postmortem survey of Beagles in 1976, Warner identified 2,142 mammary lesions, termed dysplasias, in 39 female dogs younger than 4 years old. 42 Unfortunately, histological features of these lesions were not further described to draw a comparison to the present study, but the high prevalence of lesions and the young age of the dogs stimulates continued interest in proliferative canine mammary gland lesions. A retrospective study in 1983 assessed biological behavior of mammary tumors—including hyperplasia, atypical hyperplasia, and carcinoma in situ—in mastectomized dogs with a minimum follow-up of 2 years or until tumor recurrence. Dogs with carcinoma in situ or with lesions designated as atypical had a significantly higher risk of developing invasive carcinoma than did dogs with lesions lacking atypia. 13 This finding mirrors the increased risk in women with similar IELs (ADH and DCIS). 14,19 The authors also reported coexistence of preinvasive lesions and invasive carcinoma, suggesting a progression in the development of canine mammary cancer, 13 similar to the proposed carcinogenesis in women. 4,13 In 1999, Benjamin et al reported hyperplastic and neoplastic mammary lesions in adult Beagles exposed to prenatal or early postnatal radiation. The authors emphasized the continuum between the lesions defined as hyperplasia and neoplasia and expressed difficulty in distinguishing them in several specimens. 6 In 2007, our laboratory reported a retrospective study of canine mammary IELs adjacent to tumors in mastectomy specimens. The results demonstrated a high prevalence of IELs and found that malignant tumors were significantly associated with lesions of higher grade (ADH and DCIS), once again insinuating a continuum from preinvasive epithelial proliferation to invasive carcinoma. 3
Medical pathologists attempted to stratify mammary gland IELs to emphasize this continuum and thus help determine prognosis. 28,36,39 Although human studies have continued to determine risks and establish the relationships between ADH and invasive carcinoma 9 and between DCIS and invasive carcinoma, 10,35 the distinction between atypical IELs—particularly ADH and DCIS—unfortunately remains a challenge, which leads to controversy regarding diagnostic concordance, biological significance, prognostic implication, and, therefore, the importance of maintaining these IEL categories. 5,12,32,38,40 Similarly, lesions resembling low-grade DCIS in humans have been classified as DH, or epitheliosis, in the veterinary literature. 24 An animal model of IELs is increasingly necessary to advance the understanding of carcinogenesis, methods of diagnosis, and treatment strategies.
In this study, low-grade DCIS had significantly lower immunohistochemical expression of all tested molecular biomarkers than did adjacent nonlesional mammary gland. Low-grade DCIS also had significantly lower expression of PR compared with that of the other atypical IELs. HER-2/neu overexpression was not seen in any lesion. In sum, 61 atypical IELs (83.6%) were ER-α negative, whereas 12 (16.4%) were ER-α positive.
Human invasive breast carcinomas do not express ER-α in approximately 30% of cases 27 ; most women whose tumors do not express this biomarker do not respond to endocrine therapy. 25,31 This creates two important research challenges. The first is to develop an animal model for ER-negative mammary carcinoma. The second is to evaluate preinvasive lesions to determine which might progress to ER-negative mammary carcinoma. Female dogs, like women, develop a spectrum of ER-positive and ER-negative mammary lesions. Most studies assessing ER-α immunoreactivity in canine mammary carcinoma demonstrated either significantly decreased expression 23,26,44 compared with that of benign mammary tumors or a lack of ER-α expression (between 30 and 100% of tumors were negative), 16,20,44 with one contradictory report. 18 Recently, Gama et al subtyped canine mammary carcinomas using an IHC panel based on a new human classification; almost 30% of the tumors tested by the authors were negative for ER and HER-2/neu—a phenotype associated with decreased survival rates. 11 Based on these findings and the fact that an estimated 25% of DCIS in humans lack ER-α expression, 1 the dog may have a role as a model for ER-negative breast cancer.
The expression of PR predicts the therapeutic response of human invasive mammary carcinoma to hormone therapy. 25,31 Immunoreactivity for PR was shown to decrease with increased IEL grade in humans. 2,37 In the present study, low-grade DCIS had significantly decreased PR immunoreactivity. However, this finding was not observed in the study by Millanta et al, 23 in which, incidentally, significantly lower PR expression was observed in malignant tumors. Although no lesions in the present study were completely PR negative, a paucity of PR expression was associated with ER negativity. Most of the lesions with low PR immunoreactivity and ER negativity were low-grade DCIS.
Overexpression of HER-2/neu occurs in 20 to 30% of invasive mammary carcinomas in humans, 1 and it is used to predict the therapeutic response to tamoxifen and trastuzumab. 25,31 Increased HER-2/neu expression occurred with increased IEL grade in humans. 1,2,37 In dogs, 17.6 to 19.1% of malignant mammary tumors had increased HER-2/neu immunoreactivity, 21,34 and IELs adjacent to tumors had similar overexpression, 3 consistent with a continuum. In the present study, significant HER-2/neu overexpression was not detected in IELs.
High expression of the proliferation marker Ki-67 is an indicator of poor prognosis in human invasive breast cancer. 29,31 Increased expression of Ki-67 was shown in dogs with malignant mammary tumors 30,44 as well as high-grade IELs. 3 In the present study, IELs with the greatest atypia (intermediate-grade DCIS) had increased Ki-67 expression compared with that of low-grade DCIS.
Immunohistochemistry results differed in a few regards between the present study and our retrospective study. 3 ER expression in nonlesional mammary gland of the sample population in the present report was lower than that reported by our laboratory. 3 This may be due to the use of a different ER-α antibody in this study or the different ovariohysterectomy status of dogs between the two studies or because the current population did not have palpable mammary tumors. In particular, the tumors in the previous study may have caused a field effect in which intensity of ER expression was increased in nonlesional gland adjacent to the tumor. Differences in PR expression between the two studies may reflect the use of a new antibody. The rabbit monoclonal PR antibody was selected over the previously used mouse monoclonal antibody following a study demonstrating specific immunolabeling in canine tissues. 41 However, the potential for false-positive immunoreactivity using the rabbit monoclonal antibody has been documented in human tissues. 15 The specific immunogen used to create this antibody was not available for purchase; therefore, a peptide competition study was not performed in canine tissues. The relative lack of HER-2/neu overexpression in the present study, when compared with that of our previous study, likely reflects the paucity of high-grade lesions. In our retrospective study, high-grade DCIS was the most common IEL detected adjacent to mammary tumors. 3 Although no lesions in the present study overexpressed HER-2/neu, the single high-grade DCIS was unavailable for IHC.
In summary, female dogs have a high prevalence of spontaneous IELs that are morphologically similar to human lesions. 33,36 These lesions may be ER positive or negative. Overexpression of HER-2/neu was not observed in this series; however, no lesions of high-grade DCIS were available for evaluation. The various histologic phenotypes and biomarker expression profiles of canine mammary IELs mirror those in women. These features provide support for the dog as an animal model to study carcinogenesis, diagnostic strategies, and treatment modalities for preinvasive lesions of the mammary gland. Future directions include molecular characterization of spontaneous canine mammary IELs.
Footnotes
Acknowledgements
We thank Dr José Ramos-Vara for technical advice on immunohistochemistry, the Purdue Animal Disease Diagnostic Laboratory for histology services, Dee Dusold for preparing sections for immunohistochemistry, and Brenda Johnson for assistance in tissue collection.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
This study was supported by a grant from the Department of Defense (W81XWX-04-1-0196).
