Abstract
Influenza A virus infection occurs in many species. Wild waterfowl harbor the widest variety of influenza A viruses and serve as a constant reservoir for the emergence of new viruses. Highly pathogenic avian influenza, or “fowl plague,” has been a known poultry disease for more than 130 years. It continues to emerge and reemerge, but global changes in trade and poultry production have expanded the impact and geographic range of these outbreaks. One subtype of highly pathogenic avian influenza, H5N1, has infected poultry on several continents as well as many people, leading to a human disease that is markedly different from seasonal influenza and that is associated with high mortality.
The only thing harder than preparing for a pandemic is explaining why you didn’t.—Ambassador Robert Loftis, Special Representative for Avian and Pandemic Influenza, US Department of State
Influenza in People and Animals
Influenza viruses belong to the Orthomyxoviridae family and are divided into 3 types, influenza A, B, and C, according to the antigenicity of their nucleoprotein (NP) and matrix protein. Influenza A viruses are classified into 16 HA and 9 NA subtypes according to the antigenicity of 2 spike-shaped surface proteins, hemagglutinin (HA) and neuraminidase (NA). 38 Influenza A viruses have a long history of emerging and reemerging. During the 20th century, there were 3 pandemics among humans, and now a new pandemic is threatening to occur. Many animal species can be infected with influenza A viruses, including horse, pig, dog, cat, chicken, whale, and seal. Therefore, influenza A viruses can significantly affect both animal and human health.
Highly Pathogenic Avian Influenza Viruses
For more than 130 years, highly pathogenic avian influenza viruses have existed as “fowl plagues,” causing sporadic outbreaks with high morbidity and mortality in poultry. Fowl plague was first reported in northern Italy in 1878. 24 It is believed that outbreaks may have occurred prior to this, but virologic techniques were not available at the time to isolate and identify the causative agent. Swine influenza virus was the first influenza virus isolated, followed by the isolation of human influenza virus in 1933. 28,29 In the 1940s, the HA activity of influenza virus was discovered by G K Hirst, 10 and this activity made it possible to more readily isolate and identify the virus. Fowl plague virus was demonstrated to have HA activity by D Lush 18 and was identified as an influenza A virus in 1955. 26
After the human influenza pandemics of 1957 and 1968, surveillance of the virus in avian species increased in the 1970s. Results showed that wild waterfowl harbor all subtypes of influenza A viruses, 12 and highly pathogenic viruses were predominantly limited to H5 and H7 subtypes. These were also the subtypes responsible for the majority of morbidity and mortality in domestic poultry. 17 Although sporadic outbreaks of highly pathogenic avian influenza viruses have occurred in the world since 1959, outbreaks were geographically limited to certain areas within a country, indicating they were endemic to specific regions.
However, over the last 10 years, the highly pathogenic H5N1 influenza virus has spread worldwide because of a number of factors, many of which are outlined in the first article in this series.25 Specifically, globalization combined with huge increases in intensive poultry production has facilitated spread and dissemination. Since 1970, poultry production has increased by 436%, more than 2.3 times and 7.5 times the growth of swine and beef production, respectively. Poultry is now raised in high-density layer houses to meet the increasing demand for animal protein. However, this method of intensive production facilitates rapid spread of disease through close animal contact. Breeding and genetic improvements have been used to increase meat and egg yield from poultry, potentially leading to loss of genetic diversity. 20 Genetic homogeneity may increase vulnerability to emerging and reemerging diseases because of decreased genetic plasticity compared with ancestral breeds.
The highly pathogenic H5N1 avian influenza strain responsible for the current worldwide outbreak was first isolated from a flock of sick geese in Guangdong, China, and genetically similar isolates have subsequently caused human disease. 39 From 1997 to late 2003, H5N1 virus levels remained low. However, in December 2003, a human case of H5N1 infection was reported in Vietnam, which was followed by other human cases in east and southeast Asia. Since then, the virus has spread across Asia and into Europe, Africa, and the Middle East. 40 Genetic analysis has revealed that the lineage of the 2003–2006 H5N1 epidemic virus can be traced to a virus first isolated in 1997. 15 From 2008 to 2009, human cases of H5N1 virus infection have occurred primarily in China, Vietnam, Indonesia, and Egypt. 1,36
Chickens
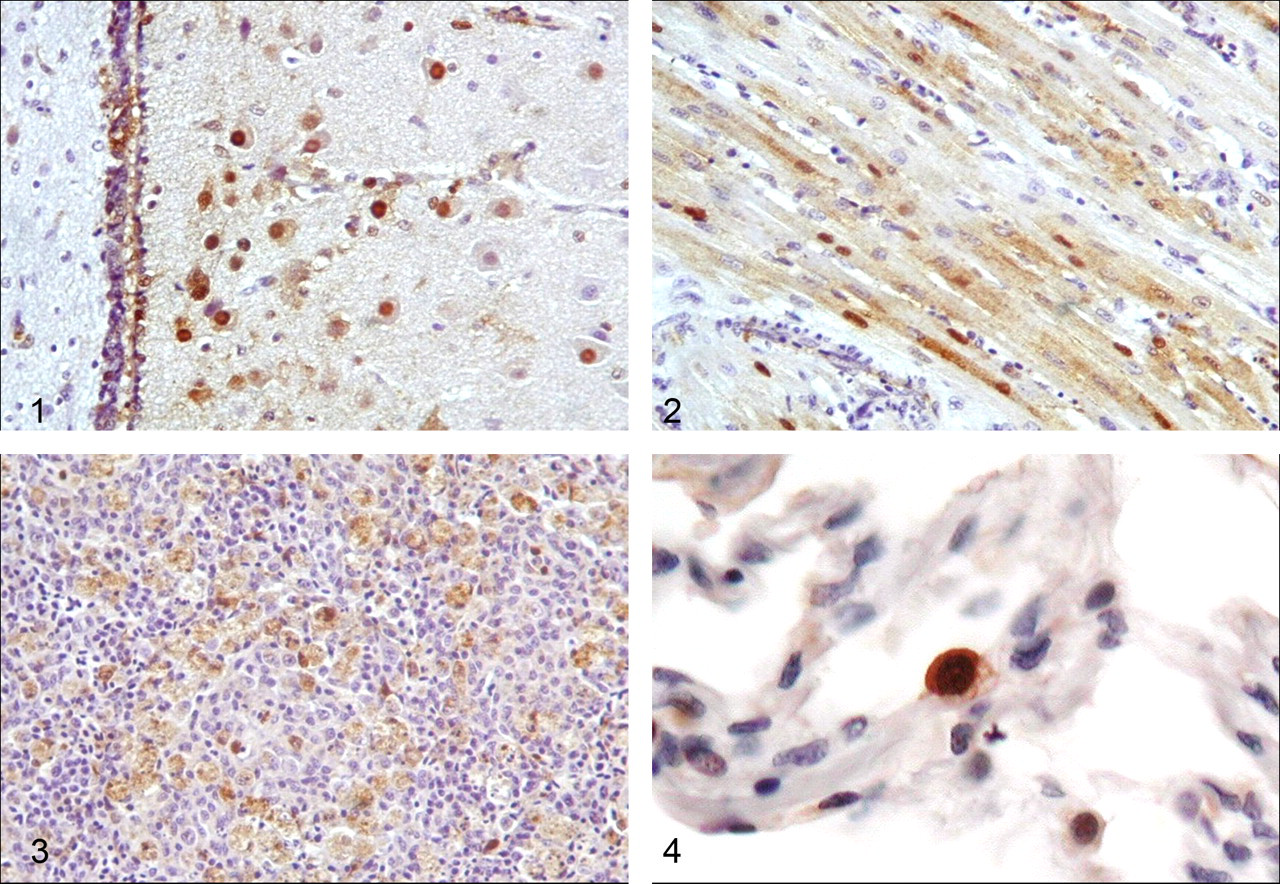
When chickens are exposed to low pathogenic influenza viruses, viral growth is limited to respiratory and intestinal organs, and the infections are not lethal. However, in the case of highly pathogenic viruses, infection results in systemic viral replication and acute or peracute death. In these chickens, immunohistochemical analyses have demonstrated extrapulmonary/intestinal infection. Although the distribution and extent of lesions differ between viral strains, one of the most prominent findings is systemic and abundant viral antigen in the vascular endothelium and perivascular parenchymal cells at the last stage of the disease. 2,13 Moreover, a distinguishing feature of many highly pathogenic virus strains is brain and cardiomyocyte tropism (Figs. 1 and 2). Elucidating the initial target cells for highly pathogenic influenza strains is vital for disease prevention, but such information is limited. Several reports describe viral replication in respiratory tissues (nasal and pulmonary) and splenic involvement in early disease stages (Fig. 3). Massive damage to the immune system may correlate with the systemic viral spreading that occurs thereafter. 27 The transmission efficiency and mortality rate in chickens associated with these highly pathogenic viruses is high.
Ducks
The influenza A virus does not commonly induce disease in ducks. Avian influenza viruses typically establish oral–fecal infection cycles in ducks, leading to asymptomatic viral shedding in feces. A long history of coevolution may have played a major role in modulating influenza A virus pathogenesis in ducks. Generally, fewer viral progeny are produced during asymptomatic infection compared with acute infection, but the duck population maintains a large influenza A virus gene pool. Although all HA and NA subtypes are detected, they are predominantly of low virulence. Highly pathogenic influenza virus is not common in the duck population.
Virologic analyses have shown that duck infection with highly pathogenic influenza A viruses typically results either in the complete absence of viral replication, restricted replication in respiratory and intestinal organs, or mild replication in multiple organs without mortality. 23 These data have been confirmed in experimental infection of ducks with highly pathogenic viruses isolated before 2002. However, H5N1 influenza viruses isolated after 2002 cause systemic and lethal infection in ducks, which is accompanied by signs of severe central nervous system dysfunction. 6,21,31,32,41 Under experimental conditions, H5N1 viruses are found systemically and viral antigen is detected in the brain. Thus, it is likely that recently isolated viruses with worldwide distribution possess different properties compared with the highly pathogenic H5 strains isolated before 2002. Although these pathological findings indicate that viral growth in brain tissue may be a major factor in determining influenza pathogenicity in ducks, we found that even an H5N1 virus isolated before 2002, which is avirulent following respiratory infection, can replicate in duck brain tissue when inoculated intracerebrally (Shinya et al, unpublished). Therefore, viral spread from the initial infection site to the brain may be a key factor in the ability of recent viruses to cause disease in ducks.
Felines
Cats are rarely reported to show seropositivity for human seasonal influenza virus. These results indicate that infection and transmission of seasonal human influenza viruses to cats are generally self-limiting. However, in the case of highly pathogenic H5N1 viruses, Felidae species, including domestic cats and tigers, became victims during outbreaks in domestic poultry. 33 Subsequent experiments have shown that these species are highly susceptible to the recently circulating H5N1 viruses. 14 Systemic infection was confirmed by pathological examination, and viral antigen was found in parenchymal cells, including immune cells, in infected organs. The main viral targets in the lungs were bronchiolar and alveolar epithelium of the lower respiratory tract. These data indicate that cats are susceptible to systemic infection if they are heavily exposed to certain types of H5N1 viruses.
Canines
Serosurvey results have demonstrated that canine species are probably susceptible to human influenza viruses, including influenza B viruses, 34 but the small number of cases indicates that infection is generally self-limiting. A report has described lethal H5N1 viral infection in a dog. 30 The dog was reported to show systemic infection after eating dead ducks during an H5N1 outbreak. Viral antigens were distributed to extrarespiratory tissues and were detected not only in the alveolar cells but also in the hepatocytes, renal tubular epithelium, and glomerulus among examined tissues. The main target of this H5N1 virus in the respiratory organs seems to be the lungs, because histopathologic examination of the dog’s lung revealed severe pulmonary damages characterized by pulmonary edema and interstitial pneumonia. However, recent studies have reported that infection and transmission efficiency of H5N1 viruses in dogs are low, and heavy exposure is required for H5N1 viruses to establish canine infection. 8,19
In contrast, the equine-derived H3N8 virus is efficiently transmitted to dogs. Since 2004, many states in the USA have reported canine influenza cases, although these outbreaks are usually sporadic and self-limiting. Most infected dogs show clinical signs related to upper respiratory tract infection, and this clinical finding was supported by histopathological findings in experimentally infected Beagle dogs that had necrotizing and hyperplastic tracheitis, bronchitis, and bronchiolitis. 4 Although bronchopneumonia and hemorrhagic pneumonia, demonstrating involvement of the lower respiratory tracts, have been occasionally described in naturally infected cases, this condition seems to occur mainly in the dogs affected by secondary bacterial infection. 3-5
Pathology of Influenza A Virus Infection in Humans
We define annual human influenza A viruses as seasonal influenza viruses. The recently circulating seasonal influenza viruses are H1N1 and H3N2 subtypes. These viruses primarily cause upper respiratory infection and are associated with fairly efficient transmission. Serious or lethal cases are rare except in people with underlying disease or those who develop secondary pneumonia. However, the infection profile of highly pathogenic H5N1 avian influenza viruses is distinct. Unlike seasonal influenza viruses, which are easy to detect in nasal and throat swab samples, highly pathogenic avian influenza viruses have been rarely isolated from nasal and throat swab samples following avian to human transmission. The main target of these viruses in people is the lower respiratory tract, and severe diffuse alveolar damage with prominent lung edema is common in human patients. 16,35 Viral antigen was frequently detected in pneumocytes, especially type II pneumocytes (Fig. 4), but in rare cases, systemic infection and associated viral antigen in the parenchyma were reported in people with certain underlying conditions. 9 This systemic antigen distribution associated with H5N1 viruses is notable and may be related to the increased cleavability of highly pathogenic influenza HA, which enables these viruses to replicate in multiple organs. 11 The infection and transmission efficiency of these H5N1 viruses among humans are still very low, and most of those infected have a history of direct contact with sick or dead birds.
Human infection by another highly pathogenic avian influenza A virus, H7N7, has been reported, and in 1 case a victim died from severe pneumonia similar to that caused by H5N1 viruses. 7 The swab sample taken from the victim’s throat was negative for viral antigen during the acute disease stage. The virus was later isolated from postmortem lung samples, which revealed that lower respiratory organs were the main targets of H7N7 viruses. However, this virus displayed an unusual infection and transmission pattern. Although 78 of 89 affected people experienced conjunctivitis with high infection and transmission efficiency, only 7 of 89 showed influenza-like illness with low infection and transmission efficiency.
We are now experiencing the first wave of worldwide infection by the newest pandemic virus, H1N1.37 Genetic analysis of this virus indicates that it originated from reassortment of several swine-derived viruses, although the original virus has not been detected in swine populations. Currently, this virus primarily causes similar symptoms to those observed in humans infected by seasonal influenza viruses, and half of the serious cases are associated with an underlying disease. However, the remaining half of serious cases occurred in the absence of underlying diseases or secondary infections and were characterized by rapidly progressing viral pneumonia with diffuse alveolar damage. 22 This disease pattern is notable in that it differs dramatically from disease caused by seasonal influenza viruses.
Summary
Influenza A viruses have great potential to alter their virulence and infect new hosts. The increasing number of outbreaks of highly pathogenic avian influenza that have occurred in recent years underscores the need for continued surveillance and vigilance, especially given the potential for rapid emergence and spread of more virulent strains, such as H5N1.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
This work was supported by the Program of Founding Research Centers for Emerging and Reemerging Infectious Diseases (K. S., Y. K.) and the Global Center of Excellence program (K. S., A. M.) sponsored by the Ministry of Education, Culture, Sports, Science and Technology of Japan.
