Abstract
The current study evaluated the test characteristics of 2 commercially available rapid antigen tests for highly pathogenic avian influenza. Diagnostic specimens were collected from free-ranging village chickens in Indonesia. A total of 174 healthy, sick, and dead birds were included in the study. The relative diagnostic test sensitivity and the relative diagnostic test specificity were calculated by using real-time reverse transcription polymerase chain reaction (RT-PCR) as the confirmatory diagnosis. The AnigenR Rapid AIV Ag test had a relative diagnostic sensitivity of 0.69 (95% confidence interval [CI]: 0.56–0.80) and a relative diagnostic specificity of 0.98 (95% CI: 0.93–0.99). The Flu DetectTM Antigen Capture test strip had a relative diagnostic sensitivity of 0.71 (95% CI: 0.58–0.82) and a relative diagnostic specificity of 0.98 (95% CI: 0.93–0.99). These tests are a valuable tool for the Indonesian avian influenza control program by reliably and quickly detecting Influenza A virus from oropharyngeal swabs from sick or dying chickens.
A critical factor in the success of controlling highly infectious, rapidly spreading epidemics is the time taken to confirm cases. Early detection of outbreaks of highly pathogenic avian influenza (HPAI) is critical for control of avian influenza in animals and for reducing the risk of pandemic human influenza. 9 The time to confirmation is dependent on a number of factors, including an effective surveillance system, appropriate logistics for sample collection and handling, and diagnostic tests that are accurate and quick to run (Boland P, Weaver J, Bhakti Usman T, et al.: 2006, Field evaluation of rapid antigen tests for highly pathogenic avian influenza. Proceedings of the 11th International Symposium on Veterinary Epidemiology and Economics 11:165. Available at: http://www.sciquest.org.nz/default.asp?pageid=69&pub=10&vol=11&iss=. Accessed February 8, 2008). Indonesia first confirmed infection with HPAI type H5N1 in poultry in January 2004. An epidemic of HPAI has since spread across much of the country. At the beginning of 2008, HPAI was well established in free-ranging poultry (Food and Agricultural Organization of the United Nations [FAO] Emergency Prevention System [EMPRES] for Transboundery Animal and Plant Pest and Diseases, EMPRESMAP. Available at: http://www.fao.org/ag/AGAinfo/programmes/en/empres/maps.html. Accessed April 2, 2008). Trained government officers conduct poultry surveillance in villages throughout the country. Rapid detection tests are used for rapid H5N1 screening of suspect cases. Ideally, rapid detection tests are easy to use, reliable, and affordable. A collaborative project was established between the FAO of the United Nations and the Indonesian Directorate General of Livestock Services to evaluate the field use of 2 commercially available rapid antigen tests for HPAI.
The study was conducted in the province of Yogyakarta on the Indonesian island of Java during the months of August and September 2007. Six teams of government officers trained in participatory disease surveillance 1,6 visited areas known to be endemic for HPAI. Diagnostic specimens were collected from free-ranging village chickens. Healthy, sick, and dead birds were included in the study. From each of the 174 birds, 3 oropharyngeal swabs were taken. Immediately, 2 rapid antigen tests, the Anigen® Rapid AIV Ag test a (hereafter Anigen test) and the Flu DetectTM Antigen Capture test strip b (hereafter Flu Detect test), were conducted according to the manufacturers' instructions. The results were recorded. The third swab was placed in BD Viral Transport Medium, c kept at 4°C, and was delivered the same day to the regional laboratory, DiseaseTM Investigation Centre Region IV. Here the specimen was held frozen at −70°C until further testing. Once all 174 specimens were received, 1-step real-time reverse transcription polymerase chain reaction (RT-PCR) tests with both matrix and hemagglutinin 5 (H5) gene primers were used to confirm or exclude H5 antigen.
The 2 rapid antigen tests were assessed for ease of use under field conditions. The relative diagnostic test sensitivity and the relative diagnostic test specificity were calculated by using real-time RT-PCR as the confirmatory diagnosis or “gold standard.” 4 Confidence intervals were calculated by using the Wilson score method without continuity correction. 7
The Anigen test was carried out in 3 simple steps and was easy to use under field conditions. The Flu Detect test required a stable horizontal surface to support the test strip and tube with extraction buffer. This test was also easy to use. The number of positive and negative rapid antigen field tests and the results of the real-time RT-PCR test on the same group of samples are shown in Table 1. Only the positive and negative results of the real-time RT-PCR test were used in the subsequent calculations of the rapid antigen field test characteristics, a total of 157 tests (53 positive and 104 negative). Of the 157 valid PCR tests, a further 2 invalid Anigen test results and 3 invalid Flu Detect results were removed from the analysis. The relative diagnostic sensitivity, the relative diagnostic specificity, and the 95% confidence intervals (CI) for both rapid tests were calculated by using the values shown in Table 2. The results are shown in Table 3.
Results of the 2 field rapid antigen tests and the real-time reverse transcription polymerase chain reaction (real-time RT-PCR) test.

Use of real-time RT-PCR as the reference test may result in misclassification errors. Large-scale proficiency programs for molecular detection of Hepatitis B virus RNA 8 and Hepatitis C virus DNA 11 by using PCR found inconsistent results. An interlaboratory comparison for the detection of Mycoplasma pneumoniae in respiratory samples by PCR showed specificities that ranged from 97.4% to 87.2% and sensitivities that ranged from 97.4% to 89.2%. 10 A recent study described a real-time RT-PCR method for the detection of Influenza A virus subtype H5 with high, but imperfect, sensitivities and specificities. 2 The possible misclassification of the real-time RT-PCR used as reference test does not invalidate the presented results, the calculated relative diagnostic sensitivity and the relative diagnostic specificity are estimates.
The relative diagnostic test sensitivity and the relative diagnostic test specificity of the Flu Detect test and the Anigen test were similar. The characteristics of the tests were calculated for chickens only; sensitivity of the tests is considered lower when used for waterfowl and wild birds. 3 Similar results were found in 2006 for the Anigen test. The sensitivity and specificity of the Anigen test were compared with other in-field antigen tests, and with virus isolation and RT-PCR as reference tests. A total of 164 sick chickens were sampled opportunistically from various sources in Indonesia, which targeted likely incidences of HPAI. The study found an Anigen test sensitivity of 76% and a specificity of 97% (Boland P, et al.: 2006, Field evaluation of rapid antigen tests). This is the first time the relative diagnostic sensitivity and the relative diagnostic specificity of the Flu Detect test has been determined under Indonesian field conditions. A 2006 study in Hong Kong found lower sensitivity values for the Flu Detect test, with virus isolation as the confirmatory test. The sensitivity for the Flu Detect test was possibly lower in that 2006 study, because the swabs collected were placed in viral transport medium, frozen, and thawed before the Flu Detect tests were conducted. 3
Two-by-two table showing the results of the 2 field rapid antigen tests compared with the real-time reverse transcription polymerase chain reaction (rate-time RT-PCR) test.*

For the Anigen Rapid AIV Ag test, 52 positive and 103 negative real-time RT-PCR test results were included. For the Flu Detect Antigen Capture test strip, 52 positive and 102 negative real-time RT-PCR test results were included.
Sensitivity and specificity of the 2 field rapid antigen tests by using real-time reverse transcription polymerase chain reaction as the reference test.*
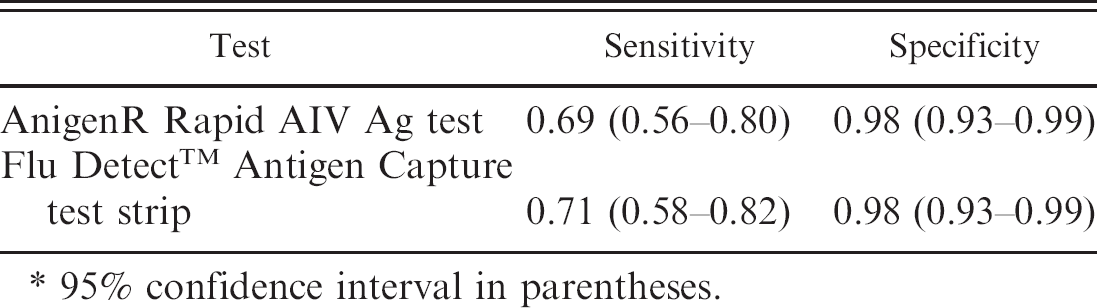
95% confidence interval in parentheses.
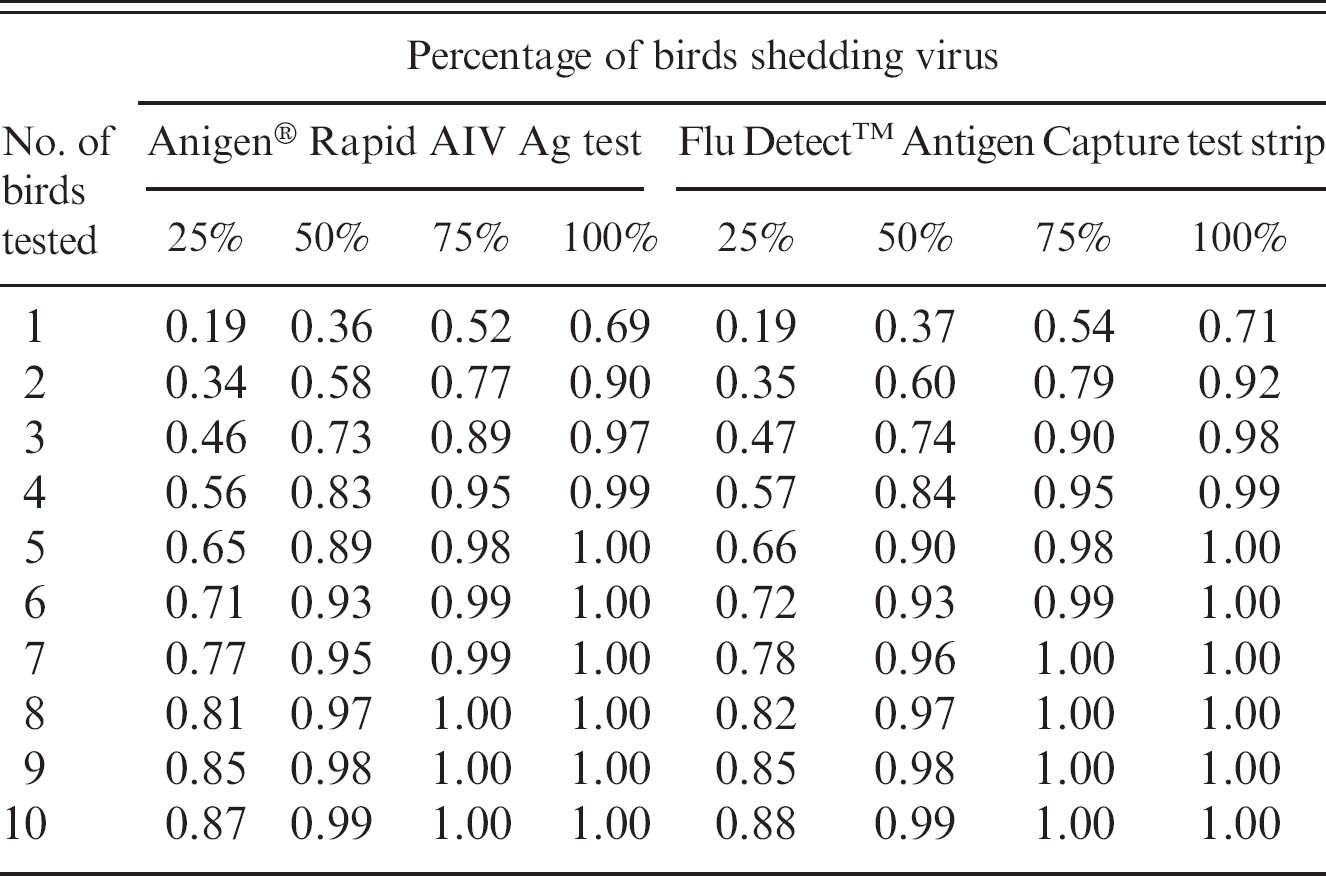
These tests can assist in facilitating rapid investigations and control interventions but should only be considered as screening tests to be followed up by confirmatory laboratory tests, such as PCR techniques or virus culture, whenever possible. The relatively low sensitivity means that individual birds are quite likely to test as false negatives; to reduce this problem, the tests should only be used on optimal clinical specimens from sick, moribund, or recently dead birds. 3 As directed by the manufacturers, tracheal and oropharyngeal swabs should be taken from behind the tongue into the oropharyngeal or trachea area. Cloacal swabs should be taken from within the cloacal area and avoid excess solid fecal material and visible blood. Furthermore, high sensitivity is needed for a successful HPAI control program. False-negative test results do not mobilize a response to control an outbreak, which may lead to possible further spread of disease. Therefore, it is recommended to test multiple animals to increase flock sensitivity (multiple tests with parallel interpretation). 4 The flock sensitivity for both tests for flocks with different infection levels and different number of birds tested are shown in Table 4. 5
Flock sensitivity using 2 field antigen tests for flocks with different levels of infection and with a different number of birds tested.
Acknowledgements. The authors would like to thank Eric Brum for his operational and technical support and the nine government officers who did all the field work: Agung Ludiro, Agung Nugroho, Antoni Kirwanto, Dradjad, Nyoman Anggraeni, Sri Imawati, Sugeng Leksomono, Wisnu Suksmono, and Yuli Astuti. This study was carried out under the FAO Avian Influenza Control Program-Indonesia and was funded by the United States Agency for International Development.
Footnotes
a.
Animal Genetics Incorporated, Kyonggi-do, Korea.
b.
Synbiotics Corp., San Diego, CA.
c.
BD, Franklin Lakes, NJ. BD Viral Transport Medium consists of: Hank's balanced salts, bovine serum albumin, L-cysteine, gelatin, sucrose, L-glutamic acid, HEPES buffer, vancomycin, amphotericin B, colistin, and phenol red: adjusted to pH 7.32.
