Abstract
Direct bird-to-human transmission, with the production of severe respiratory disease and human mortality, is unique to the Hong Kong-origin H5N1 highly pathogenic avian influenza (HPAI) virus, which was originally isolated from a disease outbreak in chickens. The pathobiology of the A/chicken/Hong Kong/ 220/97 (H5N1) (HK/220) HPAI virus was investigated in chickens, turkeys, Japanese and Bobwhite quail, guinea fowl, pheasants, and partridges, where it produced 75-100% mortality within 10 days. Depression, mucoid diarrhea, and neurologic dysfunction were common clinical manifestations of disease. Grossly, the most severe and consistent lesions included splenomegaly, pulmonary edema and congestion, and hemorrhages in enteric lymphoid areas, on serosal surfaces, and in skeletal muscle. Histologic lesions were observed in multiple organs and were characterized by exudation, hemorrhage, necrosis, inflammation, or a combination of these features. The lung, heart, brain, spleen, and adrenal glands were the most consistently affected, and viral antigen was most often detected by immunohistochemistry in the parenchyma of these organs. The pathogenesis of infection with the HK/220 HPAI virus in these species was twofold. Early mortality occurring at 1-2 days postinoculation (DPI) corresponded to severe pulmonary edema and congestion and virus localization within the vascular endothelium. Mortality occurring after 2 DPI was related to systemic biochemical imbalance, multiorgan failure, or a combination of these factors. The pathobiologic features were analogous to those experimentally induced with other HPAI viruses in domestic poultry.
The fundamental epizootiology of type A orthomyxoviruses is dependent on the interactions of avian and mammalian species, with waterfowl serving as the reservoir of influenza viruses for other birds, mammals, and humans. 42 52 54 Avian influenza viruses typically demonstrate host specificity, with interspecies transmissions being the exceptions. 48 Natural interclass transmission of these viruses is considered to be an infrequent event, 53 and influenza viruses transmitted from birds to mammals only rarely produce significant, although self-limited, disease in the aberrant host. 22 54 In 1997, zoonotic transmission of an H5N1 highly pathogenic avian influenza (HPAI) virus caused 18 cases of human infection, with six associated fatalities. 11 12 This outbreak was the first clinical infection of humans with the H5 subtype of HPAI, raising concerns that this virus could initiate the next human pandemic, but depopulation of poultry in Hong Kong averted such a disaster. 45 46
In the experimental setting, the chicken and human Hong Kong–origin H5N1 influenza viruses replicated in chickens, geese, and ducks. 43 In chickens, these isolates produced a rapidly fatal systemic disease, with median death times of 1 day postinoculation (DPI) and lesions typical of HPAI. 43 45 In experimentally infected geese, clinical disease and mortality occurred in 50% and 17% of infected animals, respectively. 43 Conversely, in ducks the H5N1 Hong Kong isolates produced innocuous infection characterized by transient shedding and no clinical disease. This limited infection is typical of avian influenza viruses (AIVs) in this subfamily of waterfowl. 16
In addition to avian species, the Hong Kong H5N1 influenza virus isolates are capable of replication in mice, rats, and pigs. 15 18 31 43 In comparison with other subtype H5 HPAI viruses, the Hong Kong–origin influenza viruses demonstrated inherent virulence for mice by causing high morbidity and mortality following intranasal inoculation without prior mouse adaptation. 15 These viruses are distinct in their ability to replicate and cause disease despite the interclass restrictions associated with other influenza viruses.
Systemic replication of AIVs in chickens and turkeys is largely defined by the presence of multiple basic amino acids at the hemagglutinin cleavage site. 28 41 56 Despite this molecular conformity among H5 and H7 HPAI viruses, considerable variation has been observed in the pathogenicity and transmissibility of these viruses in different avian species, even within the avian order Galliformes. For example, the A/turkey/Ontario/7732/66 (H5N9) influenza virus is moderately pathogenic for chickens and quail, highly pathogenic in turkeys, and apathogenic in pheasants, pigeons, and ducks. 44 Because of the differences in virulence AIVs demonstrate in domesticated avian species, it is important to consider possible ramifications of interspecies transmission of influenza viruses when different poultry species are commingled.
In this communication, we present the morbidity and mortality, gross and histologic lesions, and the immunohistochemical distribution of HK/220 viral antigen in seven species of gallinaceous birds. We assessed the basic pathobiology of the HK/220 virus in these species and compare it with that of other HPAI viruses in domestic poultry.
Materials and Methods
Virus propagation
The stock virus of A/chicken/Hong Kong/220/97 (H5N1) avian influenza virus (HK/220) was produced by second passage in 10-day-old embryonated chicken eggs. Allantoic fluid from inoculated eggs was collected and diluted 1:300 in beef heart influsion medium (BHI). Similarly, a sham inoculum was made using sterile allantoic fluid diluted 1:300 in BHI. The original HK/220 AIV was first isolated by Drs. Les Sims and Kitman Dyrting (Agriculture and Fisheries Department, Hong Kong).
Animals
Seven species of gallinaceous birds were used: specific-pathogen-free (SPF) White Leghorn (WL) chickens (Gallus domesticus; Southeast Poultry Research Laboratory [SEPRL], Athens, GA), SPF White Plymouth Rock (WR) chickens (G. domesticus; SEPRL), male Broad-breasted White turkeys (Meleagris gallopavo; British United Turkeys, Lewisburg, WV), Japanese quail (Coturnix coturnix japonicus; Poultry Science Department, University of Georgia, Athens, GA), Bobwhite quail (Colinus virginianus; Ideal Poultry, Cameron, TX), Pearl guineafowl (Numida meleagris; Privett Hatchery, Portales, NM), Ring-necked pheasant (Phasianus colchicus; Ideal Poultry), and Chukar partridges (Alectoris chukar; Ideal Poultry). All birds were inoculated at 4 weeks of age, with the exception of the turkeys, which were inoculated at 3 weeks of age. Serum samples were collected from a representative number of birds of each species prior to inoculation to ensure that the birds were serologically negative for AIV as determined with the agar gel precipitin (AGP) test. Procedures for AGP followed those previously described. 6 Each species was housed separately in Horsfal-Bauer stainless steel isolation cabinets or in self-contained isolation units (Mark 4, Controlled Isolation Systems, San Diego, CA) that were ventilated under negative pressure with HEPA-filtered air and maintained under continuous lighting. Feed and water were provided ad libitum. General care was provided as required by the Institutional Animal Care and Use Committee, as outlined in the Guide for the Care and Use of Agricultural Animals in Agricultural Research and Teaching. 13 All experiments were performed in an USDA certified biosafety level 3 agriculture facility at the SEPRL. 5
Animal inoculation and sampling
For each species, birds were divided into three groups: a control group, a modified pathotype group, and a sample group. The control group contained eight individuals intranasally (IN) inoculated with 0.05 or 0.1 ml of the sham inoculum. Two control birds were euthanatized at 2 and 14 DPI, and tissues were collected in 10% buffered formalin for histopathologic evaluation. At 14 DPI, serum was collected from two to six of the remaining control birds for AGP to ensure that controls remained serologically negative to AIV.
The pathotype group, which contained six to eight birds, and the sample group, which contained 10–18 birds, were inoculated IN with 0.05 or 0.1 ml of inoculum containing 105.8 to 106.2 EID50 of the HK/220 virus (Table 1). Inoculated birds were observed for 14 days after inoculation, during which time the clinical signs were recorded. Dead and moribund birds of these groups were necropsied for the determination of gross lesions. Birds of the sample group were euthanatized and necropsied at 1, 2, and 4 DPI or as birds became moribund. From euthanatized and select recently deceased birds, tissues for histopathologic examination were collected as described for the control group. Serum and tissues were collected from any virus-inoculated birds surviving at the termination of the experiment.
Number of birds per species intranasally inoculated with A/chicken/Hong Kong/220/97 (H5N1) influenza virus for pathogenicity assessment and tissue sampling.

Number of pathotype group dead/total number pathotype group × 100.
Median death time (range) of pathotype group.
Sample birds, moribund birds, and all birds remaining at the end of the 14-day period were euthanatized by the intravenous (IV) administration of sodium pentobarbital (100 mg/kg body weight).
Histopathology and immunohistochemistry
Tissues were fixed by submersion in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Sections were made at 7 µm and were stained with hematoxylin and eosin (HE). A duplicate 4-µm section was immunohistochemically stained by first microwaving the sections in Antigen Retrieval Citra Solution (Biogenex, San Ramon, CA) for antigen exposure. A 1:2,000 dilution of a mouse-derived monoclonal antibody (P13C11) specific for a type A influenza virus nucleoprotein (SEPRL) was applied and allowed to incubate for 2 hours at 37 C. The primary antibody was then detected by the application of biotinylated goat anti-mouse IgG secondary antibody using a biotin–streptavidin detection system (Supersensitive Multilink Immunodetection System, Biogenex). Fast Red TR (Biogenex) served as the substrate chromagen, and hematoxylin was used as a counterstain.
Results
Sham-inoculated controls
There was no mortality observed in the sham-inoculated control birds of any species. Focal subcutaneous and skeletal muscle hemorrhages, interpreted as self-inflicted trauma, were recognized infrequently in individual control Bobwhite quail (B. quail) and pheasants. Histologically, control birds of each species lacked significant lesions. With the exclusion of the chickens, guineafowl, and Japanese quail (J. quail), there was infrequent nonspecific immunohistochemical staining, which was restricted to cytoplasmic granules of few individual cells of the spleen, harderian gland, and lacrimal gland. Immunohistochemical staining of this nature has been previously interpreted as staining of mast cell granules (Swayne, personal communication). Control birds sampled at 14 DPI did not seroconvert to AIV nucleoprotein as determined by AGP.
Mortality
Intranasal administration of the HK/220 influenza virus resulted in 100% mortality in six of the seven species included in this study. The single exception was the Chukars, which experienced 75% mortality over the 14-day period. The chickens succumbed most rapidly to the infection, with all WL and WR chickens dying by 2 DPI. The percentage mortality, median death time (MDT), and temporal range of mortality for the pathotype group of each species are presented in Table 1.
Morbidity
Minimal clinical signs were observed in the WL and WR chickens, guineafowl, and J. quail. In these species, the onset of depression rapidly progressed to death within 6–8 hours. However, two J. quail (8%) displayed mild neurologic signs at 2 and 2.5 DPI. Conversely, the advancement of depression was more prolonged and was often accompanied by progressive neurologic signs in the B. quail (14%), turkeys (41%), pheasants (13%), and Chukars (28%). These signs included attenuated motor functions, such as paresis to paralysis and vestibular degradation as indicated by torticollis and nystagmus, and general behavioral aberrations (Fig. 1). The onset times of these neurologic signs initially were observed at 1.5 DPI in the turkeys, 3 DPI in the Bobwhites and pheasants, and 4 DPI in the Chukars.

Bobwhite quail, 3 DPI. Severe neurologic signs include torticollis and paralysis. Bar = 1 cm.
The development of increased fecal fluid and urates occurred in all seven species beginning at 1–1.5 DPI. The WL chickens were unique in having hematochezia as well. The turkeys, B. quail, Chukars, and pheasants, developed mucoid diarrhea with strings of urates, which has been more aptly described as “egg-albumin feces.” 27 This condition was observed in all of the inoculated birds, including the two Chukars that survived infection. For only the Chukars, the urates acquired a pale green hue in a majority of the birds. Oculonasal discharge and dyspnea were observed sporadically.
Gross lesions
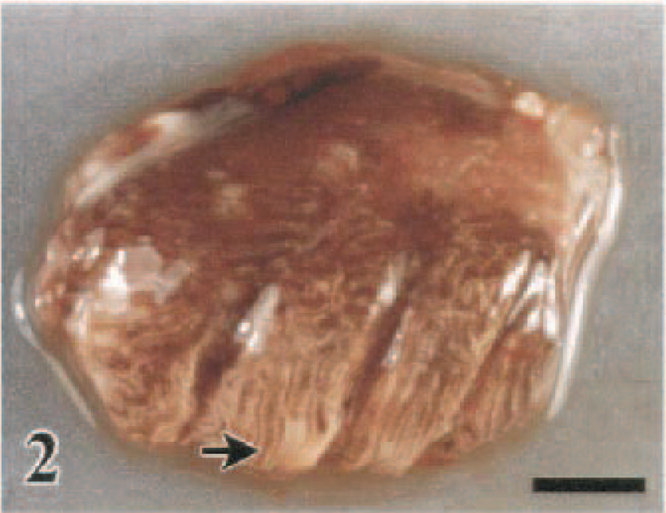
The HK/220 HPAI virus produced several consistent gross lesions among the species investigated. Foremost among these lesions was splenomegaly with parenchymal mottling, which was observed in 42% (J. quail) to 100% (WR chickens) of the birds. Although splenic lesions were observed within 1 DPI, they were most pronounced in those birds dying or sampled after 2 DPI. Renomegaly with parenchymal pallor and accentuated lobular surface architecture also was common to all seven species, occurring in 22% (Chukars) to 94% (WL chickens) of the birds. Urate accumulation in the ureters often accompanied the renal alterations. Pulmonary consolidation with edema and congestion to hemorrhage (Figs. 2, 3) was most striking in the WL chickens (94%), WR chickens (86%), J. quail (92%), and guineafowl (79%) and was less pronounced in the B. quail (29%), turkeys (59%), pheasants (26%), and Chukars (17%). There was a similar species predilection for mucosal hemorrhage in the alimentary tract, with the WL Chickens (89%), WR chickens (57%), guineafowl (58%), and J. quail (15%) having this lesion most consistently. Hemorrhages were identified predominantly at lymphoid areas of the alimentary tract, such as the esophageal–proventricular junction (Fig. 4), Peyer's patches (Fig. 5), and cecal tonsil. In the WL chickens, severe hemorrhage resulted in the accumulation of undigested blood within the enteric lumen (Fig. 6). Furthermore, hemorrhage in the cloacal bursa was exclusive of the WL chickens (83%), WR chickens (18%), and guinea fowl (53%). Mucosal hyperemia to hemorrhage at the submucosal lymphoid tissue of the palate was observed only in the guineafowl (11%). In the B. quail, turkeys, pheasants, and Chukars, enteric hemorrhages, which were largely confined to the cecal tonsil, occurred in only one or two birds of each of these species. These four species had marked distension of the enteric tract with yellow fluid, which was evident in 30% (pheasants) to 90% (B. quail) of these birds. Hemorrhages also were observed in the fascial sheaths of skeletal muscles in 21% (WR chickens) to 68% (guineafowl) of the birds (Fig. 7). A variable 9% (pheasants) to 72% (WL chickens) of the birds had petechial to ecchymotic hemorrhages on the epicardium and/or serosal surfaces of the liver and intestines (Fig. 8). Serous exudates in body cavities, such as the pericardial sac and coelom, were observed in 5% (B. quail) to 83% (pheasants) of the birds.
Lung; guineafowl, 2 DPI. Parenchymal hemorrhage along the dorsal aspect of lung and exudation of serosanguinous fluid. The parenchyma at the peripheral margin is relatively unaffected (arrow). Bar = 0.5 cm.
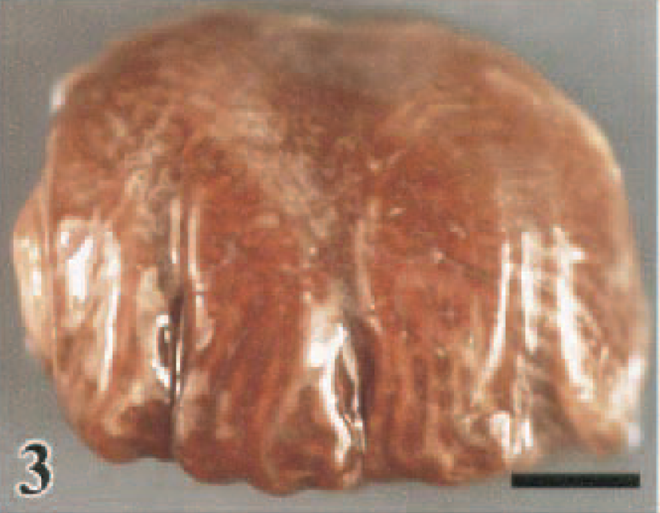
Lung; White Rock chicken, 1.5 DPI. Severe diffuse edema and congestion of pulmonary parenchyma. Bar = 0.5 cm.
Interior of proventriculus; White Leghorn chicken, 1.5 DPI. Mucosal hemorrhages at esophageal–proventricular junction (arrow) and at the papillae of the proventricular ducts. Bar = 0.5 cm.
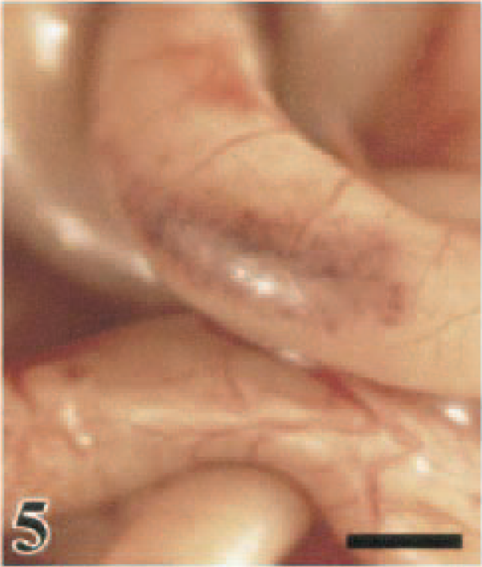
Ileum; White Leghorn chicken, 1.5 DPI. Mucosal hemorrhage in an ileal Peyer's patch is visible from the serosal surface. Bar = 0.5 cm.
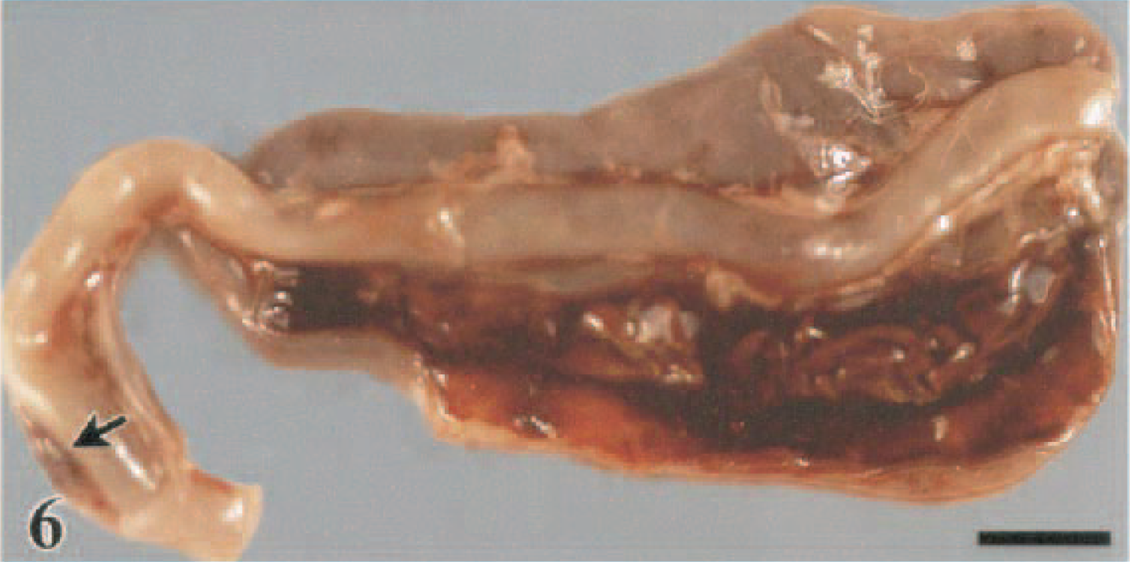
Cecal tonsil and cecae; White Leghorn chicken, 1.5 DPI. Mucosal hemorrhage in the cecal tonsil (arrow) is visible from the serosal surface. Undigested blood is admixed with contents in the cecal lumen. Bar = 0.5 cm.
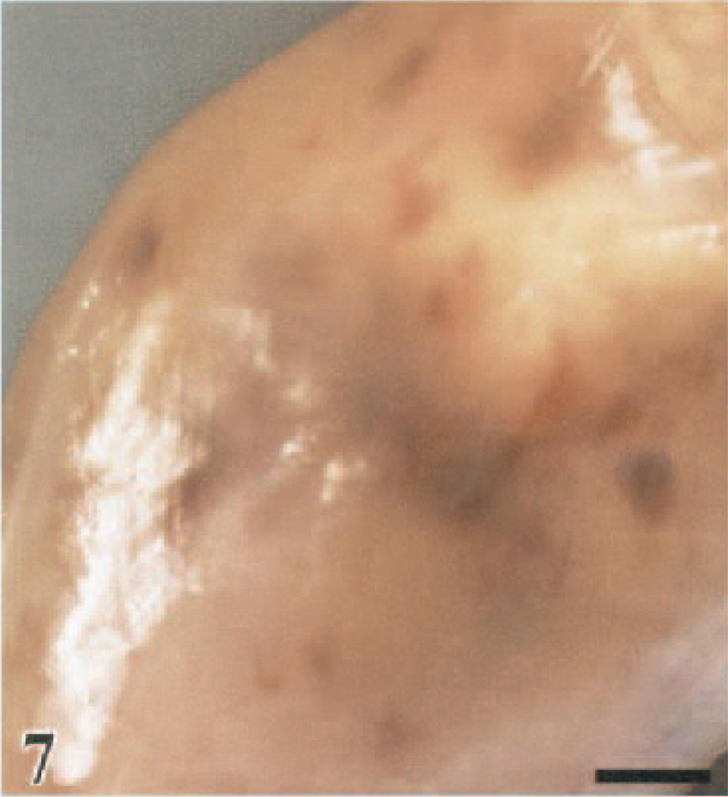
Skeletal muscle; guineafowl, 2 DPI. Multiple hemorrhages in the medial fascial plane of the gastrocnemius (pars intermedia) muscle. Bar = 0.35 cm.
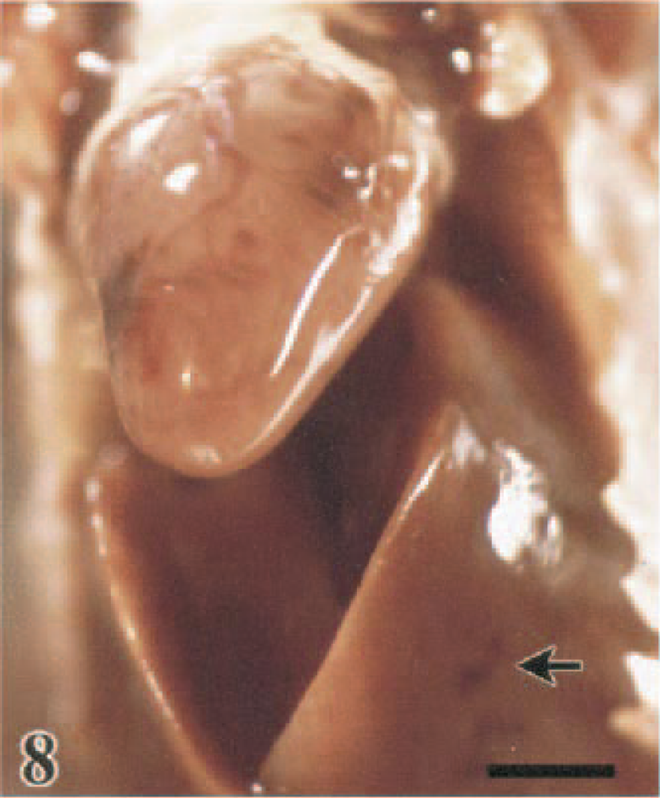
Heart and liver in situ; Japanese quail, 2.0 DPI. Petechial to purpuric hemorrhages on the epicardial surface and on the serosal surface of the liver (arrow). Bar = 0.5 cm.
Other lesions were more exclusive within a certain species, such as conjunctival hyperemia and edema in the WL chickens (53%), WR chickens (50%), and guineafowl (50%). In the WR chickens, these conjunctival lesions progressed to periorbital edema. Pancreatic lesions, characterized by multiple foci of parenchymal discoloration, were observed in the turkeys (71%), B. quail (67%), and Chukars (18%) (Fig. 9). Only the pheasants (44%) and Chukars (18%) had intense bile staining of the mucosa of the proventriculus and koilin lining of the ventriculus. Ischemic necrosis of the comb tips was observed exclusively in WL chickens (22%).
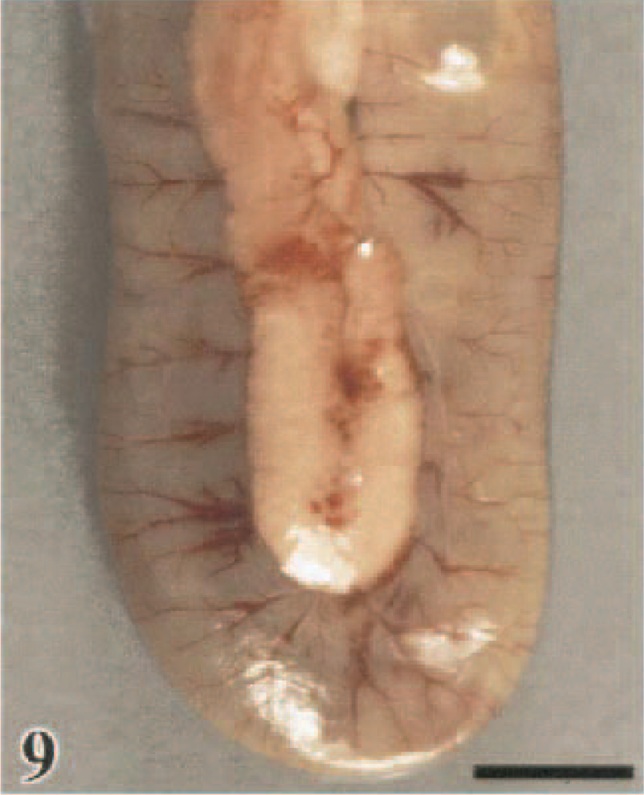
Pancreas and duodenal loop; turkey, 2.5 DPI. Multifocal reddening of the pacreatic parenchyma. The duodenum is moderately distended with fluid contents. Bar = 0.75 cm.
Histologic lesions
Respiratory tract
The prevailing lesions within the respiratory tract were confined to the nasal cavity and lung. In the nasal cavity, there was acute heterophilic rhinitis, which was often accompanied by a mucocellular exudate containing sloughed epithelial cells and heterophils, by submucosal edema, and by epithelial changes including loss of cilia and vacuolar degeneration to necrosis (Fig. 10a). These lesions were observed in 36% (Chukars) to 100% (turkeys, B. quail, and WR chickens) of the birds and were of moderate severity in the B. quail, J. quail, turkeys, and guineafowl (Table 2). Changes consistent with epithelial cell regeneration, including mild epithelial hyperplasia and cystic dilatation of mucosal glands, were confined to Chukars sampled after 5 DPI (18%).
Average severity of histologic lesions∗ in gallinaceous birds that died after intranasal inoculation with A/chicken/Hong Kong/220/97 (H5N1) influenza virus.
- = no lesions; ± = minimal; + = mild; ++ = moderate; +++ = severe.
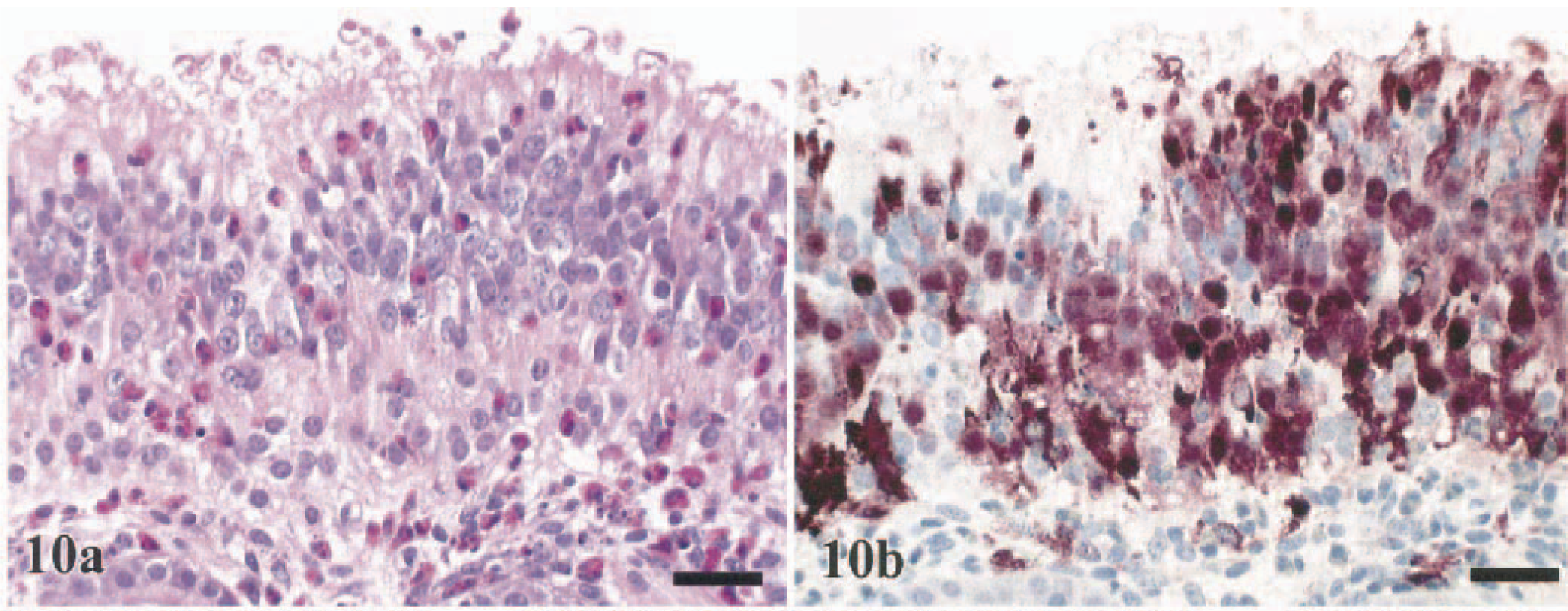
Nasal epithelium; Bobwhite quail, 1 DPI.
Severe diffuse peracute to acute exudative interstitial pneumonia with congestion to hemorrhage was most severe and most consistent in the lungs of the chickens (100%), J. quail (92%), guineafowl (100%), and B. quail (80%) sampled between 1 and 2 DPI (Table 2). Contributing lesions included marked endothelial cell hypertrophy, fibrinous microthrombosis of capillaries, and small disseminated foci of cell necrosis (Fig. 11a). Heterophilic and mononuclear infiltrates remained confined within the vascular capillary lumens and interstitium. Pulmonary alterations in the turkeys (86%), pheasants (78%), and Chukars (55%) were less exudative with mild to moderate heterophilic infiltration (Table 2). The most severe pulmonary lesions occurred at 2 DPI in the turkeys, at 2–3 DPI in the pheasants, and at 3–5 DPI in the Chukars.
Lung; White Leghorn chicken, 1.5 DPI.
The larynx, trachea, and air sacs in these species did not consistently contain significant lesions. However, the laryngeal and bronchiole-associated lymphoid tissues were exceptions, containing minimal to mild apoptotic lymphoid depletion, heterophilic inflammation, and mild degenerative changes of the overlying epithelium.
Cardiovascular system
Vascular changes indicative of acute inflammation were prevalent in all seven species. Endothelial cell hypertrophy or swelling, which was often accompanied by heterophilic pavementing to vascular exocytosis, was readily identified in small caliber vessels and capillaries and was especially prevalent in highly vascular tissues such as lung, spleen, and conjunctiva and the comb of chickens (Fig. 11a).
Endocardial hemorrhage and random multifocal to confluent myocardial degeneration to necrosis was observed in 45% (guineafowl) to 100% (pheasants) of the sampled birds (Fig. 12a). Myocardial necrosis was initially observed at 1.5–2 DPI and was mild in the majority of the species investigated (Table 2). Minimal to mild heterophilic to mononuclear inflammation was infrequently associated with myocardial necrosis in the pheasants (78%), WR chickens (43%), turkeys (29%), guineafowl (18%), and a single J. quail (8%) (Table 2). In Chukars sampled between 3 and 5 DPI, myocardial lesions were more discrete and well delineated by infiltrating macrophages (Fig. 13a). Other changes, which were sporadic and primarily limited to five pheasants (56%) and two WR chickens (25%), were mild to moderate edema of the pericardium with heterophilic inflammation.

Heart; turkey, 2.5 DPI.
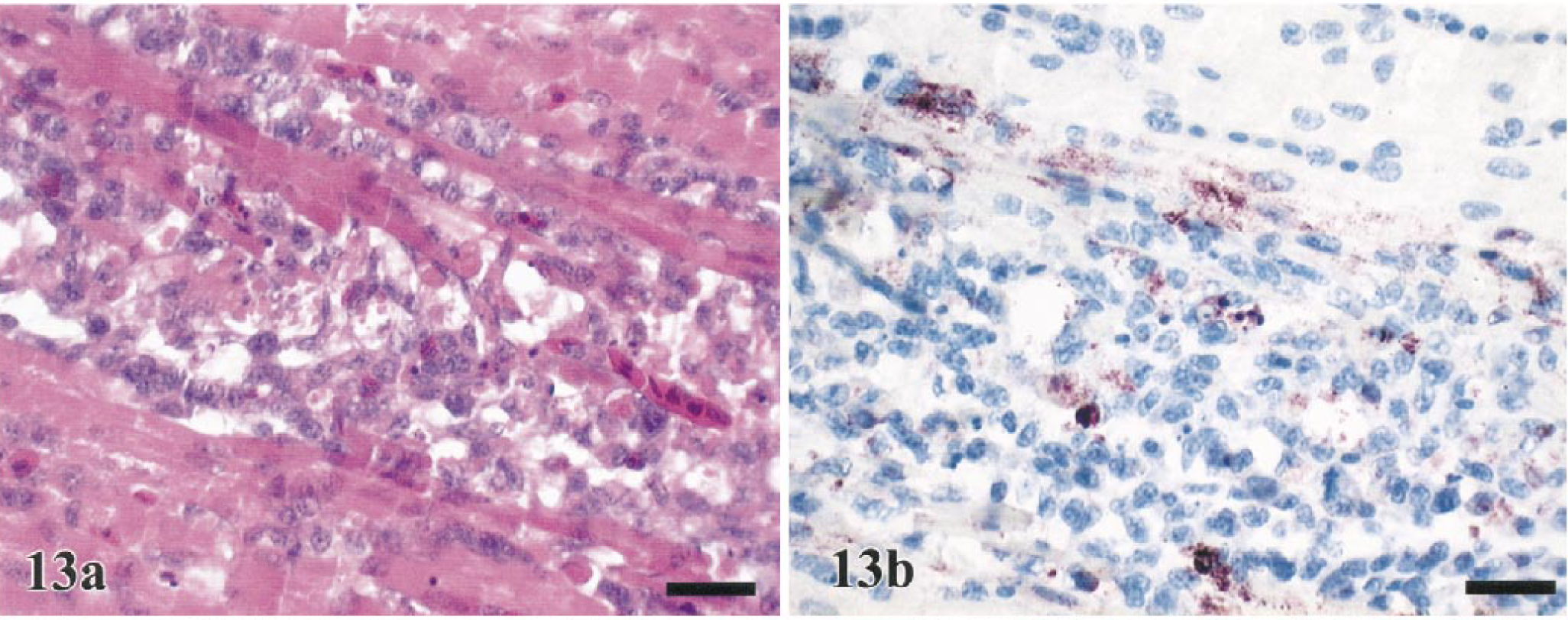
Heart; Chukar, 4 DPI.
Central and peripheral nervous systems
In the brain, dissecting vacuolation of the neuropil and white matter with dilatation of Virchow-Robbins spaces, which was interpreted as edema, was observed in 36% (guineafowl) to 71% (turkeys) of the sampled birds and was most severe in the B. quail and turkeys (Table 2). Endothelial hypertrophy accompanied the perivascular edema. Mild to moderate randomly disseminated foci of neuronal and glial cell necrosis, with sporadic involvement of ependymal cells and epithelial cells of the choroid plexus, were observed in all species (Fig. 14a) but were most consistent among the J. quail (62%), B. quail (60%), turkeys (71%), pheasants (78%), and Chukars (73%) (Table 2). Corresponding microgliosis, with occasional formation of glial nodules, and neuronophagia were observed in 46% (J. quail) to 73% (Chukars) of these five species. Mild to moderate choroiditis, consisting of vascular engorgement and heterophilic infiltration with mild exocytosis, was observed in one WL chicken, one turkey, two J. quail, seven pheasants, and four Chukars. With the exception of the Chukars, the encephalitic lesions in all species were similar in their time of onset of 1.5–2 DPI and generally progressed in severity with time. In the Chukars, lesions were delayed to 3 DPI. Five Chukars (45%), sampled at 4 and 5 DPI, had mononuclear perivascular cuffs in areas affected with malacia and gliosis (Fig. 15a).
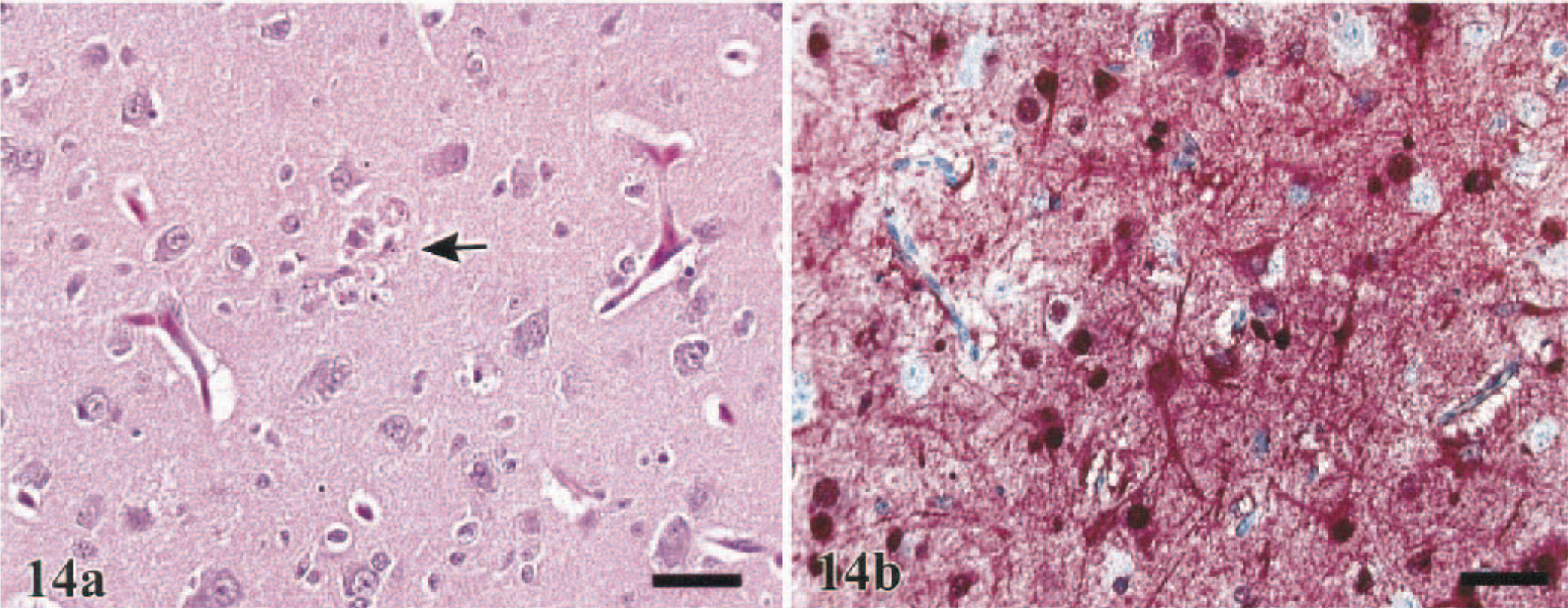
Brain, cerebrum; turkey, 2.5 DPI.

Brain, optic lobe; Chukar, 5 DPI. Marked mononuclear perivascular cuffs with few heterophils and gliosis in the surrounding neuropil. HE. Bar = 75 µm.
Peripheral autonomic ganglia, plexi of the enteric tract, and nerves in skeletal muscle only sporadically contained minimal to mild degenerative changes in neurofibers and rare heterophilic to mononuclear inflammation in five of the seven species. The chickens and guineafowl were the two species in which lesions were not observed in peripheral nerves.
Alimentary tract
Lesions in the alimentary tract were largely confined to lymphoid-associated areas, including those at the esophageal–proventricular junction, the Peyer's patches of the small intestine, and the cecal tonsils. Of these areas, the cecal tonsil was consistently the most severely affected. Enteritis could be classified as hemorrhagic in the WL chickens (100%) and WR chickens (88%) to heterophilic in the turkeys (100%), B. quail (40%), pheasants (40%), Chukars (36%), and J. quail (23%) (Fig. 16a Table 2). Often there was fibrinous to fibrinopurulent exudation into the enteric lumen accompanying the proprial heterophilic infiltration. Despite the inflammatory changes in the lamina propria, the intestinal epithelium was only minimally to mildly affected in 9% (Chukars) to 57% (turkeys) of the birds sampled, with the exception of the pheasants (Table 2). Lymphoid depletion by apoptosis to necrosis was common to all seven species but was most severe in the chickens, J. quail, turkeys, and guineafowl (Table 2). In all eight groups, the proventriculus and ventriculus intermittently contained small foci of superficial heterophilic infiltration that was irregularly accompanied by epithelial necrosis.
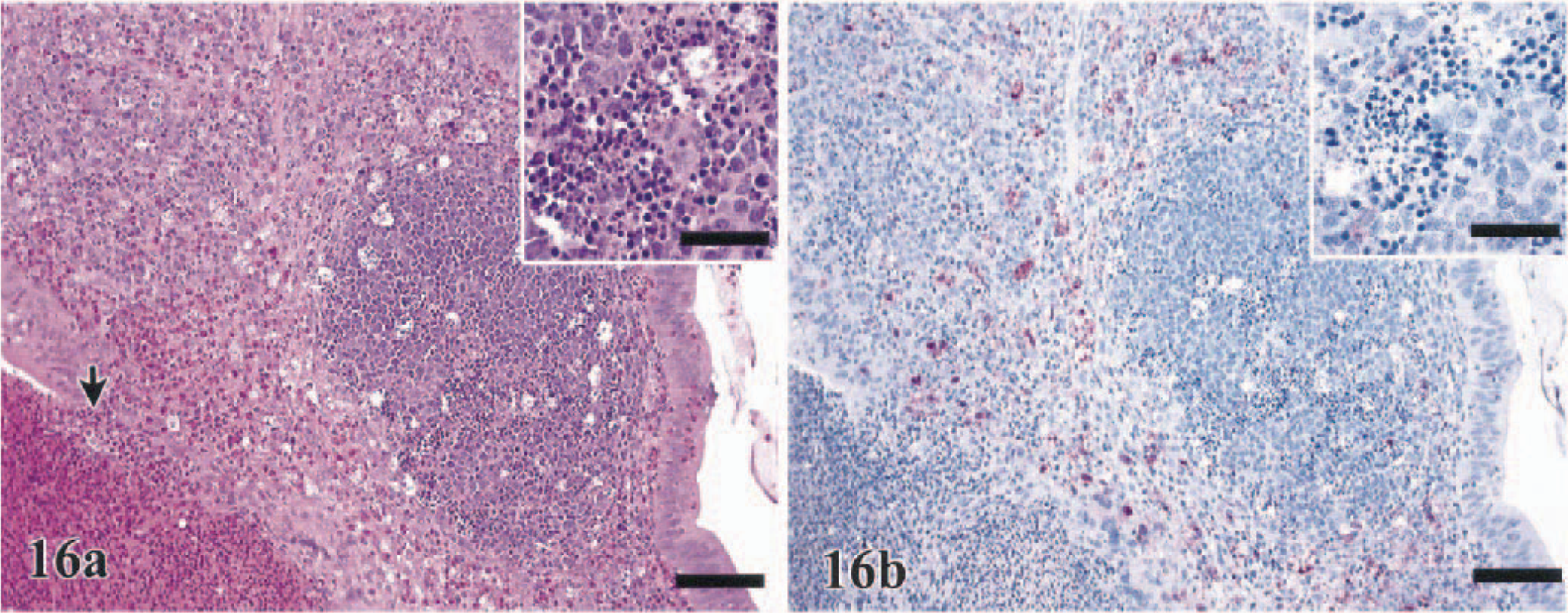
Cecal tonsil; turkey, 2 DPI. Severe heterophilic infiltration of the lamina propria with exocytosis into lumen (arrow). HE. Bar = 75 µm. Inset: Moderate apoptotic lymphocellular depletion also is present. Bar = 15 µm.
Pancreas and liver
Multifocal cellular swelling to necrosis of the pancreatic acinar epithelium occurred in 55% (J. quail) to 86% (turkeys) of sampled birds of five of the seven species (Table 2). Inflammation corresponding to destruction of the pancreatic parenchyma was poorly developed in these species and was observed in only one J. quail, one turkey, and one Chukar (Table 2). The islets of Langerhans remained largely unaffected.
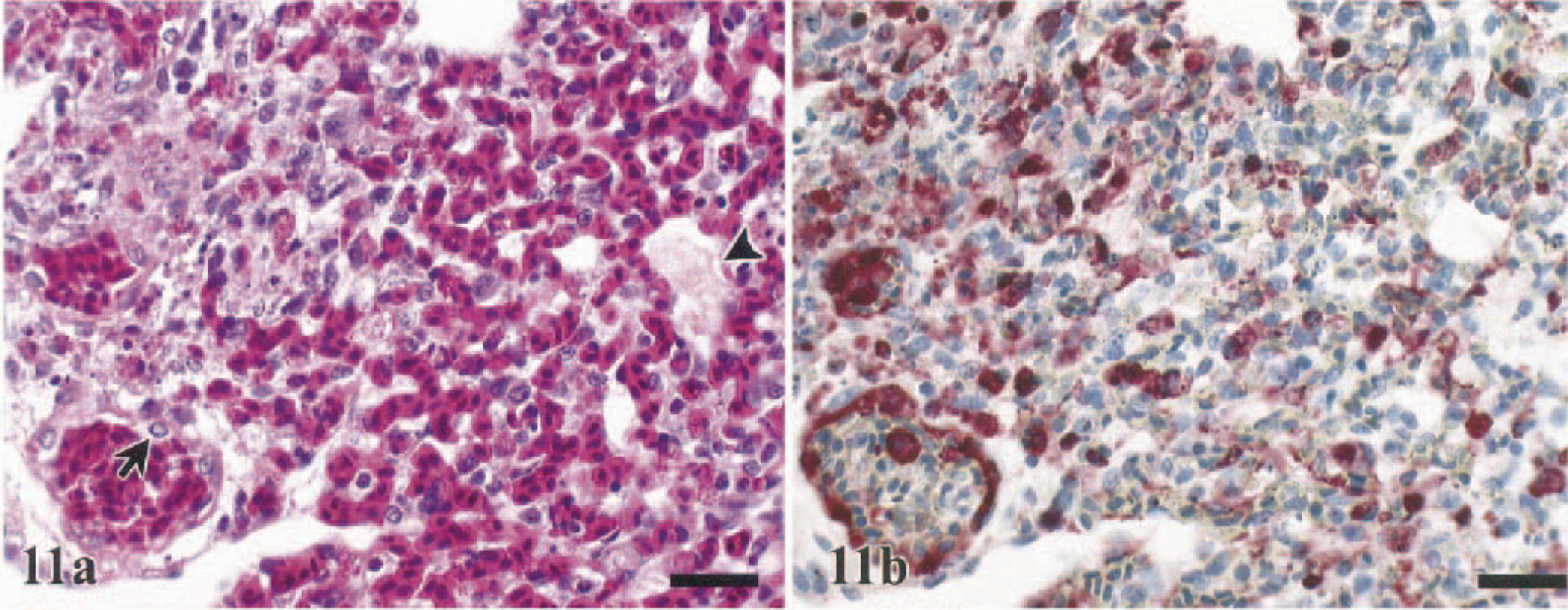
Random foci of hepatocellular necrosis was observed in 38% (WR chickens) to 100% (WL chickens) of the sampled birds and was minimal to moderate in the majority of the species investigated (Table 2). Few large foci of coagulative necrosis, indicative of infarction, were observed in the pheasants (50%), Chukars (55%), and turkeys (33%) (Fig. 17a). In all species, there also was an increase in sinusoidal cellularity that was attributed to the combined presence of pyknotic to karyorrhectic debris, Kupffer cell hypertrophy and hyperplasia, and increased numbers of heterophils in the sinusoids. Engulfed erythrocytes and/or hemosiderin were commonly recognized in the cytoplasm of Kupffer cells.
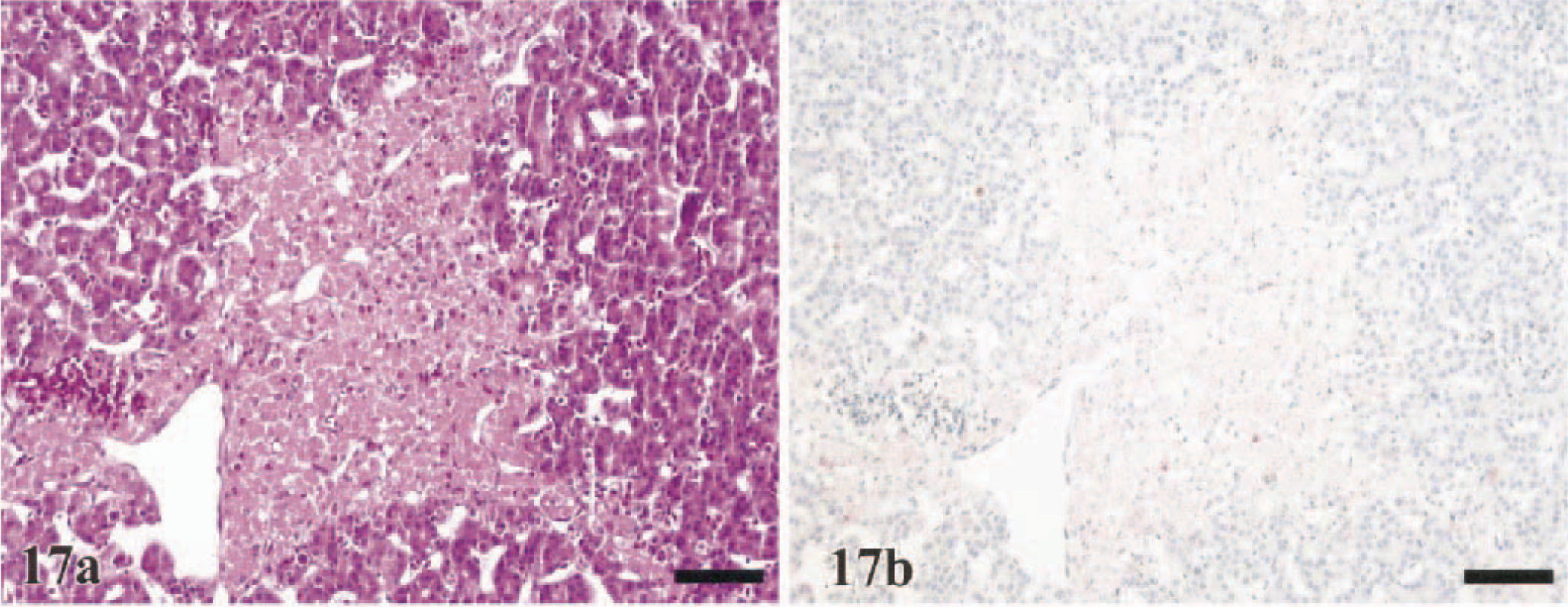
Liver; Ring-necked Pheasant, 3 DPI.
Hematopoietic system
Fifty percent (WL chickens) to 82% (guineafowl) of the sampled birds had erythroid and myeloid cellular depletion in the bone marrow. This pancellular depletion was most pronounced in the chickens, the B. quail, the J. quail, and the Chukars (Table 2). In addition, degenerate to necrotic cells in sinusoidal and extrasinusoidal compartments and macrophages containing engulfed cellular debris concurred with the decrease of cellularity (Table 2).
The spleen was one of the most rapidly affected organs; lesions were observed within 1 DPI in all species except for the Chukars, in which lesions were not observed until 2 DPI. Lymphoid depletion of the periellipsoidal and periarteriolar sheaths was moderate to marked in the majority of species (Table 2) and was observed in 71% (turkeys) to 100% (WL chickens) of the birds sampled, with the exception of the pheasants (50%). Additional lesions that were regularly observed among the seven species included sinusoidal congestion, heterophilic infiltration of sinusoids, and serofibrinous deposits, which often formed coalescent pools. Progressive lymphoid depletion was coupled with histiocytic and ellipsoidal hyperplasia, which was most conspicuous in the guineafowl, pheasants, and Chukars (Table 2). Histiocytes often contained phagocytized cellular debris and erythrocytes.
As in the spleen, the thymus, bursa, and mucosa-associated lymphoid tissue uniformly had moderate to severe lymphoid depletion with apoptosis to necrosis in remaining lymphocytes (Table 2). Both cortical and medullary regions of bursa and thymus were affected. Within 1.5–2 DPI, active lymphocellular depletion, indicated by numerous pyknotic to karryorhectic lymphocytes, was observed in 60% (B. quail) to 100% (WL chickens) of the birds of each species. Infiltrating heterophils increased proportionately with the accumulation of lymphocellular debris, especially in lymphoid areas in the alimentary tract as previously described. Reactive histiocytic hyperplasia was most apparent in the pheasants (80%) and Chukars (73%) (Table 2).
Endocrine organs
The corticotrophic cells and less consistently the chromaffin cells of the adrenal gland had mild to moderate multifocal to confluent areas of vacuolar degeneration to necrosis in 44% (Chukars) to 100% (WL chickens) of the birds sampled. Adrenal necrosis was first observed at 1 DPI in the chickens, B. quail, and J. quail, at 2 DPI in the turkeys, guineafowl, and pheasants, and at 3 DPI in the Chukars. Heterophilic infiltration lagged behind the adrenocortical necrosis was first observed at 1.5 DPI in the WR chickens (Fig. 18a), and was most widespread in the WR chickens, turkeys, pheasants, and Chukars (Table 2).

Adrenal gland; Ring-necked Pheasant 3 DPI. Severe confluent necrosis of adrenal corticotrophic and chromaffin cords. HE. Bar = 75 µm. Inset: Early heterophilic inflammation associated with necrosis. Bar = 45 µm.
The thyroid gland contained a small focus of epithelial necrosis in one guineafowl, one turkey, and one B. quail but had no significant lesions in the remaining species. The parathyroid gland, when included in the tissue samples, had mild to moderate multifocal necrosis with heterophilic inflammation.
Urogenital systems
Renal changes indicative of dehydration, such as minimal to mild dilatation of the distal segments of nephrons, were observed in the majority of birds of each group. Discrete foci of proximal convoluted tubular epithelial necrosis with minimal heterophilic inflammation was observed in only the turkeys (71%), guineafowl (45%), pheasants (50%), and Chukars (20%).
In the ovary, minimal necrosis and inflammation in the medullary interstitium and thecal layers of developing follicles were observed in the all three female pheasants, four of six female J. quail, one of two female B. quail, one of three female WL chickens, and one of four female WR chickens and guineafowl. No lesions were observed in the ovaries from the sampled Chukars. Testicular lesions were limited to minimal to mild interstitial infiltrates of heterophils and infrequent interstitial cell necrosis and were observed in all seven sampled male turkeys, one of two male WL and WR chickens, and two of four male guineafowl. Testicles of the J. quail, B. quail, pheasants, and Chukars lacked lesions. The seminiferous tubules remained unaffected.
Musculoskeletal system
In the J. quail (31%), turkeys (14%), guineafowl (9%), and pheasants (10%), skeletal muscle sampled from the breast, leg, and the extraocular sites infrequently contained degeneration to necrosis of individual myofibers. Affected myofibers were most often identified in the extraocular muscles.
Integument
Mild to moderate vasodilatation and edema were observed in the combs of the WL chickens (80%) and in the facial subcutis of the WR chickens (63%). Edema and hyperemia primarily involved the eyelids and conjunctiva in the guineafowl (63%). In the remaining species, appreciable dermal changes were infrequently observed and were minimal to mild (Table 2). However, heterophilic inflammation of the dermal pulp and degeneration to necrosis in the epithelium of the feather follicles was sporadically observed in all seven species.
Immunohistochemistry
Systemic localization of the HK/220 HPAI virus was common to all seven species investigated. In total, there was a strong parity between the demonstration of viral antigen and the identification of histologic lesions (Figs. 10b–15b, 18b). However, it was common for the distribution of viral antigen to be more widespread than the affiliated histopathologic alterations. In lymphoid organs, viral antigen was only identified in resident and infiltrating phagocytes and not in apoptotic lymphocytes (Fig. 16b). Furthermore, in the livers of a few turkeys, pheasants, and Chukars, large random foci of coagulative hepatic necrosis did not correspond to the presence of viral antigen (Fig. 17b).
Within 1 DPI, viral antigen was detected in all seven species in the epithelium of the nasal cavity and/or infraorbital sinuses, the endothelium of small vessels in the submucosa of the nasal cavity, phagocytic leukocytes in the propria of the respiratory and enteric tracts, and adrenal corticotrophic cells. In the chickens, J. quail, B. quail, turkeys, and guineafowl, viral antigen was also detected within 1 DPI in the phagocytes and capillary endothelium of the lung, phagocytes and ellipsoid-associated cells of the spleen, Kupffer cells of the liver, medullary epithelium and tingible body macrophages of the thymus, and feather follicle epithelium. With the exclusion of the guineafowl, pheasants, and Chukars, viral antigen also was detected within 1 DPI in the erythroid and myeloid precursor cells of the bone marrow, osteoclasts, neurons and glial cells of the brain, cardiac myocytes, renal tubular epithelium, bursal phagocytes, and pancreatic acinar epithelium. In the guineafowl and pheasants, antigen was initially observed in these cell types at 2 DPI. It was not until 3 DPI that antigen was identified in the parenchyma of the heart, brain, kidney, and pancreas of the Chukars; however, all other cells in these birds contained antigen by 2 DPI.
Each organ sampled had specific parenchymal cells that consistently contained viral antigen (Table 3). Despite the commonality of the cell types that harbored viral antigen, there was some discrepancy among species as to the intensity and consistency with which antigen was detected in a particular tissue. Of particular interest was the detection of viral antigen in the lungs; the extent and consistency of antigen localization in the pulmonary capillary endothelium was proportional to the demonstration of antigen in other capillary beds and small vessels in each species. Predilection for virus to infect endothelial cells throughout the body was the most diffuse and consistent in the chickens and J. quail, moderate in the guineafowl, B. quail, and turkeys, and only sporadic among the pheasants and Chukars (Table 3). Localization of viral antigen in the vascular endothelium was considered under the evaluation of the lungs and the spleen and separately from the parenchyma of other organs itemized in Table 3.
Average distribution∗ of nucleoprotein antigen, as determined by immunohistochemistry, in tissues sampled from gallinaceous birds intranasally inoculated with A/chicken/Hong Kong/220/97 (H5N1) influenza virus.
- = none; + = infrequent; ++ = common; +++ = widespread
Liver, kidney, peripheral nerves, alimentary epithelium, and skeletal muscle were consistent among the species in having infrequent HK/220 influenza virus antigen.
EACs = ellipsoid-associated cells.
The adrenal glands, lung, heart, spleen, and brain were the most concordant organs among these gallinaceous species in the consistency and extent to which they contained viral antigen (Table 3). In addition to the tissues assessed in Table 3, there was infrequent localization of viral antigen in hepatocytes, renal tubular epithelial cells, enterocytes, peripheral nerves and/or ganglia, skeletal myofibers, and satellite cells of the skeletal muscle and peripheral nerves in the species investigated. Viral antigen also was observed in the epithelium of the thyroid, parathyroid, harderian, lacrimal, and salivary glands; however, these tissues were not always included in the samples and inconsistently demonstrated viral antigen. Regardless of the tissue, the antigenic staining was invariably nuclear and also often cytoplasmic in distribution.
Discussion
Intranasal administration of the HK/220 HPAI virus to seven gallinaceous species of birds resulted in 75–100% mortality within 10 days of inoculation. This result verifies the high pathogenicity of the HK/220 virus for these species in accordance with standardized IV pathogenicity tests for influenza viruses. 1 With the earliest onset, most rapid progression of disease, and shortest MDTs in chickens, it is evident that there was maximal pathogenicity of the HK/220 HPAI virus for this species. This finding is consistent with those of previous studies suggesting species adaptation and maximal pathogenicity of AIV isolates for their corresponding gallinaceous host of origin. 2 3 44 49 55 The HK/220 virus possesses molecular characteristics common to other chicken-origin H5 and H7 influenza viruses, including a deletion in the neuraminidase stalk and increased glycosylation of the head of the hemagglutinin molecule, which may account for the maximal pathogenicity in chickens. 32
A distinct association was identified for the onset, advancement, and intensity of antigenic staining of the vascular endothelium and the rate of progression of clinical disease to death, with the chickens and Chukars forming the two extremes. In the WL and WR chickens, J. quail, B. quail, and guineafowl that died within 2–2.5 DPI, severe vascular-oriented pulmonary damage, including edema, congestion to hemorrhage, and microthrombosis of capillaries, was consistently observed. Other lesions indicative of vascular damage also were observed, including subcutaneous edema and hemorrhage, conjunctival hyperemia, and cyanosis of the comb tips (WL chickens). Viral antigen also was identified early in phagocytic leukocytes, including both heterophils and cells of monocytic lineage, which initially were localized within lymphoid aggregates of respiratory and enteric tracts. However, there was rapid distribution of viral antigen to phagocytic leukocytes of the spleen and other lymphoid organs. Primary infection of the vascular endothelium and phagocytes by the HK/220 virus resulted in rapid systemic dissemination and in peracute death by asphyxiation with the contribution of hemodynamic changes such as hypovolemia and disseminated intravascular thrombosis. This vascular phase of virus infection is akin to that described with other HPAI viruses, such as A/chicken/Pennsylvania/1370/83 (H5N2), 8 A/chicken/Victoria/1/85 (H7N7), 30 A/turkey/England/50–90/91 (H5N1), 30 and isolates from chickens in Mexico in 1995 (H5N2). 47
In birds dying between 2 and 4 DPI, the development of clinical disease, gross and histologic lesions, and extent of viral antigen contained in the parenchyma of multiple organs were largely proportional to the time of death after inoculation. Progressive neurologic dysfunction and mucoid diarrhea were the most pronounced clinical signs in each species experiencing mortality after 2 DPI. The histologic lesions of necrosis and acute inflammation corresponded to the presence of viral antigen. However, viral antigen also was observed frequently in foci lacking alterations detectable by light microscopy, which demonstrates the rapid replicative capability of this virus. In total, the pathogenesis for disease and death in birds surviving after 2 DPI is apt to be the result of a combination of factors such as myocardial conduction disturbances or insufficiency, neurologic dysfunction, adrenal insufficiency, and multiorgan failure. In addition, electrolyte imbalances resulting from pulmonary compromise and tissue necrosis may further contribute to morbidity and mortality.
The HK/220 HPAI virus demonstrated pantropic potential, with preferential virus localization in adrenocorticotrophic cells, cardiac myocytes, and neurons and glial cells of the central nervous system. Similar predilections for virus localization in the adrenal glands, 9 47 myocardium, 8 9 24 30 33 47 and parenchyma of the central nervous system 9 24 29 33 47 have been described with other HPAI viruses. Localization of HK/220 antigen in the parenchyma of other organs, such as pancreas, liver, alimentary tract, kidneys, peripheral nerves, and skeletal muscle, was less consistent among these species and more discrete in distribution, supporting the notion that virus distribution is dependent on particular host factors. 25 Furthermore, we identified novel sites of influenza viral antigen localization, including the epithelium of the parathyroid, harderian, and lacrimal glands, salivary gland epithelium, erythroid and myeloid precursors in the bone marrow, and feather follicle epithelium. It is doubtful that infection of these tissues is unique to the HK/220 virus. However, localization of viral antigen in sites such as the feather follicles may be important in terms of the epidemiology, with the possibility of transmission by contact with dander and shed feathers.
The HK/220 virus infection observed in the Chukars was distinctive. The localization of viral antigen in vascular endothelium and phagocytes was less intense in this species, and there was a significant delay in the initial detection of antigen in several tissues collected from these birds. Histologically and in accordance with the time after inoculation, the inflammatory response mounted by the Chukars to the virus was more mature and mononuclear in nature, as compared with exudative and heterophilic in the other six species, suggesting that the susceptibility of phagocytic mononuclear cells and vascular endothelium to HK/220 virus infection plays a role in the ability of species to resist virus infection. Furthermore, the extent of parenchyma containing viral antigen never reached the degree observed in the tissues of the other species investigated, especially those tissues that appeared to be most susceptible to the infection, such as the lung, heart, spleen, adrenal gland, and brain. The distribution of viral antigen reached maximal levels in birds sampled at 4 DPI. Birds sampled from 5 to 6 DPI had either stagnant or decreasing levels of viral antigen in the parenchyma of sampled organs. In the two Chukars that survived to 14 DPI, viral antigen was not detected in the collected tissues nor was virus reisolated from tissue and swab samples collected from these birds at the conclusion of the experiment (unpublished data). These birds did, however, seroconvert to the nucleoprotein antigen as determined with the AGP test, which confirms that the infection in surviving Chukars was transient in nature, as is typical of AIV infections in gallinaceous species. 16
In addition to the lesions induced directly by the HK/220 virus, there are undoubtedly secondary mechanisms that contribute to the pathogenesis of the infection in these avian species. The HK/220 virus demonstrated a high propensity for infection of phagocytic cells with the infection of monocytic cells being of particular interest. Previous studies involving the infection of macrophages with type A influenza and other viruses have demonstrated transcription upregulation or activation and subsequent release of certain cytokines and other bioactive compounds important in the mediation of inflammation. 7 17 20 23 57 In particular, tumor necrosis factor (TNF)-α is a strong mediator of endothelial activation and endothelial apoptosis, causing vasodilation, increased vascular permeability, and hypercoagulability, 4 20 35 51 and therefore may significantly contribute to the development of vascular lesions observed during the peracute course of the infection. The HK/220 virus and other HPAI viruses, likely share comparable pathogenic mechanisms with mammalian viruses that cause widespread vascular alterations, such as African horse sickness, 10 19 viral hemorrhagic disease of rabbits, 38 Dengue fever, 17 and African swine fever. 14 The pathogenesis of these diseases relates to the induction and release of cytokines from virus-infected macrophages, which can result in disseminated intravascular coagulopathy, endothelial cell activation, multiorgan failure, and shock, all of which are likely to be contributing aspects in the pathogenesis of HK/220 virus infection in the gallinaceous bird species investigated.
In addition to indirect endothelial effects, TNF-α and other cytokines are capable of causing marked lymphocytolysis via apoptosis. 14 20 40 This virus-independent or “by-stander” lymphoid depletion has been identified in various viral infections of avian 26 50 and mammalian 14 36 species, including infections with other influenza viruses in vivo and in vitro. 21 34 37 Apoptotic lymphoid depletion was uniform among the seven gallinaceous species investigated, and cytokines such as TNF-α and tumor growth factor β played a consequential role in the development of this lesion. In the current study, the severity of apoptotic depletion in lymphoid organs culminated as lymphocellular necrosis with corresponding heterophilic inflammation and histiocytic proliferation. The transition from lymphoid apoptosis to necrosis that was demonstrated in these species may result from a rapid overload of the local phagocytic cells and an attenuated phagocytic ability of virus-infected macrophages. 39 This lymphocellular depletion may have important repercussions for birds that survive infection with the HK/220 virus or other influenza viruses in that immunosuppression and increased susceptibility to other potential pathogens would result.
Footnotes
Acknowledgements
We extend special thanks to J. Beck, E. Turpin, and R. Brock for their technical assistance and support. Special recognition goes to Dr. W. Burke for the generous donation of the Japanese quail and to Dr. N. Primm for the generous donation of the turkeys that were used in this study.
