Abstract
Multiple endoscopic biopsy specimens of esophageal mucosa were received from a 13-year-old castrated male standard Poodle. The dog was presented for painful swallowing. Endoscopically, the distal aspect of the esophagus was inflamed with a polypoid mass that protruded into the esophageal lumen. Histologically, the stratified squamous epithelium overlying the mass and lining the adjacent esophageal mucosa was replaced by papillary projections covered by columnar epithelium with goblet cells supported by a fibrous stroma. This article reports a case of spontaneous esophageal adenomatous polyp with intestinal metaplasia (Barrett esophagus) and reviews the pathogenesis of esophageal metaplasia and adenocarcinoma.
Columnar-lined lower esophagus (CLE), or Barrett esophagus (BE), is a well-known and extensively studied entity in humans and has been induced experimentally in rats and dogs. 9,11 The presence of BE is the most important risk factor for the development of esophageal adenocarcinoma in humans. Although the pathogenesis of the disease is incompletely understood, the mechanism has been studied in canine, rat, and rabbit models. Experimentally, CLE has been induced in dogs by creating a lower esophageal mucosal defect and inducing acid reflux by cardioplasty and pentagastrin injection. 9 Spontaneous development of intestinal metaplasia of the esophagus, however, has not been described in the dog.
Clinical History
A 13-year-old castrated male standard Poodle presented to the Emergency Service at Tufts Cummings School of Veterinary Medicine for pain when swallowing, reluctance to eat, and hypersalivation. The dog had multiple bouts of pancreatitis over several months leading up to presentation and had clinical signs of hyperadrenocorticism (thin hair coat, comedones, and bilateral adrenal enlargement on ultrasound). No history of regurgitation was noted.
On physical exam, the dog was agitated and vocalizing; it also had abdominal distention, ptyalism, bilaterally firm submandibular lymph nodes, thin hair coat, comedones, rear limb weakness, and a grade IV/VI heart murmur. The dog also had persistent systemic hypertension (> 200 mmHg), with no response to medications (enalipril and amlodipine).
A gastrointestinal barium study was performed, and no abnormal gastric emptying or abnormal intestinal transit was found. Hematologic abnormalities included a mature neutrophilia (26.9 × 103/μl; range 4.9–16.9 × 103/μl), with mild elevation of alkaline phosphatase (311 U/L; range 12–121 U/L). Treatment was initiated to control pain, blood pressure, and odynophagia.
Endoscopic Observations
Upon endoscopic examination, the distal aspect of the esophagus was inflamed, and a broad-based sessile mass projected into the esophageal lumen (Fig. 1), approximately 1 cm orad to the cardiac sphincter. Multiple endoscopic biopsy specimens were taken from the mass and surrounding inflamed esophageal mucosa. The cardiac sphincter was open and the stomach was filled with bilious fluid. The fluid was removed, and multiple pinpoint ulcers were observed.
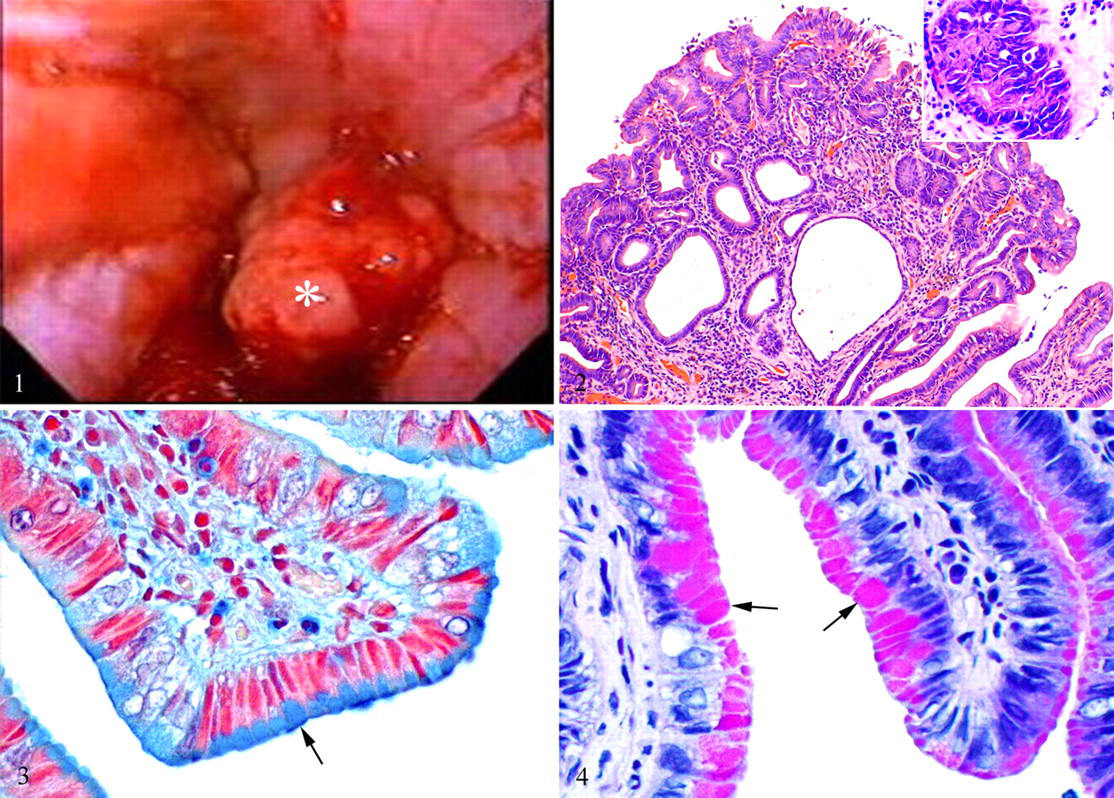
Microscopic Findings
Eight mucosal biopsy specimens of the esophageal mass and surrounding esophageal mucosa were submitted for histologic evaluation. Samples were fixed in 10% neutral-buffered formalin, routinely processed, and stained with hematoxylin and eosin (HE). Additional sections were stained with periodic acid–Schiff (PAS) and alcian blue (pH 2.5).
The biopsy specimens of the mass and from the adjacent esophageal mucosa had absence of normal stratified squamous mucosa with replacement by papillary projections of well-differentiated, regionally pleomorphic, simple columnar epithelium supported by a fibrous stroma (Fig. 2). Most of the columnar epithelial cells had moderately eosinophilic cytoplasm and oval, hyperchromatic, tightly packed basal nuclei (Fig. 2, inset). Scattered throughout the epithelium were goblet cells with pale basophilic (HE) mucinous vacuoles that were alcianophilic (Fig. 3) and PAS-positive (Fig. 4). Segmentally, epithelial cells had large, oval, hypochromatic nuclei with scant amphophilic, vacuolated cytoplasm. There were also regions with moderate anisokaryosis and anisocytosis, but mitotic figures were rare (2 per 10 high-power fields). The submucosa was infiltrated by numerous lymphocytes and plasma cells with fewer mast cells. A diagnosis was made: adenomatous polyp with distal esophageal columnar metaplasia. Owing to the poor prognosis and concurrent problems, humane euthanasia was elected; necropsy was not authorized.
Discussion
The diagnosis of BE is based on (1) the change from stratified squamous epithelium to a specialized, intestinal-type, columnar epithelium with goblet cells and (2) a change from neutral to acid mucins (sialomucins and sulfomucins). 2 There are two criteria for the diagnosis of BE: first, endoscopic evidence of columnar epithelium above the gastroesophageal junction and, second, histologic evidence of intestinal metaplasia. The term intestinal metaplasia may not be entirely correct, because true absorptive enterocytes are not observed. 10 BE most often resembles incomplete intestinal metaplasia owing to the sole presence of mucus-secreting columnar cells that contain both neutral and acid mucins. 2 It differs from complete intestinal metaplasia, which is characterized by goblet cells, Paneth cells, and absorptive enterocytes. 6 Compared to complete intestinal metaplasia, incomplete intestinal metaplasia is more frequently associated with the development of dysplasia and adenocarcinoma. 2,6 Many investigators will diagnose BE if there is any degree of columnar metaplasia in the lower esophagus with histologic evidence of goblet cells, highlighted using alcian blue stain. 14 Others believe that the presence of columnar cells alone is sufficient; they have shown that, given enough time, goblet cells will develop, thereby suggesting an early lesion of intestinal metaplasia. 3 Alcian blue reacts with acidic glycosaminoglycans, such as sulphomucin and sialomucin. 12 The dog in the present case had goblet cells within the columnar epithelium that were highlighted by alcian blue stain (Fig. 3.)
BE is associated with chronic gastroesophageal reflux and occurs in up to 10% of people with gastroesophageal reflux disease. It is the most important risk factor for developing esophageal adenocarcinoma. 10 Its pathogenesis is not fully understood, but it is thought to result from chronic mucosal irritation resulting in esophagitis, chronic inflammation, and subsequent adaptive phenotypic changes in the mucosa. Reflux of gastric contents is the most important cause of esophagitis. Most often, it is caused by decreased tone of the lower esophageal sphincter. Central nervous system depressants, hypothyroidism, pregnancy, hiatal hernia, and delayed gastric emptying are important predisposing factors. Characteristic histologic findings of esophagitis include inflammatory cells, including eosinophils, neutrophils and lymphocytes in the squamous epithelium; basal cell hyperplasia; and elongation of lamina propria papillae. Histologic changes are not correlated with the severity of signs, but they are correlated with the duration of the reflux. 10
Metaplasia is a well-known adaptive response of tissue to various stimuli. BE is one of the best-described examples of metaplasia and the progression from inflammation to metaplasia to dysplasia to neoplasia. 13 Knowledge of the sequence of events, the cell signaling, and the genes responsible for progression in BE may elucidate other neoplastic processes.
The initial injury may include any insult that irritates the distal esophagus—for example, chemical insult, dietary nitrites, bacteria such as Helicobacter pylori, genetic factors, and gastroesophageal reflux. 1 The source of reflux in this dog was not determined, and a necropsy was not performed. Under normal circumstances, without reflux, esophageal mucosal defects are repaired with regeneration of squamous epithelium. In the presence of gastroduodenal esophageal reflux of acid, or acid and bile, the regenerated epithelium is columnar. 4 Three types of regenerative columnar epithelium are possible in the lower esophagus: junctional or cardia epithelium, gastric fundic epithelium, or specialized columnar epithelium (intestinal epithelium). Only the specialized columnar epithelium, defined by intestinal-type goblet cells, carries a risk of malignancy in humans. 14
Studies of metaplasia in BE have led to the identification of key regulatory transcription factors called master switch genes. 13 Because squamous epithelium and columnar epithelium are closely related, only a few genes are responsible for their differentiation. The cell of origin of Barrett metaplasia is controversial. One possibility is that of transdifferentiation of squamous cells to the columnar phenotype. 13 The second theory is that metaplastic tissue arises from a change in esophageal stem cells (at the tips of papillae in the basal layer) to an intestinal stem cell. 1,13 Other theories on the origin of stem cells include migration of cells from the gastroesophageal junction, esophageal gland duct cell metaplasia, and bone marrow–derived stem cells. 1
A member of the p53 family of transcription factors, p63 plays a critical role in commitment of early stem cells to a basal cell progeny. In the absence of p63, progenitor cells develop into ciliated columnar cells, evidenced by the observation that p63 knockout mice completely lack stratified squamous epithelium, suggesting that p63 plays a role in the initiation and maintenance of the squamous phenotype. 13 Other potential master switch genes include homeobox genes responsible for body patterning during embryogenesis, and Cdx2 genes (caudal-type homeobox transcription factor 2). 13 These genes encode transcription factors involved in the early patterning of the posterior segment of the hindgut and in the development and maintenance of the intestinal phenotype of the epithelium. There is a sharp anterior boundary for expression of these genes at the gastroduodenal junction. In particular, Cdx2 is responsible for transcription of sucrase-isomaltase, MUC2, and lactose-phlorizin (gene products responsible for intestinal differentiation), and it has been demonstrated in Barrett metaplasia. 1,13 The mechanism for turning on the switch for this gene is unknown. 13
The progression from metaplasia to dysplasia and carcinoma is better understood. This process includes acquiring independent growth characteristics by evading negative trophic factors and apoptotic signals and by inducing angiogenesis. 1,13 The genetic mutations involved in this process in Barrett adenocarcinoma include development of aneuploidy, p53 mutations, cyclin D1 overexpression, decreased E-cadherin expression, APC gene inactivation (adenomatous polyposis coli), and loss of heterozygosity of tumor suppressor genes. 1,7,13 The stimulus for cells to develop these mutations may come from their local environment by exposure to signals they would not normally receive. Columnar cells of BE would receive signals from the surrounding squamous epithelium. 13 This is supported by the observation that (1) neoplasia is generally more common at the junction of different types of epithelium and (2) direct anastomosis of squamous epithelium to columnar epithelium in rats with induced mixed reflux of acid and bile leads to CLE with dysplasia and esophageal adenocarcinoma. 8,13 The composition of reflux is heterogeneous and includes saliva, esophageal secretions, food, gastric acid, pepsin, mucus, bile salts, trypsin, cholesterol, and lipase. 15 Experimentally, acid and pepsin damage the esophageal mucosa, and pepsin promotes regeneration with a columnar-type epithelium. Bile acids in the esophagus can also act as tumor promoters. The biological effects of bile are strongly pH dependent; more neutral pH is potentially harmful by stimulating the proliferation of cells. This is supported by the observation that patients with BE that are on long-term H2 receptor blockers and proton pump inhibitors have increased risk of developing adenocarcinoma. 1 Neutral pH also stimulates bacterial overgrowth, leading to deconjugation of bile acids, which are particularly harmful. 15 Bacterial overgrowth also leads to generation of carcinogenic N-nitroso compounds from conjugated bile salts. Neutral pH stimulates increased gastrin levels; recent data show this hormone to be mitogenic in Barrett mucosa. 15 In addition, the associated chronic inflammation produces free radicals, with upregulation of nitric oxide synthase, cyclooxygenase-2, and myeloperoxidase during the development of BE and esophageal adenocarcinoma. 15 Interestingly, the dog in this case was receiving oral H2 receptor blockers; however, the duration of treatment was unknown.
Metaplasia is often a precursor to neoplasia in humans and animals. BE is an example of columnar metaplasia of the esophagus associated with increased risk of adenocarcinoma in humans. The canine model was used early in pathogenetic studies and showed that development of columnar-lined esophagus requires mucosal damage, an incompetent lower esophageal sphincter, chronic reflux, low gastric content clearance, and increased acid. 8 Spontaneous columnar metaplasia of the esophagus has been reported in 3 cats; 5 however, to our knowledge, this is the first report of spontaneous BE with an esophageal adenomatous polyp in a dog.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
