Abstract
Background
Sepsis remains a leading cause of mortality in critically ill patients, necessitating the identification of reliable prognostic markers. The triglyceride glucose-body mass index, which combines lipid and glucose metabolism, may serve as a valuable predictor of long-term outcomes in this population.
Methods
This study employed a retrospective observational cohort design and analyzed data from the MIMIC-IV database encompassing 2424 septic patients admitted between 2008 and 2019. Patients were categorized into quartiles based on triglyceride glucose-body mass index values. The primary outcomes assessed were all-cause mortality rates at 30, 90, and 180 days as well as 1-year post-admission, and analysis was performed using multivariable Cox proportional hazards models.
Results
In total, 2424 septic patients meeting the inclusion criteria were finally included in the analyses. Lower triglyceride glucose-body mass index values have been associated with higher long-term mortality outcomes in critically ill septic patients. However, higher triglyceride glucose-body mass index quartiles were associated with a significant reduction in mortality risk at 180 days and 1 year after admission, even after adjustment for potential confounding variables. Patients in the third (hazard ratio: 0.81, 95% confidence interval: 0.67–0.98, p = 0.028) and fourth (hazard ratio: 0.73, 95% confidence interval: 0.56–0.96, p = 0.026) quartiles exhibited significantly lower hazard ratios for mortality than those in the lowest quartile. Notably, no significant associations were observed with 30-day mortality, suggesting that triglyceride glucose-body mass index values are more strongly associated with long-term outcomes.
Conclusion
In this retrospective cohort study, lower triglyceride glucose-body mass index values were associated with higher long-term mortality in critically ill septic patients. Our findings suggest that the triglyceride glucose-body mass index serves as a useful prognostic marker and can help identify patients at increased risk of mortality. These results highlight a potential association that warrants further investigation in prospective studies to clarify the role of triglyceride glucose-body mass index in sepsis management.
Keywords
Introduction
In recent years, the association between metabolic and body composition indices and health outcomes has garnered significant attention in the field of critical care medicine. 1 Among these indices, the triglyceride glucose-body mass index (TyG-BMI) has emerged as a valuable metabolic indicator that integrates fasting triglycerides (TGs), fasting blood glucose (FBG), and body mass index (BMI). Specifically, TyG-BMI combines the TG-glucose (TyG) index—a well-validated surrogate marker for insulin resistance—with BMI, which reflects adiposity. Conceptually, TyG-BMI represents the combined influence of metabolic dysregulation and excess adiposity, capturing both impaired glucose–lipid metabolism and the contribution of body mass to insulin resistance. 2 Higher TyG-BMI values typically indicate greater metabolic burden and lower insulin sensitivity. Therefore, TyG-BMI is best understood as a metabolic index augmented by adiposity, rather than a direct body composition measurement. This composite nature may make TyG-BMI a particularly relevant biomarker for critically ill patients,3–5 in whom metabolic homeostasis and nutritional reserve are crucial for recovery and survival.
Sepsis is a life-threatening condition characterized by a dysregulated host response to infection, leading to organ dysfunction and increased mortality risk. 6 The pathophysiology of sepsis involves complex interactions between inflammatory mediators, metabolic derangements, and immune responses. 7 In critically ill septic patients, the challenge of managing nutritional status becomes particularly acute as both undernutrition and obesity can adversely affect recovery outcomes. 8 As such, indices that effectively capture the interplay between body fat, metabolic health, and overall mortality risk are crucial for guiding clinical interventions and improving patient prognostication.
Emerging evidence suggests that higher TyG-BMI values are correlated with worse clinical outcomes in various populations, including those with metabolic syndrome and cardiovascular diseases. 9 However, the specific relationship between TyG-BMI and all-cause mortality (ACM) in critically ill septic patients remains inadequately explored. This knowledge gap is particularly concerning as critically ill patients often exhibit varied metabolic profiles that may influence their resilience against infection and recovery trajectories. 10 Examining ACM, rather than cause-specific mortality, is clinically significant because it provides a comprehensive assessment of the total disease burden and the combined effects of metabolic dysregulation, infection, organ failure, and treatment-related complications. In sepsis, where multiple organ systems are affected and death may result from overlapping metabolic, cardiovascular, and inflammatory pathways, ACM serves as the most objective and integrative endpoint for evaluating patient prognosis. 11
From a public health perspective, sepsis continues to be a leading contributor to global mortality, accounting for approximately one in five deaths worldwide. The high burden of ACM underscores the urgent need to identify modifiable metabolic risk factors that could improve long-term survival. Understanding how TyG-BMI influences ACM can therefore inform early risk stratification and justify targeted metabolic or nutritional interventions in the intensive care unit (ICU) setting, potentially translating into meaningful clinical action.
Moreover, stratifying patients according to TyG-BMI quartiles may help clinicians better understand the prognostic implications of metabolic health in sepsis. Previous studies have highlighted the contribution of metabolic factors to disease severity and outcomes; however, only a few have investigated TyG-BMI as an integrated predictor. 12 By analyzing its association with ACM across different timeframes, the present study aimed to assess whether TyG-BMI independently predicts mortality and identify subgroups of septic patients at differential metabolic risk. This approach may enhance the current understanding of how metabolic and nutritional factors influence survival in critical illness.
Materials and methods
Source of data
This study employed a retrospective observational cohort design with longitudinal follow-up of patients. The Medical Information Mart for Intensive Care-IV, version 3.0 (MIMIC-IV-3.0) database, which is publicly accessible, includes data on over 50,000 ICU admissions at the Beth Israel Deaconess Medical Center in Boston, Massachusetts, spanning from 2008 to 2019. 13 The MIMIC-IV database offers extensive information, including demographic data, vital signs, laboratory results, and diagnoses coded according to the International Classification of Diseases, Ninth Revision (ICD-9) and Tenth Revision (ICD-10). Access to this database was facilitated by one of the authors (Yufan Pu), who obtained the required certification (certification number: 64636894) and extracted data related to the pertinent variables for the current study. This study was conducted in accordance with the ethical principles of the Helsinki Declaration of 1975, as revised in 2024. Individual patient consent was deemed unnecessary due to the anonymized nature of the health information contained within the database. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14 Patients diagnosed with sepsis were identified using ICD-9 and ICD-10 codes. The method for identifying patients with sepsis who met the Sepsis 3.0 diagnostic criteria from the MIMIC-IV database was consistent with those reported in previously published studies. 15 The exclusion criteria were as follows: (a) ICU stay <48 h; (b) multiple admissions to the ICU for sepsis, but only data from the first admission were extracted; (c) insufficient data (missing data on TGs, FBG levels, and BMI); and (d) consumption of lipid-lowering drugs or antidiabetic treatment (e.g. insulin and oral hypoglycemics). However, detailed outpatient medication exposure prior to hospitalization is not comprehensively captured in the MIMIC-IV database; therefore, long-acting antidiabetic agents prescribed before ICU admission may not have been fully identifiable. Ultimately, a final cohort of septic patients was selected and stratified into four groups based on the TyG-BMI quartile for analysis (Figure 1).

Inclusion/exclusion criteria. MIMIC: Medical Information Mart for Intensive Care.
Variable extraction
Data extraction was conducted using PostgreSQL (version 13.7.2) and Navicat Premium (version 16), employing structured query language (SQL). The variables of interest were categorized into four primary domains: (a) demographic factors, including age, race, sex, weight, height, and BMI (the values for weight and height used to calculate BMI were the first available measurements documented within the first 24 h of ICU admission); (b) comorbid conditions, such as heart failure, diabetes, chronic kidney disease, and paraplegia; (c) laboratory markers, including red blood cell (RBC) count, white blood cell (WBC) count, and platelet count as well as hemoglobin, serum sodium, serum creatinine, high-density lipoprotein (HDL), FBG, and TG levels; and (d) severity scores at the time of admission, which included the Acute Physiology Score III (APSIII), Simplified Acute Physiology Score II (SAPS-II), Oxford Acute Severity of Illness Score (OASIS), and Sepsis-related Organ Failure Assessment (SOFA) score. The SOFA score was calculated as the worst value (i.e. the value indicating the greatest degree of organ dysfunction) recorded within the first 24 h following ICU admission. All laboratory measurements, including FBG and TGs used to calculate the TyG-BMI, were obtained within the first 24 h after ICU admission. The follow-up period commenced on the date of admission and concluded either at the time of death or at the end of the study period. The primary outcomes evaluated were ACM at 30, 90, and 180 days as well as 1 year after admission. All laboratory measurements were recorded during the initial assessment following ICU admission.
Definition of related concepts
The TyG index serves as a surrogate marker for insulin resistance and is calculated using FBG and TG levels. These laboratory values, measured within the first 24 h of ICU admission, were used for the calculation. Initially, the FBG and TG levels of the participants were measured. The TyG-BMI index was computed using the following formula: TyG index = ln [fasting TG (mg/dL) × FBG (mg/dL)/2], and the TyG-BMI was determined by multiplying the TyG index by BMI: TyG-BMI index = TyG index × BMI. This study aimed to evaluate the association between these indices and ACM in patients with sepsis, with primary endpoints assessed at 30, 90, and 180 days as well as 1 year after admission.
Statistical analyses
This study used R 4.3.0 statistical analysis software. Normally distributed continuous data were expressed as mean ± standard deviation (SD), and differences among multiple groups were analyzed using one-way analysis of variance (ANOVA), followed by post-hoc pairwise comparisons when appropriate. For comparisons between two groups, Student’s t-test was applied for normally distributed continuous variables. Non-normally distributed data were expressed as median (interquartile range (IQR)) and compared using the Mann–Whitney U test or Kruskal–Wallis test, as appropriate. Categorical data were expressed as n (%) and analyzed using the chi-square or Fisher’s exact test. Differences between the groups were considered statistically significant when the p-value was <0.05. The Kaplan–Meier (K–M) method for survival analysis was utilized to evaluate the frequency of endpoints across various groups according to the TyG-BMI index values, with their differences determined using log-rank tests. To determine the hazard ratio (HR) and 95% confidence interval (CI) among the TyG-BMI index and endpoints, Cox proportional hazards models were employed, with adjustments made for certain models. The multivariate model incorporated variables pertinent to clinical conditions and prognoses: Model 1: unadjusted; Model 2: adjusted for age, sex, BMI, and race; Model 3: adjusted for sex, age, race, BMI, respiratory failure, diabetes, congestive heart failure, paraplegia, and renal disease. The assumptions of the Cox proportional hazards model were verified prior to final analysis. The proportional hazards assumption was assessed both graphically, using log-minus-log survival plots, and statistically, using Schoenfeld residual tests. No significant violations of the proportional hazards’ assumption were detected (all p > 0.05). Model fit was further examined using deviance residuals and influential diagnostics to ensure robustness of the results. Furthermore, TyG-BMI was analyzed continuously using restricted cubic splines (RCS) to elucidate dose–effect relationships between dosage and effect in relation to the risk of outcomes.16,17 In addition, sensitivity analysis was performed after excluding patients receiving continuous insulin infusion during the first 24 h of ICU admission to evaluate the robustness of the results.
Results
Baseline characteristics of study participants
After applying the inclusion and exclusion criteria, a total of 2424 septic patients were finally included in the analysis. The observed 30-, 90-, 180-day and 1-year mortality rates were 34.60%, 48.10%, 51.70%, and 55.9%, respectively. Table 1 shows the comparison of patients in the lowest TyG-BMI quartile (Q1) with those in higher quartiles (Q2–Q4), demonstrating that patients in higher quartiles tended to be younger and predominantly male, with a greater prevalence of obesity, diabetes, and paraplegia. Furthermore, individuals in the higher TyG-BMI quartiles exhibited increased SOFA scores and distinct laboratory profiles.
Characteristics of participants categorized by TyG-BMI quartiles.

The variable “diabetes” in this table refers to a small number of patients with a previous diagnosis of diabetes who were not receiving active antidiabetic medication at the time of ICU admission and were therefore not excluded according to criterion (d) in the subsection “Source of data.” This variable was retained in the multivariate models to account for any residual confounding effect of a history of diabetes on outcomes.
TyG-BMI: triglyceride glucose-body mass index; Q: quartile; LOS: length of stay; ICU: intensive care unit; BMI: body mass index; RBC: red blood cell; WBC: white blood cell; APSIII: Acute Physiology Score III; SAPS-II: Simplified Acute Physiology Score II; OASIS: Oxford Acute Severity of Illness Score; SOFA: Sepsis-related Organ Failure Assessment; GCS: Glasgow Coma Scale
Multivariable Cox proportional hazard models
Table 2 presents the results of multivariable Cox proportional hazard models evaluating the association between TyG-BMI quartiles and mortality at different time points (30, 90, and 180 days as well as 1 year after admission) in septic patients. The HRs and 95% CIs were provided for each TyG-BMI quartile, using the lowest quartile (Q1) as the reference. No significant association was observed between TyG-BMI quartiles and 30-day mortality. The HRs for Q2, Q3, and Q4 were not statistically significant across all models. In the unadjusted model (Model 1), patients in Q3 and Q4 (Q3: HR = 0.81, 95% CI: 0.69–0.95, p = 0.008; Q4: HR = 0.79, 95% CI: 0.68–0.93, p = 0.005) showed a significantly lower risk of 90-day mortality than those in Q1. However, after adjusting for covariates (Models 2 and 3), the significance was attenuated, indicating potential confounding factors. Similar to the outcomes for 90-day mortality, patients in Q3 (HR = 0.75, 95% CI: 0.65–0.88, p < 0.001) and Q4 (HR = 0.76, 95% CI: 0.65–0.89, p < 0.001) exhibited a lower risk of 180-day mortality in the crude model. After adjustment in Model 3, Q3 remained significantly associated with lower mortality (HR = 0.81, 95% CI: 0.67–0.98, p = 0.028). The association between higher TyG-BMI quartiles and reduced 1-year mortality was consistent across all models. In the fully adjusted model (Model 3), Q3 (HR = 0.81, 95% CI: 0.67–0.97, p = 0.022) and Q4 (HR = 0.73, 95% CI: 0.56–0.96, p = 0.026) demonstrated a significant decrease in risk compared with the reference group. Overall, these findings suggest that higher TyG-BMI quartiles are associated with improved long-term survival outcomes in patients with sepsis, particularly evident for 180-day and 1-year mortality, even after controlling for demographic and clinical covariates.
Multivariable Cox proportional hazard models for TyG-BMI at different time points.

HR: hazard ratio; CI: confidence interval; BMI: body mass index; TyG-BMI: triglyceride glucose-body mass index.
Model 1: Crude.
Model 2: Adjusted for sex, age, race, and BMI.
Model 3: Adjusted for sex, age, race, BMI, congestive heart failure, respiratory failure, diabetes, paraplegia, and renal disease.
K–M survival analysis curves and HR for TyG-BMI index
In our analysis of the impact of TyG-BMI quartiles on ACM among critically ill septic patients, K–M survival curves demonstrated a significant association between TyG-BMI values and long-term mortality rates (log-rank test: 90 days, p = 0.015; 180 days, p < 0.001; 1 year, p < 0.001). This visual separation was supported by the Cox model, which showed, for instance, a significantly reduced 1-year mortality risk for patients in Q3 (HR = 0.81, 95% CI: 0.67–0.97, p = 0.022) and Q4 (HR = 0.73, 95% CI: 0.56–0.96, p = 0.026) than for patients in Q1 (Figure 2).
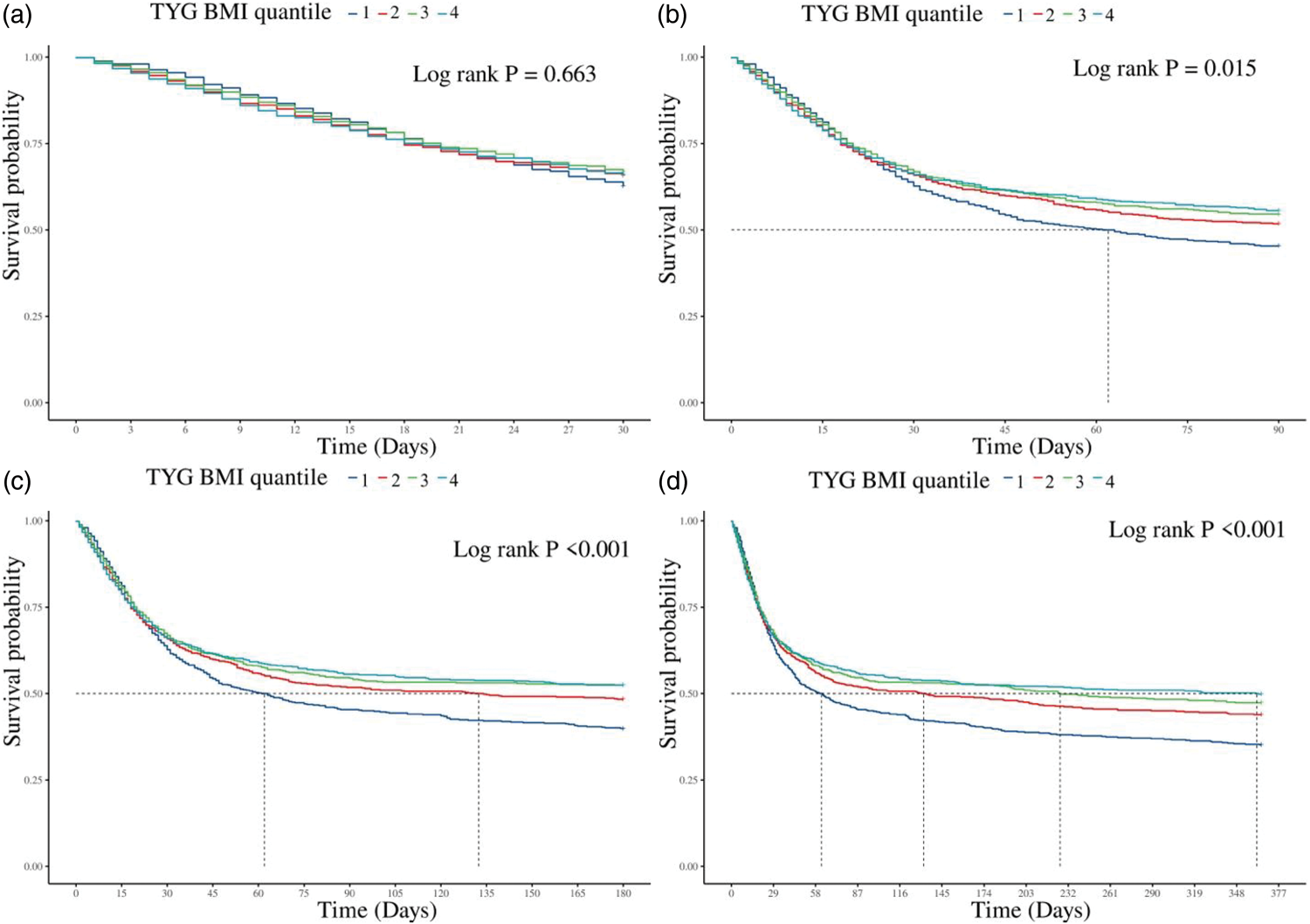
K–M survival analysis curves and cumulative incidence with TyG-BMI index for 30-, 90-, and 180-day and 1-year mortality in critically ill patients with sepsis.
RCS analysis evaluated the relationship between risk ratios and TyG-BMI values across different timeframes. Notably, the results revealed no significant nonlinear relationships for 30 days (p = 0.73) and 90 days (p = 0.11) after admission. However, a significant nonlinear relationship emerged at 180 days (p = 0.04) and 1 year (p = 0.02) after admission, indicating that lower TyG-BMI values are associated with elevated risk ratios. Beyond a certain threshold, the risk ratios stabilize, suggesting that the TyG-BMI plays a critical role in predicting long-term mortality outcomes in critically ill septic patients. These findings highlight the need for further investigation of TyG-BMI as a potential prognostic indicator in this clinical context (Figure 3).

The TyG-BMI index hazard ratio for 30-, 90-, and 180-day and 1-year mortality in critically ill patients with sepsis. (a) 30-day mortality, (b) 90-day mortality, (c) 180-day mortality and (d) 1-year mortality
Discussion
In this large retrospective cohort of critically ill septic patients, we identified a significant inverse association between the TyG-BMI index and long-term mortality. Specifically, patients in the highest TyG-BMI quartile had a 27% lower risk of 1-year ACM than those in the lowest quartile (HR = 0.73, 95% CI: 0.56–0.96), after adjusting for key confounders. This association was consistently observed for 180-day mortality but was absent for short-term (30-day) outcomes, suggesting that the metabolic and nutritional reserve captured by TyG-BMI plays a particularly important role in long-term survival and recovery after sepsis. Sepsis is a complex syndrome characterized by an overwhelming immune response to infection, often leading to organ dysfunction and high mortality rates.18–21 The TyG-BMI, which integrates TGs, glucose levels, and BMI, provides a more comprehensive picture of metabolic health, potentially offering better prognostic insights in septic patients. 3
Our study demonstrated that lower TyG-BMI quartiles were linked to a higher risk of long-term mortality, even after multivariable adjustment. This finding appears counterintuitive given that higher TyG-BMI is often associated with adverse metabolic outcomes in the general population. We hypothesize that this observation is explained by a phenomenon analogous to the “obesity paradox” described in other critical illnesses, where greater metabolic reserves potentially enhance the ability to withstand the prolonged catabolic state of sepsis.22,23 However, this interpretation remains speculative. Our study lacks direct measurements of body composition (e.g. fat-free mass), nutritional intake, or markers for hyper catabolism; therefore, the current data do not permit a direct test of the “metabolic reserve” hypothesis. The protective association we observed warrants further investigation using datasets that include these precise physiological measurements. 24
However, it is important to note that the significant effect of TyG-BMI values was not observed on short-term mortality outcomes. The lack of a significant association between TyG-BMI quartiles and 30-day mortality suggests that the initial phase of sepsis is influenced more by acute inflammatory responses and organ failure than by metabolic health. 25 This finding aligns with previous research indicating that although metabolic factors play a role in long-term recovery and resilience, immediate outcomes in sepsis are predominantly driven by the severity of infection and the host’s immune response.7,26,27
The study’s strength lies in its large sample size and robust methodology, utilizing the MIMIC-IV database, 28 which provides comprehensive clinical data over an extended period. The use of multivariable Cox proportional hazard models allowed for thorough adjustment of potential confounders, enhancing the reliability of the observed associations. 29 Additionally, the use of RCS in the analysis provided a nuanced understanding of the dose–response relationship between TyG-BMI and mortality risk, revealing a significant nonlinear association at 180 days and 1 year.30,31 This suggests that the relationship between metabolic health and sepsis outcomes is complex and involves threshold effects that warrant further investigation.
Despite these strengths, certain limitations must be acknowledged. First, the retrospective design precludes causal inference. 32 Second, although patients receiving antidiabetic therapy during ICU admission were excluded, detailed outpatient medication histories prior to hospitalization are incompletely captured in the MIMIC-IV database. Therefore, exposure to long-acting glucose-lowering agents (e.g. basal insulin analogs, GLP-1 receptor agonists, and thiazolidinediones) before ICU admission may not have been fully ascertained. Such residual exposure could have influenced early fasting glucose and TG measurements, potentially introducing nondifferential misclassification. Third, exposure measurement is subject to potential error; the lack of guaranteed fasting status for laboratory measurements and the potential inaccuracy of BMI due to fluid shifts or amputations in critically ill patients may have led to nondifferential misclassification, potentially attenuating the observed associations.
This study demonstrated that a higher TyG-BMI was independently associated with lower short- and long-term ACM in patients with sepsis, suggesting that the metabolic reserve reflected by TyG-BMI plays a protective role in critical illness. These findings extend previous evidence linking TyG-BMI to cardiovascular and metabolic outcomes,9,33 highlighting its potential prognostic relevance in critical care settings. Clinically TyG-BMI offers a simple and readily available indicator that may help identify patients at risk and guide personalized metabolic management strategies. Given its incorporation of both adiposity and insulin resistance, the index provides an integrated view of metabolic health that complements traditional severity scores.34,35
Conclusion
In this retrospective cohort study, lower TyG-BMI values were associated with higher long-term mortality in critically ill septic patients. Our findings suggest that TyG-BMI is a useful prognostic marker and can help identify patients at increased mortality risk. These results highlight a potential association that warrants further investigation in prospective studies to clarify the role of TyG-BMI in sepsis management.
Supplemental Material
sj-zip-1-imr-10.1177_03000605261420914 - Supplemental material for Association between triglyceride glucose-body mass index and all-cause mortality in patients with sepsis: Evidence from a retrospective cohort study
Supplemental material, sj-zip-1-imr-10.1177_03000605261420914 for Association between triglyceride glucose-body mass index and all-cause mortality in patients with sepsis: Evidence from a retrospective cohort study by Yufan Pu, Xiang Zhou, Xuejing Li and Jiang Xu in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605261420914 - Supplemental material for Association between triglyceride glucose-body mass index and all-cause mortality in patients with sepsis: Evidence from a retrospective cohort study
Supplemental material, sj-pdf-2-imr-10.1177_03000605261420914 for Association between triglyceride glucose-body mass index and all-cause mortality in patients with sepsis: Evidence from a retrospective cohort study by Yufan Pu, Xiang Zhou, Xuejing Li and Jiang Xu in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605261420914 - Supplemental material for Association between triglyceride glucose-body mass index and all-cause mortality in patients with sepsis: Evidence from a retrospective cohort study
Supplemental material, sj-pdf-3-imr-10.1177_03000605261420914 for Association between triglyceride glucose-body mass index and all-cause mortality in patients with sepsis: Evidence from a retrospective cohort study by Yufan Pu, Xiang Zhou, Xuejing Li and Jiang Xu in Journal of International Medical Research
Footnotes
Acknowledgments
The authors express their gratitude to the participants and staff of the MIMIC-IV 3.0 database for their invaluable contributions to this study. We thank Li Yaran for assistance in data collection. Additionally, the authors utilized an artificial intelligence tool (DeepSeek) solely for the purpose of improving language fluency and readability during the final revision of this manuscript. The content and scientific integrity of the work remain the sole responsibility of the authors.
Author contributions
Conception and design: Yufan Pu; administrative support: Jiang Xu; collection and assembly of data: Yufan Pu and Xiang Zhou; data analysis and interpretation: Yufan Pu and Xiang Zhou; manuscript writing: all authors; and final approval of manuscript: all authors.
Clinical trial number
Not applicable.
Declaration of conflicting interests
The authors report no conflict of interest.
Ethical approval
The study was performed according to the guidelines of the Helsinki Declaration (1975, as revised in 2024). The use of the Medical Information Mart for Intensive Care-IV (MIMIC-IV) database was approved by the review committee of the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center. The data are publicly available (in the MIMIC-IV database); therefore, the need for ethical approval and informed consent was waived for this study.
Funding
This work was supported by the National Natural Science Foundation of China (82105004).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
